Abstract
Social support from close relationship partners is an important resource for coping with stress, particularly during childhood. We discuss ethical challenges associated with studying stress and its social buffering in the laboratory, as well as emerging evidence regarding two potential neural substrates for the social buffering of stress: hypothalamic oxytocin activity and activation of areas in the prefrontal cortex associated with effective self-regulation. We also address the role of early-life social experiences in shaping brain development, as well as recommendations for practice and policy that would advance the ethical treatment of children and reduce social inequalities in early-life experiences and opportunities–e.g., investing in programs that prevent child maltreatment and facilitating access to high-quality child care for economically disadvantaged families. We also debate the ethical implications of using oxytocin nasal sprays to simulate the stress-reducing properties of social support and advise waiting for more evidence before recommending their use.
Keywords: social support, stress, HPA axis, cortisol, prefrontal cortex, oxytocin
Close Relationships as Stress Buffers
The importance of social relationships for most aspects of human development has been documented by psychologists for over a century. We know that human beings have a fundamental need to form emotional connections with others, and experience distress and loss when they are socially isolated or in poor-quality relationships. Furthermore, there is a growing body of evidence that social support from those we are close to has wide-ranging benefits for physical and mental health (Cohen, 2004). Social support has been defined as “a social network’s provision of psychological and material resources intended to benefit an individual’s ability to cope with stress” (Cohen, 2004, p. 676). The science of social support is increasingly moving beyond simple associations of social support with physical or mental health benefits to research designed to uncover the neurobiological mechanisms through which social support operates. Of critical importance to this review is growing evidence that social support influences physical and mental health through dampening physiological stress responses, a phenomenon termed the social buffering of stress. However, laboratory experiments studying stress and its regulation by social relationships pose a number of ethical challenges. The first is identifying stressors that are strong enough to be effective but still manageable, so that they are ethical to conduct for the sole purpose of scientific discovery. This is particularly important when examining vulnerable populations such as children and adolescents, or individuals who already experience numerous types of adversity. The second issue concerns researchers’ ethical responsibility to not only disseminate their findings very broadly so as to ultimately benefit the disadvantaged populations they study, but also to report their data carefully and acknowledge the limitations surrounding their conclusions. In the next sections we describe some of the most common study designs for examining the social buffering of stress, summarize recent findings regarding the neurobiology of social buffering, then conclude with a number of recommendations for policy and practice that would advance the ethical treatment of children in our society.
Studying Stress in the Laboratory in an Ethical Manner
Stress can be defined as a “real or interpreted threat to the physiological or psychological integrity of an individual which results in physiological and/or behavioral responses” (McEwen, 2000, p. 508). These threats can be psychological (e.g., threat of social evaluation during a job interview) or physical (e.g., infection, cold), but both types of stressors place demands on the organism that require mobilization of energy and activation of neural and physiological systems needed to cope with the threat. Mobilization of these systems is supported by the activation of the body’s stress-response systems. Along with the sympathoadrenal system (producing adrenaline) that fuels fight-or-flight responses, the hypothalamic-pituitary-adrenal (HPA) axis forms the core effector arm of the mammalian stress system. For example, in situations that threaten the self, physically or psychologically, limbic and cortical regions (e.g., amygdala, hippocampus, prefrontal cortex –PFC) relay signals over multi-synaptic pathways to the paraventricular nucleus of the hypothalamus. There, the production of corticotropin releasing hormone (CRH) augmented by arginine vasopressin (AVP) travels through a portal connection to the anterior of the pituitary gland causing the production and release of adrenocorticotropic hormone (ACTH). ACTH in circulation stimulates cells in the cortex of the adrenal gland to produce cortisol (Ulrich-Lai & Herman, 2009). Cortisol enters all the cells in the body and brain and most cells have receptors for the hormone. In this way, it produces broad effects, mobilizing energy for prolonged action, helping to lay down memory for the events that were threatening, and counteracting activity of the immune system. These actions support our ability to grapple with acutely stressful experiences, which is adaptive in the short-term. But when the system is continuously activated due to chronic stressors, excessive “wear and tear” is placed on the body (McEwen, 2008). Thus, individuals who are buffered from undue elevations in stress hormones are likely going to enjoy better health in the long run, though the biological pathways for these effects are still being characterized.
Psychobiological experiments have used laboratory-based stressors and social support manipulations to test hypotheses related to the presumed role of close relationships. For instance, the Trier Social Stress Test (Kirschbaum, Pirke, & Hellhammer, 1993) is one commonly used stress paradigm, where research volunteers are asked to deliver a speech in front of a video camera and a panel of two or three judges who are purportedly evaluating their performance. Public speaking is an ideal and ethical laboratory stressor for several reasons. First, it reliably activates neuroendocrine stress responses and is rated as stressful by older children, adolescents, and adults. Second, public speaking opportunities occur naturally in many school and work contexts. Thus, despite being dreaded by most, this experience is ethical to conduct in a laboratory setting because it is within the range of daily experiences that people might encounter. Furthermore, exposure is one of the best strategies for managing fear of public speaking, thus it could be argued that participants might even derive some benefits from these types of tasks. Social buffering studies with adults have also used the threat of mild electric shock or exposure to mildly painful stimuli to examine the effect of social partners on neural activity during these procedures (e.g., Coan, Schaefer, & Davidson, 2006; Eisenberger et al., 2011), but these mild levels of threat or pain are always rated by participants as tolerable despite being aversive –e.g., a rating of 10 on a pain scale ranging from 0 to 20. Importantly, laboratory stress, threat or pain manipulations typically take special caution to allow participants to opt out of these procedures at any time, or interrupt the task if participants appear to be experiencing excessive distress (e.g., freezing, crying), even when they do not explicitly request it.
