Abstract
Objective
To examine the effects of the updated 2012 cervical cancer screening guidelines on the rates of sexually transmitted infection (STI) screening in primary care.
Design
Retrospective chart review.
Setting
Five academic family practice units at St Michael’s Hospital in Toronto, Ont.
Participants
Female patients, aged 19 to 25, who had at least 1 visit with a physician at 1 of the 5 academic family practice units during a 12-month period before (May 1, 2011, to April 30, 2012) or after (November 1, 2012, to October 31, 2013) the release of the updated guidelines.
Main outcome measures
Number of women who received Papanicolaou tests or underwent STI screening; rates of STI screening performed during a Pap test or a periodic health examination; screening rates for HIV, syphilis, and hepatitis C; and the methods used for STI screening before and after the release of the updated guidelines.
Results
Before the release of the 2012 guidelines, 42 of 100 women had Pap tests and 40 of 100 women underwent STI screening. After the release of the guidelines, 17 of 100 women had Pap tests and 20 of 100 women received STI screening. Female patients were less likely to undergo STI screening under the 2012 guidelines compared with the 2005 guidelines (odds ratio 0.38, 95% CI 0.19 to 0.74; P = .003).
Conclusion
Implementation of the 2012 cervical cancer screening guidelines was associated with lower rates of STI screening in the primary care setting. Primary care physicians should screen at-risk women for STIs at any clinically appropriate encounter and consider using noninvasive self-sampling methods.
Résumé
Objectif
Déterminer les effets sur le taux de dépistage des maladies transmises sexuellement (MTS) de la mise à jour des directives 2012 pour le dépistage du cancer du col et ce, en contexte de soins primaires.
Type d’étude
Revue rétrospective de dossiers.
Contexte
Cinq unités universitaires de médecine familiale de l’hôpital St Michael’s de Toronto, Ont.
Participantes
Patientes de 19 à 25 ans qui avaient consulté un médecin à une des 5 unités universitaires de médecine familiale durant une période de 12 mois avant (entre le 1er mai 2011 et le 30 avril 2012) ou après (entre le 1er novembre 2012 et le 31 octobre 2013) la publication de la mise à jour des directives.
Principaux paramètres à l’étude
Nombre de femmes qui ont subi le test de Papanicolaou (Pap test) ou qui ont eu un dépistage pour les MTS; taux des dépistages effectués à l’occasion d’un Pap test ou d’un examen de santé périodique; taux de dépistage pour le SIDA, la syphilis et l’hépatite C; et méthodes utilisées pour le dépistage des MTS avant et après la publication de la mise à jour des directives.
Résultats
Avant la publication des directives de 2012, 42 % des femmes avaient eu un Pap test et 40 % un dépistage pour les MTS. Après les nouvelles directives, 17 % des femmes ont eu un Pap test et 20 % un dépistage pour les MTS. Les patientes étaient donc moins susceptibles d’avoir un dépistage pour les MTS selon les directives de 2012 par rapport à celles de 2015 (rapport de cotes 0,38, IC à 95 % 0,19 à 0,74; P = ,003).
Conclusion
La publication des directives de pratique 2012 pour le dépistage du cancer du col s’est accompagnée d’un moindre taux de dépistage pour les MTS dans un contexte de soins primaires. Les médecins de première ligne devraient dépister les MTS chez toute femme à risque, et ce, à chaque occasion appropriée de rencontre clinique; Ils devraient aussi envisager d’utiliser des méthodes d’auto-échantillonnage non invasives.
Historically, visits for Papanicolaou testing have provided family physicians with an opportunity to explore other dimensions of patients’ sexual health, including STI screening.1 Chlamydia is the most commonly reported bacterial STI in Canada, followed by gonorrhea2; prevalence for both infections is highest among women aged 15 to 24.2 The rates of chlamydia and gonorrhea in Canada have been steadily increasing.3 These infections can be long lasting and have serious consequences such as pelvic inflammatory disease, which increases the risk of chronic pelvic pain, infertility, and ectopic pregnancy.2
Before May 2012, women were advised to begin screening for cervical cancer within 3 years of the onset of sexual activity, followed by annual screening. After 3 consecutive normal Pap test results, screening was advised every 2 to 3 years until age 70.4 In May 2012, Cancer Care Ontario released new guidelines that recommended initiation of screening at age 21 for women who are or who have ever been sexually active and to screen every 3 years thereafter in the context of normal results.4 Similarly, in January 2013, the Canadian Task Force on Preventive Health Care published updated guidelines on cervical cancer screening, recommending sexually active women initiate screening at age 25 and continuing screening every 3 years thereafter.5 Anecdotally, most physicians in Ontario follow the Cancer Care Ontario cervical cancer screening guidelines.
