Abstract
The mammalian urethra is a muscular tube responsible for ensuring that urine remains in the urinary bladder until urination. In order to prevent involuntary urine leakage, the urethral musculature must be capable of constricting the urethral lumen to an extent that exceeds bladder intravesicular pressure during the urine-filling phase. The main challenge in anti-incontinence treatments involves selectively-controlling the excitability of the smooth muscles in the lower urinary tract. Almost all strategies to battle urinary incontinence involve targeting the bladder and as a result, this tissue has been the focus for the majority of research and development efforts. There is now increasing recognition of the value of targeting the urethral musculature in the treatment and management of urinary incontinence. Newly-identified and characterized ion channels and pathways in the smooth muscle of the urethra provides a range of potential therapeutic targets for the treatment of urinary incontinence. This review provides a summary of the current state of knowledge of the ion channels discovered in urethral smooth muscle cells that regulate their excitability.
Keywords: electrophysiology, ion channels, myogenic tone, patch clamp, smooth muscle, urethra, urethral innervation
Role of the Urethra in the Lower Urinary Tract
The filtration of blood by glomeruli in the kidney leads to the formation of urine, which is then transported to the urinary bladder by specialized muscular tubes called ureters that undergo finely-tuned peristaltic waves to prevent urine backflow toward the kidneys.1,2 The bladder is a hollow muscular organ capable of receiving and storing urine as it is propelled by the ureters into its interior. The compliant bladder expands as urine fills its interior and raises intravesicular pressure on the bladder walls. The impulse/desire to urinate is thought to result from the high firing rate of afferent sensory nerves stimulated by mechanoreceptors in the bladder wall, which are activated by the rise in intravesicular pressure in the bladder.3,4 These afferent sensory nerves project top the dorsal horn of the spinal cord via the pelvic nerve and connecting fibers then travel to higher brain regions (i.e., pontine micturition center and cerebrum5). The urethra is located distal to the bladder neck and connects the bladder interior to the exterior environment (Fig. 1). It is a structurally-complex multi-layered tissue comprising the lamina propria, including both mucosa and submucosa, as well as longitudinal and circular layers of smooth muscle.6 The urethra also contains striated muscle proximal to the pelvic floor, often referred to as the external urethral sphincter.7 Contraction of this muscle is commonly associated with the “guarding reflex” experienced during times of high bladder intravesicular pressure (e.g., sneezing, coughing).8-11 The adult female urethra is embedded in the anterior vaginal wall and typically ranges 3-4 cm in length and ∼0.6 cm in luminal diameter.12,13 Although the male urethra is ∼20 cm in length, it is mainly the prostatic and pre-prostatic regions (Fig. 1) that contribute to the true internal urethral sphincter.6,14
Figure 1.
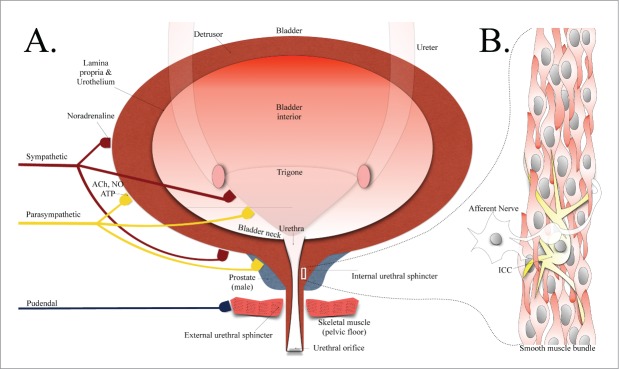
(A) A representation of the anatomy of the lower urinary tract including bladder and urethral structures, with inputs from various nervous systems illustrated. (B) A smooth muscle bundle depicting smooth muscle cells in close contact with an ICC-like cell and nerve. Abbreviations: ACh, acetylcholine; NO, nitric oxide; ATP, adenosine 5′-triphosphate; ICC, interstitial cell of Cajal.
In healthy individuals, the process of urination is a coordinated voiding mechanism involving the contraction of the detrusor muscle lining the bladder, combined with the relaxation of the urethral smooth muscle, also known as the internal urethral sphincter (Fig. 1). During the urine filling/storage phase, urine outflow from the bladder does not occur by virtue of the fact that the bladder musculature is largely relaxed and electrically quiescent, while the urethra, a conduit muscular tube extending from the base of the bladder, maintains a constant tone and is effectively closed (excellent reviews are available describing bladder and urethral physiology/pharmacology).6,13 Early studies utilizing catheters in humans reported a time-delay ranging from 5-15 seconds between the relaxation of urethral musculature and contraction of the bladder detrusor muscle.15-18
It is well-established that the inability of the urethral musculature to maintain sufficient tone can result in involuntary urine leakage.6,19-21 Damage to urethral smooth musculature may be in the form of acute trauma (e.g., surgical intervention, childbirth complications)19,20,22,23 or from aging-related diseases.13,22,23 Recently there has been an increasing recognition of the value of targeting the urethral musculature in the clinical management of urinary incontinence. The array of ion channels located in urethral smooth muscle membranes play a crucial role in determining internal urethral sphincter excitability and therefore the overall function of the urethra.6,24
Studying electrical activity in the urethra
The mammalian urethra is known to exhibit spontaneous mechanical activity, but during the urine-storage phase, muscle activity is mostly tonic in nature.6 The complex nature of urethral tissue has presented many challenges in studying its function in detail. Most current knowledge regarding the role of ion channels in the urethra has come from isometric tension recordings,25-31 sharp microelectrode recordings of intact preparations, 25,27,32,33 and the patch clamp technique.29-31,33-40
Earlier studies measuring electrical and mechanical (i.e., force) activity in rabbit urethral smooth muscle reported that the spontaneous activity was in fact myogenic in nature,25,41 resembling the activity seen in the GI tract.42 Thus, the excitability appeared to be driven by a form of pacemaker originating within the muscle itself. It is important to apply caution when interpreting data from intact preparations, such as from tension and microelectrode recordings, since (i) the urethral smooth musculature contains a mixed population of cells that includes a range of non-contractile interstitial cells of Cajal (ICC)-like cells (Fig. 1B, see focused reviews),43,44 and (ii) the urethra receives innervation from sympathetic and parasympathetic nervous systems.25,28,29,31 It is extremely difficult, therefore, to reliably isolate the role of ion channels in a given cell type using such intact preparations, particularly with limited pharmacology. The patch clamp technique,45 on the other hand, remains the only reliable method currently available to study the behavioral properties of ion channels expressed on the surface membranes of the ICCs and smooth muscle cells of the urethra. Recent advances in isolating toxins and selective pharmacological agents have greatly aided in identifying and profiling the roles of ion channels, as discussed below.
K+ Channels in Urethral Smooth Muscle Cells
The Brading research group successfully applied the patch clamp technique to urethral smooth muscle cells isolated from pigs in the late 1990s, reporting a number of different K+ channel currents34,35,46 that exhibited small and large conductance properties. One conductance in particular showed characteristics consistent with ATP-sensitive K+ (KATP) channels, and was the focus of the early studies principally using the pig model.
KATP channels
KATP channels are formed by the combination of 4 inwardly-rectifying K+ (Kir) channel subunits, comprising the pore-forming core, surrounded by a ring of 4 regulatory sulfonylurea subunits (Fig. 2 and review).47 The KATP channels identified in the urethral smooth muscle cells have a single channel conductance of 43 pS and their activation by pharmacologic agents was reported to hyperpolarize the membrane potential. Thus, K+ efflux from these channels drives the membrane potential in the negative direction, decreasing excitability. Since then, Kir6.1 and Kir6.2 subunit transcripts have been detected and it has been suggested that the KATP channel comprises a “mixed” (i.e. heterotetrameric) channel of Kir6.1 and Kir6.2 subunits, since the observed conductance value of 43 pS was intermediate between those measured for homomeric channels consisting of either Kir6.1 or 6.2 subunits.47-50 It has recently been definitively reported that the pore-forming region of the urethral smooth muscle KATP channel is in fact a heterotetrameric complex of Kir6.1 and Kir6.2 subunits, arranged in a 3:1 ratio (Fig. 2).38 In addition, transcripts for the regulatory sulfonylurea receptors SUR1 and SUR2B have also been detected, and are known to functionally co-assemble.50,51
Figure 2.
