Abstract
We previously reported results of a phase II non-comparative trial that randomized patients with glioblastoma following radiotherapy to one of two different temozolomide schedules, followed by 13-cis-retinoic acid (RA) maintenance. Here we report the results of an exploratory cohort of patients accrued with anaplastic astrocytic tumors. Patients with newly diagnosed anaplastic astrocytoma (AA) or anaplastic oligo-astrocytoma (AOA) were treated with concurrent radiotherapy (60 Gy over 6 weeks) and temozolomide (75 mg/m2), and six adjuvant 28-day cycles of either dose-dense (150 mg/m2, days 1–7, 15–21) or metronomic (50 mg/m2, days 1–28) temozolomide. Subsequently, maintenance RA (100 mg/m2, days 1–21/28) was administered until disease progression. All outcome measures were descriptive without intention to compare between treatment arms. Survival was measured by the Kaplan–Meier method. There were 31 patients (21 men, 10 women) with median age 48 years (range 28–74), median KPS 90 (range 60–100). Extent of resection was gross-total in 35 %, subtotal 23 %, and biopsy 42 %. Histology was AA in 90 %, and AOA in 10 %. MGMT promoter methylation was methylated in 20 %, unmethylated in 50 %, and uninformative in 30 % of 30 tested. Median progression-free survival was 2.1 years (95 % CI 0.95–Not Reached), and overall survival 2.9 years (95 % CI 2.0–Not Reached). We report outcomes among a homogeneously treated population with anaplastic astrocytic tumors. Survival was unexpectedly short compared to other reports. These data may be useful as a contemporary historic control for other ongoing or future randomized trials.
Keywords: Anaplastic astrocytoma, Temozolomide, Radiotherapy, Chemotherapy, Clinical trial
Introduction
The standard treatment for newly diagnosed glioblastoma (GBM) is radiotherapy (RT) with concurrent temozolomide followed by adjuvant temozolomide [1]. However, the optimal treatment for anaplastic (WHO grade III) gliomas is not as well established.
Adjuvant (post-RT) temozolomide is conventionally dosed at 150–200 mg/m2 for a 5/28-day cycle [2]. Temozolomide has demonstrated schedule-dependent anti-tumor activity and there has been significant interest in testing other dosing schedules to enhance cytotoxicity. O6-methylguanine-DNA methyltransferase (MGMT) is a DNA-repair enzyme encoded by the methylguanine methyltransferase (MGMT) gene. Protracted exposure of temozolomide, which targets the O6 position of guanine, may lead to cumulative and sustained MGMT depletion, thereby reducing temozolomide resistance [3]. More recent phase III trials have demonstrated no benefit of different schedules of temozolomide in recurrent high grade gliomas or newly diagnosed GBM [4, 5]. The trial described in the current report was designed before the results of those studies were available, and in patients with newly diagnosed anaplastic astrocytic tumors rather than GBM or recurrent tumors.
Separately, retinoids induce differentiation and apoptosis of glioma cells, as well inhibit cell proliferation and migration [6–8]. Accordingly, 13-cis-retinoic acid (RA) may help maintain disease control in treated patients with malignant glioma, as well as in those with recurrent disease [9].
In 2009, Clarke et al. published the results of a single-institution randomized non-comparative phase II trial of chemoradiation followed by 6-cycles of either “metronomic” or “dose-dense” temozolomide and then maintenance RA for newly diagnosed GBM [10]. Patients with anaplastic tumors were accrued to a separate arm to explore efficacy of these regimens in patients with other glioma subtypes. Here, we present the results from this exploratory cohort, specifically those with anaplastic astrocytic tumors (anaplastic astrocytoma, AA; or anaplastic oligo-astrocytoma, AOA).