Given that the new recommendations both delay cervical cancer screening to age 21 and recommend less frequent screening intervals, we hypothesized that asymptomatic young women would not be visiting their family physicians as frequently for Pap tests, leading to underscreening for STIs in this at-risk population. The objective of this study was to compare rates of Pap tests and STI screening before and after the release of the 2012 Ontario cervical screening guidelines. To our knowledge, there have been no studies investigating the effects of the updated cervical cancer guidelines on STI screening.
METHODS
Study overview
We conducted a retrospective electronic chart review of female patients at 5 academic family practice units at St Michael’s Hospital in Toronto, Ont. Ethics approval was obtained from the Research Ethics Board of St Michael’s Hospital.
Participants
Women aged 19 to 25 were eligible for the study. The lower age limit of 19 was used because the previous guidelines recommended initiation of screening 3 years after onset of sexual activity, and the average age at which girls in Canada begin having sexual intercourse is 16.5 years.2,6,7 The upper age limit was based on recommendations for screening in sexually active patients younger than 25 from both the Public Health Agency of Canada and the Centers for Disease Control and Prevention (CDC).2,8 The Public Health Agency of Canada recommends STI screening in at-risk groups including sexually active females younger than 25 years of age.2 Furthermore, the CDC recommends annual screening for chlamydia and gonorrhea in sexually active females younger than 25 years of age.8
Women who required specialized screening for Pap or STI tests would not be affected by the change in cervical screening guidelines. We therefore excluded women who presented with symptoms of chlamydia or gonorrhea infection, were immunocompromised (eg, were HIV positive or solid organ transplant recipients, or used long-term immunosuppressants), had a history of loop electrosurgical excision procedure, had had a cervical biopsy within 3 years before the time period, or had had an abnormal Pap test result and had not yet returned to a routine screening schedule.2 Additional exclusion criteria are found in Box 1.
Box 1. Additional detailed exclusion criteria.
|
IUD—intrauterine device, STI—sexually transmitted infection.
Derivation of cohorts
Two cohorts of patients were studied: those who had at least 1 visit with a family physician within a 12-month time period before the release of the updated guidelines (May 1, 2011, to April 30, 2012) and patients who had at least 1 visit with a family physician within a 12-month time period after the updated guidelines (November 1, 2012, to October 31, 2013). The second time period started 6 months after the release of the new guidelines to allow for translation into practice.
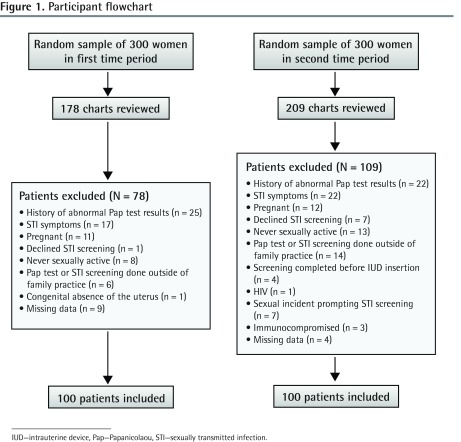
A central database at St Michael’s Hospital was used to generate a random sample of 300 women aged 19 to 25 for each time period of interest. Patients from each sample were consecutively enrolled until 100 women were included in each cohort (Figure 1). The sample size was based on detecting a hypothesized decrease in screening from 35% to 15%, with a power of 0.91.
Figure 1.
Participant flowchart
IUD—intrauterine device, Pap—Papanicolaou, STI—sexually transmitted infection.
