Abstract
Objectives
The correct identification of synovitis is critical for achieving optimal therapy results. Fluorescence optical imaging (FOI) is a novel modality based on the use of an intravenous fluorophore, which enables fluorescent imaging of the hands and wrists with increased focal optical signal intensities in areas of high perfusion and/or capillary leakage. The study objective was to determine the diagnostic utility of FOI in detecting apparent and clinically non-apparent active synovitis.
Methods
Bilateral hand and wrist joints (n=872) of 26 patients with inflammatory arthritis assessed by standard clinical examination, musculoskeletal ultrasound (MSUS) and FOI were studied. Synovitis was defined as tender and swollen joints on clinical examination, presence of synovial thickening and intra-articular Doppler signals on MSUS, and abnormal focal optical signal intensities on FOI, respectively. Subclinical synovitis was defined as being clinically non-apparent, but positively inflamed on MSUS.
Results
Depending on the standard used to define inflammation, FOI ranged from 73–83% sensitive and 83–95% specific for detecting manifest synovitis. For detecting clinically silent synovitis, the sensitivity, specificity and positive and negative predictive values of FOI were 80%, 96%, 77% and 97%, respectively.
Conclusions
The high degree of agreement between MSUS and FOI suggest its use in clinical practice, especially when MSUS is not available, in order to identify synovitis earlier and with greater confidence. FOI may be particularly useful in identifying patients with clinically non-apparent joint inflammation of the hands and/or wrists.
Keywords: Rheumatoid Arthritis, Synovitis, Ultrasonography, Inflammation, Multidisciplinary team-care
Key messages.
What is already known on this subject?
Imaging modalities such as MRI and musculoskeletal ultrasound (MSUS) are validated technologies that are known to have opened new windows of opportunity for accurately assessing inflammatory changes (synovitis).
Fluorescence optical imaging (FOI) has been approved for use in human subjects, and previous studies have reported that FOI correlates positively with MSUS and MRI in evaluating disease activity and synovitis.
What does this study add?
FOI is a time efficient and operator-independent imaging modality.
This study adds FOI's detection of clinically silent synovitis.
How might this impact on clinical practice?
FOI may be a useful tool in identifying patients with subtle synovial inflammation of the hands and wrists. It may also be used in clinical practice to accurately identify synovitis earlier and with greater confidence.
Introduction
The timely identification and diagnostic precision of arthritis detection, especially subclinical synovitis, would be of critical importance in the early inflammatory arthritis (IA) process.1–6 Imaging modalities such as MRI and musculoskeletal ultrasound (MSUS) are validated technologies that can help achieve this,6–9 and have opened new windows of opportunity for accurately accessing inflammatory changes and structural damage.8–13 In a recent report, we showed MSUS to have significantly increased the diagnostic certainty for having IA (rheumatoid arthritis (RA) in particular) and the rheumatologists’ confidence in establishing the presence and absence of inflammation.14 While having numerous benefits in the management of IA and rheumatology practice in general,12–19 MSUS is, to a certain degree, operator dependent, and may be limited in visualising very detailed altered microcirculation, perfusion and/or capillary leakage, especially in the very small finger joints.
Fluorescence optical imaging (FOI) is a time-efficient and operator-independent imaging modality that can also be performed by nurses or other non-MD (Doctor of Medicine) personnel.3 20 21 The image interpretation, however, is dependent on visual perceptions and imaging experience of the examiner. It is a novel tool that utilises an intravenous fluorophore,3 20–34 allowing imaging of the hands and wrists with increased focal optical signal intensities in areas of high perfusion, altered microcirculation and/or capillary leakage.3 20 21 Animal studies have utilised fluorophores such as indocyanine green (ICG) for various indications, which have been shown to be useful in the detection of inflammation.32 FOI has been approved for use in human subjects, and correlates positively with MSUS and MRI findings in evaluating disease activity and synovitis.25 28 30 33 Werner et al25 carried out the first comparative study of FOI with other imaging modalities and reported FOI to show a good agreement for arthritis diagnosis with power Doppler and MRI findings, achieving 76% sensitivity and 54% specificity in detecting synovitis and tenosynovitis. Meier et al,26 in contrast, found FOI sensitivity and specificity to be 40% and 85%, respectively, indicating FOI's limitations to detect mild synovitis as compared to a higher resolution MRI system, but showed more consistent results in FOI's detection of severely inflamed joints. Further studies on the value of FOI are thus needed.
Our study was designed to determine the sensitivity, specificity and predictive values of FOI as a diagnostic tool in identifying manifest synovitis, when compared with standard clinical assessments and ultrasound findings. We then investigated whether FOI could be used to complement MSUS (colour/power Doppler) for ascertaining subclinical (silent) synovitis in the hands and wrists.
