Abstract
Modern day cardiac surgery evolved upon the advent of cardiopulmonary bypass machines (CPB) in the 1950s. Following this development, cardiac surgery in recent years has improved significantly. Despite such advances and the introduction of new technologies, neurological sequelae after cardiac surgery still exist. Ischaemic stroke, delirium, and cognitive impairment cause significant morbidity and mortality and unfortunately remain common complications. Postoperative cognitive decline (POCD) is believed to be associated with the presence of new ischaemic lesions originating from emboli entering the cerebral circulation during surgery. Cardiopulmonary bypass was thought to be the reason of POCD, but randomised controlled trials comparing with off-pump surgery show contradictory results. Attention has now turned to the growing evidence that perioperative risk factors, as well as patient-related risk factors, play an important role in early and late POCD. Clearly, identifying the mechanism of POCD is challenging. The purpose of this systematic review is to discuss the literature that has investigated patient and perioperative risk factors to better understand the magnitude of the risk factors associated with POCD after cardiac surgery.
1. Introduction
Neurological complication after cardiac surgery is of a considerable concern and debate exists as to which perioperative factors may be responsible for this adverse injury. Significant advances in all aspects of intraoperative and postoperative care mean cardiac surgery is now safer than ever before [1]. However, as the complexity of surgical procedures increases and the population ages, neurological manifestations and adverse cognitive outcomes are of concern. Cognitive decline limits the ability to complete activities of daily living [2] and increase the likelihood of dependence after discharge [3]. It is therefore paramount to determine the aetiology and extent of brain injury. Such complications vary from subtle cognitive impairment to catastrophic stroke events. The complexity of the brain is demonstrated by small lesions potentially causing significant loss of function, with larger lesions on occasion causing asymptomatic outcomes.
Over time, the demographic characteristics of patients undergoing cardiac surgery have shifted to include a higher proportion of elderly patients, undergoing increasingly complex procedures. The average age of cardiac surgery patients has increased from ~64 years in 2001 to ~67 years in 2010. The number of patients with neurological disease prior to surgery has nearly doubled from 1.4% in 2001 to ~2.8% in 2010. Cardiac surgery procedures have also become more complex, with the number of patients undergoing isolated coronary artery bypass graft (CABG) decreasing by almost 20% from 2001 to 2010. Despite higher patient risk profiles, the mortality rate has fallen slightly from 4.0% in 2001/2002 to 3.1% in 2010/2011 (National Cardiac Surgery Audit, UCL, 2012).
Routine clinical examination covers crucial neurological abnormalities such as ataxia, visual defects, paresis, and hypaesthesia [4]. It also includes focal neuropsychological deficits such as apraxia, dyscalculia, and aphasia. However, more global cerebral dysfunction, such as neuropsychological decline, mood, and memory disturbances, personality changes, and decline in psychomotor speed are commonly missed because they require more explicit examination using specialised neuropsychological tests [5]. Postoperative cognitive decline (POCD) broadly refers to difficulties associated with memory and general information processing after surgery. At present POCD is not documented in the International Classification of Diseases and is not listed as a diagnosis.
2. Methods
2.1. Data Sources
A systematic literature search was conducted from searching articles from PubMed and EMBASE. Search terms were created by combining the following medical subject headings (MeSH terms): “Coronary Artery Bypass” OR “Coronary Artery Bypass, Off-Pump” OR “Valve Surgery” OR “Thoracic Surgery” OR “Cardiac Surgical Procedures” AND “Cognitive Therapy” OR “Cognition Disorders” OR “Cognition” OR “Neuropsychology” OR “Neuropsychological Tests” OR “Mild Cognitive Impairment.”
2.2. Study Selection
All studies published in English between June 1967 and August 2014 and featuring adult human subjects were eligible for review. Abstracts were excluded if they involved paediatric surgery, operations other than cardiac surgery, or no measurement of cognitive function. Case reports and studies of cardiac procedures such as angioplasty, angiography, valvuloplasty, and Transcatheter Aortic Valve Implantation (TAVI) were also excluded. Studies generating multiple publications from the same cohort were reported only once.
2.3. Quality Assessment
Abstracts involving both cardiac surgery and cognitive function were independently reviewed by two investigators (Nikil Patel and Emma M. L. Chung) and studies of adult cardiac surgery patients that assessed both before and after operative cognitive function were identified for full paper review. Where there was disagreement among investigators the full text was reviewed. Additionally, the reference lists of selected articles were evaluated for any additional articles of interest.
2.4. Analysis
Articles short-listed for full manuscript review were summarised in an Excel spreadsheet listing the study design (observational, RCT, etc.), number of patients, type(s) of surgery, outcome measures, and time point of neurocognitive assessment. Studies that included assessment of anxiety and depression were also recorded, as these conditions can impact the outcome of cognitive assessments. There was insufficient homogeneity between studies to allow a quantitative, meta-analytic approach of region of interest studies. Therefore, a critical, systematic review was undertaken.
3. Results
A total of 638 abstracts were systematically identified using our search criteria of which 426 papers were suitable for full review. Of these, 296 were observational studies and 130 were RCTs. Although over 420 original research articles were identified as having investigated cognitive decline following cardiac surgery, we found little consensus on the incidence, severity, and time course of symptoms. Differing methodologies used between studies made it difficult to directly compare study findings through systematic meta-analysis.
3.1. Time of Postoperative Testing and Cognitive Decline
Most studies evaluating cognitive decline focus on changes in executive function, learning language, visual spatial skills, attention, and memory [6]. However, neuropsychological tests vary considerably between studies and also appear to depend on the timing of neurocognitive assessment. By narrowing the search to empirical research articles that studied postoperative neuropsychological assessment as a primary outcome, the number of publications was reduced to 137 articles. Thirty-three of these articles were excluded because the total percentage of patients who declined in cognitive tests was unclear. Four articles had published the same data twice and full-texts were unavailable for 6 articles. A total of 94 studies were identified to establish the distribution of cognitive decline over several time points. Grouping studies where assessments were performed at similar time points and plotting the proportion of patients estimated to be affected by cognitive decline suggest that 40–60% of patients experience cognitive decline when tested within 2 weeks of surgery, falling to 30–40% after 8–10 weeks, recovering to 10–20% at 1 year, with proportion of patients experiencing cognitive decline increasing again at 3–5 years, Figure 1.
Figure 1.
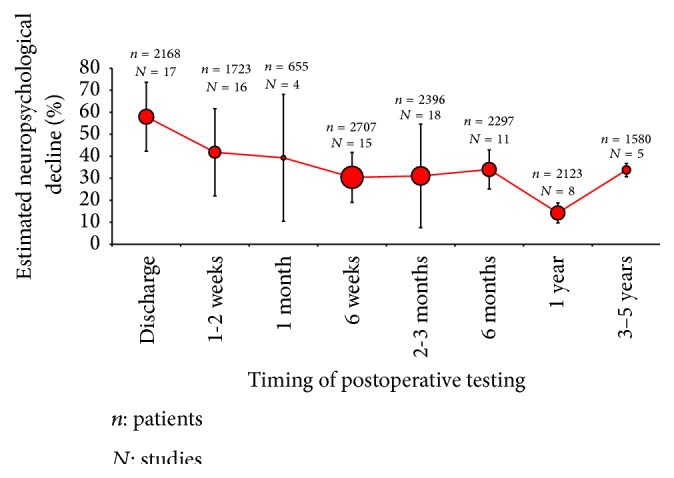
Studies attempting to quantify neuropsychological decline at various time points. The weighted mean and standard deviation (number of patients and % decline) are plotted by combining data from a total of 15649 patients and 94 studies; discharge (17 studies), 1-2 weeks (16 studies), 1 month (4 studies), 6 weeks (15 studies), 2-3 months (18 studies), 6 months (11 studies), 1 year (8 studies), and 3–5 years (5 studies).
Large variations in the estimated incidence of postoperative cognitive decline are observed, even after grouping studies where tests were performed at similar time points, Figure 1. Heterogeneity in assessment methods, patient demographics, and study design may be responsible for these variations.
3.2. Perioperative Risk Factors and Cognitive Decline
Further, we investigated perioperative risk factors associated with cognitive decline. Potential mechanisms implicated in the pathogenesis of cognitive decline investigated in previous research resulted in a total of 92 articles (see PRISMA chart in Supplementary Material available online at http://dx.doi.org/10.1155/2015/370612): anaesthesia: 15 studies; blood pressure: 5 studies; cerebral autoregulation: 4 studies; inflammatory responses: 26 studies; neuroprotective agents: 17 studies; hypothermia and rewarming: 19 studies and 6 studies, respectively.
3.3. Anaesthesia
Sedative and anaesthetic agents with N-methyl-d-aspartate receptor antagonist and γ-aminobutyric acid mediated properties can temporarily change the neurotransmission of the brain by interacting at a cellular level to achieve deep sedation during surgery [7]. Since it would be unethical to perform cardiac surgery without the use of anaesthetic agents, the impact of anaesthesia on cognition is difficult to study. Fifteen studies have investigated whether choice of anaesthesia impacts neurocognitive outcome after cardiac surgery. Of these, 8 were randomised controlled trials (RCTs), comparing 7 different types of anaesthetic agent. Studies showing an improvement, decline, and no difference in postoperative outcome are summarised in Table 1.