Patients and methods
The eligibility criteria were published previously [10]. In brief, participation required age ≥18 years, diagnosis of treatment naïve (except surgery) newly diagnosed anaplastic (WHO grade III) glioma, normal bone marrow and end organ function, and Karnofsky Performance Status (KPS) ≥60. Following maximal safe surgical resection, protocol treatment started within 6 weeks which included concurrent RT (60 Gy ± 5 % without stereotactic boost) and temozolomide (75 mg/m2 daily) followed by randomized (1:1) treatment to adjuvant temozolomide using either a “dose-dense” schedule (150 mg/m2 days 1–7 and 15–21 of 28) or metronomic (50 mg/m2 days 1–28 of 28) schedule. Patients who withdrew consent prior to randomized therapy were excluded. Following six adjuvant temozolomide 28-day cycles of either regimen, patients then received oral RA (100 mg/m2, days 1–21 of 28) until disease progression (MRI every other cycle using Macdonald criteria [11]), unacceptable toxicity (CTCAE 3.0), or withdrawal of consent. Supportive therapy including anti-emetics and growth factors were permitted per standard of care. Median progression-free and overall survival were was estimated by the Kaplan–Meier method; however, the study was not powered to compare treatment arms against each other or historic controls, and all statistical analyses were entirely exploratory in nature. When available, tumor tissue was analyzed for MGMT promoter methylation status by Oncomethylome Sciences (Amsterdam, the Netherlands). Informed consent was obtained from all individual participants included in the study (or appropriate surrogates) which was approved by the institutional review board of Memorial Sloan Kettering Cancer Center.
Results
Patient characteristics
Results among patients with GBM were published previously. Those with AA/AOA are reported here. Patients were enrolled from July 2005 through February 2011. There were 31 patients with a median age 48 years (range 20–74) and median KPS 90 (range 60–100). There were 21 men (68 %). Extent of resection, assessed by the treating investigator using the post-operative MRI, was gross-total in 11 (35 %), subtotal in 7 (23 %), and biopsy in 13 (42 %). Histology was AA in 28 (90 %). Chromosome 1p19q deletion status was assessed in 2 of 3 AOAs, and one had co-deletion. MGMT promoter methylation was assessed in 30 tumors (27 AAs, 3 AOAs), and was methylated in 6 (20 %), unmethylated in 15 (50 %), and uninformative in 9 (30 %).
Treatment
There were 14 randomized to dose-dense and 17 to metronomic adjuvant temozolomide. However, 6/31 did not proceed to adjuvant therapy because of either refusal (2) or early progression (4). Therefore, 25 received either dose-dense (11) or metronomic (14) adjuvant therapy, and 18 completed the proscribed 6 adjuvant temozolomide cycles without disease progression or unacceptable toxicity. However, 7/18 did not proceed to RA maintenance because of either refusal (5) or insurance denial coverage (2). One patient remained on adjuvant temozolomide at the time of the analysis.
Toxicity
The toxicity of concurrent RT and temozolomide has been well described and was not collected in this exploratory cohort. Adjuvant metronomic and dose-dense temozolomide was well-tolerated, with the grade 3 lymphopenia as most common serious treated related adverse event (10 events) (Table 1). Adverse events were more common in the metronomic temozolomide group. One patient discontinued treatment because of toxicity (grade 3 fatigue and GI intolerance) from dose-dense temozolomide. One patient developed a second malignancy (invasive squamous cell carcinoma), but this was considered unrelated to protocol therapy.
Table 1.
