Abstract
Purpose
We investigated the relationships between biomarkers related to endoplasmic reticulum stress proteins (glucose-regulated protein of molecular mass 78 [GRP78] and Cripto-1 [teratocarcinoma-derived growth factor 1 protein]), pathologic response, and prognosis in locally advanced rectal cancer.
Materials and Methods
All clinical stage II and III rectal cancer patients received 50.4 Gy over 5.5 weeks, plus 5-fluorouracil (400 mg/m2/day) and leucovorin (20 mg/m2/day) bolus on days 1 to 5 and 29 to 33, and surgery was performed at 7 to 10 weeks after completion of all therapies. Expression of GRP78 and Cripto-1 proteins was determined by immunohistochemistry and was assessed in 101 patients with rectal cancer treated with neoadjuvant chemoradiotherapy (CRT).
Results
High expression of GRP78 and Cripto-1 proteins was observed in 86 patients (85.1%) and 49 patients (48.5%), respectively. Low expression of GRP78 protein was associated with a significantly high rate of down staging (80.0% vs. 52.3%, respectively; p=0.046) and a significantly low rate of recurrence (0% vs. 33.7%, respectively; p=0.008) compared with high expression of GRP78 protein. Mean recurrence-free survival according to GRP78 expression could not be estimated because the low expression group did not develop recurrence events but showed a significant correlation with time to recurrence, based on the log rank method (p=0.007). GRP78 also showed correlation with overall survival, based on the log rank method (p=0.045).
Conclusion
GRP78 expression is a predictive and prognostic factor for down staging, recurrence, and survival in rectal cancer patients treated with 5-fluorouracil and leucovorin neoadjuvant CRT.
Keywords: Rectal neoplasms, Molecular chaperone GRP78, TDGF1 protein, Endoplasmic reticulum stress
Introduction
Globally, colorectal cancer is one of the leading causes of cancer deaths and approximately one third of colorectal cancer arises from the rectum [1]. Currently, multi-modality treatment with chemotherapy, radiotherapy, and surgery has improved local control of patients with locally advanced rectal cancer (LARC), thus neoadjuvant chemoradiotherapy (CRT) followed by surgical resection has been the standard treatment of LARC [2]. Despite the optimal treatment, previous studies have reported conflicting data regarding survival benefit and high recurrence rate has been reported [3]. Thus, enhanced understanding of signaling pathway and therapeutic challenge is required.
Tumor microenvironment is characterized by hypoxia, hypoglycemia, and acidosis, which results in chronic endoplasmic reticulum (ER) stress. Glucose-regulated protein of molecular mass 78 (GRP78), a major ER chaperone, is a member of the family of heat shock proteins. Under ER stress, GRP78 activates a pro-survival pathway using an autophagy mechanism, which in turn leads to the protection and survival of cancer cells, chemoresistance and radioresistance [4-6]. Cripto-1 (teratocarcinoma-derived growth factor 1 protein), an extracellular, glycosylphosphatidylinositol-anchored protein, plays essential roles in embryogenesis, stem cell maintenance, and tumorigenesis [7]. Cripto-1 is overexpressed in many kinds of human cancer tissue while low or absent in normal adult tissue [7]. Previous studies have shown that Cripto-1 binds to cell surface GRP78 and inhibits transforming growth factor (TGF) signaling pathway in human tumor, thereby resulting in cellular proliferation and tumor growth [8,9].
Many previous studies examining response indicators for rectal cancer used data from several institutes and various treatment strategies. This multi-institutional bias and variation of treatment strategy may affect data analysis. A single-institution experience with homogenous treatment and pathologic evaluation excludes some bias. We also limited the number of enrolled patients who underwent laparoscopic surgery in order to make a homogenous cohort group.
Therefore, the aim of the current study is to determine the clinical significance of GRP78 and Cripto-1 expression in patients with rectal cancer who underwent neoadjuvant CRT followed by laparoscopic surgery.