These types of laboratory procedures have allowed researchers to ask important questions about social relationships as anti-stress remedies. For instance, a landmark study in social buffering research has shown that receiving support from a romantic partner while the participant is preparing to give a speech can significantly reduce the production of cortisol assessed in saliva samples (Kirschbaum, Klauer, Filipp, & Hellhammer, 1995), even when the research participant is alone and unsupported when actually giving the speech. Findings like these pose an intriguing question for neuroscientists and psychologists alike: what are the neural pathways through which social stress buffering operates? Is it that the presence of the supportive partner makes giving the speech less scary? Or is it still scary, but some other pathway blocks stress hormone activity because the person knows that they are not alone? Research with children and adults, as well as inferences drawn from animal models of behavior have begun providing some clues regarding the underlying neurobiology, though more research is clearly needed to fully specify these pathways. We briefly review some of this research next.
The Benefits of Parental Support for Children’s Stress Reduction and Brain Development
Children develop in an environment of relationships and thrive when adults who care for them interact with them in a sensitive and responsive manner. The metaphor of “serve and return” has been used to characterize the ways in which parents, teachers and other important figures in children’s lives can promote adequate brain development by providing rich emotional, linguistic, and behavioral responses to children’s signals (National Scientific Council on the Developing Child, 2004). Responsive early caregiving also shapes children’s interpersonal skills and their ability to draw upon social support later in life. For instance, the Minnesota Longitudinal Study of Parents and Children –an ongoing 35-year longitudinal study of a high-risk poverty sample-has provided evidence that children who had experienced secure relationships with their parents during infancy were more likely to have competent social interactions with peers across childhood (assessed at ages 4-5, 8, 12), according to both teacher and self-ratings (Sroufe, Egeland, Carlson, & Collins, 2005). In turn, these measures of peer competence predicted measures of perceived social support and socio-emotional functioning in late adolescence at age 19, as well as positive, secure representations of romantic partners in young adulthood (ages 26-28). Furthermore, infants who were securely attached to their mother or primary caregiver were more likely to exhibit high emotion-regulation skills, positive affect, and high self-esteem later in childhood and observational measures showed that they were more likely to be self-reliant in the classroom.
Supportive early relationships also serve the important function of buffering children’s stress responses (Hostinar, Sullivan, & Gunnar, 2014), which allows them to confront situations that are upsetting or even frightening. In the absence of stress-buffering, these situations would produce elevated stress hormones that could shape a more fearful nervous system and lay down memories that might lead them to try to avoid those situations in the future. For example, in a series of studies (reviewed in Gunnar & Donzella, 2002), children who were distressed by experiences such as receiving their infant immunizations, meeting a clown, or being approached by a loud toy robot, did not produce elevations in cortisol if they were accompanied by a parent with whom they had a secure emotional relationship. If the attachment relationship was insecure, however, the parent’s presence was not an effective stress buffer. Parental support retains the capacity to block activation of the HPA axis into late childhood, but appears to lose potency during adolescence, perhaps in preparation for the child needing to mature and leave the natal home (Hostinar, Johnson, & Gunnar, 2015).
Presumably, these stress-buffering effects by parent support compound across the entirety of childhood and result in significantly lower exposure of the brain to circulating stress hormones, with implications for neural development. For instance, one study with elderly adults revealed that those who reported receiving greater quality of early parental care had larger hippocampal volumes, which were in turn associated with a lower cortisol output in response to the Montreal Imaging Stress Task (Engert, Buss, & Pruessner, 2010), a variation of the Trier Social Stress Test adapted for the MRI scanner. The hippocampus has inhibitory projections to the HPA axis and plays an important role in turning off cortisol production (Ulrich-Lai & Herman, 2009). Additionally, rodent models also suggest that the integrity of the hippocampus is threatened by early-life stress and high levels of circulating stress hormones, making this brain structure a likely neural substrate for carrying at least some of the effects of early experiences forward.
However, more research is needed to describe how normal variations in parenting help provide the grist that feeds the development of brain architecture, since understandably most of the work in this area has been conducted with clinical populations that experience extreme types of adverse care such as maltreatment (physical, sexual, emotional, or neglect) and complete deprivation of parental care (i.e., orphanage rearing). Indeed, a growing body of evidence supports the damaging role of child maltreatment for brain development (for reviews, see Belsky & de Haan, 2011; De Bellis, 2001). Neuroimaging studies show that these children, who are frequently diagnosed with Post-Traumatic Stress Disorder, tend to exhibit smaller total cerebral volume, as well as reductions in prefrontal cortex, prefrontal white matter, corpus callosum, and right temporal lobe volumes compared to non-maltreated children. It has been hypothesized that these effects are, at least in part, due to dysregulated patterns of stress-response system functioning (De Bellis, 2001). Maltreatment by family members often serves as a “double whammy”, not only exposing children to chronic, severe, and often unpredictable threats, but also depriving them of a blueprint for soothing social interactions that might have provided an avenue for coping with stress. Consequently, many maltreated children exhibit reduced social effectiveness and may elicit peer rejection, thus depriving them of the opportunity to experience social buffering of their stress responses. Across the lifespan, they are at an increased risk of deteriorating mental and physical health.
