Abstract
Normal bone healing is a complex process that eventually restores original structure and function to the site of trauma. However, clinical circumstances such as nonunion, critical-sized defects, systemic bone disease, and fusion procedures have stimulated a search for ways to enhance this normal healing process. Biologics are an important part of this search and many, including bone marrow aspirate concentrate, demineralized bone matrix, platelet-rich plasma, bone morphogenic proteins, and platelet-derived growth factor, are currently in clinical use. Many others, including mesenchymal stem cells, parathyroid hormone, and Nel-like molecule-1 (NELL-1) will likely be in use in the future depending on the results of preclinical and clinical trials.
Keywords: Orthopedic surgery, Biologic, Adjuvant, Bone, Graft extender, Growth factor
Introduction
Unlike some musculoskeletal tissues, bones have the ability to regenerate after injury. Under ideal biomechanical conditions, which include proper alignment and stability of the fracture site, bone will heal and remodel into its original form [1]. However, circumstances exist in the field of orthopedic surgery in which this normal healing process is inadequate. Fracture nonunion is defined as a lack of complete healing after 6 months. Depending on the site and type of fracture, rates can vary between 4.8 and 10 % [2]. In addition, critical-sized bone defects caused by trauma or excision of neoplasms derive their very definition as those that will not heal without augmentation [3]. Surgical fusion procedures, commonly performed to join two or more vertebral bodies, involve the use of bone graft in order to remodel the targeted area into one piece of bone. One type of failure of this procedure, pseudoarthrosis, has been reported to range between 0 and 56 %, depending on many factors [4]. Finally, metabolic disorders such as osteoporosis and more rare genetic disorders like osteogenesis imperfecta disrupt normal bone physiology and can result in poor bone quality and healing [5].
Due to these conditions, many techniques and materials have been developed as a way of improving bony regeneration and healing. Of particular interest and relevance to this review is the use of biologic adjuvants to augment this process. There is no consensus for the definition of what a biologic therapy is, but it usually must be derived from a living system [6]. In other fields, this usually refers to molecules involved in signaling or generating a certain immune response. However, for the purpose of this review, cell-based therapies will be included as well. While it is tempting to include the field of gene therapy under the biologic umbrella, the authors determined that this was beyond the scope of this review.
Review criteria
Articles for this review were chosen from the authors’ personal libraries and from searches of PubMed using the search terms “bone, biologic adjuvant, biologics, orthopedic surgery” alone and in various combinations. Additional references were found by reviewing the reference lists of pertinent articles and previous reviews for relevant articles. All articles were English language, full-text papers. Searches were performed up to December 2014.
Normal bone healing
Typical fracture healing is commonly described in three phases: inflammation, repair, and remodeling [7] (Fig. 1). These phases proceed in order, but with considerable overlap. Initial disruption of the bone macrostructure leads to extravasation and hematoma formation at the site. Then, inflammatory cytokines and growth factors, including transforming growth factor-β (TGF-β), platelet-derived growth factor (PDGF), and bone morphogenetic proteins (BMPs), coordinate inflammatory cell and stem cell migration [8]. In the repair phase, a fibrous callus is formed by chondrocytes and fibroblasts. This is subsequently mineralized by osteoblasts under a similar growth factor milieu. Finally, osteoclasts resorb and remodel the mineralized hard callous to approximate the original structure and function of the underlying bone. Giannoudis et al. popularized the diamond concept of bone healing, in which osteogenic cells, osteoinductive scaffolds, growth factors, and mechanical forces interact to lead to ideal bone healing [9]. This review will focus on the use of cells and growth factors to augment normal bone healing in orthopedic surgery.
Fig. 1.
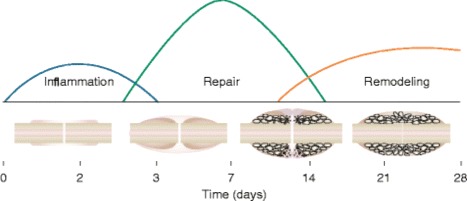
Graphical representation of the three overlapping phases of bone healing. Cytokines direct inflammatory and stem cell migration to the injured area. A fibrous callous is formed during the repair phase that is ultimately remodeled in the final phase to more closely resemble the original structure. Figure 5a from Claes L, Recknagel S, Ignatius A. Fracture healing under healthy and inflammatory conditions. Nat Rev Rheumatol. 2012;8(3):133–43
Biologic adjuvants
Platelet-derived growth factor
PDGF is responsible for various cellular responses that are critical to tissue repair in the musculoskeletal system. Some examples are chemotaxis, mesenchymal stem cell (MSC) recruitment, angiogenesis, and upregulation of vascular endothelial growth factor [10–12]. Importantly, PDGF acts as an initiator of the native bone-healing cascade at the implantation site [13]. Due to these effects, it was investigated as an adjuvant treatment of complex nonunion fractures. Recombinant human PDGF (rhPDGF) was combined with a tri-calcium phosphate (TCP) bone substitute scaffold prior to implantation. The rhPDGF saturated scaffold allowed for an initial burst release, followed by a slower release to mimic endogenous PDGF from platelets. Advantages of this type of treatment are limited systemic exposure, local delivery, and a continual dosage [14]. PDGF was further investigated in clinical trials involving hind foot and ankle fusion. A similar technique was used, combining rhPDGF with a TCP bone substitute. This resulting fusion was compared to autogenous bone grafts and was shown to be equally as effective in stimulating bone healing. This method spared the patient from any donor site morbidly or complications [14]. These studies have shown rhPDGF is a promising recent addition to the surgical treatments for bone repair. Further studies and trials are planned to optimize its therapeutic use in these applications as well as other orthopedic areas such as tendon-to-bone healing, intra-tendon, and articular cartilage regeneration [13].