Materials and Methods
1. Patients, specimens, and study designs
The study population consisted of 101 rectal cancer patients who underwent neoadjuvant CRT and laparoscopic surgery at St. Vincent’s Hospital between 2005 and 2008. The patients’ charts, radiographic findings, and postoperative pathologic slides were reviewed. Inclusion criteria were available archival paraffin-embedded tissue samples of initial colonoscopic biopsy, neoadjuvant CRT with 50.4 Gy (1.8 Gy/day in 28 fractions) over 5.5 weeks, plus 5-fluorouracil (400 mg/m2/day) and leucovorin (20 mg/m2/day) bolus on days 1 to 5 and 29 to 33, surgery performed at 7 to 10 weeks after completion of all therapies for all patients, and follow-up for at least 2 years for patients with clinical stage II-III rectal cancer. All patients had an initial work-up before CRT. Initial work-up included abdominal computed tomography (CT), chest CT, and positron emission tomography–CT, pelvis magnetic resonance imaging (MRI), and endorectal ultrasound. Initial clinical stage was decided comprehensively using these data. The patients also had a pre-surgical work-up including pelvis MRI and endorectal ultrasound. One surgical team, assisted by surgical residents in training, performed total mesorectal excision (TME) using laparoscopy. The quality of TME was classified as complete, near-complete, and incomplete. All patients had more than near-complete. Clinical stage and follow-up recurrence were performed in an independent review conducted by a radiologist, and pathologic stage, grade, and other pathological parameters were reviewed by two independent pathologists. Downstaging was defined as staging reduction from pretreatment stage (cStage) to pathologic staging (ypStage) (i.e., cIII to ypII, ypI or yp0; cII to ypI, or yp0). Pathologic response to CRT was reviewed and scored as follows: grade 0, no response; grade 1, necrosis or disappearance of tumor cells less than 2/3; grade 2, necrosis or disappearance of tumor cells more than 2/3; and grade 3, no viable cells (ypCR). We collected paraffin embedded tissue specimens from initial colonoscopic biopsies in these patients for immunohistochemistry staining.
2. Immunohistochemistry
Immunohistochemistry was performed using formalin fixed, paraffin-embedded tissue samples from initial colonoscopic biopsies to examine the expression of GRP78 and Cripto-1 proteins. The antibodies used in this study were GRP78 (Novus Biologicals, Littleton, CO) and Cripto-1 (Novus Biologicals). The tissue samples were deparaffinized in xylene and rehydrated in graded alcohols and water. Endogenous peroxidase was blocked by soaking in 3% H2O2 at 45°C for 4 minutes. The slides were microwaved in citrate buffer at 120°C for 15 minutes to unmask the antigen and treated with a protein-blocking reagent before incubation at 4°C overnight with primary antibodies at a 1:50 dilution, as recommended by the supplier. After extensive washing, the sections were incubated at room temperature for 10 minutes with Immpress anti-Rabbit immunoglobulin antibodies (Vector Laboratories Inc., Burlingame, CA) at a 1:20 dilution and subsequently with streptavidin-biotin peroxidase complexes at a 1:25 dilution. Reaction products were visualized by immersing the slides in 3,3′-diaminobenzidine tetrahydrochloride. Counterstaining was performed with hematoxylin. All series included positive and negative controls. The negative controls were prepared by omitting the primary antibodies, and known positive controls were included in each run. The results of immunohistochemical staining were evaluated independently by two pathologists in a blinded fashion. For the discordant samples, slides were reviewed jointly and a consensus was reached for the results. The results were scored according to staining intensity and staining percentage. Immunostaining intensity (a) was classified as lack of staining (0), weak staining (1), or moderate to strong staining (2); the percentage of positive tumor cells (b) was semiquantitatively divided according to three grades: no positive staining or positive staining in < 10% of tumor cells (0), 10%-50% staining (1), and > 50% staining (2). The score for each section was measured as (a)×(b), the result was defined as 0 (score 0), 1+ (score 1), 2+ (score 2), or 3+ (score 4). For statistical analysis, the expression was grouped according to low (0, 1+) or high (2+, 3+) level expression.
3. Statistical analysis
IBM SPSS ver. 21.0 (IBM Co., Armonk, NY) was used for statistical analysis. To evaluate the association between GRP78 and Cripto-1 expression and clinicopathologic factors including recurrence, correlation analysis was performed using Pearson chi-square or Fisher exact test as appropriate. Recurrence-free survival (RFS) was defined as the time from the date of operation to relapse or death for patients who died without relapse. Overall survival (OS) was defined as the time between the date of operation and death. RFS and OS were calculated using the Kaplan-Meier method. Simple variable analysis was performed using the log rank method and multivariate analysis using the Cox regression model. Survival rates and odds ratios are presented with their 95% confidence intervals and p-value of < 0.05 was considered statistically significant.
