Abstract
We investigated obesity-induced changes in kidney lipid accumulation, mitochondrial function, and endoplasmic reticulum (ER) stress in the absence of hypertension, and the potential role of leptin in modulating these changes. We compared two normotensive genetic mouse models of obesity, leptin-deficient ob/ob mice and hyperleptinemic melanocortin-4 receptor-deficient mice (LoxTB MC4R−/−), with their respective lean controls. Compared with controls, ob/ob and LoxTB MC4R−/− mice exhibit significant albuminuria, increased creatinine clearance, and high renal triglyceride content. Renal ATP levels were decreased in both obesity models, and mitochondria isolated from both models showed alterations that would lower mitochondrial ATP production. Mitochondria from hyperleptinemic LoxTB MC4R−/− mice kidneys respired NADH-generating substrates (including palmitate) at lower rates due to an apparent decrease in complex I activity, and these mitochondria showed oxidative damage. Kidney mitochondria of leptin-deficient ob/ob mice showed normal rates of respiration with no evidence of oxidative damage, but electron transfer was partially uncoupled from ATP synthesis. A fourfold induction of C/EBP homologous protein (CHOP) expression indicated induction of ER stress in kidneys of hyperleptinemic LoxTB MC4R−/− mice. In contrast, ER stress was not induced in kidneys of leptin-deficient ob/ob mice. Our findings show that obesity, in the absence of hypertension, is associated with renal dysfunction in mice but not with major renal injury. Alterations to mitochondria that lower cellular ATP levels may be involved in obesity-induced renal injury. The type and severity of mitochondrial and ER dysfunction differs depending upon the presence or absence of leptin.
Keywords: obesity, nephropathy, leptin, kidney, mitochondria, endoplasmic reticulum stress, melanocortin-4 receptor (MC4R)
obesity has emerged as a global health problem and is a major risk factor for cardiovascular diseases, such as heart failure (9, 10) and liver disease, such as non-alcoholic fatty liver disease (32, 35). Obesity may also be a risk factor for end-stage renal disease (43, 46), even after adjustment for hypertension and diabetes, and multiple studies in experimental animals (53, 61, 67, 84, 85) and humans (2, 4, 41, 91, 92) strongly implicate obesity in the development of chronic kidney disease.
With obesity, tissues are presented with elevated levels of fatty acids. Two consequences of this are 1) increased oxidation of fatty acids by the mitochondria, and 2) increased production of reactive oxygen species (ROS; superoxide, hydrogen peroxide, hydroxyl radical, lipid peroxides) in mitochondria (5, 77). Increased ROS can damage mitochondrial proteins, leading to further ROS production, and ultimately to mitochondrial dysfunction. Mitochondrial dysfunction presents in various forms, presumably due to variables such as the tissue source of the mitochondria and obesity-associated pathophysiological conditions, such as hypertension and diabetes. In addition, several protective events may be induced, including increased antioxidant defense (73, 95), activation of uncoupling proteins (54, 77), and promotion of mitochondrial biogenesis (47) and mitophagy (82).
Information on kidney mitochondrial function during obesity is limited. In mice fed a high-fat diet, kidney mitochondria respiring succinate produced more H2O2 and showed evidence of oxidative stress, while respiration and coupling appeared normal (73). However, the ability of the kidney mitochondria to oxidize fatty acids was not examined.
Obesity may also impair the protein-folding machinery within the endoplasmic reticulum (ER), a condition known as ER stress (11, 52). Disruption of the resident ER chaperone BiP/glucose-regulated protein 78 (GRP78) aggravates renal tubular injury (51). Experimentally induced ER stress in renal tubules (50) and podocytes (45) has been demonstrated to cause autophagy and result in cell injury, respectively. A recent study in obese Zucker rats, a rodent model of obesity with hypertension and metabolic syndrome, revealed activation of ER stress in the kidneys (90). However, the effect of obesity, in the absence of hypertension, on kidney ER has not, to our knowledge, been previously reported.
Obesity increases levels of the adipocyte-derived hormone leptin, which acts in the brain to regulate food intake (59). Leptin also stimulates β-oxidation of fatty acids, as well as export of lipids from tissues through signaling events involving both central (brain) and peripheral receptors (7, 13, 17, 68, 93, 99). However, the effects of leptin on kidney mitochondrial function during obesity have not, to our knowledge, been reported.
One objective of this study was to determine obesity-associated changes in triglyceride accumulation, mitochondrial function, ER stress, and kidney function in the absence of hypertension. A second objective was to test whether obesity affects the functions of renal mitochondria and ER differently in mice with high levels of leptin compared with obese mice with leptin deficiency. To accomplish these objectives, we used two genetic mouse models of obesity: 1) ob/ob mice which are obese because they do not synthesize leptin (66, 97) and 2) hyperleptinemic melanocortin-4 receptor deficient mice (LoxTB MC4R−/−) (87). The melanocortin-4 receptor (MC4R) signaling pathway also plays a central role in regulating appetite and energy homeostasis in humans and other mammals (76). Mutations in the MC4R gene are the most common genetic causes of early onset, morbid obesity in humans (24) and are associated with increased adiposity and weight gain in mice (56). Unlike genetic mutations in leptin or leptin receptors, alterations in the MC4R gene are associated with high plasma leptin levels (20, 87).
An important advantage of these obesity models, besides the fact that they have very different levels of leptin, is that they do not have increased sympathetic nervous system activity and hypertension that are usually associated with obesity. Therefore, the use of MC4R−/− and ob/ob mice permitted us to examine the impact of obesity on mitochondrial function, ER stress, and kidney function in the absence of confounding effects of hypertension. Our results show that obesity in the absence of hypertension is linked to glomerular hyperfiltration and renal dysfunction associated with increased albumin excretion. Both ob/ob and MC4R−/− mice showed reduced ATP levels in kidney tissue and mitochondrial dysfunction, although the mitochondrial alterations seen in the absence of leptin differed from those seen in the presence of the hormone. The kidneys of obese mice with high plasma leptin levels exhibited mitochondrial oxidative stress and evidence of ER stress, while the kidneys of obese mice lacking leptin did not.
MATERIALS AND METHODS
All the experimental protocols and procedures were approved by the Institutional Animal Care and Use Committee (IACUC) at the University of Mississippi Medical Center (Jackson, MS). Mice were placed in a 12:12-h dark-light cycle and given free access to food and water throughout the period of study.
Blood Pressure Recording and Urine, Blood, and Tissue Collection
Thirty-six to forty-five wk-old C57BL/6J wild-type (WT), ob/ob, LoxTB MC4R−/−, and their age-matched WT littermate control mice (n = 7/group) were used in the study. C57BL/6J WT mice were used as controls for ob/ob mice, whereas the LoxTB MC4R−/− mice that were raised in a B6 mixed background were compared against their WT littermates (WT-LoxTB) from the breeding colony. LoxTB MC4R−/− mice have a loxp-flanked transcriptional blocking (loxTB) sequence inserted between the transcriptional start site and the start codon of MC4R, which prevents the transcription of the MC4R gene (8).
Mice were implanted with telemetric blood pressure transmitters (model TA11PAC10, Data Sciences) as described previously (87). Ten days after recovery from surgery, mean arterial pressure (MAP) and heart rate (HR) were measured by telemetry continuously, 24 h/day, for 5 consecutive days.
Following blood pressure recording, mice were acclimatized in metabolic cages for 48 h before measurement of food intake and urine collection for 3 consecutive days to determine albumin and creatinine excretion. On the fifth day, mice were anesthetized with isoflurane, and 50–100 μl of blood was collected via tail snip for plasma creatinine analysis. Five days later, mice were fasted overnight, and the next day morning 50–100 μl of blood was collected again via tail snip under isoflurane anesthesia to determine fasting blood glucose, plasma insulin, leptin, and triglyceride levels. Three days later, mice were anesthetized with isoflurane and transcardially perfused with 0.1% PBS, pH 7.4. The brain, heart, liver, kidneys, perirenal, mesenteric, and epididymal fat were collected and weighed. Mitochondria were isolated from the right kidney as described below. The left kidney was divided into two portions: one portion was frozen in liquid nitrogen for biochemical and proteomic measurements, and the other portion was fixed in 10% buffered formalin to assess histological changes in the kidney.
Blood and Urine Biochemistry
Fasting blood glucose levels were measured with a glucometer (Optima ultrafast glucose strips). Urine albumin, plasma insulin, and leptin levels were determined by ELISA kits using Exocell/Albuwell M, Linco Insulin, and R&D Leptin, respectively. Urine and plasma creatinine levels were determined using HPLC. The glomerular filtration rate was estimated from creatinine clearance (μl/min) calculated from the following formula: [urine volume (μl/24 h) × urine creatinine (mg/dl)]/[plasma creatinine (mg/dl) × 1,440 min].
Histological Assessments
Coronal sections ∼5 μm thick of the kidneys were stained for hematoxylin and eosin (H&E) and periodic acid-Schiff (PAS) to analyze renal morphology and hyalinization. Renal macrophage infiltration was assessed by immunostaining for F4/80 antigen, a cell surface marker widely expressed in mouse macrophages, using anti-F4/80 antibody (AbD Serotec) as described previously (20).
Measurement of Renal Triglyceride Levels, Renal ATP Content, and Mitochondrial Protein Carbonyl Levels
Renal triglycerides were extracted using the chloroform: methanol method described by Folch et al. (26) and measured using a colorimetric triglyceride assay kit (Wako Diagnostics). ATP levels were determined in whole kidney homogenates using a bioluminescent ATP determination kit (Molecular Probes). Mitochondrial protein carbonyl levels were assayed spectrophotometrically by derivatization with 2,4-dinitrophenyl hydrazine using a protein carbonyl assay kit (Cayman Chemical).