Studies of orphanage-reared children tell a similar story, where early-life stress is associated with both dysregulated patterns of stress-response system functioning (Koss, Hostinar, Donzella, & Gunnar, 2014) and with alterations in brain structure and function (Tottenham, 2012). For instance, children adopted internationally from orphanage care exhibit increased amygdala volume and impaired prefrontal regulation of the amygdala compared to normative samples (Gee et al., 2013). These effects are mediated by cortisol, which supports the idea that stress-induced alterations of the HPA axis shape amygdala-prefrontal circuitry (Gee et al., 2013). This is a potential explanatory mechanism for heightened levels of anxiety and related emotional problems observed in this population.
Furthermore, children reared in orphanage care show alterations in oxytocin and arginine vasopressin activity, two neuropeptides known to be involved in social and affiliative behaviors. For instance, one study of 4-5-year-olds who had been adopted internationally from orphanage care roughly 3 years prior revealed that these children had lower levels of basal arginine vasopressin and did not elevate urinary oxytocin levels after a physical contact game with their mothers, while children developing under typical conditions did increase oxytocin production when they were in contact with their mothers (Wismer Fries, Ziegler, Kurian, Jacoris, & Pollak, 2005). Importantly, contact with the mother also lowered the comparison children’s cortisol levels. Children adopted from orphanages did not show this cortisol reduction with the parent, and indeed, showed an increase in cortisol after playing with their mothers, similar to what they showed when playing with a stranger (Wismer Fries, Shirtcliff, & Pollak, 2008). These findings suggest the possibility that children who did not experience a typical early relationship with a caregiver might not be able to derive the same stress relief from parent-child interactions as their non-adopted counterparts, even after spending a few years in the new family. The results also hint at the potential role of oxytocin in explaining the social-buffering of the HPA axis, as discussed in more detail next.
The Role of Oxytocin in the Social Buffering of the HPA Axis
The neuropeptide oxytocin has been implicated in many social behaviors and emotions, including empathy to a stranger’s distress, altruistic behavior, the ability to infer others’ thoughts or emotions, trust and generosity in monetary games, receiving a massage, and natural variations in both maternal and paternal behaviors with infants (reviewed in Hostinar, Sullivan, et al., 2014). Animal models of behavior have also supported a stress-attenuating role for oxytocin. For instance, administering oxytocin directly into the central nervous system inhibits the activity of the HPA axis in several mammalian species –e.g., prairie voles, guinea pigs (Hennessy, Kaiser, & Sachser, 2009). Importantly, recent evidence from an experiment with prairie voles also showed that introducing an oxytocin receptor antagonist in the paraventricular nucleus of the hypothalamus blocks the socially-buffering role of social contact for HPA stress responses in response to immobilization (Smith & Wang, 2014). In humans, intranasal administration of oxytocin paired up with social support provided in the laboratory also seems to be able to lower salivary cortisol levels produced in response to the Trier Social Stress Test (Heinrichs, Baumgartner, Kirschbaum, & Ehlert, 2003). These converging lines of evidence suggest that hypothalamic oxytocin activity might be one of the underlying mechanisms for the stress-buffering effects of social support, but more research is needed to fully characterize the neurocircuitry of social buffering in humans. We will discuss some implications and caveats derived from these findings in the final section on Implications for Practice and Policy. It is also important to note that hypothalamic oxytocin activity may not be the only underlying mechanism for social buffering, and there is growing evidence implicating other biological mediators as well. For instance, we discuss the potential involvement of the prefrontal cortex next.
The Prefrontal Cortex and Social Buffering
Most of the neuroimaging work on social buffering of negative states in humans has been conducted in adults, even though it has not always assessed HPA stress responses. These studies typically show that the presence of a close companion or of stimuli associated with them (e.g., an image of a romantic partner) can significantly reduce neural activity in regions thought to process negative affect, threat, fear, or pain. For instance, in one study of threat regulation, adult women were subjected to the threat of electric shock and had their hand held by their husband, a stranger or did not have their hand held by anyone (Coan et al., 2006). For women who rated themselves as happier and more supported in their marriages, holding their husband’s but not a stranger’s hand was correlated with lower threat-induced activation in the right anterior insula and superior frontal gyrus. It has been theorized that attachment figures serve as safety signals, activating neural regions in the prefrontal cortex that are known to be associated with the top-down control and regulation of negative emotion, like the ventromedial PFC (vmPFC) (Eisenberger et al., 2011). For instance, women who were shown images of their long-term romantic partners reported less pain in response to heat stimuli and exhibited lower levels of pain-related neural activation (dorsal anterior cingulate cortex and anterior insula) as well as increased vmPFC activation, which correlated with relationship duration and perceived partner support (Eisenberger et al., 2011). This finding can be interpreted as providing evidence that attachment figures serve as safety cues which can prime subsequent responses to distressing events, and furthermore that the prefrontal cortex is involved in the social buffering of neural responses to such distressing events.
