Abstract
Many neurological and psychiatric maladies originate from the deprivation of the human brain from estrogens. However, current hormone therapies cannot be used safely to treat these conditions commonly associated with menopause because of detrimental side-effects in the periphery. The latter also prevents the use of the hormone for neuroprotection. Here we show that a small-molecule bioprecursor prodrug, 10β,17β-dihydroxyestra-1,4-dien-3-one (DHED), converts to 17β-estradiol in the brain after systemic administration, but remains inert in the rest of the body. The localized and rapid formation of estrogen from the prodrug was revealed by a series of in vivo bioanalytical assays and through in vivo imaging in rodents. DHED treatment efficiently alleviated symptoms originated from brain estrogen deficiency in animal models of surgical menopause and provided neuroprotection in a rat stroke model. Concomitantly, we determined that 17β-estradiol formed in the brain from DHED elicited changes in gene expression and neuronal morphology identical to those obtained after direct 17β-estradiol treatment. Altogether, complementary functional and mechanistic data show that our approach is highly relevant therapeutically, because administration of the prodrug selectively produces estrogen in the brain independently from the route of administration and treatment regimen. Therefore, peripheral responses associated with the use of systemic estrogens, such as stimulation of the uterus and estrogen-responsive tumor growth, were absent. Collectively, our brain-selective prodrug approach may safely provide estrogen neuroprotection and medicate neurological and psychiatric symptoms developing from estrogen deficiency, particularly those encountered after surgical menopause, without the adverse side-effects of current hormone therapies.
Introduction
Many neurological and psychiatric symptoms originate from the deprivation of the human brain from estrogens (1). Hormone therapies, including systemically administered 17β-estradiol (E2, the main human estrogen), alleviate these conditions (2). E2 has also been shown to provide neuroprotection, as one of its best-documented non-reproductive functions, in animals (3). However, the full potential of the hormone for the treatment of estrogen-responsive central maladies cannot be realized in clinical settings until its actions are restricted to the brain. This is needed to ensure therapeutic safety, as adverse peripheral impact of estrogens has halted large-scale clinical trials investigating the long-term health benefits of hormone therapies based on equine estrogens (4). Many women discontinued these therapies or avoid starting them, which potentially increases the risks of poor brain health even in conditions where benefits of estrogen have been shown (5,6). Therefore, novel therapies providing effective and safe treatment of the brain with estrogen remain an unmet need in medicine.
Elevated circulating estrogen levels and associated risks for harmful peripheral side-effects are unavoidable with currently approved estrogen medications, even when human estrogen is prescribed (7). The development of brain-selective estrogen therapies has been, however, a formidable challenge. Efforts to discover neuroselective estrogen-receptor (ER) modulators are still focused mainly on a few well-known phytoestrogens (8), and metabolism of these compounds is variable (9). A recent development of a glucagon-like peptide-1 (GLP-1)–E2 conjugate has shown potential brain-delivery through dual hormone action to improve energy, glucose, and lipid metabolism (10). The approach targets cells that, besides ERs, also express GLP-1 receptors. Therefore, suprahypothalamic areas of the CNS involved in many estrogen deficiency–derived neurological and psychiatric symptoms (6, 11) are not affected, because ER-expressing cells that co-express GLP-1 receptors are localized only in the hypothalamus and the brainstem. An additional caveat of the method is due to key technical hurdles involved in the pharmaceutical development of effective peptide-based agents—including but not limited to manufacture, oral absorption, metabolic stability, pharmacokinetics and formulation—compared with small-molecule drugs.
Hence, selective delivery of E2 into the brain through a simple small-molecule strategy would be of immediate practical relevance. Prodrug approaches to achieve this goal have, however, remained unsuccessful. Prodrugs are inactive derivatives of therapeutic agents that are converted to the biologically active parent drug by enzymatic and/or chemical transformations in vivo (12). They are commonly developed to resolve pharmacokinetic, toxicity, formulation, and drug delivery limitations. A redox chemical delivery system, which applies the prodrug principle (12), has been proposed for enhanced delivery of E2 to the brain (13). Although this method provides increased selectivity in CNS delivery of the hormone, it still generates elevated levels of the hormone in the periphery with substantial uterotrophic side-effects and sustained high circulating estrogen levels even after acute dosing (14, 15).
Our studies on the antioxidant mechanism of estrogen neuroprotection (16) inspired the hypothesis that 10β,17β-dihydroxyestra-1,4-dien-3-one (DHED) could serve as a bioprecursor prodrug of E2 with preferential bioactivation to E2 in the brain to fulfill the unmet need for brain-selective estrogen therapy. Unlike classical prodrugs, bioprecursor prodrugs do not contain auxiliary “promoieties” but are created via a transient chemical alteration within the drug molecule itself (12). As an α,β-unsaturated carbonyl compound, DHED is a plausible substrate for a short-chain NADPH-dependent dehydrogenase/reductase (17) that is selectively expressed in the brain and linked to neuroprotection (18). Using a series of bioanalytical and imaging assessments, evaluating gene expression, morphology, and neuropharmacological responses in several rodent models of neurological and psychiatric disorders, we show here that DHED selectively converts into E2 only in the brain after systemic administration. Most importantly, we reveal the therapeutic potential of DHED on estrogen deficiency-associated neurological disorders and psychiatric conditions without encountering adverse systemic actions that are typically seen with the direct use of estrogens.
Results
DHED conversion to estrogen (E2)
DHED is a bioprecursor prodrug that is converted to E2 by a short-chain dehydrogenase/reductase (Fig. 1A). The reductive bioactivation of DHED to E2 proceeds through hydride transfer from the coenzyme NADPH to the C1 position of the C1-C2 double bond of DHED’s A-ring that is conjugated to the 3-carbonyl group. This β-addition is followed by spontaneous water elimination involving the 10(β)-OH. Our computer-aided mechanistic study fully supported this pathway of DHED’s reductive bioactivation (Fig 1B). The first, enzyme-catalyzed hydride-transfer step leads to an intermediate through a transition state (which may be considered a resonance hybrid of the substrate–coenzyme and intermediate–oxidized coenzyme complexes shown in brackets) that is endergonic, with ΔG∘ of +2.1 kcal/mol. The subsequent water elimination is immensely exergonic (ΔG∘ of −10.3 kcal/mol); hence, it proceeds spontaneously making the overall process also exergonic with ΔG∘ of −8.2 kcal/mol. The obtained phenolate form of E2 (E2−) is in acid-base equilibrium with the non-ionized (neutral) form of the hormone dictated by pH, and the neutral form predominates in equilibrium at physiological pH. Altogether, DHED’s reductive bioactivation proceeds in an opposite direction compared to the well-known oxidative process converting an androgen to estrogen catalyzed by aromatase (19). In the latter case, three sequential steps of substrate oxidation involving molecular oxygen and the transfer of six electrons are necessary for estrogen formation from androgens (Fig 1C).
Fig. 1. DHED selectively converts to the main human estrogen E2.

(A) DHED’s reductive metabolism to E2 in the brain via an NAD(P)H-dependent short-chain dehydrogenase/reductase (SDR). (B) In silico mechanistic model for DHED bioactivation to E2. Quantum-chemical calculations simulating solvation with water were applied to the complete steroid structures and a mimic of NAD(P)H ⇌ NAD(P)+, with R chosen as methyl. (C) In contrast, the aromatase-catalyzed conversion of androgens to estrogens proceeds through sequential six-electron oxidation on the C19 methyl group involving molecular oxygen.
