Abstract
Objective
Birth defects are a leading cause of infant mortality in the United States. Previous reports have highlighted black-white differences in overall infant mortality and infant mortality attributable to birth defects (IMBD). We evaluated the impact of gestational age on US racial/ethnic differences in IMBD.
Methods
We estimated the rate of IMBD (using ICD-10 codes for the underlying cause of death) using the period linked birth/infant death data for US residents for January 2003 to December 2006. We excluded infants with missing gestational age, implausible values based on Alexander’s index of birth weight for gestational age norms, or gestational ages <20 weeks or >44 weeks; we categorized gestational age into three groups: 20–33; 34–36; and 37–44 weeks. Using Poisson regression, we compared neonatal and postneonatal mortality attributable to birth defects for infants of non-Hispanic black and Hispanic mothers with that for infants of non-Hispanic white mothers stratified by gestational age.
Results
IMBD occurred in 12.2 per 10,000 live births. Among infants delivered at 37–44 weeks, blacks (and Hispanics, to a lesser degree) had significantly higher neonatal and postneonatal mortality attributable to birth defects than whites. However, among infants delivered at 20–33 or 34–36 weeks, neonatal (but not postneonatal) mortality attributable to birth defects was significantly lower among blacks compared with whites.
Conclusions
Racial/ethnic differences in IMBD were not explained in these data by differences in gestational age. Further investigation should include an assessment of possible racial/ethnic differences in severity and/or access to timely diagnosis and management of birth defects.
Keywords: infant mortality, birth defects, gestational age, race, ethnicity
Introduction
Birth defects affect approximately 3% of births in the United States1 and are responsible for 1 in 5 infant deaths.2, 3 The prevalence of preterm birth (birth at <37 completed weeks gestation) has steadily increased in recent years, and preterm birth is three times as common among non-Hispanic black compared with non-Hispanic white mothers.4 Black-white disparities in infant mortality attributable to birth defects (IMBD) have also been reported.5 US studies have shown that all-cause neonatal mortality rates are higher among term infants of black mothers compared with white mothers, but lower among preterm infants of black mothers compared with white mothers.6, 7 A recent Centers for Disease Control and Prevention (CDC) study examined differences in neonatal mortality rates due to congenital heart defects (the leading cause of IMBD) by race and gestational age.8 Another recent study focused on heterogeneous risk for infant mortality across the range of term births.9 The current study sought to expand these analyses by examining differences in both neonatal and postneonatal mortality attributable to all birth defects by race/ethnicity and gestational age.
Methods
Data source
We used the CDC National Center for Health Statistics’ period linked birth/infant death data for US residents, including live births between January 2003 and December 2006, which represented the most recent available at the time of analysis.10
Inclusion/exclusion criteria
We included records of all infants (under age 1 year) whose underlying cause of death on the death certificate was classified as a birth defect according to the International Classification of Diseases, 10th Revision, with codes Q01.0–Q99.9, excluding conditions that are considered normal conditions of prematurity (Q21.1: persistent foramen ovale, Q25.0: patent ductus arteriosus, Q33.6: lung hypoplasia, and Q53.1, 53.2, 53.9: undescended testicles) or cardiovascular conditions that were not considered structural heart defects (Q27.0–Q28.9). We restricted the analysis to infants of mothers from the three largest race/ethnicity categories – non-Hispanic white, non-Hispanic black, and Hispanic – as reported on the US birth certificate; those of other racial/ethnic groups were excluded, as were births to and infant deaths of non-US residents. All analyses were based on maternal race/ethnicity.
The gestational age measure from the birth certificate was based on a clinical estimate of elapsed time since the last menstrual period. We categorized gestational age into three groups: 20–33; 34–36; and 37–44 weeks. We excluded linked records with a missing gestational age (0.6% of the total), those with implausible gestational ages based on Alexander’s index of birth weight for gestational age norms (0.6%),6 and those with gestational ages <20 weeks or >44 weeks (1.1%).
Analytic methods
We estimated rates of neonatal (age <28 days) and postneonatal (age 28 days to under 1 year) mortality attributable to birth defects overall, and stratified by race/ethnicity and gestational age categories. We also tabulated mortality rates by individual weeks of gestational age, separately by race/ethnicity.
Because not all infant death records could be linked to the corresponding birth certificate, we weighted estimates of infant deaths according to the percentage of records linked by state and age at death. Weighting was necessary to correct in part for biases in the percentage of records linked by these characteristics -- in general, a slightly lower percentage of neonatal than postneonatal deaths was linked for these years (e.g., 98.5 vs. 99.1% for 2005).10
We used Poisson regression to estimate rate ratios (RRs) comparing neonatal and postneonatal mortality rates for infants of non-Hispanic black and Hispanic mothers with those of non-Hispanic white mothers, stratified by the three gestational age categories. Variables available on the birth certificate that we considered as potential confounders included maternal age, education, parity (number of live births), plurality, marital status, diabetes, and infant sex. These were selected based on previous literature or biologic plausibility suggesting their association with either neonatal or postneonatal mortality attributable to birth defects. Besides maternal age and education, other variables did not change the effect estimate by greater than 5% in bivariate analysis and were not included in our final models. We examined descriptive data on ‘medical complications’ and ‘labor and delivery complications’ but did not include these in analyses due to the low quality and incompleteness of these variables;11, 12 we did not adjust for birth weight due to its strong correlation with gestational age.
With the revision of the US birth certificate in 2003 and its variable introduction by states, information on maternal education was not comparable for all states with each year of linked data --- the number of states with non-comparable data for maternal education ranged from 2 US states in 2003 to 19 states and Puerto Rico in 2006 (Tables 1 & 3). Therefore, only records with maternal age and comparable data on maternal education were included in adjusted analyses. We also calculated unadjusted gestational-week specific neonatal and postneonatal mortality rate ratios.
Table 1.
