Abstract
FDA-approved high-dose interleukin-2 (IL-2) therapy and dendritic cell (DC) immunization offer time-tested treatments, but with defined issues of short in vivo half lives, toxicity, and modest clinical benefit. Complexes of IL-2 with specific monoclonal antibodies (IL-2c) exhibit improved stability in vivo with reduced toxicity and are capable of stimulating NK cell and memory phenotype CD8 T cell proliferation. Here, we demonstrate that IL-2c treatment in tumor bearing mice can enhance NK cell and tumor-specific CD8 T cell numbers. Importantly, DC immunization coupled with stabilized IL-2c infusion drastically improves the tumor-specific effector CD8 T cell response. DC+IL-2c treatment enhances number, 41BB and GITR expression, granzyme B production, cytotoxic T lymphocyte (CTL):TReg ratio, and per-cell killing capacity of CD8 T cells without increasing inhibitory molecule expression. Notably, IL-2c treatment of αCD3-stimulated human CD8 T cells resulted in higher number and granzyme B production, supporting the translational potential of this immunotherapy strategy for human malignancy. DC + IL-2c treatment enhances both endogenous NK cell and tumor antigen-specific CD8 T cell immunity to provide a marked reduction in tumor burden in multiple models of pre-existing malignancy in B6 and BALB/c mice. Depletion studies reveal contributions from both tumor-specific CD8 T cells and NK cells in control of tumor burden after DC + IL-2c treatment. Together, these data suggest that combination therapy with DC and IL-2c may be a potent treatment for malignancy.
Introduction
Chronic illnesses have increased dramatically over the last century (1), of which malignancy remains a top threat and target for many new vaccine candidates (1). Moving away from the broad-based chemotherapy of the past, current efforts focus on activating natural killer (NK) and cytotoxic T lymphocytes (CTL) for their ability to kill tumor cells directly (2, 3). Initially, the non-specific immunomodulator, interleukin-2 (IL-2) was used to enhance NK and T cell-mediated immunity to tumors (4, 5), at the expense of severe toxicity to the patient. More recently, well-tolerated dendritic cell (DC) therapy has been evaluated as a way to induce tumor antigen (TA)-specific CD8 T cells (6), but with modest potency, likely due to the relatively low CD8 T cell responses observed (7). Combinations of these two existing therapies are currently being tested to further increase CD8 T cell numbers (8), but without modifications to limit the toxicity or short in vivo half-life of IL-2 that requires long duration of therapy in specialized treatment centers.
In recent years, a more precise understanding of the success and limitations of high-dose (HD) IL-2 therapy, approved for renal cell carcinoma and metastatic melanoma (9, 10), have been highlighted. HD IL-2 therapy offers greater longevity for 16% of the patient population, at the risk of 2% mortality from treatment toxicity (11). The low efficacy of HD IL-2 in patients has been suggested to stem from poor induction of NK cell proliferation (12) and the stimulation of suppressive regulatory T (TReg) cells (13). Several investigators have since demonstrated in murine models that complexing free IL-2 with the IL-2-specific monoclonal Ab, S4B6, greatly decreases signaling to CD4+CD25+ TReg cells as well as CD25+ endothelial cells (14). The S4B6 mAb serves to redirect the bioactivity of IL-2 to CD122hi cells by competitively binding to its CD25 binding region. This unique quality decreases vascular leak syndrome (VLS), a serious side effect commonly associated with HD IL-2 therapy (14). Complexing to the IL-2-specific mAb S4B6 (IL-2c) (15) also increases its in vivo half-life since IL-2c is too large to excrete from the kidneys (15–17); this results in the in vivo proliferation of NK cells and memory-phenotype CD8 T cells (15). Additional studies, suggest that IL-2c can influence the differentiation of effector CD8 T cells responding to soluble peptide immunization (18, 19). To overcome issues with HD IL-2 associated toxicity and low CD8 T cell responses after DC vaccination, we evaluated a short immunization approach coupling DC immunization to stabilized IL-2c infusion to amplify numbers and increase function of both NK cells and endogenous TA-specific effector CD8 T cells.
Materials and Methods
Mice, Peptides, and Dendritic Cells
C57BL/6 (B6) mice were from the National Cancer Institute (Frederick, MD, USA). BALB/c mice were from Jackson Laboratories (Bar Harbor, ME, USA). Mice with TCR tg OT-I cells and SMARTA cells have been described (20, 21). The University of Iowa Animal Care and Use Committee approved animal experiments. Class I peptides used for DC pulses were Ova257-264 (SIINFEKL), AH16-14 (SPSYVYHQF), and TRP2180-188 (SVYDFFVWL) peptide at a concentration of 2μM. Class II peptides used were Ova323-339 (ISQAVHAAHAEINEAGR), Respiratory Syncitial Virus protein M226-39 (NYFEWPPHALLVRQ), and LCMV protein gp61-80 (GLKGPDIYKGVYQFKSVEFD) at the same concentration. LPS-matured peptide-coated DCs were prepared as described (22) and injected i.v. (5 × 105).
Adoptive Transfer and IL-2 Complexes
Approximately 3x104 naïve Thy1.1 OT-I CD8 T cells or 2x104 naïve Thy1.1 SMARTA CD4 T cells were transferred into naive Thy1.2+ B6 mice i.v. (23) at day −1. 5 × 105 LPS-matured/peptide-coated DCs were injected iv at day 0, followed by 1.5μg rat Ig or murine IL-2/anti-IL-2 S4B6 complexes (IL-2c) at days 4–6. Complexes were made by incubating murine IL-2 (PeproTech) with S4B6 anti–IL-2 mAb at a 2:1 molar ratio (7.5 μg/mL IL-2: 250 μg/mL S4B6) for 15 min at 37 degrees.
In Vitro Cytotoxicity Assay
Splenocytes from naïve B6 hosts were labeled with 0.08uM carboxyfluoroscein succinimidyl ester (CFSE) (no peptide) or 1uM CFSE (1μM Ova257-264). Targets (5x104 each) were mixed 1:1 ratio of CFSElo:CFSEhi with titrated numbers of effector OT-I CD8 T cell numbers from the spleens of rat Ig or IL-2c-treated mice. Killing was measured at E:T ratios of 0.3, 1,over 4 hours at 37 degrees. Percent specific lysis was calculated as 100−(100x[(% CFSElo in control well/% CFSEhi in control well)/(% CFSElo in experimental well/% CFSEhi in experimental well)].
Cell Lines and Animal Imaging
B16, B16-fLUC, 4T1-fLUC were provided by Lyse Norian (University of Iowa). B6 mice were injected s.c. in the left hind flank with 2×104 B16-fLUC cells in a 1:1 volume of PBS and matrigel (BD; cat: 356234) or 5x104 B16 cells i.v. 1x105 4T1-fLUC cells were orthotopically inoculated in mammary pad #9 of female BALB/c mice.. Bioluminescence imaging was performed on anesthetized mice on an IVIS Spectrum (Caliper) system approximately 10 minutes after i.p. injection of 1mg luciferin (Gold Biotech; St. Louis, MO). Imaging on IVIS was performed according to manufacturer’s protocols and bioluminescence was quantified in radiance.