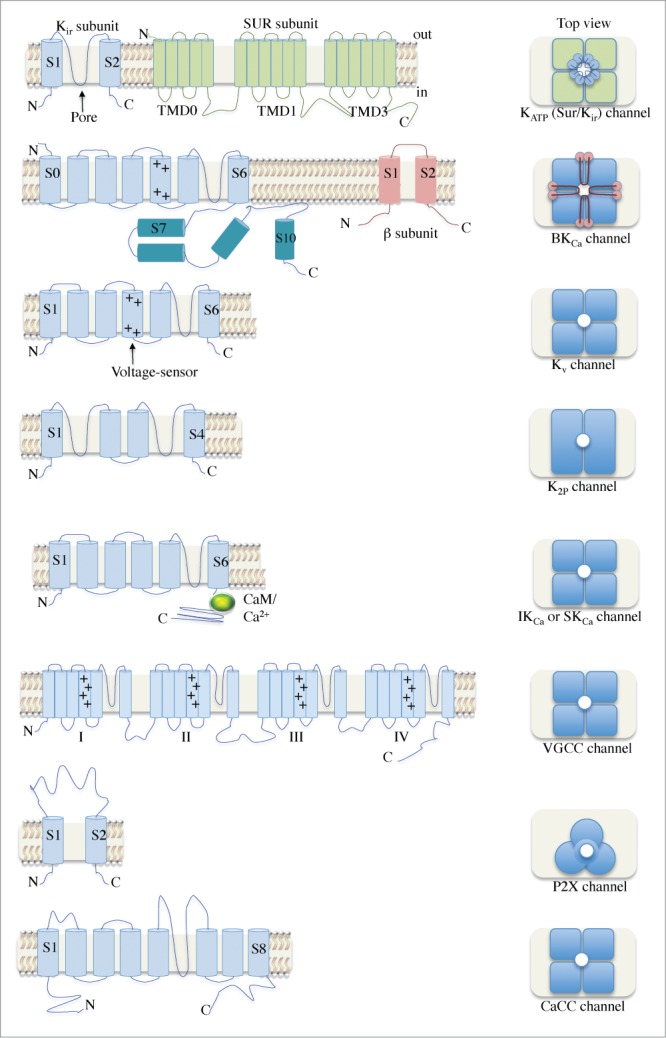
A schematic illustration of the various ion channels identified in urethral smooth muscle. The architecture of the transmembrane α and β subunits is shown in the left panel. Dimeric and trimeric arrangements can be seen for K2P and P2X channels, respectively. Most channels have tetrameric structures and several have β subunits associated. Abbreviations: N, amino-terminus; C, carboxyl-terminus; Kir, inwardly-rectifying K+ channel; SUR, sulfonylurea; TMD, transmembrane domain; KATP, ATP-sensitive K+ channel; +, positively-charged residues; BKCa, large conductance, Ca2+-activated K+ channel; Kv, voltage-gated K+ channel; CaM, calmodulin; K2P, 2-pore domain K+ channel; IKCa, intermediate conductance K+ channel; SKCa, small conductance K+ channel; VGCC, voltage-gated Ca2+ channel; CaCC, Ca2+-activated Cl− channel.
Thorough studies by Teramoto and colleagues outlining the biophysical properties of KATP channels in the pig urethra has provided valuable experimental evidence that KATP channels contribute to setting the resting membrane potential (∼ −37 mV).38,46 Important physiological implications result from the particular heterotetrameric arrangement of the channel complex identified in the urethra that distinguish it from other smooth muscle types such as those found in vascular tissues.52,53 The gating properties (i.e., activation) of the heterotetrameric KATP channel complex found in the urethra can be dynamically modulated by protein kinase C (PKC),38 which is distinct from the mechanism observed in vascular smooth muscle for KATP channels.53
Voltage and Ca2+-activated K+ channels
Following elucidation of the role of KATP channels in urethral smooth muscle cells, the next series of breakthroughs in characterizing urethral smooth muscle cell ion channel properties originated from a collaboration among the Hollywood, McHale and Thornbury laboratories investigating K+ channels and voltage-gated Ca2+ channels (VGCCs, Fig. 2). Urethral smooth muscle cells were reported to have large conductance, Ca2+-activated K+ (BKCa) channels and voltage-gated K+ (Kv) channels carrying outwardly-rectifying K+ current (Fig. 3).40 However the lack of Kv-specific inhibitors/enhancers at that time hampered further identification and profiling of the Kv conductances. The subsequent isolation and development of toxins, such as the tarantula spider venom peptide, stromatoxin 1 (ScTx),54,55 has provided a valuable selective pharmacological tool to study certain Kv channels. ScTx targets the voltage sensor domain in homotetrameric Kv channels Kv2.1, Kv2.2 and Kv4.2.30,54 In addition, ScTx has been shown to inhibit heterotetrameric Kv2.1/6.356 and Kv2.1/9.3-containing Kv channels.30,54,57,58 Using the ScTx peptide in combination with biophysical analysis, Hollywood and colleagues reported that the majority of outwardly-rectifying Kv current in urethral smooth muscle cells was carried by Kv2.1-containing channels.30 By comparing the deactivation properties of currents “native” to the urethra with those of Kv2.1 and Kv2.2 channels “cloned” from the urethra in the same report, it was suggested that the native outward current was mediated by Kv2.1 subunits co-assembled with a “silent” Kv channel subunit (i.e., either Kv 6, 8 or 9). RT-PCR and immunocytochemical analyses strongly suggested that the Kv2.1 subunit is co-expressed with a Kv9.3 subunit, in a manner similar to previous reports characterizing native Kv channels in vascular smooth muscle cells57 and more recent reports in bladder.58 Kv9.3 subunits do not form functional channels by themselves. They must be expressed in conjunction with a member of the Kv2 family.59
Figure 3.
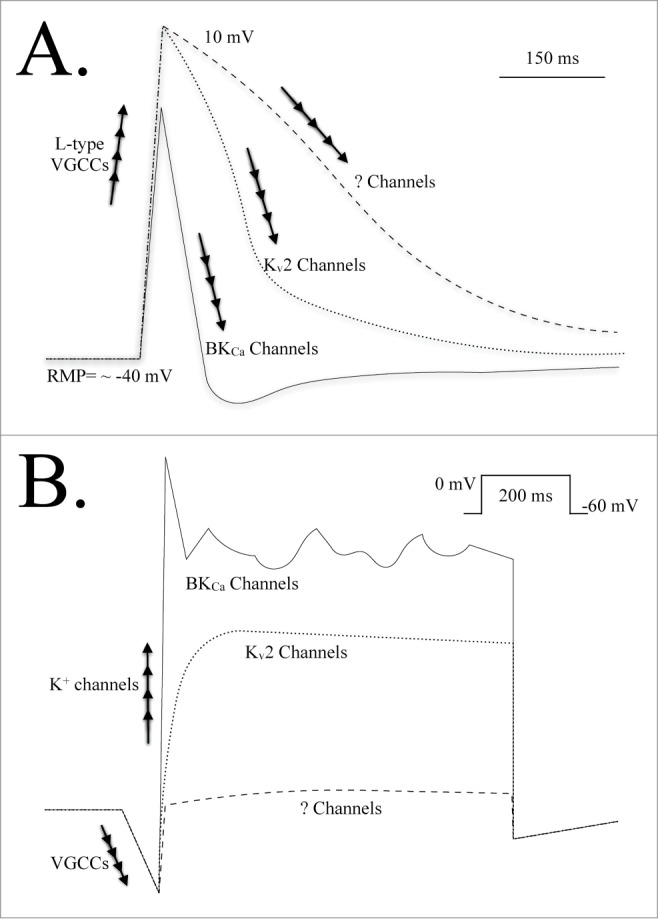
(A) A dissection of the various ion channels responsible for depolarization, repolarization and hyperpolarization of the cell membrane during an action potential event in a urethral smooth muscle cell.30,31 Under control conditions the BKCa channel is primarily responsible for reducing the duration of the action potential spike and hyperpolarizing the membrane potential; Kv2 channels do not contribute to this process.30 When BKCa channel current is suppressed, Kv2 channels are involved in repolarizing the membrane potential. When both BKCa and Kv2 channel currents are suppressed it is not yet known which conductances repolarize and stabilize the membrane potential. The conductances responsible for setting and maintaining/stabilizing the resting membrane potential have also not been reported. (B) An illustration of membrane currents evoked from a urethral smooth muscle cell in response to a step voltage clamp pulse. Depolarization of the cell membrane results in an L-type VGCC inward current. A rapidly activating, transient outward current is evident, carried by BKCa channels that requires VGCC activity and CICR from the SR. This is followed by a sustained and “noisy” outward current also carried by BKCa channels.30,31,40 The delayed-rectifier Kv2 current can be observed when BKCa channel current is inhibited. Inhibition of both BKCa and Kv2 channel currents leaves a small net outward current that has not been fully resolved. Abbreviations: L-type VGCCs, long-lasting voltage-gated Ca2+ channels; ? Channels, unknown channels; BKCa, large conductance, Ca2+-activated K+ channel; Kv2, voltage-gated K+ channels (Shab-related subfamily); mV, millivolt; ms, millisecond; RMP, resting membrane potential.