Mitochondrial Isolation and Respiration Measurements
Immediately after harvest, right kidneys from mice were washed with ice-cold PBS, pH 7.4, and thoroughly minced with a sharp scalpel. The contents were then transferred to a 5-ml capacity teflon-glass homogenizer and homogenized in 1.5 ml of ice-cold mitochondrial extraction buffer [250 mM sucrose, 10 mM HEPES, 1 mM EDTA, 1 mg/ml BSA, pH 7.4, with complete protease inhibitor and PhosStop phosphatase inhibitor cocktail (Roche Diagnostics)] by performing 8–10 strokes. Homogenates were centrifuged at 600 g for 10 min at 4°C to remove debris, and the resulting supernatants were centrifuged at 10,000 g for 10 min at 4°C to pellet the mitochondrial fraction. The pellets were resuspended in 0.5 ml of extraction buffer and centrifuged again at 10,000 g for 10 min at 4°C. The final pellets containing mitochondria were resuspended in 50 μl of mitochondrial extraction buffer, and the protein concentrations were determined by bicinchonic acid assay (Pierce).
Mitochondrial respiration measurements were performed polarographically using a Clark-type O2 electrode [Yellow Spring Instruments (YSI)] at 25°C in a 1.7-ml water-jacketed chamber (Gilson) and a YSI 5300A biological oxygen monitor. The analog output from the YSI monitor was ported to an 18-bit analog-to-digital converter in a Hansatech Oxygraph unit, and the O2 consumption measurements (mV vs. time) were collected as ASCII data using Hansatech software and transferred to Kaleidograph (Synergy Software) for extraction of the rates of O2 consumption. The reaction buffer included 250 mM sucrose, 8 mM potassium phosphate, 4 mM magnesium chloride, 1 mM EDTA, and 100 mM Tris buffer, pH 7.2. O2 consumption was initiated by the addition of respiratory substrates such as succinate (5 mM), glutamate (10 mM), malate (5 mM), palmitoyl l-carnitine (5 μM), or palmitoyl-CoA (5 μM) with l-carnitine (1 mM). The rate of O2 consumption during state-3 respiration was measured after the addition of 0.2 mM ADP. State-4 respiration was induced by adding 2 μg/ml oligomycin, to inhibit mitochondrial ATP synthase. To correct for changes in the mitochondrial content, citrate synthase (CS) activities were determined spectrophotometrically as described by Srere et al. (83). State-3 respiration rates are presented as nanomoles O2 consumed per minute per unit CS activity.
To measure complex IV (cytochrome c oxidase) activity, an aliquot of mitochondria was solubilized in 0.2% dodecylmaltoside. Oxygen consumption by complex IV was measured in the apparatus described above by methods described previously (89), using ascorbate plus N,N,N′,N′-tetramethyl-p-phenylenediamine (TMPD) to supply electrons to horse heart cytochrome c. The activities were expressed as nanomoles of electrons per minute per milligram mitochondrial protein.
Markers of ER Stress
Fifty micrograms of protein from whole kidney homogenates were separated on SDS-PAGE and transferred onto a nitrocellulose membrane. Membranes were blocked with Odyssey blocking buffer (Li-Cor) for 1 h and then probed for ER chaperone protein GRP78, C/EBP homologous protein (CHOP) and X-box binding protein-1 (XBP-1) using commercial antibodies (anti-rabbit polyclonal GRP78, 1:1,000 dilution, Cell Signaling Technology; anti-mouse monoclonal CHOP, 1:1,000 dilution, Cell Signaling Technology; and anti-rabbit polyclonal XBP-1, 1:200 dilution, Santa Cruz Biotechnology). Blots were imaged using an Odyssey imaging system, and densitometry analyses were performed to quantitate protein levels.
Statistical Analysis
Data are expressed as means ± SE. One-way ANOVA followed by Tukey's post hoc test was performed to compare between groups using GraphPad Prism 6. A P value <0.05 was considered to be statistically significant.
RESULTS
Anthropometric, Metabolic, and Cardiovascular Characteristics
The body weights of ob/ob and LoxTB MC4R−/− mice were 2.3- and 1.5-fold greater than their respective lean controls (Table 1). The wet weights of the liver, heart, and fat depots, such as visceral fat and perirenal fat, were significantly increased in both obese mouse models (Table 1). Although the brown adipose tissue depots in ob/ob mice were smaller than those of LoxTB MC4R−/− mice, the visceral and perirenal fat depots were significantly larger.
Table 1.
Anthropometric measurements of transgenic and wild-type mice
| WT (n = 7) | ob/ob (n = 7) | WT-LoxTB (n = 7) | LoxTB MC4R−/− (n = 7) | |
|---|---|---|---|---|
| Age, wk | 36–45 | 36–45 | 36–45 | 36–45 |
| Body weight, g | 34.7 ± 1.0 | 80.4 ± 3.9*‡ | 38.0 ± 2.3 | 56.9 ± 2.8† |
| Body length, cm | 10.2 ± 0.1 | 10.6 ± 0.1* | 10.2 ± 0.2 | 10.4 ± 0.1 |
| Liver, g | 1.81 ± 0.08 | 4.74 ± 0.37* | 1.70 ± 0.16 | 4.41 ± 0.75† |
| Kidney, g | 0.24 ± 0.01 | 0.28 ± 0.01* | 0.25 ± 0.02 | 0.28 ± 0.01† |
| Brain, g | 0.45 ± 0.01 | 0.41 ± 0.02* | 0.48 ± 0.01 | 0.45 ± 0.01 |
| Heart, g | 0.16 ± 0.01 | 0.23 ± 0.02* | 0.16 ± 0.01 | 0.20 ± 0.01† |
| Epididymal fat, g | 0.99 ± 0.21 | 1.36 ± 0.08* | 0.93 ± 0.18 | 1.54 ± 0.16† |
| Brown adipose tissue, g | 0.30 ± 0.03 | 0.28 ± 0.04‡ | 0.36 ± 0.03 | 0.80 ± 0.06† |
| Visceral fat, g | 0.35 ± 0.06 | 1.96 ± 0.18*‡ | 0.58 ± 0.11 | 1.16 ± 0.14† |
| Perirenal fat, g | 0.41 ± 0.08 | 3.58 ± 0.02*‡ | 0.54 ± 0.09 | 1.11 ± 0.07† |
Values are means ± SE. WT, wild-type.
P < 0.05 compared with WT.
P < 0.05 compared with WT-LoxTB.
P < 0.05 compared with LoxTB MC4R−/−.
Both ob/ob and LoxTB MC4R−/− mice exhibited elevated fasting blood glucose levels compared with their control groups, despite large increases in plasma insulin levels (Table 2). As expected (87), plasma leptin levels were markedly elevated in the LoxTB MC4R−/− mice (49.1 ± 9.8 ng/ml) compared with their littermate control mice (7.8 ± 3.0 ng/ml). In contrast, plasma leptin levels were undetectable in ob/ob mice plasma compared with WT control mice (4.9 ± 2.1 ng/ml). Despite marked obesity, ob/ob and LoxTB MC4R−/− mice were normotensive and showed normal heart rates compared with their controls (Table 2).
Table 2.
Metabolic and cardiovascular parameters of transgenic and wild-type mice
| Parameter | WT (n = 7) | ob/ob (n = 7) | WT-LoxTB (n = 7) | LoxTB MC4R−/− (n = 7) |
|---|---|---|---|---|
| Plasma leptin, ng/ml | 4.9 ± 2.1 | Undetectable | 7.8 ± 3.0 | 49.1 ± 9.8 |
| Plasma insulin, μU/ml | 6.8 ± 1.9 | 100.1 ± 16.1* | 8.2 ± 1 | 63.5 ± 12.3† |
| Fasting blood glucose, mg/dl | 110 ± 5 | 198 ± 14*‡ | 113 ± 7 | 150 ± 10† |
| Mean arterial pressure, mmHg | 111 ± 1 | 111 ± 1 | 111 ± 1 | 110 ± 9 |
| Heart rate, beats/min | 557 ± 7 | 569 ± 12 | 538 ± 4 | 555 ± 48 |
Values are means ± SE.
P < 0.05 compared with WT.
P < 0.05 compared with WT-LoxTB.
P < 0.05 compared with LoxTB MC4R−/−.
In summary, consistent with our previous studies (20, 21, 87), the melanocortin-4 receptor deficient LoxTB MC4R−/− mice as well as the ob/ob mice displayed hyperglycemia and hyperinsulinemia but normal blood pressure and heart rate. The models differ in that the LoxTB MC4R−/− mice have high plasma levels of leptin (49.1 ± 9.8 ng/ml) while the ob/ob mice have no detectable plasma leptin.
Renal Function and Architecture
Obesity caused by leptin deficiency or MC4R deficiency resulted in glomerular hyperfiltration, as shown by increases in creatinine clearance rates (Fig. 1A). Creatinine clearance in ob/ob mice was increased by 30% compared with lean controls (693 ± 61 vs. 534 ± 32 μl/min). Glomerular hyperfiltration was more pronounced in MC4R-deficient mice, in which there was a 50% increase in creatinine clearance rate compared with controls (752 ± 51 vs. 489 ± 81 μl/min). Similarly, the urine albumin excretion was also increased in both genetic mouse models of obesity (Fig. 1B). The ob/ob mice excreted about fivefold more albumin than lean controls, while LoxTB MC4R−/− mice excreted about fourfold more albumin than their lean controls.
Fig. 1.
Both transgenic obese mice models (LoxTB MC4R−/− and ob/ob mice) exhibited increased creatinine clearance (A), indicative of renal hyperfiltration, and significant albuminuria (B), a marker of renal dysfunction. Values are means ± SE. *P < 0.05 compared with wild-type (WT) Control. †P < 0.05 compared with WT-LoxTB mice.