Maternal and infant characteristics among neonatal deaths attributable to birth defects by race/ethnicity, United States, 2003–2006.
| Non-Hispanic White | Non-Hispanic Black | Hispanic | |||||||
|---|---|---|---|---|---|---|---|---|---|
|
| |||||||||
| Characteristic | Neonatal deaths attributable to birth defects (n) | Live births (n) | Mortality per 10,000 live births | Neonatal deaths attributable to birth defects (n) | Live births (n) | Mortality per 10,000 live births | Neonatal deaths attributable to birth defects (n) | Live births (n) | Mortality per 10,000 live births |
|
| |||||||||
| Total number | 7,090 | 9,071,585 | 7.8 | 2,114 | 2,312,080 | 9.1 | 3,250 | 3,761,379 | 8.6 |
|
| |||||||||
| Maternal age | |||||||||
| < 20 yrs | 590 | 666,737 | 8.9 | 344 | 397,112 | 8.7 | 415 | 535,193 | 7.8 |
| 20–34 yrs | 5,058 | 6,927,295 | 7.3 | 1,390 | 1,677,422 | 8.3 | 2,276 | 2,828,798 | 8.1 |
| > 34 yrs | 1,442 | 1,477,553 | 9.8 | 380 | 237,546 | 16 | 559 | 397,388 | 14.1 |
|
| |||||||||
| Number of previous live births | |||||||||
| 1 | 2,511 | 3,748,145 | 6.7 | 703 | 880,649 | 9.0 | 992 | 1,351,517 | 7.3 |
| 2–3 | 3,505 | 4,482,853 | 7.8 | 986 | 1,068,926 | 9.2 | 1,597 | 1,880,119 | 8.5 |
| > 3 | 989 | 799,493 | 12.4 | 395 | 343,241 | 11.5 | 633 | 516,116 | 12.3 |
| Missing | 85 | 41,094 | 20.7 | 30 | 19,264 | 15.6 | 27 | 13,627 | 19.8 |
|
| |||||||||
| Married | |||||||||
| Yes | 5,176 | 6,819,419 | 7.6 | 672 | 704,742 | 9.5 | 1,832 | 1,983,284 | 9.2 |
| No | 1,914 | 2,252,166 | 8.5 | 1,442 | 1,607,338 | 9.0 | 1,418 | 1,778,095 | 8.0 |
|
| |||||||||
| Education of Mother** | |||||||||
| <12 years | 638 | 718,851 | 8.9 | 360 | 401,449 | 9.0 | 1,118 | 1,243,939 | 9.0 |
| 12 years | 4,393 | 5,845,041 | 7.5 | 1146 | 1,331,479 | 8.6 | 1,024 | 1,369,180 | 7.5 |
| Missing | 207 | 98,907 | 20.9 | 90 | 33,929 | 26.5 | 101 | 53,347 | 18.9 |
| Exclude*** | 1,852 | 2,408,786 | 7.7 | 518 | 545,223 | 9.5 | 1007 | 1,094,913 | 9.2 |
|
| |||||||||
| Diabetes | |||||||||
| Yes | 273 | 321,749 | 8.5 | 113 | 78,842 | 14.3 | 174 | 139,928 | 12.4 |
| No | 6,744 | 8,716,520 | 7.7 | 1,986 | 2,224,794 | 8.9 | 3,055 | 3,610,343 | 8.5 |
| Missing | 73 | 33,316 | 21.9 | 15 | 8,444 | 17.8 | 20 | 11,108 | 18.0 |
|
| |||||||||
| Infant sex | |||||||||
| Male | 3,768 | 4,654,163 | 8.1 | 1,104 | 1,175,772 | 9.4 | 1,691 | 1,919,850 | 8.8 |
| Female | 3,322 | 4,417,422 | 7.5 | 1,010 | 1,136,308 | 8.9 | 1,559 | 1,841,529 | 8.5 |
|
| |||||||||
| Plurality | |||||||||
| Single | 6,485 | 8,724,539 | 7.4 | 1,956 | 2,227,016 | 8.8 | 3,091 | 3,676,458 | 8.4 |
| Multiple | 605 | 347,046 | 17.4 | 159 | 85,064 | 18.7 | 158 | 84,921 | 18.6 |
|
| |||||||||
| Infant birth weight (grams) | |||||||||
| < 1500 | 1,141 | 81,265 | 140.4 | 441 | 48,215 | 91.5 | 523 | 31,606 | 165.5 |
| 1500–2499 | 2,601 | 547,557 | 47.5 | 755 | 247,018 | 30.6 | 1,236 | 213,379 | 57.9 |
| ≥2500 | 2,391 | 8,415,792 | 2.8 | 639 | 1,992,639 | 3.2 | 1,171 | 3,504,139 | 3.3 |
| Missing | 958 | 26,971 | 355.2 | 280 | 24,208 | 115.7 | 320 | 12,255 | 261.1 |
|
| |||||||||
| Infant gestational age (weeks) | |||||||||
| 20 – 33 | 2,478 | 254,226 | 97.5 | 818 | 139,520 | 58.6 | 1,020 | 111,670 | 91.3 |
| 34 – 36 | 1,535 | 776,573 | 19.8 | 422 | 271,762 | 15.5 | 690 | 330,297 | 20.9 |
| 37 – 44 | 3,077 | 8,040,786 | 3.8 | 874 | 1,900,798 | 4.6 | 1,540 | 3,319,412 | 4.6 |
Data were based on the 1989 Revision of the U.S. Certificate of Live Birth.
Excluded data based on the 2003 Revision; states included Pennsylvania and Washington (2003–2006), Idaho, Kentucky, New York (except New York City), South Carolina, Tennessee, Florida, and New Hampshire (2004–2006), Kansas, Nebraska, Texas, Puerto Rico, Vermont (2005–2006), California (partial revision), Delaware, North Dakota, Ohio, South Dakota, and Wyoming (2006).
Table 3.
Maternal and infant characteristics among postneonatal deaths attributable to birth defects by race/ethnicity, United States, 2003–2006.