Table 1 summarizes key physicochemical, pharmacological, and biopharmaceutical properties of DHED in comparison with those of E2. DHED is inactive as an estrogen as it has no measurable affinity to the classical estrogen receptors (ERs), and does not show the well-known antioxidant activity of the hormone (16). Its lipophilicity and binding to human plasma proteins are significantly decreased, while its water-solubility is increased compared with E2. All these are great advantages over E2 in terms of bioavailability and formulation.
Table 1. Estrogen receptor affinity, antioxidant potency, plasma protein binding, and selected physicochemical properties of E2 and DHED.
Lipid peroxidation was determined using the FTC (ferric thiocyanate) and TBARS (thiobarbituric acid reactive substances) assays. logP denotes the logarithm of n-octanol/water partition coefficient as a measure of lipophilicity with values predicted by the method incorporated into the BioMedCAChe program (version 6.1). Data are averages ± SEM (n = 3). n.i. = no inhibition.
| Compound | Estrogen receptor (ER) binding IC50 (nM) | Inhibition of lipid peroxidation: IC50 (μM) | Binding to human plasma proteins (%) | Lipophilicity (logPcalc) | Intrinsic water solubility at 25 °C (μg/ml) | ||
|---|---|---|---|---|---|---|---|
| ERα | ERβ | FTC | TBARS | ||||
| E2 | 1.3 | 0.7 | 11.8 ± 1.6 | 3.9 ± 0.4 | 97.8 ± 0.3 | 4.01 | 4.2 ±0.3 |
| DHED | >10,000 | >10,000 | n.i. | n.i. | 58.0 ± 0.6 | 1.67 | 60.3 ±1.7 |
In vitro metabolism and in vivo conversion of DHED
In vitro metabolism studies were conducted by adding the prodrug to freshly prepared tissue homogenates. DHED was preferentially activated to E2 in the brain compared with estrogen-sensitive peripheral tissues, such as the uterus (Fig. 2A and fig. S1). Besides E2, we could not identify other steroid(s) formed from DHED.
Fig. 2. DHED is a brain-selective prodrug.
(A) Initial rate of in vitro E2 formation during DHED incubation (100 nM) in homogenates of various OVX rat brain structures versus rat uterus homogenate; the latter represents an estrogen-sensitive peripheral tissue. Data are averages ± SEM (n =3 per tissue). N/D, not detected. (B) Serum DHED and E2 concentrations in serum and in the brain of OVX Sprague-Dawley rats after i.v. administration of DHED (200 μg/kg). Concentrations were measured by LC-MS/MS-based bioassays. Serum concentrations are averages ± SEM (n = 3–4 animals per time point); brain concentrations are weighted averages ± SEM from measurements in seven brain regions (n = 3–4 animals per time point). E2 concentrations at time “0” originated from analyses of tissue and serum obtained from vehicle-treated animals euthanized immediately after injection. (C) Increase of E2 in brain tissue after DHED treatment arises from the metabolism of the bioprecursor prodrug. d3-DHED was administered to OVX Sprague-Dawley rats i.v. followed by tissue harvesting at 15 min post-dosing and LC–MS/MS analysis (using selected reaction monitoring, SRM). All SRM chromatograms were obtained from the analyses of hippocampal tissue and were scaled to the same ion abundance (5.0×104). The trace for the unlabeled (i.e., endogenous) E2 is obtained by SRM of m/z 506→171, the red trace (SRM: m/z 509→171) represents 16,16,17-d3-E2 formed from 16,16,17-d3-DHED in the hippocampus, and the green trace (SRM: m/z 512→171) corresponds to the 13C-labeled hormone ([13C6]E2) added as internal standard for identification and quantitation.
After intravenous (i.v.) dosing of ovariectomized (OVX) rats with DHED, appreciable level of circulating estrogens could not be detected and the prodrug disappeared rapidly from the circulation (Fig. 2B). The distribution half-life (td) was estimated to be 5.1±0.6 min. Concomitantly, E2 concentrations increased in the brain. Experiments using deuterated-(d3)-DHED demonstrated that d3-E2 was produced exclusively in the brain (Fig. 2C). Treatment with d3-DHED also did not trigger or inhibit endogenous E2 formation in the brain, and did not produce estrogen in the circulation and peripheral tissues, as shown by liquid chromatography–tandem mass spectrometry (LC–MS/MS) assays developed to distinguish different isotopic forms of the hormone. We also did not detect DHED in the brain, which indicated its rapid metabolism in this organ. Oral (p.o.) (fig. S2A,B) and subcutaneous (s.c.) (fig. S2C,D) administrations of the prodrug generated results similar to those obtained upon i.v. injection (Fig. 2B). These findings revealed that the increase of E2 in the brain after DHED treatment (Fig. 2B, fig. S2A C) originated from the conversion of the prodrug to E2 — and not from the stimulation of endogenous hormone synthesis.
Results of detailed pharmacokinetic (PK) studies measuring E2 concentrations in the brain and serum not only demonstrated a rapid uptake of DHED into the brain after systemic administration, but also indicated its complete absorption after oral administration (table S1). In vivo experiments using OVX repTOP ERE-Luc mice, in which luciferase expression is under the transcriptional control of ERs (20), also confirmed that prodrug treatments did not activate the ERE reporter construct and failed to generate bioluminescence in peripheral tissues accessible to in vivo imaging, even at high s.c. or p.o. doses (≥200 μg/kg) (fig. S3A). DHED was therefore not converted to E2 in the periphery. In contrast, mice treated s.c. with E2 at a dose of 20 μg/kg resulted in a significant increase of photon emission from the hepatic area (fig. S3B). Intracerebroventricular (i.c.v.) administration followed by ex vivo bioluminescence imaging of OVX repTOP ERE-Luc mice brain slices also revealed local estrogen formation from DHED (fig. S3C). The conversion occurred in all brain areas with the cortical areas showing the highest extent of DHED bioactivation in this model, confirming our in vitro metabolism studies in OVX rat brain tissues (Fig. 2A).
DHED treatment is neuroprotective and elicits estrogen-responsive neuropharmacological effects in rodent models of CNS diseases
We next evaluated the translational potential of DHED treatment in preclinical models of estrogen-responsive human CNS disorders, including stroke, depression, hot flushes and cognitive decline (21), in OVX rodents. Dose-dependent reduction of infarct volumes and attenuation of neurological deficits in animal models of stroke have been used frequently to quantify the extent of neuroprotection elicited by single-dose estrogens (16, 22). We used the most common paradigm, the transient middle cerebral artery occlusion (tMCAO) model followed by reperfusion, in our initial proof-of-concept studies. Neuroprotection was manifested by a dose-dependent reduction of the infarct volumes in DHED-treated animals (Fig. 3A), with concomitant attenuation of neurological deficits (Fig. 3B). The effective s.c. dose (ED50, equivalent to 50% of the maximum effect) was approximately 15 μg/kg; nearly 10-fold higher systemic E2 dose was needed to achieve the same protection against ischemic injury. These data indicate that the prodrug was significantly more potent to deliver E2 into the brain than direct administration of the hormone. Importantly, the ischemic brain retained its capacity to generate E2 from the prodrug (Fig. 3C) and, therefore, even post-infarct DHED treatment had a neuroprotective effect (Fig. 3D,E).