Despite the increases in creatinine clearance and urine albumin excretion in these two models of normotensive obesity, only subtle changes in glomerular and renal tubular architecture were observed. This was assessed by PAS staining of renal sections (Fig. 2A) as well as H&E staining (data not shown). F4/80 antigen staining, the cell surface marker for murine macrophages, did not show any major changes in macrophage infiltration in the kidneys of these normotensive obese mice compared with their lean counterparts (Fig. 2B).
Fig. 2.
Representative images of periodic acid-Schiff (PAS)-stained (A) and F4/80-stained (B) kidney sections of ob/ob and LoxTB MC4R−/− mice compared with their respective WT controls under ×400 magnification.
Renal Triglycerides, ATP Levels, and Oxidative Stress
Renal triglyceride levels.
The kidneys of ob/ob mice had a 68% greater triglyceride (TG) content compared with WT mice (8.1 ± 0.8 in ob/ob mice vs. 4.8 ± 0.2 mg TG/g kidney weight in WT mice) (Fig. 3A). Renal TG levels were 77% greater in LoxTB MC4R−/− than in their littermate lean controls (3.9 ± 0.2 vs. 2.2 ± 0.3 mg TG/g kidney wt). Both models contain elevated kidney TG compared with their respective controls.
Fig. 3.
Leptin-deficient ob/ob mice and hyperleptinemic LoxTB MC4R−/− mice showed increased lipid accumulation (A) and decreased ATP levels (B) in the kidneys. However, only the LoxTB MC4R−/− mice exhibited increased protein carbonyl levels, a measure of oxidative stress, compared with WT-LoxTB control mice (C). Values are means ± SE. *P < 0.05 compared with WT Control. †P < 0.05 compared with WT-LoxTB mice. ‡P < 0.05 compared with LoxTB MC4R−/− mice.
Renal ATP levels.
Renal ATP content was significantly lower in kidneys from both obese models compared with their lean controls (Fig. 3B). The ATP levels observed in ob/ob mice kidneys (5.95 ± 0.34 pmol/mg) were 75% of that measured in WT controls (7.86 ± 0.35 pmol/mg), whereas ATP levels in LoxTB MC4R−/− mice kidneys (4.99 ± 0.17 pmol/mg) were ∼62% of that in WT-LoxTB controls (8.00 ± 1.11 pmol/mg).
Oxidative stress.
Protein carbonylation results from the irreversible oxidation of protein side chains by hydrogen peroxide or hydroxyl radicals, both downstream products of superoxide produced by the electron transfer chain of oxidative phosphorylation (34). Hence, carbonylation provides a relative measure of the extent of oxidative damage from mitochondrial ROS. Protein carbonyl levels were increased by 20% in homogenates of LoxTB MC4R−/− kidney mitochondria (13.41 ± 0.92 nmol/mg) compared with lean controls (11.14 ± 0.18 nmol/mg) (Fig. 3C). However, there was no significant difference in the carbonylation of kidney mitochondria protein in ob/ob mice vs. lean controls.
Mitochondrial Function
Since the kidney is a highly oxidative tissue, the decreased kidney ATP levels imply that renal mitochondria in obese animals may not be meeting the ATP demands of the cells. For this reason, we examined isolated kidney mitochondria for obesity-related changes in oxidative phosphorylation.
Ob/ob mice kidney mitochondria.
compared with lean controls, no significant changes in the rate of state 3 respiration (+ADP) were observed with mitochondria isolated from ob/ob mice kidneys, using succinate, glutamate-malate, palmitoyl-carnitine, or palmitoyl-CoA plus carnitine as substrates (Fig. 4A). The finding argues against significant obesity-related declines in the activities of complex I, complex II, carnitine palmitoyl transferase I, and the dehydrogenases of the β oxidation pathway in kidney mitochondria of ob/ob mice. There was, however, an increase in the rate of state 4 respiration, compared with the lean controls (Fig. 4B). This increase is most clearly observed for state 4 respiration supported by succinate (i.e., succinate → complex II → complex III → complex IV → O2), probably because succinate allowed the most rapid rate of O2 consumption. State 4 was induced by the addition of oligomycin to block H+ transfer through ATP synthase, from the outer surface of the inner mitochondrial membrane to the inner surface. When H+ transfer through ATP synthase is blocked, the proton gradient created by the electron transfer-driven proton pumps increases, which slows both proton pumping and electron transfer; this is seen as a decreased rate of O2 consumption compared with state 3 respiration. Electron transfer slows but does not cease in the absence of proton flow through ATP synthase because secondary pathways, such as uncoupling proteins, allow protons to flow across the inner mitochondrial membrane, albeit at a slower rate. The greater rate of state 4 respiration observed in the ob/ob kidney mitochondria reflects a greater capacity for these secondary pathways of H+ flow in these mitochondria. If these secondary pathways are also in operation when proton flow through ATP synthesis is operational (i.e., during state 3 respiration), some of the protons that would normally flow through ATP synthase will instead flow through the secondary pathways. This diversion of proton flow away from ATP synthase, termed partial uncoupling, leads to a slower rate of ATP synthesis, which should contribute to the decrease in kidney ATP levels in the ob/ob mice. We cannot claim that partial uncoupling fully accounts for the decrease in tissue ATP levels because we have no measure of the rate of cellular ATP consumption.
Fig. 4.
Mitochondrial respiration studies showed impaired mitochondrial state 3 respiration in LoxTB MC4R−/− mice kidneys under complex I substrate glutamate (a glucose substrate), palmitoyl-l-carnitine (an esterified fatty acid), and nonesterified fatty acid palmitoyl CoA+l-carnitine (A). Similar impairments to mitochondrial state 4 respiration were observed in LoxTB MC4R−/− mice kidneys under fatty acid substrates palmitoyl-l-carnitine and palmitoyl CoA+l-carnitine (B). In contrast, LoxTB MC4R−/− mice kidneys demonstrate an increased respiratory control ratio (RCR) under complex II glucose substrate succinate (C) and increase in complex IV (cytochrome c oxidase) activity (D). Although no changes in state 3 respiration or complex IV activity was observed, the ob/ob mice renal mitochondria show increased state 4 respiration under complex II glucose substrate succinate (B) and diminished RCR values under glucose substrates succinate and glutamate (C) but not under fatty acid substrates. State 3 and state 4 respiration values were normalized for citrate synthase (CS) activity in renal mitochondria. Values are means ± SE. *P < 0.05 compared with WT Control. †P < 0.05 compared with WT-LoxTB mice.
The degree to which electron transfer is coupled to ATP synthesis in isolated mitochondria is typically presented as the respiratory control ratio (RCR), the ratio of the state 3 and state 4 respiration rates. Hence, the lower RCR values observed for respiration supported by succinate and by glutamate-malate (Fig. 4C) provide another indication that electron transfer is partially uncoupled from ATP synthesis in mitochondria isolated from the kidneys of ob/ob mice. A lower RCR was not apparent using fatty acids as a substrate, perhaps because of the relatively low rates of state 3 respiration obtained with these substrates.
LoxTB MC4R−/− mice kidney mitochondria.
compared with lean controls, mitochondria isolated from the kidneys of obese LoxTB MC4R−/− mice showed significantly decreased (11–43%) rates of state 3 respiration supported by substrates that produce NADH and must use the pathway of complex I complex III → complex IV (Fig. 4A; glutamate-malate, palmitoyl CoA and palmitoyl l-carnitine). In contrast, state 3 respiration supported by succinate was normal, indicating that the pathway of complex II → complex III → complex IV was unaffected. This finding suggests that the kidney mitochondria isolated from LoxTB MC4R−/− mice contain lower than normal amounts of active complex I.
In contrast to ob/ob mitochondria, the rates of state 4 respiration in LoxTB MC4R−/− kidney mitochondria were not greater than the lean controls (Fig. 4B), and the RCR values were not significantly lower than the lean controls (Fig. 4C). Hence, the kidney mitochondria isolated from high-leptin LoxTB MC4R−/− mice did not show the partial uncoupling exhibited by the kidney mitochondria isolated from leptin-deficient ob/ob mice. Since normal coupling is retained, while complex I activity appears lower, we suggest that decreased ATP production resulting from lower complex I activity contributes to the observed decrease in tissue ATP levels in the kidneys of LoxTB MC4R−/− mice.
We thought that the apparent decline in complex I activity in LoxTB MC4R−/− mitochondria could be part of a general decrease in the components of oxidative phosphorylation, as has been observed in some studies (28). To test this, we measured the activity of complex IV (cytochrome c oxidase) (Fig. 4D). Instead of a decline, we found that the mitochondria in the kidneys of high-leptin LoxTB MC4R−/− mice contained 40% greater complex IV activity, relative to control (Fig. 4D). In contrast, no significant changes in complex IV activity were observed in kidneys of leptin-deficient ob/ob mice (Fig. 4D). This result rules out a general decrease in all complexes of oxidative phosphorylation in the kidneys of high-leptin LoxTB MC4R−/− mice. However, the mechanisms underlying the apparent decline in complex I activity as well as the increase in the amount of complex IV remain to be determined.