Leukocyte Reduction System Cones and Human CD8 T cell Assays
Human CD8+ T cells were purified with the RosetteSep CD8+ T cell enrichment cocktail (Stemcell Technologies). 2x105 CD8+CD3+ T cells were plated to 96-well plates coated with 0.1 μg/mL OKT3/anti-CD28 at day 0. On day 4, additional RP10 1640 media or huIL-2 (10ng/mL; Peprotech)/MAB602 (RnD Systems) complexes were added to each well. huIL-2/MAB602 complexes were prepared by incubating huIL-2 (10ng/mL) with MAB602 (100ng/mL) for 15 minutes at 37 degrees. Ki67 positivity, total numbers of CD3+CD8+ T cells, and granzyme B expression were determined by flow cytometry on day 7.
Antibodies and Flow Cytometry
Antibodies were used with the indicated specificity and the appropriate combinations of fluorochromes. Intracellular stains were performed following manufacturer’s protocols. FoxP3/transcription factor staining buffer kit (eBioscience, cat: 00-5523-00) was purchased for transcription factor labeling. The following antibodies were purchased from BioLegend: Thy1.1 (OX-7), CD8+ (53–6.7), 41BB (17B5), PD-1 (29F.1A12), LAG3 (C9B7W). The following antibodies were purchased from eBioscience: GITR (DTA-1) and CTLA-4 (UC10-4B9). Anti-IFN-g was purchased from BD. Anti-human granzyme B and mIgG1 Abs were purchased from Invitrogen. PK136 was purchased from eBiosciences; 2.43 antibody was purchased from Santa Cruz Biotech. Anti–IL-2 mAb were purified from S4B6 hybridomas and were purchased from American Type Culture Collection.
In Vivo Tumor Protection
Intravenous and subcutaneous B16 tumor studies were performed using B16 and B16-fLUC cells at indicated concentrations. Lungs were perfused with PBS and harvested from mice at indicated day post-inoculation. Visible tumor nodules were quantified from IL-2c and Rat Ig control groups. Tumor area for B16-fLUC and 4T1-fLUC (1x105 cells/mouse) studies were determined by 2 digital caliper measurements taken on anesthetized mice.
Statistical Analysis
Unless indicated otherwise, significance of bar graphs were calculated by student’s T test using Graphpad Prism 5 for Macintosh. P-values<0.05 were considered significant. Survival curves were analyzed by the Mantel-Cox test.
Results
Tumor control, NK/NKT cell and CD8 T cell activation by IL-2c treatment
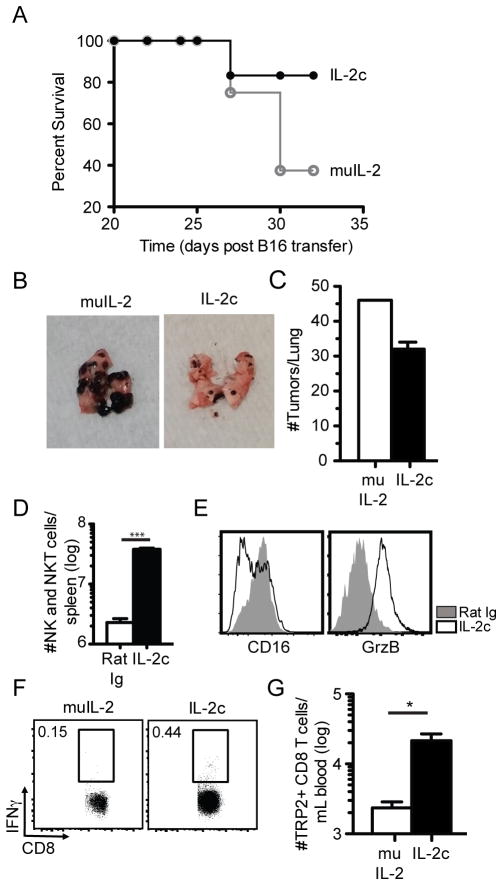
Prior studies reveal that treatment of mice with IL-2c can enhance tumor control over soluble IL-2, potentially through activation of NK and NK T cells expressing the low affinity IL-2R (15, 24). To determine the relative potency of IL-2 vs IL-2c treatment in tumor control, we used a lung metastasis model, where B6 mice were injected i.v. with B16 melanoma, and daily IL-2c or soluble IL-2 therapy was initiated 3 days later. IL-2c treatment enhanced survival of tumor bearing mice (Fig. 1A) and reduced tumor size (Fig. 1B) although no significant decreases in lung tumor numbers were observed (Fig. 1C). As previously described (15, 24), treatment with IL-2c enhanced NK and NKT cell numbers as well as activation state (CD16 and granzyme B expression (data not shown). Thus, IL-2c treatment is superior to soluble IL-2 and can enhance NK cell activation and limit tumor burden to prolong survival of mice with B16 lung metastasis.
Fig. 1. Tumor control, NK/NKT cell and CD8 T cell activation by IL-2c treatment.
Naïve B6 mice were inoculated with 5x104 B16 cells i.v. and injected with 3 doses of soluble IL-2 or IL-2c 3 days post-inoculation. (A) Survival curve of mice from both groups (N=5 mice/group). (B) Raw images of perfused lungs on day 32 post-inoculation. (C) Summary bar graphs (mean ± SEM) of total number of tumor nodules/lung (right). (D) Naïve B6 mice were injected with 3 doses of Rat Ig or IL-2c alone. NK1.1+ cells from spleen after 3 doses of IL-2c. (E) Histograms of surface phenotype on NK1.1+ cells. (F) Representative flow plots of TRP2-specific CD8 T cells in the blood via ICS. (G) Summary bar graph of TRP2-specific CD8 T cells. Data is representative from 2 independent experiments with at least 3 mice per group per experiment. * = p<0.05. *** = p<0.0005.
The Tyrosinase-Related-Protein 2 (TRP2) is a defined endogenous tumor antigen for melanoma in both humans and mice. The TRP2 epitope is presented by either H-2Kb (mouse) or HLA-A201 (human) (25, 26). In addition, TRP2-specific CD8 T cells were capable of limiting B16 melanoma in various vaccination regimens or immunotherapy (27). Of note, analyses of TRP2-specific CD8 T cells in the blood of tumor bearing mice at day 3 after completion of IL-2 therapy revealed ~10-fold higher numbers in mice receiving IL-2c treatment compared to soluble IL-2 treatment (Fig. 1D, E). To date, no preclinical studies of IL-2c have offered comprehensive data on its potential to target and expand non-mutated self-peptide tumor antigens, a trait essential to an effective tumor therapy (19, 24, 28). Although the TRP2-specific CD8 T cell response was modest in both groups, these data suggest the potential of IL-2c to amplify endogenous TA-specific CD8 T cell responses that, in addition to NK cells, could contribute to IL-2c enhanced control of B16 metastases.
