Abstract
Three out of five outbreaks of foot-and-mouth disease (FMD) since 2010 in the Republic of Korea have occurred in the winter. At the freezing temperatures, it was impossible to spray disinfectant on the surfaces of vehicles, roads, and farm premises because the disinfectant would be frozen shortly after discharge and the surfaces of the roads or machines would become slippery in cold weather. In this study, we added chemical deicers (ethylene glycol, propylene glycol, sodium chloride, calcium chloride, ethyl alcohol, and commercial windshield washer fluid) to keep disinfectants (0.2% citric acid and 4% sodium carbonate) from freezing, and we tested their virucidal efficacies under simulated cold temperatures in a tube. The 0.2% citric acid could reduce the virus titer 4 logs at −20°C with all the deicers. On the other hand, 4% sodium carbonate showed little virucidal activity at −20°C within 30 min, although it resisted being frozen with the function of the deicers. In conclusion, for the winter season, we may recommend the use of citric acid (>0.2%) diluted in 30% ethyl alcohol or 25% sodium chloride solvent, depending on its purpose.
INTRODUCTION
Foot-and-mouth disease (FMD) is an acute viral disease that affects animal with cloven hooves, such as cattle, pigs, sheep, goats, and deer. Its symptoms include increased body temperature, lameness, and blisters on the mouth, tongue, or teats or between the hooves; it affects growth, lactation, motion, and breeding (1–3). Foot-and-mouth disease virus (FMDV) belongs to the Aphthovirus genus of Picornaviridae family, and its classification has seven serotypes (O, A, Asia 1, C, SAT 1, SAT 2, and SAT 3) and about 80 subtypes (4, 5).
Since 2010, there were five FMD outbreaks (three of them lasting through the winter season) in the Republic of Korea (ROK). During those periods, liquid disinfectants could not be used sufficiently outside due to the fear of their freezing. In the ROK, January is the coldest month: its mean temperature ranges from −6°C to 7°C, and the daily low temperature often goes down to around −20°C in some northern or mountainous areas. The failure of the disinfection of the vehicles, premises, and roads in the contaminated region due to the cold weather was raised as one of the factors for not stopping the transmission in the early stage of the outbreak of 2010-2011 (6). Concerning the limitation of the conventional disinfectant, the addition of deicers in order to lower the freezing points of its use in the winter season needs to be considered. In the case of avian influenza virus, ethylene glycol or methyl alcohol is useful to decrease the freezing points and maintain the efficacy of the phenol-based disinfectant and quaternary ammonium product (7). However, the reaction test appeared to be performed at or above 4°C, which is not the real temperature outside in winter, although the concentrations of the antifreezes established for the antifreeze-disinfectant mixture were found to resist being frozen at −15°C (7).
For FMD, acid or alkali disinfectants are efficacious in inactivating the viruses, and the presence of organic matter or detergent seldom influences those effects as long as the pH value is maintained acidic or basic enough (8). Citric acid is one of the most widely used acid for FMD, and it turned out to be major component of disinfectants supplied in the ROK (9). Sodium carbonate, known as washing soda, is also a well-known alkali to inactivate FMDV. In this study, we employed six deicing chemicals (inorganic salts for the road or antifreeze for the other usages) to decrease the freezing point of 0.2% citric acid and 4% sodium carbonate and determined the virucidal effects of their mixtures for FMDV.
MATERIALS AND METHODS
Viruses and cells.
Foot-and-mouth disease virus of serotypes O and A, which were collected from the ROK in 2010, were propagated in a bovine kidney cell line (LFBK) which was grown in Dulbecco modified Eagle medium (DMEM) with high glucose (Cellgro, USA) containing 10% fetal bovine serum (FBS; Cellgro). (This cell line was kindly provided by Luis Rodriguez from ARS, USDA.)
Disinfectants and deicing chemicals.