Outcome measures
The primary outcome of interest was the number of women who received Pap testing and chlamydia and gonorrhea screening before and after the release of the updated guidelines. Chlamydia and gonorrhea screening could be performed by either physician-obtained endocervical swabs or urine-based sampling. Secondary outcomes of interest included the rates for STI screening performed at the time of a Pap test or periodic health examination, the screening rates for other types of STIs (ie, HIV, syphilis, and hepatitis C), and the method of STI screening used (eg, urine-based samples, physician-collected endocervical swabs, or self-obtained vaginal swabs). Although the CDC and the Public Health Agency of Canada do not recommend hepatitis C screening as part of routine STI screening, St Michael’s Hospital focuses on treating an inner-city population including those with a history of injection drug use and those living with HIV or AIDS; hepatitis C screening is therefore often included in the bloodwork for STI screening. The charts from each sample were independently reviewed; one author (A.F.) reviewed the first sample and another author (T.B.) reviewed the second sample.
Statistical analyses
To compare baseline demographic and clinical characteristics between the cohorts of women, χ2 and unpaired t tests were used. Odds ratios (ORs) were used to compare the rates of STI screening during the first and second time periods. Statistical analyses were performed using Microsoft Excel for Mac 2011, as well as with online calculators.9,10
RESULTS
A total of 178 charts in the first sample and 209 charts in the second sample were reviewed to generate 2 cohorts of 100 patients in the time period before and after the release of the updated guidelines (Figure 1). The mean (SD) age of patients was 22.6 (2.0) and 21.8 (2.1) years in the first and second cohorts, respectively. The mean (SD) number of visits to the family doctor was significantly different (P = .037) between the first (3.2 [2.8] visits) and second (2.4 [2.0] visits) cohorts. There was no statistically significant difference in rates of previous STIs or documented sexual activity between the 2 cohorts (Table 1).
Table 1.
Patient characteristics
| CHARACTERISTICS | BEFORE GUIDELINES (N = 100) | AFTER GUIDELINES (N = 100) | P VALUE |
|---|---|---|---|
| Mean (SD) age, y | 22.6 (2.0) | 21.8 (2.1) | .006 |
| Mean (SD) no. of visits | 3.2 (2.8) | 2.4 (2.0) | .037 |
| Previous STI, % | 9.0 | 5.0 | .267 |
| Documented sexual activity, % | 75.0 | 79.0 | .502 |
STI—sexually transmitted infection.
In the year immediately before the updated guidelines, 42.0% of women received Pap tests and 40.0% underwent screening for chlamydia and gonorrhea. During the 1-year period starting 6 months after release of the updated guidelines, 17.0% of women received Pap tests and 20.0% received STI screening for chlamydia and gonorrhea (Figure 2). The OR of undergoing STI screening under the 2012 guidelines compared with the 2005 guidelines was 0.38 (95% CI 0.19 to 0.74; P = .003).
Figure 2.
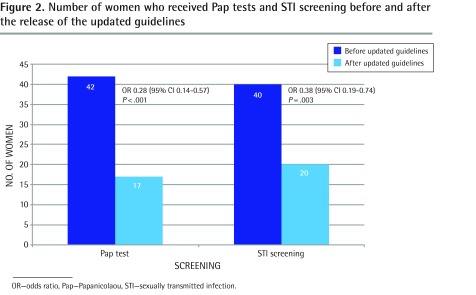
Number of women who received Pap tests and STI screening before and after the release of the updated guidelines
OR—odds ratio, Pap—Papanicolaou, STI—sexually transmitted infection.
Screening for chlamydia and gonorrhea was completed during a Pap test most of the time in the first and second study periods (70.0% and 80.0%, respectively). Additionally, in the first and second study periods, Pap testing (71.4% and 76.5%, respectively) and STI screening (62.5% and 65.0%, respectively) were commonly performed during a periodic health examination. Of note, there were also significantly fewer (P < .001) periodic health examinations in the second time period (N = 13) compared with the first time period (N = 36), but none of the women in either time period received STI screening during a periodic health examination in the absence of a Pap test.
In addition to screening for chlamydia and gonorrhea, the odds of being screened for syphilis (OR = 0.31, 95% CI 0.10 to 0.89; P = .025) and hepatitis C (OR = 0.28, 95% CI 0.07 to 0.97; P = .040) significantly decreased after the release of the new guidelines (Figure 3). A non-significant decrease in HIV screening was also found (OR = 0.48, 95% CI 0.19 to 1.23; P = .140).
Figure 3.