Methods
Study population and setting
The hand and wrist joints of patients attending the rheumatology clinic of the Karolinska University Hospital in Stockholm, Sweden, were studied, where an FOI examination was offered as part of patient care at the early arthritis clinic, when clinically justified by the referring physician. All patients were screened for allergies, and fully informed of the FOI procedure by the rheumatology nurse and rheumatologist prior to this investigation. The clinical, ultrasound and FOI examinations were carried out on the same day.
Clinical and MSUS assessment
The referred rheumatologists performed standardised clinical joint examinations. Joints were considered inflamed in the presence of tenderness and swelling, and reports were uploaded onto the patients’ journals.
One of two ultrasound machines was used to carry out the MSUS examination. Standardised presets by the manufacturer were utilised for the Siemens Accuson X150 ultrasound machine, while instrument presets according to Rezaei et al14 were used for the General Electric (GE) LOGIQ E9 ultrasound machine (Wauwatosa, Wisconsin, USA). A linear array transducer frequency of 15 MHz was utilised for brightness modulation (B-Mode) scanning, and 10 MHz for colour Doppler ultrasound, set at a pulse repetition frequency of 0.5 KHz, using a 69 Hz wall filter. For all patients, the disease activity per joint was semiquantitatively measured according to the scoring system of Ohrndorf et al.35 In this study, ultrasound was considered positive for active synovitis when synovial thickening (synovial hypertrophy) and intra-articular Doppler signals were present.
FOI assessment
Prior to the examination, hand washing was advised to prevent reflective artefacts (dust, dirt, powder etc), thus maintaining good image resolution. Patients were advised to remain as still as possible (to avoid movement artefacts) under darkroom conditions, ensuring minimal background light scatter—which may affect the charge coupled device flash exposures over the 6 min. The FOI examination followed the standardised procedure: both hands were placed on a preformed hand rest, with the index fingertips reaching maximum distance. The light protective curtaining was free from the regions of interest for full imaging coverage. An image preview was established to ensure optimal background settings, free from fluorophore spillage. Ten seconds after the initialisation of the procedure, an ICG bolus was injected by a rheumatology nursing professional (ICG—pulsion, 0.1 mg/kg/body weight intravenously).
The FOI image sequence was recorded (see online supplementary video clip (360 s)) and the electronically generated composite image was automatically obtained by means of the integrated XiraView software (V.3.6). FOI was considered positive in joints that displayed abnormal focal optical signal intensities by visual inspection of the entire image series (34 joints: 3 wrists, 5 metacarpal phalangeals (MCPs), 5 proximal interphalangeals (PIPs) and 4 distal interphalangeals (DIPs), bilaterally). Using postprocessing imaging techniques, the instrument settings were adjusted for artefact reduction, and optimum image resolution was established for each frame. For this study, only two categories of synovitis were used: either the presence of abnormal signal intensities marked as FOI (+) or the absence of abnormal signal intensities marked as FOI (–) per joint, were scored for all patients. Without prior knowledge of the clinical and MSUS results, the image series were jointly analysed for each patient, using consensus scoring by two observers: a sonographer specialised in imaging and a rheumatologist with FOI training. An independent research physician not involved in the scoring acquired the clinical joint counts, MSUS and FOI reports from the charts and patient journals for analysis.
Statistical methods
IBM SPSS V.22.0 and GraphPad software was utilised for statistical analyses. Strengths of agreement between standard clinical examination, MSUS and FOI findings were compared using κ statistics, and the metric properties (sensitivity and specificity; and positive predictive and negative predictive values (PPV, NPV)) for FOI were calculated using clinical evaluation or MSUS, respectively, as reference measures (gold standards). Subclinical (silent) synovitis was defined as being clinically non-apparent, but actively inflamed by MSUS. The number of joints also positive by FOI was then determined using the combined prevalence of clinical and MSUS as the reference measure.
Results
Patient characteristics and distribution of diagnosis
A total of 872 joints, including 3 wrist joints (radiocarpal, midcarpal and ulnarcarpal regions), 5 MCPs, 5 PIPs and 4 DIPs in 26 patients (18 female, 8 male, average age 51.5 years: range 19–84 years) with various rheumatic diseases (RA: 12, psoriatic arthritis: 2, dermatomyositis with RA overlap: 1, systemic lupus erythaematosus: 1, Sicca syndrome (Sjögrens; SS): 2, fibromyalgia: 1, idiopathic gout: 1, joint pain: 2, juvenile idiopathic arthritis and polyarthritis/undifferentiated IA 1–2 each) were examined clinically, and by MSUS and FOI (table 1).
Table 1.