Bone marrow aspirate concentrate
Initially described in 1952, bone marrow aspirate concentrate (BMAC) is a natural extension of the iliac crest bone graft (ICBG) that has been used for many years. Through aspiration of the marrow, a small percentage of mesenchymal stem cells (MSCs) can be obtained. In the past, these cells would have been expanded through culture in the lab, but the more recent impetus was to condense the harvest and implantation down to one surgical procedure. Currently, the standard is aspiration from the posterior iliac crest [15, 16], as this site is potentially psychologically less traumatic [16] and is easily accessible with the needle. Compared to harvesting from the anterior pelvis, there is no difference in the total number of cells obtained, but there are more connective tissue progenitors from a posterior aspiration [15]. After spinning the cells down, between 0.001 and 0.01 % of the cells obtained were found to be MSCs, with the total number of viable cells obtained decreasing with age [17]. As a result, the growth factors that are contained in the aspirate, PDGF, TGF-β, and VEGF are likely to be more important [18].
While often studied in conjunction with cartilage repair [19] and combined with a microfracture technique, BMAC had initially been studied in tibial shaft fractures because of the historically elevated nonunion rate. More recently, BMAC has been used successfully in osteochondritis dessicans (OCD) lesions of the talus, not only alone, but to assist in the ingrowth of allograft plugs [20–22]. The same principle has since been applied to OCD lesions in knees [23], and in addition, it has been used in many cases of nonunion in order to stimulate new bone growth [24]. However, while there are multiple level IV studies applying BMAC to use in aseptic nonunions, there is no good level 1 evidence [25].
While promising, use of BMAC is not always appropriate. In a study of aseptic nonunions, the authors found that the number of progenitor cells can vary widely and has a direct impact on the volume of formation of new bone [24]. Age and sex also play a role, as there were fewer colony forming units (CFUs) found as age increases [26]. In a separate study, BMAC was used in conjunction with TCP to fill benign cavitary lesions, and there was no reported benefit [27]. Overall, BMAC is an osteoinductive product that often must be combined with an osteoconductive material to produce effective bone formation. With high variability in samples between patients not only in aspiration volume [28] and technique [26], more research needs to be done to further define the indications of use.
Platelet-rich plasma
Platelet-rich plasma (PRP) is one of the newest innovations in orthopedic healing and repair, but most of the research has been focused on soft tissue rather than bone. While there is still debate as to whether the white blood cells (WBCs) included in the PRP preparations are beneficial or harmful to the healing response [29], PRP does contain very high concentrations of PDGF, VEGF, and FGF. Because of this, it is thought that PRP may be able to assist in many situations that require tissue healing [30].
Currently, more than 40 commercial PRP concentrations systems are available, and there is high variability in the concentrations of growth factors present, even within a given technique [31–33]. Initially thought to assist in bone healing, the results have not been promising in spinal fusion or high tibial osteotomy trials [34, 35]. However, it has been shown in a small study that the speed at which distraction osteogenesis occurs is increased with the addition of PRP, but that has not been consistent with the outcome of larger trials. PRP has been shown to decrease pain after patellar tendon autograft ACL reconstruction and rotator cuff repair, but in neither of those cases has it been shown to significantly change the overall clinical outcome [36, 37]. Currently, the American Academy of Orthopedic Surgeons (AAOS) clinical practice guidelines say that the organization is “unable to recommend for or against” the use of PRP [38] in general, but when examined solely in the setting of bone healing, PRP is not an effective adjuvant or substitute for bone graft [39]. There are simply too many confounding factors such as preparation, type of tissue being stimulated, and type of repair for the authors to recommend the use of PRP specifically to augment bone healing, though future research might show benefits.
Demineralized bone matrix
The biologic adjuvant demineralized bone matrix (DBM) is created by demineralizing ground cadaver bone and it contains BMPs, collagen, and other growth factors that are then combined with a carrier. Much like PRP, each process of DBM creation from the harvesting to the final product is unique from company to company, with no standardization across the industry [40]. Overall, most of the research has been done in spinal fusions, utilizing it as a bone graft extender to be combined with existing bone. It has not been shown to produce efficient or durable fusion when used alone. More recently, the use has been expanding, with good results seen in ankle fusions when compared with ICBG [41] and some success with femoral nonunions or segmental defects.