Table 1.
Studies comparing cognition after cardiac surgery following administration of different types of anaesthetic.
| Study | Study design | Number of patients |
Type of anaesthesia/drug | Time of assessment | Outcome |
|---|---|---|---|---|---|
| Dumas et al., 1999 [37] | RCT | 48 | Fentanyl and early extubation | 8 weeks | Improved cognition |
| Dowd et al., 2001 [38] | RCT | 78 | Propofol and lorazepam | 6–12 months | Improved cognition |
| Bottio et al., 2007 [39] | Obsv. | 50 | Epidural anaes. | 6 months | Improved cognition |
| Delphin et al., 2007 [40] | Obsv. | 91 | Sevoflurane and isoflurane | 2 hours and 1 day | Improved cognition |
| Kanbak et al., 2007 [41] | RCT | 40 | Isoflurane, sevoflurane, and desflurane | 3 and 6 days | Improved cognition |
| Hudetz et al., 2009 [42] | Obsv. | 78 | Ketamine | 1 week | Improved cognition |
| Schoen et al., 2011 [43] | RCT | 117 | Sevoflurane and propofol | 2, 4, and 6 days | Improved cognition |
| Kanbak et al., 2007 [41] | RCT | 40 | Sevoflurane and desflurane | 3 and 6 days | Decline |
| Kadoi et al., 2003 [44] | RCT | 180 | Propofol and fentanyl | 6 months | No difference |
| Silbert et al., 2006 [45] | Obsv. | 300 | Fentanyl | 1 week, 3 months, 1 year | No difference |
| Kadoi and Goto, 2007 [46] | Obsv. | 109 | Sevoflurane | 6 months | No difference |
| Lehmann et al., 2007 [47] | RCT | 66 | Sufentanil and midazolam | Discharge | No difference |
| Evered et al., 2011 [48] | Obsv. | 281 | General anaesthetics | 1 week and 3 months | No difference |
| Parra et al., 2011 [49] | Obsv. | 48 | Sevoflurane | 3 months | No difference |
| Royse et al., 2011 [50] | RCT | 180 | Desflurane andpropofol | Discharge and 3 months | No difference |
Obsv.: observational.
This research suggests that choice of anaesthetic has potential to affect cognition, particularly when tests are performed soon after surgery. However, in the majority of larger studies, the choice of anaesthetic had no impact on cognitive outcome.
3.4. Blood Pressure
A number of studies have investigated the association between low blood pressure during cardiac surgery and cognitive decline. Although normal blood pressure in conscious patients is approximately 120/80 mmHg, it is common for the blood pressure to be much lower during surgery. As the brain has a lower metabolic demand during anaesthesia, this is not thought to adversely affect tissue perfusion; however, low blood pressure may impair embolus clearance and affect the efficiency of cerebral autoregulation. A total of 5 studies have used neuropsychological tests to investigate whether mean arterial blood pressure had any impact on postoperative cognitive outcome, Table 2.
Table 2.
Studies investigating POCD associated with intraoperative blood pressure variation.
| Study | Study design |
Number of patients |
Type of intervention | Time of assessment | Outcome |
|---|---|---|---|---|---|
| Gold et al., 1995 [8] | RCT | 248 | High (80–100 mmHg) versus low (50–60 mmHg) BP | 6 months | Decline with lower BP |
| Siepe et al., 2011 [9] | RCT | 92 | High (80–90 mmHg) versus low (60–70 mmHg) BP | 2 days | Decline with lower BP |
| Gottesman et al., 2007 [51] | Obsv. | 15 | Low MAP (50–70 mmHg) | 3–5 days and 1 month | Decline with lower BP |
| Newman et al., 1995 [52] | Obsv. | 237 | Low MAP (50–60 mmHg) | Discharge | Decline with lower BP |
| Charlson et al., 2007 [10] | RCT | 412 | High MAP (57–90 mmHg) versus custom (capped at 90 mmHg) | 6 months | No difference in outcome |
Obsv.: observational.
In the study by Gold et al., a higher mean arterial pressure (80–110 mmHg) during CPB appeared to be associated with a lower stroke rate (2.4%) compared to a low mean arterial pressure between 45 and 60 mmHg (7.2%), p = 0.026. However, at 6-month follow-up the proportion of patients with neuropsychological declines (11% and 12%, resp.) were comparable [8]. In another study, Siepe et al. showed greater proportion of patients with cognitive decline two days following CABG in patients with mean arterial pressure in the range 60–70 mmHg compared to 80–90 mmHg; however cerebral oxygen saturation was similar in both groups [9]. The largest RCT by Charlson et al. found no difference in cognition between a “custom” group (average BP: 79 mmHg) and High BP group (average BP: 89 mmHg); however, the average difference in BP between groups was only 10 mmHg, which may not be a clinically significant difference [10]. Overall, studies appear to support the idea that maintenance of a sufficiently high mean arterial pressure during cardiac surgery is important for safeguarding perfusion to the brain.
3.5. Cerebral Autoregulation
Some researchers have proposed that it is not mean arterial pressure (MAP) per se that contributes to cognitive decline, but the capacity of the brain's blood flow regulation mechanisms to respond appropriately to blood pressure variations and changes in oxygen saturation. A number of studies have investigated cerebral autoregulation (CA) in response to blood pressure changes during cardiac surgery and found that a significant proportion of patients struggle to autoregulate their cerebral blood supplies intraoperatively [11]. However, only 4 studies have specifically investigated CA during cardiac surgery in conjunction with pre- and postoperative neuropsychological assessment, Table 3.
Table 3.
Studies investigating cerebral autoregulation during cardiac surgery in conjunction with neurocognitive tests.
| Study | Study design | Number of patients | Cerebral autoregulation measures | Time of assessment | Outcome |
|---|---|---|---|---|---|
| Patel et al., 1993 [53] | RCT | 70 | Xenon-133 isotope clearance, CMRO2 (cerebral metabolic rate for oxygen), CERO2 (cerebral extraction ratio for oxygen) | 6 weeks | Decline with impaired CA |
| Patel et al., 1996 [54] | RCT | 70 | CBF, CBFv, and O2 saturation were measured during 4 phases of surgery | 6 weeks | Decline with impaired CA |
| Govier et al., 1984 [55] | Obsv. | 67 | Partial pressure of arterial carbon dioxide (PaCO2), clearance of xenon-133 | Discharge | No difference |
| Newman et al., 1994 [12] | Obsv. | 215 | Xenon-133 clearance, CMRO2, cerebral AV difference (C[AV]O2) | Discharge | No difference |
Obsv.: observational.
All four studies determined pressure-flow and metabolic-flow cerebral autoregulation during cardiopulmonary bypass using the 133Xe clearance cerebral blood flow method. Two studies in Table 3 by the same author (Patel et al.) support the theory that impaired cerebral autoregulation is associated with a decline in postoperative outcome at 6 weeks, whereas two studies showed no association. The largest study by Newman et al. investigated CA in 215 patients and concluded that neuropsychological dysfunction at discharge was not explained by impaired CA; however increased oxygen extraction (measured using a thermodilution pulmonary artery catheter) was observed to be associated with a decline in some cognitive tests. They interpreted this suggesting that an imbalance in cerebral tissue oxygen supply may contribute to POCD [12]. In a recent trial it has also been proposed that some anaesthetic agents suppress autoregulatory responses more than others [13]. As far as we are aware, no studies have yet looked at the relationship between CA and POCD beyond 6 weeks.
3.6. Inflammatory Responses
All types of surgery have the risk of developing systemic inflammation; however, in cardiac surgery using CPB the blood is exposed to foreign surfaces which have potential to stimulate proinflammatory responses. Inflammation causes endothelial dysfunction, which can lead to leakage between the blood-brain barrier and tissue oedema [14]. It has been shown that cytokines (e.g., TNF-alpha, interleukin-1, and interleukin-6) have been linked to neuropathology [15, 16]. These elementary changes are hypothesised to affect the brain regardless of microembolic load received during surgery [17, 18] and potentially provide an explanation for early cognitive decline [19].
Cardiopulmonary bypass components that come into contact with the blood can be coated with biocompatible materials such as poly-2-methoxyethylacrylate, heparin, trillium, and synthetic proteins. These coatings aim to reduce inflammatory responses triggered during CPB. Heparin-coated circuits, in particular, have undergone considerable investigation in previous research. A total of 26 studies (including 7 RCTs) have used neuropsychological tests to investigate whether there is a strong association between inflammation and cognitive decline, Table 4.
Table 4.
