Abstract
A reduction of blood flow to active muscle will precipitate fatigue, and sustained isometric contractions produce intramuscular and compartmental pressures that can limit flow. The present study explored how blood flow and muscle oxygenation respond to isometric contractions at low, moderate, and maximal intensities. Over two visits, 10 males (26 ± 2 yr; means ± SD) performed 1-min dorsiflexion contractions at 30, 60, and 100% of maximal voluntary contraction (MVC) torque. Doppler ultrasound of the anterior tibial artery was used to record arterial diameter and mean blood velocity and to calculate absolute blood flow. The tissue oxygenation index (TOI) of tibialis anterior was acquired with near-infrared spectroscopy (NIRS). There was a progressive increase in blood flow at 30% MVC (peak of 289 ± 139% resting value), no change from rest until an increase in the final 10 s of exercise at 60% MVC (peak of 197 ± 102% rest), and an initial decrease (59 ± 30% resting value) followed by a progressive increase at 100% MVC (peak of 355 ± 133% rest). Blood flow was greater at 30 and 100% than 60% MVC during the last 30 s of exercise. TOI was ∼63% at rest and, within 30 s of exercise, reached steady-state values of ∼42%, ∼22%, and ∼22% for 30, 60, and 100% MVC, respectively. Even maximal contraction of the dorsiflexors is unable to cause more than a transient decrease of flow in the anterior tibial artery. Unlike dynamic or intermittent isometric exercise, our results indicate blood flow is not linearly graded with intensity or directly coupled with oxygenation during sustained isometric contractions.
Keywords: dorsiflexors, muscle blood flow, near-infrared spectroscopy
neuromuscular fatigue in response to voluntary effort can be defined as the reduced ability to exert force or power, regardless of whether or not the task can be performed successfully (4). This fatigue is task-dependent and results from central and peripheral mechanisms that act in concert or independently (1, 6). Because of the importance of blood flow to muscle metabolism, any impediment to muscle blood flow (MBF) will precipitate the development of fatigue in a variety of ways (e.g., 25). MBF is responsive to the intensity and type of muscle contraction (2, 23). Sustained isometric contractions pose particular challenges to muscle perfusion because the increased demand for MBF is opposed by increased intramuscular pressure (IMP), which will limit its delivery (5). IMP is positively related to contraction intensity (22, 24); thus, it is held that there is a critical force threshold beyond which mechanical pressure will occlude MBF (24, 25, 29, 30) and below which flow will be affected to varying degrees depending on the contraction intensity and anatomical location (compartmental pressure). Results obtained using a variety of techniques suggest that complete occlusion of MBF and perfusion occur at 50–60% of maximal voluntary contraction (MVC) force (13, 22, 25, 31). However, relatively few studies have recorded MBF directly during a sustained isometric effort, and the degree to which perfusion is compromised by high-intensity contraction remains uncertain.
In the last two decades, hemodynamic responses to isometric exercise have been assessed using four principal techniques: venous occlusion plethysmography (VOP), functional magnetic resonance imaging (fMRI), positron emission tomography (PET), and Doppler ultrasound. The first two are indirect methods that cannot measure blood flow during exercise per se (VOP; e.g., Ref. 12) or can only assess a change in flow rather than provide absolute values (fMRI; e.g., Ref. 31). In contrast, the latter two methods can provide direct measures of exercise-induced changes in muscle blood flow. PET measures perfusion directly from the muscle tissue (e.g., 14, 21), whereas Doppler ultrasound tracks changes in arterial size and mean blood velocity (MBV) to provide a direct measure of blood flow into a muscle compartment (e.g., 10, 19). The combination of Doppler ultrasound and NIRS allows for the assessment of concurrent associations between flow and oxygenation of muscle (see Ref. 18 for review).
Despite its advantages, few studies have used Doppler ultrasound to measure MBF during a sustained isometric contraction (7, 8, 13, 27, 30) with only one report conjunctly employing NIRS (9). Most of these studies examined MBF at 2–4 contraction intensities between 10 and 70% MVC and reported an increase in MBF, which was insensitive to the contraction intensity. The partial exception to this finding was a lesser increase in MBF at 50% compared with 20% MVC in males (but an equivalent increase in females; Ref. 27). As no previous study has measured MBF during maximal or even near-maximal efforts, our understanding of the relationships among contraction intensity (i.e., IMP), muscle perfusion, and oxygenation during sustained isometric contractions remains limited.
Hence, the purpose of this study was to assess real-time changes in the MBF and muscle oxygenation during a sustained MVC and compare these data to results obtained during contractions of moderate and low intensity (60 and 30% MVC, respectively). The dorsiflexor muscle group was selected for this experiment because the restrictive fascial sheath of the anterior compartment induces high levels of IMP during voluntary isometric contractions (26), and a proposed contraction intensity for complete occlusion has been identified in young males (60% MVC; Ref. 31).
MATERIALS AND METHODS
Subjects.
Ten healthy, recreationally active male volunteers (25.8 ± 2.4 yr, 174.8 ± 7.2 cm, 78.8 ± 7.4 kg) were recruited from the local university. Testing for the main experiment was performed over two visits separated by 2 to 4 days. Eight of the subjects participated in an additional experiment on a third day. The study was approved by the local university's ethics review board, and informed written consent was obtained from each subject.
Experimental setup.