The best use seems to be that it provides not only osteoinductive proteins but also physical structure that can help position any allograft used in conjunction [42]. This has been seen with the treatment of unicameral bone cysts (UBCs) where the residual void is filled with graft. Often, ICBG alone is not an option because of the limit on how much can be harvested, but when combined with DBM, a small amount can fill the entire defect. In humeral shaft nonunions, it has been successfully used to produce solid fusion of the fragments [43, 44] but compared to ICBG, there was no difference found in clinical function or healing time. Overall, DBM has been shown to produce solid fusions in multiple bony applications and allows the surgeon to avoid harvesting ICBG in some situations, which reduces donor site morbidity. However, it is worth noting that in one spine model, almost half of the products tested had a 0 % fusion rate, speaking to the vast differences in preparation and delivery that each of the unique companines provide [45].
Bone morphogenic proteins
Over the last few years, the use of BMPs has increased dramatically as they are developed and marketed. BMPs are a collection of many different proteins in the body, but only BMP-2 and BMP-7 were approved for human use. However, production of BMP-7 was stopped in May 2014 and is currently unavailable. BMP-2 is only FDA-approved for certain indications, one being anterior lumbar interbody fusion (ALIF) with a titanium or polyetheretherketone (PEEK) cage. Multiple studies have shown that the addition of BMP-2 produces good fusion rates in the lumbar spine [46, 47], and surgeons around the country have started using it off-label for multiple indications. The other major indication where BMP has shown benefit is in the treatment of open tibial shaft fractures. These can be notoriously difficult to heal and have a very high rate of nonunion secondary to multiple factors including the energy of the trauma and possible infection. However, in the BMP-2 Evaluation in Surgery for Tibial Trauma (BESTT) trial [48], the authors found accelerated fracture healing and more importantly, a decreased rate of secondary intervention such as need for reoperation when BMP was included in the bone graft at the site. This has led to the FDA approving BMP-2 for the treatment of open tibial fractures after intramedullary nailing.
The biggest concern of using BMP is safety. The initial trials were controversial, as some thought they were too closely associated with industry and that complications were underreported. As a result, the study and use of BMP has a stigma attached to it [49]. Indeed, complications can be serious, including seroma formation, severe soft tissue swelling, radiculitis, and retrograde ejaculation. However, it has been shown to be a potent former of new bone in multiple animal models [50], and further research should be done to determine accurate dosing and delivery systems to minimize the side effect profile and expand its safe use. It is currently unknown what percentage of surgeons use BMP, and what types of surgery they are using it for, though as stated, the highest percentage is likely to be in spinal fusion.
Future directions
Clinical trials
The authors searched ClinicalTrials.gov to determine recent trends in the use of biologics for bone in orthopedic surgery. The terms “fracture repair,” “bone fusion,” “critical defect bone,” and “orthopedic surgery and bone and biologic” were searched and the resulting trials reviewed. Trials that did not involve biologic treatment, did not involve orthopedic surgery, or had statuses that were unknown or terminated were excluded. Fifty-nine trials met the inclusion criteria and the number of trials involving biologics is shown in Fig. 2. In addition to giving a general overview of the state of investigation of biologics in orthopedic surgery, the authors noted two interesting trends that deserve discussion.
Fig. 2.
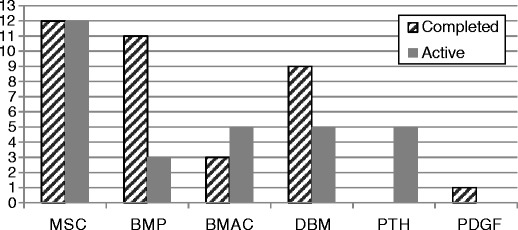
ClinicalTrials.gov was searched for relevant trials using biologic adjuvants in orthopedic surgery for bone. The number of trials investigating each category of biologic is displayed above. MSC mesenchymal stem cell, BMP bone morphogenetic protein, BMAC bone marrow aspirate concentrate, DBM demineralized bone matrix, PTH parathyroid hormone, PDGF platelet-derived growth factor
Mesenchymal stem cells
The first trend is the heavy investigation of mesenchymal stem cells. MSCs are found in many tissues in the human body, including blood, adipose tissue, skin, trabecular bone, liver, and lungs [51]. Briefly described above, stem cell migration and differentiation is thought to be an integral part of normal bone healing [52]. In contrast to the autologous MSCs contained in perioperatively harvested BMAC, many of the current MSC trials involve the use of isolated and cultured allogenic cells. Allogenic MSCs have been shown to avoid immune detection by lacking major histocompatibility complex type II (MHC II) and costimulatory molecules as well as modulating the surrounding inflammatory environment [53]. A study of rabbit fractures has shown that allogenic MSCs performed just as well as autologous MSCs in repairing bone, with no increase in immunogenicity [54]. Although the view of the future clinical utility of these cells is not uniformly optimistic [55], the successful outcomes of these trials have the potential to significantly change the field of orthopedic surgery.
