Abstract
The aim of this study was to characterize the ultrastructural effects caused by β-lactam antibiotics in Klebsiella pneumoniae isolates. Three K. pneumoniae clinical isolates were selected for the study with resistance profiles for third-generation cephalosporins, aztreonam, and/or imipenem and with different resistance genes for extended-spectrum β-lactamases (ESBL) or Klebsiella pneumoniae carbapenemase (KPC). Two K. pneumoniae isolates obtained from the microbiota, which were both resistant to amoxicillin and ampicillin, were also analyzed. In accordance with the susceptibility profile, the clinical isolates were subjected to subminimum inhibitory concentrations (sub-MICs) of cefotaxime, ceftazidime, aztreonam, and imipenem and the isolates from the microbiota to ampicillin and amoxicillin, for analysis by means of scanning and transmission electron microscopy. The K. pneumoniae isolates showed different morphological and ultrastructural changes after subjection to β-lactams tested at different concentrations, such as cell filamentation, loss of cytoplasmic material, and deformation of dividing septa. Our results demonstrate that K. pneumoniae isolates harboring different genes that encode for β-lactamases show cell alterations when subjected to different β-lactam antibiotics, thus suggesting that they possess residual activity in vitro, despite the phenotypic resistance presented in the isolates analyzed.
1. Introduction
Klebsiella pneumoniae is one of the most common Gram-negative multidrug-resistant (MDR) organisms found worldwide, and it is responsible for high morbidity and mortality both in hospitals and in other healthcare units [1].
β-Lactam antibiotics are the most commonly prescribed antibacterial agents because of the high efficiency of their mechanism of action and low toxicity [2]. Penicillin-binding proteins are the lethal targets of β-lactams in sensitive bacteria and their inhibition leads to degradation of the bacterial cell wall and eventual cell lysis [3]. Several studies have reported different bacterial morphological changes caused by subminimum inhibitory concentrations (sub-MICs) of third-generation cephalosporins and carbapenems in sensitive isolates of several bacterial species [4–6]. However, there is a lack of studies using resistant isolates.
In pathogenic bacteria, β-lactamase production is the most common and important mechanism of resistance to β-lactam antibiotics [2]. The classical β-lactamases are enzymes capable of inactivating penicillins and narrow-spectrum cephalosporins before reaching their target [7]. Most K. pneumoniae isolates carry the gene for chromosomal class A SHV-1 β-lactamase. This has limited activity against ampicillin and does not hydrolyze extended-spectrum β-lactam antibiotics [8]. The presence of this gene has been previously studied in K. pneumoniae isolates originating from the microbiota of individuals without bacterial infection [9].
The introduction into clinical practice of oxyiminocephalosporins for treating infections due to Gram-negative bacteria was soon followed by the appearance of extended-spectrum β-lactamases (ESBLs) derived from the classical β-lactamases SHV-1, TEM-1, and TEM-2. These conferred resistance to third-generation cephalosporins and monobactams such as aztreonam [2]. More than 180 variants of the gene bla SHV and more than 210 of the gene bla TEM have already been reported [10]. Another group of ESBLs, which has a high profile of hydrolysis in relation to cefotaxime and is less than 40% identical to the SHV and TEM enzymes, is the group of CTX-M enzymes. These are widely found in different species of the family Enterobacteriaceae, including K. pneumoniae [11].
Treatment of infections due to K. pneumoniae isolates producing ESBLs is limited to the use of a few available antibiotics. Carbapenems are often the last line of effective treatment against these infections [12]. However, production of the class A carbapenemase Klebsiella pneumoniae carbapenemase (KPC), which is an extended-spectrum β-lactamase, has been reported to be the primary form of carbapenem resistance in this bacterial species. Fifteen variants with dissemination in different parts of the world have been reported [10].
Production of these β-lactamases by clinical isolates of K. pneumoniae has been a major problem in many countries, and this has hampered the therapeutic options available for the treatment of bacterial infections caused by this species.
The increasing prevalence of multidrug resistance is leading towards the threat that a postantibiotic era may be about to begin, characterized by decreased effectiveness of common antibiotics and routine application of complementary therapeutic approaches for treating bacterial infections [13]. Therefore, new studies need to be developed, in which old and discarded antibiotics should be reinvestigated and reused. Even rejected antibiotics possibly should be used when necessary [14].
In this regard, some studies have tried to demonstrate the in vitro and in vivo action of therapeutic combinations of β-lactams and aminoglycosides in MDR K. pneumoniae isolates, aiming to obtain new options for treating infections due to this bacterial species [15]. However, no study has demonstrated any alterations induced by different β-lactam antibiotics in the bacterial cell structure of MDR K. pneumoniae isolates.
K. pneumoniae isolates presenting resistance to cefotaxime when subjected to sub-MICs of this antibiotic were shown to cause damage to cell surfaces. Filamentation through a dose-dependent adaptive process was observed, caused by the stressful environment that was induced by the presence of the antibiotic, thereby contributing towards the therapeutic effects [16].
Unfortunately, knowledge about the effects caused by β-lactam antibiotics on the bacterial cell structure of MDR K. pneumoniae isolates is still scarce. These isolates carry important resistance genes, such as bla SHV, bla TEM, bla CTX-M, and bla KPC. Likewise, little is known about the effect of these antibiotics on isolates from the microbiota of individuals without bacterial infection. Therefore, the present study aimed to characterize the morphological and ultrastructural effects on pathogenic and nonpathogenic K. pneumoniae isolates that carry genes encoding the classical β-lactamases ESBL and KPC, caused by β-lactam antibiotics that are widely used in clinical medicine.
2. Material and Methods
2.1. Bacterial Isolates
For this study, five K. pneumoniae isolates from Recife, PE, Brazil, were selected (Tables 1 and 2). The K21.1-F and K58.1-F isolates were obtained from the fecal microbiota of children who did not have bacterial infection. These isolates showed resistance or intermediate resistance to ampicillin and/or amoxicillin but lacked the bla SHV gene (Table 1) [9]. Three other K. pneumoniae clinical isolates obtained from public hospitals in Recife were used: K3C, K16R, and K652. The susceptibility profile and presence of the genes bla SHV and bla CTX-M had been determined in previous studies on the K3C and K16R isolates [9, 17], which showed resistance to cefotaxime, ceftazidime, and aztreonam, according to the interpretive criteria of the Clinical and Laboratory Standards Institute (CLSI) of 2013 [18].
Table 1.