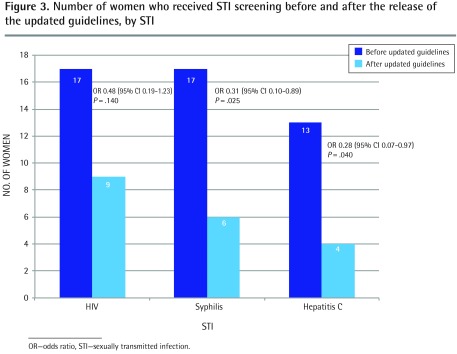
Number of women who received STI screening before and after the release of the updated guidelines, by STI
OR—odds ratio, STI—sexually transmitted infection.
Finally, there was an observed trend toward the use of urine-based rather than swab-based testing; however, this did not reach statistical significance. Cervical and vaginal swabs were used in 82.5% of STI screening before the release of the updated guidelines and 70.0% after the release of the updated guidelines. Urine-based screening was used in 17.5% of STI screening before and 30.0% after the updated guidelines (Table 2). Self-obtained vaginal swabs were not performed for any patients in either time period.
Table 2.
Method of STI screening
| METHOD | BEFORE GUIDELINES, N (%) (N = 40) | AFTER GUIDELINES, N (%) (N = 20) | ODDS RATIO (95% CI) | P VALUE |
|---|---|---|---|---|
| Swab-based testing | 33 (82.5) | 14 (70.0) | 0.50 (0.12–2.06) | .326 |
| Urine-based testing | 7 (17.5) | 6 (30.0) | 2.02 (0.49–8.44) | .326 |
| Self-collected vaginal swab | 0 (0.0) | 0 (0.0) | NA | NA |
STI—sexually transmitted infection, NA—not applicable.
DISCUSSION
We conducted a 2-year retrospective chart review spanning 1-year periods before and after the updated 2012 Ontario cervical cancer screening guidelines4 to assess the effects of the new guidelines on the rates of STI screening in primary care. In keeping with the new guidelines, which recommend delaying cervical cancer screening to age 21 and screening less frequently, it is not surprising that our study found a 60% decrease in Pap testing. However, we found an unintentional 50% decrease in screening for chlamydia and gonorrhea subsequent to the updated guidelines. This decrease in STI screening was not surprising given that STI screening for gonorrhea and chlamydia was found to occur during a Pap test and periodic health examination most of the time. Therefore, with fewer office visits for Pap tests following the new guidelines, family physicians might have fewer opportunities to screen for STIs in young women and might lack reminders to explore patients’ sexual health at other visits.
In addition to screening for chlamydia and gonorrhea, we also found a decrease in screening for syphilis and hepatitis C following the updated guidelines, which can be explained by the same factors mentioned for chlamydia and gonorrhea screening. Although not statistically significant, there was a decrease in HIV screening that was potentially of clinical significance. Additionally, most of the STI screening for gonorrhea and chlamydia was performed with traditional physician-collected endocervical swabs rather than noninvasive urine-based nucleic acid amplification testing or self-collected vaginal swabs. Because STI screening occurs during a Pap test most of the time, this can explain why there is more clinician-collected rather than self-collected sampling.
Our findings highlight an unintended and potentially harmful public health consequence of the updated Ontario cervical cancer screening guidelines. To our knowledge, there have been no other published studies investigating the effects of these updated guidelines on STI screening. Although there was evidence for the newer guidelines to support delaying cervical cancer screening to age 21 and screening less frequently, policy makers and writers of these guidelines might not have anticipated the negative effect of this recommendation on STI screening.2
Our findings merit concern considering that Canadian rates of chlamydia and gonorrhea rose by 72.0% and 53.4%, respectively, during the past decade.11 These infections, especially chlamydia, disproportionately affect younger women, further underscoring the risk imposed in this population by the effect the updated guidelines have on STI screening. It is critical that family physicians in Ontario and elsewhere are cognizant of this risk, and continue to pursue regular STI risk assessments in sexually active women younger than 25 years of age consistent with both Canadian and American guidelines.2,8
These results add to the literature describing the unintended consequences of large-scale clinical practice guidelines. Experts recognize that clinical practice guidelines risk reducing medical care to a standardized formulaic process, leading to subtle and overt long-term unintended consequences.12 Elsewhere, policy and guideline changes have had unintended consequences: after publication of the Randomized Aldactone Evaluation Study, which demonstrated that spironolactone substantially improved outcomes in patients with severe heart failure, there were increases in rates of hyperkalemia13; after routine eye examinations were delisted in Ontario, there were decreases in retinopathy screening for people with diabetes14; and when practitioners adhered to clinical practice guidelines, there was a decrease in the quality of care provided to older adults with multiple comorbidities.15
We should highlight an important development in primary care that emerged during our study period that might influence the interpretation of our results. In October 2012, the Cochrane Library published a systematic review reporting that periodic health examinations did not reduce morbidity or mortality.16 One could therefore infer that the reduction in STI screening observed in our study was also the result of a decrease in periodic health examinations. We did find that there were significantly fewer (P < .001) periodic health examinations in the second time period (N = 13) compared with the first (N = 36). However, in our study, no women received STI screening at their periodic health examination if a Pap test was not performed. It is therefore possible that fewer periodic health examinations were performed in the second time period because women were not receiving as many Pap tests.