These neural social buffering effects may have implications for peripheral stress physiology. For instance, one study by Taylor et al. (2008) used fMRI to reveal greater right ventrolateral PFC activation and lower amygdala activation during a threat regulation task for participants with high psychosocial resources. These were also the people who produced smaller cortisol responses to the Trier Social Stress Test. Psychosocial resources were defined as a composite measure of self-esteem, optimism, and a sense of psychological control/mastery. Interestingly, this study also showed that individuals with high psychosocial resources were not different in their sensitivity to detect threat, but only in the regulation of threat. Given the previously mentioned links between self-esteem and one’s social history, it is possible that psychosocial resources indexed participants’ access to social support, though this was not directly assessed in this study.
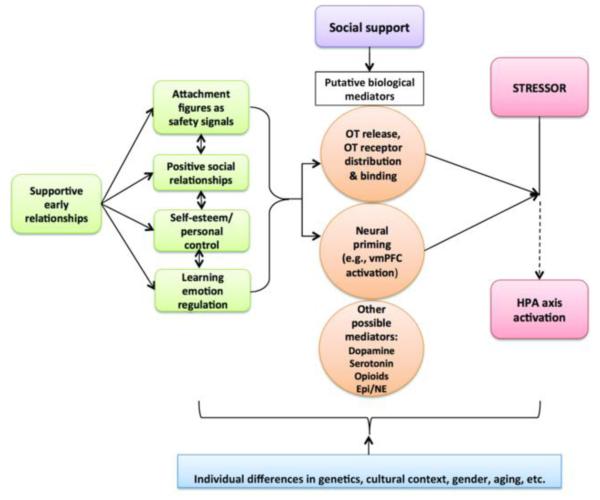
In a study that speaks to the relevance of neural buffering effects for cortisol reactivity, individuals having frequent interactions with supportive others across a 10-day period prior to testing showed both reduced salivary cortisol reactivity to the Trier Social Stress Test and diminished dorsal anterior cingulate cortex and Brodmann’s Area 8 activation (frontal cortex) in response to a social rejection paradigm (Eisenberger, Taylor, Gable, Hilmert, & Lieberman, 2007). The social rejection paradigm used in this study is called Cyberball and it is frequently used in a neuroimaging environment. It involves playing a computer game of ball tossing with two virtual (pre-programmed) others, and over the course of the game the two other players stop throwing you the ball, thus excluding you from the game. The study also found that neural reactivity to Cyberball was associated with cortisol reactivity to the Trier Social Stress Test, potentially suggesting that individuals who lack access to social support may be vulnerable to greater neural reactivity to social exclusion as well as higher HPA reactivity to a social-evaluative threat, like the Trier. Despite these emerging findings, more neuroimaging research is needed to specifically test which neural regions are activated during the buffering of the HPA axis by social stimuli. To generate new hypotheses and guide future research in this area, we recently proposed a theoretical model of the social buffering of the HPA axis (see Figure 1) across development (Hostinar, Sullivan, et al., 2014). We briefly summarize some of the main tenets of this model next.
Figure 1.
A developmental working model of the social buffering of the hypothalamic–pituitary–adrenal (HPA) axis in humans. Early-life social experiences are thought to shape the later effectiveness of social buffering of the HPA axis. Additionally, two sets of biological mechanisms are proposed to mediate the effect of social support on HPA reactivity to stressors: oxytocinergic functioning, including patterns of oxytocin release and receptor distribution; and PFC-based safety signaling/neural priming by stimuli associated with attachment figures. The role of other biological mediators (e.g., dopamine, serotonin, opioids, epinephrine, norepinephrine) has been suggested by some studies with nonhuman animals, but more research is needed to characterize their role in the social buffering of the HPA axis in humans. OT = oxytocin; vmPFC = ventromedial prefrontal cortex; Epi = epinephrine; NE = norepinephrine. Reprinted with permission from Hostinar, C. E., Sullivan, R. M., & Gunnar, M. R. (2014). Psychobiological mechanisms underlying the social buffering of the HPA axis: A review of animal models and human studies across development. Psychological Bulletin, 140(1), 256-282. Copyright 2014 by the American Psychological Association.
A Developmental Working Model of the Stress-Dampening Effects of Social Support
Two decades since the landmark social buffering finding that support from a romantic partner before Trier Social Stress Test significantly reduced cortisol reactivity in adult males (Kirschbaum et al., 1995), there are still many research gaps that prevent us from definitively understanding how the brain communicates to the body to dampen stress hormone production when social support is available. We proposed two potential underlying mechanisms: hypothalamic oxytocin activity and safety signaling by attachment figures, which involves activation of PFC areas frequently associated with the effective regulation of negative affective states. We believe that the close interactions between neural systems underlying social behavior and those implementing stress responses are likely to be evolutionarily selected characteristics of humans and other species, given the importance of conspecifics for survival. Support for this argument includes the fact that attachment to caregivers and retreating to them for safety is a nearly universal phenomenon, with human infants having strong propensities to form attachments even when caregivers are abusive. However, not all attachment figures provide the same degree of responsiveness to the infant’s needs, leading the child to form relationships of different degrees of trust with different attachment figures (Ainsworth, Bell, & Stayton, 1974). Furthermore, neuroimaging studies show that images of significant others activate the vmPFC (Eisenberger et al., 2011), which has been implicated in learning safety cues and the extinction of fear conditioning, suggesting a neural basis for the process of associating specific individuals with anti-threat and anti-stress cues.