Fig. 3. DHED treatment elicits neuroprotection in a rat model of ischemic stroke.

(A) OVX Sprague-Dawley rats were treated with DHED (0.8 to 100 μg/kg, s.c.) 1 h before tMCAO followed by 24-h reperfusion. The E2-treated group received 200 μg/kg, s.c. TCC-stained brain sections are representative of three animals per indicated treatment and from the same animals within each column. Pale-colored regions indicate areas of infarct whereas red-colored regions representing viable areas. Infarct volumes are charted as averages ± SEM (n = 3 per treatment group). (B) ND scores observed in the experiment described in (A). Data are averages ±SEM (n = 3 per treatment group). (C) Measured initial DHED to E2 conversion rates in major brain areas with tissue harvested 1 h after tMCAO. Data are averages ± SEM (n = 3 per area). (D) Infarct volumes after treatment with 100 μg/kg s.c. DHED 1 h, 2 h and 3 h after tMCAO. Data are averages ± SEM (n = 3 per treatment group). (E) ND scores observed in the experiment described in (D). P values were determined by one-way ANOVA using SNK multiple comparison test.
OVX rodents are highly responsive to estrogen, and the forced swim test (FST) using this model is widely used to screen for antidepressant-like activity (23). When administered at identical doses, DHED treatments outperformed direct administration of E2 in the FST (Fig. 4A). The high-affinity ER antagonist ICI 182,780 completely blocked this antidepressant-like effect in both treatment groups (Fig. 4B), suggesting an ER-mediated mechanism. In support, we detected a significant amount of ICI 182,780 in the brain (6.6 ±0.9 ng/g tissue) by LC–MS/MS 15 min after completion of the FST. These data confirm the ability of this ER-antagonist to enter the brain after systemic administration (24).
Fig. 4. DHED treatment elicits estrogen-responsive neuropharmacological effects.
(A) Antidepressant-like activity after DHED treatments in CD1 mice using the FST. Vehicle control, as well as E2 and DHED (both at 10 μg/kg, s.c., daily for 5 days) were evaluated 1 h after the last injection. Data are averages ± SEM (n = 5–6 per treatment group). (B) The antidepressant-like effect is reversible both in E2- and DHED-treated animals (once daily, for 5 days, at 50 μg/kg, s.c.) by the ER-antagonist ICI 182,780 (4 mg/kg, s.c.) co-injected with the test compounds. (C) Representative experiments showing tail-skin temperature changes in an OVX rat hot flush model after p.o. administrations of DHED (30 μg/kg), the orally active strong synthetic estrogen EE (200 μg/kg) used as positive control, and the vehicle control. (D) Vehicle-treated controls, as well as E2- and DHED-treated (continuously for 48-day by s.c. Alzet osmotic minipumps delivering 4 μg daily) middle-aged OVX Fisher-344 rats were subjected to the delay match-to-sample plus maze test after a two-day delay. Data are average errors made +/− SEM (n = 4–5 per treatment group). P values were determined by one-way ANOVA using SNK multiple comparison test.
We demonstrated similar efficacy of DHED treatment in additional animal models of menopause-associated neurological disorders. In a rat hot flush model (25), oral DHED treatment blunted the tail-skin temperature rise (Fig. 4C) using an orally bioavailable synthetic estrogen (17α-ethinylestradiol, EE) as a positive control. In another animal model testing cognitive impairment owing to estrogen deficiency (26), we also measured the beneficial effect of DHED treatment. Continuous, long-term s.c. administration of DHED significantly decreased working memory errors committed by OVX middle-aged rats compared to control animals in a delay match-to-sample plus maze test (Fig. 4D).
DHED treatments show markers of estrogenic effects in the brain
To further validate that DHED delivery to the brain and its subsequent conversion to E2 had functional and potentially therapeutic relevance, we assayed for clinically relevant markers of estrogenic effects on the brain (26–28). DHED treatment stimulated progesterone receptor (PR) expression in the preoptic area of the hypothalamus (Fig. 5A), increased choline acetyltransferase immunoreactive (ChAT-IR) stereological cell counts in the medial septum and vertical diagonal band (Fig. 5B), and increased the number of dendritic spines and spine heads in hippocampal CA1 neurons (Fig. 5C), all compared with vehicle control treatment. These changes in the brain were indistinguishable from those seen after direct s.c. estrogen treatment at the same concentration. ICI 182,780 abolished the effect of DHED treatment on PR induction (Fig. 5A), which suggested that this response was indeed due to estrogen formed in situ from DHED.
Fig. 5. Markers of estrogenic effects in the brain are similar after E2 and DHED treatments.
(A) PR expression in the brain. OVX Sprague-Dawley rats were treated with E2 (s.c., 50 μg/kg), DHED (s.c., 50 μg/kg) or DHED (s.c., 50 μg/kg) + ICI 182,780 (s.c., 1 mg/kg, administered 1 h before DHED), as in Fig. 4C. In situ hybridization images of PR were quantified through the measurement of optical densities. Optical densities are averages ± SEM (n = 4–8 per treatment group). (B) ChAT-IR positive neurons counted in the medial septum and vertical diagonal band. OVX Fischer 344 rats were treated with E2 or DHED continuously for 48 days at 4 μg/day by s.c. osmotic pumps. Data are average stereological ChAT-IR neuron counts ± SEM (n = 4–7 per treatment group). (C) Spine density and the number of spine heads in dendrites of CA1 neurons. OVX Fischer 344 rats were treated with DHED or E2 (10 μg s.c.) every four days for 40 days. Data are averages ± SEM (n = 5–7 per treatment group). P values in (A to C) were determined by one-way ANOVA using SNK multiple comparison test.
DHED treatment does not have uterotrophic effects or promote proliferation of cancerous tissue
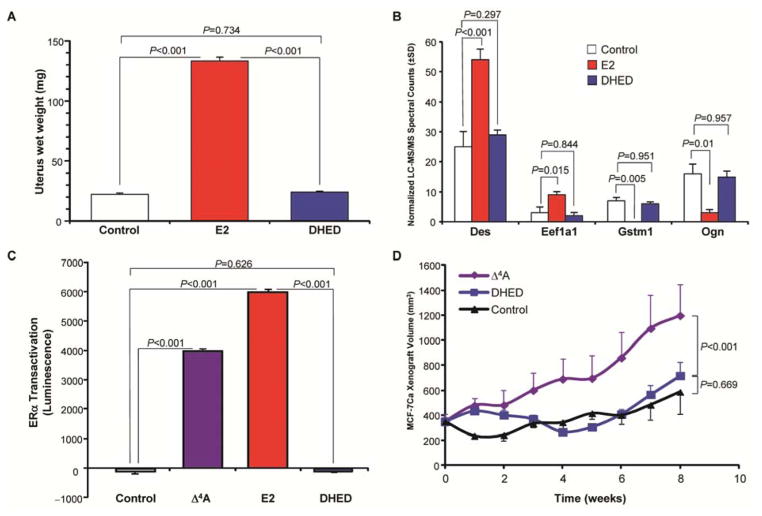
In addition to sparing the liver from estrogen exposure (fig. S3A,B), DHED treatment did not result in wet uterus weight gain (Fig. 6A), which is a uterotrophic effect of systemically administered estrogens (15, 29). This was independent of the route of administration (i.v., s.c. or p.o.) (fig. S4) and also did not occur after continuous, long-term (48-day) delivery of DHED using s.c. osmotic minipumps (fig. S4C). For these experiments, vaginal smears and standard cytology were used to confirm that all OVX animals were in diestrus before E2 and DHED treatments were initiated. During and after DHED treatment, all animals in the DHED groups remained continuously in diestrus similar to those in the OVX control group, whereas all E2-treated rats exhibited vaginal smears with many cornified cells, indicating systemic estrogen exposure. In addition, although many protein markers are up- and down-regulated by exogenous E2 (30), there were no changes in the expression of a broad panel of estrogen-regulated uterine proteins compared with control after DHED treatment (Fig. 6B, fig. S5).