The following chemicals and products were used as disinfectants or deicers: citric acid (C6H8O7; Sigma-Aldrich), sodium carbonate (Na2CO3; Sigma-Aldrich), ethylene glycol (HOCH2CH2OH; Sigma-Aldrich), propylene glycol [CH3CH(OH)CH2OH; Sigma-Aldrich], sodium chloride (NaCl; Sigma-Aldrich), calcium chloride (CaCl2; Sigma-Aldrich), ethyl alcohol (CH3CH2OH; Sigma-Aldrich), and windshield washer fluid (SK Networks, ROK) containing 38 to ∼39% (vol/vol) methyl alcohol (CH3OH). The disinfectants were diluted in sterilized WHO hard water containing calcium chloride (0.305 g; Sigma-Aldrich) and magnesium chloride (MgCl2·6H2O; 0.139 g; Sigma-Aldrich) in 1 liter of deionized water. For soiling the mixture of the disinfectant and deicer, 5% FBS (Cellgro) was used as a solvent.
Physiochemical and efficacy test of the mixture of deicer and disinfectant.
At first, each kind of chemical deicers was diluted in hard water and subject to a freezing temperature (−20°C) for 24 h to find a range of its concentrations at which those solutions are not frozen (data not shown). Thereafter, 0.2% citric acid and 4% sodium carbonate were diluted in those solution and then examined for their changes in pH values, physical state, and sedimentation after being kept at 37°C, −20°C, and −40°C for 24 h (Table 1). From these results, the optimal concentration of each of the deicers was determined, and, at these levels, the mixtures of deicer and disinfectant were tested for their virucidal effects on FMD virus, of serotype O, at various temperatures (37°C, 4°C, and −20°C) for various periods (5 min and 30 min).
TABLE 1.
Physiochemical state and pH values of deicer-disinfectant mixtures kept for 24 h at various temperaturesa
| Disinfectant | Temp (°C) | Parameter | Result with: |
||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Windshield washer fluid (vol/vol) |
Ethylene glycol (vol/vol) |
Propylene glycol (vol/vol) |
Sodium chloride (wt/vol) |
Calcium chloride (wt/vol) |
Ethyl alcohol (vol/vol) |
||||||||||
| 70% | 80% | 90% | 30% | 40% | 50% | 30% | 40% | 20% | 25% | 30% | 25% | 30% | |||
| 0.2% citric acid | 37 | State | L | L | L | L | L | L | L | L | L | L | L | L | L |
| Sediment | − | − | − | − | − | − | − | − | − | − | − | − | − | ||
| pH | 2.87 | 2.92 | 2.99 | 2.62 | 2.64 | 2.65 | 2.67 | 2.72 | 1.84 | 1.73 | 0.90 | 2.72 | 2.80 | ||
| −20 | State | L | L | L | L | L | L | L | L | L | L | L | L | L | |
| Sediment | − | − | − | − | − | − | − | − | − | − | − | − | − | ||
| pH | 3.10 | 3.15 | 3.23 | 2.84 | 2.84 | 2.85 | 2.83 | 2.86 | 2.03 | 1.86 | 0.99 | 2.96 | 3.12 | ||
| −40 | State | F | L | L | L | L | L | F | L | F | L | L | F | L | |
| Sediment | − | − | − | − | − | − | − | − | − | − | − | − | − | ||
| pH | 3.31 | 3.34 | 3.40 | 3.05 | 3.02 | 2.99 | 2.96 | 2.98 | 2.23 | 2.16 | 1.15 | 3.33 | 3.36 | ||
| 4% sodium carbonate | 37 | State | L | L | L | L | L | L | L | L | L | L | L | L | L |
| Sediment | − | − | − | − | − | − | − | − | − | − | +++ | − | − | ||
| pH | 11.80 | 11.88 | 11.94 | 11.46 | 11.51 | 11.56 | 11.63 | 11.74 | 10.62 | 10.53 | 5.06 | 11.78 | 11.94 | ||
| −20 | State | F | L | L | F | L | L | F | L | F | L | L | F | L | |
| Sediment | − | − | + | − | − | − | − | − | − | − | +++ | + | ++ | ||
| pH | 11.71 | 11.78 | 11.63 | 11.39 | 11.46 | 11.51 | 11.58 | 11.71 | 10.37 | 10.26 | 5.03 | 11.59 | 11.84 | ||
| −40 | State | F | F | F | F | L | L | F | F | F | F | L | F | F | |
| Sediment | − | − | + | − | − | − | − | − | + | + | +++ | + | ++ | ||
| pH | 11.41 | 11.63 | 11.71 | 11.48 | 11.34 | 11.54 | 11.45 | 11.66 | 9.90 | 9.70 | 5.01 | 11.67 | 11.75 | ||
L, liquid; F, frozen. The number of plus signs corresponds to the degree of sedimentation. −, no sedimentation.