| Non-Hispanic White | Non-Hispanic Black | Hispanic | |||||||
|---|---|---|---|---|---|---|---|---|---|
|
| |||||||||
| Characteristic | Postneonatal deaths attributable to birth defects (n) | Live births (n) | Mortality per 10,000 live births | Postneonatal deaths attributable to birth defects (n) | Live births (n) | Mortality per 10,000 live births | Postneonatal deaths attributable to birth defects (n) | Live births (n) | Mortality per 10,000 live births |
|
| |||||||||
| Total number | 2,967 | 9,071,585 | 3.3 | 1,224 | 2,312,080 | 5.3 | 1,435 | 3,761,379 | 3.8 |
|
| |||||||||
| Maternal age | |||||||||
| < 20 yrs | 289 | 666,737 | 4.3 | 223 | 397,112 | 5.6 | 240 | 535,193 | 4.5 |
| 20–34 yrs | 2,146 | 6,927,295 | 3.1 | 794 | 1,677,422 | 4.7 | 991 | 2,828,798 | 3.5 |
| > 34 yrs | 531 | 1,477,553 | 3.6 | 207 | 237,546 | 8.7 | 204 | 397,388 | 5.1 |
|
| |||||||||
| Number of previous live births | |||||||||
| 1 | 1,103 | 3,748,145 | 2.9 | 398 | 880,649 | 4.5 | 470 | 1,351,517 | 3.5 |
| 2–3 | 1,494 | 4,482,853 | 3.3 | 582 | 1,068,926 | 5.4 | 722 | 1,880,119 | 3.8 |
| > 3 | 341 | 799,493 | 4.3 | 232 | 343,241 | 6.8 | 230 | 516,116 | 4.5 |
| Missing | 29 | 41,094 | 7.1 | 12 | 19,264 | 6.2 | 13 | 13,627 | 9.5 |
|
|
|||||||||
| Married | |||||||||
| Yes | 2,100 | 6,819,419 | 3.1 | 373 | 704,742 | 5.3 | 711 | 1,983,284 | 3.6 |
| No | 866 | 2,252,166 | 3.9 | 851 | 1,607,338 | 5.3 | 724 | 1,778,095 | 4.1 |
|
| |||||||||
| Education of Mother** | |||||||||
| <12 years | 351 | 718,851 | 4.9 | 242 | 401,449 | 6.0 | 557 | 1,243,939 | 4.5 |
| 12 years | 1,818 | 5,845,041 | 3.1 | 673 | 1,331,479 | 5.0 | 442 | 1,369,180 | 3.2 |
| Missing | 55 | 98,907 | 5.6 | 15 | 33,929 | 4.4 | 35 | 53,347 | 6.6 |
| Exclude*** | 743 | 2,408,786 | 3.1 | 294 | 545,223 | 5.4 | 401 | 1,094,913 | 3.7 |
|
| |||||||||
| Diabetes | |||||||||
| Yes | 149 | 321,749 | 4.6 | 79 | 78,842 | 10.0 | 74 | 139,928 | 5.3 |
| No | 2,792 | 8,716,520 | 3.2 | 1,137 | 2,224,794 | 5.1 | 1,356 | 3,610,343 | 3.8 |
| Missing | 25 | 33,316 | 7.5 | 8 | 8,444 | 9.5 | 5 | 11,108 | 4.5 |
|
| |||||||||
| Infant sex | |||||||||
| Male | 1,550 | 4,654,163 | 3.3 | 645 | 1,175,772 | 5.5 | 693 | 1,919,850 | 3.6 |
| Female | 1,417 | 4,417,422 | 3.2 | 579 | 1,136,308 | 5.1 | 742 | 1,841,529 | 4.0 |
|
| |||||||||
| Plurality | |||||||||
| Single | 2,789 | 8,724,539 | 3.2 | 1,152 | 2,227,016 | 5.2 | 1,373 | 3,676,458 | 3.7 |
| Multiple | 177 | 347,046 | 5.1 | 73 | 85,064 | 8.6 | 63 | 84,921 | 7.4 |
|
| |||||||||
| Infant birth weight (grams) | |||||||||
| < 1500 | 238 | 81,265 | 29.3 | 133 | 48,215 | 27.6 | 133 | 31,606 | 42.1 |
| 1500–2499 | 913 | 547,557 | 16.7 | 446 | 247,018 | 18.1 | 455 | 213,379 | 21.3 |
| ≥2500 | 1,760 | 8,415,792 | 2.1 | 612 | 1,992,639 | 3.1 | 824 | 3,504,139 | 2.4 |
| missing | 55 | 26,971 | 20.4 | 32 | 24,208 | 13.2 | 23 | 12,255 | 18.8 |
|
| |||||||||
| Infant gestational age (weeks) | |||||||||
| 20 – 33 | 399 | 254,226 | 15.7 | 212 | 139,520 | 15.2 | 212 | 111,670 | 19.0 |
| 34 – 36 | 545 | 776,573 | 7.0 | 249 | 271,762 | 9.2 | 271 | 330,297 | 8.2 |
| 37 – 44 | 2,023 | 8,040,786 | 2.5 | 763 | 1,900,798 | 4.0 | 952 | 3,319,412 | 2.9 |
Data were based on the 1989 Revision of the U.S. Certificate of Live Birth.
Excluded data based on the 2003 Revision; states included Pennsylvania and Washington (2003–2006), Idaho, Kentucky, New York (except New York City), South Carolina, Tennessee, Florida, and New Hampshire (2004–2006), Kansas, Nebraska, Texas, Puerto Rico, Vermont (2005–2006), California (partial revision), Delaware, North Dakota, Ohio, South Dakota, and Wyoming (2006).
We estimated infant mortality rates and RRs for all birth defects combined as well as for several major birth defect categories, as grouped by the ICD-10 classification scheme. For regression analyses, we included the cleft lip/cleft palate category with the digestive system defects, and we did not examine the “eye, ear, face, and neck” or “other” categories due to sparse numbers.
Results
Overall IMBD
The analysis included 15,145,044 live births in the United States during 2003–2006; 9,071,585 births to non-Hispanic white mothers, 2,312,080 births to non-Hispanic black mothers, and 3,761,379 births to Hispanic mothers. A total of 18,080 infant deaths had a birth defect listed as the underlying cause (18% of all infant deaths) after exclusion of the specific ICD codes specified above, and the overall rate of IMBD was 12.2 per 10,000 live births. Race-specific IMBD rates were 11.1, 14.4, and 12.5 per 10,000 live births for non-Hispanic white, non-Hispanic black, and Hispanic mothers, respectively. Congenital heart defects were noted as the underlying cause of death in 31% of IMBD, followed by chromosomal abnormalities, not elsewhere classified (22%), and central nervous system defects (15%), which include neural tube defects. The prevalence of preterm birth among live births was 11% for non-Hispanic white, 18% for non-Hispanic black, and 12% for Hispanic mothers.
Neonatal Mortality Attributable to Birth Defects
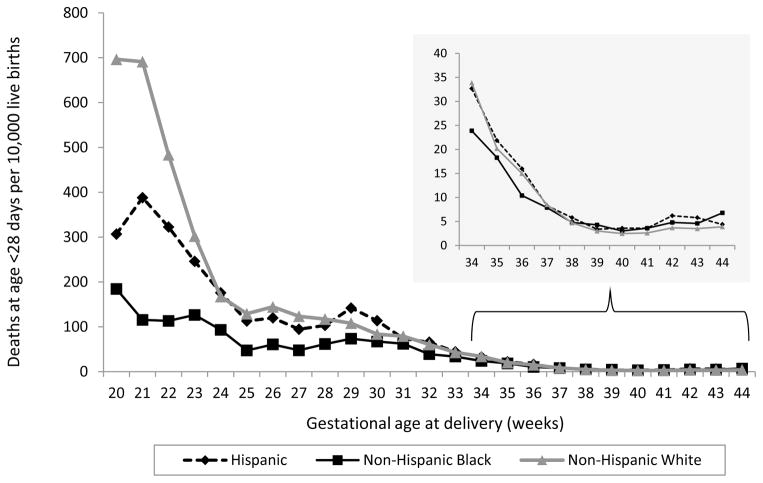
Nearly 69% of IMBD occurred in the neonatal period (Table 1). Neonatal mortality attributable to birth defects ranged from 7.8 per 10,000 live births among non-Hispanic white mothers to 9.1 per 10,000 live births among non-Hispanic black mothers. Across racial/ethnic categories, neonatal mortality attributable to birth defects was higher among both younger and older mothers, mothers with higher parity, multiple births, and mothers with diabetes, fewer years of education, or tobacco use. However, the most substantial elevations were observed for low birth weight or preterm infants. For example, among non-Hispanic white mothers, neonatal mortality attributable to birth defects was substantially higher at 97.5 per 10,000 live births for infants delivered at 20–33 weeks compared with 3.8 per 10,000 for infants delivered at 37–44 weeks. Congenital heart defects were the underlying cause of 24% of neonatal deaths attributable to birth defects.