Patient distribution according to methods of assessment and diagnosis
| Baseline characteristics | All patients Mean (±SD) (N=26) | RA (N=12) | PsA (N=2) | DM (RA) overlap (1), SLE (1), Sicca syndrome (2) (N=4) | FM (1), idiopathic gout (1), joint pain (2) (N=4) | JIA (1), polyarthritis (1), UIA (2) (N=4) |
|---|---|---|---|---|---|---|
| Gender (female): N (%) | 18 (69.2) | 9 (75) | 1 (50) | 3 (75) | 3 (75) | 2 (50) |
| Age (years) | 51.5 (±17.5) | 54.3 (±16.3) | 47 (±1) | 45.3 (±12.7) | 44 (±15) | 58.8 (±23.5) |
| Symptom duration (months) | 22.3 (±26.5) | 20.3 (±25.5) | 43.5 (±40.5) | 23.3 (±11.7) | 31.3 (±33.4) | 7.5 (±3.2) |
| Anti-CCP status: | ||||||
| Positive: N (%) | 15 (57.7) | 9 (75) | 0 (0) | 3 (75) | 2 (50) | 1 (25) |
| Negative: N (%) | 11 (42.3) | 3 (25) | 2 (100) | 1 (25) | 2 (50) | 3 (75) |
| RF status: | ||||||
| Positive: N (%) | 9 (34.6) | 5 (41.7) | 1 (50) | 3 (75) | 0 (0) | 1 (25) |
| Negative: N (%) | 17 (65.4) | 7 (58.3) | 1 (50) | 1 (25) | 4 (100) | 3 (75) |
| ESR levels (mm/h) | 22.3 (±18.6) | 28.3 (±21.1) | 6 (±2) | 35.3 (±10.9) | 6.8 (±2.2) | 15.3 (±9.0) |
| CRP levels (mg/L) | 11.9 (±23.2) | 20.3 (±31.5) | 1.8 (±1.2) | 10.3 (±8.6) | 1.5 (±0.9) | 3.5 (±1.8) |
| Smoking: previous and current status: N (%) | 16 (61.5) | 7 (58.3) | 1 (50) | 3 (75) | 2 (50) | 1 (25) |
| X-rays (bone erosions): N (%) | 7 (29.9) | 4 (33.3) | 1 (50) | 0 (0) | 1 (25) | 1 (25) |
| Clinical assessment | ||||||
| Hand and/or wrist inflammation: N (%) | 18 (69.2) | 10 (83.3) | 2 (100) | 2 (50) | 2 (50) | 2 (50) |
| Hand and wrist Joints swollen |
5.5 (±7.5) | 9.7 (±9.1) | 2 (±1) | 1.8 (±2) | 1.3 (±1.3) | 2.8 (±2.8) |
| MSUS | ||||||
| Hand and/or wrist inflammation: N (%) | 23 (88.5) | 12 (100) | 2 (100) | 3 (75) | 3 (75) | 3 (75) |
| MSUS synovitis Joint count |
9.3 (±9.7) | 12.2 (±9.8) | 14.5 (±11.5) | 7.8 (±10.6) | 3 (±2.5) | 6 (±6.4) |
| Fluorescence optical imaging | ||||||
| Hand and/or wrist inflammation: N (%) | 22 (84.6) | 11 (91.7) | 2 (100) | 3 (75) | 3 (75) | 3 (75) |
| FOI synovitis Joint count |
8.8 (±8.8) | 11.8 (±10.0) | 16 (±6) | 6 (±7.6) | 3 (±2.5) | 4.8 (±4.0) |
Anti-CCP, anti-cyclic citrullinated peptide; CRP, C reactive protein; DM, dermatomyositis; ESR, erythrocyte sedimentation rate; FM, fibromyalgia; FOI, fluorescence optical imaging; JIA, juvenile idiopathic arthritis; MSUS, musculoskeletal ultrasound; PsA, psoriatic arthritis; RA, rheumatoid arthritis; RF, rheumatoid factor; SLE, systemic lupus erythaematosus; SS, Sjögrens Syndrome; UIA, undifferentiated inflammatory arthritis.
The majority of patients (17/26) had symptom duration less than 1 year. Mean (±SD) symptom duration was 22.3 (±26.5), the shortest being 2 months and the longest 84 months. Anti-cyclic citrullinated peptide (anti-CCP) and rheumatoid factor (RF) were positive in 15 (58%) and 11 (42%), respectively. Of the 12 patients diagnosed with RA, 7 (75%) were anti-CCP positive and 5 (42%) RF positive. The mean (±SD) results for the erythrocyte sedimentation rate and C reactive protein levels were 22.3 (±18.6) mm/h and 11.9 (±23.2) mg/L for the total population, respectively. Of the 26 patients selected for this study, 16 (62%) were either previous or current smokers. The hand and wrist radiographs revealed the presence of bone erosions in 30% (7/26), of whom 57% (4/7) were diagnosed with erosive RA. The number and percentage of patients diagnosed as having inflammation were 18 (69%) by clinical examination, 23 (89%) by ultrasound and 22 (85%) by FOI, with the mean (±SD) number of joints inflamed being 5.5 (±7.5), 9.3 (±9.7) and 8.8 (±8.8) by clinical, MSUS and by FOI examination, respectively (table 1).