IL-2c treatment massively enhances dendritic cell primed CD8 T cell responses
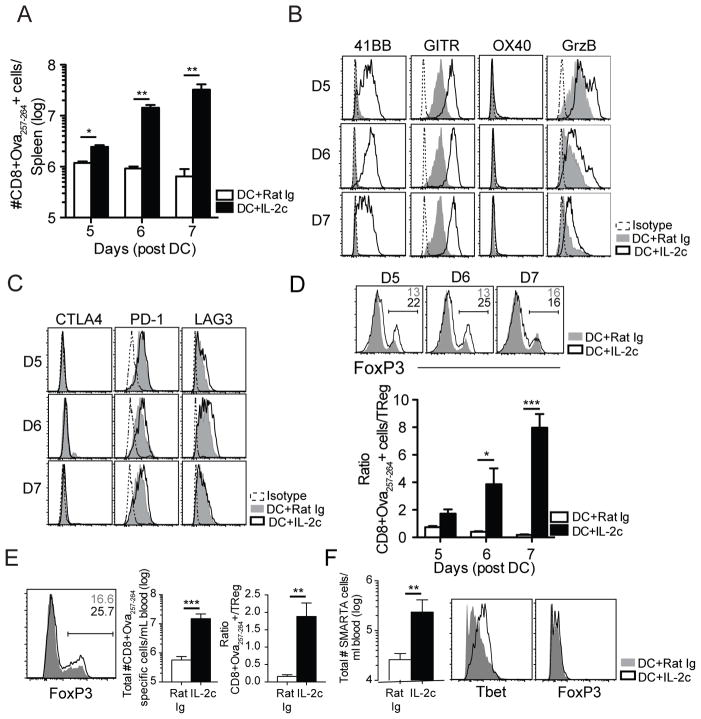
To initially determine if IL-2c treatment could enhance the CD8 T cell response to a relevant antigen-specific vaccination, we combined a DC immunization with Ova257-264 peptide and IL-2c treatment. IL-2c has been shown to stimulate proliferation in memory phenotype CD8 T cells and NK cells expressing the low affinity IL-2R (CD122, CD132) (29). Since expression of the high affinity IL-2R (CD25, CD122, CD132) on newly activated CD8 T cells is largely extinguished by 4 days post DC immunization (30), Rat Ig control or IL-2c were administered on D4-6 post DC priming. Strikingly, IL-2c treatment markedly enhanced the number of DC-primed endogenous Ova257-264-specific CD8 T cells by greater than 50-fold at day 7 after immunization (Fig. 2A). Previously, Pipkin et al. demonstrated that IL-2 signaling induced a transcriptional program promoting effector differentiation and cytolytic potential of activated CD8 T cells (18). Thus, we monitored expression of inducible costimulatory molecules that have previously been shown to enhance differentiation of effector T cells (31–33). IL-2c treatment upregulated 41BB and GITR expression on Ova257-264-specific CD8 T cells, but failed to upregulate OX40 (Fig. 2B). These results demonstrate the capacity of IL-2c to massively enhance accumulation of DC-primed effector CD8 T cells and selectively upregulate costimulatory molecules.
Fig. 2. IL-2c treatment massively enhances DC-primed CD8 T cell responses.
Naïve B6 mice were immunized i.v. with 5x105 DCs followed by IL-2c i.p. from D4–6 post DC-immunization (A) Total numbers of Ova257-264-specific CD8 T cells in the spleen of Rat Ig or IL-2c groups from D5–7. (B) Representative histograms of 41BB, GITR, OX40 and Granzyme B expression on Ova257-264-specific CD8 T cells from Rat Ig or IL-2c-treated cells. (C) Representative histograms of CTLA-4, PD-1, and LAG3 expression on Ova257-264-specific CD8 T cells from Rat Ig or IL-2c treated mice from D5–7. (D) Representative histograms of FoxP3+ CD4 T cells after Rat Ig or IL-2c (top). Ratio of total number of Ova257-264-specific CD8 T cells over total CD4+FoxP3+ cell numbers in the spleen (bottom). (E) Same as (D) except DCs were pulsed with Ova257-264, Ova323-339, gp61-80, and M226-39. Representative histograms of FoxP3+ CD4 T cells (left), summary bar graph of total Ova257 CD8 T cell response (center), and ratio of Ova257-264-specific CD8 T cells:TRegs in spleen (right). (F) 2x104 SMARTA cells were adoptively transferred into naïve B6 recipients and administered DC-gp61±IL-2c. SMARTA cells were quantified in the spleen at D7 post-DC immunization (left). Representative histograms of Tbet and FoxP3 positive SMARTAs in Rat Ig compared to IL-2c treated mice. *** = p<0.0005. Results shown are representative of 3 independent experiments with n=3/group/day for each repeat.
Similar to NK cells, Ova257-264-specific CD8 T cells responding to IL-2c also expressed more granzyme B (Fig. 2B), but did not further upregulate expression of the inhibitory receptors CTLA4, PD-1 or LAG3 compared to DC primed CD8 T cells in the absence of IL-2c treatment (Fig. 2C). To address additional functions important in tumor control, we utilized an adoptive transfer model of OT-I T cell receptor transgenic (tg) cells. This TCR tg approach with DC priming allowed us to measure CD8 T cell number and function, independent of changes in TCR repertoire in the translational context of DC immunization. Similar to the endogenous CD8 T cell response, IL-2c treatment amplified DC-Ova257-264 primed OT-I CD8 T cell numbers (Fig. S1A), 41BB, GITR and granzyme B expression (Fig. S1B), and did not alter CTLA4, PD-1 or LAG3 expression (Fig. S1C). As many human tumor-associated Ags are derived from non-mutated self-proteins expressed at low levels, antigen sensitivity (also termed functional avidity) is an important consideration when developing T cell-mediated tumor immunotherapies (34). Indeed, high T cell avidity was shown to correlate with T cell recognition of tumors and their therapeutic efficacy (35). IL-2c treatment enhanced antigen sensitivity of monoclonal OT-I cells for IFN-γ production by approximately 2-fold (Fig. S1D), indicated by a decreased effective concentration (EC50) (Fig. S1E). Consistent with the sustained expression of granzyme B and enhanced sensitivity to antigen, OT-I cells generated by DC-prime and IL-2c treatment exhibited enhanced in vitro cytolytic activity (Fig S1F, G). These data demonstrate an increase in both Ag sensitivity for cytokine production and per-cell killing capacity by CD8 T cells following DC+IL-2c treatment without increasing inhibitory receptor expression.
HD IL-2 therapy has been shown to induce TRegs that contribute to poor disease outcomes (13, 36). IL-2c treatment did result in a modest increase in the frequency of FoxP3 expressing TReg cells (Fig. 2D, top); however, it has been suggested that in the context of immunotherapy, the ratio of effector CD8 T cells: TReg may be the critical parameter determining the success of treatment (37). Our results show that amplification of the Ova257-264-specific CD8 T cell response by IL-2c was much greater than the increase in TReg cells, and the ratio of effector Ova257-264-specific CD8 T cells:TReg therefore increased substantially during the treatment (Fig. 2D, bottom). To examine whether a complex mixture of class I and II peptide epitopes on DCs might induce a greater TReg response, we peptide-pulsed DCs with Ova257-264 and class II peptide epitopes Ova323-339, gp61-80, and M226-39. We subsequently treated with IL-2c D4–6 post-DC immunization and quantified CD4+FoxP3+ T cells, which yielded a similar TReg induction and enhanced CTL/TReg ratio as seen in IL-2c treated mice that received DC pulsed with Ova257-264 alone (Fig. 2E). Furthermore, we tested whether DC+IL-2c would directly polarize naive CD4 T cells to an effector T helper 1 (Th1) or suppressive TReg phenotype. We adoptively transferred 2x104 transgenic SMARTA CD4 T cells into naïve B6 recipients that were subsequently treated with DC-gp61-80+IL-2c from D4–6. Activating CD4 T cells directly using DC+IL-2c enhanced the quantity of CD4 T cells by 10-fold and polarized them to a Th1 phenotype, without inducing a discernable TReg population (Fig. 2F). These data demonstrate that IL-2c treatment in the context of DC-immunization results in preferential expansion of effector CD8 T cells over TReg cells, and can also be used to enhance Th1 CD4 T cells against a defined epitope.