Studies investigating whether biomarkers associated with inflammation and/or interventions aimed at reducing inflammation are associated with changes in cognition after surgery.
| Study | Study design |
Number of patients |
Marker for cerebral damage | Time of assessment | Outcome |
|---|---|---|---|---|---|
| Fitch et al., 1999 [20] | RCT | 35 | Inhibition of complement activation by specific antibody and no antibody | Discharge | Improved cognition |
| Heyer et al., 2002 [21] | RCT | 99 | Inhibition of complement activation by heparin-coated CPB | 5 days and 6 weeks | Improved cognition |
| Baufreton et al., 2005 [19] | RCT | 30 | Inhibition of complement activation by heparin-coated CPB | Discharge | Improved cognition |
| Skrabal et al., 2006 [22] | RCT | 39 | PMEA-coated circuits and noncoated circuits | 7–10 days | Improved cognition |
| Wimmer-Greinecker et al., 1998 [56] | Obsv. | 76 | >S-100 and NSE | 5 days and 2 months | Decline |
| Jönsson et al., 1999 [57] | Obsv. | 132 | >S-100 | 2 weeks and 2 months | Decline |
| Kilminster et al., 1999 [58] | Obsv. | 130 | >S-100 | 6–8 weeks | Decline |
| Rasmussen et al., 1999 [59] | Obsv. | 35 | >NSE | Discharge and 3 months | Decline |
| Derkach et al., 2000 [60] | RCT | 27 | >S-100 and NSE (deep and mild hypothermic) | 6 months | Decline |
| Diegeler et al., 2000 [61] | RCT | 40 | >S-100 (on- and off-pump) | 1 week | Decline |
| Georgiadis et al., 2000 [62] | Obsv. | 190 | >S-100 | Discharge | Decline |
| Lloyd et al., 2000 [63] | RCT | 125 | >S-100 (on- and off-pump) | 3 months | Decline |
| Basile et al., 2001 [64] | Obsv. | 16 | >S-100 and NSE | 6 months | Decline |
| Rasmussen et al., 2002 [65] | Obsv. | 15 | >NSE | Discharge and 3 months | Decline |
| Farsak et al., 2003 [66] | Obsv. | 50 | >S-100 | Discharge | Decline |
| Mathew et al., 2003 [67] | Obsv. | 460 | Reduced preoperative endotoxin immunity | 6 weeks | Decline |
| Jönsson et al., 2004 [68] | Obsv. | 56 | >S-100 | 6 months | Decline |
| Kofke et al., 2004 [69] | Obsv. | 28 | Apo epsilon 4 allele, >S-100 | 8 and 24 hrs | Decline |
| Snyder-Ramos et al., 2004 [70] | Obsv. | 64 | >S-100 and NSE | Throughout 7 days | Decline |
| Kálmán et al., 2006 [71] | Obsv. | 14 | >Cytokine interleukin-6 | 1 week and 6 months | Decline |
| Ramlawi et al., 2006 [72] | Obsv. | 42 | >C-reactive protein | 6 hours and 4 days | Decline |
| Lazibat et al., 2012 [24] | Obsv. | 62 | >S-100 | 2 days | Decline |
| Bayram et al., 2013 [25] | Obsv. | 64 | >S-100 | 1 week | Decline |
| Westaby et al., 2001 [26] | Obsv. | 1001 | >S-100 and NSE | 5 days and 3 months | No difference |
| Mathew et al., 2005 [73] | Obsv. | 440 | Statin treatment | 6 weeks | No difference |
| Plaschke et al., 2013 [74] | Obsv. | 151 | Preoperative serum anticholinergic activity | 3 months | No difference |
NSE: neuron-specific enolase, PMEA: poly-2-methoxyethylacrylate, and Obsv.: observational.
All studies that have randomised patients to receive a heparin-coated CPB system found neuropsychological outcome was better in patients receiving the heparin-coated circuit [19–22]. In studies investigating inflammatory responses, a consensus panel has concluded that “the use of surface-modified circuits might be effective at attenuating the systemic inflammatory response to CPB and improving outcome” [23]. Many markers associated with susceptibility to brain ischaemia such as S-100 beta and neuron-specific enolase (NSE) have been suggested to be associated with an increased risk of cognitive decline [24–26]. Inflammation may also play an important role in our understanding of long-term cognitive function. Biomarkers for inflammation tend to be higher in patients with chronic cardiovascular disease [25]. Overall, the role of inflammation in the pathogenesis of cognitive decline appears to warrant further investigation [27].
3.7. Neuroprotective Agents
A number of neuroprotective agents have been investigated to assess whether these could be administered to help preserve neurocognitive function. The results of 17 studies investigating whether neuroprotective agents reduce the incidence of POCD are summarised in Table 5.
Table 5.
RCTs investigating the efficacy of neuroprotection, or neuroprotective agents, in reducing cognitive decline after cardiac surgery.
| Study | Number of patients | Type of neuroprotective drug | Time of assessment | Outcome |
|---|---|---|---|---|
| Grieco et al., 1996 [75] | 29 | GM-100 (ganglioside) or placebo | 1 week and 6 months | Improved cognition |
| Arrowsmith et al., 1998 [76] | 171 | Remacemide or placebo | 2 months | Improved cognition |
| Svensson et al., 2002 [77] | 403 | Mannitol, thiopental, MgSO4, lidocaine | 2-3 weeks | Improved cognition |
| Wang et al., 2002 [29] | 118 | Lidocaine or placebo | 9 days | Improved cognition |
| Uebelhack et al., 2003 [78] | 64 | Piracetam or placebo | 3 days | Improved cognition |
| Szalma et al., 2006 [79] | 98 | Piracetam or placebo | 6 weeks | Improved cognition |
| Haljan et al., 2009 [80] | 32 | Erythropoietin or placebo | Discharge and 2 months | Improved cognition |
| Hudetz et al., 2009 [42] | 52 | Ketamine or placebo | 1 week | Improved cognition |
| Zhang et al., 2011 [81] | 200 | Benzyl alcohols or saline (placebo) | Discharge and 3 months | Improved cognition |
| Kong et al., 2002 [82] | 245 | Chlormethiazole/administration or placebo | 4–7 weeks | No difference |
| Taggart et al., 2003 [83] | 150 | Imidazoles: low dose (10 mg) or high dose (100 mg) or placebo | 5 days and 3 months | No difference |
| Mathew et al., 2004 [84] | 914 | Pexelizumab bolus, bolus plus infusion, or placebo | 4 days and 1 month | No difference |
| Mathew et al., 2005 [73] | 440 | Hydroxymethylglutaryl-CoA reductase inhibitors | 6 weeks | No difference |
| Hogue et al., 2007 [85] | 174 | 17-beta estradiol or placebo | 4–6 weeks | No difference |
| Mathew et al., 2009 [31] | 241 | Lidocaine or placebo | 6 weeks and 1 year | No difference |
| Mitchell et al., 2009 [32] | 158 | Lidocaine or placebo | 10 weeks and 25 weeks | No difference |
| Holinski et al., 2011 [86] | 88 | Piracetam or placebo | 3 days | No difference |
One of the most commonly used neuroprotective agents is Lidocaine, which featured in 4 of the 17 studies. Lidocaine is thought to inhibit inflammatory responses during cardiac surgery by modulation of inflammatory mediators, reduction in cerebral metabolism, and deceleration of ischaemic ion fluxes [28]. Two studies showed improved outcome with the use of the drug [29, 30], while two studies showed no difference [31, 32]. Currently, no trials have demonstrated a reproducible clinically significant benefit conferred by the use of any particular neuroprotective drug.
3.8. Hypothermia and Rewarming
The patient's temperature during cardiac surgery has long been thought to play a role in neurological outcome. Several studies have focused their trials on whether reducing the metabolic demand of the brain through hypothermia is neuroprotective. Based on our literature search, 41 studies investigating the effects of temperature were identified. Seventeen studies were excluded from the final result due to lack of clarity in neuropsychological assessments and outcomes. Results from a total of 19 studies investigating the effect of temperature on pre- and postoperative neuropsychological tests are summarised in Table 6.
Table 6.
Studies investigating POCD associated with temperature during cardiac surgery.