FOI predictive values from sensitivity, specificity and prevalence
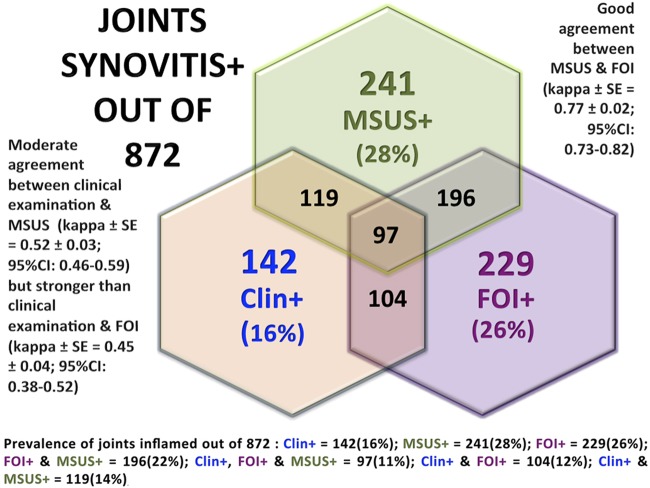
Of the 872 joints evaluated, the prevalence of inflammation detection was 142 (16%), 241 (28%) and 229 (26%) by clinical examination, MSUS and FOI, respectively (figure 1).
Figure 1.
Strengths of agreement and prevalence of synovitis (+) joints by clinical examination (clin+), musculoskeletal ultrasound (MSUS+) and fluorescence optical imaging (FOI+) in 872 hand and wrist joints of 26 inflammatory arthritis patients.
Clinical examination and/or MSUS as a reference measure
Of the 142 (16%) clinically inflamed joints, 84% (119/142) were also positive by MSUS versus 73% (104/142) by FOI (figure 1). Of the 730 non-inflamed joints (84% of 872) by clinical examination, 83% (608/730) were non-inflamed by MSUS and 83% (605/730) were non-inflamed by FOI (figure 2). The PPV in the current setting, where the large majority of examined joints were not inflamed, was low, as expected (<50%), for MSUS and FOI, yielding a 97% and 95% NPV, respectively (figure 2). Of the 16% (142/872) of clinically inflamed joints using MSUS (28% prevalence) as a reference, there was a PPV of 63% in FOI detection of positive joints and 89% NPV for detecting clinically non-inflamed joints. Of the 241 inflamed joints by MSUS, 196 (81%) were inflamed by FOI, and only 119 (49%) joints were inflamed clinically. The sensitivity of 81% and PPV of 87% was achieved in FOI's detection of active synovitis in 28% of the MSUS positive joints. A PPV of 73% was indicated when using both clinical and MSUS (14% prevalence) as reference measures (figures 1 and 2) Of the 631 non-inflamed (72% of 872) joints by MSUS, 598 (95%) were also non-inflamed by FOI. The specificity of FOI was 95% and the NPV 93%, for non-inflamed MSUS joints (figure 2).
Figure 2.
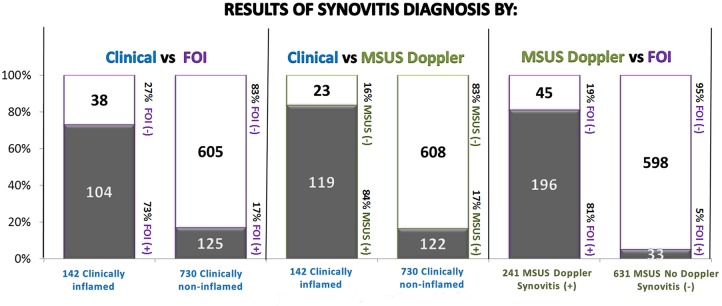
The distribution of hand and wrist joint synovitis diagnosis by clinical examination and imaging (MSUS and FOI) methods (FOI, fluorescence optical imaging; MSUS, musculoskeletal ultrasound).
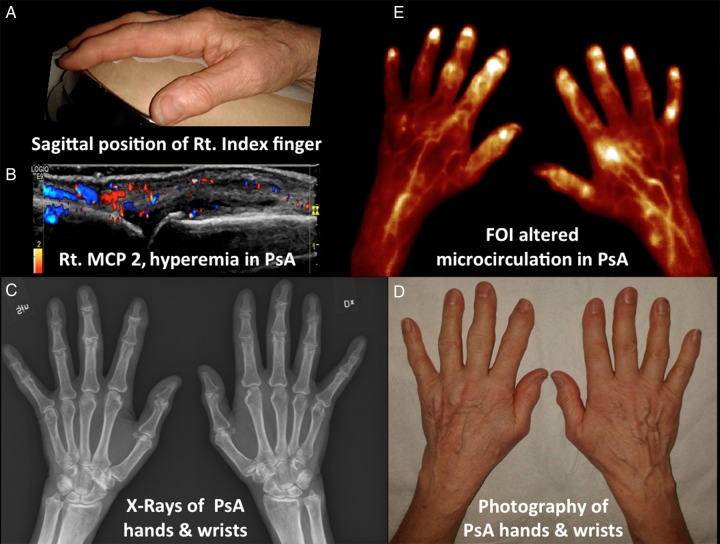
Figure 3.