Direct translation of this approach to humans would require generation of IL-2c with human IL-2 and mAb. To address this possibility, we generated IL-2c with human IL-2 and the MAB602 anti-human IL-2-specific antibody, complexes of which have been shown to stimulate proliferation of memory-phenotype CD8 T cells in mice (16). IL-2c MAB602 treatment enhanced Ki67 expression, as a measure of proliferation (Fig. S2A, B), cell recovery (Fig. S2C) and granzyme B expression (Fig. S2D) of anti-CD3/CD28 stimulated human CD8 T cells. Thus, IL-2c treatment is a potentially viable means to amplify human CD8 T cells responding to antigen-stimulation.
IL-2c treatment enhances CD8 T cell responses to endogenous tumor antigens and control of lung metastasis
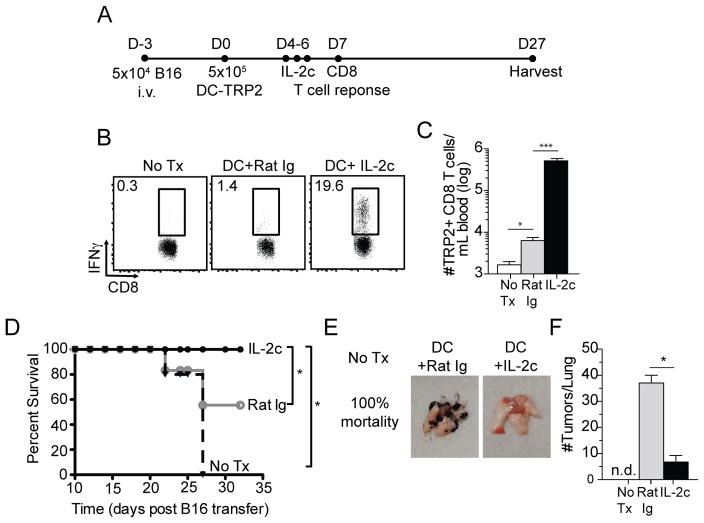
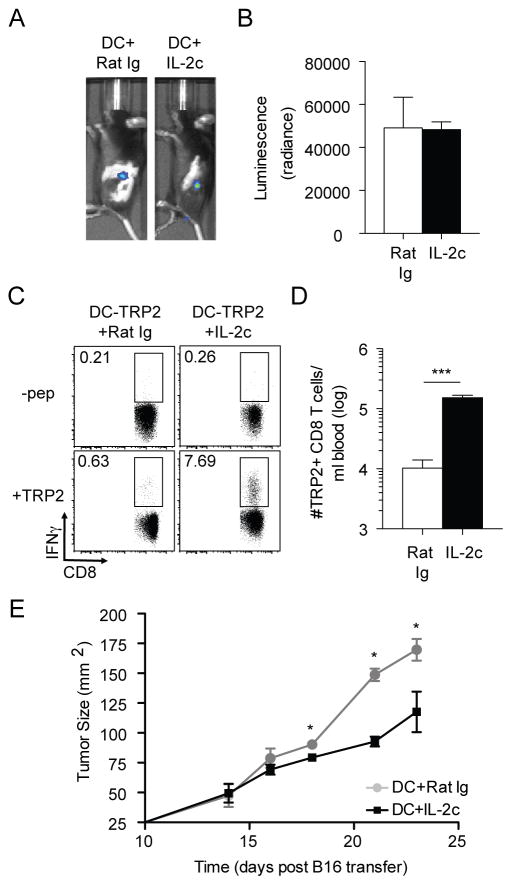
The prior results (Fig. 2) were obtained with a foreign antigen in non-tumor bearing mice. To address the potency of IL-2c to amplify a DC-primed TA-specific CD8 T cell response in tumor bearing mice, B16 cells were injected i.v. and DC-TRP2 was initiated 3 days later followed by IL-2c treatment of some mice at days 4–6 post DC immunization (Fig. 3A). IL-2c dramatically enhanced the frequency (Fig. 3B) and total numbers (Fig. 3C) of TRP2-specific CD8 T cells over DC-TRP2 priming alone or in mice with no treatment. Of note, all mice that received DC + IL-2c survived the 32-day study period whereas 100% of mice receiving no therapy and 50% of mice receiving DC-TRP2 without IL-2c succumbed to tumor (Fig. 3D). Mice that received DC-TRP2 + IL-2c exhibited reduced tumor size (Fig. 3E) and substantially reduced tumor numbers at day 30 compared to surviving mice that received DC-TRP2 without IL-2c (Fig. 3F). The reduction of tumor numbers was a specific improvement of the DC + IL-2c treatment over IL-2c treatment alone (Fig. 1). These data suggest that IL-2c treatment, in conjunction with DC-TRP2 priming, can enhance control of pre-existing lung metastasis.
Fig. 3. Therapeutic vaccine strategy significantly improves CD8 T cell-mediated protection to established tumors.
Naïve B6 mice were inoculated with 5x104 B16 tumors i.v. and treated with PBS, DC+Rat Ig, or DC+IL-2c on D3 post-inoculation. (A) Experimental approach. (B) Endogenous TRP2-specific CD8 T cells were quantified per ml of blood at D7 post-DC via ICS. (C) Summary bar graph (mean ± SEM). (D) Survival curves for all treatment groups. (E) Representative images of perfused lungs at D30 post-transfer of B16 melanoma cells. (F) Summary bar graphs (mean ± SEM) of total number of tumors per lung for all treatment groups. Data are representative of 2 independent experiments with at least four mice per treatment group. *** = p<0.0005; n.d., no data.
Importantly, late intervention with DC + IL-2c was capable of inducing an equally potent TA-specific CD8 T cell response. To ensure a high tumor burden, 2x105 B16 cells were injected i.v. in naïve B6 mice, followed by DC-TRP2 + IL-2c 21 days later (Fig. S3A). TRP2-specific CD8 T cells were measured in the blood at d7 post-DC immunization with a 30-fold increase in frequency (Fig. S3B) and 40-fold increase in total numbers (Fig. S3C). DC + IL-2c treatment initiated as late as 21 days after tumor injection was able to substantially reduce the number of tumor nodules in the lung (Fig. S3D, E). These data suggest that DC+IL-2c treatment has the potential to effectively amplify immune responses and control tumor load in advanced cancer patients.