Fig. 6. DHED treatment avoids uterotrophic effect and proliferation of cancerous breast tissue.
(A) OVX Swiss-Webster mice injected s.c. for 5 consecutive days with vehicle or 50 μg/kg of E2 or DHED every day. Data are averages ± SEM (n = 11 per treatment group). P values were determined by one-way ANOVA using SNK multiple comparison test. (B) Expression data for representative estrogen-sensitive uterine proteins desmin (Des), elongation factor 1-α isoform 1 (Eef1a1), glutathione S-transferase μ isoform 1 (Gstm1), and the mimecan precursor (Ogn) from the uteri of OVX Swiss-Webster mice treated with vehicle, E2, or DHED (once daily for 5 consecutive days, at 50 μg/kg, s.c. every day). Data are expressed as normalized LC–MS/MS spectral counts (average ± SD, n = 5 per treatment group). P values were determined by one-way ANOVA using SNK multiple comparison test. (C) ERα transactivation in MCF-7Ca breast cancer cells incubated with vehicle or 1 nM of the estrogen prohormone Δ4-androstenedione (Δ4A, positive control), E2, or DHED. Luminescence data are averages ± SEM (n = 4 per treatment). P values were determined by one-way ANOVA using SNK multiple comparison test. (D) Tumor volumes in nude mice with human MCF7-Ca xenografts after daily s.c. injections of vehicle, Δ4A (100 μg/kg) or DHED (100 μg/kg) for 8 weeks. Data are averages ± SEM (n = 8 per treatment group). P values were determined by two-way ANOVA with treatment as the main factor and repeated measurements in time followed by SNK multiple comparison test.
Another undesired consequence of increasing estrogen in the peripheral circulation is its effect on cancerous breast tissue. E2 and the aromatase substrate Δ4-androstenedione (Δ4A) induced the expected cancer-promoting ERα transactivation in cultured human MCF-7Ca cells (31), whereas DHED did not (Fig. 6C). Unlike Δ4A, DHED treatment also did not stimulate the growth of aromatase-transfected MCF-7Ca breast cancer cells in vivo in a nude mouse xenograft model (Fig. 6D), indicating that DHED indeed remained inert in these cells.
Discussion
Estrogen-based therapies have remained the most effective remedies to treat menopausal indications; however, they are neither appropriate nor desirable for all symptomatic women. For example, estrogens are contraindicated after preventive oophorectomy (1, 32), which is increasingly performed in gynecological oncology. Therefore, a growing population of women will not have effective and safe treatment against neurological and psychiatric maladies triggered by estrogen deficiency of the brain owing to surgical menopause (33). To address this major unmet medical need, we explored a new prodrug, 10β,17β-dihydroxyestra-1,4-dien-3-one (DHED), that remains inert in the body—only to be quickly converted to estrogen in the brain. Evidence provided here indicates that the reductive bioactivation of DHED to estrogen is brain-selective in female animal models, which could offer unique benefits to the treatment of human diseases associated with brain estrogen-deficiency or for those benefiting from estrogen-mediated neuroprotection.
Adverse consequences of estrogen deficiency in the human brain have been seen inevitably and most dramatically upon bilateral oophorectomy performed before reproductive senescence, which precipitates premature menopause. Symptoms of premature menopause and also of menopause resulting from reproductive senescence include, among others, hot flushes, depression, cognitive impairment, and an increased risk of ischemic stroke (5, 6, 34). Consequently, we chose widely accepted animal models of these human conditions to evaluate our DHED-based brain-selective intervention for a direct, side-by-side comparison with estrogen therapy. Collectively, our results indicate that DHED enters the brain of OVX rodents where it is rapidly metabolized to the main estrogen, E2. In estrogen-sensitive peripheral tissues and in the circulation, DHED is not converted to E2 and, thus, remains inert. Rapid metabolism of this prodrug to the hormone in the brain also explained why brain levels of DHED were undetectable. Overall, the significant elevation of brain E2 levels after DHED treatments originated solely from the metabolism of the inactive bioprecursor and not from DHED-induced E2 biosynthesis.
In addition to abolishing affinity to ER and to other steroid receptors, the unique structural attributes (i.e., a cyclohexadienone A-ring and the oxidatively added 10β-hydroxyl group) also favorably change key physicochemical and biopharmaceutical properties of DHED in comparison with those of E2. The benefits of these changes are shown in this study through rodent models demonstrating delivery of E2 to the brain without toxic and uterotrophic systemic side effects seen for exogenous estrogen treatments. Compared to DHED treatment, an order of magnitude higher dose of estrogen was needed to generate a similar extent of neuroprotection and neuropharmacological responses in animal models of estrogen deficiency. This finding highlights brain-selective E2 therapy by DHED as a better “drug economy”; in other words, it focuses the biologically active estrogen to the brain—unlike systemic treatment with E2 that distributes the hormone throughout the entire body.
DHED-based E2 therapy was more efficacious than direct E2 administration in a cerebrovascular stroke model. This improvement is attributed to the favorable physicochemical properties of DHED for blood-brain barrier transport from the circulation and based on mechanisms by which estrogens known to protect neurons against ischemic injury. As expected from in vitro experiments indicating DHED bioactivation by the ischemic tissue, we also observed significant neuroprotection even when the animals received DHED up to 2 h after the stroke. These additional results may expand the potential benefits of brain-selective estrogen therapy from stroke prevention in surgically menopausal women to effective post-stroke medication for all patients.
E2 has been shown to alleviate depression-like condition precipitated by a hypoestrogenic state in mice (23), and the antidepressant activity of the hormone is due to a multimodal mechanism of action involving the regulation of several pathways and functions (11). DHED treatment profoundly reduced behavioral immobility, which was reversible by an ER antagonist, the latter indicating that, similar to E2 treatment, an estrogen was responsible for the observed pharmacological response. In our preclinical evaluation, DHED administration was also effective in an established rat model of hot flushes (25), which are prevalent and long-lasting during the menopause transition (35).
A drop in E2 levels following surgical menopause also may affect key parts of the brain responsible for memory and concentration, including the hippocampus that has been shown to be associated with cognitive impairment (21, 36). In young adult and middle-aged female rats, ovarian hormone loss induces cognitive impairment that can be reversed by systemic estrogen treatment (26). In our study, middle-aged rats committed significantly fewer working memory errors in a DMS plus maze test after chronic administration of DHED than the OVX control animals. The increase in brain E2 after systemic DHED treatment appeared to have no direct effect on hippocampal estrogen biosynthesis. In subsequent experiments complementing the neuropharmacological assessments, we showed that E2 generated from DHED in the brain produced the expression of characteristic markers indicating central estrogenic effects, and DHED-treated groups were indistinguishable from the E2-treated control groups.