Results
1. Characteristics and pathological response of patients
This study included 69 male and 32 female patients with a median age of 62 years (range, 53 to 69 years). Clinical stages II and III accounted for 51.5% and 48.5% in each group. The down-staging rate of this neoadjuvant CRT was 56.4% (57 patients) and ypCR was 19.8% (20 patients). Down-staging after neoadjuvant CRT presented 52 cII patients to 17 ypI and 9 yp0, and 49 cIII patients to 15 ypII, 5 ypI and 11 yp0. At a median follow-up period of 51 months (range, 38 to 64 months), 81 patients (80.2%) were alive, and two cases were non-cancer-related deaths. Overall recurrence rate was 31.7% (32 patients). Eight patients (7.9%) had loco-regional recurrence, and the rest of patients with recurrence had distant recurrence only. The RFS and OS did not reach the median. Three-year RFS was 66.3% and 3-year OS was 78.2%. Patients’ characteristics and their response to neoadjuvant CRT are shown in Tables 1 and 2.
Table 1.
Patients' characteristics
| Characteristic | No. of patients (n=101) |
|---|---|
| Age (yr) | 62 (53-69) |
| Gender (malefemale) | 69:32 |
| ECOG PS (1:0) | 45:56 |
| Differentiation | |
| Well | 18 |
| Moderate | 76 |
| Poor | 7 |
| CEA (ng/mL) | 3.48 (1.83-6.87) |
| LDH (IU/L) | 285.0 (235.0-321.5) |
Values are presented as median (range) or number. ECOGPS, Eastern Cooperative Oncology Group performance status; CEA, carcinoembryonic antigen; LDH, lactate dehydrogenase.
Table 2.
Treatment response of neoadjuvant chemoradiotherapy in patients with locally advanced rectal cancer
| Variable | Clinical stage | Pathologic stage | |
|---|---|---|---|
| T stage | |||
| 0 | 0 | 20 (19.8) | |
| 1 | 0 | 5 (5.0) | |
| 2 | 2 (2.0) | 23 (22.8) | |
| 3 | 96 (95.0) | 50 (49.5) | |
| 4 | 3 (3.0) | 3 (3.0) | |
| N stage | |||
| 0 | 52 (51.5) | 71 (70.3) | |
| 1 | 32 (31.7) | 24 (23.8) | |
| 2 | 17 (16.8) | 6 (5.9) | |
| TNM stage | |||
| 0 | 0 | 20 (19.8) | |
| I | 0 | 22 (21.8) | |
| II | 52 (51.5) | 29 (28.7) | |
| III | 49 (48.5) | 30 (29.7) | |
| Pathologic response | |||
| Grade 0 | 19 (18.8) | ||
| Grade 1 | 33 (32.7) | ||
| Grade 2 | 29 (28.7) | ||
| Grade 3 | 20 (19.8) | ||
Values are presented as number (%).
2. GRP78 and Cripto-1 protein expression in pretreatmentbiopsy specimens
High expression of GRP78 and Cripto-1 proteins was observed in 86 patients (85.1%) and 49 patients (48.5%), respectively. Expression of GRP78 and Cripto-1 proteins is shown in Table 3 and Fig. 1. Expression of GRP78 and Cripto-1 did not show significant correlation (p=0.686).
Table 3.
Immunohistochemistry results of endoplasmic reticulum stress-related proteins
| Variable | Intensity (a) | Portion (b) | Score (axb) |
|---|---|---|---|
| GRP78 | |||
| 0 | 0 | 0 | - |
| 1 | 15 (14.9) | 16 (15.8) | 15 (14.9) |
| 2 | 86 (85.1) | 85 (84.2) | 1 (1.0) |
| 4 | 85 (84.2) | ||
| Cripto-1 | |||
| 0 | 32 (31.7) | 32 (31.7) | 32 (31.7) |
| 1 | 31 (30.7) | 30 (29.7) | 20 (19.8) |
| 2 | 38 (37.6) | 39 (38.6) | 21 (20.8) |
| 4 | - | - | 28 (27.7) |
Values are presented as number (%). GRP78, glucose-regulated protein of molecular mass 78.
Fig. 1.