Adverse events attributable to temozolomide
| Adverse events | Metronomic temozolomide (n = 17)
|
Dose-dense temozolomide (n = 14)
|
||
|---|---|---|---|---|
| Grade 3 | Grade 4 | Grade 3 | Grade 4 | |
| Elevated liver enzymes | 1 | – | – | – |
| Constipation | 1 | – | – | – |
| Abdominal pain | – | – | 1 | – |
| Fatigue | 2 | – | 2 | – |
| Lymphopenia | 6 | 1 | 4 | 2 |
| Neutropenia | – | 1 | – | – |
| Thrombocytopenia | 1 | 1 | – | – |
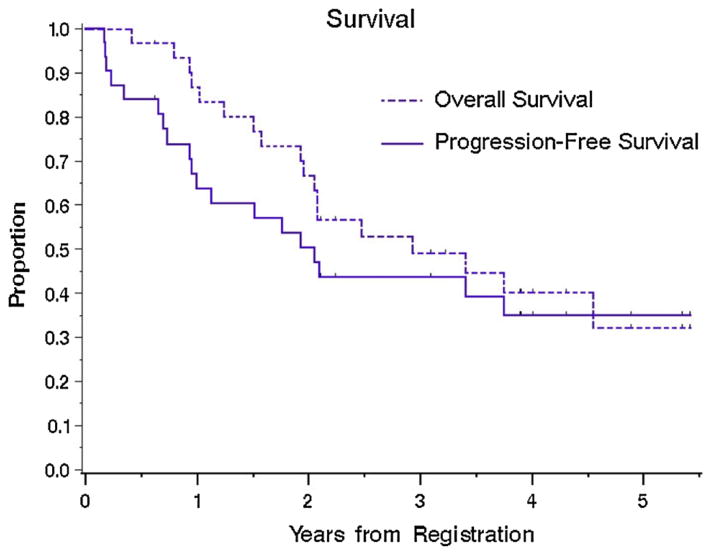
Survival
After a median follow up of 2.4 years (range, 0.4–5.4 years) and 18 deaths, the median overall survival (OS) was 2.9 years (95 % CI 2.0–Not Reached) and 1-year OS rate was 87 % (95 % CI 75–99 %) (Fig. 1, n = 31). Median progression-free survival (PFS) was 2.1 years (95 % CI 0.95–Not Reached) and 1-year PFS rate was 64 % (95 % CI 47–81 %). Outcome per arm is shown in Table 2 and Supplemental Fig. 1. The arms were not powered for comparison.
Fig. 1.
Overall survival (OS) and Progression-free survival (PFS) for all patients
Table 2.
Survival by arm
| Cohort | Metronomic Temozolomide (n = 17) | Dose-dense temozolomide (n = 14) |
|---|---|---|
| Median OS | 2.1 years (95 % CI, 1.5–4.6) | Not Reached |
| 1-year OS | 88 % (95 % CI, 73–100) | 85 % (95 % CI, 66–100) |
| Median PFS | 1.5 years (95 % CI, 0.7–3.4) | 3.8 y (95 % CI, 0.3–No Upper Limit) |
| 1-year PFS | 59 % (95 % CI, 35–82) | 71 % (95 % CI, 47–95) |
Discussion
Following maximal surgical resection, chemoradiotherapy with temozolomide is the standard of care for newly diagnosed GBM [1]. We explored this approach in a prospective clinical trial for patients with anaplastic astrocytic tumors. An ongoing phase III study (CATNON—Concurrent and adjuvant temozolomide for 1p19q non-codeleted anaplastic gliomas) will definitively address the benefit of this approach in a similar population of patients, but results will not mature for several years.
Comparison with prior studies (Table 3) [12–19] is difficult because of heterogeneity of histology and treatment approaches. For example, a German phase III study (NOA-04) of anaplastic gliomas included patients with pure oligodendrogliomas, and also used sequential rather than concurrent radiotherapy and chemotherapy [14]; it was also published when results were relatively immature [20]. Moreover, in NOA-04, although the median PFS for patients with all histologies receiving either RT or chemotherapy was ≥2.5 years, median PFS for those with AA was <1 year in patients receiving RT alone and 1.5 years in those receiving chemotherapy alone. By contrast, our data represent a reasonably homogenous population with mature survival data.
Table 3.