Imaging methods of photography, MSUS, X-rays and FOI in evaluating a patient with PsA. (A) Photograph of sagittal position of right index finger. (B) Hyperaemia of right MCP 2 indicated by intra-articular colour Doppler activity including extra-articular tissue involvement in PsA. Active synovitis is apparent as synovial thickening, Doppler signals and effusion. Bone erosion and osteophyte were evident in the metacarpal head of MCP 2. (C) X-ray of bilateral hands and wrists in PsA. Bone erosions, osteophytes and joint space narrowing noted. Sesamoid bones are also seen. (D) Photography of bilateral hands and wrists of PsA with bony swelling apparent. (E) FOI using ‘temperature’ pallet settings demonstrating increased abnormal focal optical signal intensities in areas of capillary leakage, altered microcirculation and perfusion. The right wrist (radial), IP 1, MCP 1 and 2, PIP 5 and DIPs 2–5 showed synovitis positive on the right and left. IP 1, PIP 2 and 3 and DIPs 2 and 3 showed abnormal signal intensities on the left. Tenosynovitis of bilateral thumbs and left index finger noted. Also apparent is fluorophore perfused skin tissue variation (DIP, distal interphalangeal; FOI, fluorescence optical imaging; IP, interphalangeal; MSUS, musculoskeletal ultrasound; MCP, metacarpal-phalangeal; PsA, psoriatic arthritis; PIP, proximal interphalangeal).
Strengths of agreement between clinical examination, MSUS and FOI
The strengths of agreement between clinical examination and MSUS were moderate (κ±SE=0.524±0.033; 95% CI 0.459 to 0.589) and somewhat stronger than the agreement between clinical examination and FOI (κ±SE=0.450±0.035; 95% CI 0.381 to 0.519; figure 1). The strength of agreement between MSUS and FOI in the detection of active synovitis was good (κ±SE=0.773±0.024; 95% CI 0.725 to 0.821; figure 1).
Detection of subclinical inflammation by FOI
In the absence of clinical inflammation (730/872), 17% of joints (125/730) were inflamed by FOI while 17% (122/730) were inflamed by MSUS (table 2).
Table 2.
Subclinical inflammation
| Clinically (−) |
|||
|---|---|---|---|
| MSUS Doppler (+) | MSUS Doppler (−) | Total | |
| FOI (synovitis+) | 98 | 27 | 125 |
| FOI (synovitis−) | 24 | 581 | 605 |
| Total | 122 | 608 | 730 (out of 872) |
FOI, fluorescence optical imaging; MSUS, musculoskeletal ultrasound.
Of the 122 clinically non-inflamed joints that were MSUS positive, 98 (80%) were also inflamed by FOI. Thus, the sensitivity of FOI for detecting clinically silent synovitis when defined as a positive MSUS in the absence of clinical inflammation was 80% and the PPV in this material 77% (table 2). These joints comprised mainly of the radial, mid-carpal and ulnar carpal wrist joints. Of the 608 joints that were non-inflamed clinically and non-inflamed by MSUS, 4% (27/608) had inflammation by FOI, which comprised mainly of the DIP and PIP joints, yielding a specificity of 96% (581/608) and a NPV of 97%.
Discussions
FOI is an emerging imaging modality that is relatively quick and reliable and technically feasible. The use of the intravenous fluorophore ICG provides good image resolution during the ‘venous wash-out’ and arterial phases of absorbed microcirculation, and has been shown to be an appropriate tracer to enhance inflamed joints.33 In very small joints such as the DIPs, FOI may provide greater microcirculatory information than MSUS, as observed in our study, where 4% (27/608) of the FOI positive joints were not reported to be inflamed by MSUS.
A limitation of FOI is the near infrared light reflective properties of fluorophore signalling in patients with increased adiposity (thick hands), especially in the wrist region. These limitations may be overcome with adjustments of instrument settings and filtres, to improve tissue absorption and light reflection at varied wavelengths. Fischer et al23 carried out MRI and a prototype of ICG-enhanced FOI in healthy volunteers compared with clinically active patients with RA, showing a high correlation (r=0.84) between the FOI and MRI results. Werner et al proposed a standardised approach of scaling and image interpretation of the three different phases, which showed a substantial intrareader and inter-reader reliability of κ=0.78.35 The correct interpretations of images are important for establishing accurate diagnostics. The FOI images need to be viewed by an experienced reader in its entirety (360 s, see online supplementary video clip), in order to reduce false positive or negative results and interobserver variability. Some consider the first phase or at least the first 240 s to be sufficient for diagnostics. The results for inter-reader and intrareader agreement by Meier et al26 suggested that the basic interpretation of optical images was moderately consistent among readers with varied medical experience. They also recommended further training and standardised grading schemes to improve intergroup reproducibility for synovitis assessment. The added advantage of our FOI study was that we used consensus scoring between the two readers to eliminate observer variability and discordant outcomes.