Parathyroid hormone
Another interesting trend identified in the trial search is the use of teraperatide, which is recombinant parathyroid hormone (PTH). Physiologically, PTH acts on osteoblasts and osteoclasts to resorb bone, increasing serum calcium. However, when given in a pulsatile fashion during treatment of osteoporosis, it increases bone mineral density (BMD) and improves bone architechture [56]. This likely occurs due to preferential osteoblast differentiation over osteoclast bone resorption [57]. Limited clinical studies of off-label use have shown positive effects on bone repair [58]. Therefore, the results of these clinical trials involving bone repair and fusion have the potential to add another treatment option in these difficult cases.
Nel-like molecule-1
Protein kinase C-binding protein Nel-like molecule-1 (NELL-1) is a growth factor originally identified in patients with craniosyntosis [59]. Since its discovery, animal studies involving overexpression as well as deficiency have confirmed its importance in bone formation [60–63]. Investigators have attempted to utilize the osteoinductive effects of NELL-1 to enhance bone repair in experimental animal models. Rat and mouse explant studies demonstrated its ability to induce and accelerate bone growth [64, 65]. NELL-1 has also been investigated in many in vivo models, including a rat critical-sized femoral defect model and rat and sheep spine models, showing positive effects on bone growth and repair [66–68]. The exact actions of NELL-1 in the signaling cascade are still being defined, but it is thought to act through Runx2 [62, 69, 70] and interact with BMPs and the sonic hedgehog (SHH) signaling pathway [71–74]. This protein has demonstrated enormous potential in pre-clinical studies and further progress towards a clinical use is anticipated.
Coating implants with biologics
In addition to fracture repair and fusion procedures, biologic materials are being investigated as a coating material for orthopedic implants. Implants are an important part of orthopedic surgery but are susceptible to several drawbacks that accompany placing a foreign material in the body such as nonintegration, inflammation and immune response, and infection. The rationale for coating implants is to reduce these risks. In both oral and orthopedic implants, animal models show that ceramic coatings improve osteointegration. Biologic coatings such as extracellular matrix peptides, collagen, BMPs, and other growth factors have been investigated but are still in the early phases of pre-clinical study [75, 76]. Continued research study is needed to develop biologic implant coatings for routine human use.
Conclusion
The field of orthopedic surgery currently uses several biologic adjuvants in the treatment of a vast array of bone pathologies. Research is actively seeking ways to refine and optimize currently used treatments while simultaneously investigating promising new molecules. These advances combined with breakthroughs in other fields such as materials science, tissue engineering, and surgical technique ensure the continued dynamic evolution and progress of the field as a whole.
Compliance with Ethics Guidelines
Conflict of Interest
Benjamin Smith, Todd Goldstein, and Charles Ekstein declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Footnotes
This article is part of the Topical Collection on Biological Adjuvants in Orthopedic Surgery
References
- 1.Claes L, Recknagel S, Ignatius A. Fracture healing under healthy and inflammatory conditions. Nat Rev Rheumatol. 2012;8(3):133–43. doi: 10.1038/nrrheum.2012.1. [DOI] [PubMed] [Google Scholar]
- 2.Bennett MH, Stanford RE, Turner R. Hyperbaric oxygen therapy for promoting fracture healing and treating fracture non-union. Cochrane Database Syst Rev. 2012;11 doi: 10.1002/14651858.CD004712.pub4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Spicer PP, Kretlow JD, Young S, Jansen JA, Kasper FK, Mikos AG. Evaluation of bone regeneration using the rat critical size calvarial defect. Nat Protoc. 2012;7(10):1918–29. doi: 10.1038/nprot.2012.113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Gruskay JA, Webb ML, Grauer JN. Methods of evaluating lumbar and cervical fusion. Spine J. 2014;14(3):531–9. doi: 10.1016/j.spinee.2013.07.459. [DOI] [PubMed] [Google Scholar]