In current clinical practice, chronic hormone therapies require the use of a progestin to counterbalance the detrimental impact of estrogens on the endometrium. Progestins, however, have been shown to interfere with the beneficial effect of estrogens in the brain (37). Consistent with no change in circulating E2 levels after DHED treatments, OVX rodents did not display E2-induced uterotrophic effects when dosed with DHED. All systemically estrogen-treated animals in our experiments showed the expected uterotrophic effect. The lack of uterine response upon DHED treatment was confirmed using proteomic markers of E2 activity. Overall, uterine protein expression was not different between the DHED- and vehicle-treated groups. Thus, the lack of DHED’s uterotrophic activity would obviate the need for co-administration of a progestin.
The induction of aromatase-dependent breast cancer also has been considered a serious risk of current estrogen therapies often contraindicating their use to manage symptoms of menopause clinically (33). However, DHED-treated cells did not show ERα transactivation in vitro or stimulate MCF-7Ca xenografts in vivo. In further support of the inherent inertness and safety of DHED, we found that the liver was not exposed to estrogen after DHED treatment even at high p.o. doses (≥ 200 μg/kg). In contrast, s.c. administration of E2 even at 1/10th of DHED’s dose showed the expected profound ER-activation in the liver—a common hepatic side-effect of current estrogen therapies.
Collectively, our comprehensive and multidisciplinary preclinical studies have revealed that E2’s activity may be effectively confined to the brain of female rodents through the unique metabolism of a systemically administered bioprecursor prodrug. DHED has excellent oral bioavailability, which is a requirement along with additional drug-like properties, such as appropriate lipophilicity, reduced plasma-protein binding and increased solubility in water, for successful pharmaceutical development. Importantly, our study indicates the translational potential of DHED for brain-selective estrogen therapy, especially to remedy neurological and psychiatric symptoms of early and surgical menopause, and potentially also in a wide range of human diseases associated with estrogen deficiency or benefiting from estrogen-mediated protection of the brain.
Materials and Methods
Study design
This preclinical study was designed to test the ability of DHED to selectively metabolize to E2 in the brain and thereby provide brain-selective estrogen therapy. In vitro metabolism studies were performed using homogenates prepared from both healthy and diseased rat tissues. Pharmacokinetic studies (with i.v., s.c. and p.o. dosing) measuring prodrug and estrogen concentrations, respectively, in target tissues and serum were carried out for proof-of-concept. Complementary in vivo imaging was done using various routes of prodrug administration (s.c., p.o., i.c.v.). Confirmatory pharmacological assessments and assessment of markers revealing estrogenic effects were performed in animal models known to respond to exogenously administered E2. These paradigms enabled the direct comparison between the prodrug (DHED) and the parent drug (E2) to highlight the distinguishing and unique properties of DHED in terms of brain-selective estrogen therapy (endpoints). For each experiment, sample sizes were chosen to minimize the number of OVX animals needed while obtaining sufficient statistical power. Treatment duration and dosage regimen were selected based on published experiments involving estrogens after considering drug-like properties of DHED from in silico predictions and experimental measurements. Assays were carried out by blinded investigators.
Chemicals
The method used for the synthesis of DHED was based on a procedure adapted from the literature (38) using E2 (Steraloids), m-chloroperbenzoic acid (Jansen Chimica) oxidant and dibenzoyl peroxide (Aldrich) radical initiator in refluxing CH2Cl2 under nitrogen for 3 h, while being irradiated with a 60-W tungsten lamp. Column chromatographic purification (silicagel, CH2Cl2:ethyl acetate 7:3, v/v), and crystallization from toluene routinely afforded ~ 45% yield. [melting point: 215–217 °C; 1H-NMR resonances having diagnostic δ values (in ppm), using CD3OD solvent: 7.1 (d, J = 10.4 Hz, 1H, H1), 6.0 (dd, J = 10.4 Hz, 2.1 Hz, 1H, H2), 5.9 (t, J = 2.2 Hz, 1H, H4), 3.3 (t, J = 8.1 Hz, 1H, 17α-H), and 0.9 (s, 18-CH3); ESI-MS: m/z 289.3 (M+H)+]. We synthesized the deuterated analogs (d3- and d5-DHED) according to a previously reported microwave-assisted procedure (39). All other chemicals and reagents were purchased from commercial vendors.
Characterization of DHED
Steroid receptor-binding experiments were performed by Caliper Life Sciences. Inhibition of lipid peroxidation was assayed in triplicate by the ferric thiocyanate (FTC) and thiobarbituric acid reactive substances (TBARS) methods (40). Binding to human plasma proteins were measured by rapid equilibrium dialysis (with a single-use dialysis plate with inserts, Thermo) according to the manufacturer’s protocol. The logarithm of n-octanol/water partition coefficient (logP) was predicted by the method incorporated into the BioMedCAChe program (version 6.1, Fujitsu America, Inc) and was verified experimentally by the shake-flask technique. Intrinsic water solubility (Sw) was measured in deionized water at 25 °C and using an equilibrium solubility method adopted from an earlier procedure (41) by performing quantitative LC–MS/MS analyses (40).
In vitro metabolic conversions of DHED to E2
Initial in vitro conversion rates in freshly prepared 20% w/v OVX rat tissue homogenates were measured by the procedure and analytical method described previously (16). Assays were performed in triplicate after addition of d5-E2 from stock solution to reach internal standard concentration equivalent to 2 μM in the sample. Liquid-liquid extraction and quantitation by isotope dilution LC–MS/MS were done as reported before (40).
Animal treatments and monitoring
Saline and corn or sesame oil vehicles were used for i.v. and s.c. injections, respectively, while compounds were delivered by gavage in a saline or phosphate-buffered saline (PBS) vehicle upon p.o. administration. PBS also served as a vehicle for i.c.v. injection. E2, ethinyl estradiol (EE), and DHED stock solutions were prepared in dimethyl sulfoxide (DMSO) or ethanol, which were admixed to the vehicles. Concentrations in the stock solutions and mixing ratios were chosen to keep the percentage of the organic co-solvent at a minimum, while avoiding the precipitation of the compounds. For DHED, this represented ≤3% (v/v) of DMSO or ethanol in the administration-ready solutions. For continuous s.c. administration by Alzet osmotic pumps (Model 2ML4 rated for delivery at 0.25 ± 0.05 μl/h for 28 days; Durect Corporation), E2 or DHED were dissolved in propylene glycol. For control rats, pumps were filled only with propylene glycol. Pump insertion was done 18 days after ovariectomy under vaporized isoflurane anesthesia according to the surgical procedure recommended by the manufacturer. We replaced the pumps with freshly-filled new ones after 15 days of delivery; this ensured that all animals had their assigned substrate delivered for the duration of behavioral testing and through euthanasia 48 days after the surgical insertion of the first pump. During the infusion period, we examined vaginal smears according to a previously reported procedure (42). Transgenic Operative Products srl performed the experiments involving repTOP ERE-Luc mice, for which the animals were fed with a special estrogen-free diet (43).