Limitations
We acknowledge several limitations in our study design and analysis. First, there are inherent limitations to a retrospective chart review including the lack of a control group and randomization, as well as the potential for selection bias. We also recognize that our study was limited to 5 primary care sites in 1 urban academic setting and therefore the results might not be generalizable to other settings. Indeed, the family practice units at St Michael’s Hospital are urban academic centres, providing care to an inner-city population, and for that reason providers might have had a heightened sense of awareness of STIs. It is also important to note that the mean age between the 2 cohorts was statistically different; however, given that the difference was less than 1 year, it is likely not clinically significant.
Another limitation might be the relatively short time period of the study. We were limited to a 12-month time range before the release of the guidelines owing to the implementation of an electronic medical record 13 months before the release of the updated guidelines. We acknowledge that we might not have captured women who underwent STI screening immediately before or after the given time period. Additionally, the 6-month time period between the release of the updated guidelines and the second time period under study is likely conservative for knowledge translation and uptake of clinical practice guidelines. Therefore, it is possible that this study underestimated the true reduction in STI screening after the release of the updated cervical cancer screening guidelines.
Another drawback of our study was the lack of consistent documentation of sexual activity in patients’ charts, which could have contributed to the lower overall rates of STI screening in both samples. Nevertheless, more than 70% of patients in each sample were sexually active and there was no significant difference in documented sexual activity between the 2 cohorts.
Finally, data were abstracted only once and by 2 different abstractors at the 2 time intervals. The abstractors worked together to ensure consistency in data abstraction and compared results on a number of charts. Inclusion and exclusion criteria resulted in a different proportion of exclusions for different reasons in each time period but the overall number of exclusions in each time period was similar.
Despite these limitations, we are confident in the validity of our principal findings demonstrating that the implementation of the 2012 Ontario cervical cancer screening guidelines was associated with lower rates of STI screening in the primary care setting. With the change in cervical cancer screening guidelines, it is important for family physicians to find innovative ways to ensure that STI screening is addressed in at-risk populations. New interventions, such as urine-based samples or self-administered vaginal swabs, analyzed by nucleic acid amplification tests, might be increasingly important to screen for STIs in asymptomatic women. Self-sampling is highly acceptable to young women and highly sensitive and specific.17–20 Furthermore, noninvasive sampling allows women with STIs who are not receiving Pap tests or who are not presenting with gynecological complaints to be easily screened. Although young women might not visit their family physicians as often for routine Pap tests, particularly in those younger than age 21, these women are at higher risk of STIs. With the use of noninvasive sampling, STI screening programs have the capability of extending beyond the clinic setting into community settings including high schools, universities, shopping malls, and street out-reach sites.
Conclusion
Although clinical practice guidelines set out to improve quality of care, preventive health care is often complex and interconnected and therefore a change in guidelines should not be implemented in isolation. With this in mind, family physicians should screen at-risk women for STIs at any clinically appropriate encounter, irrespective of Pap testing, and consider using noninvasive self-sampling methods.
Acknowledgments
We thank Dr Sheila Dunn for her work in conceptualizing this manuscript and for her comments on earlier drafts. Dr Glazier is supported as a clinician scientist in the Department of Family and Community Medicine at the University of Toronto and at St Michael’s Hospital.
EDITOR’S KEY POINTS
The updated cervical cancer screening guidelines recommend delaying cervical cancer screening until age 21 and screening less frequently. With these changes, asymptomatic young women might visit their family physicians less frequently for Papanicolaou tests, leading to underscreening of sexually transmitted infections (STIs) in this at-risk population.