Intriguingly, while it was firmly established that Kv2 channels carried the majority of the delayed rectifier Kv current in the urethra, these channels apparently contributed very little to action potential repolarisation (Fig. 3A).30,31 Rather, the repolarisation phase of the action potential was heavily dependent on K+ efflux via BKCa channels.30,31,40 However, if BKCa channels were pre-inhibited, the role of the Kv2 channels became apparent, suggesting that Kv2.1-containing channels may represent a possible “fail-safe” brake mechanism for repolarising the cellular membrane potential if the BKCa channels were not properly activated, or otherwise suppressed. Isometric tension studies indeed demonstrated an increase in excitability when the Kv current was inhibited in urethral tissue, particularly when BKCa channels were pre-inhibited.30,31 However, it is worth noting that it has not yet been determined if Kv2.1-containing channels and/or BKCa channels are also present in ICCs within urethral smooth muscle, which could contribute to the observed effect(s).
BKCa channels are unique members of the K+ channel family due to their dual Ca2+- and voltage-sensitive features (see reviews).60,61 In addition, their large single channel conductance makes them formidable players in the regulation of urethral smooth muscle excitability. While most of the transmembrane region of the BKCa channel α subunit superficially resembles that of the Kv channel subunits (Fig. 2), the BKCa channel has a considerably larger C-terminal regulatory domain that is key to its overall function in smooth muscle. This region contains Ca2+-binding sites and can be dynamically regulated by intracellular signaling pathways (e.g., phosphorylation).61,62 While not yet directly reported, it is likely that the BKCa channels expressed in the urethral smooth muscle plasma membrane co-associate with the smooth muscle BKCa channel β subunit, BKCa β1 (Fig. 2), which has been identified in bladder smooth muscle cells.63,64
Electrophysiological studies in urethral smooth muscle cells using the perforated patch (amphotericin-mediated), voltage clamp configuration have provided evidence for an interesting transient-like or A-type59 outward K+ current during membrane depolarization (Fig. 3B) that was not carried by Kv channels.30,31,40 It was determined that this transiently-activating current was in fact carried by BKCa channels (i.e., iberiotoxin-sensitive), and required Ca2+ influx via L-type voltage-gated calcium channels (VGCCs), leading to Ca2+-induced Ca2+ release (CICR) via ryanodine receptors (RyRs) on the sarcoplasmic reticulum (SR) store during depolarization of the cell membrane.31 RyR-mediated Ca2+ release was found to be crucial for the transient activation of BKCa current and this proved important for repolarising the membrane potential, thus shortening the duration of action potential spike events. It was also reported that this transient current could be inhibited by muscarinic receptor activation (i.e., M3), by altering the SR store Ca2+ release mechanisms and mitigating the contribution of the BKCa current to action potential repolarization.31 These data also suggested that the BKCa current could be suppressed during parasympathetic stimulation, as evidenced by broadened action potential events and isometric tension studies. Such findings also supported the idea that the delayed rectifier Kv current was functionally important during BKCa current suppression, as may occur during parasympathetic stimulation.
Possible roles for other K+ channels in urethral smooth muscle
Comprehensive investigation into urethral smooth muscle cell ion channels has lagged behind its bladder smooth muscle cell counterpart, particularly with regard to K+ channels. The reasons for this are multifactorial, and exacerbated by the difficulties associated with obtaining adequate urethral tissue from small rodents including rats and mice. The more readily available bladder tissue, however, has provided a valuable reference point for candidate ion channels in urethral smooth muscle since both bladder and urethra cell types superficially and electrically resemble each other.6,30,40,65,66
The residual urethral smooth muscle delayed rectifier Kv current (i.e., non- Kv2) has not yet been fully characterized. However, parallel studies in bladder smooth muscle cells indicate that this current may be carried by Kv7-containing subunits in either homo- or heteromeric combinations.67,68 While this possibility would present an alternative therapeutic Kv target in urethral smooth muscle, the role of this third conductance would appear to be less significant than those mediated by BKCa or Kv2 channels during action potential events, based on results obtained using currently-available pharmacology.30,31,40
Studies involving bladder smooth muscle cells have definitively reported expression of BKCaβ1 subunits in functional BKCa channel complexes.64,69 Interestingly, BKCaβ4 subunit transcript and protein also appear to be present in bladder smooth muscle cells, although its role, if any, has not yet been elucidated.64,70 It has not yet been reported in urethral smooth muscle cells.
It is certainly worth noting that the K2P channels (Fig. 2) TASK-1 and TASK-2 have been reported by Sang Don Koh and co-workers to be present in bladder smooth muscle cells. These “leak” K+ channels were found to be functionally important in terms of regulating the resting membrane potential.71 Similar investigations focusing on urethral smooth muscle cells would constitute a logical next step in understanding the mechanisms responsible for setting the resting membrane potential. It has also been reported that there is a role for small-conductance, Ca2+-activated K+ (SKCa) channels in bladder smooth muscle cells using patch clamp recordings in combination with intact preparations.72,73 These channels have also been implicated in bladder ICC function.74 Intermediate-conductance, calcium-activated K+ (IKCa) channels have not yet been found to be functionally important in bladder smooth muscle cells,75 although they have been detected at the transcriptional and protein levels.64,72
VGCCs in Urethral Smooth Muscle
Most forms of mammalian smooth muscle employ Ca2+ entry through VGCCs as a key process to elevate intracellular Ca2+ levels.76,77 This has certainly proved to be consistent with action potential spike events observed in urethral smooth muscle cells.30,31,33,39,78
It is well-established that the L-type (long-lasting) VGCCs activate at more depolarized potentials than their T-type (transient-lasting) counterpart.76 It was also previously demonstrated that L-type VGCC current was crucial for generating and maintaining urethral tone in intact preparations.6,79 However, characterization of VGCCs in the individual smooth muscle cells has required extensive biophysical and pharmacological studies utilizing the patch clamp technique. In the early 2000s, urethral smooth muscle cell VGCC currents were examined and it was reported that human and rabbit urethral smooth muscle cells functionally contained both L- and T-type VGCC conductances.33,39,78,80 This was followed up by studies demonstrating that the L-type VGCC current was essential not only for the upstroke in the action potential, but for the transient nature of the BKCa channel current.30,31 While detailed biophysical data are available for the L- and T-type VGCCs, the channel subunit isoforms and regulatory subunits have not yet been identified or described.
P2X Channels
Studies examining purinergic stimulation of urethral smooth muscle have been controversial for a number of reasons. Briefly, ATP activates purinergic receptors that include P2X (ionotropic) ion channels, and the G protein-coupled purinergic P2Y (metabotropic) receptors (see excellent reviews on purinergic receptors).81,82 Both sub-families of these purinergic receptors are known to be expressed in urethral smooth muscle tissue, which further complicates the interpretation of data arising from intact preparations, as discussed previously. This helped contribute to apparently conflicting reports by several groups characterizing purinergic stimulation as both inhibitory83-85 and excitatory28,29 under their respective recording conditions.