Given the differences by gestational age, we did not present the overall effect estimates for weeks 20–44. White infants born early preterm (20–33 completed weeks’ gestation) had the highest rates of neonatal mortality for nearly all birth defect categories (Table 2). The rate of neonatal mortality attributable to birth defects was significantly lower among black infants delivered at 20–33 or 34–36 weeks gestation compared with whites (for example, for all birth defects combined, the neonatal mortality rates for infants of black and white mothers delivered at 20–33 weeks were 58.6 and 97.5 per 10,000, respectively; RR: 0.6 [95% CI: 0.5,0.6]). The black-white difference was reversed among infants delivered at 37–44 weeks, with neonatal mortality higher among infants of black compared with white mothers (for all birth defects combined, the neonatal mortality rates for infants of black and white mothers delivered at 37–44 weeks gestation were 4.6 and 3.8 per 10,000, respectively; RR: 1.2 [95% CI: 1.1, 1.3]). The neonatal RRs for each birth defect category were similar to those for all birth defects combined, with blacks having lower mortality rates when born preterm, but higher rates when born at term.
Table 2.
Rates of neonatal mortality attributable to birth defects by race/ethnicity, and adjusted† rate ratios comparing infants of non-Hispanic black and Hispanic mothers with those of non-Hispanic white mothers, United States, 2003–2006.
| Non-Hispanic White (N=9,071,585) |
Non-Hispanic Black (N=2,312,080) |
Hispanic (N=3,761,379) |
Non-Hispanic Black/ Non-Hispanic White |
Hispanic/ Non-Hispanic White |
|
|---|---|---|---|---|---|
|
Birth defect category, neonatal deaths (Gestational age in weeks) |
Rate/10,000 live births |
Rate/10,000 live births |
Rate/10,000 live births |
Rate Ratio (95% CI) | Rate Ratio (95% CI) |
| All birth defects, N=12,454* | |||||
| (20 – 33 weeks) | 97.5 | 58.6 | 91.3 | 0.6 (0.5–0.6) | 0.9 (0.8–1.0) |
| (34 – 36 weeks) | 19.8 | 15.5 | 20.9 | 0.8 (0.7–0.9) | 1.0 (0.9–1.1) |
| (37 – 44 weeks) | 3.8 | 4.6 | 4.6 | 1.2 (1.1–1.3) | 1.2 (1.1–1.2) |
| Central nervous system defects, N=2,062 | |||||
| (20 – 33 weeks) | 19.0 | 8.9 | 19.1 | 0.5 (0.4–0.6) | 1.0 (0.8–1.2) |
| (34 – 36 weeks) | 3.0 | 2.1 | 3.7 | 0.5 (0.4–0.7) | 1.2 (0.9–1.5) |
| (37 – 44 weeks) | 0.5 | 0.6 | 1.0 | 0.9 (0.7–1.2) | 1.6 (1.4–2.0) |
| Chromosomal abnormalities, N=2,891 | |||||
| (20 – 33 weeks) | 21.0 | 13.5 | 21.0 | 0.6 (0.5–0.8) | 1.0 (0.8–1.2) |
| (34 – 36 weeks) | 4.8 | 3.6 | 4.9 | 0.7 (0.6–1.0) | 1.2 (1.0–1.5) |
| (37 – 44 weeks) | 0.9 | 1.1 | 1.0 | 1.3 (1.1–1.5) | 1.2 (1.0–1.4) |
| Congenital heart defects, N=2,943 | |||||
| (20 – 33 weeks) | 14.8 | 8.0 | 13.5 | 0.6 (0.4–0.7) | 0.9 (0.7–1.1) |
| (34 – 36 weeks) | 4.2 | 2.7 | 3.8 | 0.6 (0.5–0.9) | 0.8 (0.6–1.0) |
| (37 – 44 weeks) | 1.3 | 1.5 | 1.2 | 1.2 (1.0–1.4) | 0.9 (0.8–1.0) |
| Digestive system/cleft defects, N=145 | |||||
| (20 – 33 weeks) | 1.1 | 1.1 | 1.4 | 0.8 (0.4–1.8) | 0.7 (0.3–1.7) |
| (34 – 36 weeks) | 0.2 | 0.3 | 0.1 | 1.4 (0.5–4.3) | 0.7 (0.2–2.6) |
| (37 – 44 weeks) | 0.0 | 0.1 | 0.0 | 1.9 (1.0–3.7) | 0.7 (0.3–1.6) |
| Genitourinary system defects, N=1,340 | |||||
| (20 – 33 weeks) | 12.6 | 7.2 | 9.9 | 0.5 (0.4–0.7) | 0.7 (0.6–0.9) |
| (34 – 36 weeks) | 3.0 | 2.9 | 3.4 | 0.9 (0.7–1.2) | 1.0 (0.8–1.3) |
| (37 – 44 weeks) | 0.2 | 0.3 | 0.4 | 1.2 (0.9–1.7) | 1.5 (1.1–1.9) |
| Musculoskeletal system defects, N=1,462 | |||||
| (20 – 33 weeks) | 10.4 | 6.5 | 8.1 | 0.6 (0.4–0.8) | 0.6 (0.5–0.8) |
| (34 – 36 weeks) | 2.4 | 1.8 | 2.5 | 0.8 (0.5–1.1) | 0.9 (0.6–1.2) |
| (37 – 44 weeks) | 0.5 | 0.7 | 0.5 | 1.4 (1.1–1.8) | 1.0 (0.8–1.3) |
| Respiratory system defects, N=199 | |||||
| (20 – 33 weeks) | 2.5 | 1.2 | 1.5 | 0.5 (0.3–0.9) | 0.7 (0.4–1.3) |
| (34 – 36 weeks) | 0.3 | 0.3 | 0.4 | 0.8 (0.3–2.1) | 1.2 (0.6–2.7) |
| (37 – 44 weeks) | 0.0 | 0.0 | 0.0 | 1.1 (0.5–2.6) | 1.1 (0.5–2.3) |
Adjusted for maternal age and education. Education data from the birth certificate were missing or lacked comparable data for 3,775(30.3%) of mothers.