This study found a 60% decrease in Pap testing and a 50% decrease in screening for chlamydia and gonorrhea subsequent to the updated guidelines. This decrease in STI screening was not surprising given that screening for these STIs usually occurred during a Pap test or periodic health examination. Therefore, with fewer office visits for Pap tests, family physicians might have fewer opportunities to screen for STIs and explore patients’ sexual health at other visits.
Family physicians need to find innovative ways to ensure that STI screening is addressed in at-risk populations. New interventions (eg, self-administered vaginal swabs) might be increasingly important to screen for STIs in asymptomatic women.
POINTS DE REPÈRE DU RÉDACTEUR
Les nouvelles directives de pratique sur le dépistage du cancer du col recommandent de le faire moins souvent et seulement à partir de 21 ans. En raison de ces changements, les jeunes femmes asymptomatiques risquent de ne pas consulter leur médecin aussi fréquemment pour un test de Papanicolaou (Pap test), avec comme conséquences une diminution du dépistage des maladies transmises sexuellement (MTS) chez cette population à risque.
Cette étude a montré qu’à la suite de la mise à jour des directives, il y a eu une réduction de 60 % des Pap tests et de 50 % du dépistage pour la chlamydia et la gonorrhée. Cette diminution du dépistage des MTS n’est pas surprenante étant donné que ce type de dépistage est généralement effectué à l’occasion du Pap test ou d’un examen de santé périodique. Comme il y a moins de visites pour le Pap test, le médecin de famille aura probablement moins d’occasions de vérifier la santé sexuelle de ses patientes lors des autres visites.
Les médecins de famille devront trouver de nouvelles façons de s’assurer que le dépistage des MTS est effectué chez les patientes à risque. De nouvelles interventions (p. ex. des frottis vaginaux auto-administrés) pourraient devenir de plus en plus importantes pour dépister les MTS chez des patientes asymptomatiques.
Footnotes
This article has been peer reviewed.
Cet article fait l’objet d’une révision par des pairs.
Contributors
All authors contributed to the concept and design of the study; data gathering, analysis, and interpretation; and preparing the manuscript for submission.
Competing interest
None declared
References
- 1.Murphy J, Kennedy E, Dunn S, Fung Kee, Fung M, Gzik D, McLachlin CM, et al. Cervical screening. Evidence-based series 15-9. Toronto, ON: Cancer Care Ontario; 2011. Available from: www.cancercare.on.ca/common/pages/UserFile.aspx?fileId=124511. Accessed 2015 Aug 21. [Google Scholar]
- 2.Public Health Agency of Canada [website] Canadian guidelines on sexually transmitted infections. Ottawa, ON: Public Health Agency of Canada; 2015. Available from: www.phac-aspc.gc.ca/std-mts/sti-its/cgsti-ldcits/index-eng.php. Accessed 2015 Aug 21. [Google Scholar]
- 3.Public Health Agency of Canada [website] Report on sexually transmitted infections in Canada: 2010. Ottawa, ON: Public Health Agency of Canada; 2012. Available from: http://publications.gc.ca/collections/collection_2013/aspc-phac/HP37-10-2010-eng.pdf. Accessed 2015 Aug 21. [Google Scholar]
- 4.Murphy J, Kennedy EB, Dunn S, McLachlin CM, Fung Kee Fung M, Gzik D, et al. Cervical screening: a guideline for clinical practice in Ontario. J Obstet Gynaecol Can. 2012;34(5):453–8. doi: 10.1016/S1701-2163(16)35242-2. [DOI] [PubMed] [Google Scholar]
- 5.Canadian Task Force on Preventive Health Care. Dickinson J, Tsakonas E, Conner Gorber S, Lewin G, Shaw E, et al. Recommendations on screening for cervical cancer. CMAJ. 2013;185(1):35–45. doi: 10.1503/cmaj.121505. Epub 2013 Jan 7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Rotermann M. Sex, condoms and STDs among young people. Health Rep. 2005;16(3):39–45. [PubMed] [Google Scholar]