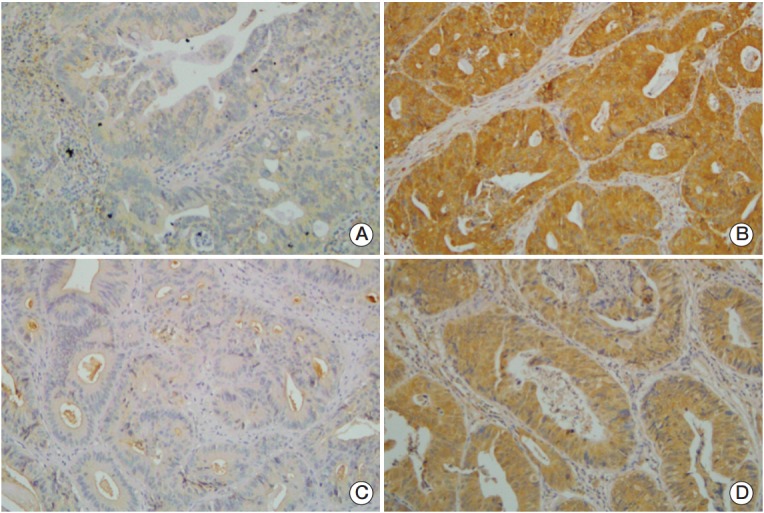
These are representative tissue sections used in immunohistochemical analyses. (A) Low expression of glucose-regulated protein of molecular mass 78 (GRP78, ×400). (B) High cytoplasmic expression of GRP78 (×400). (C) Low expression of Cripto-1 (×400). (D) High cytoplasmic expression of Cripto-1 (×400).
3. Association between expression of these biomarkers and clinical parameters
Associations between expression of these biomarkers and clinical parameters are shown in Table 4. GRP78 and Cripto-1 protein expressions did show significant association with clinical parameters.
Table 4.
Correlation between expressions of endoplasmic reticulum stress-related proteins and clinicopathologic factors
| Variable | No. | GRP78 |
Cripto-1 |
||||
|---|---|---|---|---|---|---|---|
| Low | High | p-value | Low | High | p-value | ||
| Age (yr) | 0.431 | 0.292 | |||||
| ≤ 65 | 65 | 11 | 54 | 36 | 29 | ||
| > 65 | 36 | 4 | 32 | 16 | 20 | ||
| Histologic differentiation | 0.623 | 0.317 | |||||
| Well | 18 | 4 | 14 | 11 | 7 | ||
| Moderate | 76 | 10 | 66 | 36 | 40 | ||
| Poor | 7 | 1 | 6 | 5 | 2 | ||
| Clinical T stage | 0.289 | 0.193 | |||||
| 2 | 2 | 1 | 1 | 1 | 1 | ||
| 3 | 96 | 14 | 82 | 51 | 45 | ||
| 4 | 3 | 0 | 3 | 0 | 3 | ||
| Clinical N stage | 0.905 | 0.375 | |||||
| 0 | 52 | 7 | 45 | 27 | 25 | ||
| 1 | 32 | 5 | 27 | 14 | 18 | ||
| 2 | 17 | 3 | 14 | 11 | 6 | ||
| cTNM stage | 0.686 | 0.928 | |||||
| II | 52 | 7 | 45 | 27 | 25 | ||
| III | 49 | 8 | 41 | 25 | 24 | ||
GRP78, glucose-regulated protein of molecular mass 78; cTNM stage, clinical tumor, node, metastasis stage.
4. Correlations between biomarkers and treatment response to neoadjuvant CRT
Table 5 shows the correlations between biomarker expressions and responses of neoadjuvant CRT. GRP78 was the biomarker shown to be related to chemoradiosensitivity. Low expression of GRP78 protein was associated with a significantly high rate of down staging compared with high expression of GRP78 protein (80.0% vs. 52.3%, respectively; p=0.046) (Fig. 2A). Expression of Cripto-1 protein did not show correlation with down staging or ypCR of CRT.
Table 5.
Responses and recurrence after neoadjuvant chemoradiotherapy according to expressions of endoplasmic reticulum stress-related proteins
| Variable | GRP78 expression |
Cripto-1 expression |
||||
|---|---|---|---|---|---|---|
| Low (n=15) | High (n=86) | p-value | Low (n=52) High (n=49) | p-value | ||
| Pathologic response | 0.406 | 0.809 | ||||
| Grade 0 | 2 | 17 | 11 | 8 | ||
| Grade 1 | 3 | 30 | 18 | 15 | ||
| Grade 2 | 5 | 24 | 13 | 16 | ||
| Grade 3 | 5 | 15 | 10 | 10 | ||
| Down staging (%) | 80 | 52.3 | 0.046 | 53.8 | 59.2 | 0.589 |
| ypCR rate (%) | 33.3 | 17.4 | 0.154 | 19.2 | 20.4 | 0.882 |
| Recurrence rate (%) | 0 | 37.2 | 0.004 | 32.7 | 30.6 | 0.882 |
GRP78, glucose-regulated protein of molecular mass 78; ypCR, pathologic complete response.