An important tenet of our model is that early caregiving experiences shape the later effectiveness for the social buffering of stress. As discussed above, childhood social experiences lay the foundation for later social competence with peers and romantic partners, who can become attachment figures in their own right, which in turn promotes positive emotional outcomes and higher levels of self-esteem and personal control. Children who experience adverse rearing conditions (e.g., maltreatment, deprivation of parental care in orphanage settings) show alterations in the structure and function of brain regions involved in emotional processing, like the amygdala, and sometimes exhibit reduced social effectiveness, which may negatively influence their ability to draw upon social support in times of stress. We argue that experiencing the stress-relieving effects of caregivers early on is critical for teaching the organism to seek social partners and may condition certain neural activation patterns that promote affiliative behaviors across development, thus serving as positive feedback signals in the process of becoming disposed to exhibit a buffering of stress by social stimuli later in development. A final point is that there are likely developmental changes in the social buffering of stress and perhaps also in the neurobiology involved. We hypothesize that the oxytocin-based social buffering mechanism is operative very early on, whereas PFC-based safety signaling likely has a more protracted development, requiring some level of neural maturation before emerging. However, more empirical work will be needed to fully test these ideas.
Recommendations for Policy and Practice to Promote the Ethical Treatment of Children
The influential and enduring role of supportive early childhood experiences for healthy brain and behavioral development suggests that society as a whole and individuals directly involved in child rearing have a moral obligation to promote the ethical treatment of children. In addition to this moral imperative, there are also utilitarian considerations drawn from economic analyses which have shown very high returns for investments in young disadvantaged children (for a review, see Heckman & Mosso, 2014). This is because the capacity for learning and neurobehavioral change is highest in the early years and decreases across development. Additionally, it has been highlighted that early experiences not only lay the foundation for lifelong mental and physical health, but they are very influential in skill development, which forms the basis for adult productivity (Heckman & Mosso, 2014). Additionally, it is more cost-effective for a society to prevent mental and physical diseases through programs that improve parenting and reduce childhood stress than to grapple with the consequences of numerous deleterious outcomes later.
As discussed above, the family climate is one of the most influential factors for children’s neurobehavioral development and as such a number of specific recommendations have been proposed for advancing child wellbeing through interventions that improve caregiving (for a more comprehensive list of recommendations, see the report by the National Scientific Council on the Developing Child, 2004). Firstly, interventions to prevent child maltreatment and improve parenting among at-risk families have been suggested as a highly effective way to improve mental health for both children and adults in the general population (De Bellis, 2001). The observation that parents at risk for abusing or neglecting their children often lack adequate social support has prompted psychologists to examine ways in which social support interventions may help buffer these parents against stress, improve their functioning, and serve child-protective goals as well (Thompson, 2014). The primary obstacles to this approach are that social isolation experienced by at-risk parents often has multiple complex causes and, furthermore, that social support afforded to them might be insufficient for preventing maltreatment unless it is also coupled with social monitoring and firm guidance to prevent abusive or neglectful behaviors towards their children (Thompson, 2014).
Despite these challenges, some solutions have been proposed for preventing child maltreatment. For instance, home visitation programs have shown initial promise in this arena (Thompson & Ontai, 2000). Such programs –e.g. The Maternal, Infant, and Early Childhood Home Visiting (MIECHV) program supported by the U.S. Department of Health and Human Services-provide in-home assistance to at-risk families, with the goal of preventing child abuse and neglect, encouraging positive parenting, and promoting child development and school readiness. Given the long-term societal benefits that can be derived from promoting the wellbeing of children and families, we believe that funding should be increased for evidence-based programs of this kind. Additionally, augmenting existing practices of Child Protective Services with mental health treatments for both parents and children has been recommended as an avenue for not only ameliorating children’s current circumstances but also preventing the intergenerational transmission of maltreatment –i.e., the elevated risk that maltreated children will grow up to abuse or neglect their own offspring (De Bellis, 2001). Multi-generational interventions that involve grandparents as an additional way to recruit social support for struggling parents may also be an effective intervention strategy (Thompson, 2014).
