Introduction
Venous thromboembolism (VTE) is the third cause of morbidity and mortality in western countries with an incidence rate of 1–3 per 1,000 individuals/year1. VTE has a multifactorial aetiology in which a genetic predisposition related to one or more thrombophilic traits interacts with acquired conditions and circumstantial risk factors2.
Major transient risk factors for VTE are recent surgery or trauma, fracture, pregnancy or puerperium, prolonged immobilisation, active cancer, and severe medical diseases. Oral contraceptive use and long distance travel have also been associated with an increased risk of VTE. VTE is usually defined as “provoked” when it occurs in the presence of one of more of these conditions3. However, in 25–50% of cases VTE occurs in the absence of any triggering factor and, in this case, it is classified as “unprovoked” VTE4.
The unprovoked nature of a VTE has been found to be an independent and strong predictor of recurrence5, and some evidence suggests the need for prolonged antithrombotic treatment in patients with unprovoked VTE6.
In recent years, new direct oral anticoagulants (DOAC) have been developed, including factor IIa and FXa inhibitors and several trials in patients with acute VTE showed a comparable efficacy in the prevention of VTE recurrence and a significantly lower bleeding risk in patients receiving DOAC compared to those receiving a vitamin K antagonist (VKA)7.
Unfortunately information on the efficacy and safety of DOAC in patients with unprovoked or provoked VTE is still limited since, to date, no study specifically enrolling patients according to the nature of VTE has been published. However, some studies have provided separate data for patients with unprovoked and provoked VTE. Thus, the aim of this study was to perform a systematic review and meta-analysis of literature data on this topic.
Material and methods
We carried out a systematic literature search of randomised controlled trials comparing DOAC with standard VKA treatment in the management of patients with acute VTE. Medline, ISI Web of Science, SCOPUS, Cochrane database and EMBASE databases were searched up to April 2014. Research was supplemented by manually reviewing abstract books from congresses of the International Society on Thrombosis and Haemostasis (ISTH) and the American Society of Hematology (ASH) (2003–2013) and the reference lists of all retrieved articles. Only studies providing data on safety and/or efficacy outcome separately for patients with unprovoked or provoked VTE were included. The efficacy outcome was defined by the prevention of recurrent VTE or VTE-related death. The safety outcome was represented by the occurrence of major or clinically relevant non-major bleeding. Search results were reported according to Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) guidelines8. Data about the study (year of publication, study type) and patients’ characteristics (number of subjects studied, mean age, gender) were extracted from each randomised controlled trial. Any discrepancies between reviewers were resolved by discussion or by the opinion of a third reviewer, as necessary. Formal quality score adjudication was not used, since previous investigations failed to demonstrate its usefulness9. Statistical analysis was performed using Review Manager (Version 5.2, The Cochrane Collaboration, Copenhagen, Denmark) provided by The Cochrane Collaboration. Pooled risk ratios (RR) and 95% confidence intervals (CI) were calculated using a random effects model. Statistical heterogeneity was evaluated through the use of Cochran’s Q and of I2 statistics. The presence of publication bias was explored using funnel plots of effect size against standard error. Visual inspection for funnel plot asymmetry was performed to look for a possible small-study effect10. We also performed sub-analyses of results according to the type of DOAC used (anti-FIIa or anti-FXa) and according to the duration of the treatment (6 months vs 12 months).