| Study | Study design |
Number of patients |
Mean temperature (Celsius) | Time of assessment | Outcome |
|---|---|---|---|---|---|
| Grimm et al., 2000 [87] | RCT | 144 | (1) Normothermia: 37°C (2) Hypothermia: 32°C |
1 week and 4 months | Improved cognition (with normothermia) |
|
| |||||
| Shaaban-Ali et al., 2002 [88] | RCT | 60 | (1) Normothermia: 34°C (2) Hypothermia: 28°C |
5 days | Improved cognition (with normothermia) |
|
| |||||
| Nathan et al., 1995 [89] | Obsv. | 30 | Maintain ≤ 34°C | 1 week | Improved cognition (with hypothermia) |
|
| |||||
| Grocott et al., 2002 [90] | Obsv. | 300 | Post-op hypothermia only | 6 weeks | Improved cognition (with hypothermia) |
|
| |||||
| Kadoi et al., 2004 [91] | RCT | 60 | (1) Normothermia: 37°C (2) Hypothermia: 32°C |
1 month | Improved cognition (with hypothermia) |
|
| |||||
| Boodhwani et al., 2006 [92] | RCT | 448 | (1) Normothermia: 37°C (2) Hypothermia: 34°C |
1 week | Improved cognition (with hypothermia) |
|
| |||||
| Hiraoka et al., 2012 [93] | Obsv. | 11 | Hypothermia: 20–22°C | 3 weeks and 6 months | Improved cognition (with hypothermia) |
|
| |||||
| McLean et al., 1994 [94] | RCT | 155 | (1) Hyperthermia: >34°C (2) Hypothermia: <28°C |
5 days and 3 months | No difference |
|
| |||||
| Regragui et al., 1996 [95] | RCT | 97 | (1) Normothermia: 37°C (2) Hypothermia: 28°C & 32°C |
6 weeks | No difference |
|
| |||||
| Heyer et al., 1997 [96] | RCT | 99 | (1) Normothermia: 34°C (2) Hypothermia: 28°C |
Discharge and 6 weeks | No difference |
|
| |||||
| Kneebone et al., 1998 [97] | Obsv. | 50 | (1) Normothermia: 37°C (2) Hypothermia: 30–32°C |
1 week | No difference |
|
| |||||
| Reich et al., 1999 [98] | Obsv. | 149 | (1) Deep hypothermia: 12–15°C (<25 mins) (2) Deep hypothermia: 12–15°C (>25 mins) |
1 month | No difference |
|
| |||||
| Kaukinen et al., 2000 [99] | RCT | 36 | (1) Normothermia: 36-37°C (2) Hypothermia: 28°C |
5 days and 11–23 months | No difference |
|
| |||||
| Górna et al., 2001 [100] | Obsv. | 33 | No full text | 3–10 days | No difference |
|
| |||||
| Grigore et al., 2001 [101] | RCT | 300 | (1) Normothermia: 35.5–36.5°C (2) Hypothermia: 28–30°C |
6 weeks | No difference |
|
| |||||
| Kaukuntla et al., 2004 [102] | Obsv. | 60 | (1) Normothermia: 35°C (2) Differential temperature management |
1 and 8 weeks | No difference |
|
| |||||
| Reich et al., 2004 [103] | Obsv. | 61 | Monitoring during deep hypothermic arrest (28°C) | Discharge | No difference |
|
| |||||
| Boodhwani et al., 2007 [33] | RCT | 268 | (1) Normothermia: 37°C (2) Hypothermia: 34°C |
Discharge and 3 months | No difference |
|
| |||||
| Kunihara et al., 2007 [104] | Obsv. | 26 | (1) Normothermia: 34°C (2) Hypothermia: 22°C |
1 week | No difference |
Obsv.: observational.
Some studies suggest that hypothermia is more effective than normothermia in protecting the brain during surgery; however, other studies report no obvious difference between “mild hypothermia” and “normothermia” in terms of neuropsychological performance at discharge (49% and 45%, resp.) and at 3 months (4% and 8%, resp.) [33].
Some researchers have proposed that the brain could be susceptible to insult during rewarming from hypothermia, particularly if cerebral autoregulation mechanisms are unable to compensate for a sudden increase in metabolic activity associated with changes in temperature. Six studies have been conducted to examine the effect of rewarming rate on POCD, and all of these have shown a benefit in postoperative outcome associated with slower rewarming, Table 7.
Table 7.
Studies investigating POCD associated with the rate of rewarming during cardiac surgery.
| Study | Study design |
Number of patients |
Mean temperature (Celsius) | Time of assessment | Outcome |
|---|---|---|---|---|---|
| Mora et al., 1996 [105] | RCT | 138 | (1) Rewarm 1-2°C (per increase) (2) Rewarm 3–5°C (per increase) |
1–3 days, 7–10 days, and 1 month | Improved cognition with slower rewarm |
|
| |||||
| Nathan et al., 2001 [106] | Obsv. | 294 | (1) Rewarm to 34°C (1°C per increase) (2) Rewarm to 37°C (3°C per increase) |
1 week and 3 months | Improved cognition with slower rewarm |
|
| |||||
| Grigore et al., 2002 [107] | Obsv. | 100 | (1) Rewarm to 32°C (max within 3 mins) (2) Rewarm to 37°C (max within 3 mins) |
6 weeks | Improved cognition with slower rewarm |
|
| |||||
| Kawahara et al., 2003 [108] | RCT | 100 | (1) Rewarm 1-2°C (per increase) (2) Rewarm 4-5°C (per increase) |
1 month | Improved cognition with slower rewarm |
|
| |||||
| Nathan et al., 2007 [109] | RCT | 223 | (1) Rewarm to 34°C (1°C per increase) (2) Rewarm to 37°C (3°C per increase) |
1 week | Improved cognition with slower rewarm |
|
| |||||
| Sahu et al., 2009 [110] | RCT | 80 | (1) Rewarm 1–3°C (per increase) (2) Rewarm 3–5°C (per increase) |
5 days | Improved cognition with slower rewarm |
Obsv.: observational.
4. Conclusion
Neuropsychological function is a soft outcome measure and has proved challenging to quantify postoperatively. Although neuropsychological tests theoretically provide a highly sensitive means of quantifying changes in cognition, differences in test batteries, timing of assessment, and criteria for defining neuropsychological decline generate considerable heterogeneity in the data, which limits our ability to compare the results of different studies. Depending on the timing of the neurocognitive tests and the definition used for determining decline, the reported incidence of neurocognitive decline after cardiac surgery varied extensively. The outcome suggests that 50–70% of patients experience cognitive decline when tested within one week of surgery, falling to 30–50% after 8–10 weeks, recovering to 10–20% at 1 year, and then declines again at 3–5 years. Currently, there is no widely accepted clinical definition of cognitive decline; therefore, it is possible that arbitrary definitions of decline have resulted in an overestimation of the incidence of decline. At present, there is no evidence to suggest that the long-term incidence of cognitive decline differs from that of nonoperative controls. Estimating long-term cognitive decline can be difficult, as normal ageing and dementia interfere with studies with older populations.
Further research is required to develop a more dynamic and nuanced picture of interactions between underlying pre- and perioperative risk factors. It is apparent that studies investigating isolated perioperative factors are insufficient to explain complex interactions between temperature, cerebral autoregulation, oxygen saturation, and brain metabolism. To date, isolated interventions and neuroprotective drugs aimed at improving cognitive outcome have proved to be largely ineffective. Literature examining underlying and perioperative risk factors associated with the pathogenesis of cognitive decline suggests that there is no single causative factor responsible for POCD. It seems likely that the causes are multifactorial, due to emboli, impaired perfusion, chronic cardiovascular disease, and inflammatory responses.
Interpreting the risk factors associated with postoperative cognitive decline, it seems that efforts to protect the brain during surgery are intrinsically linked with the need to control the progression of cardiovascular disease, especially in older patients. It is possible that patients may be exceeding a “threshold” of preexisting vulnerability where the brain's ability to compensate for injuries or inflammation during surgery is absent. It is also important to address that cardiac surgery equipment advances are a confounder within this review and should be considered. In summary, the literature examining underlying risk factors and perioperative risk factors associated with the pathogenesis of cognitive decline suggests that there is no single factor responsible for postoperative cognitive decline or single intervention capable of protecting the brain during surgery. Overall, the pathogenesis of cognitive decline following surgery still remains unclear.
Several factors have been associated with brain injury including hypoperfusion, arrhythmias, rapid rewarming, and inflammation (local or global) [34–36]. The process of preventing such brain injury involves prevention of such events occurring; however, to date no single intervention has successfully prevented cognitive decline, signalling an increased likelihood of a multifactorial aetiology. The advent of new technologies to prevent physiological stress on the brain has focussed on neuroprotective agents or perfusionist strategies. Although prevention has an important role, it would be ideal to develop methods of protecting or restoring neurocognitive decline to nearer preoperative baselines.
Cardiac surgery is a triumph of modern day medicine, and its acceptance as a safe procedure is widespread. Unfortunately, postoperative cognitive issues remain a consideration. As cardiac surgery procedures are now being challenged by less invasive methods, perhaps intraoperative transcranial Doppler monitoring, neuropsychological tests, and neuroimaging will play an increasingly important role in optimising treatment.
Supplementary Material
To help with the commentary of the systematic review and ensure the transparent and complete reporting, a PRISMA chart has been attached to view the full selection process of the studies included. The diagram depicts the flow of information through different phases of the systematic review, mapping out the number of records identified, included and excluded.
Acknowledgments
The authors would like to thank the Department of Cardiovascular Sciences, University of Leicester, and the Department of Medical Physics, University Hospitals of Leicester NHS Trust. Nikil Patel is funded by the National Institute of Health Research (NIHR) Leicester Cardiovascular Biomedical Research Unit. Jatinder S. Minhas is an Academic Clinical Fellow in Stroke Medicine funded by the NIHR.
Conflict of Interests
The authors declare that they have no conflict of interests.
Authors' Contribution
Nikil Patel and Emma M. L. Chung conducted the literature search and data extraction. Nikil Patel, Emma M. L. Chung, and Jatinder S. Minhas drafted the paper.
