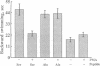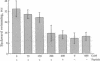Abstract
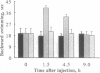
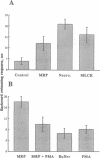
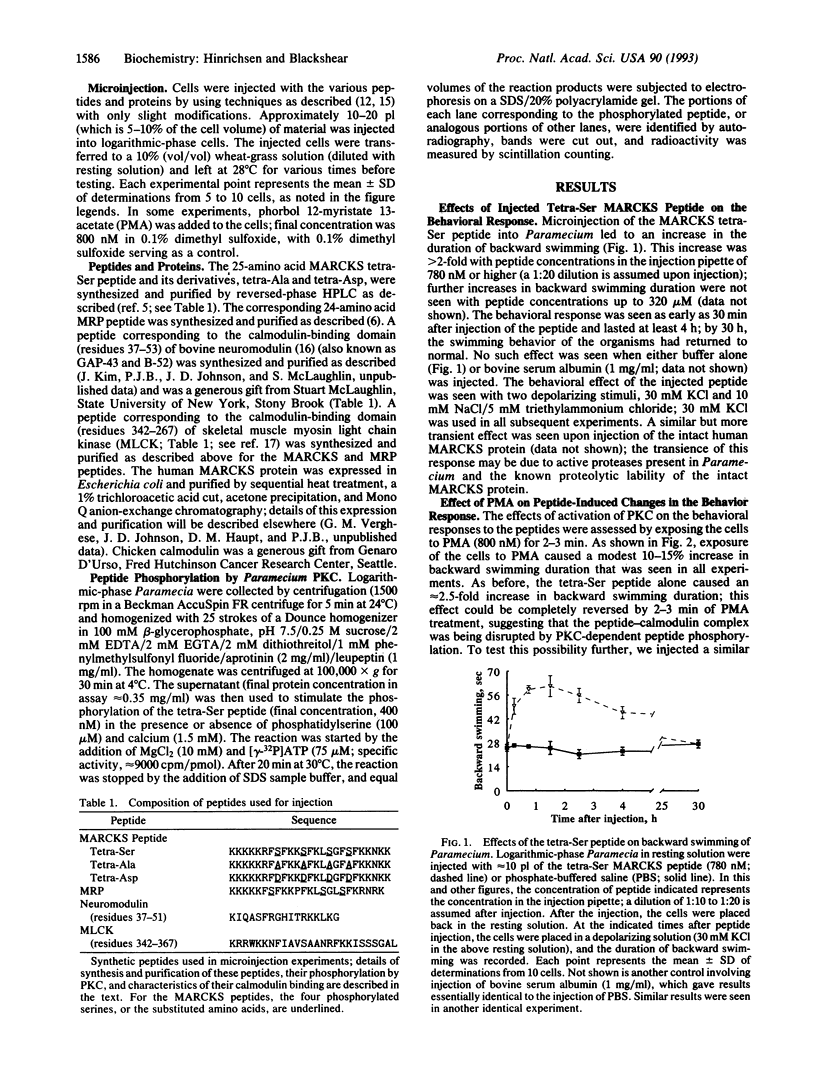
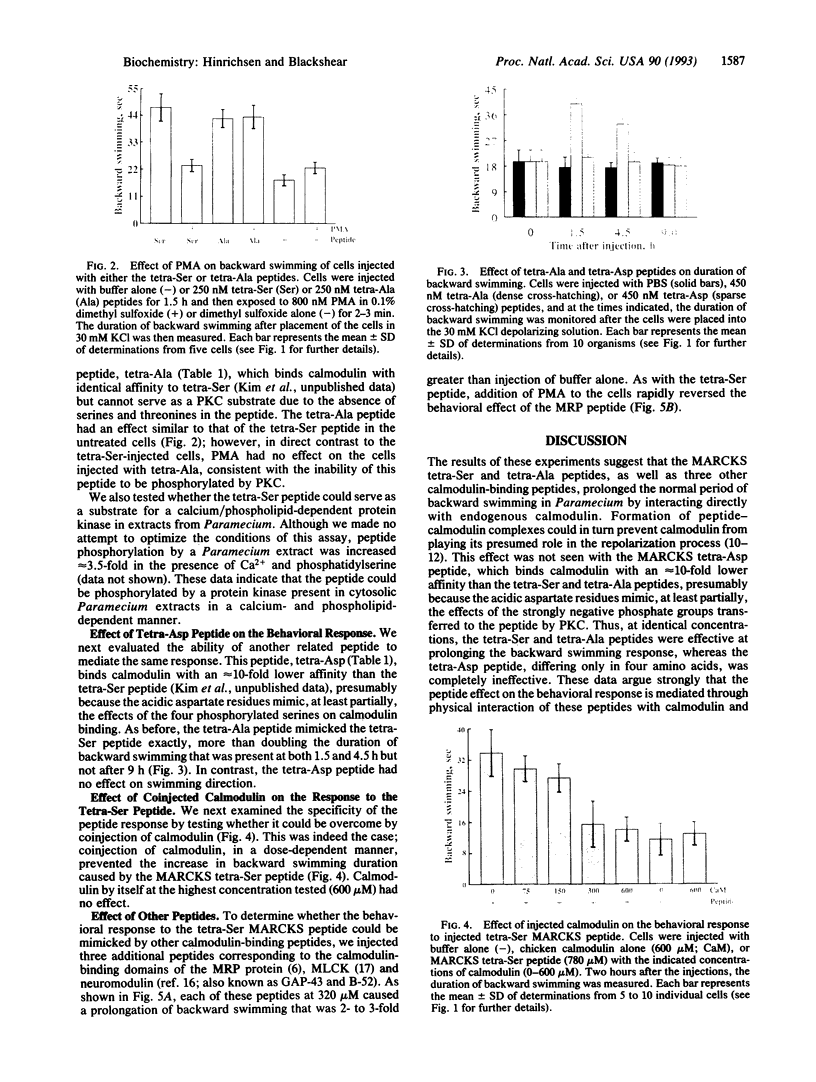
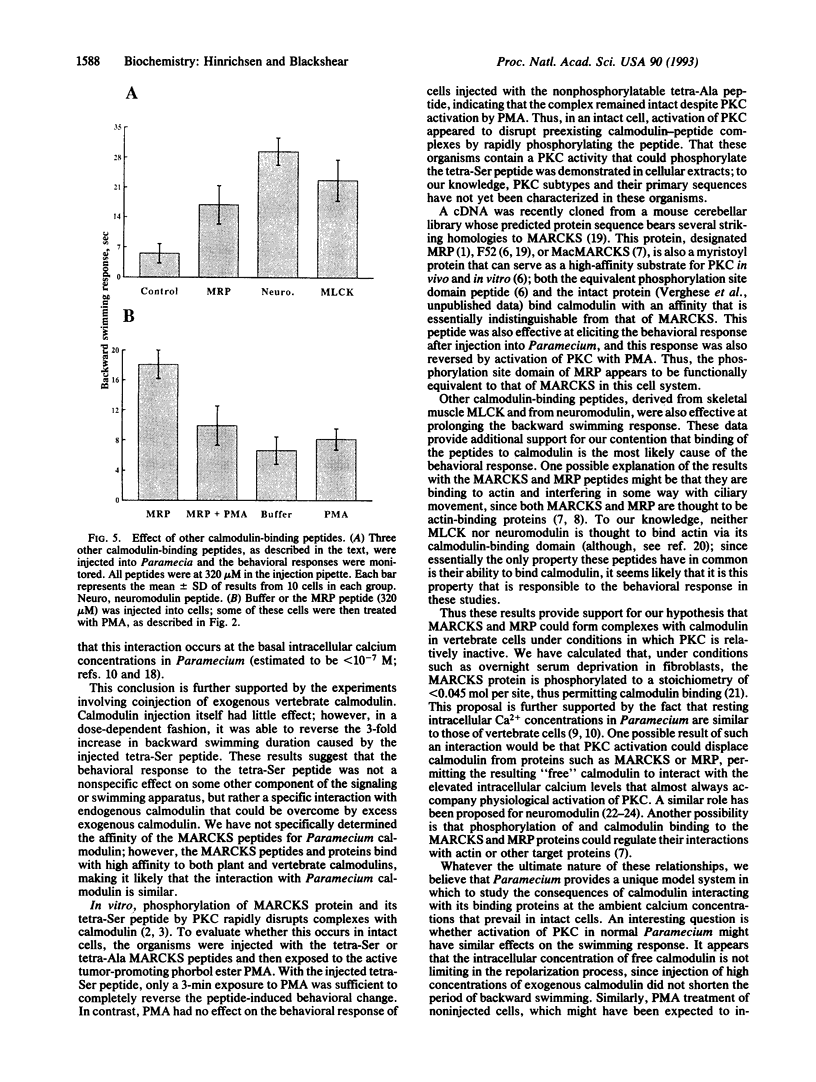
We used the freshwater protozoan Paramecium tetraurelia to investigate the potential regulation by protein kinase C of calmodulin interactions with binding peptides in intact cells. In these organisms, an action potential results in membrane depolarization and a period of backward swimming; repolarization and a return to forward swimming requires the presence of normal calmodulin. We postulated that injection of high-affinity calmodulin binding peptides might interfere with repolarization and thus prolong the period of membrane depolarization. Synthetic peptides spanning the protein kinase C phosphorylation site/calmodulin-binding domains of the myristoylated alanine-rich C-kinase substrate (MARCKS) and the MARCKS-related protein (also known as F52 or MacMARCKS) were injected into cells; these caused a 2- to 3-fold increase in the duration of backward swimming. Similar changes were seen with two other calmodulin-binding peptides. This behavioral response could be prevented by coinjecting calmodulin. Activation of Paramecium protein kinase C with an active phorbol ester completely reversed (within 3 min) the behavioral effects of the normal MARCKS and MARCKS-related protein peptides. Injection of a nonphosphorylatable peptide, in which alanines were substituted for serines, resulted in the usual behavioral response; however, this was not reversed by phorbol ester treatment. The corresponding aspartate-substituted peptide, which has a 10-fold lower affinity for calmodulin, did not prolong backward swimming. These data suggest that these peptides can form complexes with calmodulin at the calcium concentrations that prevail in intact Paramecium cells and that such complexes can be disrupted by protein kinase C phosphorylation of the peptides.