Results
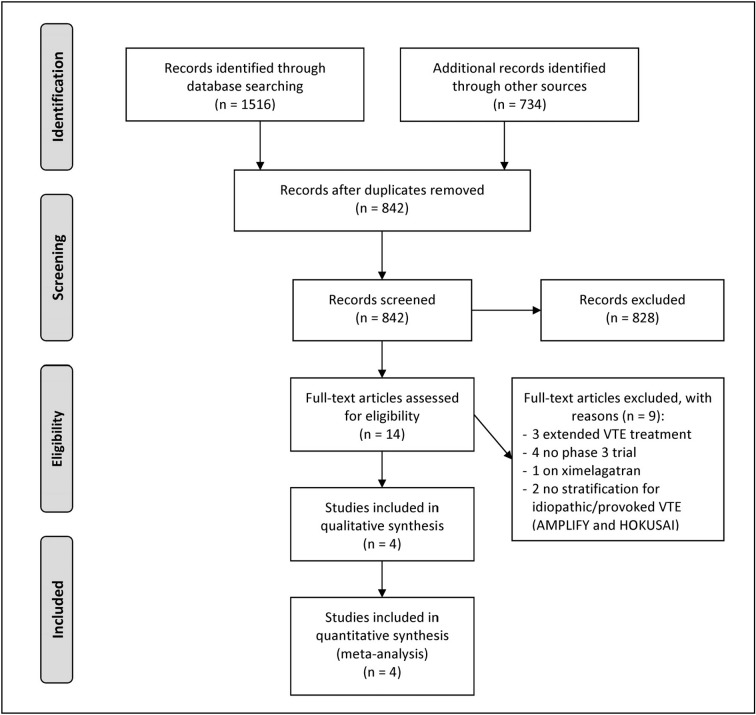
The search provided 842 results, of which 828 were excluded because they were reviews, case-reports or judged off the topic after scanning the title and/or the abstract. Another nine studies were excluded after evaluation of the full-length paper (Figure 1).
Figure 1.
PRISMA flow diagram.
Thus, five randomised controlled trials11–15 including a total of 21,628 patients (10,821 receiving DOAC and 10,807 VKA) with acute VTE were included in the analysis. Of these, two studies11,12 evaluated dabigatran as the experimental drug, two rivaroxaban13,14 and one edoxaban15.
Study characteristics
The principal characteristics of the studies included in this analysis are shown in Table I.
Table I.
Characteristics of the studies included.
| Study | Drug | Dosage | Duration | Study population | Age (mean) | Males | N. (%) of patients with unprovoked VTE |
|---|---|---|---|---|---|---|---|
| RECOVER I & II11,12 | Dabigratan (anti-FIIa) | 150 mg/BID | 6 months | DOAC group: 2,553 VKA group: 2,554 |
54 years | 59% | DOAC group: 893 (34.9%) VKA group: 924 (36.2%) |
| EINSTEIN-DVT13 | Rivaroxaban (anti-FXa) | 15 mg/BID (3 weeks) + 20 mg/OD | 12 months | DOAC group: 1,731 VKA group: 1,718 |
56 years | 57% | DOAC group: 1,055 (60.9%) VKA group: 1,083 (63.0%) |
| EINSTEIN-PE14 | Rivaroxaban (anti-FXa) | 15 mg/BID (3 weeks) + 20 mg/OD | 12 months | DOAC group: 2,419 VKA group: 2,413 |
58 years | 53% | DOAC group: 1,566 (64.7%) VKA group: 1,551 (64.3%) |
| HOKUSAI 201315 | Edoxaban (anti-FXa) | 60 mg/OD | 12 months | DOAC group: 4,118 VKA group: 4,122 |
56 years | 57% | DOAC group: 2713 (65.9%) VKA group: 2697 (65.4%) |
BID: twice daily; OD: once daily; DOAC: direct oral anticoagulant; VKA: Vitamin K antagonist.
Of note, in the RECOVER-II study the stratification according to the nature of VTE (unprovoked vs provoked) was provided in the frame of a pooled analysis including data from the RECOVER-I study. Thus, these two studies have been analysed as a unique dataset.
The number of patients varied from 3,449 to 8,240, the mean age from 54 to 58 years, the prevalence of male gender from 53% to 59%, the treatment duration from 6 to 12 months and the prevalence of unprovoked VTE from 35.6% to 65.6%. Overall, a total of 12,482 out of 21,628 enrolled patients (57.7%) had an unprovoked VTE at the time of the randomisation and the prevalence of patients with unprovoked VTE was similar in the DOAC and VKA groups (57.5% vs 57.9%, P=0.61).