All tests were conducted using the subject's dominant (right) leg. Subjects were seated in a custom-built isometric dynamometer with their right ankle positioned at 30° of plantar flexion and an angle of 90° at both the knee and hip joints (17). The distal aspect of the right thigh was clamped to minimize hip flexion during isometric dorsiflexion contractions. Straps across the toes and dorsum of the foot secured the limb to the dynamometer footplate.
Ultrasound imaging (8–10 MHz transducer, GE/Vingmed System Five) was used to measure anterior tibial artery diameter and Doppler ultrasound (4.7 MHz; 60° insonation angle, and full-width pulsed wave gate; GE System Five) was used to quantify MBV through the vessel. The ultrasound probe was positioned on the anterior aspect of the right leg, distal and lateral to the tibial tuberosity, to record from the anterior tibial artery below the point at which it emerges through the interosseous membrane. Local tissue oxygenation levels were recorded from the tibialis anterior using NIRS (Hamamatsu NIRO 300, Hamamatsu Photonics KK, Tokyo, Japan). The emitting and detecting optodes of the NIRS unit were placed over the belly of the TA muscle, ∼8 cm inferior and 2 cm lateral to the tibial tuberosity after the optimum position of the ultrasound probe had been determined and marked. The optodes were held within a black rubber housing that maintained a constant optode spacing of 4 cm. The opaque housing was affixed to the skin with black tape to minimize any loss of near-infrared light, as well as to prevent the intrusion of outside light.
Blood pressure (mean arterial pressure; MAP) was monitored with a pneumatic finger cuff (Ohmeda 2300 Finapres BP Monitor, Louisville, CO) on the middle finger of the left hand, which rested in the subject's lap. Heart rate (HR) was monitored using a three-lead echocardiogram with Ag-AgCl soft cloth electrodes (1 × 1 cm; 3M, London, ON, Canada). Electromyographic activity (EMG) of the dorsiflexors was recorded with Ag-AgCl electrodes (1.5 × 1 cm; Kendall-LTP, Chicopee, MA) arranged in a monopolar configuration. The recording electrode was positioned on the belly of the TA superior to the NIRS housing, whereas the reference electrode was placed on the distal tendon of the tibialis anterior muscle. Finally, a ground electrode was placed on the patella.
Experimental procedures.
Once a suitable image of the artery was found, 2 min of baseline data (Pre) were collected before exercise began. During pilot testing, it was discovered that sudden, intense dorsiflexion contractions caused the anterior tibial artery to shift position in relation to the probe in some subjects. To improve tracking of the artery, subjects used torque feedback displayed on an oscilloscope to gradually ramp up to the desired contraction intensity over a 5-s period.
On day 1, subjects performed a MVC and continued to contract maximally for 60 s. Strong verbal encouragement and visual torque feedback were provided throughout the minute. Three minutes after the end of the sustained contraction, subjects performed another ramped MVC. This MVC was used to assess the recovery of torque and was maintained for ∼2 s once torque reached a plateau.
The experimental setup and procedures were similar for day 2, except subjects were instructed to ramp their torque to a line on the oscilloscope that corresponded to 30% of their day 1 MVC and to match that target line as closely as possible for 60 s. Following the brief MVC in the recovery period, the subject rested for 15 min and then repeated the same test sequence, but at 60% of his day 1 MVC. During the rest period between the 30% and 60% tasks, HR, MAP, MBV, and tissue oxygenation index (TOI) measures all returned to baseline resting values.
To determine the minimal oxygen saturation value in the dorsiflexors, a subset of participants (n = 8) participated in an additional experiment on day 3. Participants were seated in the same position as previous experimental sessions; however, a blood pressure cuff was placed around their right thigh to occlude blood flow to the leg. The NIRS unit was affixed to the subject's leg in the same location as in previous sessions. Blood flow was occluded to the leg by inflating the cuff beyond 240 mmHg, and this occlusion was maintained until oxygen saturation researched a stable minimum level.
Data analysis and statistics.
HR, MAP, and MBV data were stored on personal computer and analyzed off-line (second-by-second) using Chart software (version 5; ADInstruments, Colorado Springs, CO). In addition to storage of these data on the computer, ultrasound data from each trial were recorded on S-VHS tape for the off-line measurement of the anterior tibial artery diameter. Using electronic calipers within the ultrasound software, a single investigator measured vessel diameter during the baseline period (pre-exercise) and at 5, 15, 25, 35, 45, and 55 s of exercise. At each time point, three separate diameter measures were taken, and the mean value was used in further analyses. To calculate mean blood flow during exercise, diameter data were combined with MBV (10 s of velocity data centered about the diameter measurement) using the following equation: [MBF (ml/min) = MBV (cm/s) × πr2 (cm2) × 60], where r is the radius of the anterior tibial artery. Subsequently, vascular conductance was calculated as MBF ÷ MAP.
NIRS oxygen saturation (tissue oxygen index, or TOI) data were also recorded and transferred to a personal computer for off-line analysis. TOI represents the ratio of oxygenated to total hemoglobin (expressed as a percentage). The half-time of oxygen desaturation (from Pre to the minimum value reached during exercise) was calculated to assess the rate of decrease in oxygen saturation at the onset of exercise. Torque and EMG data were analyzed using Spike 2 software (version 4.13; Cambridge Electronic Design, Cambridge, UK). Maximal torque was classified as the peak value attained at the onset of the sustained 100% MVC contraction. To match cardiovascular measures, torque was averaged over 10-s intervals during exercise. These mean torque values were then normalized to the peak value attained at the onset of the sustained 100% MVC contraction on day 1. Similar to torque data, root mean square EMG amplitude was averaged over 10-s intervals during exercise. EMG data during the 100% MVC contraction were normalized to the mean value over the first 10 s. Because the submaximal contractions (30 and 60% MVC) were performed on a separate day (and, hence, electrode placement was not identical), EMG data during the 30 and 60% MVC contractions were normalized to the greatest value over 1 s from the recovery MVC performed 3 min after the 30% MVC contraction.