MICs of K. pneumoniae isolates obtained in hospital and sub-MICs used for analysis by electron microscopy.
| Isolates | Cefotaxime | Ceftazidime | Aztreonam | ||||||
|---|---|---|---|---|---|---|---|---|---|
| (μg mL−1) | (μg mL−1) | (μg mL−1) | |||||||
| ESBL | MICsa | Sub-MICsb | MICsa | Sub-MICsb | MICsa | Sub-MICsb | |||
| TEMc | SEMd | TEMc | SEMd | TEMc | SEMd | ||||
| K3C | >256 | 64; 32 | 32 | 128 | 32; 16 | 32 | 64 | 32; 16 | 32 |
| K16R | >256 | 64; 32 | 32 | 32 | 16; 8 | 16 | 16 | 4 | 4 |
|
| |||||||||
| Isolates | Cefotaxime | Ceftazidime | Imipenem | ||||||
| (μg mL−1) | (μg mL−1) | (μg mL−1) | |||||||
| KPC | MICsa | Sub-MICsb | MICsa | Sub-MICsb | MICsa | Sub-MICsb | |||
| TEMc | SEMd | TEMc | SEMd | TEMc | SEMd | ||||
|
| |||||||||
| K652 | ≥256 | 98 | 98 | ≥256 | 27 | 27 | ≥128 | 16 | 16 |
aMICs: minimum inhibitory concentrations; bsub-MICs: sub-minimum inhibitory concentrations; cTEM: transmission electron microscopy; and dSEM: scanning electron microscopy.
Table 2.
MICs of K. pneumoniae isolates obtained of microbiota and sub-MICs used for analysis by electron microscopy.
| Isolates | Ampicillin (μg mL−1) | Amoxicillin (μg mL−1) | ||||
|---|---|---|---|---|---|---|
| MICsa | Sub-MICsb | MICsa | Sub-MICsb | |||
| TEMc | SEMd | TEMc | SEMd | |||
| K58.1-F | 16 | 0.5; 2; 8 | 0.5; 8 | 32 | 0.5; 4; 16 | 0.5; 16 |
| K21.1-F | 128 | 0.5; 16; 32; 64 | 16; 32; 64 | >128 | 0.5; 4; 16; 64; 128 | 0.5; 16; 128 |
aMICs: minimum inhibitory concentrations; bsub-MICs: sub-minimum inhibitory concentrations; cTEM: transmission electron microscopy; and dSEM: scanning electron microscopy.
The K3C isolate possesses the bla SHV-11 gene and the K16R isolate has the bla SHV-1 and bla CTX-M2 genes [9, 17]. The K652 isolate, which was studied for the first time in the present work, was obtained by means of surveillance testing on rectal swabs from individuals without clinical symptoms of bacterial infection who were admitted to a public hospital in 2011. This isolate was identified and its susceptibility profile in relation to different antimicrobials was determined using the VITEK 2 automated system (BioMérieux). It was manually confirmed in relation to cefotaxime, ceftazidime, and imipenem (Sigma-Aldrich) by means of broth macrodilution testing, in accordance with the 2013 CLSI recommendations [18].
2.2. DNA and PCR Extraction
The genomic DNA of K. pneumoniae isolates was extracted using the Wizard Genomic DNA Purification Kit (Promega), in accordance with the manufacturer's instructions. The presence of the bla TEM gene was investigated by means of PCR in the macrobiota isolates and in the three clinical isolates using the T1 primer 5′-ATAAAATTCTTGAAGACGAAA-3′ and the T2 primer 5′-GACAGTTACCAATGCTTAATC-3′ [19]. The K652 isolate was also evaluated by means of PCR for the presence of the bla SHV and bla KPC genes using the primers 5′-GGTTATGCGTTATATTCGCC-3′ and 5′-TTAGCGTTGCCAGTGCTC-3 [20] and using KPC1F: 5′-GCTACACCTAGCTCCACCTTC-3′ and KPC1R: 5′-ACAGTGGTTGGTAATCCATGC-3′ [21], respectively.
2.3. DNA Sequencing
Sequencing of the PCR products from the bla TEM and bla KPC genes was performed using the Applied Biosystems/Hitachi automated sequencer, after purification of the PCR amplicons using the PureLink Micro Kit (Invitrogen), in accordance with the manufacturer's instructions. The bla TEM gene was sequenced in the K3C and K16R isolates because of phenotypic resistance that was presented in relation to the third-generation cephalosporins that were tested. Only the bla KPC gene was sequenced in K652 isolate, due to phenotypic resistance to carbapenems that was presented. The primers were the same as for PCR, plus TEMup21: 5′-TCCCTTTTTTGCGGCATTTTGC-3′ and TEMdwn280: 5′-CAGTGAGGCACCTATCTC-3′ [22], for the bla TEM gene, and KPC F: 5′-GAGCTGAACTCCGCCATC-3′ and KPC R: 5′-TATTTTTCCGAGATGGGTGAC-3′ [23], for the bla KPC gene.
The analyses on the DNA sequence and the multiple alignments were performed using the DNAstar software and BLAST [24]. The sequences for the bla TEM-1, bla TEM-15, and bla KPC-2 genes were deposited in the GenBank database under the access numbers KF906436, KF906435, and KF906437, respectively.
2.4. Transmission Electron Microscopy (TEM)
To analyze the action of antibiotics on K. pneumoniae isolates, these isolates were subjected to different sub-MICs of ampicillin, amoxicillin, ceftazidime, cefotaxime, aztreonam, and imipenem (Sigma-Aldrich) at 37°C for 6 hours, according to the origin and susceptibility profile of each isolate, using clinically relevant concentrations (Tables 1 and 2) [25, 26]. In all the processes, a control for the isolate was included under the same conditions and at the same dilutions, without the presence of the antibiotic. After growth, the bacterial cells were centrifuged and fixed using 2.5% glutaraldehyde and 4% paraformaldehyde (Sigma-Aldrich). The cells were postfixed in 1% osmium tetroxide and then contrasted in 5% uranyl acetate (Electron Microscopy Science). Dehydration was carried out using acetone (Sigma-Aldrich) followed by infiltration and embedment of the material in epon 812 resin (Electron Microscopy Science). The samples were viewed under a transmission electron microscope (Zeiss EM109).
2.5. Scanning Electron Microscopy (SEM)
The K. pneumoniae isolates were subjected to antimicrobial agents using the concentrations and criteria described above (Tables 1 and 2). After growth, the bacterial cells were gently centrifuged and fixed using 2.5% glutaraldehyde (Sigma-Aldrich). Postfixation was performed using 1% osmium tetroxide (Electron Microscopy Science) followed by dehydration using ethanol (Sigma-Aldrich). After dehydration, the material was dried in preparation for metallization and viewing of the bacterial cells under a scanning electron microscope (JEOL JSM-5600 LV).