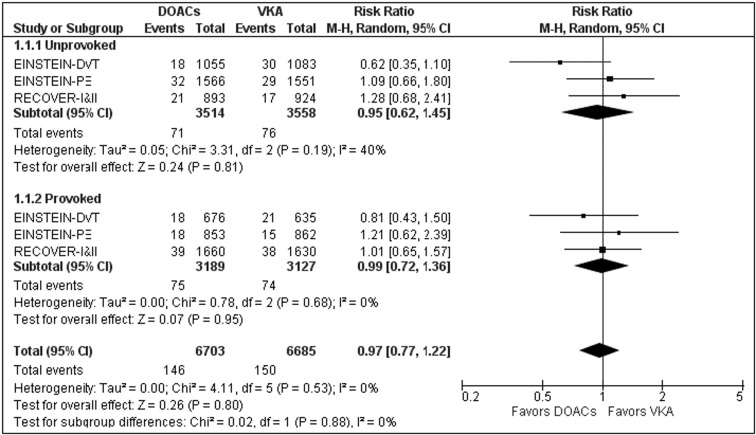
All five studies11–15 provided efficacy outcome data stratified according to the nature of VTE (unprovoked vs provoked) and showed that DOAC had a similar efficacy to that of VKA in both unprovoked and provoked VTE. As shown in Figure 2, among patients with unprovoked VTE, recurrent VTE/VTE-related death occurred in 2.6% of patients receiving DOAC and in 2.8% of those receiving VKA (RR: 0.93, 95% CI: 0.74–1.17, P=0.51, I2: 11%, P=0.34). Similarly, among patients with a provoked VTE, recurrent VTE/VTE-related death occurred in 2.5% of patients receiving DOAC and in 2.6% of those receiving VKA (RR: 0.94, 95% CI: 0.72–1.22, P=0.65, I2: 0%, P=0.79). Similar results were obtained when the analyses were stratified according to the type of DOAC (anti-FIIa or anti-FXa) and to the duration of anticoagulant treatment (Table II).
Figure 2.
Forest plot of the primary efficacy outcome (recurrent VTE or VTE-related death) for DOAC vs VKA in patients with unprovoked (Panel A) or provoked (Panel B) VTE.
DOAC: direct oral anticoagulant; VKA: Vitamin K antagonist; M-H: Mantel-Haenszel effect; CI: confidence interval.
Table II.
Risk ratio of recurrent venous thromboembolism (VTE) or death related to VTE in patients with unprovoked or provoked VTE receiving direct oral anticoagulants (DOAC) or vitamin K antagonists stratified according to the type of DOAC (anti-FIIa or anti-FXa).
| Type of drug | Unprovoked VTE | Provoked VTE | ||||
|---|---|---|---|---|---|---|
|
| ||||||
| N. of studies | Population | RR (95% CI) | N. of studies | Population | RR (95% CI) | |
| Anti-FIIa (dabigatran) | 2 | DOAC group: 893 VKA group: 924 |
1.28 (0.68–2.41) | 2 | DOAC group: 1,660x VKA group: 924 |
1.01 (0.65–1.57) |
| Anti-FXa (rivaroxaban and edoxaban) | 3 | DOAC group: 5,607 VKA group: 5,616 |
0.89 (0.70–1.13) | 3 | DOAC group: 2,661 VKA group: 2,637 |
0.91 (0.66–1.26) |
DOAC: direct oral anticoagulant; VKA: vitamin K antagonist; RR: risk ratio; CI: confidence interval.
NOTE: Treatment lasted 6 months in the two studies on dabigatran, whereas it lasted up to 12 months in the two studies on rivaroxaban and in the study on edoxaban. Thus, the sub-group analysis according to treatment duration provided exactly the same results as those reported in the table above.