Two-way repeated-measures ANOVAs, with contraction intensity and time as within-subject variables, were used to compare HR, MAP, arterial diameter, MBV, MBF, vascular conductance, and TOI among contraction intensities (SPSS version 21). All measures except MAP had a significant intensity × time interaction. Thus, at each intensity, results of paired-samples t-tests were compared with a two-tailed Dunnett's table to determine which time points in exercise were different from Pre. For MAP data during exercise, a Bonferroni correction factor was applied to the multiple time comparisons in post hoc testing. One-way repeated-measures ANOVAs were used to compare the half-time of oxygen desaturation among contraction intensities. Separate one-way repeated-measures ANOVAs were used to test for a main effect of time on torque and EMG during the 100% MVC contraction, as well as EMG during the 30 and 60% MVC contractions. Paired-samples t-tests were used to compare the torque of each recovery MVC to the peak value of the 100% MVC contraction on day 1. All data are reported in the text as group mean ± SD. The significance level was P < 0.05.
RESULTS
Torque and EMG.
Maximal torque was 39.4 ± 4.4 Nm at the onset of the 100% MVC contraction. Torque declined progressively during the sustained maximal contraction (P < 0.001) and was 60% of the initial value by the final 10 s of exercise (Fig. 1). As intended, submaximal torques of 30 and 60% MVC were held constant throughout exercise (Fig. 1). Peak torque of the recovery MVC was equivalent to the maximal value at the onset of the 100% MVC contraction for all levels of contraction (P > 0.05; 103.1 ± 4.8, 100.1 ± 7.1, 96.1 ± 6.2% for 30, 60, and 100% MVC, respectively). EMG activity did not change during the 30 or 100% MVC contractions but increased significantly during the 60% MVC contraction (P < 0.001; Fig. 1).
Fig. 1.
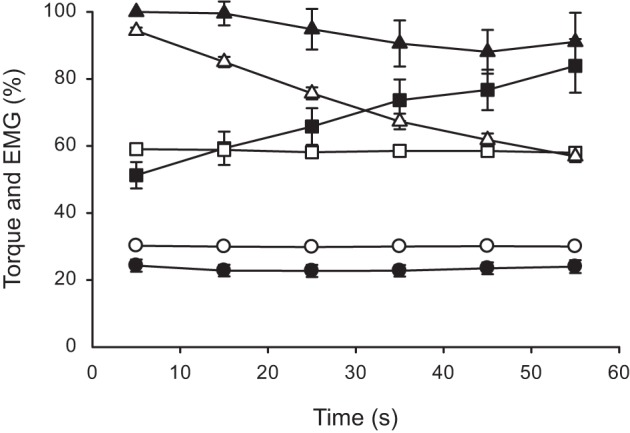
Normalized dorsiflexor torque and tibialis anterior electromyographic activity (EMG) during sustained isometric contractions at different levels of maximal torque. Values are expressed as means ± SE. Contractions were performed at 30 (○), 60 (□), and 100% (△) of maximum voluntary contraction (MVC) torque. Open symbols are torque, whereas solid symbols are EMG data. Torque data are normalized to the peak value obtained at the start of 100% MVC contraction. EMG data during the 30 and 60% MVC contractions are normalized to the value obtained during the brief MVC performed in the recovery period following the 30% MVC contraction. For the 100% MVC contraction, EMG data are normalized to the mean value from the first 10 s of exercise.
HR and MAP.
Heart rate responses to the exercise were graded with contraction intensity. The two-way repeated-measures ANOVA revealed main effects of intensity and time, as well as an interaction between the two variables (all P < 0.001). Specifically, HR was unchanged at 30% MVC but elevated by 26.2 ± 14.7% at 60% MVC and even higher (59.8 ± 14.4%) at 100% MVC (Fig. 2A). A significant main effect of time indicated an increase of MAP during exercise (P < 0.001), but no differences were observed among the three contraction intensities (Fig. 2B).
Fig. 2.