Overall birth defect category includes all subcategories listed in the table plus eye, ear, face, and neck defects (N=5), and defects not included in any other subcategory (N=1,407).
Similar to the black-white differences, among term infants, Hispanics had a significantly higher neonatal mortality rate attributable to birth defects than whites overall (RR: 1.2 [95% CI: 1.1, 1.2]) and in most defect categories; however, the differences in the 20–36 weeks gestational age categories were not statistically significant.
We assessed the race/ethnicity-specific rates of neonatal mortality attributable to birth defects graphically by gestational week (Figure 1). We also examined these patterns for each different birth defect category, and the striking neonatal picture was similar for all birth defect groups (individual graphs by defect category not shown).
Figure 1.
Neonatal mortality rate attributable to birth defects
Postneonatal Mortality Attributable to Birth Defects
Postneonatal mortality attributable to birth defects ranged from 3.3 per 10,000 live births among non-Hispanic white mothers to 5.3 per 10,000 live births among non-Hispanic black mothers (Table 3). The same maternal characteristics associated with an increased rate of neonatal mortality were also associated with an increased rate of postneonatal mortality, including gestational age.
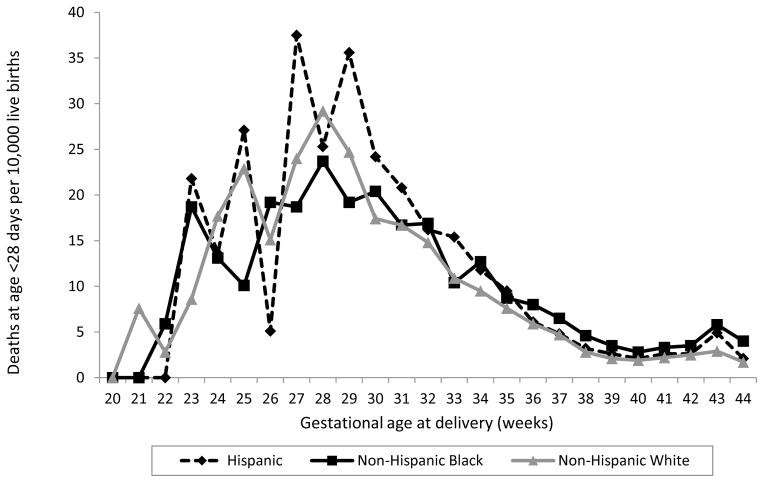
Comparing infants of black and white mothers, the rates of postneonatal mortality attributable to birth defects (Table 4) were quite different from the rates of neonatal mortality attributable to birth defects (Table 2). Uniquely for postneonatal mortality, blacks born at 34–36 weeks had a higher rate than whites born late preterm (for all birth defects combined, RR: 1.3 [95% CI: 1.1, 1.6]; for chromosomal abnormalities, RR: 2.2 [95% CI: 1.5, 3.3]), and there was no significant difference in the postneonatal mortality rate among blacks and whites born at 20–33 weeks (with RRs of 0.9 and 1.1 for all birth defects combined and chromosomal abnormalities, respectively). However, similarly to neonatal mortality, among term infants, blacks had a significantly higher postneonatal mortality rate than whites (for all birth defects combined, RR: 1.5 [95% CI: 1.4, 1.7]; for chromosomal abnormalities, RR: 2.2 [95% CI: 1.8, 2.7]). Comparing postneonatal mortality attributable to birth defects between Hispanics and whites, there were no statistically significant differences in any gestational age group for all birth defects combined, with RRs ranging from 1.0 to 1.1 (Table 4); however, Hispanics had significantly higher rates for some defect groups, including central nervous system defects (at term) and chromosomal abnormalities (at any gestational age except 20–33 weeks).
Table 4.
Rates of postneonatal mortality attributable to birth defects by race/ethnicity, and adjusted† rate ratios comparing infants of non-Hispanic black and Hispanic mothers with those of non-Hispanic white mothers, United States, 2003–2006.
| Non-Hispanic White (N=9,071,585) |
Non-Hispanic Black (N=2,312,080) |
Hispanic (N=3,761,379) |
Non-Hispanic Black/ Non-Hispanic White |
Hispanic/ Non-Hispanic White |
|
|---|---|---|---|---|---|
|
Birth defect category, postneonatal deaths (Gestational age in weeks) |
Rate/10,000 live births |
Rate/10,000 live births |
Rate/10,000 live births |
Rate Ratio (95% CI) | Rate Ratio (95% CI) |
| All birth defects, N=5,626* | |||||
| (20 – 33 weeks) | 15.7 | 15.2 | 19.0 | 0.9 (0.7–1.1) | 1.1 (0.9–1.3) |
| (34 – 36 weeks) | 7.0 | 9.2 | 8.2 | 1.3 (1.1–1.6) | 1.1 (0.9–1.3) |
| (37 – 44 weeks) | 2.5 | 4.0 | 2.9 | 1.5 (1.4–1.7) | 1.0 (0.9–1.1) |
| Central nervous system defects, N=653 | |||||
| (20 – 33 weeks) | 2.1 | 2.9 | 3.1 | 1.2 (0.8–2.0) | 1.1 (0.7–1.8) |
| (34 – 36 weeks) | 0.8 | 0.8 | 1.0 | 1.1 (0.7–1.8) | 1.0 (0.6–1.6) |
| (37 – 44 weeks) | 0.2 | 0.4 | 0.4 | 1.4 (1.0–1.9) | 1.3 (1.0–1.8) |
| Chromosomal abnormalities, N=1,005 | |||||
| (20 – 33 weeks) | 2.1 | 2.4 | 2.8 | 1.1 (0.7–1.9) | 1.5 (0.9–2.4) |
| (34 – 36 weeks) | 1.1 | 2.0 | 1.6 | 2.2 (1.5–3.3) | 1.8 (1.2–2.7) |
| (37 – 44 weeks) | 0.4 | 0.9 | 0.6 | 2.2 (1.8–2.7) | 1.3 (1.0–1.6) |
| Congenital heart defects, N=2,586 | |||||
| (20 – 33 weeks) | 6.3 | 5.2 | 7.1 | 0.8 (0.6–1.1) | 1.0 (0.8–1.4) |
| (34 – 36 weeks) | 3.2 | 3.6 | 3.0 | 0.9 (0.7–1.3) | 0.8 (0.6–1.0) |
| (37 – 44 weeks) | 1.3 | 1.9 | 1.3 | 1.4 (1.2–1.6) | 0.9 (0.8–1.1) |
| Digestive system/cleft defects, N=213 | |||||
| (20 – 33 weeks) | 0.9 | 1.4 | 1.2 | 1.3 (0.6–2.6) | 1.1 (0.5–2.5) |
| (34 – 36 weeks) | 0.2 | 0.6 | 0.5 | 2.4 (1.2–4.7) | 1.5 (0.7–3.1) |
| (37 – 44 weeks) | 0.1 | 0.1 | 0.1 | 1.1 (0.5–2.1) | 1.2 (0.7–2.1) |
| Genitourinary system defects, N=74 | |||||
| (20 – 33 weeks) | 0.3 | 0.4 | 1.3 | 1.1 (0.3–4.1) | 3.7(1.3–10.4) |
| (34 – 36 weeks) | 0.1 | 0.1 | 0.1 | 1.5 (0.5–5.3) | 1.0 (0.2–3.9) |
| (37 – 44 weeks) | 0.0 | 0.0 | 0.0 | 1.5 (0.5–4.7) | 1.0 (0.4–3.1) |
| Musculoskeletal system defects, N=507 | |||||
| (20 – 33 weeks) | 1.5 | 1.3 | 1.3 | 0.8 (0.4–1.5) | 1.0 (0.5–2.0) |
| (34 – 36 weeks) | 0.8 | 0.9 | 1.0 | 1.1 (0.6–1.8) | 1.0 (0.6–1.6) |
| (37 – 44 weeks) | 0.2 | 0.3 | 0.2 | 1.2 (0.8–1.6) | 0.8 (0.6–1.1) |
| Respiratory system defects, N=89 | |||||
| (20 – 33 weeks) | 0.5 | 0.2 | 0.4 | 0.3 (0.1–1.3) | 0.4 (0.1–1.8) |
| (34 – 36 weeks) | 0.1 | 0.3 | 0.0 | 0.9 (0.3–2.4) | 0.2 (0.0–1.3) |
| (37 – 44 weeks) | 0.0 | 0.1 | 0.0 | 1.0 (0.5–2.1) | 0.7 (0.3–1.6) |