3. Results
3.1. Antimicrobial Susceptibility
The susceptibility profiles of the K3C, K16R, K21.1-F, and K58.1-F isolates had previously been determined in other studies [9, 17] and are described in Tables 1 and 2. The K. pneumoniae K652 isolate showed resistance to all the antibiotics tested using the VITEK 2 automated system (ampicillin, ampicillin/sulbactam, aztreonam, cephalosporins, cefepime, cefotaxime, cefoxitin, ceftazidime, ciprofloxacin, ertapenem, gentamicin, imipenem, meropenem, and piperacillin/tazobactam), except for amikacin and tigecycline. The macrodilution broth test confirmed the resistance of this isolate to cefotaxime, ceftazidime, and imipenem, with MICs of 256 μg mL−1.
3.2. Presence of bla SHV, bla TEM, and bla KPC Genes
The PCR analyses identified the presence of bla TEM gene in the three clinical isolates of K. pneumoniae and in the K21.1-F isolate from the microbiota. Additionally, the bla SHV and bla KPC genes were also identified by means of PCR in the K. pneumoniae isolate K652.
3.3. Sequencing of the bla TEM and bla KPC Genes
The bla TEM-1 gene was identified in the K. pneumoniae isolate K16R, which also had the bla CTX-M2 gene [17]. The bla TEM gene found in the K3C isolate showed a nucleotide change in relation to the bla TEM-1 gene at positions 512 G→A and 914 G→A, leading to replacement of Glu-104→Lys and Gly-238→Ser. The bla KPC-2 gene was identified in the K. pneumoniae isolate K652.
3.4. Ultrastructural and Morphological Analyses
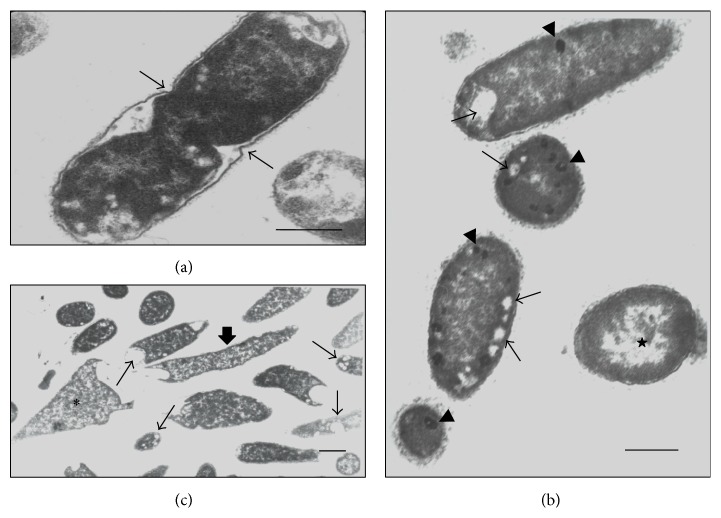
The cells of the control K. pneumoniae isolates, which were analyzed without having been subjected to β-lactam antibiotics, showed morphology that had been preserved, with an intact cell wall and cytoplasmic contents of electron-dense appearance on analysis by means of TEM. The presence of ribosomes and genetic material distributed in the bacterial cytoplasm could be seen. The cells undergoing a division process showed formation of septa without irregularities (Figures 1(a), 2(a), and 3(a)). The SEM analysis showed conserved rod-shaped bacterial morphology and an average cell length of 1.5 to 3 μm (Figures 4(a), 4(b), 5(a), and 6(a)).
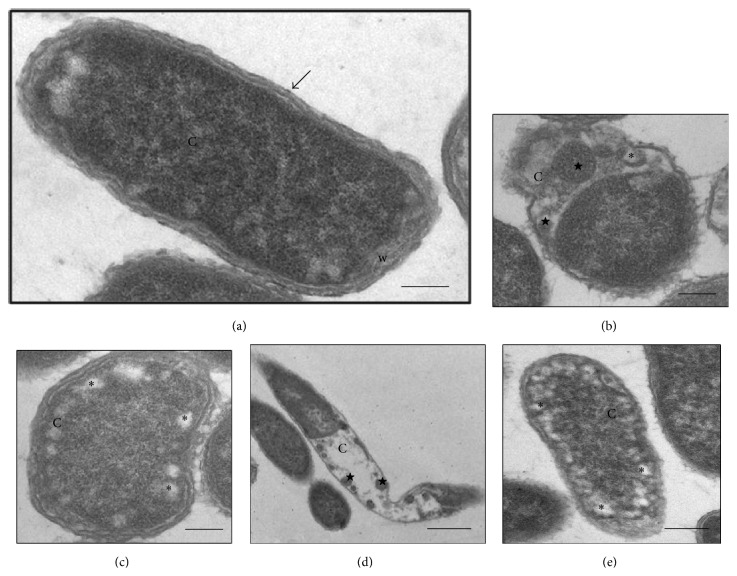
Figure 1.
(a–e) Transmission electron micrographs of isolate K3C from K. pneumoniae. (a) bacterial cell without treatment (control)—preserved morphology, cytoplasmic membrane (arrow), cell wall (w), and cytoplasm (C) intact. (b-c) Cells subjected to CTX (64 μg mL−1)—decreased cytoplasmic material (C), presence of membrane compartments (stars), electron-lucent spaces (asterisks) and disorganization of the membrane and cell wall. (d) Cell subjected to CAZ (32 μg mL−1)—elongated aspect morphology with loss of cytoplasmic material (C) and the presence of small membrane compartments (stars). (e) Cell subjected to ATM (32 μg mL−1)—normal morphology in the presence of electron-lucent spaces (asterisks) in the cytoplasm (C). Bars = 0.5 μm. CAZ: ceftazidime; CTX: cefotaxime; and ATM: aztreonam.
Figure 2.
(a–h) Transmission electron micrographs of isolate K16R from K. pneumoniae. (a) Untreated bacterial cell—preserved morphology, cytoplasmic membrane (arrow), cell wall (w), and cytoplasm (C) intact. (b) Cell subjected to CTX (64 μg mL−1)—presence of large electron-lucent space due to increased periplasmic space (stars) and reduced cytoplasmic material (C). (c–e) Cells subjected to CAZ (16 μg mL−1). (c-d) Filamentous cells and the presence of membrane compartments containing cytoplasmic material (star). (e) Elongated cell showing loss of cytoplasmic material (star). (f–h) Cell subjected to ATM (4 μg mL−1)—loss of cytoplasmic material (C) and presence of membrane compartments (stars). Bars = 0.5 μm. CAZ: ceftazidime; CTX: cefotaxime; and ATM: aztreonam.