Markers of ER Stress
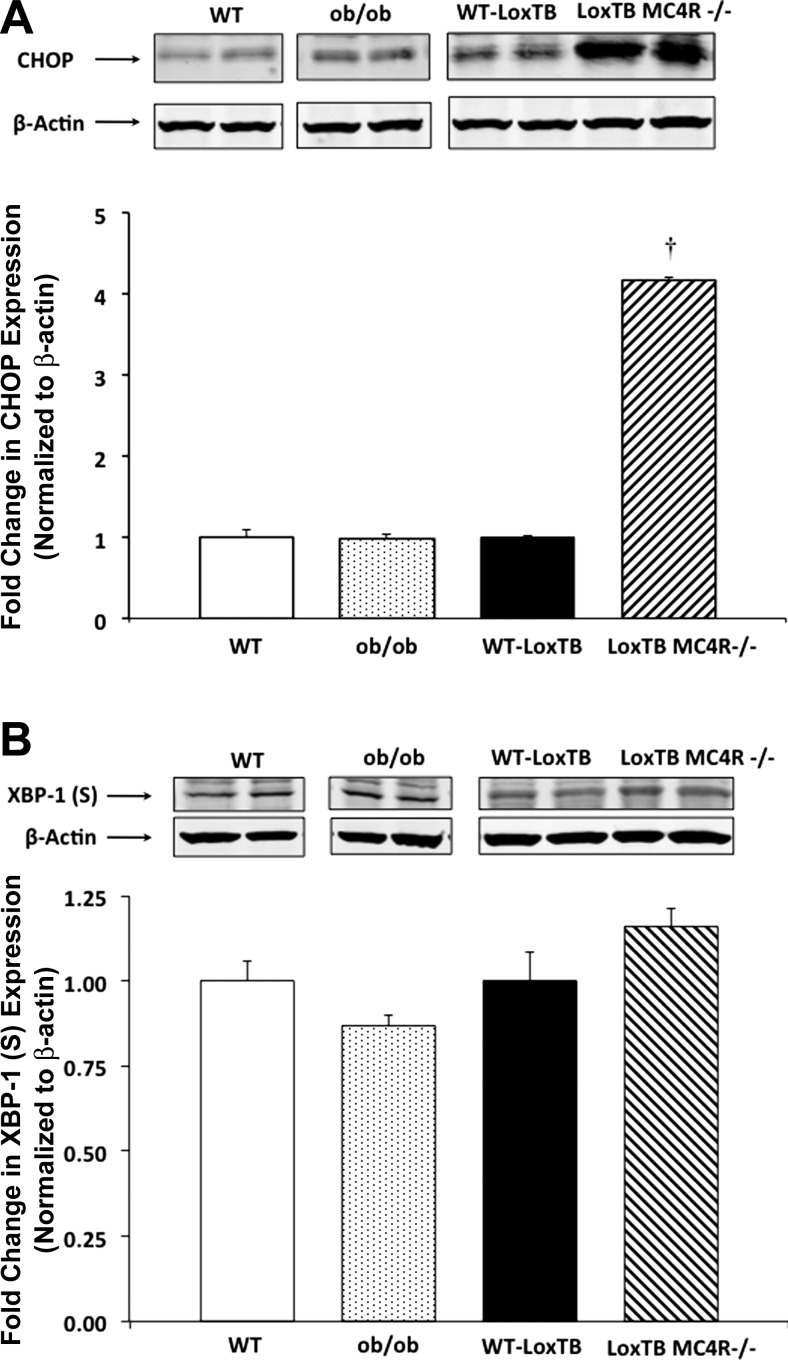
ER stress frequently accompanies mitochondrial dysfunction. Immunoblots of ob/ob and LoxTB MC4R−/− mice kidney homogenates were analyzed by densitometry for obesity-related changes in the expression of ER stress markers. We observed a fourfold induction of the proapoptotic protein CHOP (63) in LoxTB MC4R−/− mice kidneys (Fig. 5A), suggesting long-term ER stress. In terms of common markers of the cellular response to ER stress, a small increase was noted in the expression of the spliced form of X-box binding protein 1 (s-XBP-1) (75) in these kidneys (Fig. 5B), but the expression of the ER protein-folding chaperone GRP78 (70) remained unchanged (data not shown). No changes in the levels of ER stress and stress response markers were detected in ob/ob mice renal homogenates compared with lean controls.
Fig. 5.
Western blot and densitometry analysis showed increased expression of proapoptotic endoplasmic reticulum (ER) stress response marker CHOP (A) in LoxTB MC4R−/− mice kidneys with subtle, but not statistically significant, changes in the expression of other ER stress marker XBP-1 (spliced form; B) in LoxTB MC4R−/− and ob/ob mice kidneys with respect to their corresponding lean WT controls. Representative Western blots (n = 3) of ER stress markers [CHOP (A) and XBP-1 (B)] with β-actin used as loading control are shown at the top of each panel, and densitometry values (normalized to β-actin) of ER stress markers are shown at the bottom. Values are means ± SE. †P < 0.05 compared with WT-LoxTB.
DISCUSSION
Obesity, in the absence of hypertension, is associated with altered kidney function but not major renal injury. Previous studies in experimental animals and in humans have shown that obesity is associated with the development of chronic kidney injury, although the pathogenic mechanisms remain unclear. Metabolic, hormonal, and hemodynamic factors have all been proposed to contribute to renal injury in obese subjects. Hemodynamic changes include increased arterial pressure and glomerular hypertension associated with glomerular hyperfiltration (4, 38, 39).
Emerging evidence from animal models of diet-induced obesity strongly support the idea that obesity impairs renal architecture and function (18, 19, 73). For example, studies by Ruggiero et al. (73) showed that obesity induced by a high-fat diet (HFD) feeding in mice is associated with glomerular hypertrophy, mesangial expansion, and renal fibrosis. Similarly, studies conducted in HFD-fed obese mice by Decleves et al. (18, 19) also demonstrated that diet-induced obesity induces renal hypertrophy, alterations in renal lipid metabolism, and activation of inflammatory markers in the kidney. However, the obesity models used in these HFD studies also present with elevated arterial pressure (23, 36, 40), a well-recognized risk factor for the development of renal disease (39). This leads to the question of how obesity itself, with its associated metabolic changes, causes alterations in renal architecture and function, compared with obesity plus increased blood pressure (19, 72, 73).
In the present study, we used two genetic models of obesity, hyperleptinemic MC4R-deficient mice and leptin-deficient ob/ob mice. Both models have normal or slightly reduced blood pressures despite severe obesity and multiple metabolic abnormalities including insulin resistance, hyperinsulinemia, and dyslipidemia. Studies have shown that a functional central nervous system (CNS) leptin-melanocortin pathway is essential for obesity to cause sympathetic nervous system activation and hypertension in rodents as well as in humans (33, 37, 79). Therefore, LoxTB MC4R−/− and ob/ob mice provide an opportunity to study the impact of obesity and associated metabolic abnormalities on kidney function in the absence of hypertension (Table 3).
Table 3.
Summary of the findings from ob/ob and LoxTB MC4R−/− mice models compared with the findings from diet-induced obese animal models reported in the literature
| Parameter | ob/ob Mice Model | LoxTB MC4R−/− Mice Model | Diet-Induced Obesity (Based on Literature) |
|---|---|---|---|
| Leptin levels | None | Very high | Higher than normal (3) |
| Leptin CNS sympathetic effects | No | No | Yes (23, 79) |
| Leptin peripheral effects | No | Yes | Yes (15, 16) |
| Sympathetic activation | No | No | Yes (23, 39, 40) |
| Blood pressure | Normal | Normal | High (23, 37, 40) |
| Renal lipid accumulation | Yes | Yes | Yes (19) |
| Altered renal function | Yes | Yes | Yes (5, 18, 55, 95) |
| Renal injury | No | No | Yes (18, 19, 73) |
| Kidney mitochondrial dysfunction | Normal respiration rates but partial uncoupling, which may lower ROS production as well as ATP levels | Decreased respiration of substrates that require complex I may lower ATP levels; normal coupling | Normal respiration from succinate; normal coupling (73) |
| Kidney ATP levels | Lower than normal | Lower than normal | |
| Mitochondrial oxidative stress | No | Yes | Yes (62, 73) |
| Proapoptotic signaling in kidney | No | Yes (increased CHOP expression) | Yes (increased CHOP expression) (78, 90) |
CNS, central nervous system; ROS, reactive oxygen species.
Our results indicate that obesity, even when blood pressure is not elevated, leads to glomerular hyperfiltration and increased albumin excretion in ob/ob and LoxTB MC4R−/− mice. However, we found no major macrophage infiltration or substantial structural changes in the kidneys of LoxTB MC4R−/− or ob/ob mice. Therefore, a surprising finding is that severe obesity with its associated metabolic abnormalities in mice may not cause major renal injury in the absence of sympathetic activation and hypertension. Increased sympathetic nervous system activity and hypertension, interacting synergistically with obesity-induced metabolic changes, may be required for obesity to cause major renal injury. This explanation is consistent with the finding that tight pharmacological control of hypertension appears to protect against development and progression of chronic kidney disease in obese type 2 diabetic patients even when metabolic abnormalities and obesity are poorly controlled (14).
Obesity promotes renal TG accumulation. Previous studies in leptin-deficient ob/ob mice have documented excessive fat accumulation in visceral organs (94, 96) and pancreatic islets (99). However, administration of leptin attenuates fat accumulation in isolated rodent hearts (7) and skeletal muscle (58). Thus we initially hypothesized that the high leptin levels in LoxTB MC4R−/− mice would protect the kidneys from excessive fat deposition despite obesity. This hypothesis turned out to be incorrect. Both the leptin-deficient and the leptin-abundant mouse models of obesity showed significant hypertrophy of the kidney (and also of the liver) plus an expansion of fat pads surrounding the peritoneum and kidney. In fact, both obese mouse models showed similar increases in kidney fat (∼1.7-fold increase for the LoxTB MC4R−/− mice and ∼1.8-fold increase for the ob/ob mice compared with their respective lean controls). It is interesting to note that in both mouse models of genetic obesity, the changes in kidney function, including increased creatinine clearance and albumin excretion, occurred in proportion to the percent increase in intrarenal fat deposition. This observation suggests, but does not prove, a causal link between the renal fat accumulation and altered renal function.
Obesity is associated with decreased kidney ATP levels and altered mitochondrial function. Both obese mouse models exhibited significantly decreased kidney ATP levels, compared with their lean controls, which implies less efficient oxidative phosphorylation in kidney mitochondria. This was tested by examining the respiratory activity of isolated kidney mitochondria.