Fig. 2.
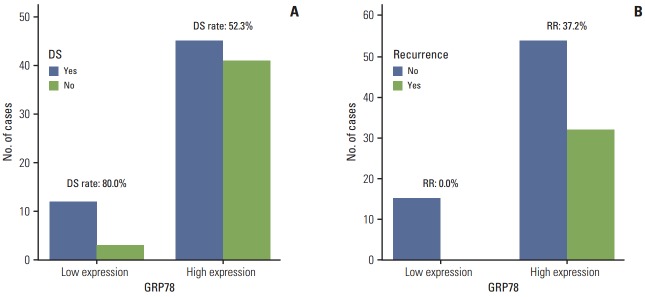
(A) Down staging (DS) rate according to expression of glucose-regulated protein of molecular mass 78 (GRP78) protein (low expression of GRP78 [80.0%] vs. high expression of GRP78 [52.3%], respectively; p=0.046). (B) Recurrence rate (RR) according to expression of GRP78 protein (low expression of GRP78 [0%] vs. high expression of GRP78 [37.2%], respectively; p=0.004).
5. Association between tumor recurrence and biomarkers
Table 5 and Fig. 2B show the correlations between biomarker expressions and recurrence. Three-year RFS rate was 100% in the low expression group of GRP78 protein and 60.5% in the high expression group of GRP78 protein. Low expression of GRP78 protein was associated with a significantly low rate of recurrence compared with high expression of GRP78 protein (0% vs. 33.7%, respectively; p=0.008). None of the patients with low GRP78 expression showed recurrence. RFS in the patients after curative surgery in relation to down staging, ypCR, and the protein expressions of GRP78 and Cripto-1 are shown in Table 6. Mean RFS according to GRP78 expression could not be estimated because the low expression group did not develop recurrence events but showed a significant correlation with recurrence rate, based on the log-rank method (p=0.007) (Fig. 3). And expression of Cripto-1 protein did not show significant correlation with RFS, based on the log-rank method (p=0.457). Clinical parameters including cStage, down-staging, and ypCR showed significant correlation with RFS. Initial carcinoembryonic antigen and lactate dehydrogenase levels also did not show statistically significant correlation with tumor recurrence. For the clinical parameters except GRP78, multivariate analysis for time to recurrence showed two independent prognostic factors, clinical stage and down stage (0.006 and 0.002). As shown in Table 6, GRP78 showed correlation with OS, based on the log-rank method (p=0.045). cStage also showed correlation with OS (p=0.023).
Table 6.
Univariate analysis for several factors related to recurrence and survival
| Variable | No. | Relapse free survival (mo) |
Overall survival (mo) |
|||
|---|---|---|---|---|---|---|
| Relapse free survival (mo) | p-value | Mean (95% CI) | p-value | |||
| Age (yr) | ≤ 65 | 65 | 56.1 (49.4-62.7) | 0.613 | 75.1 (69.7-80.5) | 0.16 |
| > 65 | 36 | 53.5 (43.9-63.1) | 64.1 (56.3-71.9) | |||
| Clinical stage | II | 52 | 60.5 (53.8-67.3) | 0.046 | 78.0 (72.6-83.5) | 0.023 |
| III | 49 | 48.1 (39.9-56.3) | 67.0 (59.8-74.1) | |||
| Down staging | Yes | 57 | 64.4 (58.5-70.2) | 0 | 75.7 (70.6-80.8) | 0.072 |
| No | 44 | 43.1 (34.2-52.0) | 68.3 (60.4-76.2) | |||
| Pathologic complete response | Yes | 20 | 68.4 (60.9-75.9) | 0.031 | 77.5 (70.2-84.8) | 0.261 |
| No | 81 | 51.8 (45.5-58.2) | 71.5 (66.1-77.0) | |||
| GRP78 | Low | 15 | Not estimate | 0.007 | Not estimate | 0.045 |
| High | 86 | Not estimate | Not estimate | |||
| Cripto-1 | Low | 52 | 51.7 (44.1-59.2) | 0.737 | 73.0 (66.2-79.9) | 0.860 |
| High | 49 | 56.3 (48.7-64.0) | 67.5 (62.2-72.8) | |||
| Carcinoembryonic antigen (ng/mL) | ≤ 5.0 | 67 | 53.0 (46.5-59.5) | 0.978 | 71.6 (65.6-77.6) | 0.472 |
| > 5.0 | 34 | 55.1 (45.8-64.4) | 74.5 (67.5-81.4) | |||
| Lactate dehydrogenase (IU/L) | ≤ 300 | 61 | 53.8 (46.5-61.1) | 0.663 | 70.0 (63.4-76.7) | 0.166 |
| > 300 | 40 | 56.8 (48.6-65.1) | 71.7 (66.4-76.9) | |||
CI, confidence interval; GRP78, glucose-regulated protein of molecular mass 78.