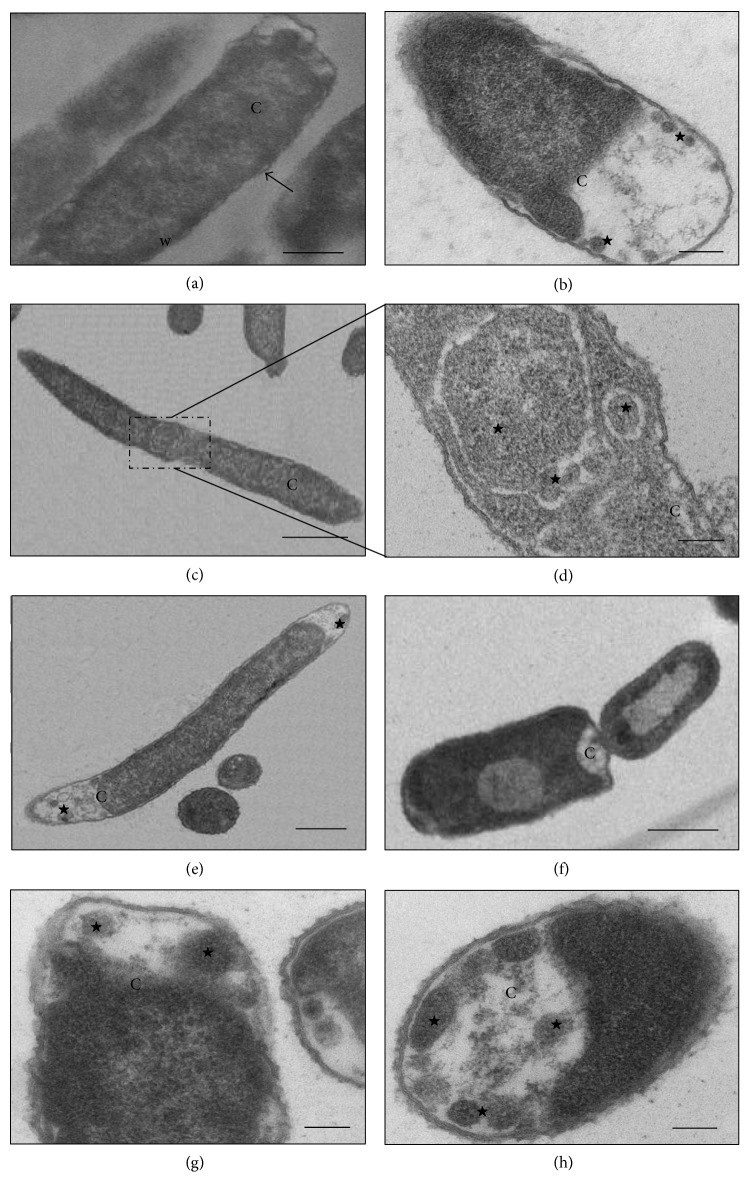
Figure 3.
(a–h) Transmission electron micrographs of isolate K652 of K. pneumoniae. (a) Untreated bacterial cell—cell wall with preserved morphology (w) and cytoplasm (C) intact, besides regular septation (arrowheads). (b-c) Cell subjected to CTX (98 μg mL−1)—presence of cells not grown and rounded (stars) at the ends of cells with normal morphology. (d–f) Cells subjected to CAZ (27 μg mL−1)—consecutive formation of septa (arrowheads) providing elongation of bacterial morphology. Observe disorganization of membrane and cell wall with elastic aspect. (g-h) Cell subjected to IMP (16 μg mL−1)—morphology of elongated appearance with formation of consecutive septa (arrowheads). Bars = 0.5 μm. CAZ: ceftazidime; CTX: cefotaxime; and IMP: imipenem.
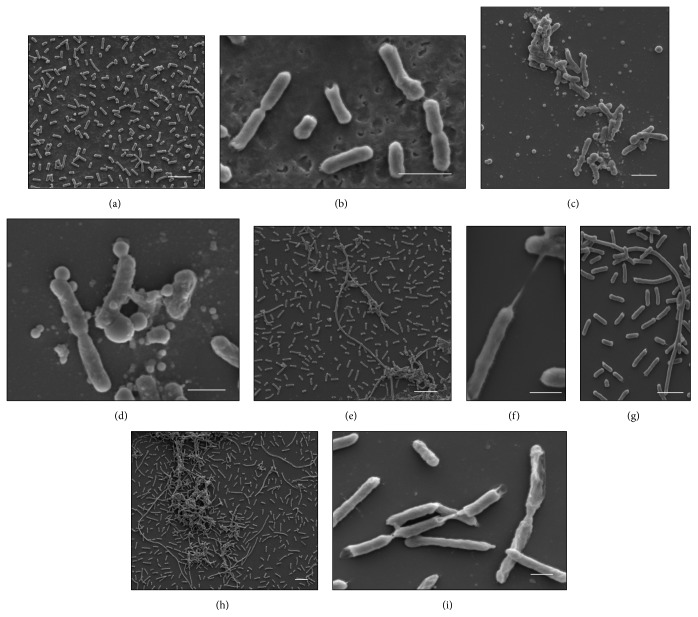
Figure 4.
(a–i) Scanning electron micrographs of isolate K652. (a-b) Control cells with preserved morphology. (c-d) Cells subjected to CTX (98 μg mL−1). Observe spherical cells that are not grown being formed from cells with preserved morphology. (e–g) Cells subjected to CAZ (27 μg mL−1). Observe elongated cells forming bacterial filaments. (h-i) Cells subjected to IMP (16 μg mL−1). Observe elongated cells due to unfinished successive divisions. Bars = 2 μm. CAZ: ceftazidime; CTX: cefotaxime; and IMP: imipenem.
Figure 5.
(a–d) Scanning electron micrographs of isolate K16R. (a) Control cells with preserved morphology. (b) Cells subjected to CTX (32 μg mL−1). (c) Cells subjected to CAZ (16 μg mL−1). (d) Cells subjected to ATM (4 μg mL−1). Bars = 2 μm. CAZ: ceftazidime; CTX: cefotaxime; and ATM: aztreonam.