In our experience, we found FOI favourable in detecting clinically non-apparent inflammation in joints that were MSUS positive. According to the overall clinical assessment of the 26 patients, 18 (69%) were diagnosed with some form of hand and wrist inflammation, while ultrasound showed inflammation in 23 (89%) and FOI in 22 (85%) of the patients studied. We also found FOI to be beneficial in the management of patients with disparate rheumatic diseases who showed no clinical signs of hand or wrist swelling but had joint inflammation on MSUS and FOI. Although FOI was superior to clinical examination in detecting wrist inflammation, it was not as obvious, as seen on MSUS, in mild wrist synovitis. We have, however, recognised some ‘blotchy’ signal patterns as possible indications for positive signals of the wrist in mild disease state, by adjusting the filtres and pallet settings to the ‘temperature’ setting. If wrist tenosynovitis is involved, it may warrant an MSUS for confirmation.36 The depth and thickness of the wrist is what makes this region more difficult as compared with smaller joints where the image resolution is superior to ultrasound in terms of detailed microcirculatory tissue information (figure 3). Thus, our study suggests the use of FOI in patients with suspected early IA.
Limitations of our study include the relatively small sample size and missing some data. The intravenous injection of fluorophore ICG rarely causes allergic reactions and therefore requires special attention to precautionary measures and a clinical setting equipped to deal with such events. Also strong in our study was the inclusion of a mix of rheumatic disorders, and the fact that individual joints were evaluated using MSUS as a validated and feasible reference measure for confirmation of ambiguous or uncertain FOI signal patterns. Another strength of this study was that an agreement was established between the two readers (consensus scoring) and the combined report was used for this analysis. For further studies, we will focus on larger study populations and on patients grouped according to diagnosis, X-rays/other imaging modalities and biochemistry results. Treatment monitoring of patients on dose reduction therapeutic strategies,37 38 (RF van Vollenhoven. Reducing dose of etanercept effective in reducing RA flares in select patients. Ann Rheum 2014. Submitted for publication) using digital activity software (DACT-FOI), and supplementary hand and wrist views may also be assessed in future FOI studies.
In conclusion, we found the sensitivity of FOI for detecting clinically manifest inflammation in individual joints of the hands and wrists to range between 73–84%, and the specificity from 83–95%, suggesting a slightly lower sensitivity but similar specificity compared with MSUS. For detecting subclinical synovitis in the hands and wrists, 80% (98/142) sensitivity and 96% (557/581) specificity were seen. These metrics suggest that FOI may be a useful tool in identifying patients with subtle synovial inflammation of the hands and/or wrists. The good strength of agreement between MSUS and FOI also suggests that the latter may be used in clinical practice, particularly where ultrasound is not available, in order to accurately identify synovitis earlier and with greater confidence.
Supplementary Material
Acknowledgments
The authors would like to acknowledge to all participating patients and staff of the Rheumatology Clinic at the Karolinska University Hospital, and all at ClinTRID, who have contributed to this study in one way or the other.
Footnotes
Contributors: RvV, EaK, NG and YK were responsible for the conception and design of this FOI study. HR designed the musculoskeletal ultrasound performance. AK carried out all the nursing protocols; and together with EaK and YK, performed the FOI examinations. All FOI image series (360 image frames per patient) were postprocessed by YK for analysis. YK and EaK performed the image interpretations separately, followed by an FOI report in combination. NG acquired all the data for analysis and interpreted the statistics together with YK, EaK and RvV. All the authors contributed equally towards the preparation of this article, and critically revised it for important intellectual content and unanimously approved the final manuscript for submission.
Competing interests: RvV has received research support and/or honoraria from AbbVie, Biotest, BMS, UCB and Vetex, all of which are not related to this study. HR has received lecture honoraria from General Electric and AbbVie, neither of which are related to this study.
Provenance and peer review: Not commissioned; externally peer reviewed.
Data sharing statement: No additional data are available.