Mitochondria isolated from kidneys of leptin-deficient ob/ob mice respired both fatty acid and non-fatty acid substrates at rates similar to lean controls (Fig. 4). However, electron transfer was partially uncoupled from ATP synthesis in the ob/ob mice, as explained in results. The partial uncoupling of oxidative phosphorylation likely contributes to the observed 25% decline in whole kidney ATP levels, also as explained in results. Partial uncoupling has also been observed in mitochondria isolated from the livers of ob/ob mice (57, 80).
In contrast to ob/ob mice, the RCR values measured for kidney mitochondria of LoxTB MC4R−/− mice indicated normal coupling of electron transfer to ATP synthesis (Fig. 4). In addition, mitochondria isolated from kidneys of hyperleptinemic LoxTB MC4R−/− mice showed normal rates of respiration of succinate, but decreased respiration rates of substrates that require complex I activity, such as glutamate-malate and fatty acids (Fig. 4). A deficiency in complex I activity could decrease mitochondrial ATP production and thereby contribute to the decrease in kidney ATP levels in the LoxTB MC4R−/− mice (Fig. 3B). It is also likely that the increased oxidative damage observed in these mitochondria may impair mitochondrial ATP production by lowering the activity of mitochondrial enzymes and complexes.
The kidney mitochondria of the obese mouse models in this study show dysfunctions that can at least partially explain the finding of lower tissue ATP levels. However, the types of mitochondrial dysfunction seen with high leptin and leptin deficiency are distinctly different (Table 3).
Mitochondria of obese mice with sustained high levels of leptin, comparable to those found in LoxTB MC4R−/− mice, have not previously been studied. However, mice fed a HFD for an extended time do show a sustained increase in plasma leptin, although the absolute amount of leptin is far lower than that of LoxTB MC4R−/− mice (3). Ruggiero et al. (73) analyzed kidney mitochondria isolated from male C57BL/6 mice fed a HFD for 12–16 wk. Their findings are more similar to what we have found for the hyperleptinemic LoxTB MC4R−/− mice than for the leptin-deficient ob/ob mice (Table 3). In the HFD-fed mice of Ruggiero et al. (73), the RCR measured during the respiration of succinate was either slightly elevated or unchanged, indicating retention of normal coupling. This is similar to the hyperleptinemic LoxTB MC4R−/− mice (Fig. 4). After 16 wk, the expression of complex I in the HFD mice was ∼20% less than in the low-fat diet mice, although the result was not significant (74). This is similar to our finding of a loss of complex I activity in the kidney mitochondria of hyperleptinemic mice.
High leptin levels are associated with an increase in oxidized protein in kidney mitochondria. Previous studies have supported a role for oxidative stress in promoting renal injury during obesity hypertension (22, 62, 73, 74, 81). However, the role of leptin in obesity-related renal oxidative stress is less clear. In this study, we found that mitochondria from the kidneys of leptin-deficient ob/ob mice showed no significant elevation in protein carbonylation, indicating that the mitochondria were not producing ROS in excess of mitochondrial antioxidant systems. The lack of increased oxidative damage in ob/ob mitochondria is likely due to the observed partial uncoupling, which lessens the production of superoxide and downstream ROS during respiration (6). Livers of ob/ob mice have indications of oxidative damage, but the principal finding in these studies is increased protein nitration, possibly due to increased mitochondrial nitric oxide production (25, 28, 29). Increased protein nitration is not solely induced by low leptin levels, however, since it also occurs in the high-leptin environment produced by a HFD (73).
In hyperleptinemic LoxTB MC4R−/− mice, the carbonylation of kidney mitochondrial proteins was increased, indicating the production of ROS in excess of antioxidant capacity. Obese animal models in which leptin levels are elevated have generally shown increased production of mitochondrial ROS (1, 15, 16, 69, 71, 73, 86). This is often attributed to the increased oxidation of fatty acids, which is promoted by leptin (7, 13, 17, 68, 93, 99). Consistent with our finding of increased proton carbonylation, kidney mitochondria of mice made obese by a HFD release more hydrogen peroxide during state 3 respiration than mitochondria from mice fed a control diet (73).
Obesity in the presence of high leptin levels is associated with ER/mitochondrial stress. Increased ROS and depletion of energy slow protein folding in the ER, a condition known as ER stress (44). Chronic ER stress leads to apoptosis via the expression of proapoptotic ER stress proteins, such as CHOP, and loss of cells contributes to tissue dysfunction (44, 50). A response to ER stress, termed the unfolded protein response (UPR), involves increased expression of protein-folding agents, such as the ER chaperone termed GRP78 (11). Studies in obese animals have demonstrated activation of this ER stress response in the liver (65), brain (64), and kidney (78). In most of these studies, however, it has been difficult to separate the effects of obesity on ER stress from the effects of hypertension, which can increase ER stress (64, 78). In this study, we examined the effect of obesity on kidney ER stress without the complications of hypertension in two mouse models with very different levels of leptin.
The apparent lack of ER stress in kidneys of leptin-deficient ob/ob mice is consistent with the lack of mitochondrial protein carbonylation, since mitochondrial ROS are primary contributors to both ER stress and protein carbonylation. In contrast, the kidneys of hyperleptinemic LoxTB MC4R−/− mice showed a marked increase in the levels of CHOP, a strong indication of advanced ER stress (63). Consistent with our findings, induction of the ER stress marker CHOP has also been documented in in vitro (48, 49) and in vivo (78, 90) models of obesity-induced nephropathy. Together, these findings unveil a potential role of ER stress in obesity-induced renal dysfunction during MC4R deficiency.
In terms of a response to ER stress, only a slight increase in the spliced form of XBP-1 (s-XBP-1) was observed, and there was no increase in the expression of ER chaperone GRP78. This unique pattern of the UPR, evidenced by a solitary increase in CHOP, in conjunction with increased mitochondrial protein carbonylation in the high-leptin MC4R-deficient mice kidneys, suggests the involvement of mitochondrial stress (42, 98), a condition similar to ER stress. In support of this possibility, elevated mitochondrial ROS has been shown to increase the expression of CHOP in adipocytes (12). Similarly, reduced complex I activity, another key finding in the LoxTB MC4R−/− mice kidneys, induces the expression of CHOP in cybrid cells (27). Therefore, the increased levels of CHOP in MC4R-deficient mice kidneys could be a response to one or more of the following: 1) ER UPR, 2) mitochondrial UPR, 3) mitochondrial ROS, or 4) decreased complex I activity. However, our results appear to indicate that high leptin levels in the kidney promote apoptosis or cell cycle arrest via induction of CHOP more than high leptin promotes the UPR in the ER.
One potential criticism of our studies is that LoxTB MC4R−/− mice are not only obese and hyperleptinemic, but also have MC4R deficiency which could have effects on kidney function independently of obesity and increased leptin levels. However, in adult mammals MC4R is expressed mainly in the CNS, especially in the hypothalamus, brain stem, and spinal cord intermediolateral nucleus (88). Although MC4R may be expressed in several peripheral tissues during fetal development, current evidence from Northern blotting, in situ hybridization, and radioligand binding studies suggests that MC4R is not expressed, or expressed at very low levels, in the kidneys of adult rodents (88). Therefore, the effects of MC4R deficiency on kidney function are likely to be related to indirect effects caused by altered CNS function. As discussed previously, MC4R deficiency in the CNS causes hyperphagia, obesity, and hyperleptinemia, which are not associated with increased sympathetic activity or hypertension. Abnormalities of kidney function observed in LoxTB MC4R−/− mice may therefore be caused mainly by obesity and associated metabolic effects, including hyperleptinemia. However, we cannot completely rule out the possibility that MC4R deficiency in the kidneys during fetal development may have contributed to some of the renal dysfunction observed in LoxTB MC4R−/− mice.
Summary and Perspectives
Using two genetic mice models of obesity, MC4R-deficient (hyperleptinemic) mice and leptin-deficient ob/ob mice, we demonstrated that obesity, in the absence of hypertension, is associated with glomerular hyperfiltration and increased albumin excretion, but not with major renal injury. Our findings also help to define which obesity-related kidney changes are associated with leptin signaling and which are not. For example, obesity-related changes in renal TG accumulation and renal function were similar in the presence or absence of leptin. Similarly, mitochondrial dysfunction and a decline in renal ATP were present in both obesity models, regardless of the presence or absence of leptin; however, the type of mitochondrial dysfunction differed significantly depending on the leptin status. In mitochondria from kidneys of hyperleptinemic LoxTB MC4R−/− mice, oxidative phosphorylation and respiration of fatty acids are impaired by an apparent deficiency in complex I, while in the leptin-deficient kidneys of ob/ob mice oxidative phosphorylation is impaired by partial uncoupling of electron transfer from ATP synthesis. Another major difference was that mitochondrial oxidative stress and ER/mitochondrial stress in obese mice were evident only in the presence of leptin. Nevertheless, the absence of the MC4R in hyperleptinemic LoxTB MC4R−/− mice must also be taken into consideration when our results are analyzed.
Table 3 compares our findings from the ob/ob and LoxTB MC4R−/− mice models with findings from diet-induced obese animal models, which have elevated leptin. There are similarities and differences. As noted, mice made obese by a HFD are hypertensive. All of the obese models show altered renal function, but only the hypertensive obese mice exhibit substantial renal injury. In high-leptin mice fed a HFD and hyperleptinemic LoxTB MC4R−/− mice, kidney oxidative phosphorylation remains coupled, while in leptin-deficient ob/ob mice it is partially uncoupled. The kidneys of both groups of mice with high levels of leptin show evidence of oxidative stress in mitochondria as well as increased expression of the proapoptotic protein CHOP. Neither of these abnormalities occurs in obese ob/ob mice with leptin deficiency.
Resistance to overt renal injury in these two normotensive genetic models of obesity may also be due, in part, to other factors besides lack of increased blood pressure. Previous studies have shown that blockade of nitric oxide synthesis, for example, renders mice much more susceptible to diabetes-induced nephropathy (60). However, blockade of nitric oxide synthesis also increased arterial pressure in these previous studies, making it difficult to assess the contribution of hemodynamic changes to the renal injury. Further studies are needed to determine whether increases in arterial pressure may interact synergistically with metabolic changes to mediate obesity-induced ER stress, mitochondrial dysfunction, oxidative stress, and other changes that may contribute to renal injury in obese subjects.