Full text
PDF

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alexander K. A., Cimler B. M., Meier K. E., Storm D. R. Regulation of calmodulin binding to P-57. A neurospecific calmodulin binding protein. J Biol Chem. 1987 May 5;262(13):6108–6113. [PubMed] [Google Scholar]
- Blackshear P. J., Haupt D. M., Stumpo D. J. Insulin activation of protein kinase C: a reassessment. J Biol Chem. 1991 Jun 15;266(17):10946–10952. [PubMed] [Google Scholar]
- Blackshear P. J. The MARCKS family of cellular protein kinase C substrates. J Biol Chem. 1993 Jan 25;268(3):1501–1504. [PubMed] [Google Scholar]
- Blackshear P. J., Verghese G. M., Johnson J. D., Haupt D. M., Stumpo D. J. Characteristics of the F52 protein, a MARCKS homologue. J Biol Chem. 1992 Jul 5;267(19):13540–13546. [PubMed] [Google Scholar]
- Blumenthal D. K., Takio K., Edelman A. M., Charbonneau H., Titani K., Walsh K. A., Krebs E. G. Identification of the calmodulin-binding domain of skeletal muscle myosin light chain kinase. Proc Natl Acad Sci U S A. 1985 May;82(10):3187–3191. doi: 10.1073/pnas.82.10.3187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brehm P., Eckert R. Calcium entry leads to inactivation of calcium channel in Paramecium. Science. 1978 Dec 15;202(4373):1203–1206. doi: 10.1126/science.103199. [DOI] [PubMed] [Google Scholar]
- Graff J. M., Rajan R. R., Randall R. R., Nairn A. C., Blackshear P. J. Protein kinase C substrate and inhibitor characteristics of peptides derived from the myristoylated alanine-rich C kinase substrate (MARCKS) protein phosphorylation site domain. J Biol Chem. 1991 Aug 5;266(22):14390–14398. [PubMed] [Google Scholar]