References
- 1.Gerlag DM, Raza K, van Baarsen LGM et al. . EULAR recommendations for terminology and research in individuals at risk of rheumatoid arthritis: report from the Study Group for Risk Factors for Rheumatoid Arthritis. Ann Rheum Dis 2012;71:638–41. 10.1136/annrheumdis-2011-200990 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Van Vollenhoven R, Naggy G, Tak PP. Early start and stop of biologics: has the time come? BMC Med 2014;12:25 10.1186/1741-7015-12-25 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kisten Y, Györi N, Rezaei H et al. . The utility of fluorescence optical imaging for detecting clinically silent synovitis of the hands & wrists [abstract]. Arthritis Rheum 2013;65(Suppl 10):1970. [Google Scholar]
- 4.O'Dell JR. Treating rheumatoid arthritis early: a window of opportunity? Arthritis Rheum 2002;46:283–5. 10.1002/art.10092 [DOI] [PubMed] [Google Scholar]
- 5.Raza K, Saber TP, Kvien TK et al. . Timing the therapeutic window of opportunity in early rheumatoid arthritis: proposal for definitions of disease duration in clinical trials. Ann Rheum Dis 2012;71:1921–3. 10.1136/annrheumdis-2012-201893 [DOI] [PubMed] [Google Scholar]
- 6.Deane KD, Norris JM, Holers VM. Pre-clinical rheumatoid arthritis: identification, evaluation and future directions for investigation. Rheum Dis Clin North Am 2010;36:213–41. 10.1016/j.rdc.2010.02.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Tins BJ, Butler R. Imaging in rheumatology: reconciling radiology and rheumatology. Insights Imaging 2014;4:799–810. 10.1007/s13244-013-0293-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Østergaard M. Can imaging be used for inflammatory arthritis screening? Semin Musculoskelet Radiol 2012;16:401–9. 10.1055/s-0032-1329893 [DOI] [PubMed] [Google Scholar]
- 9.Filippucci E, Di Geso L, Grassi W. Progress in imaging in rheumatology. Nat Rev Rheumatol 2014;10:628–34. 10.1038/nrrheum.2014.145 [DOI] [PubMed] [Google Scholar]
- 10.McQueen FM. Imaging in early rheumatoid arthritis. Best Pract Res Clin Rheumatol 2013;27:499–522. 10.1016/j.berh.2013.09.005 [DOI] [PubMed] [Google Scholar]
- 11.Døhn UM, Ejberg BJ, Court-Payen M et al. . Are bone erosions detected by magnetic resonance imaging and ultrasonography true erosions? A comparison with computed tomography in rheumatoid arthritis metacarpophalangeal joints. Arthritis Res Ther 2006;8:R110 10.1186/ar1995 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Szkudlarek M, Court-Payen M, Strandberg C et al. . Power Doppler ultrasonography for assessment of synovitis in the metacarpophalangeal joints of patients with rheumatoid arthritis: a comparison with dynamic magnetic resonance imaging. Arthritis Rheum 2001;44:2018–23. [DOI] [PubMed] [Google Scholar]
- 13.Backhaus M, Burmester GR, Sandrock D et al. . Prospective two year follow up study comparing novel and conventional imaging procedures in patients with arthritic finger joints. Ann Rheum Dis 2002;61:895–904. 10.1136/ard.61.10.895 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Rezaei H, Torp-Pedersen S, Af Klint E et al. . Diagnostic utility of musculoskeletal ultrasound in patients with suspected arthritis—a probabilistic approach. Arthritis Res Ther 2014;16:448 10.1186/s13075-014-0448-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hammer HB, Kvien TK. Comparisons of 7 to 78-joint ultrasonography scores: all different joint combinations show equal response to adalimumab treatment in patients with rheumatoid arthritis. Arthritis Res Ther 2011;13:R78 10.1186/ar3341 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Klauser A, Frauscher F, Schirmer M et al. . The value of contrast-enhanced color Doppler ultrasound in the detection of vascularization of finger joints in patients with rheumatoid arthritis. Arthritis Rheum 2002;46:647–53. 10.1002/art.10136 [DOI] [PubMed] [Google Scholar]
- 17.Brown AK. Using ultrasonography to facilitate best practice in diagnosis and management of RA. Nat Rev Rheumatol 2009;5:698–706. 10.1038/nrrheum.2009.227 [DOI] [PubMed] [Google Scholar]
- 18.Szkudlarek M, Klarlund M, Narvestad E et al. . Ultrasonography of the metacarpophalangeal and proximal interphalangeal joints in rheumatoid arthritis: a comparison with magnetic resonance imaging, conventional radiography and clinical examination. Arthritis Res Ther 2006;8:R52 10.1186/ar1904 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Terslev L, Torp-Pedersen S, Savnik A et al. . Doppler ultrasound and magnetic resonance imaging of synovial inflammation of the hand in rheumatoid arthritis: a comparative study. Arthritis Rheum 2003;48:2434–41. 10.1002/art.11245 [DOI] [PubMed] [Google Scholar]