Prior studies in WHO grade III gliomas
| Design | Histology | N | Treatment | PFS (y) | OS (y) | Reference/Authors |
|---|---|---|---|---|---|---|
| Phase III | AG | 190 | RT + PCV |
– | 4.08 | Prados et al. (RTOG 9404) [13] |
| RT/BUdR +PCV | 4.50 | |||||
| Retrospective | AA | 109 | RT → PCV | 2.87 | NR | Brandes et al. [15] |
| RT → TMZ | 3.01 | NR | ||||
| Phase III | AA | 193 | RT | 1.03 | 1.99 | Hildebrand et al. (EORTC 26882) [16] |
| RT + DBD/BCNU | 1.23 | 2.28 | ||||
| Retrospective | AA, AOA | 60 | RT | 0.58 | 1.08 | Combs et al. [17] |
| RT/TMZ | 0.50 | 1.25 | ||||
| Phase III | AA, AO, AOA | 274 | RT | 2.55 | 6.01 | Wick et al. (NAO-04) [14] |
| PCV/TMZ | 2.66 | 6.88 | ||||
| Phase II | AA, AO | 33 | RT/TMZ → TMZ | 4.06 | 5.50 | Kim et al. [18] |
| Phase II | AA, AO, AOA | 32 | TMZ, 13-cRA | 3.15 | NR | Grauer et al. (RNOP-05) [12] |
| Retrospective | AA | 97 | RT/TMZ → TMZ | 3.0 | 4.2 | Minniti et al. [19] |
N number of patients; AG anaplastic glioma; AA anaplastic astrocytoma; AOA anaplastic oligoastrocytoma; AO anaplastic oligodendroglioma; RT radiation therapy; PCV procarbazine, lomustine, vincristine; BUdR bromodeoxyuridine; DBD dibromodulcitol; BCNU bichloroethylni-trosourea/carmustine; TMZ temozolomide; 13-cRA 13-cis-retinoic acid
The most striking finding may be the relatively low proportion of patients who actually completed the entire planned course of treatment. For example, 12 (39 %) did not complete all 6 cycles of adjuvant temozolomide because of either early confirmed (3) or suspected (2) disease progression, tumor (2) or treatment (1) related side effects, non-compliance (1), refusal (2), or loss to follow up (1) These findings highlight the high degree of variability in biologic function of the anaplastic glioma group. While grade III histology is often expected to confer a better prognosis than GBM, these data emphasize that only a subgroup of patients with WHO grade III tumors actually have a better outcome and many have or behave as if they have a WHO grade IV (GBM) tumor. The relatively low frequency of MGMT promoter methylation (20 %) may partially explain the poor outcome. In addition, 42 % of our patients had only a biopsy for diagnosis and those patients may have harbored WHO grade IV disease in the unsampled tissue—an important issue in studies that do not require at least subtotal resection for eligibility. However, we reviewed the images for all patients that underwent biopsy only. Among 13 patients, all had unresectable tumors because of involvement of deep structures or motor or language cortex, thus limiting the sample available for diagnostic and molecular evaluations. Regarding radiographic features, only 3 patients had heterogeneously enhancing lesions typical for GBM. Three others had non-enhancing tumors. The remainder had largely non-enhancing tumors with faint or punctate areas of enhancement consistent with anaplastic astrocytoma, one of whom underwent re-operation after chemoradiation which re-confirmed anaplastic histology.
Our sample size was too small to delineate the different effects of the extent of surgical resection, varying histology (astrocytoma vs mixed), MGMT methylation, 1p19q deletion, and the typical prognostic factors such as age and KPS. However, our patients were relatively young and had an excellent performance status, making it less likely those factors confounded results. While GBM has four (at least) well-described molecular profile categories, such an analysis has not yet separated the almost certain subgroups of anaplastic glioma [21]. Recent studies have indicated that isocitrate dehydrogenase 1 (IDH1) mutation may confer a favorable prognosis in patients with anaplastic gliomas, but its definitive role remains to be seen [19, 22]. A limitation of our study is missing IDH mutational information. IDH mutational testing was not performed routinely at the time this study was conducted. In addition, finite tissue was available from those who underwent biopsy, and molecular analysis was focused on MGMT promoter methylation, the biomarker of interest at the time. Accordingly, the challenges in obtaining archival tissue from external sites, and the costs associated with analysis, proved impractical for a post hoc analysis of a small exploratory cohort of a larger trial. Moreover, we also did not analyze tissue for other potentially prognostic biomarkers, such as Glioma CpG island methylator phenotype [23, 24] or myriad others. Varying representation of different subgroups within large and small cohort studies may well explain the highly variable PFS and OS reported for this group of patients.