Adjusted for maternal age and education. Education data from the birth certificate were missing or lacked comparable data for 3,775(30.3%) of mothers.
Overall birth defect category includes all subcategories listed in the table plus eye, ear, face, and neck defects (N=4), and defects not included in any other subcategory (N=495).
We also assessed the race/ethnicity-specific rates of postneonatal mortality attributable to birth defects graphically by gestational week (Figure 2).
Figure 2.
Postneonatal mortality rate attributable to birth defects
Discussion
Overall racial/ethnic differences in IMBD cannot be explained by differences in gestational age. For both early and late preterm infants, we found neonatal mortality attributable to birth defects to be significantly less likely among non-Hispanic blacks than among non-Hispanic whites. Previous reports support our findings that infants born to black mothers had higher IMBD rates than infants born to white mothers,5, 13 and that term infants born to black and Hispanic mothers had higher IMBD rates compared to those born to whites.9
Similar patterns have been reported for all-cause neonatal mortality by gestational age group in the United States during 1989–2001.7 The reason for the lower rate of all-cause neonatal mortality among preterm infants of black mothers compared with white mothers is unclear. One possibility is that live-born infants who die shortly after birth might be misclassified as fetal deaths, particularly those born at early gestational ages.7 The fetal mortality rate in the United States is approximately twice as high among blacks as among whites,14, 15 and differences by race in reporting fetal deaths versus early neonatal deaths might exist. Recent research has shown variation by state in classification of neonatal death at <24 hours versus fetal death for infants at the limits of viability (i.e., gestation of <24 weeks or birth weight <500 g).16 Whether such variation might also occur by race/ethnicity is unknown.
Also unclear is whether factors specific to birth defects contribute to the differences in black and white neonatal mortality patterns by gestational age. Potentially, differences in prevalence of specific types of birth defects might explain the differences in mortality patterns. Whereas several categories of birth defects vary by race/ethnicity, no study has identified overall racial/ethnic differences in all birth defects combined.17–20 There are also many birth defects for which no racial/ethnic differences have been observed; for instance, among studies that included birth defect prevalence among live births, stillbirths, and pregnancy terminations, no racial difference was observed for prevalence of hypoplastic left heart syndrome, the most common specific cause of death attributable to congenital heart defects.17, 19 Differences in maternal serum screening uptake might also differ by race/ethnicity, which could contribute to differences in prenatal diagnosis of birth defects.21, 22 Excess postneonatal mortality attributable to chromosomal defects was observed for non-Hispanic blacks and for all gestational age categories of Hispanics compared with non-Hispanic whites. This is consistent with the finding that median age at death for people with Down syndrome is much lower among blacks and other races than among whites;23–26 no similar study has assessed median age at death for Down syndrome by Hispanic ethnicity.
Improved technologies and neonatal care might have shifted some deaths to the postneonatal period. For instance, in the last decade, a Norwood procedure with a Sano shunt instead of a Blalock-Taussig shunt has become a common alternative for neonates with single-ventricle physiology such as hypoplastic left heart syndrome. A recent randomized, multi-center, clinical trial found that subjects with a Sano shunt had decreased short-term mortality rates as compared to those with a Blalock-Taussig shunt.27
In addition to gestational age and race/ethnicity, we found associations of IMBD with maternal age, parity, plurality, and diabetes in both the neonatal and postneonatal periods. These associations are consistent with previous observations on infant mortality from all causes.28, 29 Our estimate of the IMBD prevalence (18%) is lower than the typically referenced 20%28 due to our noted ICD exclusions within the larger group of “all congenital malformations”.
Efforts to reduce infant mortality rates due to birth defects should include both: 1) strategies to decrease mortality among those infants living with birth defects through timely and appropriate medical and surgical treatment, and 2) prevention of birth defects, where possible, by addressing modifiable known risk factors such as folic acid use, avoidance of teratogenic medicines, prevention of certain infections by getting vaccinated and potential risk factors such as pregestational diabetes, obesity, and maternal smoking.30–33
This analysis included over 15 million births to non-Hispanic white, non-Hispanic black, and Hispanic mothers in the United States from 2003–2006. Use of the linked birth/infant death files allowed us to assess infant mortality attributable to specific causes while stratifying or adjusting for variables included on the birth certificate.28 These data also allowed us to ensure that the same maternal race/ethnicity variable (from the US birth certificate) was used for the numerator (infant deaths) and the denominator (total live births); this was essential given the focus of our analysis on racial/ethnic differences.