Overall, our findings indicate that obesity, independently of leptin signaling, can cause kidney mitochondrial dysfunction even in the absence of hypertension, although increased leptin associated with MC4R deficiency may contribute to oxidative stress and a proapoptotic response via ER or mitochondrial stress.
GRANTS
This research was supported by National Heart, Lung, and Blood Institute Grant (PO1HL51971), the National Institute of General Medical Sciences (P20GM104357), and an American Heart Association (Greater Southeast Affiliate) Postdoctoral Fellowship Grant (09POST2300079) to S. Munusamy and a Scientist Development Grant (AHA58161) to J. M. do Carmo.
DISCLOSURES
No conflicts of interest, financial or otherwise, are declared by the authors.
AUTHOR CONTRIBUTIONS
Author contributions: S.M., J.M.d.C., J.P.H., and J.E.H. provided conception and design of research; S.M. and J.M.d.C. performed experiments; S.M. and J.M.d.C. analyzed data; S.M., J.P.H., and J.E.H. interpreted results of experiments; S.M. prepared figures; S.M. drafted manuscript; S.M., J.M.d.C., J.P.H., and J.E.H. edited and revised manuscript; J.P.H. and J.E.H. approved final version of manuscript.
ACKNOWLEDGMENTS
We thank Haiyan Zhang for technical assistance and Stephanie Lucas for help with proofreading and submission of the manuscript.
REFERENCES
- 1.Abdurrachim D, Ciapaite J, Wessels B, Nabben M, Luiken JJ, Nicolay K, Prompers JJ. Cardiac diastolic dysfunction in high-fat diet fed mice is associated with lipotoxicity without impairment of cardiac energetics in vivo. Biochim Biophys Acta 1842: 1525–1537, 2014. [DOI] [PubMed] [Google Scholar]
- 2.Agnani S, Vachharajani V, Gupta R, Atray N, Vachharajani T. Does treating obesity stabilize chronic kidney disease? BMC Nephrol 6: 7, 2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ahren B, Mansson S, Gingerich RL, Havel PJ. Regulation of plasma leptin in mice: influence of age, high-fat diet, and fasting. Am J Physiol Regul Integr Comp Physiol 273: R113–R120, 1997. [DOI] [PubMed] [Google Scholar]
- 4.Alicic RZ, Patakoti R, Tuttle KR. Direct and indirect effects of obesity on the kidney. Adv Chronic Kidney Dis 20: 121–127, 2013. [DOI] [PubMed] [Google Scholar]
- 5.Aon MA, Bhatt N, Cortassa SC. Mitochondrial and cellular mechanisms for managing lipid excess. Front Physiol 5: 282, 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Aon MA, Cortassa S, O'Rourke B. Redox-optimized ROS balance: a unifying hypothesis. Biochim Biophys Acta 1797: 865–877, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Atkinson LL, Fischer MA, Lopaschuk GD. Leptin activates cardiac fatty acid oxidation independent of changes in the AMP-activated protein kinase-acetyl-CoA carboxylase-malonyl-CoA axis. J Biol Chem 277: 29424–29430, 2002. [DOI] [PubMed] [Google Scholar]
- 8.Balthasar N, Dalgaard LT, Lee CE, Yu J, Funahashi H, Williams T, Ferreira M, Tang V, McGovern RA, Kenny CD, Christiansen LM, Edelstein E, Choi B, Boss O, Aschkenasi C, Zhang CY, Mountjoy K, Kishi T, Elmquist JK, Lowell BB. Divergence of melanocortin pathways in the control of food intake and energy expenditure. Cell 123: 493–505, 2005. [DOI] [PubMed] [Google Scholar]
- 9.Birse RT, Bodmer R. Lipotoxicity and cardiac dysfunction in mammals and Drosophila. Crit Rev Biochem Mol Biol 46: 376–385, 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Boren J, Taskinen MR, Olofsson SO, Levin M. Ectopic lipid storage and insulin resistance: a harmful relationship. J Intern Med 274: 25–40, 2013. [DOI] [PubMed] [Google Scholar]
- 11.Borradaile NM, Han X, Harp JD, Gale SE, Ory DS, Schaffer JE. Disruption of endoplasmic reticulum structure and integrity in lipotoxic cell death. J Lipid Res 47: 2726–2737, 2006. [DOI] [PubMed] [Google Scholar]
- 12.Carrière A, Carmona MC, Fernandez Y, Rigoulet M, Wenger RH, Pénicaud L, Casteilla L. Mitochondrial reactive oxygen species control the transcription factor CHOP-10/GADD153 and adipocyte differentiation. J Biol Chem 279: 40462–40469, 2004. [DOI] [PubMed] [Google Scholar]
- 13.Ceddia RB. Direct metabolic regulation in skeletal muscle and fat tissue by leptin: implications for glucose and fatty acids homeostasis. Int J Obes (Lond) 29: 1175–1183, 2005. [DOI] [PubMed] [Google Scholar]
- 14.Chadban S, Howell M, Twigg S, Thomas M, Jerums G, Cass A, Campbell D, Nicholls K, Tong A, Mangos G, Stack A, MacIsaac RJ, Girgis S, Colagiuri R, Colagiuri S, Craig J, CARI. The CARI guidelines. Prevention and management of chronic kidney disease in type 2 diabetes. Nephrology 15, Suppl 1: S162–S194, 2010. [DOI] [PubMed] [Google Scholar]
- 15.Ciapaite J, Bakker SJ, Van Eikenhorst G, Wagner MJ, Teerlink T, Schalkwijk CG, Fodor M, Ouwens DM, Diamant M, Heine RJ, Westerhoff HV, Krab K. Functioning of oxidative phosphorylation in liver mitochondria of high-fat diet fed rats. Biochim Biophys Acta 1772: 307–316, 2007. [DOI] [PubMed] [Google Scholar]
- 16.Ciapaite J, van den Broek NM, Te Brinke H, Nicolay K, Jeneson JA, Houten SM, Prompers JJ. Differential effects of short- and long-term high-fat diet feeding on hepatic fatty acid metabolism in rats. Biochim Biophys Acta 1811: 441–451, 2011. [DOI] [PubMed] [Google Scholar]
- 17.Cortes VA, Cautivo KM, Rong S, Garg A, Horton JD, Agarwal AK. Leptin ameliorates insulin resistance and hepatic steatosis in Agpat2-/- lipodystrophic mice independent of hepatocyte leptin receptors. J Lipid Res 55: 276–288, 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Decleves AE, Mathew AV, Cunard R, Sharma K. AMPK mediates the initiation of kidney disease induced by a high-fat diet. J Am Soc Nephrol 22: 1846–1855, 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Decleves AE, Zolkipli Z, Satriano J, Wang L, Nakayama T, Rogac M, Le TP, Nortier JL, Farquhar MG, Naviaux RK, Sharma K. Regulation of lipid accumulation by AMP-activated kinase [corrected] in high fat diet-induced kidney injury. Kidney Int 85: 611–623, 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.do Carmo JM, Tallam LS, Roberts JV, Brandon EL, Biglane J, da Silva AA, Hall JE. Impact of obesity on renal structure and function in the presence and absence of hypertension: evidence from melanocortin-4 receptor-deficient mice. Am J Physiol Regul Integr Comp Physiol 297: R803–R812, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.do Carmo JM, Bassi M, da Silva AA, Hall JE. Control of cardiovascular, metabolic and respiratory functions during prolonged obesity in leptin-deficient and diet-induced obese mice. Hypertension 15: 109, 2008. [Google Scholar]
- 22.Ebenezer PJ, Mariappan N, Elks CM, Haque M, Francis J. Diet-induced renal changes in Zucker rats are ameliorated by the superoxide dismutase mimetic TEMPOL. Obesity (Silver Spring) 17: 1994–2002, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Erdos B, Broxson CS, Cudykier I, Basgut B, Whidden M, Landa T, Scarpace PJ, Tumer N. Effect of high-fat diet feeding on hypothalamic redox signaling and central blood pressure regulation. Hypertens Res 32: 983–988, 2009. [DOI] [PubMed] [Google Scholar]
- 24.Farooqi IS, Keogh JM, Yeo GS, Lank EJ, Cheetham T, O'Rahilly S. Clinical spectrum of obesity and mutations in the melanocortin 4 receptor gene. N Engl J Med 348: 1085–1095, 2003. [DOI] [PubMed] [Google Scholar]
- 25.Finocchietto PV, Holod S, Barreyro F, Peralta JG, Alippe Y, Giovambattista A, Carreras MC, Poderoso JJ. Defective leptin-AMP-dependent kinase pathway induces nitric oxide release and contributes to mitochondrial dysfunction and obesity in ob/ob mice. Antioxid Redox Signal 15: 2395–2406, 2011. [DOI] [PubMed] [Google Scholar]
- 26.Folch J, Lees M, Stanley GHS. A simple method for the isolation and purification of total lipids from animal tissues. J Biol Chem 226: 497–509, 1957. [PubMed] [Google Scholar]
- 27.Fujita Y, Ito M, Nozawa Y, Yoneda M, Oshida Y, Tanaka M. CHOP (C/EBP homologous protein) and ASNS (asparagine synthetase) induction in cybrid cells harboring MELAS and NARP mitochondrial DNA mutations. Mitochondrion 7: 80–88, 2007. [DOI] [PubMed] [Google Scholar]
- 28.Garcia-Ruiz I, Rodriguez-Juan C, Diaz-Sanjuan T, del Hoyo P, Colina F, Munoz-Yague T, Solis-Herruzo JA. Uric acid and anti-TNF antibody improve mitochondrial dysfunction in ob/ob mice. Hepatology 44: 581–591, 2006. [DOI] [PubMed] [Google Scholar]