Only one study15 provided data on the safety outcome stratified according to the nature of VTE (unprovoked vs provoked) and showed that the rate of major or clinically relevant non-major bleeding was similar in patients with provoked VTE treated with edoxaban or VKA (9.7% vs 10.9%; RR: 0.87, 95% CI: 0.67–1.15). In contrast, among the patients with unprovoked VTE, a lower bleeding rate was reported in patients treated with edoxaban than in patients treated with VKA (8.0% vs 10.0%; RR: 0.78, 95% CI: 0.66–0.94).
Given the low number of included studies, funnel plot analysis could not be used to evaluate the publication bias and meta-regression analysis could not be performed to adjust for potential confounders.
Discussion
Although data from a number of high-quality, randomised controlled trials and from large meta-analyses suggested that DOAC and VKA have similar efficacy in the treatment of acute VTE7 this is the first study, to the best of our knowledge, providing separate analyses according to the nature of VTE (unprovoked vs provoked). The study shows that DOAC have a similar efficacy as VKA in both these two groups of VTE patients.
Patients with unprovoked or provoked VTE have different characteristics with the former group having a higher mean age and a higher proportion of males. Furthermore these patients have a different risk of VTE recurrence. These findings confirm the different nature of unprovoked and provoked VTE. Thus, data on the efficacy and safety of DOAC in these two groups of patients are warranted.
Unfortunately, no study specifically assessing the efficacy of DOAC in the treatment of unprovoked VTE has been published to date, and so the results of the present meta-analysis are the only available data on this issue. However, few data about safety were reported in the randomised controlled trials included in this analysis and we were not, therefore, able to perform a risk-benefit assessment on the use of DOAC in patients with unprovoked or provoked VTE.
Our findings may have some important clinical implications. Consistent data suggest that the unprovoked nature of VTE is a strong predictor of recurrence5. Results of the ELATE and PREVENT trials highlighted the importance of considering unprovoked VTE as a chronic disease and suggested the benefit of a more intensive and prolonged anticoagulant treatment in this clinical setting6. Results of our meta-analysis suggest that DOAC have a similar efficacy in patients with unprovoked or provoked events. Furthermore, three large, randomised controlled trials and a recent meta-analysis suggested that DOAC were effective for the extended treatment of VTE and may reduce the risk of all-cause mortality without increasing the risk of major bleeding complications. Thus, patients with an unprovoked VTE may benefit from treatment with these compounds.
Our meta-analysis has some potential limitation. First it is based on a retrospective analysis of aggregate data and patients were not randomised according to the nature of VTE. Thus, a different prevalence of potential confounders in patients randomised to DOAC or VKA could not be definitively excluded. Furthermore, the prevalence of unprovoked VTE was highly variable in the studies included, ranging from about 30% in the two RECOVER trials11,12 to more than 60% in the three trials on anti-FX drugs13–15, which might affect the reproducibility of results in a general population of patients with VTE.
In conclusion, despite all inherent limitations, the results of the present meta-analysis suggest that DOAC have an efficacy similar to that of VKA in the treatment of both unprovoked and provoked VTE. We were not able to collect information on the safety of these new compounds in these two groups of patients. Thus, other adequately designed and sized studies are warranted to confirm our preliminary findings and provide information on the safety of DOAC in different settings.
Footnotes
Authorship contributions
MNDD conceived and designed the study, analysed the data, interpreted the results and drafted the manuscript; RLi performed the literature search, extracted data and drafted the manuscripting; PA and ADM performed the literature revision and acquired data; FD interpreted the results and critically revised the manuscript. All Authors read and approved the final version of the manuscript.
Disclosure of conflicts of interest
MNDDi Minno and FD have received grants and honoraria for research unrelated to the present study. None of the other Authors have anything to declare.