- 5.Siddiqui NA, Owen JM. Clinical advances in bone regeneration. Curr Stem Cell Res Ther. 2013;8(3):192–200. doi: 10.2174/1574888X11308030003. [DOI] [PubMed] [Google Scholar]
- 6.Morrow T, Felcone LH. Defining the difference: what makes biologics unique. Biotechnol Healthc. 2004;1(4):24–9. [PMC free article] [PubMed] [Google Scholar]
- 7.Kalfas IH. Principles of bone healing. Neurosurg Focus. 2001;10(4):E1. doi: 10.3171/foc.2001.10.4.2. [DOI] [PubMed] [Google Scholar]
- 8.Schindeler A, Mcdonald MM, Bokko P, Little DG. Bone remodeling during fracture repair: the cellular picture. Semin Cell Dev Biol. 2008;19(5):459–66. doi: 10.1016/j.semcdb.2008.07.004. [DOI] [PubMed] [Google Scholar]
- 9.Giannoudis PV, Einhorn TA, Marsh D. Fracture healing: the diamond concept. Injury. 2007;38(Suppl 4):S3–6. doi: 10.1016/S0020-1383(08)70003-2. [DOI] [PubMed] [Google Scholar]
- 10.Hollinger JO, Hart CE, Hirsh SN, Lynch S, Friedlander GE. Recombinant human platelet-derived growth factor: biology and clinical applications. J Bone Joint Surg Am. 2008; Suppl 1:48–54. [DOI] [PubMed]
- 11.Caplan AL, Correa D. PDGF in bone formation and regeneration: new insights into a novel mechanism involving MSCs. J Orthop Res. 2011;29(12):1795–803. doi: 10.1002/jor.21462. [DOI] [PubMed] [Google Scholar]
- 12.Heldin CH, Westermark B. Mechanism of action and in vivo role of platelet-derived growth factor. Physiol Rev. 1999;79(4):1283–316. doi: 10.1152/physrev.1999.79.4.1283. [DOI] [PubMed] [Google Scholar]
- 13.DiGiovanni CW, Petricek JM. The evolution of rhPDGF-BB in musculoskeletal repair and its role in foot and ankle fusion surgery. Foot Ankle Clin N Am. 2010;15:621–40. doi: 10.1016/j.fcl.2010.07.001. [DOI] [PubMed] [Google Scholar]
- 14.Young CS, Ladd PA, Browning CF, et al. Release, biological potency, and biochemical integrity of recombinant human platelet-derived growth factor-BB (rhPDGF-BB) combined with Augment(TM) Bone Graft or GEM 21S beta-tricalcium phosphate (beta-TCP) J Control Release. 2009;140(3):250–5. doi: 10.1016/j.jconrel.2009.06.030. [DOI] [PubMed] [Google Scholar]
- 15.Pierini M, Di Bella C, Dozza B, et al. The posterior iliac crest outperforms the anterior iliac crest when obtaining mesenchymal stem cells from bone marrow. J Bone Joint Surg Am. 2013;95(12):1101–7. doi: 10.2106/JBJS.L.00429. [DOI] [PubMed] [Google Scholar]
- 16.Bierman HR. Bone marrow aspiration the posterior iliac crest, an additional safe site. Calif Med. 1952;77(2):138–9. [PMC free article] [PubMed] [Google Scholar]
- 17.Pittenger MF, Mackay AM, Beck SC, et al. Multilineage potential of adult human mesenchymal stem cells. Science. 1999;284(5411):143–7. doi: 10.1126/science.284.5411.143. [DOI] [PubMed] [Google Scholar]
- 18.McCarrel T, Fortier L. Temporal growth factor release from platelet-rich plasma, trehalose lyophilized platelets, and bone marrow aspirate and their effect on tendon and ligament gene expression. J Orthop Res. 2009;27:1033–42. doi: 10.1002/jor.20853. [DOI] [PubMed] [Google Scholar]
- 19.Fortier LA, Potter HG, Rickey EJ, et al. Concentrated bone marrow aspirate improves full-thickness cartilage repair compared with microfracture in the equine model. J Bone Joint Surg Am. 2010;92(10):1927–37. doi: 10.2106/JBJS.I.01284. [DOI] [PubMed] [Google Scholar]
- 20.Murawski CD, Duke GL, Deyer TW, Kennedy JG. Bone marrow aspirate concentrate (BMAC) as a biological adjunct to osteochondral lesions of the talus. Tech Foot Ankle Surg. 2011;10(1):18–27. doi: 10.1097/BTF.0b013e31820b4c2f. [DOI] [Google Scholar]
- 21.Kennedy JG, Murawski CD. The treatment of osteochondral lesions of the talus with autologous osteochondral transplantation and bone marrow aspirate concentrate: surgical technique. Cartilage. 2011;2:327–36. doi: 10.1177/1947603511400726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Giannini S, Buda R, Vannini F, Cavallo M, Grigolo B. One-step bone marrow-derived cell transplantation in talar osteochondral lesions. Clin Orthop Relat Res. 2009;467(12):3307–20. doi: 10.1007/s11999-009-0885-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Buda R, Vannini F, Cavallo M, Grigolo B, Cenacchi A, Giannini S. Osteochondral lesions of the knee: a new one-step repair technique with bone-marrow-derived cells. J Bone Joint Surg Am. 2010;92(Suppl 2):2–11. doi: 10.2106/JBJS.J.00813. [DOI] [PubMed] [Google Scholar]