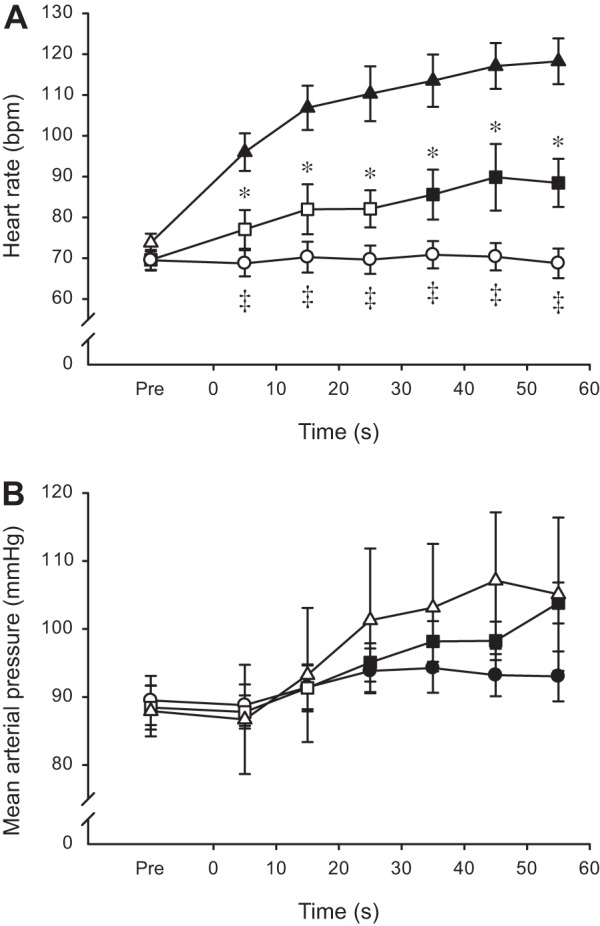
Heart rate (A) and mean arterial pressure (B) during sustained isometric contractions at different levels of maximal torque. Values are expressed as means ± SE. Contractions were performed at 30 (○), 60 (□), and 100% (△) of MVC torque. Solid symbols indicate data points that are significantly different from Pre (P < 0.05). Asterisks (*) denote time points when 60% MVC differs significantly from 100% MVC (P < 0.05). Double daggers (‡) denote time points when 30% is significantly different from both 60 and 100% MVC (P < 0.05).
Diameter, MBV, MBF, and vascular conductance.
Anterior tibial artery diameter was affected by both contraction intensity (P = 0.002) and time (P < 0.001). There was no change in diameter at 30% MVC, but the diameter was significantly compressed during the early stages of the contractions at 60 and 100% MVC, which led to differences among the three levels of contraction intensity (Fig. 3A). The patterns of change were nearly identical for MBV (Fig. 3B) and MBF (Fig. 3C), so only MBF data are described here. There were main effects of intensity and time and an interaction between the two variables (all P < 0.001), and consequently, the three contraction levels showed distinctive MBF responses to exercise. Blood flow began to increase from the onset at 30% MVC, whereas there was an initial reduction in flow at 60 and 100% MVC (not statistically significant at 60% MVC). After this transient decrease during the first 10 s, flow increased slowly at 60% MVC, but rapidly at 100% MVC, such that flow was similar at 30 and 100% MVC, and both had greater flow than 60% MVC in the second half of exercise (Fig. 3C). Changes to vascular conductance (Fig. 4) were remarkably similar to those seen for MBV and MBF. That is, there was an immediate increase at 30% MVC but an initial reduction at 60 and 100% MVC. Conductance was significantly greater at 30 than 60% MVC throughout exercise, whereas 100% was less than 30% for the initial 20 s, but similar to 30% and greater than 60% for the second half of the fatigue protocol. After cessation of the contraction, there was a large and immediate hyperemic response. The peak value for MBF after 30% MVC (185.5 ± 53.9 ml/min) was lower (P < 0.001) than the equivalent values after 60 and 100% MVC (280.3 ± 67.0 and 253.8 ± 49.1 ml/min, respectively) (data not shown).
Fig. 3.
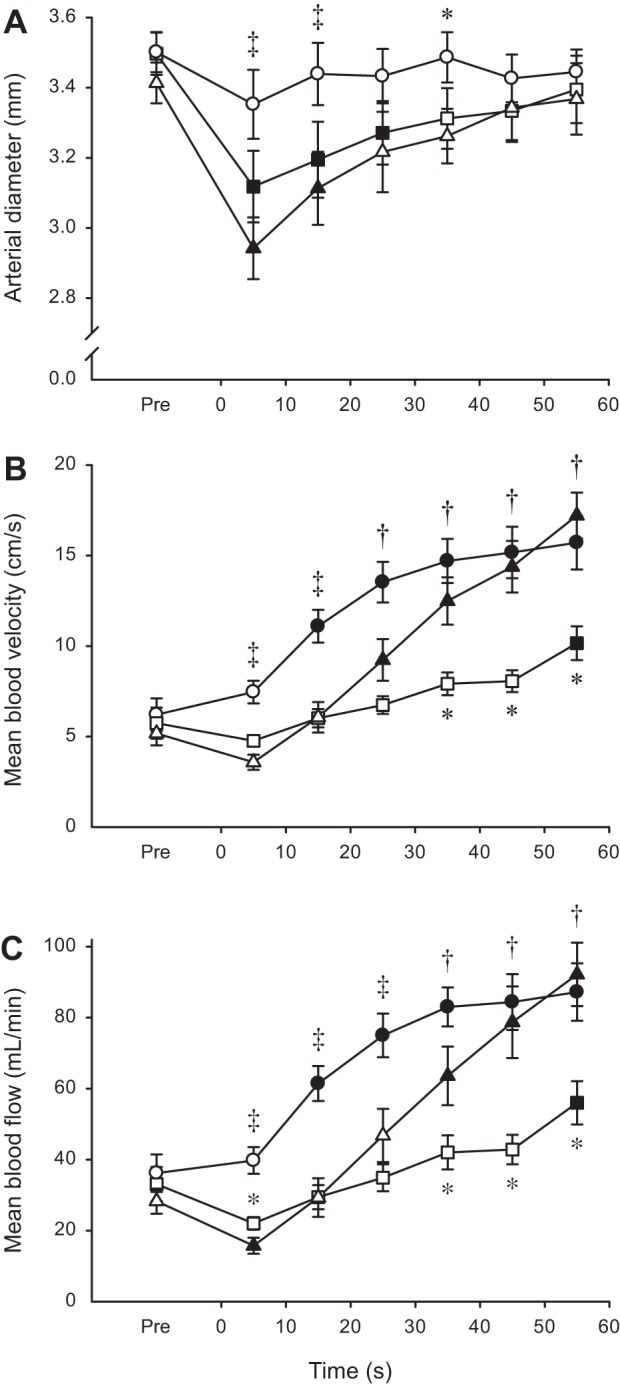
Anterior tibial artery diameter (A), mean blood velocity (B), and mean blood flow (C) during sustained isometric contractions at different levels of maximal torque. Values are expressed as means ± SE. Contractions were performed at 30 (○), 60 (□), and 100% (△) of MVC torque. Solid symbols indicate data points that are significantly different from Pre (P < 0.05). Asterisks (*) denote time points when either 30 or 60% MVC differs significantly from 100% MVC (P < 0.05). Daggers (†) denote time points when 30% is significantly different from 60% MVC, whereas double daggers (‡) denote time points when 30% is significantly different from both 60 and 100% MVC (P < 0.05).