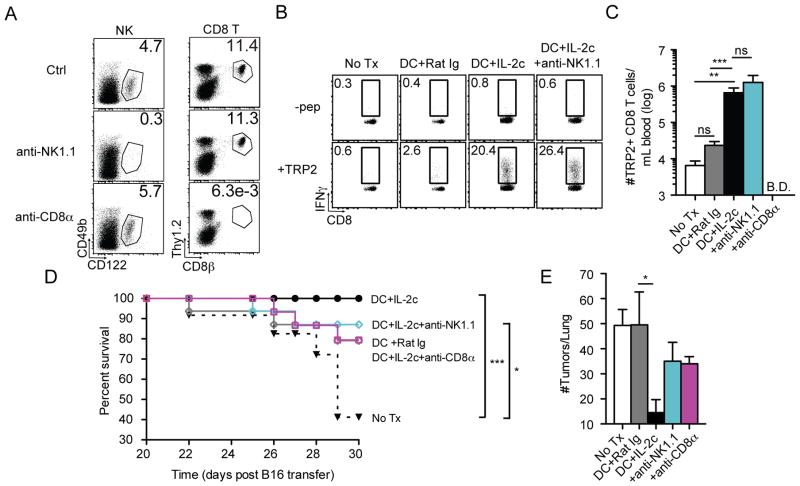
To address the relative contribution of CD8 T cells and NK cells in tumor control, mice were injected i.v. with B16 melanoma, followed by CD8 or NK cell specific-depleting antibodies (Fig. 4A). One week after B16 injection, some mice were immunized with DC-TRP2 and some mice in each group received IL-2c treatment at day 4–6 post-DC immunization. The presence or absence of NK cells had no impact on the number of TRP2-specific CD8 T cells, whereas CD8 depleted mice contained no detectable TRP2-specific CD8 T cells (Fig. 4B, C and data not shown). Again, all mice that received DC-TRP2 + IL-2c treatment survived the experimental period whereas 60% of untreated mice succumbed (Fig. 4D). Some mice in DC-TRP2 + IL-2c groups depleted of either NK cells or CD8 T cells also succumbed during the experimental period (Fig. 4D), suggesting that both effector populations contributed to enhanced survival. Depletion of CD8 T cells or NK cells in DC-TRP2 + IL-2c treated mice resulted in higher numbers of tumors in the surviving mice compared to mice containing both cell populations (Fig. 4E). Thus, DC-TRP2 + IL-2c treatment enhances survival and control of pre-existing lung metastases through a combination of amplified tumor-specific CD8 T cells and activated NK cells.
Fig. 4. Depletion of either NK cells or CD8 T cells reduces protection.
Naïve B6 mice were inoculated with 5x104 B16 tumors i.v. and treated with PBS, DC+Rat Ig, or DC+IL-2c±PK136 or 2.43. (A) Confirmation of NK and CD8 T cell depletion at D4 post-DC immunization. (B) Raw flow plots of TRP2+ CD8 T cells in each treatment group. (C) Summary bar graph (mean ± SEM). (D) Survival curves for all treatment groups. (E) Summary bar graph of number of tumors/lung in each treatment group at D30 post-B16 inoculation. Data are representative of 2 independent experiments with at least four mice per treatment group. * = p<0.05; ** = p<0.005; *** = p<0.0005 n.s., not significant.
DC-prime + IL-2c treatment in control of solid tumors
Melanoma initiates as a localized primary tumor in the skin. To address the potential that DC-TRP2 + IL-2c treatment could control a solid tumor, B16 melanoma cells expressing firefly luciferase were injected subcutaneously into the flank of B6 mice and tumor size was evaluated by bioluminescence imaging. Mice were randomized into groups with similar sized tumors (Fig. 5A, B) at day 7 after tumor inoculation and subsequently immunized with DC-TRP2 + IL-2c treatment. IL-2c treatment amplified DC-primed TRP2-specific CD8 T cells in solid tumor bearing mice (Fig. 5C, D) and substantially slowed the rate of tumor growth (Fig. 5E) compared to mice given DC-TRP2 alone. Thus, DC-TRP2 immunization can be enhanced by IL-2c treatment for control of a solid tumor.
Fig. 5. DC-TRP2+IL-2c treatment reduces solid tumor burden.
Naïve B6 mice were inoculated with 5x104 B16 tumors s.c. and treated with DC±IL-2c 7 days later. (A) Raw whole-mouse IVIS images at D7 post-B16 inoculation. (B) Same a (A), but bioluminescence measurement of B16-fLUC tumor take in recipient mice before treatment intervention. (C) Raw flow plots of endogenous TRP2-specific CD8 T cells per ml of blood at D7 post-treatment via ICS. (D) Summary bar graphs (mean ± SEM) of total number of TRP2+ CD8 T cells per ml of blood. (E) Growth kinetics curves quantified in mm2. Data are representative of 2 independent experiments with at least five mice per treatment group.* = p<0.05; n.s., not significant.
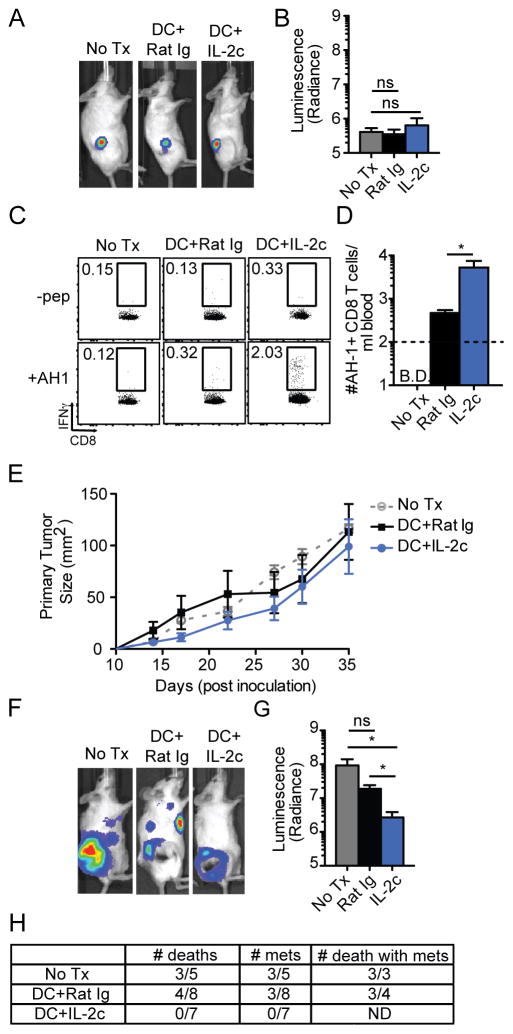
The results so far evaluated the impact of IL-2c treatment on DC-primed CD8 T cells and tumor control in B6 mice. The 4T1 breast cancer cell line establishes a solid tumor after orthotopic transplant in BALB/c mice and spontaneously metastasizes in a manner similar to human breast cancer, leading to host mortality (38, 39). The AH1 peptide, derived from the endogenous retroviral gp100 antigen serves as a tumor specific antigen for the 4T1 tumor (40). To extend our analyses to an additional mouse strain, epitope and tumor model, we inoculated female BALB/c mice with 4T1 tumor cells expressing firefly luciferase (4T1-fLUC) and monitored tumor growth by bioluminescence. Mice were randomized into three groups with equivalent tumor burdens (Fig. 6A, B) at day 7 post-tumor inoculation. Some mice were then immunized with DC-AH1 ± IL-2c. IL-2c treatment enhanced the AH1-specific CD8 T cell response (Fig. 6C, D) as observed previously for response to Ova257-264 or TRP2 peptide. In contrast to the prior study with B16 melanoma, DC or DC + IL-2c treatment had little impact on the growth rate of the primary 4T1-fLUC tumor (Fig. 6E). However, DC + IL-2c treatment, prevented the development of metastases visible by whole mouse bioluminescence imaging and thus, reduced the overall live tumor burden (Fig. 6F, G). Furthermore, none of the mice in the DC + IL-2c group succumbed during the study period, whereas 60% of control mice and 50% of DC-AH1 alone mice succumbed (Fig. 6H). Metastases were readily apparent in untreated mice or mice given DC-AH1 without IL-2c. Importantly, 100% of mice in the untreated control group and 75% in the DC-AH1 group that succumbed to the 4T1 tumor exhibited detectable metastases (Fig. 6H). Strikingly, there was no visible metastasis in the DC-AH1 group that received IL-2c, consistent with the lack of mortality from this group (Fig. 6H). Of note, IL-2c alone did not prevent metastases in the experimental time frame (Fig S4A, B) Thus, DC-AH1+IL-2c treatment had specific efficacy against metastases in the 4T1 model even with minimal control of the primary tumor site.