Recent patch clamp studies undertaken by Sergeant and coworkers has provided clarifying evidence that purinergic compounds released by nerves in urethral smooth muscle tissue activate the cation-selective P2X receptors expressed on smooth muscle cell membranes and depolarize the membrane potential.28,29 This was found to be in contrast to the ATP-induced activation of ICCs from urethral tissue, which was mediated by P2Y receptors.29 This area of research is ongoing and will likely prove to be more difficult than traditional ion channel studies due to the complex array of purinergic receptors, some of which can “desensitize” to a stimulus during the course of an experiment.81,82
CaCC Channels
Early studies using intact preparations including microelectrode recordings strongly indicated a functional role for Ca2+-activated Cl− channels (CaCCs) in regulating the excitability and function of urethral smooth muscle.25,41 Patch clamp studies on cells isolated from the urethra provided evidence that the CaCC currents were tightly-linked with the pace-making mechanism exhibited by ICCs.78 While CaCC currents could be activated upon depolarization of urethral smooth muscle cells, the vast majority of cells isolated from human and rabbit urethra show no spontaneous activity, unlike ICCs.6,80,86 The most likely candidate CaCC expressed in urethral smooth muscle cells is anoctamine-1 (ANO-1), also known as TMEM16A.87 The possible functional role(s) of ANO-1 in urethral smooth muscle is now undergoing greater scrutiny.88
Concluding Remarks and Future Directions
Selectively-regulating the excitability and tone of the smooth muscles in the lower urinary tract remains the most difficult challenge in battling urinary incontinence. Over the last 2 decades, research examining the functionality of urethral smooth muscle, which provides a crucial “internal sphincter” mechanism, has greatly advanced. It is well-established that the behaviors of ion channels expressed at the surface membrane are closely-tied to smooth muscle excitability and tone. Most therapies for urinary incontinence currently available attempt to selectively-target bladder function and achieve only limited success. These treatments have notorious system-wide side-effects that promote poor patient-compliance and reduce quality of life. Unfortunately, this is a consequence of the limited range and bioavailability of therapeutic targets in the bladder, combined with a lack of tissue-specific available pharmacologic tools. Novel developments in understanding the respective roles of several K+ channels (BKCa, Kv2 and Kv7 families) in regulating bladder excitability has generated interest in developing selective K+ channel agonists for overactive bladder treatments. While it is likely that this would reduce detrusor smooth muscle excitability, parallel advances in urethral smooth muscle studies indicate that similar channels are also present in this tissue, and that enhancing their activity may promote undesired relaxation in the urethra.
Recently a range of novel therapeutic targets have been identified in urethral smooth muscle cells that include several members of the K+ channel families, VGCCs, purinergic receptors and CaCCs. In addition, several members of their signaling pathways have been reported (e.g., RyRs, muscarinic receptors), adding to the richness of potential therapeutic targets. In addition to directly targeting these channels and their respective signaling pathways with activators and/or inhibitors, other approaches to manipulate ion channel behavior may be available. For instance, determining that a specific subunit is contained in a heterotetrameric ion channel (e.g., Kv2.1/9.3 channels or Kir6.1/6.2), or an associated accessory subunit (BKCaβ1), may serve as a signature and provide new opportunities to selectively-target ion channel complexes expressed primarily in the bladder and/or urethra. Additionally, selectively-targeting established signaling pathways may promote desired ion channel behavior. Such a strategy, for instance, is exploited by sildenafil, the active ingredient in Viagra™, in erectile tissues (i.e. PDE5 inhibition).
Strategies involving molecular biology (e.g., genetic) can now be explored with greater confidence in urethral smooth muscle in the form of directly and selectively upregulating/downregulating ion channel expression (e.g., plasmid-infused). Similarly, by targeting tissue-specific ion channel subunit chaperones and regulatory proteins (e.g., BKCa channel chaperones) it is possible to selectively increase or decrease a particular ion channel expression at the surface membrane.61,89,90 Controlling BKCa channel protein turnover is a powerful determinant of the cell excitability,61 but more study will be required to understand the trafficking and chaperoning mechanisms of these channel proteins in urethra and bladder smooth muscle before a useful strategy can be utilized.
Given the prominent role of BKCa channels in regulating urethral smooth muscle cell excitability, it is likely that this channel will become a primary therapeutic target in the near future, similar to current bladder research and development projects. In the short term, it is likely that successful strategies will make use of the cell's existing machinery that dynamically regulate ion channel excitability (e.g., PDEs, PKC,38 PKG,62). PDE4 has recently emerged as an upstream BKCa channel modulator in bladder smooth muscle cells,91 and thus may be a candidate for similar modulation in urethral smooth muscle. While there has been much recent progress in understanding urethral smooth muscle regulation, it is disappointing that no clear candidate protein/channel has emerged that is distinguishable from bladder smooth muscle. Although K2P, Kv7 and BKCaβ4 protein expression has been reported in bladder, it remains to be seen whether they will be found to be functionally present in urethral smooth muscle.
Disclosure of Potential Conflicts of Interest
No potential conflicts of interest were disclosed.
Acknowledgments
The author thanks Drs Andrew P Braun, Michael P Walsh and Alejandro Moreno-Domínguez for their critical evaluation and proof-reading of the manuscript.
References
- 1. Burdyga T, Wray S. Action potential refractory period in ureter smooth muscle is set by Ca sparks and BK channels. Nature [Internet] 2005. [cited 2014 Mar 3]; 436:559-62. Available from: http://www.ncbi.nlm.nih.gov/pubmed/16049489; PMID:16049489; http://dx.doi.org/ 10.1038/nature03834 [DOI] [PubMed] [Google Scholar]
- 2. Lang RJ, Takano H, Davidson ME, Suzuki H, Klemm MF. Characterization of the spontaneous electrical and contractile activity of smooth muscle cells in the rat upper urinary tract. J Urol [Internet] 2001. [cited 2014 Mar 14]; 166:329-34. Available from: http://www.ncbi.nlm.nih.gov/pubmed/11435895; PMID:11435895; http://dx.doi.org/ 10.1016/S0022-5347(05)66154-X [DOI] [PubMed] [Google Scholar]