References
- 1.Selnes O. A., McKhann G. M., Borowicz L. M., Jr., Grega M. A. Cognitive and neurobehavioral dysfunction after cardiac bypass procedures. Neurologic Clinics. 2006;24(1):133–145. doi: 10.1016/j.ncl.2005.10.001. [DOI] [PubMed] [Google Scholar]
- 2.Phillips-Bute B., Mathew J. P., Blumenthal J. A., et al. Association of neurocognitive function and quality of life 1 year after Coronary Artery Bypass Graft (CABG) surgery. Psychosomatic Medicine. 2006;68(3):369–375. doi: 10.1097/01.psy.0000221272.77984.e2. [DOI] [PubMed] [Google Scholar]
- 3.Steinmetz J., Christensen K. B., Lund T., Lohse N., Rasmussen L. S. Long-term consequences of postoperative cognitive dysfunction. Anesthesiology. 2009;110(3):548–555. doi: 10.1097/ALN.0b013e318195b569. [DOI] [PubMed] [Google Scholar]
- 4.Brott T., Adams H. P., Jr., Olinger C. P., et al. Measurements of acute cerebral infarction: a clinical examination scale. Stroke. 1989;20(7):864–870. doi: 10.1161/01.str.20.7.864. [DOI] [PubMed] [Google Scholar]
- 5.Murkin J. M., Newman S. P., Stump D. A., Blumenthal J. A. Statement of consensus on assessment of neurobehavioral outcomes after cardiac surgery. Annals of Thoracic Surgery. 1995;59(5):1289–1295. doi: 10.1016/0003-4975(95)00106-U. [DOI] [PubMed] [Google Scholar]
- 6.Rudolph J. L., Schreiber K. A., Culley D. J., et al. Measurement of post-operative cognitive dysfunction after cardiac surgery: a systematic review. Acta Anaesthesiologica Scandinavica. 2010;54(6):663–677. doi: 10.1111/j.1399-6576.2010.02236.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Eckenhoff R. G., Johansson J. S., Wei H., et al. Inhaled anesthetic enhancement of amyloid-beta oligomerization and cytotoxicity. Anesthesiology. 2004;101(3):703–709. doi: 10.1097/00000542-200409000-00019. [DOI] [PubMed] [Google Scholar]
- 8.Gold J. P., Charlson M. E., Williams-Russo P., et al. Improvement of outcomes after coronary artery bypass: a randomized trial comparing intraoperative high versus low mean arterial pressure. The Journal of Thoracic and Cardiovascular Surgery. 1995;110(5):1302–1314. doi: 10.1016/s0022-5223(95)70053-6. [DOI] [PubMed] [Google Scholar]
- 9.Siepe M., Pfeiffer T., Gieringer A., et al. Increased systemic perfusion pressure during cardiopulmonary bypass is associated with less early postoperative cognitive dysfunction and delirium. European Journal of Cardio-Thoracic Surgery. 2011;40(1):200–207. doi: 10.1016/j.ejcts.2010.11.024. [DOI] [PubMed] [Google Scholar]
- 10.Charlson M. E., Peterson J. C., Krieger K. H., et al. Improvement of outcomes after coronary artery bypass II: a randomized trial comparing intraoperative high versus customized mean arterial pressure. Journal of Cardiac Surgery. 2007;22(6):465–472. doi: 10.1111/j.1540-8191.2007.00471.x. [DOI] [PubMed] [Google Scholar]
- 11.Ono M., Joshi B., Brady K., et al. Risks for impaired cerebral autoregulation during cardiopulmonary bypass and postoperative stroke. British Journal of Anaesthesia. 2012;109(3):391–398. doi: 10.1093/bja/aes148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Newman M. F., Croughwell N. D., Blumenthal J. A., et al. Effect of aging on cerebral autoregulation during cardiopulmonary bypass. Association with postoperative cognitive dysfunction. Circulation. 1994;90(5):II243–II249. [PubMed] [Google Scholar]
- 13.Tanaka T., Kai S., Koyama T., et al. General anesthetics inhibit erythropoietin induction under hypoxic conditions in the mouse brain. PLoS ONE. 2011;6(12) doi: 10.1371/journal.pone.0029378.e29378 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Abbott N. J. Inflammatory mediators and modulation of blood-brain barrier permeability. Cellular and Molecular Neurobiology. 2000;20(2):131–147. doi: 10.1023/A:1007074420772. [DOI] [PubMed] [Google Scholar]
- 15.Terrando N., Eriksson L. I., Kyu Ryu J., et al. Resolving postoperative neuroinflammation and cognitive decline. Annals of Neurology. 2011;70(6):986–995. doi: 10.1002/ana.22664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Cibelli M., Fidalgo A. R., Terrando N., et al. Role of interleukin-1β in postoperative cognitive dysfunction. Annals of Neurology. 2010;68(3):360–368. doi: 10.1002/ana.22082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Reinsfelt B., Ricksten S.-E., Zetterberg H., Blennow K., Fredén-Lindqvist J., Westerlind A. Cerebrospinal fluid markers of brain injury, inflammation, and blood-brain barrier dysfunction in cardiac surgery. Annals of Thoracic Surgery. 2012;94(2):549–555. doi: 10.1016/j.athoracsur.2012.04.044. [DOI] [PubMed] [Google Scholar]
- 18.Reinsfelt B., Westerlind A., Blennow K., Zetterberg H., Ricksten S.-E. Open-heart surgery increases cerebrospinal fluid levels of Alzheimer-associated amyloid β . Acta Anaesthesiologica Scandinavica. 2013;57(1):82–88. doi: 10.1111/j.1399-6576.2012.02769.x. [DOI] [PubMed] [Google Scholar]
- 19.Baufreton C., Allain P., Chevailler A., et al. Brain injury and neuropsychological outcome after coronary artery surgery are affected by complement activation. Annals of Thoracic Surgery. 2005;79(5):1597–1605. doi: 10.1016/j.athoracsur.2004.08.061. [DOI] [PubMed] [Google Scholar]
- 20.Fitch J. C. K., Rollins S., Matis L., et al. Pharmacology and biological efficacy of a recombinant, humanized, single-chain antibody C5 complement inhibitor in patients undergoing coronary artery bypass graft surgery with cardiopulmonary bypass. Circulation. 1999;100(25):2499–2506. doi: 10.1161/01.cir.100.25.2499. [DOI] [PubMed] [Google Scholar]
- 21.Heyer E. J., Lee K. S., Manspeizer H. E., et al. Heparin-bonded cardiopulmonary bypass circuits reduce cognitive dysfunction. Journal of Cardiothoracic and Vascular Anesthesia. 2002;16(1):37–42. doi: 10.1053/jcan.2002.29659. [DOI] [PubMed] [Google Scholar]
- 22.Skrabal C. A., Khosravi A., Westphal B., Steinhoff G., Liebold A. Effects of poly-2-methoxyethylacrylate (PMEA)-coating on CPB circuits. Scandinavian Cardiovascular Journal. 2006;40(4):224–229. doi: 10.1080/14017430600833124. [DOI] [PubMed] [Google Scholar]
- 23.Shann K. G., Likosky D. S., Murkin J. M., et al. An evidence-based review of the practice of cardiopulmonary bypass in adults: a focus on neurologic injury, glycemic control, hemodilution, and the inflammatory response. The Journal of Thoracic and Cardiovascular Surgery. 2006;132(2):283–290.e3. doi: 10.1016/j.jtcvs.2006.03.027. [DOI] [PubMed] [Google Scholar]
- 24.Lazibat I., Sutlić Ž., Brkić K., Nevajda B., Šikić J., Meštrović A. H. Predictors of short-term neurocognitive outcome following coronary revascularisation (CABG) depending on the use of cardiopulmonary bypass. Collegium Antropologicum. 2012;36(3):827–833. [PubMed] [Google Scholar]
- 25.Bayram H., Hidiroglu M., Cetin L., et al. Comparing S-100 beta protein levels and neurocognitive functions between patients undergoing on-pump and off-pump coronary artery bypass grafting. Journal of Surgical Research. 2013;182(2):198–202. doi: 10.1016/j.jss.2012.10.047. [DOI] [PubMed] [Google Scholar]
- 26.Westaby S., Saatvedt K., White S., Katsumata T., van Oeveren W., Halligan P. W. Is there a relationship between cognitive dysfunction and systemic inflammatory response after cardiopulmonary bypass? Annals of Thoracic Surgery. 2001;71(2):667–672. doi: 10.1016/s0003-4975(00)02405-x. [DOI] [PubMed] [Google Scholar]
- 27.Carnevale D., Mascio G., Ajmone-Cat M. A., et al. Role of neuroinflammation in hypertension-induced brain amyloid pathology. Neurobiology of Aging. 2012;33(1):205.e19–205.e29. doi: 10.1016/j.neurobiolaging.2010.08.013. [DOI] [PubMed] [Google Scholar]
- 28.Mitchell S., Gorman D. The pathophysiology of cerebral arterial gas embolism. The Journal of Extra-Corporeal Technology. 2002;34(1):18–23. [PubMed] [Google Scholar]
- 29.Wang D., Wu X., Li J., Xiao F., Liu X., Meng M. The effect of lidocaine on early postoperative cognitive dysfunction after coronary artery bypass surgery. Anesthesia and Analgesia. 2002;95(5):1134–1141. doi: 10.1097/00000539-200211000-00002. [DOI] [PubMed] [Google Scholar]