Fig. 4.
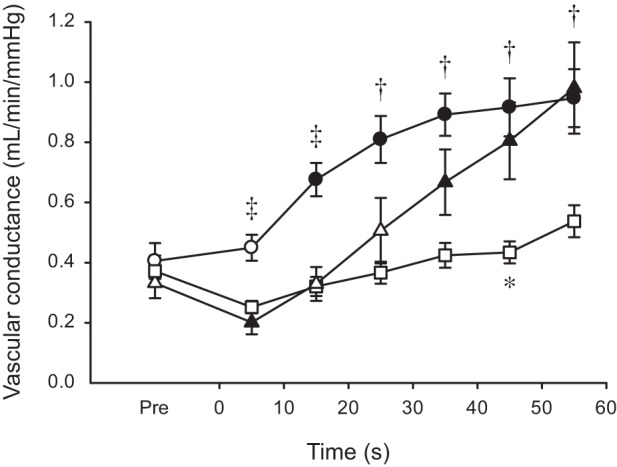
Anterior tibial artery vascular conductance during sustained isometric contractions at different levels of maximal torque. Values are expressed as means ± SE. Contractions were performed at 30 (○), 60 (□), and 100% (△) of MVC torque. Solid symbols indicate data points that are significantly different from Pre (P < 0.05). The asterisk (*) denotes the time point when 60% MVC differs significantly from 100% MVC (P < 0.05). Daggers (†) denote time points when 30% is significantly different from 60% MVC, whereas double daggers (‡) denote time points when 30% is significantly different from both 60 and 100% MVC (P < 0.05).
Oxygen saturation.
The rate of decrease in TOI at the onset of exercise, as assessed by the half-time of desaturation, was progressively faster (P < 0.001), with increasing contraction intensity (21.8 ± 2.9, 16.0 ± 1.6, 12.3 ± 1.2 s for 30, 60, and 100% MVC, respectively). Results of the two-way ANOVA indicate TOI was affected by both intensity and time and that the two factors interacted with one another (all P < 0.001). With the exception of the first 10 s at 30% MVC, oxygen saturation was lower than baseline throughout exercise (Fig. 5). Saturation was higher at 30% compared with both 60 and 100% MVC at all time points and higher at 60 than 100% MVC during the first 20 s of exercise (Fig. 5). Finally, with maintained muscle ischemia (day 3), oxygen saturation reached a nadir of 14.3 ± 6.4%. This value was significantly lower than the minimal levels reached during the 60 and 100% MVC fatiguing contractions (P < 0.05). The large postexercise increase in MBF was reflected in the saturation data. The peak value was lower after 30 than 100% MVC (109.2 ± 6.9% and 120.2 ± 9.0% of the resting value, respectively; P < 0.05), whereas 60% MVC (116.4 ± 11.4) was equivalent to the other intensities (data not shown).
Fig. 5.
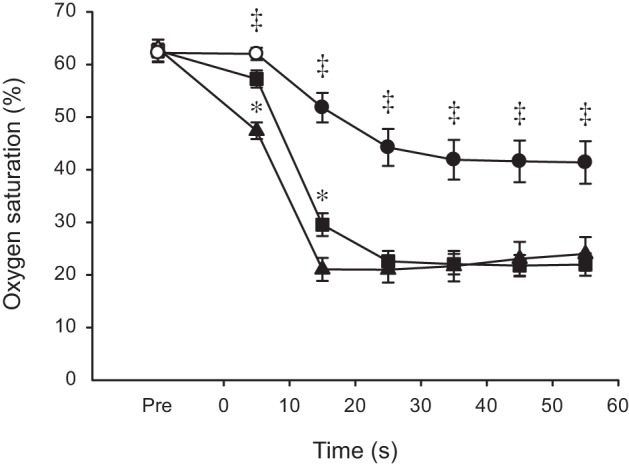
Dorsiflexor oxygen saturation during sustained isometric contractions at different levels of maximal torque. Values are expressed as means ± SE. Contractions were performed at 30 (○), 60 (□), and 100% (△) of MVC torque. Solid symbols indicate data points that are significantly different from Pre (P < 0.05). Asterisks (*) denote time points when 60% MVC differs significantly from 100% MVC (P < 0.05). Double daggers (‡) denote time points when 30% is significantly different from both 60 and 100% MVC (P < 0.05).