Because of the evident effect measure modification by gestational age, of not only the magnitude but also the direction of the effect, adjusting for gestational age or its correlates (such as birth weight), as has been done in previous studies, would likely obscure important differences in infant mortality rates by gestational age and race/ethnicity.
Limitations
This study has several limitations. Gestational age can be inaccurate on the birth certificate and may be less accurate among preterm births.6 Although we excluded cases with implausible gestational age/birth weight combinations, we likely included some births for which gestational age was misclassified. The quality of other covariate data available on the birth certificate is also a limitation. Education data from the birth certificate were missing or lacked comparable data for 30% of mothers, limiting the sample size for adjusted analyses, although the adjusted results were very similar to crude results that included the full data set. This analysis included deaths due to birth defects listed as the underlying cause of death and did not identify those listing the defect as only a contributing cause. This likely resulted in an underestimation of the mortality rate due to birth defects. However, this limitation would only impact our analyses of racial/ethnic differences in mortality rates if there were differential reporting of the underlying cause of death among racial/ethnic groups. Finally, there was an underlying assumption of random missingness on the weighting scheme to account for unlinked birth records, and a violation of this assumption might impact these results; however, given the high percentage of linked records, the unweighted results were similar to our weighted results.
Conclusion
The reasons why infants of black mothers and, to a lesser extent, Hispanic mothers, have higher rates of IMBD than infants of white mothers are unclear. Similarly, we cannot explain why preterm infants of black mothers with birth defects fare better than preterm infants of white mothers, when stratified by gestational age. Further investigation should include assessments of possible racial/ethnic differences in other factors that contribute to infant mortality – such as severity of the defect, access to timely diagnosis, and the quality of medical care provided. In addition, racial/ethnic differences in the frequency of prenatal diagnosis – and the subsequent termination of pregnancies affected by severe birth defects – can contribute to differences in the live birth prevalence of severe defects.34 Lastly, racial/ethnic differences in the classification of fetuses at the limits of viability as live births (and subsequent infant deaths) or fetal deaths, could have influenced our results. Given that stillbirths are more common among US black mothers than white,14, 15 the occurrence of more neonatal deaths among very preterm infants of white mothers than among very preterm infants of black mothers may be reflective of a racial/ethnic difference in the classification of deliveries at the limits of viability.
What’s Known on this Subject
Birth defects are associated with preterm birth and are a major contributor to infant mortality. There are persistent black-white differences in overall infant mortality and infant mortality attributable to birth defects.
What This Study Adds
Among infants delivered at 37–44 weeks, blacks and Hispanics had significantly higher neonatal and postneonatal mortality attributable to birth defects than whites. Among infants delivered at 20–33 or 34–36 weeks, neonatal mortality attributable to birth defects was significantly lower among blacks.
Acknowledgments
Funding: This research was supported in part by an appointment to the Research Participation Program at the Centers for Disease Control and Prevention administered by the Oak Ridge Institute for Science and Education through an interagency agreement between the U.S. Department of Energy and CDC.
The authors acknowledge the contributions of Huichao Chen, Quanhe Yang, Adolfo Correa, and Owen Devine to an earlier version of this project.
Abbreviations
- IMBD
infant mortality attributable to birth defects
Footnotes
Financial disclosure: The authors have no financial relationships relevant to this article to disclose.
Conflicts of interest: None.
Disclaimer: The findings and conclusions in this report are those of the authors and do not necessarily represent the official position of the Centers for Disease Control and Prevention.
Contributor’s Statement
- Substantial contribution to conception and design, acquisition of data, and/or analysis and interpretation of data;
- Drafting the article and/or revising it critically for important intellectual content; and
- Final approval of the version submitted for publication.
References
- 1.CDC. Update on overall prevalence of major birth defects—Atlanta, Georgia, 1978–2005. MMWR Morb Mortal Wkly Rep. 2008;57:1–5. [PubMed] [Google Scholar]
- 2.Petrini J, Damus K, Russell R, Poschman K, Davidoff MJ, Mattison D. Contribution of birth defects to infant mortality in the United States. Teratology. 2002;66 (Suppl 1):S3–6. doi: 10.1002/tera.90002. [DOI] [PubMed] [Google Scholar]
- 3.Martin JA, Kung HC, Mathews TJ, Hoyert DL, Strobino DM, Guyer B, et al. Annual summary of vital statistics: 2006. Pediatrics. 2008;121(4):788–801. doi: 10.1542/peds.2007-3753. [DOI] [PubMed] [Google Scholar]
- 4.Martin JH, BE, Sutton PD, et al. National vital statistics reports. National Center for Health Statistics; 2009. Births: Final data for 2006. [Google Scholar]
- 5.Yang Q, Chen H, Correa A, Devine O, Mathews TJ, Honein MA. Racial differences in infant mortality attributable to birth defects in the United States, 1989–2002. Birth Defects Res A Clin Mol Teratol. 2006;76(10):706–713. doi: 10.1002/bdra.20308. [DOI] [PubMed] [Google Scholar]
- 6.Alexander GR, Kogan M, Bader D, Carlo W, Allen M, Mor J. US birth weight/gestational age-specific neonatal mortality: 1995–1997 rates for whites, hispanics, and blacks. Pediatrics. 2003;111(1):e61–66. doi: 10.1542/peds.111.1.e61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.CDC. Racial/ethnic disparities in neonatal mortality--United States, 1989–2001. MMWR Morb Mortal Wkly Rep. 2004;53(29):655–658. [PubMed] [Google Scholar]