- 3. De Groat WC, Ryall RW. Reflexes to sacral parasympathetic neurones concerned with micturition in the cat. J Physiol [Internet] 1969. [cited 2014 Mar 14]; 200:87-108. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=1350419&tool=pmcentrez&ren-dertype=abstract; PMID:5248885 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.De Groat WC, Ryall RW. Recurrent inhibition in sacral parasympathetic pathways to the bladder. J Physiol [Internet] 1968. [cited 2014 Mar 14]; 196:579-91. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=1351765&tool=pmcentrez&rendertype=abstract; PMID:5243140 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Birder LA. Nervous network for lower urinary tract function. Int J Urol [Internet] 2013 [cited 2014 Jul 22]; 20:4-12. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=4036083&tool=pmcentrez&rendertype=abstract; http://dx.doi.org/ 10.1111/j.1442-2042.2012.03210.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Brading AF. Spontaneous activity of lower urinary tract smooth muscles: correlation between ion channels and tissue function. J Physiol [Internet] 2006. [cited 2014 Mar 3]; 570:13-22. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=1464291&tool=pmcentrez&rendertype=abstract; PMID:16210349; http://dx.doi.org/ 10.1113/jphysiol.2005.097311 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Thor KB, de Groat WC. Neural control of the female urethral and anal rhabdosphincters and pelvic floor muscles. Am J Physiol Regul Integr Comp Physiol [Internet] 2010. [cited 2014 Mar 26]; 299:R416-38. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=2928615&tool=pmcentrez&rendertype=abstract; PMID:20484700 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Thor KB, Hisamitsu T, Roppolo JR, Tuttle P, Nagel J, deGroat WC. Selective inhibitory effects of ethylketocyclazocine on reflex pathways to the external urethral sphincter of the cat. J Pharmacol Exp Ther [Internet] 1989. [cited 2014 Mar 26]; 248:1018-25. Available from: http://www.ncbi.nlm.nih.gov/pubmed/2539452; PMID:2539452 [PubMed] [Google Scholar]
- 9. Thor KB, Muhlhauser MA. Vesicoanal, urethroanal, and urethrovesical reflexes initiated by lower urinary tract irritation in the rat. Am J Physiol [Internet] 1999. [cited 2014 Mar 22]; 277:R1002-12. Available from: http://www.ncbi.nlm.nih.gov/pubmed/10516238; PMID:10516238 [DOI] [PubMed] [Google Scholar]
- 10. Thor KB. Targeting serotonin and norepinephrine receptors in stress urinary incontinence. Int J Gynaecol Obstet [Internet] 2004. [cited 2014 Mar 26]; 86 Suppl 1:S38-52. Available from: http://www.ncbi.nlm.nih.gov/pubmed/15302566; PMID:15302566; http://dx.doi.org/ 10.1016/j.ijgo.2004.04.028 [DOI] [PubMed] [Google Scholar]
- 11. Al-Noah Z, McKenna D, Langdale C, Thor KB, Marson L, Burgard E, Kullmann FA. Nitrergic relaxations and phenylephrine contractions are not compromised in isolated urethra in a rat model of diabetes. Auton Neurosci 2014; 183:58-65; PMID:24656892; 10897320http://www.ncbi.nlm.nih.gov/pubmed/24656892 [DOI] [PubMed] [Google Scholar]
- 12. Keane DP, O’Sullivan S. Urinary incontinence: anatomy, physiology and pathophysiology. Baillieres Best Pract Res Clin Obstet Gynaecol [Internet] 2000. [cited 2014 Mar 14]; 14:207-26. Available from: http://www.ncbi.nlm.nih.gov/pubmed/10897320; PMID:10897320; http://dx.doi.org/ 10.1053/beog.1999.0072 [DOI] [PubMed] [Google Scholar]
- 13. Canda AE, Cinar MG, Turna B, Sahin MO. Pharmacologic targets on the female urethra. Urol Int [Internet] 2008. [cited 2014 Jul 28]; 80:341-54. Available from: http://www.ncbi.nlm.nih.gov/pubmed/18587243; http://dx.doi.org/ 10.1159/000132690 [DOI] [PubMed] [Google Scholar]
- 14. Brading AF, Teramoto N, Dass N, McCoy R. Morphological and physiological characteristics of urethral circular and longitudinal smooth muscle. Scand J Urol Nephrol Suppl [Internet] 2001. [cited 2014 Mar 26]; 12-8; discussion 106–25. Available from: http://www.ncbi.nlm.nih.gov/pubmed/11409610; PMID:11409610; http://dx.doi.org/ 10.1080/003655901750174818 [DOI] [PubMed] [Google Scholar]
- 15. Tanagho EA, Smith DR. The anatomy and function of the bladder neck. Br J Urol [Internet] 1966. [cited 2014 Jul 28]; 38:54-71. Available from: http://www.ncbi.nlm.nih.gov/pubmed/4952316; http://dx.doi.org/ 10.1111/j.1464-410X.1966.tb09679.x [DOI] [PubMed] [Google Scholar]
- 16. Tanagho EA, Miller ER, Meyers FH, Corbett RK. Observations on the daynamics of the bladder neck 1. Br J Urol [Internet] 1966. [cited 2014 Jul 28]; 38:72-84. Available from: http://doi.wiley.com/10.1111/j.1464-410X.1966.tb09680.x; PMID:4952318; http://dx.doi.org/ 10.1111/j.1464-410X.1966.tb09680.x [DOI] [PubMed] [Google Scholar]
- 17. Tanagho EA, Smith DR, Guthrie TH. Pathophysiology of functional ureteral obstruction. J Urol [Internet] 1970. [cited 2014 Jul 28]; 104:73-88. Available from: http://www.ncbi.nlm.nih.gov/pubmed/5426713 [DOI] [PubMed] [Google Scholar]
- 18. Tanagho EA, Smith DR. Clinical evaluation of a surgical technique for the correction of complete urinary incontinence. J Urol [Internet] 1972. [cited 2014 Jul 28]; 107:402-11. Available from: http://www.ncbi.nlm.nih.gov/pubmed/4551624; PMID:4551624 [DOI] [PubMed] [Google Scholar]
- 19. Lapides J. Evacuation of the bladder via abdominal urethrostomy. J Urol [Internet] 1985. [cited 2014 Jul 28]; 133:253. Available from: http://www.ncbi.nlm.nih.gov/pubmed/4038523; PMID:4038523 [DOI] [PubMed] [Google Scholar]
- 20. Lapides J, Tank ES. Urinary complications following abdominal perineal resection. Cancer [Internet] 1971. [cited 2014 Jul 28]; 28:230-5. Available from: http://www.ncbi.nlm.nih.gov/pubmed/4106734; PMID:4106734; http://dx.doi.org/ [DOI] [PubMed] [Google Scholar]
- 21. Lapides J. Structure and function of the internal vesical sphincter. J Urol [Internet] 1958. [cited 2014 Jul 28]; 80:341-53. Available from: http://www.ncbi.nlm.nih.gov/pubmed/13588737; PMID:13588737 [DOI] [PubMed] [Google Scholar]
- 22. Osman NI, Mangera A, Chapple CR. A systematic review of surgical techniques used in the treatment of female urethral stricture. Eur Urol [Internet] 2013. [cited 2014 Jul 28]; 64:965-73. Available from: http://www.europeanurology.com/article/S0302-2838(13)00795-1/fu-lltext/a-systematic-review -of-surgical-techniques-used-in-the-treat-ment-of-female-urethral-stricture [DOI] [PubMed] [Google Scholar]
- 23. Kirkali Z, Canda AE. Superficial urothelial cancer in the prostatic urethra. ScientificWorldJ [Internet] 2006. [cited 2014 Jul 28]; 6:2603-10. Available from: http://www.ncbi.nlm.nih.gov/pubmed/17619737; http://dx.doi.org/ 10.1016/j.eururo.2013.07.038 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Brading AF, McCloskey KD. Mechanisms of disease: specialized interstitial cells of the urinary tract–an assessment of current knowledge. Nat Clin Pract Urol [Internet] 2005. [cited 2014 Mar 21]; 2:546-54. Available from: http://www.ncbi.nlm.nih.gov/pubmed/16474598; PMID:16474598; http://dx.doi.org/ 10.1038/ncpuro0340 [DOI] [PubMed] [Google Scholar]
- 25. Ito Y, Kimoto Y. The neural and non-neural mechanisms involved in urethral activity in rabbits. J Physiol [Internet] 1985. [cited 2014 Mar 26]; 367:57-72. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=1193053&tool=pmcentrez&rendertype=abstract; PMID:4057107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Mattiasson A, Andersson KE, Andersson PO, Larsson B, Sjögren C, Uvelius B. Nerve-mediated functions in the circular and longitudinal muscle layers of the proximal female rabbit urethra. J Urol [Internet] 1990. [cited 2014 Mar 26]; 143:155-60. Available from: http://www.ncbi.nlm.nih.gov/pubmed/2294247; PMID:2294247 [DOI] [PubMed] [Google Scholar]