Fig. 3.
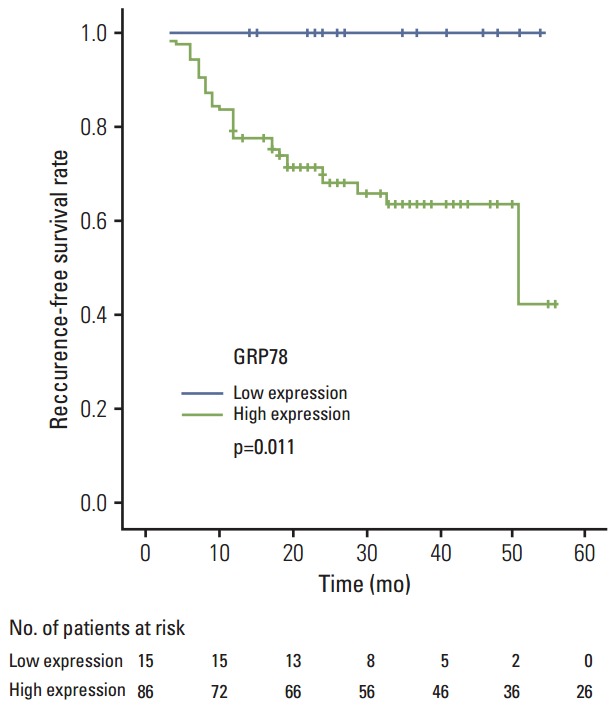
Kaplan-Meier survival curves are calculated using expression patterns of glucose-regulated protein of molecular mass 78 (GRP78) for recurrence-free survival.
Discussion
The current study attempted to evaluate the clinical significance of GRP78 and Cripto-1 expression in patients with LARC who underwent neoadjuvant CRT followed by laparoscopic surgery. Our results suggested the predictive and prognostic role of GRP78. In patients with low expression of GRP78, higher response rate (down staging) and prolonged survival were demonstrated and no recurrence was observed.
Emerging data have shown the relationship of ER stress and human cancer, in terms of tumorigenesis, aggressiveness, and resistance to anti-tumor treatment including chemotherapy and radiotherapy [4,5]. Increased expression of GRP78 is a part of coordinated protein response in order to release ER stress, thereby resulting in maintenance of ER function and prevention of cell death. However, few studies have reported on GRP78 in rectal cancer. Low GRP78 expression was reported to show correlation with tumor regression in LARC patients who underwent neoadjuvant CRT [10]. A recent study suggested GRP78 as a therapeutic target in colorectal cancer. The study demonstrated that a fusion protein containing GRP binding peptide inhibited growth and induced apoptosis in colorectal cancer cells and the antitumor activity was shown as well in a xenograft model [11]. Our results showed low expression of GRP78 protein in rectal cancer and its correlation with down staging and OS. Therefore GRP78 protein is a possible predictive marker of neoadjuvant CRT and a prognostic marker of LARC patients.