Figure 6.
(a–d) Scanning electron micrographs of isolate K3C. (a) Control cells with preserved morphology. (b) Cells subjected to CTX (32 μg mL−1). (c) Cells subjected to CAZ (32 μg mL−1). (d) Cells subjected to ATM (32 μg mL−1). Bars = 2 μm. CAZ: ceftazidime; CTX: cefotaxime; and ATM: aztreonam.
3.5. K16R Isolate
Although the K16R isolate presented resistance to cefotaxime, ceftazidime, and aztreonam and had the bla SHV-1, bla TEM-1, and bla CTX-M2 genes, subjection of this isolate to sub-MICs of these antimicrobials caused cellular events that differed from those of the control cells. After subjection to cefotaxime at concentrations of 32 and 64 μg mL−1, cells with increased periplasmic space over their entire extent with greater amounts of space at their ends were observed, thus suggesting that the cytoplasm had decreased in volume or retracted (Figure 2(b)). No significant morphological changes to cell size were observed in the SEM analysis (Figure 5(b)).
Subjection of the K16R isolate to ceftazidime at concentrations of 8 and 16 μg mL−1 allowed identification of morphological and cellular organizational changes by means of TEM and SEM, in which several elongated cells were viewed. Through TEM, it could be seen that this event occurred because of unfinished cell division, and this was identified through the presence of several consecutive division septa that were incomplete. Some cells also showed membrane cell spaces inside the bacterial cytoplasm and cytoplasmic content identical to the rest of the cell, with ribosomes and genetic material equally distributed (Figures 2(c)–2(e)). This suggests that cell division did not happen, thus enabling formation of membrane compartments within the bacterial cells. Structural disorganization of the continuity of the cytoplasmic membrane of some cells and decreased amounts of bacterial cytoplasm could also be observed. Through SEM, it was possible to identify cells that were more than 15 μm in length (Figure 5(c)).
After the K16R isolate was subjected to application of 4 μg mL−1 of aztreonam, some cells presented large electron-lucent spaces in the bacterial cytoplasm with decreased cytoplasmic mass (Figures 2(f)–2(h)). No morphological or cell size changes were identified through SEM (Figure 5(d)).
3.6. K3C Isolate
The K. pneumoniae isolate K3C also has phenotypic resistance to cefotaxime, ceftazidime, and aztreonam and has the bla SHV-11 and bla TEM-15 genes. After this isolate was subjected to 32 and 64 μg mL−1 of cefotaxime, some cells showed a decrease in cytoplasmic material with formation of membrane compartments within the cells, which presented internal material containing ribosomes. These cells showed cytoplasmic membrane disorganization. Several cells also contained in their cytoplasm a large number of nonmembrane electron-lucent structures, near the cell membrane, at both tested concentrations (Figures 1(b) and 1(c)). No morphological change was identified through SEM (Figure 6(b)).
Various filamentous cells were seen through TEM and SEM after the K3C isolate was subjected to 16 and 32 μg mL−1 of ceftazidime. Through TEM, it could be seen that cell elongation was followed by a decrease in cytoplasmic material in the middle of the dividing cell or at the ends of cells that had already separated. The presence of a large electron-lucent space between the cytoplasmic membrane and cell wall of these cells highlighted the presence of cell compartments separated by membranes that contained cytoplasmic material (Figure 1(d)). Through SEM, cells of up to 18 μm in length could be seen (Figure 6(c)).
The cellular events induced by aztreonam in this isolate, at concentrations of 16 and 32 μg mL−1, were discreet. Some cells showed large numbers of nonmembrane electron-lucent spaces in the bacterial cytoplasm, similar to those found after treatment with cefotaxime (Figure 1(e)). However, most of the cells showed conservation of morphology with preservation of the cytoplasmic membrane and cell wall. No morphological changes were observed through SEM (Figure 6(d)).
3.7. K652 Isolate
The K652 isolate of K. pneumoniae has the bla KPC-2 gene and shows high phenotypic resistance to carbapenems and third-generation cephalosporins. Subjection of this isolate to both ceftazidime at 27 μg mL−1 and imipenem at 16 μg mL−1 caused cell filamentation, as seen in analyses using SEM. Cells of more than 50 mm in length could be seen (Figures 4(e)–4(i)). The number of elongated cells seemed to be greater after subjection to imipenem than to ceftazidime (Figures 3(d)–3(h)). The TEM analysis on the K652 isolate subjected to these antibiotics showed that most of the cells showed formation of consecutive division septa, without completing the division process. Many cells also showed elongation and disorganization of the cytoplasmic membrane and cell wall at the site of cell division, thus providing a resilient appearance to these cellular components.
Through SEM, various rod-shaped bacilli that gave rise to nondeveloped cells in the form of cocci could be observed after the K652 isolate was subjected to 98 μg mL−1 of cefotaxime (Figures 4(c) and 4(d)). The TEM confirmed that some spherical cells were being formed from rod-shaped cells through changes to the density of the bacterial cytoplasm (Figures 3(b) and 3(c)).
3.8. K58.1-F and K21.1-F Isolates
The results from the TEM and SEM analyses on the K. pneumoniae isolates from the microbiota, after subjection to amoxicillin and ampicillin, were quite similar to those from the isolates obtained from the hospital environment when the latter were subjected to cefotaxime and aztreonam.
The K58.1-F isolate showed phenotypic resistance to amoxicillin and intermediate resistance to ampicillin, when exposed to sub-MICs of these antibiotics (Table 2). This was demonstrated by the enlarged electron-lucent spaces at the ends of the cells, in comparison with the control, as seen through TEM, thus suggesting that there was a decrease in the cytoplasmic content. Large quantities of electron-lucent spaces and nonmembrane electron-dense structures were also identified in the bacterial cytoplasm of several cells (Figures 7(b) and 7(c)). These cellular events became more pronounced with higher concentrations of each drug, thus demonstrating that there was a dose-dependent correlation.