- 27. Hashitani H, Yanai Y, Kohri K, Suzuki H. Heterogeneous CPA sensitivity of spontaneous excitation in smooth muscle of the rabbit urethra. Br J Pharmacol [Internet] 2006. [cited 2014 Mar 26]; 148:340-9. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=1751569&tool=pmcentrez&rendertype=abstract; PMID:16582935 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Bradley E, Kadima S, Drumm B, Hollywood MA, Thornbury, McHale NG, Sergeant GP. Novel excitatory effects of adenosine triphosphate on contractile and pacemaker activity in rabbit urethral smooth muscle. J Urol [Internet] 2010. [cited 2014 Mar 21]; 183:801-11. Available from: http://www.ncbi.nlm.nih.gov/pubmed/20022059; PMID:20022059; http://dx.doi.org/ 10.1016/j.juro.2009.09.075 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Bradley E, Kadima S, Kyle B, Hollywood MA, Thornbury KD, McHale NG, Sergeant GP. P2X receptor currents in smooth muscle cells contribute to nerve mediated contractions of rabbit urethral smooth muscle. J Urol [Internet] 2011. [cited 2014 Mar 21]; 186:745-52. Available from: http://www.ncbi.nlm.nih.gov/pubmed/21683405; PMID:21683405; http://dx.doi.org/ 10.1016/j.juro.2011.03.140 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Kyle B, Bradley E, Ohya S, Sergeant GP, McHale NG, Thornbury KD, Hollywood MA. Contribution of Kv2.1 channels to the delayed rectifier current in freshly dispersed smooth muscle cells from rabbit urethra. Am J Physiol Cell Physiol [Internet] 2011. [cited 2014 Mar 3]; 301:C1186-200. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3213918&tool=pmcentrez&rendertype=abstract; PMID:21813710 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Kyle BD, Bradley E, Large R, Sergeant GP, McHale NG, Thornbury KD, Hollywood MA. Mechanisms underlying activation of transient BK current in rabbit urethral smooth muscle cells and its modulation by IP3-generating agonists. Am J Physiol Cell Physiol [Internet] 2013. [cited 2014 Mar 2]; 305:C609-22. Available from: http://www.ncbi.nlm.nih.gov/pubmed/23804200; PMID:23804200; http://dx.doi.org/ 10.1152/ajpcell.00025.2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Hashitani H, Edwards FR. Spontaneous and neurally activated depolarizations in smooth muscle cells of the guinea-pig urethra. J Physiol [Internet] 1999. [cited 2014 Mar 26]; 514 (Pt 2:459-70. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=2269076&tool=pmcentrez&rendertype=abstract; PMID:9852327; http://dx.doi.org/ 10.1111/j.1469-7793.1999.459ae.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Bradley JE, Anderson UA, Woolsey SM, Thornbury KD, McHale NG, Hollywood MA. Characterization of T-type calcium current and its contribution to electrical activity in rabbit urethra. Am J Physiol Cell Physiol [Internet] 2004. [cited 2014 Mar 26]; 286:C1078-88. Available from: http://www.ncbi.nlm.nih.gov/pubmed/15075207; PMID:15075207 [DOI] [PubMed] [Google Scholar]
- 34. Teramoto N, Brading AF. Activation by levcromakalim and metabolic inhibition of glibenclamide-sensitive K channels in smooth muscle cells of pig proximal urethra. Br J Pharmacol [Internet] 1996. [cited 2014 Mar 26]; 118:635-42. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=1909732&tool=pmcentrez&rendertype=abstract; PMID:8762088 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Teramoto N, Brading AF. Nicorandil activates glibenclamide-sensitive K +channels in smooth muscle cells of pig proximal urethra. J Pharmacol Exp Ther [Internet] 1997. [cited 2014 Mar 26]; 280:483-91. Available from: http://www.ncbi.nlm.nih.gov/pubmed/8996232; PMID:8996232 [PubMed] [Google Scholar]
- 36. Teramoto N, McMurray G, Brading AF. Effects of levcromakalim and nucleoside diphosphates on glibenclamide-sensitive K+ channels in pig urethral myocytes. Br J Pharmacol [Internet] 1997. [cited 2014 Mar 26]; 120:1229-40. Available from: http://www.pubmedcentral.nih.gov /article-render.fcgi ?art-id=1564596&tool=pmcentrez&rendertype=abstract; PMID:9105697 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Teramoto N, Brading AF, Ito Y. Glibenclamide-sensitive K+ channels underlying levcromakalim-induced relaxation in pig urethra. Eur J Pharmacol [Internet] 1999. [cited 2014 Mar 26]; 365:291-300. Available from: http://www.ncbi.nlm.nih.gov/pubmed/9988114; http://dx.doi.org/ 10.1016/S0014-2999(98)00885-1; PMID:9988114 [DOI] [PubMed] [Google Scholar]
- 38. Teramoto N, Zhu H-L, Shibata A, Aishima M, Walsh EJ, Nagao M, Cole WC. ATP-sensitive K+ channels in pig urethral smooth muscle cells are heteromultimers of Kir6.1 and Kir6.2. Am J Physiol Renal Physiol [Internet] 2009. [cited 2014 Mar 26]; 296:F107-17. Available from: http://www.ncbi.nlm.nih.gov/pubmed/18945825; PMID:18945825 [DOI] [PubMed] [Google Scholar]
- 39. Hollywood MA, Woolsey S, Walsh IK, Keane PF, McHale NG, Thornbury KD. T- and L-type Ca2+ currents in freshly dispersed smooth muscle cells from the human proximal urethra. J Physiol [Internet] 2003. [cited 2014 Mar 26]; 550:753-64. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=2343068&tool=pmcentrez&rendertype=abstract; http://dx.doi.org/ 10.1113/jphysiol.2003.043695; PMID:12807987 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Hollywood MA, McCloskey KD, McHale NG, Thornbury KD. Characterization of outward K(+) currents in isolated smooth muscle cells from sheep urethra. Am J Physiol Cell Physiol [Internet] 2000. [cited 2014 Mar 26]; 279:C420-8. Available from: http://www.ncbi.nlm.nih.gov/pubmed/10913009; PMID:10913009 [DOI] [PubMed] [Google Scholar]
- 41. Hashitani H, Van Helden DF, Suzuki H. Properties of spontaneous depolarizations in circular smooth muscle cells of rabbit urethra. Br J Pharmacol [Internet] 1996. [cited 2014 Mar 28]; 118:1627-32. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=1909835&tool=pmcentrez&rendertype=abstract; PMID:8842424 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Sanders KM, Ward SM, Koh SD. Interstitial cells: regulators of smooth muscle function. Physiol Rev [Internet] 2014. [cited 2014 Jul 21]; 94:859-907. Available from: http://www.ncbi.nlm.nih.gov/pubmed/24987007; http://dx.doi.org/ 10.1152/physrev.00037.2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. McHale NG, Hollywood MA, Sergeant GP, Shafei M, Thornbury KT, Ward SM. Organization and function of ICC in the urinary tract. J Physiol [Internet] 2006. [cited 2014 Mar 26]; 576:689-94. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=1890397&tool=pmcentrez&rendertype=abstract; PMID:16916908; http://dx.doi.org/ 10.1113/jphysiol.2006.116657 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. McCloskey KD. Interstitial cells of Cajal in the urinary tract. Handb Exp Pharmacol [Internet] 2011. [cited 2014 Mar 21]; :233-54. Available from: http://www.ncbi.nlm.nih.gov/pubmed/21290229; http://dx.doi.org/ 10.1007/978-3-642-16499-6 [DOI] [PubMed] [Google Scholar]
- 45. Hamill OP, Marty A, Neher E, Sakmann B, Sigworth FJ. Improved patch-clamp techniques for high-resolution current recording from cells and cell-free membrane patches. Pflugers Arch [Internet] 1981. [cited 2014 Mar 19]; 391:85-100. Available from: http://www.ncbi.nlm.nih.gov/pubmed/6270629; PMID:6270629; http://dx.doi.org/ 10.1007/BF00656997 [DOI] [PubMed] [Google Scholar]
- 46. Teramoto N, Creed KE, Brading AF. Activity of glibenclamide-sensitive K +channels under unstimulated conditions in smooth muscle cells of pig proximal urethra. Naunyn Schmiedebergs Arch Pharmacol [Internet] 1997. [cited 2014 Mar 26]; 356:418-24. Available from: http://www.ncbi.nlm.nih.gov/pubmed/9303582; PMID:9303582; http://dx.doi.org/ 10.1007/PL00005071 [DOI] [PubMed] [Google Scholar]
- 47. Teramoto N. Physiological roles of ATP-sensitive K+ channels in smooth muscle. J Physiol [Internet] 2006. [cited 2014 Mar 26]; 572:617-24. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=1779997&tool=pmcentrez&rendertype=abstract; PMID:16484295; http://dx.doi.org/ 10.1113/jphysiol.2006.105973 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Teramoto N, Yunoki T, Tanaka K, Takano M, Masaki I, Yonemitsu Y, Sueishi K, Ito Y. The effects of caffeine on ATP-sensitive K(+) channels in smooth muscle cells from pig urethra. Br J Pharmacol [Internet] 2000. [cited 2014 Mar 26]; 131:505-13. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=1572345&tool=pmcentrez&rendertype=abstract; PMID:11015301 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Teramoto N. [Molecular and electrophysiological investigation of ATP-sensitive K+ channels in lower urinary tract function: the aims for clinical treatment of unstable detrusor]. Nihon Yakurigaku Zasshi [Internet] 2003. [cited 2014 Mar 26]; 121:317-24. Available from: http://www.ncbi.nlm.nih.gov/pubmed/12784733; PMID:12784733 [DOI] [PubMed] [Google Scholar]