Drug distribution in vivo
In vivo microdialysis experiments were performed as described previously (44). Measurements from serum and brain tissue were made after dissecting the brain into regions, homogenization, liquid-liquid extraction, dansylation and using 13C-labeled internal standards (13C6-E2 and13C6-E1; each at 100 pg/ml serum and 1.7 ng/g wet tissue) for quantification by isotope-dilution LC–MS/MS (45). DHED analyses were carried out in a separate LC–MS/MS assay using a Supelco Discovery HS C18 column, 50 mm × 2.1 mm, packed with 5-μm particles (Supelco). The elution was isocratic at 0.25 ml/min flow rate with a mixture of acetonitrile and water (23:77 v/v) containing 0.5% (v/v) acetic acid. DHED and the added internal reference compound (d5-DHED) in the effluent were detected by ion trap (LTQ) or triple-quadrupole (TSQ Quantum Ultra, Thermo Scientific) mass spectrometers equipped with ESI sources operated in the positive ionization mode. Quantifications were performed, similar to those of estrogens (45), by isotope-dilution MS/MS. Protein contents in tissues were measured by a dye-binding assay (Thermo Scientific). Non-compartmental pharmacokinetic (PK) analyses (46) were performed by nonlinear curve fitting using Scientist for Windows (Micromath).
Focal ischemia/transient middle cerebral artery occlusion–reperfusion injury model
Experiments were performed as previously described (47) and the infarct volumes measured after 1 h of tMCAO followed by 24-h reperfusion. Vehicle, E2, and DHED were injected s.c. 1 h before tMCAO in the stroke prevention study, while DHED injections (100 μg/kg, s.c.) also were administered immediately upon starting reperfusion (i.e., 1 h after tMCAO), as well as 1 h and 2 h thereafter (2 h and 3 h after tMCAO, respectively). The harvested brains were kept in ice-cold saline for 5 min, after which seven coronal slices (2 mm thickness) were cut from each brain for 15-min incubation in 2% 2,3,5-triphenyltetrazolium chloride (TCC) solution at 37 °C. Infarct volume was determined from measurements performed by using the ImageJ software (U.S. National Institutes of Health). Neurological deficits were assessed and scored as follows: 0, normal, i.e., no neurological deficit observed; 1, mild impairment (the animal failed to extend the contralateral forepaw on lifting the whole body by the tail); 2, moderate impairment (the animal circled contralaterally); 3, severe impairment (the animal leaned contralaterally when resting).
Computational study
All computations were performed with the SCIGRESS molecular modeling suit (version 2.6; Fujitsu–FQS Poland). Structures for ground and transition states were calculated by using the PM6 and PM6-TS semi-empirical quantum chemical parameterizations of the Molecular Orbital Package (MOPAC, Stewart Computational Chemistry) (48). The conductor-like screening model (COSMO) was employed to simulate continuum solvation in water (with the ε permittivity parameter set to 78.4) (49). Thermodynamic quantities were calculated with the THERMO program of MOPAC.
Statistical analysis
We applied Shapiro-Wilks tests, visual inspections of histograms, normal Q-Q plots and box plots to confirm normal distribution of data. Statistical evaluations were done by one-way ANOVA in all but one experiment (effect on MCF-7Ca breast cancer xenografts in athymic nude mice, where we used repeated measures two-way ANOVA). All of our follow-up, two-group comparisons employed Student-Newman-Keuls (SNK) post hoc tests when a significant omnibus ANOVA (with two-tailed α set to 0.05) was found, noting that type-I error correction was not necessary with orthogonal planned comparisons (50). P<0.05 was considered statistically significant.
Supplementary Material
Fig. S1. DHED is converted to E2 in vitro in brain, but not uterine tissue of OVX Sprague-Dawley rats.
Fig. S2. Estrogen concentrations are increased in brain but not in circulation after DHED treatments.
Fig. S3. Bioluminescence imaging in repTOP ERE-Luc mice.
Fig. S4. Lack of uterotrophic effect after DHED treatment is independent of route of administration and treatment regimen.
Fig. S5. Additional expression data for estrogen-sensitive proteins from the uteri of OVX mice treated with E2 and DHED.
Table S1. Non-compartmental single-dose PK analysis of extracted E2 concentrations in brain over time.
Acknowledgments
We thank P. Fryèák, B. Blazics, B. Ughy, S. M. Stevens Jr., M. Lane, S. White, and B. Cornelius for their expert contribution or excellent technical assistance. L.P. is grateful to M.J. Forster and M. Arad for their help with statistical analyses.
Funding: The project was supported in part by the National Institutes of Health (NS044765 [L.P.], RR012023 [L.P], AG031535 [I.M. and L.P.], AG027956 [P.K. and L.P.], AG028084 [H.B.-N.], AG031421 [K.P.-T.], and MH100700 [T.D.G. and L.P.]), the Felix and Carmen Sabates Missouri Endowed Chair in Vision Research, the Vision Research Foundation of Kansas City (P.K.) and the Robert A. Welch Foundation (endowment BK-0031; L.P.).
Footnotes
Author contributions: L.P., H.A.B-N., C.D.C., A.B., P.J.S., I.M., P.K. and K.P.-T. designed the research; L.P. performed computational chemistry and statistical analyses; K. P.-T. synthesized and characterized DHED and its labeled analogs, as well as led the in vitro metabolism studies; V.N. completed pharmacokinetics, in vivo microdialyses and the forced swim tests; S.S. ran the LC MS/MS and plasma protein-binding assays; P.G. performed the stroke study; G.S. executed the breast cancer stimulation assessments; K.J.M. collected the data on hippocampal dendritic spines and heads; J.S.T. did the maze test and measured ChAT-IR; I.M. completed the hot flush and PR-expression experiments. L.P., H.A.B-N., J.S.T., K.J.M., C.D.C., T.D.G., I.M., P.K., and K.P-T. analyzed and interpreted data. L.P. and K.P.-T. co-wrote the manuscript, with contribution by P.K. All authors have read and commented on the final manuscript.
Competing interests: L.P. and K.P.-T. have equity interests in AgyPharma LLC. L.P. and K.P.-T. hold US Patents 7,026,306 and 7,300,926 on the use of DHED.
Data and materials availability: DHED must be obtained through an MTA.