- 24.Hernigou P, Poignard A, Beaujean F, Rouard H. Percutaneous autologous bone-marrow grafting for nonunions. Influence of the number and concentration of progenitor cells. J Bone Joint Surg Am. 2005;87:1430–7. doi: 10.2106/JBJS.D.02215. [DOI] [PubMed] [Google Scholar]
- 25.WG D l, Einhorn TA, Koval K, et al. Bone grafts and bone graft substitutes in orthopaedic trauma surgery. A critical analysis. J Bone Joint Surg Am. 2007;89(3):649–58. doi: 10.2106/JBJS.F.00465. [DOI] [PubMed] [Google Scholar]
- 26.Payne KA, Didiano DM, Chu CR. Donor sex and age influence the chondrogenic potential of human femoral bone marrow stem cells. Osteoarthritis Cartilage. 2010;18(5):705–13. doi: 10.1016/j.joca.2010.01.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Damron TA, Lisle J, Craig T, Wade M, Silbert W, Cohen H. Ultraporous β-tricalcium phosphate alone or combined with bone marrow aspirate for benign cavitary lesions: comparison in a prospective randomized clinical trial. J Bone Joint Surg Am. 2013;95(2):158–66. doi: 10.2106/JBJS.K.00181. [DOI] [PubMed] [Google Scholar]
- 28.Muschler GF, Boehm C, Easley K. Aspiration to obtain osteoblast progenitor cells from human bone marrow: the influence of aspiration volume. J Bone Joint Surg Am. 1997;79(11):1699–709. doi: 10.2106/00004623-199711000-00012. [DOI] [PubMed] [Google Scholar]
- 29.DeLong JM, Russell RP, Mazzocca AD. Platelet-rich plasma: the PAW classification system. Arthroscopy. 2012;28(7):998–1009. doi: 10.1016/j.arthro.2012.04.148. [DOI] [PubMed] [Google Scholar]
- 30.Hall MP, Band PA, Meislin RJ, Jazrawi LM, Cardone DA. Platelet-rich plasma: current concepts and application in sports medicine. J Am Acad Orthop Surg. 2009;17(10):602–8. doi: 10.5435/00124635-200910000-00002. [DOI] [PubMed] [Google Scholar]
- 31.Castillo TN, Pouliot MA, Kim HJ, Dragoo JL. Comparison of growth factor and platelet concentration from commercial platelet-rich plasma separation systems. Am J Sports Med. 2011;39(2):266–71. doi: 10.1177/0363546510387517. [DOI] [PubMed] [Google Scholar]
- 32.Mazzucco L, Balbo V, Cattana E, Guaschino R, Borzini P. Not every PRPgel is born equal: evaluation of growth factor availability for tissues through four PRP-gel preparations. Fibrinet, RegenPRP-Kit, Plateltex and one manual procedure. Vox Sang. 2009;97(2):110–8. doi: 10.1111/j.1423-0410.2009.01188.x. [DOI] [PubMed] [Google Scholar]
- 33.Mazzocca AD, Mccarthy MB, Chowaniec DM, et al. Platelet-rich plasma differs according to preparation method and human variability. J Bone Joint Surg Am. 2012;94(4):308–16. doi: 10.2106/JBJS.K.00430. [DOI] [PubMed] [Google Scholar]
- 34.Carreon LY, Glassman SD, Anekstein Y, Puno RM. Platelet gel (AGF) fails to increase fusion rates in instrumented posterolateral fusions. Spine (Phila Pa 1976) 2005;30(9):E243–6. doi: 10.1097/01.brs.0000160846.85397.44. [DOI] [PubMed] [Google Scholar]
- 35.Dallari D, Savarino L, Stagni C, et al. Enhanced tibial osteotomy healing with use of bone grafts supplemented with platelet gel or platelet gel and bone marrow stromal cells. J Bone Joint Surg Am. 2007;89(11):2413–20. doi: 10.2106/JBJS.F.01026. [DOI] [PubMed] [Google Scholar]
- 36.Nakamura N. Platelet-rich plasma added to the patellar tendon harvest site during anterior cruciate ligament reconstruction enhanced healing. J Bone Joint Surg Am. 2013;95(10):942. doi: 10.2106/JBJS.9510.ebo450. [DOI] [PubMed] [Google Scholar]
- 37.Mall NA, Tanaka MJ, Choi LS, Paletta GA. Factors affecting rotator cuff healing. J Bone Joint Surg Am. 2014;96(9):778–88. doi: 10.2106/JBJS.M.00583. [DOI] [PubMed] [Google Scholar]
- 38.American Academy of Orthopaedic Surgeons: treatment of osteoarthritis of the knee: evidence-based guideline, 2nd Edition. Available at: http://www.aaos.org/research/guidelines/TreatmentofOsteoarthritisoftheKneeGuideline.pdf. Accessed 15 Dec 2014.