Second, the average quality of care for infants and children in the United States could be improved. Parental leave policies based on the Family and Medical Leave Act (FMLA) currently only provide 12 weeks of unpaid job-protected leave to care for a newborn, and only about half of American workers are eligible or able to take advantage of this benefit (Waldfogel, 2001). Unlike laws in other developed countries which guarantee paid leave and sometimes allow as much as 10-12 months of paid parental leave (Waldfogel, 2001), these FMLA provisions do not account for the critical need to allow parents and children time to form strong bonds in the first year of life. We also believe that policies guaranteeing the option of paid parental leave for all American workers would be the ethical and equitable solution to reduce current inequalities, whereby impoverished families and their children are disproportionately affected by status quo parental leave provisions. It is easy to envision how individuals from high socioeconomic strata might be able to secure superior caregiving conditions for their children regardless of parental leave policies, whereas lower income families might be left with few options. These early-life disparities in the quality of care received might then reverberate across development and accentuate social inequities in mental or physical health and academic or work success across the lifespan.
An additional way of reducing socioeconomic disparities would be to improve the quality of out-of-home child care facilities by increasing wages and benefits for child care workers, while also providing subsidies to allow disadvantaged families to access high-quality child care. Increasing wages for child care workers would minimize staff turnover and allow more consistency in caregivers, which would facilitate the formation of emotional bonds to specific individuals. We need not look further than some of the research on children adopted internationally from orphanage care reviewed previously to observe the long-term sequelae of inconsistent caregivers and of neglect caused by a high children-to-caregiver ratio. Even though this type of adverse rearing is admittedly much more extreme and severe than most poor-quality day care arrangements, the research findings reviewed here serve as a cautionary tale for how persistent the role of early experience can be, which may perhaps generalize to milder forms of deficient care.
Would it Be Ethical to Use Nasal Oxytocin Sprays to Simulate the Stress-Buffering Properties of Social Support?
The neuroscientific evidence we reviewed regarding hypothalamic oxytocin as a neural substrate for the social buffering of HPA stress reactivity might also inspire a number of clinical and practical applications. For instance, the availability of oxytocin nasal sprays, coupled with the emerging evidence that oxytocin may have a stress-attenuating role in humans, might prompt some to recommend its use as a pharmacological agent for stress reduction and enhancing social closeness. However, the possibility of pharmacologically manipulating feelings of social connectedness, trust, and belonging through use of oxytocin nasal sprays raises ethical concerns that are not negligible. The potential for misuse in order to harm individuals or influence them to act against their own best interest cannot be ruled out, and will need to be carefully weighed against any potential benefits in stress reduction, if enough evidentiary support will accumulate from future research to support such purported benefits. At the moment, we must caution that the scientific evidence is insufficient to warrant any such recommendations. The number of studies showing reductions in salivary cortisol levels in humans after use of the oxytocin nasal spray is extremely limited, the samples studied are fairly small, and these studies do not account for individual differences in genetics (e.g., oxytocin receptor polymorphisms) that may affect oxytocinergic function and, consequently, may influence individual responses to supplemental oxytocin in unpredictable ways. A second concern is that of potential receptor downregulation –i.e., many neurohormones have self-regulating feedback loops such that their number of receptors can decrease in response to increasing levels in order to maintain adequate levels of functioning. Thus, an unintended consequence of using synthetic oxytocin to reduce stress might be to decrease the number of available receptors and, over time, diminish cells’ sensitivity to oxytocin. Third, even though there are a number of ongoing clinical trials examining the extent to which oxytocin might alleviate symptoms of social dysfunction in disorders such as autism or schizophrenia, this work will not tell us about the effect on individuals without these disorders. Indeed, recent research has begun suggesting that oxytocin treatment may be a “double-edged sword”, having a number of unanticipated side effects (Miller, 2013). For instance, intranasal oxytocin sprays seem to promote friendly behaviors towards familiar individuals, but prompt unfriendly behavior towards strangers and ethnocentrism. These effects will need to be weighed against the potential benefits in stress reduction, if the latter will ultimately be substantiated by future research. Lastly, the vast majority of studies administering oxytocin intranasally in humans have been conducted with adults, and little is known about its long-term effects on the developing brain. Prominent researchers who are studying the role of oxytocin in developing animals have shown that the effect of such treatments depend on their dosage, duration, and developmental timing (Bales & Perkeybile, 2012), suggesting that more evidence is needed and additional caution needs to be exercised before proposing oxytocin use in children and adolescents. There are many unanswered questions in this area and more research is needed to characterize the role of both endogenous and exogenously-administered oxytocin in social behavior and in the social buffering of stress responses.
Final Remarks
Stress is an inherent aspect of life, and likely one that cannot –and arguably should not-be eradicated. This is because mild and manageable forms of stress can be beneficial, allowing us to mobilize energy for grappling with diverse challenges and to adapt to a constantly changing environment. It is the chronic, severe and unmanageable types of stress that take a toll on physical and mental health, particularly when they occur during childhood (e.g., child maltreatment). Thus, interventions recruiting social support in its various forms for the purpose of dampening physiological stress reactivity for children and adults constitute an important avenue to pursue. However, we must also acknowledge that there are numerous obstacles to harnessing the benefits of social support. For instance, a 2014 public survey conducted by NPR, the Robert Wood Johnson Foundation and the Harvard School of Public Health titled “The Burden of Stress in America” revealed that 45% of adults in the United States reported that stress makes it more difficult to get along with their family members. We have also reviewed evidence that children exposed to early-life social neglect may not experience the full benefits of support later in life. Thus, it seems that populations experiencing chronic stress, who are in greatest need of social support, seem to also face the greatest barriers to accessing it. Research has made undeniable advances in understanding the detrimental effects of stress on mental and physical health, as well as the potentially protective qualities of social support and the neurobiological mechanisms through which it may operate. The next challenge for researchers, practitioners, and policymakers will be to understand and reduce the barriers to accessing and benefitting from social support during stressful times, especially for vulnerable populations. Furthermore, as discussed above, society has both a moral imperative and long-term economic incentives to provide increased assistance to disadvantaged families with the goal of reducing family stress, preventing child maltreatment, promoting positive parenting, and supporting child development and school readiness. These measures will likely bring us closer to leveling the playing field of early-life experiences and minimizing social inequalities in mental and physical health across the lifespan.