Ten microliters of the refrigerated virus supernatant of serotype O or A with titers of >107 50% tissue culture infective doses (TCID50)/ml was carefully added to 10 μl of deicer-disinfectant solution in the microtube on the thermorack (Eppendorf, Germany), which had been equilibrated to the designated temperatures (37°C, 4°C, and −20°C) for 24 h inside the incubator or freezer. The mixture was quickly and carefully mixed by pipetting up and down so that the virus would not to be touched to the walls of the tube by itself. Thereafter, the mixture was placed back into the incubator or freezer for the desired period, and then 980 μl of neutralizing medium (5% FBS) was added to inactivate the virucidal effect of the disinfectant (1:50). The cytotoxicity of the disinfectants and deicers on the cells was found to be neutralized in this process. The infectivity of the virus before and after the treatment was measured by titration in the LFBK cells on the 96-well microtiter plate, where the virus-induced cytopathic effect (CPE) was checked for 48 h and a TCID50 was determined based on the method of Reed and Muench (10). For the selected deicers (sodium chloride and ethyl alcohol), we did the disinfection test against the two different viruses of serotypes O and A under the condition of soiling of the deicer-disinfectant mixture with 5% FBS.
RESULTS
Effects of deicers on the physiochemical property of the disinfectants.
A range of concentrations of each deicer at which its mixture with disinfectant resists being frozen at −20°C was determined (Table 1). For 0.2% citric acid, the pH values range from 0.9 to 3.4, depending on the various temperatures and the combination with deicer; calcium chloride most greatly influenced the pH of the solution by lowering it to 0.9 to ∼0.99, and sodium chloride also decreased the pH of the solution, to around 1.73 to ∼2.23 (Table 1). At −40°C, in the cases of lower concentrations of deicer, 0.2% citric acid was frozen slightly. For 4% sodium carbonate, the pH values range from 9.70 to 11.94 depending on the various temperatures and the combination with deicer, except for calcium chloride, which precipitates with sodium carbonate and decreases the pH of the solution to 5.01 to ∼5.06 (Table 1). This sodium carbonate-deicer mixture appeared to be frozen more easily at −40°C than did citric acid, although the frozen state was not so solid. In higher concentrations of windshield washer fluid and ethyl alcohol at lower temperatures, 4% sodium carbonate produced some sediments (Table 1). From these results, we chose the optimal concentration of the deicers to test virucidal efficacy at various temperatures: for ethylene glycol, 40% (vol/vol); for propylene glycol, 40% (vol/vol); for sodium chloride, 25% (wt/vol); for calcium chloride, 30% (wt/vol); for ethyl alcohol, 30% (vol/vol); and for windshield washer fluid, 80% (vol/vol).
Efficacies of the disinfectant-deicer mixtures for inactivating virus.