DISCUSSION
The major findings of the study were 1) blood flow was not occluded at the level of the conduit artery during any of the contraction intensities, and, indeed, it was increased overall from rest during the majority of the latter half of the sustained contractions; 2) blood flow was not linearly graded according to contraction intensity; 3) despite continued or increased blood flow through the conduit artery during the contraction, local muscle oxygenation levels declined rapidly and considerably; and 4) the pattern and extent of muscle deoxygenation were similar at moderate (60% MVC) and maximal contraction intensities.
Occlusion caused by intramuscular pressure.
In a variety of muscle groups (e.g., elbow flexors, knee extensors, plantar flexors, and dorsiflexors), data collected indirectly using isotope clearance (22), VOP (25), or fMRI (31) suggest that the IMP generated during isometric exercise will occlude MBF at a relative contraction strength of 50–65% MVC (60% MVC in the dorsiflexors of young males; Ref. 31). We expected that ultrasound of the narrow (3.5 ± 0.2 mm) anterior tibial artery would enable confirmation of this result via direct measurement of blood flow during a sustained isometric contraction. However, our data illustrate that even a maximal contraction of the dorsiflexors fails to generate sufficient IMP to cause more than transient reductions (<10 s) in diameter and blood flow through the artery (14% and 40%, respectively). Nevertheless, the similarity of NIRS-derived oxygen saturation at 60 and 100% MVC during exercise (Fig. 5) and the comparable postexercise hyperemic response of MBF and TOI may (see below for a conflicting perspective) support the suggestion (31) that IMP at 60% MVC is sufficient to occlude blood flow to the contracting muscle, most likely at the lower-order arterioles or the capillary bed (15, 28).
Blood flow and oxygen saturation.
It has been established that, during dynamic exercise, blood flow increases to meet the demands of the working muscle (2, 19, 23). However, the results of this study and other studies (7, 13, 27, 30, 31) suggest that the same relationship does not exist during sustained isometric exercise. In the present study, there was a progressive increase in blood flow at 30% MVC, no change from rest until an increase in the final 10 s of exercise at 60% MVC, and an initial decrease followed by a progressive increase at 100% MVC (Fig. 3). Thus, our results illustrate a nonlinear gradation and differential time course of blood flow among low (30% MVC), moderate (60% MVC), and maximal (100% MVC) contractions.
In accordance with data collected at comparable contraction intensities in other muscle groups (7–9, 13, 27), a sustained 30% MVC contraction of the dorsiflexors induced a progressive increase in blood flow in the conduit artery, presumably to attempt to meet the energy demands of the task. However, despite the increase in blood flow, oxygen saturation was ∼30% lower than the resting value for the final 45 s of contraction. Hicks et al. (9) reported a comparable reduction in oxygen saturation during wrist flexion at 30% MVC, albeit after a greater delay than in the present study. The absence of even a transient reduction in artery diameter or blood flow suggests that IMP had a negligible impact at this contraction intensity.
Unlike comparable increases in blood flow reported at low (25 or 30% MVC) and moderate contraction intensities (50 or 70% MVC) in the much larger femoral artery (7) or the brachial artery of females (13), we saw marked differences in anterior tibial artery flow between 30 and 60% MVC. Thompson et al. (27) demonstrated that sex-related differences in strength impact blood flow, as they found equivalent increases in brachial artery flow at 20 and 50% MVC for females (similar to Refs. 7 and 13), whereas their data collected in males mirror our findings at 30 and 60% MVC. Despite presumed occlusion of flow to the muscle tissue (31), a 1-min contraction at 60% MVC failed to alter anterior tibial artery blood flow until an increase during the final 10 s of exercise. This suggests that 1) the energy demands of the contraction are almost met by anaerobic pathways and oxidative phosphorylation fueled by the oxygen trapped in the muscle at the onset of exercise, or 2) flow to the muscle is not completely occluded (more on this below). Increases in HR and MAP that occurred within the first 20–40 s of exercise indicate a pressor response. The magnitude was rather modest as it did not translate to an increase in blood flow until the end of exercise.
In contrast, the maximal contraction induced a pressor response large enough to increase anterior tibial artery blood flow above resting levels after 30 s of exercise despite the 40% reduction in flow observed during the first 10 s. Although we did not measure IMP in this study, the initial transient decreases in anterior tibial artery diameter and blood flow, as well as vascular conductance, suggest that IMP at the onset of exercise was sufficiently high to overcome the exercise-induced demand for increased MBF. Unlike the submaximal tasks during which torque was held constant, the maximal contraction resulted in a linear decrease in torque from 100 to 60%. The presumed decline in IMP is likely to have facilitated the ability of the concurrent metaboreflex-induced pressor response to increase blood flow to the muscle. Torque remained above 60% MVC for the entire minute, so the elevated flow in the conduit artery may not have aided (entered) the contracting muscle (31). However, oxygen saturation reached a steady-state minimum value (∼21%), which failed to match the minimum achieved with muscle ischemia (14%). This suggests that the enhanced blood flow was reaching the muscle tissue because the reliance on oxidative phosphorylation increases markedly during a sustained 1-min dorsiflexor MVC (16). Moreover, despite greater torque production (energy demand) and blood flow in the conduit artery at 100 compared with 60% MVC, TOI was equivalent beyond 20 s of contraction (Fig. 5). NIRS-derived oxygen saturation is believed to be representative of the balance between O2 delivery and extraction, so the data suggest that some of the added blood available at 100% MVC reached the contracting muscle, or else the TOI score would have been lower than at 60% MVC. However, it cannot be ruled out that there is a technological component to the results. That is, the NIRS signal represents a weighted average of venous and arterial blood (3), and an exercise-induced shift in the balance could influence the TOI value.