- 39.Hsu WK, Mishra A, Rodeo SR, et al. Platelet-rich plasma in orthopaedic applications: evidence-based recommendations for treatment. J Am Acad Orthop Surg. 2013;21(12):739–48. doi: 10.5435/JAAOS-21-12-739. [DOI] [PubMed] [Google Scholar]
- 40.Peterson B, Whang PG, Iglesias R, Wang JC, Lieberman JR. Osteoinductivity of commercially available demineralized bone matrix: preparations in a spine fusion model. J Bone Joint Surg Am. 2004;86(10):2243–50. doi: 10.2106/00004623-200410000-00016. [DOI] [PubMed] [Google Scholar]
- 41.Michelson JD, Curl LA. Use of demineralized bone matrix in hindfoot arthrodesis. Clin Orthop Relat Res. 1996;325:203–8. doi: 10.1097/00003086-199604000-00024. [DOI] [PubMed] [Google Scholar]
- 42.Muschler GF, Matsukura Y, Nitto H, et al. Selective retention of bone marrowderived cells to enhance spinal fusion. Clin Orthop Relat Res. 2005;432:242–51. doi: 10.1097/01.blo.0000149812.32857.8b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Hierholzer C, Sama D, Toro JB, Peterson M, Helfet DL. Plate fixation of ununited humeral shaft fractures: effect of type of bone graft on healing. J Bone Joint Surg Am. 2006;88(7):1442–7. doi: 10.2106/JBJS.E.00332. [DOI] [PubMed] [Google Scholar]
- 44.Taylor NL, Crow SA, Heyworth BE, Rosenwasser MP. Treatment of humeral nonunions with allograft, demineralized bone matrix, and plate fixation. Osteosynthesis Trauma Care. 2005;13(2):105–12. doi: 10.1055/s-2005-836534. [DOI] [Google Scholar]
- 45.Bae H, Zhao L, Zhu D, Kanim LE, Wang JC, Delamarter RB. Variability across ten production lots of a single demineralized bone matrix product. J Bone Joint Surg Am. 2010;92(2):427–35. doi: 10.2106/JBJS.H.01400. [DOI] [PubMed] [Google Scholar]
- 46.Burkus JK, Gornet MF, Dickman CA, Zdeblick TA. Anterior lumbar interbody fusion using rhBMP-2 with tapered interbody cages. J Spinal Disord Tech. 2002;15(5):337–49. doi: 10.1097/00024720-200210000-00001. [DOI] [PubMed] [Google Scholar]
- 47.Burkus JK, Transfeldt EE, Kitchel SH, Watkins RG, Balderston RA. Clinical and radiographic outcomes of anterior lumbar interbody fusion using recombinant human bone morphogenetic protein-2. Spine (Phila Pa 1976) 2002;27(21):2396–408. doi: 10.1097/00007632-200211010-00015. [DOI] [PubMed] [Google Scholar]
- 48.Govender S, Csimma C, Genant HK, et al. Recombinant human bone morphogenetic protein-2 for treatment of open tibial fractures: A prospective, controlled, randomized study of four hundred and fifty patients. J Bone Joint Surg Am. 2002;84:2123–34. doi: 10.1302/0301-620X.84B5.12795. [DOI] [PubMed] [Google Scholar]
- 49.Carragee EJ, Hurwitz EL, Weiner BK. A critical review of recombinant human bone morphogenetic protein-2 trials in spinal surgery: Emerging safety concerns and lessons learned. Spine J. 2011;11(6):471–91. doi: 10.1016/j.spinee.2011.04.023. [DOI] [PubMed] [Google Scholar]
- 50.Wozney JM, Rosen V. Bone morphogenetic protein and bone morphogenetic protein gene family in bone formation and repair. Clin Orthop Relat Res. 1998;346:26–37. doi: 10.1097/00003086-199801000-00006. [DOI] [PubMed] [Google Scholar]
- 51.Undale AH, Westendorf JJ, Yaszemski MJ, Khosla S. Mesenchymal stem cells for bone repair and metabolic bone diseases. Mayo Clin Proc. 2009;84(10):893–902. doi: 10.4065/84.10.893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Jones E, Yang X. Mesenchymal stem cells and bone regeneration: current status. Injury. 2011;42(6):562–8. doi: 10.1016/j.injury.2011.03.030. [DOI] [PubMed] [Google Scholar]
- 53.Ryan JM, Barry FP, Murphy JM, Mahon BP. Mesenchymal stem cells avoid allogeneic rejection. J Inflamm (Lond). 2005;2(8). [DOI] [PMC free article] [PubMed]
- 54.Udehiya RK, Amarpal, Aithal HP, et al. Comparison of autogenic and allogenic bone marrow derived mesenchymal stem cells for repair of segmental bone defects in rabbits. Res Vet Sci. 2013;94(3):743–52. doi: 10.1016/j.rvsc.2013.01.011. [DOI] [PubMed] [Google Scholar]
- 55.Charron D, Suberbielle-boissel C, Tamouza R, Al-daccak R. Anti-HLA antibodies in regenerative medicine stem cell therapy. Hum Immunol. 2012;73(12):1287–94. doi: 10.1016/j.humimm.2012.06.010. [DOI] [PubMed] [Google Scholar]
- 56.Tella SH, Gallagher JC. Biological agents in management of osteoporosis. Eur J Clin Pharmacol. 2014;70(11):1291–301. doi: 10.1007/s00228-014-1735-5. [DOI] [PubMed] [Google Scholar]