Fig. 6. DC-prime + IL-2c treatment in control of solid tumors.
Naïve BALB/c mice were orthotopically inoculated with 1x105 4T1-fLUC cells in mammary pad no. 9 and treated with PBS, DC+Rat Ig, or DC+IL-2c on day 7 post-inoculation. (A) Representative whole-mouse IVIS images of recipient mice in each treatment group. (B) Bioluminescence measurement of 4T1-fLUC take in recipient mice before treatment intervention. (C) Raw flow plots of endogenous AH1-specific CD8 T cells per ml of blood at D7 post-DC immunization via ICS. (D) Summary bar graphs (mean ± SEM) of total number of AH1+ CD8 T cells per ml of blood. (E) Tumor growth kinetics quantified in mm2. (F) Whole-mouse bioluminescence measurement at day 35 post-4T1 inoculation. (E) Table of numbers of deaths, metastases, and death with metastases. Data are representative of 2 independent experiments with at least five mice per treatment group. * = p<0.05
Discussion
The immune system is naturally equipped with the means to eliminate tumor cells; however, unmanipulated endogenous responses appear insufficient for full tumor control. To this end, multiple approaches have been tested as immunotherapies against cancer. To be effective, immunotherapies would ideally activate highly functional cytotoxic NK and TA-specific CD8 T cells with the capability to eradicate tumor cells without concurrent activation of regulatory cells that could dampen immunity. Although currently approved HD IL-2 infusion can modestly increase CD8 T cell numbers, induction of TReg cells and high toxicity following therapy, remain important limitations of this treatment (13, 41). DC-based immunotherapy can additionally stimulate TA-specific CD8 T cells, but the relatively low magnitude T cell responses after DC-immunization may underlie the modest impact on clinical outcomes in malignancy (7). Thus, single therapeutic modalities appear to lack potency and the field has turned to combination immunotherapies with the potential to enhance clinical outcome (32, 42). However, combination DC with soluble IL-2 treatment does not address the toxicity or long-duration of IL-2 therapy (8). Here, we evaluate a strategy combining IL-2 with a specific monoclonal antibody to generate relatively stable, non-toxic IL-2c that can be used to augment the ability of DC immunization to enhance the magnitude and function of TA-specific effector CD8 T cells, while also amplifying NK cell function and numbers.
CD25 expression is maintained for only 3 days following DC immunization alone (30) and then responding CD8 T cells express only the low affinity IL-2R (CD122, CD132)(43). Based on these data, we combined initial DC-immunization with IL-2c delivered from day 4–6 after DC-immunization to amplify TA-specific CD8 T cells, while minimizing suppressive mechanisms and toxicity. This rationale was based on literature indicating that a specific IL-2c (IL-2 complexed with the S4B6 mAb) signals primarily through the low affinity IL-2R and not the high affinity IL-2R expressed on some TReg and vascular endothelium (14, 16). Our results demonstrate the superior efficacy of DC-TA + IL-2c immunization strategies for tumor control over IL-2c or DC immunization alone. The combination of DC-TA-+ IL-2c infusion not only activates and expands NK cell, but also enhances the number, antigen sensitivity, cytotoxicity, and CTL/TReg ratio of effector TA-specific CD8 T cells without increasing inhibitory receptor expression that could blunt activation (44). These quantitative and qualitative improvements in innate and adaptive cytotoxic cell types translate to better control of tumor burden in multiple models of pre-existing malignancy. Notably, we show that DC+IL-2c enhances number and function of endogenous TA-specific CD8 T cells for significant reduction of tumor burden. Moreover, by enhancing non-mutated self peptide-specific CD8 T cell responses, DC+IL-2c treatment can be effectively used against tumors that do not undergo high rates of mutation and express neoantigens (45, 46); thus, providing a viable treatment option for cancer patients carrying tumors that traditionally exhibit the highest resistance to immunotherapies (47).
In many cases, primary tumors can be limited through surgical intervention (48). However, metastatic disease is more difficult to detect and prevent (49). Perhaps the most striking finding from our data is the enhanced capacity of the DC-TA + IL-2c combination therapy to deal with pre-existing metastases (B16 i.v. model) or prevent detectable metastases (4T1 model). Whether this results solely from enhanced numbers or specific functional attributes of DC-TA + IL-2c generated NK cells or TA-specific CD8 T cells remains a question of great interest. For example, TA-specific endogenous CD8 T cells likely emerge from a pre- immune repertoire that has been limited in number and affinity to avoid autoimmunity (50, 51). The decreased affinity for TA may be partially overcome by the enhanced antigen-sensitivity and increased cytolytic capacity observed in T cells stimulated by combination DC-TA + IL-2c treatment. Additionally, the enhanced expression of late co-stimulatory molecules (41BB and GITR) by IL-2c treatment has the potential to render responding TA-specific CD8 T cells and NK cells resistant to suppressive mechanisms within tumors (32). Furthermore, DC + IL-2c treatment dramatically enhanced TA-specific CD8 T cell:TReg ratios, a relationship that has been suggested to be critical for successful immunotherapy (37). Finally, it will be of great interest to determine if additional combination with checkpoint blockade will further increase the capacity of DC + IL-2c immunotherapy to combat malignancy. The model systems described here should be ideally suited to address these questions.
Our results establish the proof of principle for combining DC-TA immunization with IL-2c infusion for tumor control. Importantly, complexes of human IL-2 and anti-human mAb can also enhance the proliferation, accumulation and function of human CD8 T cells. As DC therapy is currently FDA-approved (7), and IL-2c has been shown in multiple preclinical studies to address serious pitfalls with currently approved HD IL-2 therapy (9), we consider this DC + IL-2c immunization strategy to be a viable candidate for FDA approval as an immunotherapy for cancer patients.
Supplementary Material
Acknowledgments
We thank members of the Harty lab for helpful discussion.
Footnotes
Conflict of Interest: The authors declare no conflict of interest.
Author Contributions: M.T.K. performed all mouse and human cell experiments and analyzed all data, M.J.R. assisted with functional avidity assay and reviewed manuscript, B.P.G. and L.A.N. provided tumor cell lines and expertise on tumor models. J.T.H., V.P.B. and M.T.K. designed overall study and drafted final manuscript.