- 50. Yunoki T, Teramoto N, Ito Y. Functional involvement of sulphonylurea receptor (SUR) type 1 and 2B in the activity of pig urethral ATP-sensitive K+ channels. Br J Pharmacol [Internet] 2003. [cited 2014 Mar 26]; 139:652-60. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=1573868&tool=pmcentrez&rendertype=abstract; PMID:12788825; http://dx.doi.org/ 10.1254/fpj.121.317 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Teramoto N, Tomoda T, Yunoki T, Brading AF, Ito Y. Modification of ATP-sensitive K+ channels by proteolysis in smooth muscle cells from pig urethra. Life Sci [Internet] 2002. [cited 2014 Mar 26]; 72:475-85. Available from: http://www.ncbi.nlm.nih.gov/pubmed/12467888; PMID:12467888; http://dx.doi.org/ 10.1016/S0024-3205(02)02284-1 [DOI] [PubMed] [Google Scholar]
- 52. Cole WC, Clément-Chomienne O. ATP-sensitive K+ channels of vascular smooth muscle cells. J Cardiovasc Electrophysiol [Internet] 2003. [cited 2014 Jul 29]; 14:94-103. Available from: http://www.ncbi.nlm.nih.gov/pubmed/12625619; PMID:12625619; http://dx.doi.org/ 10.1046/j.1540-8167.2003.02376.x [DOI] [PubMed] [Google Scholar]
- 53. Cole WC, Malcolm T, Walsh MP, Light PE. Inhibition by protein kinase C of the K(NDP) subtype of vascular smooth muscle ATP-sensitive potassium channel. Circ Res [Internet] 2000. [cited 2014 Jul 29]; 87:112-7. Available from: http://www.ncbi.nlm.nih.gov/pubmed/10903994; PMID:10903994; http://dx.doi.org/ 10.1161/01.RES.87.2.112 [DOI] [PubMed] [Google Scholar]
- 54. Escoubas P, Diochot S, Célérier M-L, Nakajima T, Lazdunski M. Novel tarantula toxins for subtypes of voltage-dependent potassium channels in the Kv2 and Kv4 subfamilies. Mol Pharmacol [Internet] 2002. [cited 2014 Mar 26]; 62:48-57. Available from: http://www.ncbi.nlm.nih.gov/pubmed/12065754; PMID:12065754; http://dx.doi.org/ 10.1124/mol.62.1.48 [DOI] [PubMed] [Google Scholar]
- 55. Escoubas P, Rash L. Tarantulas: eight-legged pharmacists and combinatorial chemists. Toxicon [Internet] 2004. [cited 2014 Mar 26]; 43:555-74. Available from: http://www.ncbi.nlm.nih.gov/pubmed/15066413; PMID:15066413; http://dx.doi.org/ 10.1016/j.toxicon.2004.02.007 [DOI] [PubMed] [Google Scholar]
- 56. Moreno-Domínguez A, Cidad P, Miguel-Velado E, López-López JR, Pérez-García MT. De novo expression of Kv6.3 contributes to changes in vascular smooth muscle cell excitability in a hypertensive mice strain. J Physiol [Internet] 2009. [cited 2014 Mar 26]; 587:625-40. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=2670085&tool=pmcentrez&rendertype=abstract; PMID:19074965; http://dx.doi.org/ 10.1113/jphysiol.2008.165217 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Zhong XZ, Abd-Elrahman KS, Liao C-H, El-Yazbi AF, Walsh EJ, Walsh MP, Cole WC. Stromatoxin-sensitive, heteromultimeric Kv2.1/Kv9.3 channels contribute to myogenic control of cerebral arterial diameter. J Physiol [Internet] 2010. [cited 2014 Mar 26]; 588:4519-37. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3008855&tool=pmcentrez&rendertype=abstract; PMID:20876197; http://dx.doi.org/ 10.1113/jphysiol.2010.196618 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Hristov KL, Chen M, Soder RP, Parajuli SP, Cheng Q, Kellett WF, Petkov G V. KV2.1 and electrically silent KV channel subunits control excitability and contractility of guinea pig detrusor smooth muscle. Am J Physiol Cell Physiol [Internet] 2012. [cited 2014 Mar 3]; 302:C360-72. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3328844&tool=pmcentrez&rendertype=abstract; PMID:21998137 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Cox RH. Molecular determinants of voltage-gated potassium currents in vascular smooth muscle. Cell Biochem Biophys [Internet] 2005. [cited 2014 Mar 28]; 42:167-95. Available from: http://www.ncbi.nlm.nih.gov/pubmed/15858231; PMID:15858231; http://dx.doi.org/ 10.1385/CBB:42:2:167 [DOI] [PubMed] [Google Scholar]
- 60. Toro L, Li M, Zhang Z, Singh H, Wu Y, Stefani E. MaxiK channel and cell signalling. Pflugers Arch [Internet] 2013. [cited 2014 Feb 21]; Available from: http://www.ncbi.nlm.nih.gov/pubmed/24077696 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Kyle BD, Braun AP. The regulation of BK channel activity by pre- and post-translational modifications. Front Physiol [Internet] 2014. [cited 2014 Aug 3]; 5 Available from: http://journal.frontiersin.org/Journal/10.3389/fphys.2014.00316/impact#impact [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Kyle BD, Hurst S, Swayze RD, Sheng J, Braun AP. Specific phosphorylation sites underlie the stimulation of a large conductance, Ca(2+)-activated K(+) channel by cGMP-dependent protein kinase. FASEB J [Internet] 2013. [cited 2014 Mar 3]; 27:2027-38. Available from: http://www.ncbi.nlm.nih.gov/pubmed/23407708; PMID:23407708; http://dx.doi.org/ 10.1096/fj.12-223669 [DOI] [PubMed] [Google Scholar]
- 63. Chen M, Petkov G V. Identification of large conductance calcium activated potassium channel accessory beta4 subunit in rat and mouse bladder smooth muscle. J Urol [Internet] 2009. [cited 2014 Mar 21]; 182:374-81. Available from: http://www.ncbi.nlm.nih.gov/pubmed/19447431; PMID:19447431; http://dx.doi.org/ 10.1016/j.juro.2009.02.109 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Petkov G V. Role of potassium ion channels in detrusor smooth muscle function and dysfunction. Nat Rev Urol [Internet] 2012. [cited 2014 Mar 21]; 9:30-40. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3759241&tool=pmcentrez&rendertype=abstract; http://dx.doi.org/ 10.1038/nrurol.2011.194 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Heppner TJ, Bonev AD, Nelson MT. Ca(2+)-activated K+ channels regulate action potential repolarization in urinary bladder smooth muscle. Am J Physiol Cell Physiol [Internet] 1997. [cited 2014 Jul 7]; 273:C110-117. Available from: http://ajpcell.physiology.org/content/273/1/C110; PMID:9252448 [DOI] [PubMed] [Google Scholar]
- 66. Thorneloe KS, Nelson MT. Properties and molecular basis of the mouse urinary bladder voltage-gated K+ current. J Physiol [Internet] 2003. [cited 2014 Jul 28]; 549:65-74. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=2342925&tool=pmcentrez&rendertype=abstract; PMID:12679374; http://dx.doi.org/ 10.1113/jphysiol.2003.039859 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Afeli SAY, Malysz J, Petkov G V. Molecular expression and pharmacological evidence for a functional role of kv7 channel subtypes in Guinea pig urinary bladder smooth muscle. PLoS One [Internet] 2013. [cited 2014 Mar 21]; 8:e75875. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3779188&tool=pmcentrez&rendertype=abstract; PMID:24073284; http://dx.doi.org/ 10.1371/journal.pone.0075875 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Anderson UA, Carson C, Johnston L, Joshi S, Gurney AM, McCloskey KD. Functional expression of KCNQ (Kv 7) channels in guinea pig bladder smooth muscle and their contribution to spontaneous activity. Br J Pharmacol [Internet] 2013. [cited 2014 Mar 21]; 169:1290-304. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3746117&tool=pmcentrez&rendertype=abstract; PMID:23586426 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Petkov G V, Bonev AD, Heppner TJ, Brenner R, Aldrich RW, Nelson MT. Beta1-subunit of the Ca2+-activated K+ channel regulates contractile activity of mouse urinary bladder smooth muscle. J Physiol [Internet] 2001. [cited 2014 Mar 21]; 537:443-52. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=2278973&tool=pmcentrez&rendertype=abstract; PMID:11731577; http://dx.doi.org/ 10.1111/j.1469-7793.2001.00443.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Hristov KL, Chen M, Kellett WF, Rovner ES, Petkov GV. Large-conductance voltage- and Ca2+-activated K+ channels regulate human detrusor smooth muscle function. Am J Physiol Cell Physiol [Internet] 2011. [cited 2014 Mar 3]; 301:C903-12. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3191561&tool=pmcentrez&rendertype=abstract; PMID:21697543; http://dx.doi.org/ 10.1152/ajpcell.00495.2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Beckett EAH, Han I, Baker SA, Han J, Britton FC, Koh SD. Functional and molecular identification of pH-sensitive K+ channels in murine urinary bladder smooth muscle. BJU Int [Internet] 2008. [cited 2014 Mar 27]; 102:113-24. Available from: http://www.ncbi.nlm.nih.gov/pubmed/18394011; PMID:18394011; http://dx.doi.org/ 10.1111/j.1464-410X.2008.07541.x [DOI] [PubMed] [Google Scholar]