- 29.Garcia-Ruiz I, Rodriguez-Juan C, Diaz-Sanjuan T, Martinez MA, Munoz-Yague T, Solis-Herruzo JA. Effects of rosiglitazone on the liver histology and mitochondrial function in ob/ob mice. Hepatology 46: 414–423, 2007. [DOI] [PubMed] [Google Scholar]
- 30.Granell S, Mohammad S, Ramanagoudr-Bhojappa R, Baldini G. Obesity-linked variants of melanocortin-4 receptor are misfolded in the endoplasmic reticulum and can be rescued to the cell surface by a chemical chaperone. Mol Endocrinol 24: 1805–1821, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Granell S, Serra-Juhe C, Martos-Moreno GA, Diaz F, Perez-Jurado LA, Baldini G, Argente J. A novel melanocortin-4 receptor mutation MC4R-P272L associated with severe obesity has increased propensity to be ubiquitinated in the ER in the face of correct folding. PLoS One 7: e50894, 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Green CJ, Hodson L. The influence of dietary fat on liver fat accumulation. Nutrients 6: 5018–5033, 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Greenfield JR, Miller JW, Keogh JM, Henning E, Satterwhite JH, Cameron GS, Astruc B, Mayer JP, Brage S, See TC, Lomas DJ, O'Rahilly S, Farooqi IS. Modulation of blood pressure by central melanocortinergic pathways. N Engl J Med 360: 44–52, 2009. [DOI] [PubMed] [Google Scholar]
- 34.Grimsrud PA, Xie H, Griffin TJ, Bernlohr DA. Oxidative stress and covalent modification of protein with bioactive aldehydes. J Biol Chem 283: 21837–21841, 2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Gusdon AM, Song KX, Qu S. Nonalcoholic fatty liver disease: pathogenesis and therapeutics from a mitochondria-centric perspective. Oxid Med Cell Longev 2014: 637027, 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Hall JE, Brands MW, Dixon WN, Smith MJ Jr. Obesity-induced hypertension. Renal function and systemic hemodynamics. Hypertension 22: 292–299, 1993. [DOI] [PubMed] [Google Scholar]
- 37.Hall JE, da Silva AA, do Carmo JM, Dubinion J, Hamza S, Munusamy S, Smith G, Stec DE. Obesity-induced hypertension: role of sympathetic nervous system, leptin, and melanocortins. J Biol Chem 285: 17271–17276, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Hall JE, Henegar JR, Dwyer TM, Liu J, Da Silva AA, Kuo JJ, Tallam L. Is obesity a major cause of chronic kidney disease? Adv Ren Replace Ther 11: 41–54, 2004. [DOI] [PubMed] [Google Scholar]
- 39.Hall ME, do Carmo JM, da Silva AA, Juncos LA, Wang Z, Hall JE. Obesity, hypertension, and chronic kidney disease. Int J Nephrol Renovasc Dis 7: 75–88, 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Henegar JR, Bigler SA, Henegar LK, Tyagi SC, Hall JE. Functional and structural changes in the kidney in the early stages of obesity. J Am Soc Nephrol 12: 1211–1217, 2001. [DOI] [PubMed] [Google Scholar]
- 41.Hill CJ, Cardwell CR, Maxwell AP, Young RJ, Matthews B, O'Donoghue DJ, Fogarty DG. Obesity and kidney disease in type 1 and 2 diabetes: an analysis of the National Diabetes Audit. QJM 106: 933–942, 2013. [DOI] [PubMed] [Google Scholar]
- 42.Horibe T, Hoogenraad NJ. The chop gene contains an element for the positive regulation of the mitochondrial unfolded protein response. PLoS One 2: e835, 2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Hsu CY, McCulloch CE, Iribarren C, Darbinian J, Go AS. Body mass index and risk for end-stage renal disease. Ann Intern Med 144: 21–28, 2006. [DOI] [PubMed] [Google Scholar]
- 44.Inagi R. Endoplasmic reticulum stress as a progression factor for kidney injury. Curr Opin Pharmacol 10: 156–165, 2010. [DOI] [PubMed] [Google Scholar]
- 45.Inagi R, Nangaku M, Onogi H, Ueyama H, Kitao Y, Nakazato K, Ogawa S, Kurokawa K, Couser WG, Miyata T. Involvement of endoplasmic reticulum (ER) stress in podocyte injury induced by excessive protein accumulation. Kidney Int 68: 2639–2650, 2005. [DOI] [PubMed] [Google Scholar]
- 46.Iseki K, Ikemiya Y, Kinjo K, Inoue T, Iseki C, Takishita S. Body mass index and the risk of development of end-stage renal disease in a screened cohort. Kidney Int 65: 1870–1876, 2004. [DOI] [PubMed] [Google Scholar]
- 47.Jain SS, Paglialunga S, Vigna C, Ludzki A, Herbst EA, Lally JS, Schrauwen P, Hoeks J, Tupling AR, Bonen A, Holloway GP. High-fat diet-induced mitochondrial biogenesis is regulated by mitochondrial-derived reactive oxygen species activation of CaMKII. Diabetes 63: 1907–1913, 2014. [DOI] [PubMed] [Google Scholar]
- 48.Katsoulieris E, Mabley JG, Samai M, Green IC, Chatterjee PK. Alpha-Linolenic acid protects renal cells against palmitic acid lipotoxicity via inhibition of endoplasmic reticulum stress. Eur J Pharmacol 623: 107–112, 2009. [DOI] [PubMed] [Google Scholar]
- 49.Katsoulieris E, Mabley JG, Samai M, Sharpe MA, Green IC, Chatterjee PK. Lipotoxicity in renal proximal tubular cells: relationship between endoplasmic reticulum stress and oxidative stress pathways. Free Radic Biol Med 48: 1654–1662, 2010. [DOI] [PubMed] [Google Scholar]
- 50.Kawakami T, Inagi R, Takano H, Sato S, Ingelfinger JR, Fujita T, Nangaku M. Endoplasmic reticulum stress induces autophagy in renal proximal tubular cells. Nephrol Dial Transplant 24: 2665–2672, 2009. [DOI] [PubMed] [Google Scholar]
- 51.Kimura K, Jin H, Ogawa M, Aoe T. Dysfunction of the ER chaperone BiP accelerates the renal tubular injury. Biochem Biophys Res Commun 366: 1048–1053, 2008. [DOI] [PubMed] [Google Scholar]
- 52.Kitamura M. Endoplasmic reticulum stress and unfolded protein response in renal pathophysiology: Janus faces. Am J Physiol Renal Physiol 295: F323–F334, 2008. [DOI] [PubMed] [Google Scholar]
- 53.Lezcano EJ, Inigo P, Larraga AM, Barranquero C, Gimenez I, Osada J. Caloric restriction or telmisartan control dyslipidemia and nephropathy in obese diabetic Zucker rats. Diabetol Metab Syndr 6: 10, 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Mailloux RJ, Harper ME. Uncoupling proteins and the control of mitochondrial reactive oxygen species production. Free Radic Biol Med 51: 1106–1115, 2011. [DOI] [PubMed] [Google Scholar]
- 55.Marciniak C, Marechal X, Montaigne D, Neviere R, Lancel S. Cardiac contractile function and mitochondrial respiration in diabetes-related mouse models. Cardiovasc Diabetol 13: 118, 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Meehan TP, Tabeta K, Du X, Woodward LS, Firozi K, Beutler B, Justice MJ. Point mutations in the melanocortin-4 receptor cause variable obesity in mice. Mamm Genome 17: 1162–1171, 2006. [DOI] [PubMed] [Google Scholar]
- 57.Melia HP, Andrews JF, McBennett SM, Porter RK. Effects of acute leptin administration on the differences in proton leak rate in liver mitochondria from ob/ob mice compared to lean controls. FEBS Lett 458: 261–264, 1999. [DOI] [PubMed] [Google Scholar]
- 58.Minokoshi Y, Kim YB, Peroni OD, Fryer LG, Muller C, Carling D, Kahn BB. Leptin stimulates fatty-acid oxidation by activating AMP-activated protein kinase. Nature 415: 339–343, 2002. [DOI] [PubMed] [Google Scholar]
- 59.Morris DL, Rui L. Recent advances in understanding leptin signaling and leptin resistance. Am J Physiol Endocrinol Metab 297: E1247–E1259, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Nakagawa T, Sato W, Glushakova O, Heinig M, Clarke T, Campbell-Thompson M, Yuzawa Y, Atkinson MA, Johnson RJ, Croker B. Diabetic endothelial nitric oxide synthase knockout mice develop advanced diabetic nephropathy. J Am Soc Nephrol 18: 539–550, 2007. [DOI] [PubMed] [Google Scholar]
- 61.Nakano D, Diah S, Kitada K, Hitomi H, Mori H, Masaki T, Kobori H, Nishiyama A. Short-term calorie restriction in early life attenuates the development of proteinuria but not glucose intolerance in type 2 diabetic OLETF rats. ISRN Endocrinol 2011: 768637, 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Noeman SA, Hamooda HE, Baalash AA. Biochemical study of oxidative stress markers in the liver, kidney and heart of high fat diet induced obesity in rats. Diabetol Metab Syndr 3: 17, 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Oyadomari S, Mori M. Roles of CHOP/GADD153 in endoplasmic reticulum stress. Cell Death Differ 11: 381–389, 2004. [DOI] [PubMed] [Google Scholar]
- 64.Ozcan L, Ergin AS, Lu A, Chung J, Sarkar S, Nie D, Myers MG Jr, Ozcan U. Endoplasmic reticulum stress plays a central role in development of leptin resistance. Cell Metab 9: 35–51, 2009. [DOI] [PubMed] [Google Scholar]
- 65.Ozcan U, Cao Q, Yilmaz E, Lee AH, Iwakoshi NN, Ozdelen E, Tuncman G, Gorgun C, Glimcher LH, Hotamisligil GS. Endoplasmic reticulum stress links obesity, insulin action, and type 2 diabetes. Science 306: 457–461, 2004. [DOI] [PubMed] [Google Scholar]