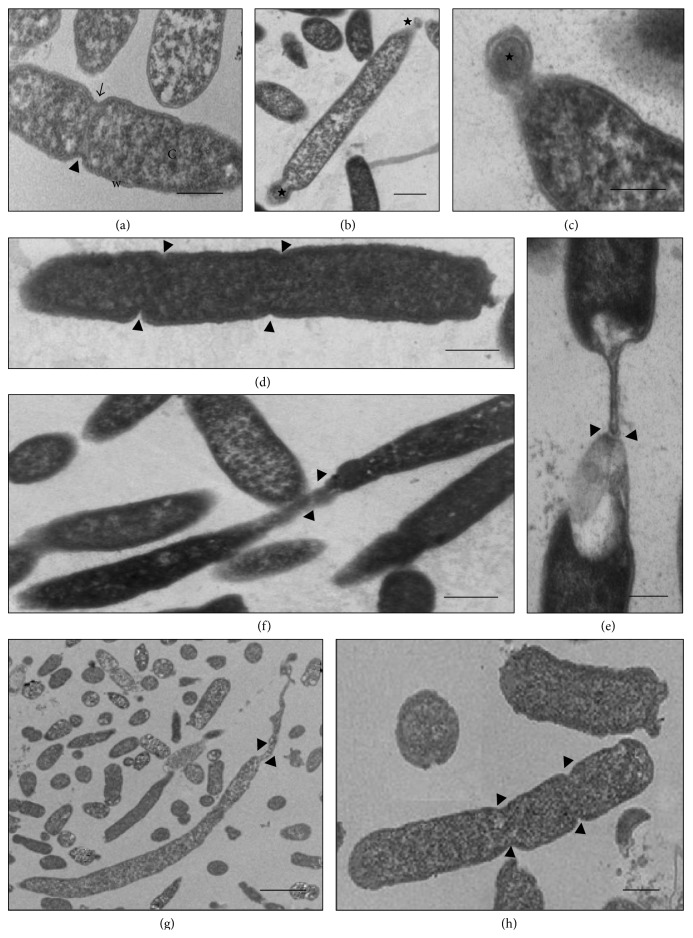
Figure 7.
(a–c) Transmission electron micrographs of isolate K21.1-F and K58.1-F of K. pneumoniae. (a) Isolate K21.1-F submitted to 0.5 μg mL−1 ampicillin. Observe irregular formation of septa in a dividing cell (arrow). (b) Isolate K58.1-F subjected to 0.5 μg mL−1 of ampicillin. Cells present electron-lucent (fine arrow), and electron-dense spaces with different sizes and shape throughout the bacterial cytoplasm (arrowheads), in addition to reduction of cytoplasmic contents (star). (c) Isolate K58.1-F subjected to 4 μg mL−1 of amoxicillin. The cells exhibit altered morphologies with cell elongation (large arrow) and undefined forms (asterisk), with the presence of electron-lucent structures with shape and varying sizes scattered throughout the cell cytoplasm (fine arrow). Bars = 0.5 μm.
Although the K21.1-F isolate showed higher MICs for amoxicillin and ampicillin than that of the K58.1-F isolate, the TEM analysis after the isolates were subjected to sub-MICs of these antibiotics showed that the results from the two isolates were similar. Large quantities of electron-lucent spaces and nonmembrane electron-dense structures were identified in the bacterial cytoplasm of several cells of the K21.1-F isolate, even at the lowest concentration tested (0.5 μg mL−1) (Figure 7(a)). Several cells with irregular formation of septa during bacterial division, but without loss of cytoplasmic content even at higher concentrations, were simultaneously observed (Figure 7). The SEM analysis did not show any significant changes to any of the antibiotic concentrations tested, in relation to the K21.1-F and K58.1-F isolates.
4. Discussion
Production of β-lactamases is one of the main mechanisms of resistance among isolates of the Enterobacteriaceae family. ESBL and KPC in particular are often found in K. pneumoniae clinical isolates, thus conferring a phenotype that is resistant to the main β-lactams used in clinical medicine.
The bla TEM-15, bla CTX-M2, and bla KPC-2 genes, found, respectively, in the K3C, K16R, and K652 isolates, encode enzymes of ESBL and carbapenemase type. These genes have previously been reported in different bacterial species [27–29]. The changes of Glu→Lys at position 104 and Gly→Ser at position 238 of the bla TEM-15 gene are known to favor increased substrate fixation and an enzyme-active site, thus giving rise to an enzyme with increased affinity and catalytic efficiency for oxyiminocephalosporins [29]. ESBL CTX-M belonging to the CTX-M2 phylogenetic group is one of the CTX-M-type enzymes most found in Enterobacteriaceae isolates in Brazil [28], with high hydrolysis power compared with third-generation cephalosporins, especially cefotaxime.
KPC β-lactamases efficiently hydrolyze penicillins, cephalosporins, carbapenems, and aztreonam and are inhibited by clavulanic acid and tazobactam [28]. The K652 isolate presents the resistance gene bla KPC-2. This gene was first detected in Brazil in K. pneumoniae isolates obtained from patients hospitalized in Recife, PE [27]. Since then, other studies have demonstrated the presence of this gene in K. pneumoniae isolates obtained from several other states in Brazil [27, 28].
Cases of infections by isolates producing these enzymes give rise to considerable concern regarding the most appropriate antibiotic therapy. This is because bacterial growth often does not change, and cell death due to subjection to β-lactams does not occur. In this context, the present study was designed to demonstrate the in vitro effect of imipenem, penicillin, third-generation cephalosporins and aztreonam on MDR K. pneumoniae, and microbiota isolates obtained from individuals under different clinical conditions. This variability made it possible to analyze the effects of β-lactam antibiotics in isolates with distinct genetic and phenotypic resistance profiles, which would have the capacity to evolve differently in their host organisms.
Subjection of the K3C, K16R, and K652 isolates to different concentrations of ceftazidime, and of the K652 isolate to imipenem, caused cell filamentation in all the isolates analyzed, with formation of several consecutive septa and disorganization of the cell membrane and cell wall in several cells. This induction of cell filamentation was previously reported in other studies using sensitive and resistant isolates from different bacterial species, including Serratia marcescens, K. pneumoniae, and P. aeruginosa subjected to third-generation cephalosporins, carbapenems, and monobactams. These studies showed that inactivation of different penicillin-binding proteins may be associated with inability to complete the cell division process after replication of the bacterial mass [4, 5, 16, 30].
In our study, the presence of bacterial filaments and cell disorganization caused by K. pneumoniae isolates after subjection to ceftazidime and imipenem makes us believe that these antibiotics have some residual activity, despite the presence of genes that code for ESBL and KPC. However, even though the β-lactam antibiotics tested here were capable of causing changes to the K. pneumoniae isolates used in this study, these antibiotics were clearly incapable of interfering with the survival of isolates with a resistant phenotype.