Supplementary Material
Acknowledgments
Financial support was received from Merck (formerly Schering Plough) for MGMT analyses and for partial study costs. K.S.P. and A.S.R. were supported in part by P30 CA008748.
Footnotes
Electronic supplementary material The online version of this article (doi:10.1007/s11060-015-1771-8) contains supplementary material, which is available to authorized users.
Conflict of interest ABL previously received honoraria from Merck/Schering-Plough for speaking and consulting. LEA is currently a full time employee of F-Hoffman-La Roche Ltd. LN, KSP, ASR, JTH, EP, SGB, and LMD have no relevant disclosures.
References
- 1.Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ, Belanger K, Brandes AA, Marosi C, Bogdahn U, Curschmann J, Janzer RC, Ludwin SK, Gorlia T, Allgeier A, Lacombe D, Cairncross JG, Eisenhauer E, Mirimanoff RO. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med. 2005;352:987–996. doi: 10.1056/NEJMoa043330. [DOI] [PubMed] [Google Scholar]
- 2.Newlands ES, Blackledge GR, Slack JA, Rustin GJ, Smith DB, Stuart NS, Quarterman CP, Hoffman R, Stevens MF, Brampton MH, et al. Phase I trial of temozolomide (CCRG 81045: M&B 39831: NSC 362856) Br J Cancer. 1992;65:287–291. doi: 10.1038/bjc.1992.57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Tolcher AW, Gerson SL, Denis L, Geyer C, Hammond LA, Patnaik A, Goetz AD, Schwartz G, Edwards T, Reyderman L, Statkevich P, Cutler DL, Rowinsky EK. Marked inactivation of O6-alkylguanine-DNA alkyltransferase activity with protracted temozolomide schedules. Br J Cancer. 2003;88:1004–1011. doi: 10.1038/sj.bjc.6600827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Brada M, Stenning S, Gabe R, Thompson LC, Levy D, Rampling R, Erridge S, Saran F, Gattamaneni R, Hopkins K, Beall S, Collins VP, Lee SM. Temozolomide versus procarbazine, lomustine, and vincristine in recurrent high-grade glioma. J Clin Oncol. 2010;28:4601–4608. doi: 10.1200/JCO.2009.27.1932. [DOI] [PubMed] [Google Scholar]
- 5.Gilbert MR, Wang M, Aldape KD, Stupp R, Hegi ME, Jaeckle KA, Armstrong TS, Wefel JS, Won M, Blumenthal DT, Mahajan A, Schultz CJ, Erridge S, Baumert B, Hopkins KI, Tzuk-Shina T, Brown PD, Chakravarti A, Curran WJ, Jr, Mehta MP. Dose-dense temozolomide for newly diagnosed glioblastoma: a randomized phase III clinical trial. J Clin Oncol. 2013;31:4085–4091. doi: 10.1200/JCO.2013.49.6968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bouterfa H, Picht T, Kess D, Herbold C, Noll E, Black PM, Roosen K, Tonn JC. Retinoids inhibit human glioma cell proliferation and migration in primary cell cultures but not in established cell lines. Neurosurgery. 2000;46:419–430. doi: 10.1097/00006123-200002000-00029. [DOI] [PubMed] [Google Scholar]
- 7.Rodts GE, Jr, Black KL. Trans retinoic acid inhibits in vivo tumour growth of C6 glioma in rats: effect negatively influenced by nerve growth factor. Neurol Res. 1994;16:184–186. doi: 10.1080/01616412.1994.11740223. [DOI] [PubMed] [Google Scholar]
- 8.Costa SL, Paillaud E, Fages C, Rochette-Egly C, Plassat JL, Jouault H, Perzelova A, Tardy M. Effects of a novel synthetic retinoid on malignant glioma in vitro: inhibition of cell proliferation, induction of apoptosis and differentiation. Eur J Cancer. 2001;37:520–530. doi: 10.1016/s0959-8049(00)00430-5. [DOI] [PubMed] [Google Scholar]