- 72. Parajuli SP, Soder RP, Hristov KL, Petkov G V. Pharmacological activation of small conductance calcium-activated potassium channels with naphtho[1,2-d]thiazol-2-ylamine decreases guinea pig detrusor smooth muscle excitability and contractility. J Pharmacol Exp Ther [Internet] 2012. [cited 2014 Mar 21]; 340:114-23. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3251021&tool=pmcentrez&rendertype=abstract; http://dx.doi.org/ 10.1124/jpet.111.186213 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Soder RP, Parajuli SP, Hristov KL, Rovner ES, Petkov G V. SK channel-selective opening by SKA-31 induces hyperpolarization and decreases contractility in human urinary bladder smooth muscle. Am J Physiol Regul Integr Comp Physiol [Internet] 2013. [cited 2014 Mar 21]; 304:R155-63. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3543661&tool=pmcentrez&rendertype=abstract; PMID:23174857 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. Lee H, Koh BH, Peri LE, Sanders KM, Koh SD. Functional expression of SK channels in murine detrusor PDGFR+ cells. J Physiol [Internet] 2013. [cited 2014 Mar 27]; 591:503-13. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3577524&tool=pmcentrez&rendertype=abstract; PMID:23148317; http://dx.doi.org/ 10.1113/jphysiol.2012.241505 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Afeli SAY, Rovner ES, Petkov G V. SK but not IK channels regulate human detrusor smooth muscle spontaneous and nerve-evoked contractions. Am J Physiol Renal Physiol [Internet] 2012. [cited 2014 Mar 21]; 303:F559-68. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3423111&tool=pmcentrez&rendertype=abstract [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Harraz OF, Altier C. STIM1-mediated bidirectional regulation of Ca(2+) entry through voltage-gated calcium channels (VGCC) and calcium-release activated channels (CRAC). Front Cell Neurosci [Internet] 2014. [cited 2014 Mar 21]; 8:43. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3932444&tool=pmcentrez&rendertype=abstract; PMID:24605083; http://dx.doi.org/ 10.3389/fncel.2014.00043 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77. Harraz OF, ABD El-Rahman RR, Bigdely-Shamloo K, Wilson SM, Brett SE, Romero M, Gonzales AL, Earley S, Vigmond E, Nygren A, et al. CaV3.2 Channels and the Induction of Negative Feedback In Cerebral Arteries. Circ Res [Internet] 2014. [cited 2014 Aug 1]; :CIRCRESAHA.114.304056–. Available from: http://circres.ahajournals.org/content/early/2014/08/01/CIRCRESAHA.114.304056.abstract; PMID:25085940 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78. Sergeant GP, Hollywood MA, McCloskey KD, Thornbury KD, McHale NG. Specialised pacemaking cells in the rabbit urethra. J Physiol [Internet] 2000. [cited 2014 Mar 21]; 526 Pt 2:359-66. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=2270007&tool=pmcentrez&rendertype=abstract; PMID:10896724; http://dx.doi.org/ 10.1111/j.1469-7793.2000.t01-2-00359.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79. Bridgewater M, MacNeil HF, Brading AF. Regulation of tone in pig urethral smooth muscle. J Urol [Internet] 1993. [cited 2014 Aug 6]; 150:223-8. Available from: http://www.ncbi.nlm.nih.gov/pubmed/7685424 [DOI] [PubMed] [Google Scholar]
- 80. Sergeant GP, Hollywood MA, McHale NG, Thornbury KD. Spontaneous Ca2+ activated Cl- currents in isolated urethral smooth muscle cells. J Urol [Internet] 2001. [cited 2014 Mar 21]; 166:1161-6. Available from: http://www.ncbi.nlm.nih.gov/pubmed/11490316; http://dx.doi.org/ 10.1016/S0022-5347(05)65939-3 [DOI] [PubMed] [Google Scholar]
- 81. Burnstock G. Historical review: ATP as a neurotransmitter. Trends Pharmacol Sci [Internet] 2006. [cited 2014 Mar 26]; 27:166-76. Available from: http://www.ncbi.nlm.nih.gov/pubmed/16487603; http://dx.doi.org/ 10.1016/j.tips.2006.01.005 [DOI] [PubMed] [Google Scholar]
- 82. North RA, Jarvis MF. P2X receptors as drug targets. Mol Pharmacol [Internet] 2013. [cited 2014 Mar 28]; 83:759-69. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3608433&tool=pmcentrez&rendertype=abstract; http://dx.doi.org/ 10.1124/mol.112.083758 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83. Pinna C, Puglisi L, Burnstock G. ATP and vasoactive intestinal polypeptide relaxant responses in hamster isolated proximal urethra. Br J Pharmacol [Internet] 1998. [cited 2014 Mar 28]; 124:1069-74. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=1565480&tool=pmcentrez&rendertype=abstract; PMID:9720775 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84. Pinna C, Glass R, Knight GE, Bolego C, Puglisi L, Burnstock G. Purine- and pyrimidine-induced responses and P2Y receptor characterization in the hamster proximal urethra. Br J Pharmacol [Internet] 2005. [cited 2014 Mar 28]; 144:510-8. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=1576028&tool=pmcentrez&rendertype=abstract; PMID:15655529 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85. Hernández M, Knight GE, Wildman SSP, Burnstock G. Role of ATP and related purines in inhibitory neurotransmission to the pig urinary bladder neck. Br J Pharmacol [Internet] 2009. [cited 2014 Mar 28]; 157:1463-73. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=2765313&tool=pmcentrez&rendertype=abstract [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86. Cotton KD, Hollywood MA, McHale NG, Thornbury KD. Ca2+ current and Ca(2+)-activated chloride current in isolated smooth muscle cells of the sheep urethra. J Physiol [Internet] 1997. [cited 2014 Mar 26]; 505 (Pt 1:121-31. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=1160098&tool=pmcentrez&rendertype=abstract; PMID:9409476; http://dx.doi.org/ 10.1111/j.1469-7793.1997.121bc.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87. Bulley S, Jaggar JH. Cl(-) channels in smooth muscle cells. Pflugers Arch 2013; 466(5):861-72; http://www.ncbi.nlm.nih.gov/pubmed/24077695 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88. Sancho M, García-Pascual A, Triguero D. Presence of the Ca2+-activated chloride channel anoctamin 1 in the urethra and its role in excitatory neurotransmission. Am J Physiol Renal Physiol [Internet] 2012. [cited 2014 Mar 27]; 302:F390-400. Available from: http://www.ncbi.nlm.nih.gov/pubmed/22114201; PMID:22114201 [DOI] [PubMed] [Google Scholar]
- 89. Kyle BD, Ahrendt E, Braun AP, Braun JEA. The large conductance, calcium-activated K+ (BK) channel is regulated by cysteine string protein. Sci Rep [Internet] 2013. [cited 2014 Mar 3]; 3:2447. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3744087&tool=pmcentrez&rendertype=abstract; PMID:23945775; http://dx.doi.org/ 10.1038/srep02447 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90. Ahrendt E, Kyle B, Braun AP, Braun JEA. Cysteine String Protein Limits Expression of the Large Conductance, Calcium-Activated K(+) (BK) Channel. PLoS One [Internet] 2014; 9:e86586. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3903548&tool=pmcentrez&rendertype=abstract; PMID:24475152; http://dx.doi.org/ 10.1371/journal.pone.0086586 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91. Xin W, Li N, Cheng Q, Petkov G V. BK Channel-Mediated Relaxation of Urinary Bladder Smooth Muscle: A Novel Paradigm for Phosphodiesterase Type 4 Regulation of Bladder Function. J Pharmacol Exp Ther [Internet] 2014. [cited 2014 Mar 21]; 349:56-65. Available from: http://www.ncbi.nlm.nih.gov/pubmed/24459245; PMID:24459245; http://dx.doi.org/ 10.1124/jpet.113.210708 [DOI] [PMC free article] [PubMed] [Google Scholar]