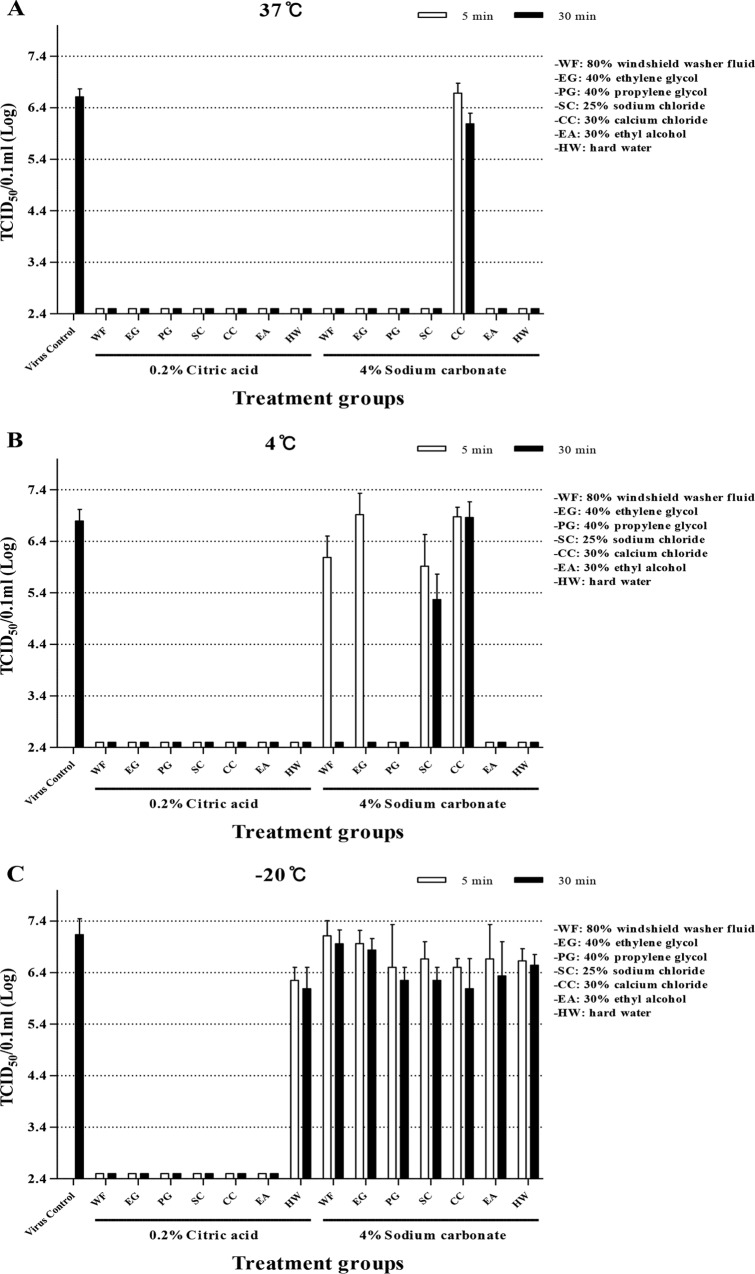
The mixtures of 0.2% citric acid and all the deicers could reduce the virus titer at least 4 log10 in 5 min at 37°C, 4°C, and −20°C (Fig. 1). But 4% sodium carbonate did not appear to neutralize any infectivity of the viruses in 30 min at −20°C, even with deicers. At 4°C, 4% sodium carbonate mixed with windshield washer fluid, ethylene glycol, sodium chloride, and calcium chloride was less effective in neutralizing the virus than the 4% sodium carbonate. The sodium carbonate-calcium chloride mixture, of which the pH is close to 6.0, appeared to lose its efficacy almost completely irrespective of the temperature conditions (Fig. 1).
FIG 1.
Inactivation of FMDV of serotype O by disinfectant-deicer mixtures at various temperatures. The virus was titrated after treatment for 5 min and 30 min at 37°C (A), 4°C (B), and −20°C (C). Each reaction was tested more than twice, and all the values were averaged; the lowest measurable titer was 2.5 log10 TCID50/0.1 ml.