- 30.Svensson L. G., Nadolny E. M., Penney D. L., et al. Prospective randomized neurocognitive and S-100 study of hypothermic circulatory arrest, retrograde brain perfusion, and antegrade brain perfusion for aortic arch operations. Annals of Thoracic Surgery. 2001;71(6):1905–1912. doi: 10.1016/s0003-4975(01)02570-x. [DOI] [PubMed] [Google Scholar]
- 31.Mathew J. P., Mackensen G. B., Phillips-Bute B., et al. Randomized, double-blinded, placebo controlled study of neuroprotection with lidocaine in cardiac surgery. Stroke. 2009;40(3):880–887. doi: 10.1161/strokeaha.108.531236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Mitchell S. J., Merry A. F., Frampton C., et al. Cerebral protection by lidocaine during cardiac operations: a follow-up study. Annals of Thoracic Surgery. 2009;87(3):820–825. doi: 10.1016/j.athoracsur.2008.12.042. [DOI] [PubMed] [Google Scholar]
- 33.Boodhwani M., Rubens F., Wozny D., Rodriguez R., Nathan H. J. Effects of sustained mild hypothermia on neurocognitive function after coronary artery bypass surgery: a randomized, double-blind study. The Journal of Thoracic and Cardiovascular Surgery. 2007;134(6):1443–1452.e1. doi: 10.1016/j.jtcvs.2007.08.016. [DOI] [PubMed] [Google Scholar]
- 34.Newman M. F., Mathew J. P., Grocott H. P., et al. Central nervous system injury associated with cardiac surgery. The Lancet. 2006;368(9536):694–703. doi: 10.1016/s0140-6736(06)69254-4. [DOI] [PubMed] [Google Scholar]
- 35.McKhann G. M., Grega M. A., Borowicz L. M., Jr., Baumgartner W. A., Selnes O. A. Stroke and encephalopathy after cardiac surgery: an update. Stroke. 2006;37(2):562–571. doi: 10.1161/01.str.0000199032.78782.6c. [DOI] [PubMed] [Google Scholar]
- 36.Grocott H. P. Pharmacologic neuroprotection: the search continues. Journal of Extra-Corporeal Technology. 2007;39(4):296–301. [PMC free article] [PubMed] [Google Scholar]
- 37.Dumas A., Dupuis G. H., Searle N., Cartier R. Early versus late extubation after coronary artery bypass grafting: effects on cognitive function. Journal of Cardiothoracic and Vascular Anesthesia. 1999;13(2):130–135. doi: 10.1016/s1053-0770(99)90074-5. [DOI] [PubMed] [Google Scholar]
- 38.Dowd N. P., Karski J. M., Cheng D. C., et al. Fast-track cardiac anaesthesia in the elderly: effect of two different anaesthetic techniques on mental recovery. British Journal of Anaesthesia. 2001;86(1):68–76. doi: 10.1093/bja/86.1.68. [DOI] [PubMed] [Google Scholar]
- 39.Bottio T., Bisleri G., Piccoli P., Negri A., Manzato A., Muneretto C. Heart valve surgery in a very high-risk population: a preliminary experience in awake patients. Journal of Heart Valve Disease. 2007;16(2):187–194. [PubMed] [Google Scholar]
- 40.Delphin E., Jackson D., Gubenko Y., et al. Sevoflurane provides earlier tracheal extubation and assessment of cognitive recovery than isoflurane in patients undergoing off-pump coronary artery bypass surgery. Journal of Cardiothoracic and Vascular Anesthesia. 2007;21(5):690–695. doi: 10.1053/j.jvca.2006.12.008. [DOI] [PubMed] [Google Scholar]
- 41.Kanbak M., Saricaoglu F., Akinci S. B., et al. The effects of isoflurane, sevoflurane, and desflurane anesthesia on neurocognitive outcome after cardiac surgery: a pilot study. The Heart Surgery Forum. 2007;10(1):E36–E41. doi: 10.1532/hsf98.20061076. [DOI] [PubMed] [Google Scholar]
- 42.Hudetz J. A., Iqbal Z., Gandhi S. D., et al. Ketamine attenuates post-operative cognitive dysfunction after cardiac surgery. Acta Anaesthesiologica Scandinavica. 2009;53(7):864–872. doi: 10.1111/j.1399-6576.2009.01978.x. [DOI] [PubMed] [Google Scholar]
- 43.Schoen J., Husemann L., Tiemeyer C., et al. Cognitive function after sevoflurane- vs propofol-based anaesthesia for on-pump cardiac surgery: a randomized controlled trial. British Journal of Anaesthesia. 2011;106(6):840–850. doi: 10.1093/bja/aer091. [DOI] [PubMed] [Google Scholar]
- 44.Kadoi Y., Saito S., Kunimoto F., Goto F., Fujita N. Comparative effects of propofol versus fentanyl on cerebral oxygenation state during normothermic cardiopulmonary bypass and postoperative cognitive dysfunction. Annals of Thoracic Surgery. 2003;75(3):840–846. doi: 10.1016/S0003-4975(02)04498-3. [DOI] [PubMed] [Google Scholar]
- 45.Silbert B. S., Scott D. A., Evered L. A., et al. A comparison of the effect of high- and low-dose fentanyl on the incidence of postoperative cognitive dysfunction after coronary artery bypass surgery in the elderly. Anesthesiology. 2006;104(6):1137–1145. doi: 10.1097/00000542-200606000-00007. [DOI] [PubMed] [Google Scholar]
- 46.Kadoi Y., Goto F. Sevoflurane anesthesia did not affect postoperative cognitive dysfunction in patients undergoing coronary artery bypass graft surgery. Journal of Anesthesia. 2007;21(3):330–335. doi: 10.1007/s00540-007-0537-7. [DOI] [PubMed] [Google Scholar]
- 47.Lehmann A., Schmidt M., Zeitler C., Kiessling A.-H., Isgro F., Boldt J. Bispectral index and electroencephalographic entropy in patients undergoing aortocoronary bypass grafting. European Journal of Anaesthesiology. 2007;24(9):751–760. doi: 10.1017/S0265021506002249. [DOI] [PubMed] [Google Scholar]
- 48.Evered L., Scott D. A., Silbert B., Maruff P. Postoperative cognitive dysfunction is independent of type of surgery and anesthetic. Anesthesia and Analgesia. 2011;112(5):1179–1185. doi: 10.1213/ANE.0b013e318215217e. [DOI] [PubMed] [Google Scholar]
- 49.Parra V. M., Sadurní M., Doñate M., et al. Neuropsychological dysfunction after cardiac surgery: cerebral saturation and bispectral index: a longitudinal study. Revista Medica de Chile. 2011;139(12):1553–1561. doi: 10.4067/s0034-98872011001200004. [DOI] [PubMed] [Google Scholar]
- 50.Royse C. F., Andrews D. T., Newman S. N., et al. The influence of propofol or desflurane on postoperative cognitive dysfunction in patients undergoing coronary artery bypass surgery. Anaesthesia. 2011;66(6):455–464. doi: 10.1111/j.1365-2044.2011.06704.x. [DOI] [PubMed] [Google Scholar]
- 51.Gottesman R. F., Hillis A. E., Grega M. A., et al. Early postoperative cognitive dysfunction and blood pressure during coronary artery bypass graft operation. Archives of Neurology. 2007;64(8):1111–1114. doi: 10.1001/archneur.64.8.noc70028. [DOI] [PubMed] [Google Scholar]
- 52.Newman M. F., Kramer D., Croughwell N. D., et al. Differential age effects of mean arterial pressure and rewarming on cognitive dysfunction after cardiac surgery. Anesthesia and Analgesia. 1995;81(2):236–242. doi: 10.1097/00000539-199508000-00005. [DOI] [PubMed] [Google Scholar]
- 53.Patel R. L., Turtle M. R. J., Chambers D. J., Newman S., Venn G. E. Hyperperfusion and cerebral dysfunction. Effect of differing acid-base management during cardiopulmonary bypass. European Journal of Cardio-Thoracic Surgery. 1993;7(9):457–464. doi: 10.1016/1010-7940(93)90274-F. [DOI] [PubMed] [Google Scholar]
- 54.Patel R. L., Turtle M. R., Chambers D. J., James D. N., Newman S., Venn G. E. Alpha-stat acid-base regulation during cardiopulmonary bypass improves neuropsychologic outcome in patients undergoing coronary artery bypass grafting. Journal of Thoracic and Cardiovascular Surgery. 1996;111(6):1267–1279. doi: 10.1016/s0022-5223(96)70230-1. [DOI] [PubMed] [Google Scholar]
- 55.Govier A. V., Reves J. G., McKay R. D., et al. Factors and their influence on regional cerebral blood flow during nonpulsatile cardiopulmonary bypass. Annals of Thoracic Surgery. 1984;38(6):592–600. doi: 10.1016/s0003-4975(10)62316-8. [DOI] [PubMed] [Google Scholar]
- 56.Wimmer-Greinecker G., Matheis G., Brieden M., et al. Neuropsychological changes after cardiopulmonary bypass for coronary artery bypass grafting. Thoracic and Cardiovascular Surgeon. 1998;46(4):207–212. doi: 10.1055/s-2007-1010226. [DOI] [PubMed] [Google Scholar]
- 57.Jönsson H., Johnsson P., Ailing C., Bäckström M., Bergh C., Blomquist S. S100beta after coronary artery surgery: release pattern, source of contamination, and relation to neuropsychological outcome. Annals of Thoracic Surgery. 1999;68(6):2202–2208. doi: 10.1016/s0003-4975(99)00851-6. [DOI] [PubMed] [Google Scholar]