Cripto-1, a cell surface protein, plays an important role during embryogenesis and disappears in adult tissues, except in stem cells. Cripto-1 reappears in tumorous conditions, which promotes cell proliferation, epithelial to mesenchymal transition, migration, invasion, and angiogenesis. Cripto-1 interacts with an extensive range of signaling molecules, including TGF, Wnt/β-catenin, erbB4, Notch, and GRP78, which accounts for its diverse biological effects [7,12]. In the interaction between GRP78 and Cripto-1, Cripto-1 signaling in human cancer necessitates GRP78 and Cripto-GRP78 complex inhibits TGF signaling, which has important functions during development and tumorigenesis [8,9]. Furthermore, cells with coexpression of GRP78 and Cripto-1 grow more rapidly compared to cells with expression of either protein [9]. Few research studies on Cripto-1 in rectal cancer have been reported as well. A previous study reported that Cripto-1 was expressed in 71% of primary or metastatic colorectal cancers whereas in only 2%-7% of normal colonic mucosa [13]. Cripto-1 expression was reported in premalignant lesions such as colon adenoma, suggesting its role in tumorigenesis [14]. In our study, 48.5% of patients showed high expression of Cripto-1. Its correlation with tumor invasiveness and metastasis has been reported in some cancers [15,16]. GRP78 is induced by physiologic stress, which perturbs ER function and homeostasis, protecting against pathologic conditions such as hypoxia and glycolytic condition [17]. Tumor microenvironment characterized by glucose deprivation and severe hypoxia leads to the ER stress condition. All tumor tissue including stromal cells as well as cancer cells was affected by ER stress condition. Therefore it is difficult to interpret the expression of GRP78 because GRP78 was expressed generally and strongly in tumor tissues. We had to carefully evaluate the expression of GRP78. Under the condition of low GRP78 expression, the environment of tumor tissue is less hypoxic and glycolytic. Consequently tumor of low GRP78 expression is more radiosensitive. We already reported that GLUT1 expression as a hypoxic biomarker is a predictive and prognostic factor for pathologic complete response and recurrence in rectal cancer patients [18]. We thought that GRP78 is also a hypoxia related biomarker.
This current study has limitations. Preoperative biopsied tissue was used in our study and there is some risk in regarding biopsied tissue as a representative sample for the whole tumor. Median RFS has not yet been reached, thus, further follow up and a large number of study subjects is required in order to properly assess the prognostic roles of GRP78 and Cripto-1.
In conclusion, this current study indicates that GRP78 and Cripto-1 were highly expressed in the majority of LARC patients and high expression of GRP78 was related to poor response and poor prognosis; this suggests correlation of GRP78 with chemoresistance and radioresistance. So far in rectal cancer, little is known about GRP78 and Cripto-1 and predictive molecular markers in CRT remain elusive. Prediction of tumor response is important in selection of promising candidates for preoperative CRT and candidates who need additional adjuvant therapy after surgery. GRP78 is an important prognostic marker due to correlation with OS. Although further studies are needed, including further follow-up, standardized validation, and in vitro/in vivo research, this study might improve our insight regarding molecular signaling pathway in rectal cancer and predictive markers in terms of CRT.
Conclusion
GRP78 expression is a predictive and prognostic factor for down staging, recurrence, and OS in rectal cancer patients treated with 5-flurouracil and leucovorin neoadjuvant CRT.
Acknowledgments
The authors wish to acknowledge the financial support of the Catholic Medical Center Research Foundation made in the program year of 2013.
Footnotes
Conflict of interest relevant to this article was not reported.
References
- 1.Naishadham D, Lansdorp-Vogelaar I, Siegel R, Cokkinides V, Jemal A. State disparities in colorectal cancer mortality patterns in the United States. Cancer Epidemiol Biomarkers Prev. 2011;20:1296–302. doi: 10.1158/1055-9965.EPI-11-0250. [DOI] [PubMed] [Google Scholar]
- 2.Kockerling F, Reymond MA, Altendorf-Hofmann A, Dworak O, Hohenberger W. Influence of surgery on metachronous distant metastases and survival in rectal cancer. J Clin Oncol. 1998;16:324–9. doi: 10.1200/JCO.1998.16.1.324. [DOI] [PubMed] [Google Scholar]