- 20.Kisten Y, Györi N, Rezaei H et al. . Clinically “silent” synovitis: detected by fluorescence optical imaging of the hands & wrists [abstract]. Ann Rheum Dis 2014;73(Suppl 2):1139 10.1136/annrheumdis-2014-eular.2215 [DOI] [Google Scholar]
- 21.Györi N, Kisten Y, Rezaei H et al. . The diagnostic utility of fluorescence optical imaging in evaluating synovitis of the hands and wrists [abstract]. Ann Rheum Dis 2014;73:671 10.1136/annrheumdis-2014-eular.2484 [DOI] [Google Scholar]
- 22.Bremer C, Werner S, Langer HE. Assessing activity of rheumatic arthritis with fluorescence optical imaging. Eur Musculoskelet Rev 2010;4:96–100. [Google Scholar]
- 23.Fischer T, Ebert B, Voigt J et al. . Detection of rheumatoid arthritis using non-specific contrast enhanced fluorescence imaging. Acad Radiol 2010;17:375–81. 10.1016/j.acra.2009.09.016 [DOI] [PubMed] [Google Scholar]
- 24.Werner SG, Ohrndorf S, Langer HE et al. . Comparison of Xiralite with clinical examination and ultrasonography—Poster tours [abstract]. Ann Rheum Dis 2011;70(Suppl 3):361. [Google Scholar]
- 25.Werner SG, Lander HE, Ohrndorf S et al. . Inflammation assessment in patients with arthritis using a novel in vivo fluorescence optical imaging technology. Ann Rheum Dis 2012;71:504–10. 10.1136/annrheumdis-2010-148288 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Meier R, Thürmel K, Moog M et al. . Detection of synovitis in the hands of patients with rheumatologic disorders: diagnostic performance of optical imaging in comparison magnetic resonance imaging. Arthritis Rheum 2012;64:2489–98. 10.1002/art.34467 [DOI] [PubMed] [Google Scholar]
- 27.Hermann KGA, Ohrndorf S, Werner SG et al. . Imaging modalities in psoriatic arthritis. Z Rheumatol 2013;72:771–8. 10.1007/s00393-013-1188-8 [DOI] [PubMed] [Google Scholar]
- 28.Meier R, Thürmel K, Noël PB et al. . Synovitis in patients with early inflammatory arthritis monitored with quantitative analysis of dynamic contrast—enhanced optical imaging and MR imaging. Radiology 2014;270:176–85. 10.1148/radiol.13130039 [DOI] [PubMed] [Google Scholar]
- 29.Mohajerani P, Meier R, Noël PB et al. . Spatiotemporal analysis for indocyanine green-aided imaging of rheumatoid arthritis in hand joints. J Biomed Opt 2013;18:097004 10.1117/1.JBO.18.9.097004 [DOI] [PubMed] [Google Scholar]
- 30.Werner S, Langer HE, Scott P et al. . Indocyanine green enhanced fluorescence optical imaging in early and very early arthritis patients: a comparative study with magnetic resonance imaging. Arthritis Rheum 2013;65:3036–44. 10.1002/art.38175 [DOI] [PubMed] [Google Scholar]
- 31.Golov D, Meier R, Rummeny E et al. . Optical imaging of rheumatoid arthritis. Int J Clin Rheumatol 2011;6:67–75. 10.2217/ijr.10.105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Gompels LL, Lim NH, Vincent T et al. . In vivo optical imaging in arthritis—an enlightening future? Rheumatology (Oxford) 2010;49:1436–46. 10.1093/rheumatology/keq012 [DOI] [PubMed] [Google Scholar]
- 33.Meier R, Krug C, Golovko D et al. . Indocyanine green–enhanced imaging of antigen-induced FOI in early and very early arthritis 3043 arthritis with an integrated optical imaging/radiography system. Arthritis Rheum 2010;62:2322–7. 10.1002/art.27542 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Bahner ML, Werner SG, Cziumplik M et al. . Treatment monitoring of fluorescence optical imaging (FOI) using automated, computer based quantitative assessment. Ann Rheum Dis 2013; 72(Suppl 3):1012 10.1136/annrheumdis-2013-eular.3055 [DOI] [Google Scholar]
- 35.Ohrndorf S, Backhaus M. Advances in sonographic scoring of rheumatoid arthritis . Ann Rheum Dis 2013;72(Suppl 2):69–75. 10.1136/annrheumdis-2012-202197 [DOI] [PubMed] [Google Scholar]
- 36.Naredo E, D'Agostino MA, Wakefield RJ et al. . OMERACT Ultrasound Task Force. Ann Rheum Dis 2013;72:1328–34. 10.1136/annrheumdis-2012-202092 [DOI] [PubMed] [Google Scholar]
- 37.Smolen JS, Emery P, Fleischmann R et al. . Adjustment of therapy in rheumatoid arthritis on the basis of achievement of stable low disease activity with adalimumab plus methotrexate or methotrexate alone: the randomised controlled OPTIMA trial. Lancet 2014;383:321–32. 10.1016/S0140-6736(13)61751-1 [DOI] [PubMed] [Google Scholar]
- 38.Goekoop-Ruiterman YP, de Vries-Bouwstra JK, Allaart CF et al. . Clinical and radiographic outcomes of four different treatment strategies in patients with early rheumatoid arthritis (the BeSt study): a randomized, controlled trial. Arthritis Rheum 2005;52:3381–90. 10.1002/art.21405 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.