Because of the great need for effective therapeutic options for treating MDR bacterial isolates, new therapeutic combinations have been tested. Hirsch et al. conducted a study that showed that meropenem and amikacin in combination were able to inhibit in vitro and in vivo growth of resistant K. pneumoniae isolates that produced KPC-2 and KPC-3 [15]. In the present study, it was found that imipenem and ceftazidime alone were able to promote in vitro cellular changes that included loss of cytoplasmic material, filamentation, and disorganization of the cell membrane and cell wall at the division site. Understanding the effect of antibiotics alone on MDR K. pneumoniae isolates is extremely important because this enables understanding of how the development of therapeutic combinations can effectively contribute towards treating the infections caused by these isolates. In our study, resistant K. pneumoniae isolates subjected to clinically relevant concentrations β-lactam antibiotics seemed to have responses similar to those of sensitive cells at low concentrations of these antibiotics.
Deloney and Schiller [31] used Helicobacter pylori-sensitive isolates subjected to sub-MICs of β-lactam antibiotics, including ampicillin and aztreonam, and showed that the bacterial cells became filamentous when subjected to aztreonam and spherical when subjected to other β-lactams. Our data showed that aztreonam was unable to induce cell filamentation in the clinical isolates tested. However, the appearance of electron-lucent spaces suggested that loss or retraction of cytoplasmic contents occurred, in the same way that occurred when microbial isolates were subjected to ampicillin and amoxicillin.
Our results demonstrate that K. pneumoniae isolates harboring different genes that encode for β-lactamases can undergo ultrastructural changes when subjected to sub-MICs of β-lactam drugs, thus suggesting that this antimicrobials have residual activity in vitro, despite the phenotypic resistance presented in the isolates analyzed.
Acknowledgments
This study is financially supported by Programa Estratégico de Apoio à Pesquisa em Saúde-PAPES IV (Process no. 407826/2012-2), Fundação Oswaldo Cruz (FIOCRUZ). The authors thank the Programa de Desenvolvimento Tecnológico em Insumos para Saúde (PDTIS) of Fundação Oswaldo Cruz (FIOCRUZ) and the Núcleo de Plataformas Tecnológicas of Centro de Pesquisas Aggeu Magalhães (CpqAM)/FIOCRUZ, especially MSc. Viviane Carvalho and Dr. Cássia Docena for the technical assistance.
Conflict of Interests
The authors declare that there is no conflict of interests regarding the publication of this paper.
References
- 1.Mammina C., Bonura C., Aleo A., et al. Sequence type 101 (ST101) as the predominant carbapenem-non-susceptible Klebsiella pneumoniae clone in an acute general hospital in Italy. International Journal of Antimicrobial Agents. 2012;39(6):543–545. doi: 10.1016/j.ijantimicag.2012.02.012. [DOI] [PubMed] [Google Scholar]
- 2.Hæggman S., Löfdahl S., Paauw A., Verhoef J., Brisse S. Diversity and evolution of the class A chromosomal beta-lactamase gene in Klebsiella pneumoniae . Antimicrobial Agents and Chemotherapy. 2004;48(7):2400–2408. doi: 10.1128/aac.48.7.2400-2408.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Stefanova M. E., Tomberg J., Olesky M., Höltje J.-V., Gutheil W. G., Nicholas R. A. Neisseria gonorrhoeae penicillin-binding protein 3 exhibits exceptionally highy carboxypeptidase and β-lactam binding activities. Biochemistry. 2003;42(49):14614–14625. doi: 10.1021/bi0350607. [DOI] [PubMed] [Google Scholar]
- 4.Buijs J., Dofferhoff A. S. M., Mouton J. W., Wagenvoort J. H., van der Meer J. Concentration-dependency of β-lactam-induced filament formation in Gram-negative bacteria. Clinical Microbiology and Infection. 2008;14(4):344–349. doi: 10.1111/j.1469-0691.2007.01940.x. [DOI] [PubMed] [Google Scholar]
- 5.Horii T., Kobayashi M., Sato K., Ichiyama S., Ohta M. An in-vitro study of carbapenem-induced morphological changes and endotoxin release in clinical isolates of Gram-negative bacilli. Journal of Antimicrobial Chemotherapy. 1998;41(4):435–442. doi: 10.1093/jac/41.4.435. [DOI] [PubMed] [Google Scholar]
- 6.Jackson J. J., Kropp H. β-lactam antibiotic-induced release of free endotoxin: in vitro comparison of penicillin-binding protein (PBP) 2-specific imipenem and PBP 3-specific ceftazidime. Journal of Infectious Diseases. 1992;165(6):1033–1041. doi: 10.1093/infdis/165.6.1033. [DOI] [PubMed] [Google Scholar]
- 7.Robin F., Delmas J., Chanal C., Sirot D., Sirot J., Bonnet R. TEM-109 (CMT-5), a natural complex mutant of TEM-1 β-lactamase combining the amino acid substitutions of TEM-6 and TEM-33 (IRT-5) Antimicrobial Agents and Chemotherapy. 2005;49(11):4443–4447. doi: 10.1128/aac.49.11.4443-4447.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Nelson E. C., Segal H., Elisha B. G. Outer membrane protein alterations and bla TEM-1 variants: their role in β-lactam resistance in Klebsiella pneumoniae . Journal of Antimicrobial Chemotherapy. 2003;52(6):899–903. doi: 10.1093/jac/dkg486. [DOI] [PubMed] [Google Scholar]
- 9.Veras D. L., Alves L. C., Brayner F. A., et al. Prevalence of the blaSHV gene in Klebsiella pneumoniae Isolates obtained from hospital and community infections and from the microbiota of healthy individuals in Recife, Brazil. Current Microbiology. 2011;62(5):1610–1616. doi: 10.1007/s00284-011-9899-z. [DOI] [PubMed] [Google Scholar]
- 10.Jacoby G., Bush K. β-Lactamase Classification and Amino Acid Sequences for TEM, SHV and OXA Extended-Spectrum and Inhibitor Resistant Enzymes. 2013, http://www.lahey.org/studies.