References
- 1.Bachmann MF, Dyer MR. Therapeutic vaccination for chronic diseases: a new class of drugs in sight. Nature reviews Drug discovery. 2004;3:81–88. doi: 10.1038/nrd1284. [DOI] [PubMed] [Google Scholar]
- 2.Knutson KL, Disis ML. Tumor antigen-specific T helper cells in cancer immunity and immunotherapy. Cancer immunology, immunotherapy : CII. 2005;54:721–728. doi: 10.1007/s00262-004-0653-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Steer HJ, Lake RA, Nowak AK, Robinson BW. Harnessing the immune response to treat cancer. Oncogene. 2010;29:6301–6313. doi: 10.1038/onc.2010.437. [DOI] [PubMed] [Google Scholar]
- 4.Smith KA. Interleukin-2: inception, impact, and implications. Science. 1988;240:1169–1176. doi: 10.1126/science.3131876. [DOI] [PubMed] [Google Scholar]
- 5.Blattman JN, Grayson JM, Wherry EJ, Kaech SM, Smith KA, Ahmed R. Therapeutic use of IL-2 to enhance antiviral T-cell responses in vivo. Nature medicine. 2003;9:540–547. doi: 10.1038/nm866. [DOI] [PubMed] [Google Scholar]
- 6.Palucka K, Banchereau J. Cancer immunotherapy via dendritic cells. Nature reviews Cancer. 2012;12:265–277. doi: 10.1038/nrc3258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Janikashvili N, Larmonier N, Katsanis E. Personalized dendritic cell-based tumor immunotherapy. Immunotherapy. 2010;2:57–68. doi: 10.2217/imt.09.78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Baek S, Kim YM, Kim SB, Kim CS, Kwon SW, Kim Y, Kim H, Lee H. Therapeutic DC vaccination with IL-2 as a consolidation therapy for ovarian cancer patients: a phase I/II trial. Cellular & molecular immunology. 2015;12:87–95. doi: 10.1038/cmi.2014.40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Stone R. Anti-cancer drug IL-2 may finally be approved. Science. 1992;255:528. doi: 10.1126/science.1736353. [DOI] [PubMed] [Google Scholar]
- 10.McDermott DF, Atkins MB. Interleukin-2 therapy of metastatic renal cell carcinoma--predictors of response. Seminars in oncology. 2006;33:583–587. doi: 10.1053/j.seminoncol.2006.06.004. [DOI] [PubMed] [Google Scholar]
- 11.Petrella T, Quirt I, Verma S, Haynes AE, Charette M, Bak K. Single-agent interleukin-2 in the treatment of metastatic melanoma: a systematic review. Cancer treatment reviews. 2007;33:484–496. doi: 10.1016/j.ctrv.2007.04.003. [DOI] [PubMed] [Google Scholar]
- 12.Shook DR, Campana D. Natural killer cell engineering for cellular therapy of cancer. Tissue antigens. 2011;78:409–415. doi: 10.1111/j.1399-0039.2011.01796.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sim GC, Martin-Orozco N, Jin L, Yang Y, Wu S, Washington E, Sanders D, Lacey C, Wang Y, Vence L, Hwu P, Radvanyi L. IL-2 therapy promotes suppressive ICOS+ Treg expansion in melanoma patients. The Journal of clinical investigation. 2014;124:99–110. doi: 10.1172/JCI46266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Krieg C, Letourneau S, Pantaleo G, Boyman O. Improved IL-2 immunotherapy by selective stimulation of IL-2 receptors on lymphocytes and endothelial cells. Proceedings of the National Academy of Sciences of the United States of America. 2010;107:11906–11911. doi: 10.1073/pnas.1002569107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Boyman O, Kovar M, Rubinstein MP, Surh CD, Sprent J. Selective stimulation of T cell subsets with antibody-cytokine immune complexes. Science. 2006;311:1924–1927. doi: 10.1126/science.1122927. [DOI] [PubMed] [Google Scholar]
- 16.Letourneau S, van Leeuwen EM, Krieg C, Martin C, Pantaleo G, Sprent J, Surh CD, Boyman O. IL-2/anti-IL-2 antibody complexes show strong biological activity by avoiding interaction with IL-2 receptor alpha subunit CD25. Proceedings of the National Academy of Sciences of the United States of America. 2010;107:2171–2176. doi: 10.1073/pnas.0909384107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Phelan JD, Orekov T, Finkelman FD. Cutting edge: mechanism of enhancement of in vivo cytokine effects by anti-cytokine monoclonal antibodies. J Immunol. 2008;180:44–48. doi: 10.4049/jimmunol.180.1.44. [DOI] [PubMed] [Google Scholar]
- 18.Pipkin ME, Sacks JA, Cruz-Guilloty F, Lichtenheld MG, Bevan MJ, Rao A. Interleukin-2 and inflammation induce distinct transcriptional programs that promote the differentiation of effector cytolytic T cells. Immunity. 2010;32:79–90. doi: 10.1016/j.immuni.2009.11.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Mostbock S, Lutsiak ME, Milenic DE, Baidoo K, Schlom J, Sabzevari H. IL-2/anti-IL-2 antibody complex enhances vaccine-mediated antigen-specific CD8+ T cell responses and increases the ratio of effector/memory CD8+ T cells to regulatory T cells. J Immunol. 2008;180:5118–5129. doi: 10.4049/jimmunol.180.7.5118. [DOI] [PubMed] [Google Scholar]
- 20.Hogquist KA, Jameson SC, Heath WR, Howard JL, Bevan MJ, Carbone FR. T cell receptor antagonist peptides induce positive selection. Cell. 1994;76:17–27. doi: 10.1016/0092-8674(94)90169-4. [DOI] [PubMed] [Google Scholar]
- 21.Oxenius A, Bachmann MF, Zinkernagel RM, Hengartner H. Virus-specific MHC-class II-restricted TCR-transgenic mice: effects on humoral and cellular immune responses after viral infection. European journal of immunology. 1998;28:390–400. doi: 10.1002/(SICI)1521-4141(199801)28:01<390::AID-IMMU390>3.0.CO;2-O. [DOI] [PubMed] [Google Scholar]
- 22.Schmidt NW, Podyminogin RL, Butler NS, Badovinac VP, Tucker BJ, Bahjat KS, Lauer P, Reyes-Sandoval A, Hutchings CL, Moore AC, Gilbert SC, Hill AV, Bartholomay LC, Harty JT. Memory CD8 T cell responses exceeding a large but definable threshold provide long-term immunity to malaria. Proceedings of the National Academy of Sciences of the United States of America. 2008;105:14017–14022. doi: 10.1073/pnas.0805452105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Badovinac VP, Haring JS, Harty JT. Initial T cell receptor transgenic cell precursor frequency dictates critical aspects of the CD8(+) T cell response to infection. Immunity. 2007;26:827–841. doi: 10.1016/j.immuni.2007.04.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kamimura D, Sawa Y, Sato M, Agung E, Hirano T, Murakami M. IL-2 in vivo activities and antitumor efficacy enhanced by an anti-IL-2 mAb. J Immunol. 2006;177:306–314. doi: 10.4049/jimmunol.177.1.306. [DOI] [PubMed] [Google Scholar]