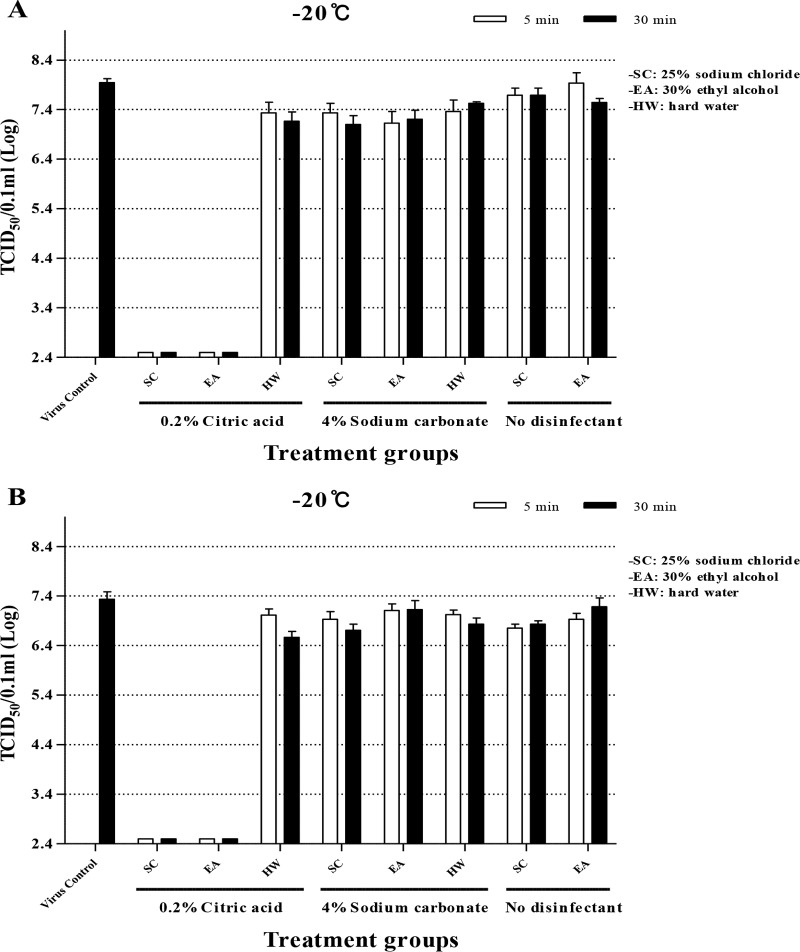
Further disinfection tests were done with two different viruses of serotypes O and A by employing soiling, exclusively for sodium chloride and ethyl alcohol because of their usefulness as additives to the disinfectants (Fig. 2). There appeared to be no difference in the pattern of the reduction of virus titer after treatment between two different viruses, and the soiling with organic matter did not seem to influence the results. The two substances, sodium chloride and ethyl alcohol, themselves did not inactivate the viruses (Fig. 2).
FIG 2.
Inactivation of FMDV of serotypes O and A by disinfectant-deicer mixtures soiled with 5% FBS at −20°C. The virus was titrated after treatment for 5 min and 30 min at −20°C for serotype O (A) and serotype A (B). Each reaction was tested in duplicate more than twice, and all the values were averaged; the lowest measurable titer was 2.5 log10 TCID50/0.1 ml .
DISCUSSION
The efficacy of 0.2% citric acid in inactivating FMDV appeared to be persistent with the addition of the deicers even at various temperatures: 37°C, 4°C, and −20°C. In all cases, at least a 4-log reduction in virus titer was found in 5 min. Because the volume of the reaction was in a microscale and it had to be diluted 1:50, the discernible range to quantify the reduction of the virus titer was limited in comparing the effects of the various temperatures more thoroughly in this study (Fig. 1 and 2). However, to neutralize the cytotoxicity of the deicers and to handle the disinfection reaction under an isothermal condition at −20°C more effectively, the reaction volume needed to be small.
Soiling with 5% FBS did not appear to affect the virucidal effect of the 0.2% citric acid-deicer mixtures. The robustness of the efficacy of acid for disinfecting FMDV had been already proven (1, 8); as long as the addition of ox serum, feces, or soil suspension addition did not affect the pH, the reduction of the virus titer in the acid or alkali was almost same at 4°C (8).
On the other hand, 4% sodium carbonate was not so effective at inactivating the virus at freezing temperatures (Fig. 1 and 2). Although deicer-sodium carbonate mixtures resist being frozen at −20°C, this does not improve the virucidal effect of the disinfectant at that temperature. The two deicers (sodium chloride and calcium chloride) decreased the disinfection efficacy of the sodium carbonate by chemical reactions among them which decrease the pH of the disinfectant (Fig. 1). Reportedly, concerning the rate of virus inactivation using sodium carbonate (4%, pH 11.0), it took 3 min and 30 min to achieve a 5-log reduction in virus titer at 20°C and 4°C, respectively (8). Similarly, in this study, it only took at the most 5 min to get a 4-log reduction in the titer for type O virus with 4% sodium carbonate at 4°C (Fig. 1).