References and Notes
- 1.Rocca WA, Shuster LT, Grossardt BR, Maraganore DM, Gostout BS, Geda YE, Melton LJ., III Long-term effects of bilateral oophorectomy on brain aging: unanswered questions from the Mayo Clinic Cohort Study of Oophorectomy and Aging. Women’s Health. 2009;5:39–48. doi: 10.2217/17455057.5.1.39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Nachtigall L. Does combined hormone replacement therapy improve the health-related quality of life of postmenopausal women? Nat Clin Pract Endocrinol Metab. 2009;5:136–137. doi: 10.1038/ncpendmet1079. [DOI] [PubMed] [Google Scholar]
- 3.McEwen BC. Genome and hormones: Gender differences in physiology - Invited review: Estrogens effects on the brain: multiple sites and molecular mechanisms. J Appl Physiol. 2001;91:2785–2801. doi: 10.1152/jappl.2001.91.6.2785. [DOI] [PubMed] [Google Scholar]
- 4.Prentice RL, Anderson GL. The Women’s Health Initiative: Lessons learned. Annu Rev Public Health. 2007;29:131–150. doi: 10.1146/annurev.publhealth.29.020907.090947. [DOI] [PubMed] [Google Scholar]
- 5.Rocca WA, Grossardt BR, Miller VM, Shuster LT, Brown RD. Premature menopause or early menopause and risk of ischemic stroke. Menopause. 2012;19:272–277. doi: 10.1097/gme.0b013e31822a9937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bailey ME, Wang ACJ, Hao J, Janssen WGM, Hara Y, Dumitriu D, Hof PR, Morrison JH. Interactive effects of age and estrogen on cortical neurons: implications for cognitive aging. Neuroscience. 2011;191:148–158. doi: 10.1016/j.neuroscience.2011.05.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Files JA, Ko MG, Pruthi S. Bioidentical hormone therapy. Mayo Clin Proc. 2010;86:673–680. doi: 10.4065/mcp.2010.0714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Yao J, Zhao LQ, Mao ZS, Chen SH, Wong KC, To J, Brinton RD. Potentiation of brain mitochondrial function by S-equol and R/S-equol estrogen receptor beta-selective phytoSERM treatments. Brain Res. 2013;1514:128–141. doi: 10.1016/j.brainres.2013.02.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.de Cremoux P, This P, Leclercq G, Jacquot Y. Controversies concerning the use of phytoestrogens in menopause management: Bioavailability and metabolism. Maturitas. 2010;65:334–339. doi: 10.1016/j.maturitas.2009.12.019. [DOI] [PubMed] [Google Scholar]
- 10.Finan B, Yang B, Ottaway N, Stemmer K, Muller TD, Yi CX, Habegger K, Schriever SC, Garcia-Caceres C, Kabra DG, Hembree J, Holland J, Raver C, Seeley RJ, Hans W, Irmler M, Beckers J, de Angelis MH, Tiano JP, Mauvais-Jarvis F, Perez-Tilve D, Pfluger P, Zhang LS, Gelfanov V, DiMarchi TD, Tschoep MH. Targeted estrogen delivery reverses the metabolic syndrome. Nat Medicine. 2012;18:1847–1856. doi: 10.1038/nm.3009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Österlund MK. Underlying mechanisms mediating the antidepressant effects of estrogens. Biochim Biophys Acta Gen Subj. 2010;1800:1136–1144. doi: 10.1016/j.bbagen.2009.11.001. [DOI] [PubMed] [Google Scholar]
- 12.Ettmayer P, Amidon GL, Clement B, Testa B. Lessons learned from marketed and investigational prodrugs. J Med Chem. 2003;47:2393–2404. doi: 10.1021/jm0303812. [DOI] [PubMed] [Google Scholar]
- 13.Tapfer MK, Sebestyen L, Kurucz I, Horvath K, Szelenyi I, Bodor N. New evidence for the selective, long-lasting central effects of the brain-targeted estradiol, Estredox. Pharmacol Biochem Behav. 2004;77:423–429. doi: 10.1016/j.pbb.2003.11.005. [DOI] [PubMed] [Google Scholar]
- 14.Sarkar DK, Friedman SJ, Yen SS, Frautschy SA. Chronic inhibition of hypothalamic-pituitary-ovarian axis and body weight gain by brain-directed delivery of estradiol-17β in female rats. Neuroendocrinology. 1989;50:204–210. doi: 10.1159/000125223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Prokai-Tatrai K, Szarka S, Nguyen V, Sahyouni F, Walker C, White S, Talamantes T, Prokai L. “All in the mind”? Brain-targeting chemical delivery system of 17β-estradiol (Estredox) produces significant uterotrophic side effect. Pharm Anal Acta. 2013;S7:002. doi: 10.4172/2153-2435.S7-002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Prokai L, Prokai-Tatrai K, Perjesi P, Zharikova AD, Perez E, Liu R, Simpkins JW. Quinol-based cyclic antioxidant mechanism in estrogen neuroprotection. Proc Natl Acad Sci USA. 2003;100:11741–11746. doi: 10.1073/pnas.2032621100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bray JE, Marsden BD, Oppermann U. The human short-chain dehydrogenase/reductase (SDR) superfamily: A bioinformatics summary. Chem Biol Interact. 2009;178:99–109. doi: 10.1016/j.cbi.2008.10.058. [DOI] [PubMed] [Google Scholar]
- 18.Maser E. Neuroprotective role for carbonyl reductase? Biochem Biophys Res Commun. 2006;340:1019–1022. doi: 10.1016/j.bbrc.2005.12.113. [DOI] [PubMed] [Google Scholar]
- 19.Grogan J, Shou M, Zhou D, Chen S, Korzekwa KR. Use of aromatase (CYP 19) metabolite ratios to characterize electron transfer from NADPH-Cytochrome P450 reductase. Biochemistry. 1993;32:12007–12012. doi: 10.1021/bi00096a010. [DOI] [PubMed] [Google Scholar]
- 20.Ciana P, Raviscioni M, Mussi P, Vegeto E, Que I, Parker MG, Lowik C, Maggi A. In vivo imaging of transcriptionally active estrogen receptors. Nat Med. 2003;9:82–86. doi: 10.1038/nm809. [DOI] [PubMed] [Google Scholar]
- 21.Schuster LT, Rhodes DJ, Gostout BS, Grossardt BR, Rocca WA. Premature menopause or early menopause: Long-term health consequences. Maturitas. 2010;65:161–166. doi: 10.1016/j.maturitas.2009.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Merchenthaler I, Shughrue PJ. Neuroprotection by estrogen in animal models of ischemia and Parkinson’s disease. Drug Dev Res. 2006;66:172–181. [Google Scholar]
- 23.Bekku N, Yoshimura H. Animal model of menopausal depressive-like state in female mice: prolongation of immobility time in the forced swimming test following ovariectomy. Psychopharmacol. 2005;183:300–307. doi: 10.1007/s00213-005-0179-0. [DOI] [PubMed] [Google Scholar]
- 24.Alfinito PD, Chen X, Atherton J, Cosmi S, Deecher DC. ICI 182,780 penetrates brain and hypothalamic tissue and has functional effects in the brain after systemic dosing. Endocrinology. 2008;149:5219–5226. doi: 10.1210/en.2008-0532. [DOI] [PubMed] [Google Scholar]
- 25.Merchenthaler I, Funkhouser JM, Carver JM, Lundeen SG, Ghosh K, Winneker RC. The effect of estrogens and antiestrogens in a rat model for hot flush. Maturitas. 1998;30:307–316. doi: 10.1016/s0378-5122(98)00045-0. [DOI] [PubMed] [Google Scholar]
- 26.Bimonte-Nelson HA, Acosta JI, Talboom JS. Neuroscientists as cartographers: Mapping the crossroads of gonadal hormones, memory and age using animal models. Molecules. 2010;15:6050–6105. doi: 10.3390/molecules15096050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Shughrue PJ, Lane MV, Merchenthaler I. Regulation of progesterone receptor messenger ribonucleic acid in the rat medial preoptic nucleus by estrogenic and antiestrogenic compounds: An in situ hybridization study. Endocrinology. 1997;138:5476–5484. doi: 10.1210/endo.138.12.5595. [DOI] [PubMed] [Google Scholar]