- 11.Saladin M., Cao V. T. B., Lambert T., et al. Diversity of CTX-M β-lactamases and their promoter regions from Enterobacteriaceae isolated in three Parisian hospitals. FEMS Microbiology Letters. 2002;209(2):161–168. doi: 10.1016/s0378-1097(02)00484-6. [DOI] [PubMed] [Google Scholar]
- 12.Lascols C., Peirano G., Hackel M., Laupland K. B., Pitout J. D. D. Surveillance and molecular epidemiology of Klebsiella pneumoniae isolates that produce carbapenemases: first report of OXA-48-like enzymes in North America. Antimicrobial Agents and Chemotherapy. 2013;57(1):130–136. doi: 10.1128/aac.01686-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Cress B. F., Englaender J. A., He W., Kasper D., Linhardt R. J., Koffas M. A. G. Masquerading microbial pathogens: capsular polysaccharides mimic host-tissue molecules. FEMS Microbiology Reviews. 2014;38(4):660–697. doi: 10.1111/1574-6976.12056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Bush K., Courvalin P., Dantas G., et al. Tackling antibiotic resistance. Nature Reviews Microbiology. 2011;9(12):894–896. doi: 10.1038/nrmicro2693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hirsch E. B., Guo B., Chang K.-T., et al. Assessment of antimicrobial combinations for Klebsiella pneumoniae carbapenemase-producing K. pneumoniae . Journal of Infectious Diseases. 2013;207(5):786–793. doi: 10.1093/infdis/jis766. [DOI] [PubMed] [Google Scholar]
- 16.Rajeshwari H., Nagveni S., Oli A., Parashar D., Chandrakanth K. R. Morphological changes of Klebsiella pneumoniae in response to Cefotaxime: a scanning electron microscope study. World Journal of Microbiology and Biotechnology. 2009;25(12):2263–2266. doi: 10.1007/s11274-009-0126-z. [DOI] [Google Scholar]
- 17.Lopes A. C. S., Veras D. L., Lima A. M. S., Melo R. D. C. A., Ayala J. blaCTX-M-2 and blaCTX-M-28 extended-spectrum β-lactamase genes and class 1 integrons in clinical isolates of Klebsiella pneumoniae from Brazil. Memorias do Instituto Oswaldo Cruz. 2010;105(2):163–167. doi: 10.1590/s0074-02762010000200009. [DOI] [PubMed] [Google Scholar]
- 18.Clinical and Laboratory Standards Institute. Document. M100-S15. Wayne, Pa, USA: CLSI; 2010. Performance standards for antimicrobial susceptibility testing: fifteenth informational supplement. [Google Scholar]
- 19.Mabilat C., Goussard S. PCR detection and identification of genes for extended-spectrum β-lactamases. In: Persing D. H., Smith T. F., Tenover F. C., White T. J., editors. Diagnostic Molecular Microbiology: Principles and Applications. Washington, DC, USA: American Society for Microbiology; 1993. pp. 553–559. [Google Scholar]
- 20.Rasheed J. K., Jay C., Metchock B., et al. Evolution of extended-spectrum β-lactam resistance (SHV-8) in a strain of Escherichia coli during multiple episodes of bacteremia. Antimicrobial Agents and Chemotherapy. 1997;41(3):647–653. doi: 10.1128/aac.41.3.647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Moland E. S., Black J. A., Ourada J., Reisbig M. D., Hanson N. D., Thomson K. S. Occurrence of newer β-lactamases in Klebsiella pneumoniae isolates from 24 U.S. Hospitals. Antimicrobial Agents and Chemotherapy. 2002;46(12):3837–3842. doi: 10.1128/aac.46.12.3837-3842.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Jones C. H., Ruzin A., Tuckman M., Visalli M. A., Petersen P. J., Bradford P. A. Pyrosequencing using the single-nucleotide polymorphism protocol for rapid determination of TEM- and SHV-type extended-spectrum β-lactamases in clinical isolates and identification of the novel β-lactamase genes bla SHV-48, bla SHV-105, and bla TEM-155 . Antimicrobial Agents and Chemotherapy. 2009;53(3):977–986. doi: 10.1128/aac.01155-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Yigit H., Queenan A. M., Anderson G. J., et al. Novel carbapenem-hydrolyzing-lactamase, KPC-1, from a carbapenem-resistant strain of Klebsiella pneumoniae . Antimicrobial Agents and Chemotherapy. 2001;45(4):1151–1161. doi: 10.1128/aac.45.4.1151-1161.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.BLAST. Basic Local Alignment Search Tool, http://blast.ncbi.nlm.nih.gov/Blast.cgi.
- 25.Adamis G., Papaioannou M. G., Giamarellos-Bourboulis E. J., Gargalianos P., Kosmidis J., Giamarellou H. Pharmacokinetic interactions of ceftazidime, imipenem and aztreonam with amikacin in healthy volunteers. International Journal of Antimicrobial Agents. 2004;23(2):144–149. doi: 10.1016/j.ijantimicag.2003.07.001. [DOI] [PubMed] [Google Scholar]
- 26.Ahsman M. J., Wildschut E. D., Tibboel D., Mathot R. A. Pharmacokinetics of cefotaxime and desacetylcefotaxime in infants during extracorporeal membrane oxygenation. Antimicrobial Agents and Chemotherapy. 2010;54(5):1734–1741. doi: 10.1128/AAC.01696-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Monteiro J., Santos A. F., Asensi M. D., Peirano G., Gales A. C. First report of KPC-2-producing Klebsiella pneumoniae strains in Brazil. Antimicrobial Agents and Chemotherapy. 2009;53(1):333–334. doi: 10.1128/aac.00736-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Peirano G., Seki L. M., Val Passos V. L., Pinto M. C. F. G., Guerra L. R., Asensi M. D. Carbapenem-hydrolysing β-lactamase KPC-2 in Klebsiella pneumoniae isolated in Rio de Janeiro, Brazil. Journal of Antimicrobial Chemotherapy. 2009;63(2):265–268. doi: 10.1093/jac/dkn484. [DOI] [PubMed] [Google Scholar]
- 29.Sirot D., Recule C., Chaibi E. B., et al. A complex mutant of TEM-1 β-lactamase with mutations encountered in both IRT-4 and extended-spectrum TEM-15, produced by an Escherichia coli clinical isolate. Antimicrobial Agents and Chemotherapy. 1997;41(6):1322–1325. doi: 10.1128/aac.41.6.1322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Gunkel A.-G., Hechler U., Martin H. H. State of penicillin-binding proteins and requirements for their bactericidal interaction with β-lactam antibiotics in Serratia marcescens highly resistant to extended-spectrum β-lactams. Journal of General Microbiology. 1991;137(2):243–252. doi: 10.1099/00221287-137-2-243. [DOI] [PubMed] [Google Scholar]
- 31.Deloney C. R., Schiller N. L. Competition of various β-lactam antibiotics for the major penicillin-binding proteins of Helicobacter pylori: antibacterial activity and effects on bacterial morphology. Antimicrobial Agents and Chemotherapy. 1999;43(11):2702–2709. doi: 10.1128/aac.43.11.2702. [DOI] [PMC free article] [PubMed] [Google Scholar]