- 66.Pelleymounter MA, Cullen MJ, Baker MB, Hecht R, Winters D, Boone T, Collins F. Effects of the obese gene product on body weight regulation in ob/ob mice. Science 269: 540–543, 1995. [DOI] [PubMed] [Google Scholar]
- 67.Podkowka-Sieczka R, Wieczorowska-Tobis K, Niemir ZI, Styszynski A, Breborowicz A, Oreopoulos DG. The effect on renal structure and function of late-life-introduced caloric restriction (CR) in rats. Int Urol Nephrol 41: 211–217, 2009. [DOI] [PubMed] [Google Scholar]
- 68.Prieur X, Tung YC, Griffin JL, Farooqi IS, O'Rahilly S, Coll AP. Leptin regulates peripheral lipid metabolism primarily through central effects on food intake. Endocrinology 149: 5432–5439, 2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Raffaella C, Francesca B, Italia F, Marina P, Giovanna L, Susanna I. Alterations in hepatic mitochondrial compartment in a model of obesity and insulin resistance. Obesity (Silver Spring) 16: 958–964, 2008. [DOI] [PubMed] [Google Scholar]
- 70.Rao RV, Peel A, Logvinova A, del Rio G, Hermel E, Yokota T, Goldsmith PC, Ellerby LM, Ellerby HM, Bredesen DE. Coupling endoplasmic reticulum stress to the cell death program: role of the ER chaperone GRP78. FEBS Lett 514: 122–128, 2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Rindler PM, Plafker SM, Szweda LI, Kinter M. High dietary fat selectively increases catalase expression within cardiac mitochondria. J Biol Chem 288: 1979–1990, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Roberts CK, Barnard RJ, Sindhu RK, Jurczak M, Ehdaie A, Vaziri ND. Oxidative stress and dysregulation of NAD(P)H oxidase and antioxidant enzymes in diet-induced metabolic syndrome. Metabolism 55: 928–934, 2006. [DOI] [PubMed] [Google Scholar]
- 73.Ruggiero C, Ehrenshaft M, Cleland E, Stadler K. High-fat diet induces an initial adaptation of mitochondrial bioenergetics in the kidney despite evident oxidative stress and mitochondrial ROS production. Am J Physiol Endocrinol Metab 300: E1047–E1058, 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Sabuhi R, Ali Q, Asghar M, Al-Zamily NR, Hussain T. Role of the angiotensin II AT2 receptor in inflammation and oxidative stress: opposing effects in lean and obese Zucker rats. Am J Physiol Renal Physiol 300: F700–F706, 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Samali A, Fitzgerald U, Deegan S, Gupta S. Methods for monitoring endoplasmic reticulum stress and the unfolded protein response. Int J Cell Biol 2010: 830307, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Santini F, Maffei M, Pelosini C, Salvetti G, Scartabelli G, Pinchera A. Melanocortin-4 receptor mutations in obesity. Adv Clin Chem 48: 95–109, 2009. [PubMed] [Google Scholar]
- 77.Schrauwen P, Schrauwen-Hinderling V, Hoeks J, Hesselink MK. Mitochondrial dysfunction and lipotoxicity. Biochim Biophys Acta 1801: 266–271, 2010. [DOI] [PubMed] [Google Scholar]
- 78.Sharkey D, Fainberg HP, Wilson V, Harvey E, Gardner DS, Symonds ME, Budge H. Impact of early onset obesity and hypertension on the unfolded protein response in renal tissues of juvenile sheep. Hypertension 53: 925–931, 2009. [DOI] [PubMed] [Google Scholar]
- 79.Simonds SE, Pryor JT, Ravussin E, Greenway FL, Dileone R, Allen AM, Bassi J, Elmquist JK, Keogh JM, Henning E, Myers MG Jr, Licinio J, Brown RD, Enriori PJ, O'Rahilly S, Sternson SM, Grove KL, Spanswick DC, Farooqi IS, Cowley MA. Leptin mediates the increase in blood pressure associated with obesity. Cell 159: 1404–1416, 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Singh A, Wirtz M, Parker N, Hogan M, Strahler J, Michailidis G, Schmidt S, Vidal-Puig A, Diano S, Andrews P, Brand MD, Friedman J. Leptin-mediated changes in hepatic mitochondrial metabolism, structure, and protein levels. Proc Natl Acad Sci USA 106: 13100–13105, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Sleeman P, Patel NN, Lin H, Walkden GJ, Ray P, Welsh GI, Satchell SC, Murphy GJ. High fat feeding promotes obesity and renal inflammation and protects against post cardiopulmonary bypass acute kidney injury in swine. Crit Care 17: R262, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Song M, Chen Y, Gong G, Murphy E, Rabinovitch PS, Dorn GW 2nd. Super-suppression of mitochondrial reactive oxygen species signaling impairs compensatory autophagy in primary mitophagic cardiomyopathy. Circ Res 115: 348–353, 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Srere PA. [1] Citrate synthase: [EC 4.1.3.7. Citrate oxaloacetate-lyase (CoA-acetylating)]. In: Methods in Enzymology, edited by John ML. New York: Academic, 1969, p. 3–11. [Google Scholar]
- 84.Stern JS, Gades MD, Wheeldon CM, Borchers AT. Calorie restriction in obesity: prevention of kidney disease in rodents. J Nutr 131: 913S–917S, 2001. [DOI] [PubMed] [Google Scholar]
- 85.Stevenson FT, Wheeldon CM, Gades MD, van Goor H, Stern JS. Hyperphagia as a mediator of renal disease initiation in obese Zucker rats. Obes Res 9: 492–499, 2001. [DOI] [PubMed] [Google Scholar]
- 86.Sverdlov AL, Elezaby A, Behring JB, Bachschmid MM, Luptak I, Tu VH, Siwik DA, Miller EJ, Liesa M, Shirihai OS, Pimentel DR, Cohen RA, Colucci WS. High fat, high sucrose diet causes cardiac mitochondrial dysfunction due in part to oxidative post-translational modification of mitochondrial complex II. J Mol Cell Cardiol 78: 165–173, 2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Tallam LS, Stec DE, Willis MA, da Silva AA, Hall JE. Melanocortin-4 receptor-deficient mice are not hypertensive or salt-sensitive despite obesity, hyperinsulinemia, and hyperleptinemia. Hypertension 46: 326–332, 2005. [DOI] [PubMed] [Google Scholar]
- 88.Tao YX. The melanocortin-4 receptor: physiology, pharmacology, pathophysiology. Endocr Rev 31: 506–543, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Varanasi L, Hosler J. Alternative initial proton acceptors for the D pathway of Rhodobacter sphaeroides cytochrome c oxidase. Biochemistry 50: 2820–2828, 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Wang C, Wu M, Arvapalli R, Dai X, Mahmood M, Driscoll H, Rice KM, Blough E. Acetaminophen attenuates obesity-related renal injury through ER-mediated stress mechanisms. Cell Physiol Biochem 33: 1139–1148, 2014. [DOI] [PubMed] [Google Scholar]
- 91.Wang Y, Chen X, Song Y, Caballero B, Cheskin LJ. Association between obesity and kidney disease: A systematic review and meta-analysis. Kidney Int 73: 19–33, 2007. [DOI] [PubMed] [Google Scholar]
- 92.Wang YL, Shu KH, Yang MF, Yang WC, Wu MJ, Lin TM, Chen CH. The impact of body weight management in chronic kidney disease patients with obesity. J Ren Nutr 23: 372–379, 2013. [DOI] [PubMed] [Google Scholar]
- 93.Wein S, Ukropec J, Gasperikova D, Klimes I, Sebokova E. Concerted action of leptin in regulation of fatty acid oxidation in skeletal muscle and liver. Exp Clin Endocrinol Diabetes 115: 244–251, 2007. [DOI] [PubMed] [Google Scholar]
- 94.Wiegman CH, Bandsma RH, Ouwens M, van der Sluijs FH, Havinga R, Boer T, Reijngoud DJ, Romijn JA, Kuipers F. Hepatic VLDL production in ob/ob mice is not stimulated by massive de novo lipogenesis but is less sensitive to the suppressive effects of insulin. Diabetes 52: 1081–1089, 2003. [DOI] [PubMed] [Google Scholar]
- 95.Yang S, Zhu H, Li Y, Lin H, Gabrielson K, Trush MA, Diehl AM. Mitochondrial adaptations to obesity-related oxidant stress. Arch Biochem Biophys 378: 259–268, 2000. [DOI] [PubMed] [Google Scholar]
- 96.Ye Q, Danzer CF, Fuchs A, Vats D, Wolfrum C, Rudin M. Longitudinal evaluation of hepatic lipid deposition and composition in ob/ob and ob/+ control mice. NMR Biomed 26: 1079–1088, 2013. [DOI] [PubMed] [Google Scholar]
- 97.Zhang Y, Proenca R, Maffei M, Barone M, Leopold L, Friedman JM. Positional cloning of the mouse obese gene and its human homologue. Nature 372: 425–432, 1994. [DOI] [PubMed] [Google Scholar]
- 98.Zhao Q, Wang J, Levichkin IV, Stasinopoulos S, Ryan MT, Hoogenraad NJ. A mitochondrial specific stress response in mammalian cells. EMBO J 21: 4411–4419, 2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Zhou YT, Shimabukuro M, Koyama K, Lee Y, Wang MY, Trieu F, Newgard CB, Unger RH. Induction by leptin of uncoupling protein-2 and enzymes of fatty acid oxidation. Proc Natl Acad Sci USA 94: 6386–6390, 1997. [DOI] [PMC free article] [PubMed] [Google Scholar]