- Graff J. M., Stumpo D. J., Blackshear P. J. Characterization of the phosphorylation sites in the chicken and bovine myristoylated alanine-rich C kinase substrate protein, a prominent cellular substrate for protein kinase C. J Biol Chem. 1989 Jul 15;264(20):11912–11919. [PubMed] [Google Scholar]
- Graff J. M., Young T. N., Johnson J. D., Blackshear P. J. Phosphorylation-regulated calmodulin binding to a prominent cellular substrate for protein kinase C. J Biol Chem. 1989 Dec 25;264(36):21818–21823. [PubMed] [Google Scholar]
- Hartwig J. H., Thelen M., Rosen A., Janmey P. A., Nairn A. C., Aderem A. MARCKS is an actin filament crosslinking protein regulated by protein kinase C and calcium-calmodulin. Nature. 1992 Apr 16;356(6370):618–622. doi: 10.1038/356618a0. [DOI] [PubMed] [Google Scholar]
- Hinrichsen R. D., Burgess-Cassler A., Soltvedt B. C., Hennessey T., Kung C. Restoration by calmodulin of a Ca2+-dependent K+ current missing in a mutant of Paramecium. Science. 1986 Apr 25;232(4749):503–506. doi: 10.1126/science.2421410. [DOI] [PubMed] [Google Scholar]
- Hinrichsen R. D., Pollock M., Hennessey T., Russell C. An intragenic suppressor of a calmodulin mutation in Paramecium: genetic and biochemical characterization. Genetics. 1991 Nov;129(3):717–725. doi: 10.1093/genetics/129.3.717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hinrichsen R., Wilson E., Lukas T., Craig T., Schultz J., Watterson D. M. Analysis of the molecular basis of calmodulin defects that affect ion channel-mediated cellular responses: site-specific mutagenesis and microinjection. J Cell Biol. 1990 Dec;111(6 Pt 1):2537–2542. doi: 10.1083/jcb.111.6.2537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li J., Aderem A. MacMARCKS, a novel member of the MARCKS family of protein kinase C substrates. Cell. 1992 Sep 4;70(5):791–801. doi: 10.1016/0092-8674(92)90312-z. [DOI] [PubMed] [Google Scholar]