- 3.De Caluwe L, Van Nieuwenhove Y, Altendorf-Hofmann A, Ceelen WP. Preoperative chemoradiation versus radiation alone for stage II and III resectable rectal cancer. Cochrane Database Syst Rev. 2013;2: doi: 10.1002/14651858.CD006041.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Lee AS. GRP78 induction in cancer: therapeutic and prognostic implications. Cancer Res. 2007;67:3496–9. doi: 10.1158/0008-5472.CAN-07-0325. [DOI] [PubMed] [Google Scholar]
- 5.Pyrko P, Schonthal AH, Hofman FM, Chen TC, Lee AS. The unfolded protein response regulator GRP78/BiP as a novel target for increasing chemosensitivity in malignant gliomas. Cancer Res. 2007;67:9809–16. doi: 10.1158/0008-5472.CAN-07-0625. [DOI] [PubMed] [Google Scholar]
- 6.Feng XP, Yi H, Li MY, Li XH, Yi B, Zhang PF, et al. Identification of biomarkers for predicting nasopharyngeal carcinoma response to radiotherapy by proteomics. Cancer Res. 2010;70:3450–62. doi: 10.1158/0008-5472.CAN-09-4099. [DOI] [PubMed] [Google Scholar]
- 7.Strizzi L, Bianco C, Normanno N, Salomon D. Cripto-1: a multifunctional modulator during embryogenesis and oncogenesis. Oncogene. 2005;24:5731–41. doi: 10.1038/sj.onc.1208918. [DOI] [PubMed] [Google Scholar]
- 8.Kelber JA, Panopoulos AD, Shani G, Booker EC, Belmonte JC, Vale WW, et al. Blockade of Cripto binding to cell surface GRP78 inhibits oncogenic Cripto signaling via MAPK/PI3K and Smad2/3 pathways. Oncogene. 2009;28:2324–36. doi: 10.1038/onc.2009.97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Shani G, Fischer WH, Justice NJ, Kelber JA, Vale W, Gray PC. GRP78 and Cripto form a complex at the cell surface and collaborate to inhibit transforming growth factor beta signaling and enhance cell growth. Mol Cell Biol. 2008;28:666–77. doi: 10.1128/MCB.01716-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kikuchi M, Mikami T, Sato T, Tokuyama W, Araki K, Watanabe M, et al. High Ki67, Bax, and thymidylate synthase expression well correlates with response to chemoradiation therapy in locally advanced rectal cancers: proposal of a logistic model for prediction. Br J Cancer. 2009;101:116–23. doi: 10.1038/sj.bjc.6605105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Li Z, Zhao C, Li Z, Zhao Y, Shan S, Shi T, et al. Reconstructed mung bean trypsin inhibitor targeting cell surface GRP78 induces apoptosis and inhibits tumor growth in colorectal cancer. Int J Biochem Cell Biol. 2014;47:68–75. doi: 10.1016/j.biocel.2013.11.022. [DOI] [PubMed] [Google Scholar]
- 12.Nagaoka T, Karasawa H, Castro NP, Rangel MC, Salomon DS, Bianco C. An evolving web of signaling networks regulated by Cripto-1. Growth Factors. 2012;30:13–21. doi: 10.3109/08977194.2011.641962. [DOI] [PubMed] [Google Scholar]
- 13.Ciardiello F, Kim N, Saeki T, Dono R, Persico MG, Plowman GD, et al. Differential expression of epidermal growth factor-related proteins in human colorectal tumors. Proc Natl Acad Sci U S A. 1991;88:7792–6. doi: 10.1073/pnas.88.17.7792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Saeki T, Stromberg K, Qi CF, Gullick WJ, Tahara E, Normanno N, et al. Differential immunohistochemical detection of amphiregulin and cripto in human normal colon and colorectal tumors. Cancer Res. 1992;52:3467–73. [PubMed] [Google Scholar]
- 15.Ertoy D, Ayhan A, Sarac E, Karaagaoglu E, Yasui W, Tahara E, et al. Clinicopathological implication of cripto expression in early stage invasive cervical carcinomas. Eur J Cancer. 2000;36:1002–7. doi: 10.1016/s0959-8049(00)00033-2. [DOI] [PubMed] [Google Scholar]
- 16.Shike M, Winawer SJ, Greenwald PH, Bloch A, Hill MJ, Swaroop SV. Primary prevention of colorectal cancer: the WHO Collaborating Centre for the prevention of colorectal cancer. Bull World Health Organ. 1990;68:377–85. [PMC free article] [PubMed] [Google Scholar]
- 17.Lee AS. The glucose-regulated proteins: stress induction and clinical applications. Trends Biochem Sci. 2001;26:504–10. doi: 10.1016/s0968-0004(01)01908-9. [DOI] [PubMed] [Google Scholar]
- 18.Shim BY, Jung JH, Lee KM, Kim HJ, Hong SH, Kim SH, et al. Glucose transporter 1 (GLUT1) of anaerobic glycolysis as predictive and prognostic values in neoadjuvant chemoradiotherapy and laparoscopic surgery for locally advanced rectal cancer. Int J Colorectal Dis. 2013;28:375–83. doi: 10.1007/s00384-012-1542-3. [DOI] [PubMed] [Google Scholar]