- 58.Kilminster S., Treasure T., McMillan T., Holt D. W. Neuropsychological change and S-100 protein release in 130 unselected patients undergoing cardiac surgery. Stroke. 1999;30(9):1869–1874. doi: 10.1161/01.str.30.9.1869. [DOI] [PubMed] [Google Scholar]
- 59.Rasmussen L. S., Christiansen M., Hansen P. B., Moller J. T. Do blood levels of neuron-specific enolase and S-100 protein reflect cognitive dysfunction after coronary artery bypass? Acta Anaesthesiologica Scandinavica. 1999;43(5):495–500. doi: 10.1034/j.1399-6576.1999.430502.x. [DOI] [PubMed] [Google Scholar]
- 60.Derkach D. N., Okamoto H., Takahashi S. Neuronal and astroglial injuries in patients undergoing coronary artery bypass grafting and aortic arch replacement during hypothermic cardiopulmonary bypass. Anesthesia and Analgesia. 2000;91(5):1066–1072. doi: 10.1097/00000539-200011000-00005. [DOI] [PubMed] [Google Scholar]
- 61.Diegeler A., Hirsch R., Schneider F., et al. Neuromonitoring and neurocognitive outcome in off-pump versus conventional coronary bypass operation. Annals of Thoracic Surgery. 2000;69(4):1162–1166. doi: 10.1016/s0003-4975(99)01574-x. [DOI] [PubMed] [Google Scholar]
- 62.Georgiadis D., Berger A., Kowatschev E., et al. Predictive value of S-100beta and neuron-specific enolase serum levels for adverse neurologic outcome after cardiac surgery. Journal of Thoracic and Cardiovascular Surgery. 2000;119(1):138–147. doi: 10.1016/s0022-5223(00)70229-7. [DOI] [PubMed] [Google Scholar]
- 63.Lloyd C. T., Ascione R., Underwood M. J., Gardner F., Black A., Angelini G. D. Serum S-100 protein release and neuropsychologic outcome during coronary revascularization on the beating heart: a prospective randomized study. The Journal of Thoracic and Cardiovascular Surgery. 2000;119(1):148–154. doi: 10.1016/s0022-5223(00)70230-3. [DOI] [PubMed] [Google Scholar]
- 64.Basile A. M., Fusi C., Conti A. A., et al. S-100 protein and neuron-specific enolase as markers of subclinical cerebral damage after cardiac surgery: preliminary observation of a 6-month follow-up study. European Neurology. 2001;45(3):151–159. doi: 10.1159/000052114. [DOI] [PubMed] [Google Scholar]
- 65.Rasmussen L. S., Christiansen M., Eliasen K., Sander-Jensen K., Moller J. T. Biochemical markers for brain damage after cardiac surgery—time profile and correlation with cognitive dysfunction. Acta Anaesthesiologica Scandinavica. 2002;46(5):547–551. doi: 10.1034/j.1399-6576.2002.460512.x. [DOI] [PubMed] [Google Scholar]
- 66.Farsak B., Gunaydin S., Yorgancioglu C., Zorlutuna Y. Elevated levels of s-100β correlate with neurocognitive outcome after cardiac surgery. Journal of Cardiovascular Surgery. 2003;44(1):31–35. [PubMed] [Google Scholar]
- 67.Mathew J. P., Grocott H. P., Phillips-Bute B., et al. Lower endotoxin immunity predicts increased cognitive dysfunction in elderly patients after cardiac surgery. Stroke. 2003;34(2):508–513. doi: 10.1161/01.str.0000053844.09493.58. [DOI] [PubMed] [Google Scholar]
- 68.Jönsson H., Johnsson P., Bäckström M., Alling C., Dautovic-Bergh C., Blomquist S. Controversial significance of early S100B levels after cardiac surgery. BMC Neurology. 2004;4, article 24 doi: 10.1186/1471-2377-4-24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Kofke W. A., Konitzer P., Meng Q. C., Guo J., Cheung A. The effect of apolipoprotein E genotype on neuron specific enolase and S-100β levels after cardiac surgery. Anesthesia & Analgesia. 2004;99(5):1323–1325. doi: 10.1213/01.ane.0000135345.03635.6a. [DOI] [PubMed] [Google Scholar]
- 70.Snyder-Ramos S. A., Gruhlke T., Bauer H., et al. Cerebral and extracerebral release of protein S100B in cardiac surgical patients. Anaesthesia. 2004;59(4):344–349. doi: 10.1111/j.1365-2044.2004.03663.x. [DOI] [PubMed] [Google Scholar]
- 71.Kálmán J., Juhász A., Bogáts G., et al. Elevated levels of inflammatory biomarkers in the cerebrospinal fluid after coronary artery bypass surgery are predictors of cognitive decline. Neurochemistry International. 2006;48(3):177–180. doi: 10.1016/j.neuint.2005.10.007. [DOI] [PubMed] [Google Scholar]
- 72.Ramlawi B., Rudolph J. L., Mieno S., et al. C-Reactive protein and inflammatory response associated to neurocognitive decline following cardiac surgery. Surgery. 2006;140(2):221–226. doi: 10.1016/j.surg.2006.03.007. [DOI] [PubMed] [Google Scholar]
- 73.Mathew J. P., Grocott H. P., McCurdy J. R., II, et al. Preoperative statin therapy does not reduce cognitive dysfunction after cardiopulmonary bypass. Journal of Cardiothoracic and Vascular Anesthesia. 2005;19(3):294–299. doi: 10.1053/j.jvca.2005.03.004. [DOI] [PubMed] [Google Scholar]
- 74.Plaschke K., Hauth S., Jansen C., et al. The influence of preoperative serum anticholinergic activity and other risk factors for the development of postoperative cognitive dysfunction after cardiac surgery. Journal of Thoracic and Cardiovascular Surgery. 2013;145(3):805–811. doi: 10.1016/j.jtcvs.2012.07.043. [DOI] [PubMed] [Google Scholar]
- 75.Grieco G., d'Hollosy M., Culliford A. T., Jonas S. Evaluating neuroprotective agents for clinical anti-ischemic benefit using neurological and neuropsychological changes after cardiac surgery under cardiopulmonary bypass. Methodological strategies and results of a double-blind, placebo-controlled trial of GM1 ganglioside. Stroke. 1996;27(5):858–874. doi: 10.1161/01.str.27.5.858. [DOI] [PubMed] [Google Scholar]
- 76.Arrowsmith J. E., Harrison M. J. G., Newman S. P., Stygall J., Timberlake N., Pugsley W. B. Neuroprotection of the brain during cardiopulmonary bypass: a randomized trial of remacemide during coronary artery bypass in 171 patients. Stroke. 1998;29(11):2357–2362. doi: 10.1161/01.str.29.11.2357. [DOI] [PubMed] [Google Scholar]
- 77.Svensson L. G., Nadolny E. M., Kimmel W. A. Multimodal protocol influence on stroke and neurocognitive deficit prevention after ascending/arch aortic operations. Annals of Thoracic Surgery. 2002;74(6):2040–2046. doi: 10.1016/s0003-4975(02)04023-7. [DOI] [PubMed] [Google Scholar]
- 78.Uebelhack R., Vohs K., Zytowski M., Schewe H.-J., Koch C., Konertz W. Effect of piracetam on cognitive performance in patients undergoing bypass surgery. Pharmacopsychiatry. 2003;36(3):89–93. doi: 10.1055/s-2003-39981. [DOI] [PubMed] [Google Scholar]
- 79.Szalma I., Kiss Á., Kardos L., et al. Piracetam prevents cognitive decline in coronary artery bypass: a randomized trial versus placebo. Annals of Thoracic Surgery. 2006;82(4):1430–1435. doi: 10.1016/j.athoracsur.2006.05.005. [DOI] [PubMed] [Google Scholar]
- 80.Haljan G., Maitland A., Buchan A., et al. The erythropoietin neuroprotective effect: assessment in CABG surgery (TENPEAKS): a randomized, double-blind, placebo controlled, proof-of-concept clinical trial. Stroke. 2009;40(8):2769–2775. doi: 10.1161/strokeaha.109.549436. [DOI] [PubMed] [Google Scholar]
- 81.Zhang Z., Ma P., Xu Y., et al. Preventive effect of gastrodin on cognitive decline after cardiac surgery with cardiopulmonary bypass: a double-blind, randomized controlled study. Journal of Huazhong University of Science and Technology: Medical Science. 2011;31(1):120–127. doi: 10.1007/s11596-011-0162-4. [DOI] [PubMed] [Google Scholar]
- 82.Kong R. S., Butterworth J., Aveling W., et al. Clinical trial of the neuroprotectant clomethiazole in coronary artery bypass graft surgery: a randomized controlled trial. Anesthesiology. 2002;97(3):585–591. doi: 10.1097/00000542-200209000-00011. [DOI] [PubMed] [Google Scholar]
- 83.Taggart D. P., Browne S. M., Wade D. T., Halligan P. W. Neuroprotection during cardiac surgery: a randomised trial of a platelet activating factor antagonist. Heart. 2003;89(8):897–900. doi: 10.1136/heart.89.8.897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Mathew J. P., Fontes M. L., Tudor I. C., et al. A multicenter risk index for atrial fibrillation after cardiac surgery. The Journal of the American Medical Association. 2004;291(14):1720–1729. doi: 10.1001/jama.291.14.1720. [DOI] [PubMed] [Google Scholar]