- 28.Conrad CD, McLaughlin KJ, Huynh TN, El-Ashmawy M, Sparks M. Chronic stress and a cyclic regimen of estradiol administration separately facilitate spatial memory: Relationship with hippocampal CA1 spine density and dendritic complexity. Behav Neurosci. 2012;126:142–156. doi: 10.1037/a0025770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Owens JW, Ashby J. Critical review and evaluation of the uterotrophic bioassay for the identification of possible estrogen agonists and antagonists: In support of the validation of the OECD uterotrophic protocols for the laboratory rodent. Crit Rev Toxicol. 2002;32:445–520. doi: 10.1080/20024091064291. [DOI] [PubMed] [Google Scholar]
- 30.Prokai L, Stevens SM, Jr, Rauniyar N, Nguyen V. Rapid label-free identification of estrogen-induced differential protein expression in vivo from mouse brain and uterine tissue. J Proteome Res. 2009;8:3862–3871. doi: 10.1021/pr900083v. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Yue W, Zhou D, Chen S, Brodie A. A new nude mouse model for postmenopausal breast cancer using MCF-7 cells transfected with the human aromatase gene. Cancer Res. 1994;54:5092–5095. [PubMed] [Google Scholar]
- 32.Finch A, Metcalfe KA, Chiang JK, Elit L, McLaughlin J, Springate C, Demsky R, Murphy J, Rosen B, Narod SA. The impact of prophylactic salpingo-oophorectomy on menopausal symptoms and sexual function in women who carry a BRCA mutation. Gyn Oncol. 2011;121:163–168. doi: 10.1016/j.ygyno.2010.12.326. [DOI] [PubMed] [Google Scholar]
- 33.Rocca WA, Brandon BR, Grossardt R, Shusterd LT. Oophorectomy, menopause, estrogen treatment, and cognitive aging: Clinical evidence for a window of opportunity. Brain Res. 2011;1379:188–198. doi: 10.1016/j.brainres.2010.10.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Nelson HD. Menopause. Lancet. 2008;371:760–770. doi: 10.1016/S0140-6736(08)60346-3. [DOI] [PubMed] [Google Scholar]
- 35.Avis NE, Crawford SL, Greendale G, Bromberger JT, Everson-Rose SA, Gold EB, Hess R, Joffe H, Kravitz HM, Tepper PG, Thurston RC. Study of Women’s Health Across the Nation (SWAN), Duration of menopausal vasomotor symptoms over the menopause transition. JAMA Internal Med. 2015;175:531–539. doi: 10.1001/jamainternmed.2014.8063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Vierk R, Brandt N, Rune GM. Hippocampal estradiol synthesis and its significance for hippocampal synaptic stability in male and female animals. Neuroscience. 2014;274:24–32. doi: 10.1016/j.neuroscience.2014.05.003. [DOI] [PubMed] [Google Scholar]
- 37.Braden BB, Talboom JS, Crain ID, Simard AR, Lukas RJ, Prokai L, Scheldrup MR, Bowman BL, Bimonte-Nelson HA. Medroxyprogesterone acetate impairs memory and alters the GABAergic system in aged surgically menopausal rats. Neurobiol Learn Mem. 2011;93:444–453. doi: 10.1016/j.nlm.2010.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Solaja BA, Milic DR, Gasic MJ. A novel m-CPBA oxidation: p-quinols and epoxyquinols from phenols. Tetrahedron Lett. 1996;37:3765–3768. [Google Scholar]
- 39.Prokai-Tatrai K, Rivera-Portalatin NM, Rauniyar N, Prokai L. A facile microwave-assisted synthesis of p-quinols by lead(IV)acetate oxidation. Lett Org Chem. 2007;4:265–267. [Google Scholar]
- 40.Rivera-Portalatin NM, Serano-Vera JL, Prokai-Tatrai K, Prokai L. Comparison of estrogen-derived ortho-quinone and para-quinol concerning induction of oxidative stress. J Steroid Biochem Mol Biol. 2007;105:71–75. doi: 10.1016/j.jsbmb.2006.11.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Hurwitz AR, Liu ST. Determination of aqueous solubility and pKa values of estrogens. J Pharm Sci. 1977;66:624–627. doi: 10.1002/jps.2600660504. [DOI] [PubMed] [Google Scholar]
- 42.Goldman JM, Murr AS, Cooper RL. The rodent estrous cycle: Characterization of vaginal cytology and its utility in toxicological studies. Birth Defects Res B Dev Reprod Toxicol. 2007;80:84–97. doi: 10.1002/bdrb.20106. [DOI] [PubMed] [Google Scholar]
- 43.Stell A, Belcredito S, Ciana P, Maggi A. Molecular imaging provides novel insights on estrogen receptor activity in mouse brain. Mol Imaging. 2008;7:283–292. [PMC free article] [PubMed] [Google Scholar]
- 44.Prokai L, Prokai-Tatrai K, Zharikova AD, Nguyen V, Perjesi P, Stevens SM., Jr Centrally-acting and metabolically stable thyrotropin-releasing hormone analogues upon replacement of histidine with substituted pyridinium. J Med Chem. 2004;47:6025–6033. doi: 10.1021/jm020531t. [DOI] [PubMed] [Google Scholar]
- 45.Szarka S, Nguyen V, Prokai L, Prokai-Tatrai K. Separation of dansylated 17α-estradiol, 17β-estradiol, and estrone on a single HPLC column for simultaneous quantitation by LC-MS/MS. Anal Bioanal Chem. 2013;405:3399–3406. doi: 10.1007/s00216-013-6710-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Gibaldi M, Perrier D. Pharmacokinetics. 2. Marcel Dekker; New York: 1982. [Google Scholar]
- 47.Garg P, Duncan RS, Kaja S, Zabaneh A, Chapman KD, Koulen P. Lauroylethanolamide and linoleoylethanolamide improve functional outcome in a rodent model for stroke. Neurosci Lett. 2011;492:134–138. doi: 10.1016/j.neulet.2011.01.073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Stewart JJP. Optimization of parameters for semiempirical methods V: Modification of NDDO approximations and application to 70 elements. J Mol Model. 2007;13:1173–1213. doi: 10.1007/s00894-007-0233-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Klamt A, Schüürmann G. COSMO: A new approach to dielectric screening in solvents with explicit expressions for the screening energy and its gradient. J Chem Soc Perkin Trans. 1993;2:799–805. [Google Scholar]
- 50.Keppel G, Wickens TD. Design and Analysis: A Researcher’s Handbook. 4. Pearson Prentice Hall; Upper Saddle River, NJ: 2004. [Google Scholar]
- 51.Acosta JI, Mayer L, Talboom JS, Zay C, Scheldrup M, Castillo J, Demers LM, Enders CK, Bimonte-Nelson HA. Premarin improves memory, prevents scopolamine-induced amnesia and increases number of basal forebrain choline acetyltransferase positive cells in middle-aged surgically menopausal rats. Horm Behav. 2009;55:454–464. doi: 10.1016/j.yhbeh.2008.11.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Fig. S1. DHED is converted to E2 in vitro in brain, but not uterine tissue of OVX Sprague-Dawley rats.
Fig. S2. Estrogen concentrations are increased in brain but not in circulation after DHED treatments.
Fig. S3. Bioluminescence imaging in repTOP ERE-Luc mice.
Fig. S4. Lack of uterotrophic effect after DHED treatment is independent of route of administration and treatment regimen.
Fig. S5. Additional expression data for estrogen-sensitive proteins from the uteri of OVX mice treated with E2 and DHED.
Table S1. Non-compartmental single-dose PK analysis of extracted E2 concentrations in brain over time.