In summary, we may recommend the use of sodium chloride and ethyl alcohol as antifreezing substances to be added to citric acid for disinfecting FMDV. Because sodium chloride is used as a deicer widely in the winter season, this may be more appropriate to disinfect the roads and wheels of vehicles in the affected area. Because ethyl alcohol is used widely in the laboratory and citric acid is less harmful to human and animals than other acids, their mixture may be applied to clothes, vehicles, and buildings in the affected premises. To increase the effect of citric acid, the use of detergent and/or an increase in the concentration of citric acid might be considered. However, the chloride component corrodes metal, which can cause problems when applied to automobiles and other metal materials. In addition, the evaporation of the alcohol also needs to be considered for their usage. So some precautions need to be taken for their use in the field with citric acid. Concerning other deicers which are effective for citric acid, propylene glycol is more environmentally friendly than ethylene glycol in terms of toxicity to humans, but both of these do not seem to be cost-effective compared to sodium chloride and ethyl alcohol; although windshield washer fluid can be cost-effective and user-friendly, its potential harmfulness to humans is substantial.
In conclusion, considering the limitations of the deicers or antifreezing substances tested for disinfecting FMDV in this study or avian influenza virus (7), we may need to test more candidate chemicals or materials to enable a disinfectant to be more efficacious in the winter season.
ACKNOWLEDGMENTS
This study was supported by Animal and Plant Quarantine Agency (QIA).
We thank the reviewers and Jeesoo Kim for their critical reading and valuable comments.
REFERENCES
- 1.Bachrach HL. 1968. Foot-and-mouth disease. Annu Rev Microbiol 22:201–244. doi: 10.1146/annurev.mi.22.100168.001221. [DOI] [PubMed] [Google Scholar]
- 2.Burrows R, Mann J, Garland A, Greig A, Goodridge D. 1981. The pathogenesis of natural and simulated natural foot-and-mouth disease infection in cattle. J Comp Pathol 91:599–609. doi: 10.1016/0021-9975(81)90089-X. [DOI] [PubMed] [Google Scholar]
- 3.Thomson G, Vosloo W, Bastos AD. 2003. Foot and mouth disease in wildlife. Virus Res 91:145–161. [DOI] [PubMed] [Google Scholar]
- 4.Pereira H. 1981. Foot-and-mouth disease, p 333–363. In Gibbs EPG. (ed), Virus diseases of food animals, vol 2 Academic Press, New York, NY. [Google Scholar]
- 5.Sellers RF. 1969. The nature and the control of foot-and-mouth disease. Int J Dairy Technol 22:90–93. doi: 10.1111/j.1471-0307.1969.tb01049.x. [DOI] [Google Scholar]
- 6.Yoon H, Yoon SS, Kim YJ, Moon OK, Wee SH, Joo YS, Kim B. 2015. Epidemiology of the foot-and-mouth disease serotype O epidemic of November 2010 to April 2011 in the Republic of Korea. Transbound Emerg Dis 62:252–263. doi: 10.1111/tbed.12109. [DOI] [PubMed] [Google Scholar]
- 7.Davison S, Benson CE, Ziegler AF, Eckroade RJ. 1999. Evaluation of disinfectants with the addition of antifreezing compounds against nonpathogenic H7N2 avian influenza virus. Avian Dis 43:533–537. doi: 10.2307/1592653. [DOI] [PubMed] [Google Scholar]
- 8.Sellers RF. 1968. The inactivation of foot-and mouth disease virus by chemicals and disinfectants. Vet Rec 83:504–506. doi: 10.1136/vr.83.20.504. [DOI] [PubMed] [Google Scholar]
- 9.Kim H-M, Shim I-S, Baek Y-W, Han H-J, Kim P-J, Choi K. 2013. Investigation of disinfectants for foot-and-mouth disease in the Republic of Korea. J Infect Public Health 6:331–338. doi: 10.1016/j.jiph.2013.04.002. [DOI] [PubMed] [Google Scholar]
- 10.Reed LJ, Muench H. 1938. A simple method of estimating fifty percent endpoints. Am J Hyg 27:493–497. [Google Scholar]