Acknowledgements
First author’s effort on this manuscript was supported by a grant from the National Institute of Child Health and Human Development (F32HD078048), whereas the second author was supported by a grant from the National Science Foundation (1439258). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health or National Science Foundation.
Contributor Information
Camelia E. Hostinar, Institute for Policy Research, Northwestern University
Megan R. Gunnar, Institute of Child Development, University of Minnesota
References
- Ainsworth MDS, Bell SM, Stayton DJ. Infant–mother attachment and social development: Socialisation as a product of reciprocal responsiveness to signals. In: Richards MPM, editor. The integration of a child into a social world. Cambridge University Press; New York, NY: 1974. pp. 99–137. [Google Scholar]
- Bales KL, Perkeybile AM. Developmental experiences and the oxytocin receptor system. Hormones and Behavior. 2012;61(3):313–9. doi: 10.1016/j.yhbeh.2011.12.013. doi:10.1016/j.yhbeh.2011.12.013. [DOI] [PubMed] [Google Scholar]
- Belsky J, de Haan M. Annual Research Review: Parenting and children’s brain development: The end of the beginning. Journal of Child Psychology and Psychiatry, and Allied Disciplines. 2011;52(4):409–28. doi: 10.1111/j.1469-7610.2010.02281.x. doi:10.1111/j.1469-7610.2010.02281.x. [DOI] [PubMed] [Google Scholar]
- Coan JA, Schaefer HS, Davidson RJ. Lending a hand: Social regulation of the neural response to threat. Psychological Science. 2006;17(12):1032–9. doi: 10.1111/j.1467-9280.2006.01832.x. doi:10.1111/j.1467-9280.2006.01832.x. [DOI] [PubMed] [Google Scholar]
- Cohen S. Social relationships and health. The American Psychologist. 2004;59(8):676–84. doi: 10.1037/0003-066X.59.8.676. doi:10.1037/0003-066X.59.8.676. [DOI] [PubMed] [Google Scholar]
- De Bellis MD. Developmental traumatology: The psychobiological development of maltreated children and its implications for research, treatment, and policy. Development and Psychopathology. 2001;13(3):539–564. doi: 10.1017/s0954579401003078. doi:10.1017/S0954579401003078. [DOI] [PubMed] [Google Scholar]
- Eisenberger NI, Master SL, Inagaki TK, Taylor SE, Shirinyan D, Lieberman MD, Naliboff BD. Attachment figures activate a safety signal-related neural region and reduce pain experience. Proceedings of the National Academy of Sciences of the United States of America. 2011;108(28):11721–6. doi: 10.1073/pnas.1108239108. doi:10.1073/pnas.1108239108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eisenberger NI, Taylor SE, Gable SL, Hilmert CJ, Lieberman MD. Neural pathways link social support to attenuated neuroendocrine stress responses. NeuroImage. 2007;35(4):1601–12. doi: 10.1016/j.neuroimage.2007.01.038. doi:10.1016/j.neuroimage.2007.01.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Engert V, Buss C, Pruessner JC. Investigating the association between early life parental care and stress responsivity in adulthood. Developmental Neuropsychology. 2010;35(5):570–581. doi: 10.1080/87565641.2010.494752. doi:10.1080/875656412010494752. [DOI] [PubMed] [Google Scholar]
- Gee DG, Gabard-Durnam LJ, Flannery J, Goff B, Humphreys KL, Telzer EH, Tottenham N. Early developmental emergence of human amygdala-prefrontal connectivity after maternal deprivation. Proceedings of the National Academy of Sciences of the United States of America. 2013;110(39):15638–43. doi: 10.1073/pnas.1307893110. doi:10.1073/pnas.1307893110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gunnar MR, Donzella B. Social regulation of the cortisol levels in early human development. Psychoneuroendocrinology. 2002;27(1-2):199–220. doi: 10.1016/s0306-4530(01)00045-2. [DOI] [PubMed] [Google Scholar]
- Heckman JJ, Mosso S. The economics of human development and social mobility. Annual Review of Economics. 2014;6:689–733. doi: 10.1146/annurev-economics-080213-040753. doi:10.1146/annurev-economics-080213-040753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heinrichs M, Baumgartner T, Kirschbaum C, Ehlert U. Social support and oxytocin interact to suppress cortisol and subjective responses to psychosocial stress. Biological Psychiatry. 2003;54(12):1389–1398. doi: 10.1016/s0006-3223(03)00465-7. doi:10.1016/S0006-3223(03)00465-7. [DOI] [PubMed] [Google Scholar]
- Hennessy MB, Kaiser S, Sachser N. Social buffering of the stress response: Diversity, mechanisms, and functions. Frontiers in Neuroendocrinology. 2009;30(4):470–82. doi: 10.1016/j.yfrne.2009.06.001. doi:10.1016/j.yfrne.2009.06.001. [DOI] [PubMed] [Google Scholar]