Methodological considerations.
We observed exercise-induced increases in blood flow at the level of the conduit artery, but measurement of gross flow at this site does not indicate how much of the added blood actually perfused the contracting muscle. The concurrent examination of muscle oxygenation allowed us to speculate on perfusion, but the combination of Doppler ultrasound and NIRS does not approach the precision of PET, which can show the distribution of blood in a muscle during a sustained isometric contraction (14, 20, 21).
Although both appropriate and necessary for the current research question, the 1-min contraction duration (imposed by the ability to maintain a constant torque at 60% MVC) precluded a more complete investigation of competing mechanisms during sustained low-intensity contraction.
Perspectives and Significance
The results from this study indicate that, even during a maximal contraction of a distal muscle group housed in a restrictive fascial sheath, increases in IMP do not greatly impede blood flow at the level of a narrow conduit artery. Equivalent oxygen saturation during exercise as well as blood flow after exercise at 60 and 100% MVC may support the proposed (31) occlusion of blood flow to the dorsiflexor muscle tissue at 60% MVC, but downstream of our recording site. However, data from the same research group that showed a marked increase in oxidative phosphorylation during a 1-min dorsiflexor MVC in young males (16) lead us to interpret our blood flow and oxygen saturation data as evidence that some blood reaches contracting muscle fibers even at MVC. Finally, our results show, in contrast to dynamic (2, 19) or intermittent isometric (11, 32) exercise, blood flow is not linearly graded with exercise intensity nor directly linked with muscle oxygenation during sustained isometric contractions. Thus, similar to neuromuscular fatigue itself, blood flow responses to sustained isometric exercise demonstrate clear task dependency.
GRANTS
This work was supported by the Natural Sciences and Engineering Research Council of Canada
DISCLOSURES
No conflicts of interest, financial or otherwise, are declared by the authors.
AUTHOR CONTRIBUTIONS
Author contributions: C.J.M., E.O., and C.L.R. conception and design of research; C.J.M. and E.O. performed experiments; C.J.M. and E.O. analyzed data; C.J.M., M.D.A., E.O., J.K.S., and C.L.R. interpreted results of experiments; C.J.M. prepared figures; C.J.M., M.D.A., and E.O. drafted manuscript; C.J.M., M.D.A., E.O., J.K.S., and C.L.R. edited and revised manuscript; C.J.M., M.D.A., E.O., J.K.S., and C.L.R. approved final version of manuscript.
ACKNOWLEDGMENTS
The authors thank Dr. Jason Loeppky for his assistance with statistical analyses.
REFERENCES
- 1.Allen DG, Lamb GD, Westerblad H. Skeletal muscle fatigue: cellular mechanisms. Physiol Rev 88: 287–332, 2008. [DOI] [PubMed] [Google Scholar]
- 2.Andersen P, Saltin B. Maximal perfusion of skeletal muscle in man. J Physiol 366: 233–249, 1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bickler PE, Feiner JR, Rollins MD. Factors affecting the performance of 5 cerebral oximeters during hypoxia in healthy volunteers. Anes Analg 117: 813–823, 2013. [DOI] [PubMed] [Google Scholar]
- 4.Bigland-Ritchie B, Rice CL, Garland SJ, Walsh ML. Task-dependent factors in fatigue of human voluntary contractions. In: Fatigue, edited by Gandevia SC, Enoka RM, McComas AJ, Stuart DG, and Thomas CK. New York: Plenum, 1995, p. 361–380. [DOI] [PubMed] [Google Scholar]
- 5.Degens H, Salmons S, Jarvis JC. Intramuscular pressure, force and blood flow in rabbit tibialis anterior muscles during single and repetitive contractions. Eur J Appl Physiol Occup Physiol 78: 13–19, 1998. [DOI] [PubMed] [Google Scholar]
- 6.Enoka RM, Duchateau J. Muscle fatigue: what, why and how it influences muscle function. J Physiol 586: 11–23, 2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Gaffney FA, Sjogaard G, Saltin B. Cardiovascular and metabolic responses to static contraction in man. Acta Physiol Scand 138: 249–258, 1990. [DOI] [PubMed] [Google Scholar]
- 8.Griffin L, Garland SJ, Ivanova T, Hughson RL. Blood flow in the triceps brachii muscle in humans during sustained submaximal isometric contractions. Eur J Appl Physiol 84: 432–437, 2001. [DOI] [PubMed] [Google Scholar]
- 9.Hicks A, McGill S, Hughson RL. Tissue oxygenation by near-infrared spectroscopy and muscle blood flow during isometric contractions of the forearm. Can J Appl Physiol 24: 216–230, 1999. [DOI] [PubMed] [Google Scholar]
- 10.Hughson RL, Shoemaker JK, Tschakovsky ME, Kowalchuk JM. Dependence of muscle V̇o2 on blood flow dynamics at onset of forearm exercise. J Appl Physiol 81: 1619–1626, 1996. [DOI] [PubMed] [Google Scholar]