References
- 1.White RH. The epidemiology of venous thromboembolism. Circulation. 2003;107:I4–8. doi: 10.1161/01.CIR.0000078468.11849.66. [DOI] [PubMed] [Google Scholar]
- 2.Coppola A, Tufano A, Cerbone AM, et al. Inherited thrombophilia: implications for prevention and treatment of venous thromboembolism. Semin Thromb Hemost. 2009;35:683–94. doi: 10.1055/s-0029-1242722. [DOI] [PubMed] [Google Scholar]
- 3.White RH. Identifying risk factors for venous thromboembolism. Circulation. 2012;125:2051–3. doi: 10.1161/CIRCULATIONAHA.112.102814. [DOI] [PubMed] [Google Scholar]
- 4.Di Minno MN, Tufano A, Guida A, et al. Abnormally high prevalence of major components of the metabolic syndrome in subjects with early-onset idiopathic venous thromboembolism. Thromb Res. 2011;127:193–7. doi: 10.1016/j.thromres.2010.12.005. [DOI] [PubMed] [Google Scholar]
- 5.Baglin T, RL, Brown K, et al. Incidence of recurrent venous thromboembolism in relation to clinical and thrombophilic risk factors. Lancet. 2003;362:523–6. doi: 10.1016/S0140-6736(03)14111-6. [DOI] [PubMed] [Google Scholar]
- 6.Goldhaber S. Prevention of recurrent idiopathic venous thromboembolism. Circulation. 2004;110:IV20–4. doi: 10.1161/01.CIR.0000150641.65000.f2. [DOI] [PubMed] [Google Scholar]
- 7.van der Hulle T, Kooiman J, den Exter PL, et al. Effectiveness and safety of novel oral anticoagulants as compared with vitamin K antagonists in the treatment of acute symptomatic venous thromboembolism: a systematic review and meta-analysis. J Thromb Haemost. 2014;12:320–8. doi: 10.1111/jth.12485. [DOI] [PubMed] [Google Scholar]
- 8.Moher D, Liberati A, Tetzlaff J, et al. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLOS Medicine. 2009;6:e1000097. doi: 10.1371/journal.pmed.1000097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Juni P, Witschi A, Bloch R, et al. The hazards of scoring the quality of clinical trials for meta-analysis. JAMA. 1999;282:1054–60. doi: 10.1001/jama.282.11.1054. [DOI] [PubMed] [Google Scholar]
- 10.Sterne JA, Egger M, Smith GD. Systematic reviews in health care: investigating and dealing with publication and other biases in meta-analysis. BMJ. 2001;323:101–5. doi: 10.1136/bmj.323.7304.101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Schulman S, Kearon C, Kakkar AK, et al. Dabigatran versus warfarin in the treatment of acute venous thromboembolism. N Engl J Med. 2009;361:2342–52. doi: 10.1056/NEJMoa0906598. [DOI] [PubMed] [Google Scholar]
- 12.Schulman S, Kakkar AK, Goldhaber SZ, et al. Treatment of acute venous thromboembolism with dabigatran or warfarin and pooled analysis. Circulation. 2014;129:764–72. doi: 10.1161/CIRCULATIONAHA.113.004450. [DOI] [PubMed] [Google Scholar]
- 13.EINSTEIN Investigators. Bauersachs R, Berkowitz SD, et al. Oral rivaroxaban for symptomatic venous thromboembolism. N Engl J Med. 2010;363:2499–510. doi: 10.1056/NEJMoa1007903. [DOI] [PubMed] [Google Scholar]
- 14.EINSTEIN-PE Investigators. Büller HR, Prins MH, et al. Oral rivaroxaban for the treatment of symptomatic pulmonary embolism. N Engl J Med. 2012;366:1287–97. doi: 10.1056/NEJMoa1113572. [DOI] [PubMed] [Google Scholar]
- 15.Hokusai-VTE Investigators. Büller HR, Décousus H, et al. Edoxaban versus warfarin for the treatment of symptomatic venous thromboembolism. N Engl J Med. 2013;369:1406–15. doi: 10.1056/NEJMoa1306638. [DOI] [PubMed] [Google Scholar]