- Liu Y. C., Storm D. R. Regulation of free calmodulin levels by neuromodulin: neuron growth and regeneration. Trends Pharmacol Sci. 1990 Mar;11(3):107–111. doi: 10.1016/0165-6147(90)90195-e. [DOI] [PubMed] [Google Scholar]
- McIlroy B. K., Walters J. D., Blackshear P. J., Johnson J. D. Phosphorylation-dependent binding of a synthetic MARCKS peptide to calmodulin. J Biol Chem. 1991 Mar 15;266(8):4959–4964. [PubMed] [Google Scholar]
- Meiri K. F., Gordon-Weeks P. R. GAP-43 in growth cones is associated with areas of membrane that are tightly bound to substrate and is a component of a membrane skeleton subcellular fraction. J Neurosci. 1990 Jan;10(1):256–266. doi: 10.1523/JNEUROSCI.10-01-00256.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Preston R. R., Kink J. A., Hinrichsen R. D., Saimi Y., Kung C. Calmodulin mutants and Ca2(+)-dependent channels in Paramecium. Annu Rev Physiol. 1991;53:309–319. doi: 10.1146/annurev.ph.53.030191.001521. [DOI] [PubMed] [Google Scholar]
- Schaefer W. H., Hinrichsen R. D., Burgess-Cassler A., Kung C., Blair I. A., Watterson D. M. A mutant Paramecium with a defective calcium-dependent potassium conductance has an altered calmodulin: a nonlethal selective alteration in calmodulin regulation. Proc Natl Acad Sci U S A. 1987 Jun;84(11):3931–3935. doi: 10.1073/pnas.84.11.3931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Skene J. H. Axonal growth-associated proteins. Annu Rev Neurosci. 1989;12:127–156. doi: 10.1146/annurev.ne.12.030189.001015. [DOI] [PubMed] [Google Scholar]
- Skene J. H. GAP-43 as a 'calmodulin sponge' and some implications for calcium signalling in axon terminals. Neurosci Res Suppl. 1990;13:S112–S125. doi: 10.1016/0921-8696(90)90040-a. [DOI] [PubMed] [Google Scholar]
- Umekage T., Kato K. A mouse brain cDNA encodes a novel protein with the protein kinase C phosphorylation site domain common to MARCKS. FEBS Lett. 1991 Jul 29;286(1-2):147–151. doi: 10.1016/0014-5793(91)80961-2. [DOI] [PubMed] [Google Scholar]