- Hostinar CE, Johnson AE, Gunnar MR. Parent support is less effective in buffering cortisol stress reactivity for adolescents compared to children. Developmental Science. 2015 doi: 10.1111/desc.12195. doi:10.1111/desc.12195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hostinar CE, Sullivan RM, Gunnar MR. Psychobiological mechanisms underlying the social buffering of the hypothalamic-pituitary-adrenocortical axis: A review of animal models and human studies across development. Psychological Bulletin. 2014;140(1):256–82. doi: 10.1037/a0032671. doi:10.1037/a0032671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kirschbaum C, Klauer T, Filipp SH, Hellhammer DH. Sex-specific effects of social support on cortisol and subjective responses to acute psychological stress. Psychosomatic Medicine. 1995;57(1):23–31. doi: 10.1097/00006842-199501000-00004. Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/7732155. [DOI] [PubMed] [Google Scholar]
- Kirschbaum C, Pirke K-M, Hellhammer DH. The “Trier Social Stress Test” -A tool for investigating psychobiological stress responses in a laboratory setting. Neuropsychobiology. 1993;28:76–81. doi: 10.1159/000119004. [DOI] [PubMed] [Google Scholar]
- Koss KJ, Hostinar CE, Donzella B, Gunnar MR. Social deprivation and the HPA axis in early development. Psychoneuroendocrinology. 2014;50:1–13. doi: 10.1016/j.psyneuen.2014.07.028. doi:10.1016/j.psyneuen.2014.07.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McEwen BS. Central effects of stress hormones in health and disease: Understanding the protective and damaging effects of stress and stress mediators. European Journal of Pharmacology. 2008;583(2-3):174–85. doi: 10.1016/j.ejphar.2007.11.071. doi:10.1016/j.ejphar.2007.11.071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller G. The promise and perils of oxytocin. Science. 2013 Jan;339:267–269. doi: 10.1126/science.339.6117.267. [DOI] [PubMed] [Google Scholar]
- National Scientific Council on the Developing Child Young children develop in an environment of relationships. 2004 Retrieved from http://www.developingchild.net.
- Smith AS, Wang Z. Hypothalamic oxytocin mediates social buffering of the stress response. Biological Psychiatry. 2014;76(4):281–8. doi: 10.1016/j.biopsych.2013.09.017. doi:10.1016/j.biopsych.2013.09.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sroufe LA, Egeland B, Carlson EA, Collins WA. The development of the person: The Minnesota study of risk and adaptation from birth to adulthood. Guilford Press; New York, NY: 2005. [Google Scholar]
- Taylor SE, Burklund LJ, Eisenberger NI, Lehman BJ, Hilmert CJ, Lieberman MD. Neural bases of moderation of cortisol stress responses by psychosocial resources. Journal of Personality and Social Psychology. 2008;95(1):197–211. doi: 10.1037/0022-3514.95.1.197. doi:10.1037/0022-3514.95.1.197. [DOI] [PubMed] [Google Scholar]
- Thompson RA. Social support and child protection: Lessons learned and learning. Child Abuse & Neglect. 2014 doi: 10.1016/j.chiabu.2014.06.011. doi:10.1016/j.chiabu.2014.06.011. [DOI] [PubMed] [Google Scholar]
- Thompson RA, Ontai L. Striving to do well what comes naturally: Social support, developmental psychopathology, and social policy. Development and Psychopathology. 2000;12(4):657–675. doi: 10.1017/s0954579400004065. doi:10.1017/S0954579400004065. [DOI] [PubMed] [Google Scholar]
- Tottenham N. Human amygdala development in the absence of species-expected caregiving. Developmental Psychobiology. 2012;54(6):598–611. doi: 10.1002/dev.20531. doi:10.1002/dev.20531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ulrich-Lai YM, Herman JP. Neural regulation of endocrine and autonomic stress responses. Nature Reviews. Neuroscience. 2009;10(6):397–409. doi: 10.1038/nrn2647. doi:10.1038/nrn2647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Waldfogel J. International policies toward parental leave and child care. Future of Children. 2001;11(1):98–111. [PubMed] [Google Scholar]
- Wismer Fries AB, Shirtcliff EA, Pollak SD. Neuroendocrine dysregulation following early social deprivation in children. Developmental Psychobiology. 2008;50(6):588–99. doi: 10.1002/dev.20319. doi:10.1002/dev.20319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wismer Fries AB, Ziegler TE, Kurian JR, Jacoris S, Pollak SD. Early experience in humans is associated with changes in neuropeptides critical for regulating social behavior. Proceedings of the National Academy of Sciences of the United States of America. 2005;102(47):17237–40. doi: 10.1073/pnas.0504767102. doi:10.1073/pnas.0504767102. [DOI] [PMC free article] [PubMed] [Google Scholar]