- 8.CDC. Racial differences by gestational age in neonatal deaths attributable to congenital heart defects --- United States, 2003–2006. MMWR Morb Mortal Wkly Rep. 2010;59(37):1208–1211. [PubMed] [Google Scholar]
- 9.Reddy UM, Bettegowda VR, Dias T, Yamada-Kushnir T, Ko CW, Willinger M. Term pregnancy: a period of heterogeneous risk for infant mortality. Obstet Gynecol. 2011;117(6):1279–1287. doi: 10.1097/AOG.0b013e3182179e28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.NCHS. Public-use data file documentation: 2006 period linked birth/infant death data set [online] Hyattsville, MD: National Center for Health Statistics; [Google Scholar]
- 11.Lydon-Rochelle MT, Holt VL, Cardenas V, Nelson JC, Easterling TR, Gardella C, et al. The reporting of pre-existing maternal medical conditions and complications of pregnancy on birth certificates and in hospital discharge data. Am J Obstet Gynecol. 2005;193(1):125–134. doi: 10.1016/j.ajog.2005.02.096. [DOI] [PubMed] [Google Scholar]
- 12.Gore DC, Chez RA, Remmel RJ, Harahan M, Mock M, Yelverton R. Unreliable medical information on birth certificates. J Reprod Med. 2002;47(4):297–302. [PubMed] [Google Scholar]
- 13.CDC. Trends in infant mortality attributable to birth defects--United States, 1980–1995. MMWR Morb Mortal Wkly Rep. 1998;47(37):773–778. [PubMed] [Google Scholar]
- 14.CDC. Racial/ethnic trends in fetal mortality--United States, 1990–2000. MMWR Morb Mortal Wkly Rep. 2004;53(24):529–532. [PubMed] [Google Scholar]
- 15.Willinger M, Ko CW, Reddy UM. Racial disparities in stillbirth risk across gestation in the United States. Am J Obstet Gynecol. 2009;201(5):469 e461–468. doi: 10.1016/j.ajog.2009.06.057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ehrenthal DB, Wingate MS, Kirby RS. Variation by state in outcomes classification for deliveries less than 500 g in the United States. Matern Child Health J. 2011;15(1):42–48. doi: 10.1007/s10995-010-0566-y. [DOI] [PubMed] [Google Scholar]
- 17.Botto LD, Correa A, Erickson JD. Racial and temporal variations in the prevalence of heart defects. Pediatrics. 2001;107(3):E32. doi: 10.1542/peds.107.3.e32. [DOI] [PubMed] [Google Scholar]
- 18.Carmichael SL, Shaw GM, Kaidarova Z. Congenital malformations in offspring of Hispanic and African-American women in California, 1989–1997. Birth Defects Res A Clin Mol Teratol. 2004;70(6):382–388. doi: 10.1002/bdra.20012. [DOI] [PubMed] [Google Scholar]
- 19.Canfield MA, Honein MA, Yuskiv N, Xing J, Mai CT, Collins JS, et al. National estimates and race/ethnic-specific variation of selected birth defects in the United States, 1999–2001. Birth Defects Res A Clin Mol Teratol. 2006;76(11):747–756. doi: 10.1002/bdra.20294. [DOI] [PubMed] [Google Scholar]
- 20.Kucik JE, Alverson CJ, Gilboa SM, Correa A. Racial/Ethnic Variations in the Prevalence of Selected Major Birth Defects, Metropolitan Atlanta, 1994–2005. Public Health Reports. doi: 10.1177/003335491212700106. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Press N, Browner CH. Characteristics of women who refuse an offer of prenatal diagnosis: data from the California maternal serum alpha fetoprotein blood test experience. Am J Med Genet. 1998;78(5):433–445. [PubMed] [Google Scholar]
- 22.Li DK, Karlberg K, Wi S, Norem C. Factors influencing women’s acceptance of prenatal screening tests. Prenat Diagn. 2008;28(12):1136–1143. doi: 10.1002/pd.2142. [DOI] [PubMed] [Google Scholar]
- 23.CDC. Racial disparities in median age at death of persons with Down syndrome. MMWR Morb Mortal Wkly Rep. 2001;50:463–465. [PubMed] [Google Scholar]
- 24.Yang Q, Rasmussen SA, Friedman JM. Mortality associated with Down’s syndrome in the USA from 1983 to 1997: a population-based study. Lancet. 2002;359(9311):1019–1025. doi: 10.1016/s0140-6736(02)08092-3. [DOI] [PubMed] [Google Scholar]
- 25.Rasmussen SA, Wong LY, Correa A, Gambrell D, Friedman JM. Survival in infants with Down syndrome, Metropolitan Atlanta, 1979–1998. J Pediatr. 2006;148(6):806–812. doi: 10.1016/j.jpeds.2006.01.010. [DOI] [PubMed] [Google Scholar]
- 26.Shin M, Besser LM, Kucik JE, Lu C, Siffel C, Correa A. Prevalence of Down syndrome among children and adolescents in 10 regions of the United States. Pediatrics. 2009;124(6):1565–1571. doi: 10.1542/peds.2009-0745. [DOI] [PubMed] [Google Scholar]
- 27.Ohye RG, Sleeper LA, Mahony L, Newburger JW, Pearson GD, Lu M, et al. Comparison of shunt types in the Norwood procedure for single-ventricle lesions. N Engl J Med. 2010;362(21):1980–1992. doi: 10.1056/NEJMoa0912461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Mathews TJ, Menacker F, MacDorman MF. Infant mortality statistics from the 2002 period: linked birth/infant death data set. Natl Vital Stat Rep. 2004;53(10):1–29. [PubMed] [Google Scholar]
- 29.MacDorman MF, Mathews TJ. Infant deaths - United States, 2000–2007. MMWR Surveill Summ. 2011;60 (Suppl):49–51. [PubMed] [Google Scholar]
- 30.Jenkins KJ, Correa A, Feinstein JA, Botto L, Britt AE, Daniels SR, et al. Noninherited risk factors and congenital cardiovascular defects: current knowledge: a scientific statement from the American Heart Association Council on Cardiovascular Disease in the Young: endorsed by the American Academy of Pediatrics. Circulation. 2007;115(23):2995–3014. doi: 10.1161/CIRCULATIONAHA.106.183216. [DOI] [PubMed] [Google Scholar]
- 31.Waller DK, Shaw GM, Rasmussen SA, Hobbs CA, Canfield MA, Siega-Riz AM, et al. Prepregnancy obesity as a risk factor for structural birth defects. Arch Pediatr Adolesc Med. 2007;161(8):745–750. doi: 10.1001/archpedi.161.8.745. [DOI] [PubMed] [Google Scholar]
- 32.Correa A, Gilboa SM, Besser LM, Botto LD, Moore CA, Hobbs CA, et al. Diabetes mellitus and birth defects. Am J Obstet Gynecol. 2008;199(3):237 e231–239. doi: 10.1016/j.ajog.2008.06.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Hackshaw A, Rodeck C, Boniface S. Maternal smoking in pregnancy and birth defects: a systematic review based on 173 687 malformed cases and 11.7 million controls. Hum Reprod Update. 2011 doi: 10.1093/humupd/dmr022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Cragan JD, Gilboa SM. Including prenatal diagnoses in birth defects monitoring: Experience of the Metropolitan Atlanta Congenital Defects Program. Birth Defects Res A Clin Mol Teratol. 2009;85(1):20–29. doi: 10.1002/bdra.20508. [DOI] [PubMed] [Google Scholar]