- 7.Rotermann M. Sexual behaviour and condom use of 15- to 24-year-olds in 2003 and 2009/2010. Health Rep. 2012;23(1):41–5. [PubMed] [Google Scholar]
- 8.Centers for Disease Control and Prevention [website] STD and HIV screening recommendations. Atlanta, GA: Centers for Disease Control and Prevention; 2014. Available from: www.cdc.gov/std/prevention/screeningReccs.htm. Accessed 2015 Aug 21. [Google Scholar]
- 9.Hutchon DJR. Calculator for confidence intervals of odds ratio in an unmatched case control study. Darlington, Engl: David J.R. Hutchon; 2001. Available from: www.hutchon.net/ConfidOR.htm. Accessed 2015 Aug 21. [Google Scholar]
- 10.2-Way contingency table analysis. Kissimmee, FL: John C. Pezzullo; 2013. Statpages. Available from: http://statpages.org/ctab2x2.html. Accessed 2015 Aug 21. [Google Scholar]
- 11.Public Health Agency of Canada . The Chief Public Health Officer’s report on the state of public health in Canada, 2013. Infectious disease—the never-ending threat. Ottawa, ON: Public Health Agency of Canada; 2013. Available from: www.phac-aspc.gc.ca/cphorsphc-respcacsp/2013/sti-its-eng.php. Accessed 2015 Aug 21. [Google Scholar]
- 12.Shackelton RJ, Marceau LD, Link CL, McKinlay JB. The intended and unintended consequences of clinical guidelines. J Eval Clin Pract. 2009;15(6):1035–42. doi: 10.1111/j.1365-2753.2009.01201.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Juurlink DN, Mamdani MM, Lee DS, Kopp A, Austin PC, Laupacis A, et al. Rates of hyperkalemia after publication of the Randomized Aldactone Evaluation Study. N Engl J Med. 2004;351(6):543–51. doi: 10.1056/NEJMoa040135. [DOI] [PubMed] [Google Scholar]
- 14.Kiran T, Kopp A, Moineddin R, Victor JC, Campbell RJ, Shah BR, et al. Unintended consequences of delisting routine eye exams on retinopathy screening for people with diabetes in Ontario, Canada. CMAJ. 2013;185(3):E167–73. doi: 10.1503/cmaj.120862. Epub 2013 Jan 7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Boyd CM, Darer J, Boult C, Fried LP, Boult L, Wu AW. Clinical practice guidelines and quality of care for older patients with multiple comorbid diseases: implications for pay for performance. JAMA. 2005;294(6):716–24. doi: 10.1001/jama.294.6.716. [DOI] [PubMed] [Google Scholar]
- 16.Krogsbøll LT, Jørgensen KJ, Grønhøj, Larsen C, Gøtzsche PC. General health checks in adults for reducing morbidity and mortality from disease. Cochrane Database Syst Rev. 2012;(10):CD009009. doi: 10.1002/14651858.CD009009.pub2. [DOI] [PubMed] [Google Scholar]
- 17.Chernesky MA, Hook EW, 3rd, Martin DH, Lane J, Johnson R, Jordan JA, et al. Women find it easy and prefer to collect their own vaginal swabs to diagnose Chlamydia trachomatis or Neisseria gonorrhoeae infections. Sex Transm Dis. 2005;32(12):729–33. doi: 10.1097/01.olq.0000190057.61633.8d. [DOI] [PubMed] [Google Scholar]
- 18.Fang J, Husman C, DeSilva L, Chang R, Peralta L. Evaluation of self-collected vaginal swab, first void urine, and endocervical swab specimens for the detection of Chlamydia trachomatis and Neisseria gonorrhoeae in adolescent females. J Pediatr Adolesc Gynecol. 2008;21(6):355–60. doi: 10.1016/j.jpag.2008.03.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Gaydos CA, Quinn TC. Urine nucleic acid amplification tests for the diagnosis of sexually transmitted infections in clinical practice. Curr Opin Infect Dis. 2005;18(1):55–66. doi: 10.1097/00001432-200502000-00010. [DOI] [PubMed] [Google Scholar]
- 20.Taylor D, Lunny C, Wong T, Gilbert M, Li N, Lester R, et al. Self-collected versus clinician-collected sampling for sexually transmitted infections: a systematic review and meta-analysis protocol. Syst Rev. 2013;2:93. doi: 10.1186/2046-4053-2-93. [DOI] [PMC free article] [PubMed] [Google Scholar]