- 85.Hogue C. W., Jr., Freedland K., Hershey T., et al. Neurocognitive outcomes are not improved by 17β-estradiol in postmenopausal women undergoing cardiac surgery. Stroke. 2007;38(7):2048–2054. doi: 10.1161/strokeaha.106.480426. [DOI] [PubMed] [Google Scholar]
- 86.Holinski S., Claus B., Alaaraj N., et al. Cerebroprotective effect of piracetam in patients undergoing open heart surgery. Annals of Thoracic and Cardiovascular Surgery. 2011;17(2):137–142. doi: 10.5761/atcs.oa.10.01545. [DOI] [PubMed] [Google Scholar]
- 87.Grimm M., Czerny M., Baumer H., et al. Normothermic cardiopulmonary bypass is beneficial for cognitive brain function after coronary artery bypass grafting—a prospective randomized trial. European Journal of Cardio-Thoracic Surgery. 2000;18(3):270–275. doi: 10.1016/s1010-7940(00)00510-8. [DOI] [PubMed] [Google Scholar]
- 88.Shaaban-Ali M., Harmer M., Vaughan R. S., et al. Changes in serum S100β protein and mini-mental state examination after cold (28°C) and warm (34°C) cardiopulmonary bypass using different blood gas strategies (alpha-stat and pH-stat) Acta Anaesthesiologica Scandinavica. 2002;46(1):10–16. doi: 10.1046/j.0001-5172.2001.00000.x. [DOI] [PubMed] [Google Scholar]
- 89.Nathan H. J., Munson J., Wells G., Mundi C., Balaa F., Wynands J. E. The management of temperature during cardiopulmonary bypass: effect on neuropsychological outcome. Journal of Cardiac Surgery. 1995;10(4):481–487. doi: 10.1111/j.1540-8191.1995.tb00681.x. [DOI] [PubMed] [Google Scholar]
- 90.Grocott H. P., Mackensen G. B., Grigore A. M., et al. Postoperative hyperthermia is associated with cognitive dysfunction after coronary artery bypass graft surgery. Stroke. 2002;33(2):537–541. doi: 10.1161/hs0202.102600. [DOI] [PubMed] [Google Scholar]
- 91.Kadoi Y., Saito S., Takahashi K.-I., Fujita N., Goto F. Jugular venous oxygen saturation during mild hypothermic versus normothermic cardiopulmonary bypass in elderly patients. Surgery Today. 2004;34(5):399–404. doi: 10.1007/s00595-003-2736-7. [DOI] [PubMed] [Google Scholar]
- 92.Boodhwani M., Rubens F. D., Wozny D., et al. Predictors of early neurocognitive deficits in low-risk patients undergoing on-pump coronary artery bypass surgery. Circulation. 2006;114(1):I461–I466. doi: 10.1161/CIRCULATIONAHA.105.001354. [DOI] [PubMed] [Google Scholar]
- 93.Hiraoka K., Kawatsu S., Mori E., Saiki Y. Total aortic arch replacement using hypothermic circulatory arrest with antegrade selective cerebral perfusion: are there cerebral deficits other than frank stroke? General Thoracic and Cardiovascular Surgery. 2012;60(6):345–349. doi: 10.1007/s11748-012-0057-8. [DOI] [PubMed] [Google Scholar]
- 94.McLean R. F., Wong B. I., Naylor C. D., et al. Cardiopulmonary bypass, temperature, and central nervous system dysfunction. Circulation. 1994;90(5):II250–II255. [PubMed] [Google Scholar]
- 95.Regragui I., Birdi I., Izzat M. B., et al. The effects of cardiopulmonary bypass temperature on neuropsychologic outcome after coronary artery operations: a prospective randomized trial. The Journal of Thoracic and Cardiovascular Surgery. 1996;112(4):1036–1045. doi: 10.1016/s0022-5223(96)70105-8. [DOI] [PubMed] [Google Scholar]
- 96.Heyer E. J., Adams D. C., Delphin E., et al. Cerebral dysfunction after coronary artery bypass grafting done with mild or moderate hypothermia. Journal of Thoracic and Cardiovascular Surgery. 1997;114(2):270–277. doi: 10.1016/S0022-5223(97)70155-7. [DOI] [PubMed] [Google Scholar]
- 97.Kneebone A. C., Andrew M. J., Baker R. A., Knight J. L. Neuropsychologic changes after coronary artery bypass grafting: use of reliable change indices. Annals of Thoracic Surgery. 1998;65(5):1320–1325. doi: 10.1016/s0003-4975(98)00158-1. [DOI] [PubMed] [Google Scholar]
- 98.Reich D. L., Uysal S., Sliwinski M., et al. Neuropsychologic outcome after deep hypothermic circulatory arrest in adults. The Journal of Thoracic and Cardiovascular Surgery. 1999;117(1):156–163. doi: 10.1016/s0022-5223(99)70481-2. [DOI] [PubMed] [Google Scholar]
- 99.Kaukinen L., Porkkala H., Kaukinen S., et al. Release of brain-specific creatine kinase and neuron-specific enolase into cerebrospinal fluid after hypothermic and normothermic cardiopulmonary bypass in coronary artery surgery. Acta Anaesthesiologica Scandinavica. 2000;44(4):361–368. doi: 10.1034/j.1399-6576.2000.440402.x. [DOI] [PubMed] [Google Scholar]
- 100.Górna R., Kustrzycki W., Kiejna A., Rymaszewska J. Assessment of short-term neuropsychologic changes after normothermic versus hypothermic coronary artery bypass grafting. Psychiatria Polska. 2001;35(5):781–795. [PubMed] [Google Scholar]
- 101.Grigore A. M., Mathew J., Grocott H. P., et al. Prospective randomized trial of normothermic versus hypothermic cardiopulmonary bypass on cognitive function after coronary artery bypass graft surgery. Anesthesiology. 2001;95(5):1110–1119. doi: 10.1097/00000542-200111000-00014. [DOI] [PubMed] [Google Scholar]
- 102.Kaukuntla H., Walker A., Harrington D., Jones T., Bonser R. S. Differential brain and body temperature during cardiopulmonary bypass—a randomised clinical study. European Journal of Cardio-thoracic Surgery. 2004;26(3):571–579. doi: 10.1016/j.ejcts.2004.05.042. [DOI] [PubMed] [Google Scholar]
- 103.Reich D. L., Horn L. M., Hossain S., Uysal S. Using jugular bulb oxyhemoglobin saturation to guide onset of deep hypothermic circulatory arrest does not affect post-operative neuropsychological function. European Journal of Cardio-Thoracic Surgery. 2004;25(3):401–408. doi: 10.1016/j.ejcts.2003.11.041. [DOI] [PubMed] [Google Scholar]
- 104.Kunihara T., Tscholl D., Langer F., Heinz G., Sata F., Schäfers H.-J. Cognitive brain function after hypothermic circulatory arrest assessed by cognitive P300 evoked potentials. European Journal of Cardio-Thoracic Surgery. 2007;32(3):507–513. doi: 10.1016/j.ejcts.2007.06.006. [DOI] [PubMed] [Google Scholar]
- 105.Mora C. T., Henson M. B., Weintraub W. S., et al. The effect of temperature management during cardiopulmonary bypass on neurologic and neuropsychologic outcomes in patients undergoing coronary revascularization. The Journal of Thoracic and Cardiovascular Surgery. 1996;112(2):514–522. doi: 10.1016/s0022-5223(96)70280-5. [DOI] [PubMed] [Google Scholar]
- 106.Nathan H. J., Wells G. A., Munson J. L., Wozny D. Neuroprotective effect of mild hypothermia in patients undergoing coronary artery surgery with cardiopulmonary bypass: a randomized trial. Circulation. 2001;104(1):i85–i91. doi: 10.1161/hc37t1.094710. [DOI] [PubMed] [Google Scholar]
- 107.Grigore A. M., Grocott H. P., Mathew J. P., et al. The rewarming rate and increased peak temperature alter neurocognitive outcome after cardiac surgery. Anesthesia & Analgesia. 2002;94(1):4–10. doi: 10.1097/00000539-200201000-00002. [DOI] [PubMed] [Google Scholar]
- 108.Kawahara F., Kadoi Y., Saito S., Goto F., Fujita N. Slow rewarming improves jugular venous oxygen saturation during rewarming. Acta Anaesthesiologica Scandinavica. 2003;47(4):419–424. doi: 10.1034/j.1399-6576.2003.00063.x. [DOI] [PubMed] [Google Scholar]
- 109.Nathan H. J., Rodriguez R., Wozny D., et al. Neuroprotective effect of mild hypothermia in patients undergoing coronary artery surgery with cardiopulmonary bypass: five-year follow-up of a randomized trial. The Journal of Thoracic and Cardiovascular Surgery. 2007;133(5):1206–1211. doi: 10.1016/j.jtcvs.2006.09.112. [DOI] [PubMed] [Google Scholar]
- 110.Sahu B., Chauhan S., Kiran U., et al. Neurocognitive function in patients undergoing coronary artery bypass graft surgery with cardiopulmonary bypass: the effect of two different rewarming strategies. Journal of Cardiothoracic and Vascular Anesthesia. 2009;23(1):14–21. doi: 10.1053/j.jvca.2008.07.010. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
To help with the commentary of the systematic review and ensure the transparent and complete reporting, a PRISMA chart has been attached to view the full selection process of the studies included. The diagram depicts the flow of information through different phases of the systematic review, mapping out the number of records identified, included and excluded.