- 9.Wismeth C, Hau P, Fabel K, Baumgart U, Hirschmann B, Koch H, Jauch T, Grauer O, Drechsel L, Brawanski A, Bogdahn U, Steinbrecher A. Maintenance therapy with 13-cis retinoid acid in high-grade glioma at complete response after first-line multimodal therapy—a phase-II study. J Neurooncol. 2004;68:79–86. doi: 10.1023/b:neon.0000024748.26608.2f. [DOI] [PubMed] [Google Scholar]
- 10.Clarke JL, Iwamoto FM, Sul J, Panageas K, Lassman AB, DeAngelis LM, Hormigo A, Nolan CP, Gavrilovic I, Karimi S, Abrey LE. Randomized phase II trial of chemoradiotherapy followed by either dose-dense or metronomic temozolomide for newly diagnosed glioblastoma. J Clin Oncol. 2009;27:3861–3867. doi: 10.1200/JCO.2008.20.7944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Macdonald DR, Cascino TL, Schold SC, Jr, Cairncross JG. Response criteria for phase II studies of supratentorial malignant glioma. J Clin Oncol. 1990;8:1277–1280. doi: 10.1200/JCO.1990.8.7.1277. [DOI] [PubMed] [Google Scholar]
- 12.Grauer O, Pascher C, Hartmann C, Zeman F, Weller M, Proescholdt M, Brawanski A, Pietsch T, Wick W, Bogdahn U, Hau P. Temozolomide and 13-cis retinoic acid in patients with anaplastic gliomas: a prospective single-arm monocentric phase-II study (RNOP-05) J Neurooncol. 2011;104:801–809. doi: 10.1007/s11060-011-0548-y. [DOI] [PubMed] [Google Scholar]
- 13.Prados MD, Seiferheld W, Sandler HM, Buckner JC, Phillips T, Schultz C, Urtasun R, Davis R, Gutin P, Cascino TL, Greenberg HS, Curran WJ., Jr Phase III randomized study of radiotherapy plus procarbazine, lomustine, and vincristine with or without BUdR for treatment of anaplastic astrocytoma: final report of RTOG 9404. Int J Radiat Oncol Biol Phys. 2004;58:1147–1152. doi: 10.1016/j.ijrobp.2003.08.024. [DOI] [PubMed] [Google Scholar]
- 14.Wick W, Hartmann C, Engel C, Stoffels M, Felsberg J, Stock-hammer F, Sabel MC, Koeppen S, Ketter R, Meyermann R, Rapp M, Meisner C, Kortmann RD, Pietsch T, Wiestler OD, Ernemann U, Bamberg M, Reifenberger G, von Deimling A, Weller M. NOA-04 randomized phase III trial of sequential radiochemotherapy of anaplastic glioma with procarbazine, lomustine, and vincristine or temozolomide. J Clin Oncol. 2009;27:5874–5880. doi: 10.1200/JCO.2009.23.6497. [DOI] [PubMed] [Google Scholar]
- 15.Brandes AA, Nicolardi L, Tosoni A, Gardiman M, Iuzzolino P, Ghimenton C, Reni M, Rotilio A, Sotti G, Ermani M. Survival following adjuvant PCV or temozolomide for anaplastic astrocytoma. Neuro Oncol. 2006;8:253–260. doi: 10.1215/15228517-2006-005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hildebrand J, Gorlia T, Kros JM, Afra D, Frenay M, Omuro A, Stupp R, Lacombe D, Allgeier A, van den Bent MJ. Adjuvant dibromodulcitol and BCNU chemotherapy in anaplastic astrocytoma: results of a randomised European Organisation for Research and Treatment of Cancer phase III study (EORTC study 26882) Eur J Cancer. 2008;44:1210–1216. doi: 10.1016/j.ejca.2007.12.005. [DOI] [PubMed] [Google Scholar]