- 11.Hunter SK, Griffith EE, Schlachter KM, Kufahl TD. Sex differences in time to task failure and blood flow for an intermittent isometric fatiguing contraction. Muscle Nerve 39: 42–53, 2009. [DOI] [PubMed] [Google Scholar]
- 12.Hunter SK, Schletty JM, Schlachter KM, Griffith EE, Polichnowski AJ, Ng AV. Active hyperemia and vascular conductance differ between men and women for an isometric fatiguing contraction. J Appl Physiol 101: 140–150, 2006. [DOI] [PubMed] [Google Scholar]
- 13.Kagaya A, Homma S. Brachial arterial blood flow during static handgrip exercise of short duration at varying intensities studied by a Doppler ultrasound method. Acta Physiol Scand 160: 257–265, 1997. [DOI] [PubMed] [Google Scholar]
- 14.Kalliokoski KK, Laaksonen MS, Takala TO, Knuuti J, Nuutila P. Muscle oxygen extraction and perfusion heterogeneity during continuous and intermittent static exercise. J Appl Physiol 94: 953–958, 2003. [DOI] [PubMed] [Google Scholar]
- 15.Laaksonen MS, Kalliokoski KK, Kyrolainen H, Kemppainen J, Teras M, Sipila H, Nuutila P, Knuuti J. Skeletal muscle blood flow and flow heterogeneity during dynamic and isometric exercise in humans. Am J Physiol Heart Circ Physiol 284: H979–H986, 2003. [DOI] [PubMed] [Google Scholar]
- 16.Lanza IR, Befroy DE, Kent-Braun JA. Age-related changes in ATP-producing pathways in human skeletal muscle in vivo. J Appl Physiol 99: 1736–1744, 2005. [DOI] [PubMed] [Google Scholar]
- 17.Marsh E, Sale D, McComas AJ, Quinlan J. Influence of joint position on ankle dorsiflexion in humans. J Appl Physiol 51: 160–167, 1981. [DOI] [PubMed] [Google Scholar]
- 18.McCully KK, Hamaoka T. Near-infrared spectroscopy: what can it tell us about oxygen saturation in skeletal muscle? Exerc Sport Sci Rev 28: 123–127, 2000. [PubMed] [Google Scholar]
- 19.Radegran G. Ultrasound Doppler estimates of femoral artery blood flow during dynamic knee extensor exercise in humans. J Appl Physiol 83: 1383–1388, 1997. [DOI] [PubMed] [Google Scholar]
- 20.Rudroff T, Kalliokoski KK, Block DE, Gould JR, Klingensmith WC III, Enoka RM. PET/CT imaging of age- and task-associated differences in muscle activity during fatiguing contractions. J Appl Physiol 114: 1211–1219, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Rudroff T, Weissman JA, Bucci M, Seppanen M, Kaskinoro K, Heinonen I, Kalliokoski KK. Positron emission tomography detects greater blood flow and less blood flow heterogeneity in the exercising skeletal muscles of old compared with young men during fatiguing contractions. J Physiol 592: 337–349, 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Sadamoto T, Bonde-Petersen F, Suzuki Y. Skeletal muscle tension, flow, pressure, and EMG during sustained isometric contractions in humans. Eur J Appl Physiol Occup Physiol 51: 395–408, 1983. [DOI] [PubMed] [Google Scholar]
- 23.Saltin B, Radegran G, Koskolou MD, Roach RC. Skeletal muscle blood flow in humans and its regulation during exercise. Acta Physiol Scand 162: 421–436, 1998. [DOI] [PubMed] [Google Scholar]
- 24.Sejersted OM, Hargens AR, Kardel KR, Blom P, Jensen O, Hermansen L. Intramuscular fluid pressure during isometric contraction of human skeletal muscle. J Appl Physiol 56: 287–295, 1984. [DOI] [PubMed] [Google Scholar]
- 25.Sjogaard G, Savard G, Juel C. Muscle blood flow during isometric activity and its relation to muscle fatigue. Eur J Appl Physiol Occup Physiol 57: 327–335, 1988. [DOI] [PubMed] [Google Scholar]
- 26.Sylvest O, Hvid N. Pressure measurements in human striated muscles during contraction. Acta Rheumatol Scand 5: 216–222, 1959. [DOI] [PubMed] [Google Scholar]
- 27.Thompson BC, Fadia T, Pincivero DM, Scheuermann BW. Forearm blood flow responses to fatiguing isometric contractions in women and men. Am J Physiol Heart Circ Physiol 293: H805–H812, 2007. [DOI] [PubMed] [Google Scholar]
- 28.van Beekvelt MC, van Engelen BG, Wevers RA, Colier WN. In vivo quantitative near-infrared spectroscopy in skeletal muscle during incremental isometric handgrip exercise. Clin Physiol Funct Imaging 22: 210–217, 2002. [DOI] [PubMed] [Google Scholar]
- 29.Vollestad NK, Wesche J, Sejersted OM. Gradual increase in leg oxygen uptake during repeated submaximal contractions in humans. J Appl Physiol 68: 1150–1156, 1990. [DOI] [PubMed] [Google Scholar]
- 30.Wesche J. The time course and magnitude of blood flow changes in the human quadriceps muscles following isometric contraction. J Physiol 377: 445–462, 1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Wigmore DM, Damon BM, Pober DM, Kent-Braun JA. MRI measures of perfusion-related changes in human skeletal muscle during progressive contractions. J Appl Physiol 97: 2385–2394, 2004. [DOI] [PubMed] [Google Scholar]
- 32.Wigmore DM, Propert K, Kent-Braun JA. Blood flow does not limit skeletal muscle force production during incremental isometric contractions. Eur J Appl Physiol 96: 370–378, 2006. [DOI] [PubMed] [Google Scholar]