- 25.Bloom MB, Perry-Lalley D, Robbins PF, Li Y, el-Gamil M, Rosenberg SA, Yang JC. Identification of tyrosinase-related protein 2 as a tumor rejection antigen for the B16 melanoma. The Journal of experimental medicine. 1997;185:453–459. doi: 10.1084/jem.185.3.453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wang RF, Appella E, Kawakami Y, Kang X, Rosenberg SA. Identification of TRP-2 as a human tumor antigen recognized by cytotoxic T lymphocytes. The Journal of experimental medicine. 1996;184:2207–2216. doi: 10.1084/jem.184.6.2207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Cho HI, Celis E. Optimized peptide vaccines eliciting extensive CD8 T-cell responses with therapeutic antitumor effects. Cancer research. 2009;69:9012–9019. doi: 10.1158/0008-5472.CAN-09-2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Tomala J, Chmelova H, Mrkvan T, Rihova B, Kovar M. In vivo expansion of activated naive CD8+ T cells and NK cells driven by complexes of IL-2 and anti-IL-2 monoclonal antibody as novel approach of cancer immunotherapy. J Immunol. 2009;183:4904–4912. doi: 10.4049/jimmunol.0900284. [DOI] [PubMed] [Google Scholar]
- 29.Votavova P, Tomala J, Kovar M. Increasing the biological activity of IL-2 and IL-15 through complexing with anti-IL-2 mAbs and IL-15Ralpha-Fc chimera. Immunology letters. 2014;159:1–10. doi: 10.1016/j.imlet.2014.01.017. [DOI] [PubMed] [Google Scholar]
- 30.Valenzuela J, Schmidt C, Mescher M. The roles of IL-12 in providing a third signal for clonal expansion of naive CD8 T cells. J Immunol. 2002;169:6842–6849. doi: 10.4049/jimmunol.169.12.6842. [DOI] [PubMed] [Google Scholar]
- 31.Watts TH. TNF/TNFR family members in costimulation of T cell responses. Annual review of immunology. 2005;23:23–68. doi: 10.1146/annurev.immunol.23.021704.115839. [DOI] [PubMed] [Google Scholar]
- 32.Myers L, Lee SW, Rossi RJ, Lefrancois L, Kwon BS, Mittler RS, Croft M, Vella AT. Combined CD137 (4-1BB) and adjuvant therapy generates a developing pool of peptide-specific CD8 memory T cells. International immunology. 2006;18:325–333. doi: 10.1093/intimm/dxh371. [DOI] [PubMed] [Google Scholar]
- 33.Lee SW, Park Y, Song A, Cheroutre H, Kwon BS, Croft M. Functional dichotomy between OX40 and 4-1BB in modulating effector CD8 T cell responses. J Immunol. 2006;177:4464–4472. doi: 10.4049/jimmunol.177.7.4464. [DOI] [PubMed] [Google Scholar]
- 34.Alexander-Miller MA. High-avidity CD8+ T cells: optimal soldiers in the war against viruses and tumors. Immunologic research. 2005;31:13–24. doi: 10.1385/IR:31:1:13. [DOI] [PubMed] [Google Scholar]
- 35.Moore TV, Lyons GE, Brasic N, Roszkowski JJ, Voelkl S, Mackensen A, Kast WM, Le Poole IC, Nishimura MI. Relationship between CD8-dependent antigen recognition, T cell functional avidity, and tumor cell recognition. Cancer immunology, immunotherapy : CII. 2009;58:719–728. doi: 10.1007/s00262-008-0594-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ganesan AP, Johansson M, Ruffell B, Yagui-Beltran A, Lau J, Jablons DM, Coussens LM. Tumor-infiltrating regulatory T cells inhibit endogenous cytotoxic T cell responses to lung adenocarcinoma. J Immunol. 2013;191:2009–2017. doi: 10.4049/jimmunol.1301317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Sato E, Olson SH, Ahn J, Bundy B, Nishikawa H, Qian F, Jungbluth AA, Frosina D, Gnjatic S, Ambrosone C, Kepner J, Odunsi T, Ritter G, Lele S, Chen YT, Ohtani H, Old LJ, Odunsi K. Intraepithelial CD8+ tumor-infiltrating lymphocytes and a high CD8+/regulatory T cell ratio are associated with favorable prognosis in ovarian cancer. Proceedings of the National Academy of Sciences of the United States of America. 2005;102:18538–18543. doi: 10.1073/pnas.0509182102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Pulaski BA, Ostrand-Rosenberg S. Mouse 4T1 breast tumor model. In: Coligan John E, et al., editors. Current protocols in immunology. Unit 20–22. Chapter 20. 2001. [DOI] [PubMed] [Google Scholar]
- 39.Tao K, Fang M, Alroy J, Sahagian GG. Imagable 4T1 model for the study of late stage breast cancer. BMC cancer. 2008;8:228. doi: 10.1186/1471-2407-8-228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Scrimieri F, Askew D, Corn DJ, Eid S, Bobanga ID, Bjelac JA, Tsao ML, Allen F, Othman YS, Wang SC, Huang AY. Murine leukemia virus envelope gp70 is a shared biomarker for the high-sensitivity quantification of murine tumor burden. Oncoimmunology. 2013;2:e26889. doi: 10.4161/onci.26889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Ahmadzadeh M, Rosenberg SA. IL-2 administration increases CD4+ CD25(hi) Foxp3+ regulatory T cells in cancer patients. Blood. 2006;107:2409–2414. doi: 10.1182/blood-2005-06-2399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Al-Lazikani B, Banerji U, Workman P. Combinatorial drug therapy for cancer in the post-genomic era. Nature biotechnology. 2012;30:679–692. doi: 10.1038/nbt.2284. [DOI] [PubMed] [Google Scholar]
- 43.Starbeck-Miller GR, Xue HH, Harty JT. IL-12 and type I interferon prolong the division of activated CD8 T cells by maintaining high-affinity IL-2 signaling in vivo. The Journal of experimental medicine. 2014;211:105–120. doi: 10.1084/jem.20130901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Kyi C, Postow MA. Checkpoint blocking antibodies in cancer immunotherapy. FEBS letters. 2014;588:368–376. doi: 10.1016/j.febslet.2013.10.015. [DOI] [PubMed] [Google Scholar]
- 45.Castle JC, Kreiter S, Diekmann J, Lower M, van de Roemer N, de Graaf J, Selmi A, Diken M, Boegel S, Paret C, Koslowski M, Kuhn AN, Britten CM, Huber C, Tureci O, Sahin U. Exploiting the mutanome for tumor vaccination. Cancer research. 2012;72:1081–1091. doi: 10.1158/0008-5472.CAN-11-3722. [DOI] [PubMed] [Google Scholar]
- 46.Schumacher TN, Schreiber RD. Neoantigens in cancer immunotherapy. Science. 2015;348:69–74. doi: 10.1126/science.aaa4971. [DOI] [PubMed] [Google Scholar]
- 47.Mellman I, Coukos G, Dranoff G. Cancer immunotherapy comes of age. Nature. 2011;480:480–489. doi: 10.1038/nature10673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Shien T, Kinoshita T, Shimizu C, Hojo T, Taira N, Doihara H, Akashi-Tanaka S. Primary tumor resection improves the survival of younger patients with metastatic breast cancer. Oncology reports. 2009;21:827–832. [PubMed] [Google Scholar]
- 49.Steeg PS. Tumor metastasis: mechanistic insights and clinical challenges. Nature medicine. 2006;12:895–904. doi: 10.1038/nm1469. [DOI] [PubMed] [Google Scholar]
- 50.Bouneaud C, Kourilsky P, Bousso P. Impact of negative selection on the T cell repertoire reactive to a self-peptide: a large fraction of T cell clones escapes clonal deletion. Immunity. 2000;13:829–840. doi: 10.1016/s1074-7613(00)00080-7. [DOI] [PubMed] [Google Scholar]
- 51.Pardoll D. Does the Immune System See Tumors as Foreign or Self? Annual review of immunology. 2003;21:32. doi: 10.1146/annurev.immunol.21.120601.141135. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.