- 57.Kaback LA, Soung do Y, Naik A, et al. Teriparatide (1–34 human PTH) regulation of osterix during fracture repair. J Cell Biochem. 2008;105(1):219–26. doi: 10.1002/jcb.21816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Mancilla EE, Brodsky JL, Mehta S, Pignolo RJ, Levine MA. Teriparatide as a systemic treatment for lower extremity non-union fractures: a case series. Endocr Pract. 2014;1–22. [DOI] [PubMed]
- 59.Ting K, Vastardis H, Mulliken JB, et al. Human NELL-1 expressed in unilateral coronal synostosis. J Bone Miner Res. 1999;14(1):80–9. doi: 10.1359/jbmr.1999.14.1.80. [DOI] [PubMed] [Google Scholar]
- 60.Zhang X, Kuroda S, Carpenter D, et al. Craniosynostosis in transgenic mice overexpressing Nell-1. J Clin Invest. 2002;110(6):861–70. doi: 10.1172/JCI15375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Zhang X, Cowan CM, Jiang X, et al. Nell-1 induces acrania-like cranioskeletal deformities during mouse embryonic development. Lab Invest. 2006;86(7):633–44. doi: 10.1038/labinvest.3700430. [DOI] [PubMed] [Google Scholar]
- 62.Zhang X, Ting K, Bessette CM, et al. Nell-1, a key functional mediator of Runx2, partially rescues calvarial defects in Runx2(+/−) mice. J Bone Miner Res. 2011;26(4):777–91. doi: 10.1002/jbmr.267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Zhang X, Ting K, Pathmanathan D, et al. Calvarial cleidocraniodysplasia-like defects with ENU-induced Nell-1 deficiency. J Craniofac Surg. 2012;23(1):61–6. doi: 10.1097/SCS.0b013e318240c8c4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Cowan CM, Cheng S, Ting K, et al. Nell-1 induced bone formation within the distracted intermaxillary suture. Bone. 2006;38(1):48–58. doi: 10.1016/j.bone.2005.06.023. [DOI] [PubMed] [Google Scholar]
- 65.Aghaloo T, Cowan CM, Chou YF, et al. Nell-1-induced bone regeneration in calvarial defects. Am J Pathol. 2006;169(3):903–15. doi: 10.2353/ajpath.2006.051210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Lu SS, Zhang X, Soo C, et al. The osteoinductive properties of Nell-1 in a rat spinal fusion model. Spine J. 2007;7(1):50–60. doi: 10.1016/j.spinee.2006.04.020. [DOI] [PubMed] [Google Scholar]
- 67.Li W, Zara JN, Siu RK, et al. Nell-1 enhances bone regeneration in a rat critical-sized femoral segmental defect model. Plast Reconstr Surg. 2011;127(2):580–7. doi: 10.1097/PRS.0b013e3181fed5ae. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Siu RK, Lu SS, Li W, et al. Nell-1 protein promotes bone formation in a sheep spinal fusion model. Tissue Eng Part A. 2011;17(7–8):1123–35. doi: 10.1089/ten.tea.2010.0486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Bokui N, Otani T, Igarashi K, et al. Involvement of MAPK signaling molecules and Runx2 in the NELL1-induced osteoblastic differentiation. FEBS Lett. 2008;582(2):365–71. doi: 10.1016/j.febslet.2007.12.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Truong T, Zhang X, Pathmanathan D, Soo C, Ting K. Craniosynostosis-associated gene nell-1 is regulated by runx2. J Bone Miner Res. 2007;22(1):7–18. doi: 10.1359/jbmr.061012. [DOI] [PubMed] [Google Scholar]
- 71.Aghaloo T, Cowan CM, Zhang X, et al. The effect of NELL1 and bone morphogenetic protein-2 on calvarial bone regeneration. J Oral Maxillofac Surg. 2010;68(2):300–8. doi: 10.1016/j.joms.2009.03.066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Zhu S, Song D, Jiang X, Zhou H, Hu J. Combined effects of recombinant human BMP-2 and Nell-1 on bone regeneration in rapid distraction osteogenesis of rabbit tibia. Injury. 2011;42(12):1467–73. doi: 10.1016/j.injury.2011.05.040. [DOI] [PubMed] [Google Scholar]
- 73.Xia L, Xu Y, Chang Q, et al. Maxillary sinus floor elevation using BMP-2 and Nell-1 gene-modified bone marrow stromal cells and TCP in rabbits. Calcif Tissue Int. 2011;89(1):53–64. doi: 10.1007/s00223-011-9493-1. [DOI] [PubMed] [Google Scholar]
- 74.James AW, Pang S, Askarinam A, et al. Additive effects of sonic hedgehog and Nell-1 signaling in osteogenic versus adipogenic differentiation of human adipose-derived stromal cells. Stem Cells Dev. 2012;21(12):2170–8. doi: 10.1089/scd.2011.0461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Goodman SB, Yao Z, Keeney M, Yang F. The future of biologic coatings for orthopaedic implants. Biomaterials. 2013;34(13):3174–83. doi: 10.1016/j.biomaterials.2013.01.074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Junker R, Dimakis A, Thoneick M, Jansen JA. Effects of implant surface coatings and composition on bone integration: a systematic review. Clin Oral Implants Res. 2009;20(Suppl 4):185–206. doi: 10.1111/j.1600-0501.2009.01777.x. [DOI] [PubMed] [Google Scholar]