- 17.Combs SE, Nagy M, Edler L, Rausch R, Bischof M, Welzel T, Debus J, Schulz-Ertner D. Comparative evaluation of radiochemotherapy with temozolomide versus standard-of-care postoperative radiation alone in patients with WHO grade III astrocytic tumors. Radiother Oncol. 2008;88:177–182. doi: 10.1016/j.radonc.2008.03.005. [DOI] [PubMed] [Google Scholar]
- 18.Kim YH, Park CK, Cho WH, Kim IA, Moon S, Choe G, Park SH, Kim IH, Kim DG, Jung HW, Lee MM, Bae SH, Cha SH, Kim CY. Temozolomide during and after radiation therapy for WHO grade III gliomas: preliminary report of a prospective multicenter study. J Neurooncol. 2011;103:503–512. doi: 10.1007/s11060-010-0404-5. [DOI] [PubMed] [Google Scholar]
- 19.Minniti G, Scaringi C, Arcella A, Lanzetta G, Di Stefano D, Scarpino S, Bozzao A, Pace A, Villani V, Salvati M, Esposito V, Giangaspero F, Enrici RM. IDH1 mutation and MGMT methylation status predict survival in patients with anaplastic astrocytoma treated with temozolomide-based chemoradiotherapy. J Neurooncol. 2014;118:377–383. doi: 10.1007/s11060-014-1443-0. [DOI] [PubMed] [Google Scholar]
- 20.DeAngelis LM. Anaplastic glioma: how to prognosticate outcome and choose a treatment strategy. [corrected] J Clin Oncol. 2009;27:5861–5862. doi: 10.1200/JCO.2009.24.5985. [DOI] [PubMed] [Google Scholar]
- 21.Verhaak RG, Hoadley KA, Purdom E, Wang V, Qi Y, Wilkerson MD, Miller CR, Ding L, Golub T, Mesirov JP, Alexe G, Lawrence M, O’Kelly M, Tamayo P, Weir BA, Gabriel S, Winckler W, Gupta S, Jakkula L, Feiler HS, Hodgson JG, James CD, Sarkaria JN, Brennan C, Kahn A, Spellman PT, Wilson RK, Speed TP, Gray JW, Meyerson M, Getz G, Perou CM, Hayes DN Cancer Genome Atlas Research N. Integrated genomic analysis identifies clinically relevant subtypes of glioblastoma characterized by abnormalities in PDGFRA, IDH1, EGFR, and NF1. Cancer cell. 2010;17:98–110. doi: 10.1016/j.ccr.2009.12.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hartmann C, Hentschel B, Wick W, Capper D, Felsberg J, Simon M, Westphal M, Schackert G, Meyermann R, Pietsch T, Reifenberger G, Weller M, Loeffler M, von Deimling A. Patients with IDH1 wild type anaplastic astrocytomas exhibit worse prognosis than IDH1-mutated glioblastomas, and IDH1 mutation status accounts for the unfavorable prognostic effect of higher age: implications for classification of gliomas. Acta Neuropathol. 2010;120:707–718. doi: 10.1007/s00401-010-0781-z. [DOI] [PubMed] [Google Scholar]
- 23.Noushmehr H, Weisenberger DJ, Diefes K, Phillips HS, Pujara K, Berman BP, Pan F, Pelloski CE, Sulman EP, Bhat KP, Verhaak RG, Hoadley KA, Hayes DN, Perou CM, Schmidt HK, Ding L, Wilson RK, Van Den Berg D, Shen H, Bengtsson H, Neuvial P, Cope LM, Buckley J, Herman JG, Baylin SB, Laird PW, Aldape K. Identification of a CpG island methylator phenotype that defines a distinct subgroup of glioma. Cancer Cell. 2010;17:510–522. doi: 10.1016/j.ccr.2010.03.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.van den Bent MJ, Gravendeel LA, Gorlia T, Kros JM, Lapre L, Wesseling P, Teepen JL, Idbaih A, Sanson M, Smitt PA, French PJ. A hypermethylated phenotype is a better predictor of survival than MGMT methylation in anaplastic oligodendroglial brain tumors: a report from EORTC study 26951. Clin Cancer Res. 2011;17:7148–7155. doi: 10.1158/1078-0432.CCR-11-1274. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.