Abstract
Purpose
The purpose of this study was to determine the effect of the protein stabilizer octanoic acid on blastocyst development, implantation, and fetal growth in a murine model.
Methods
One-cell mouse embryos were collected and individually cultured in medium supplemented with recombinant human serum albumin for 96 h at 5 % oxygen in an EmbryoScope. Embryos were randomly allocated to four octanoic acid groups (0, 400, 800, or 1200 μM). Blastocyst development and cell cycle timings were calculated at 96 h of culture, and experiments were repeated in triplicate. Blastocysts were stained and fixed at 96 h for differential cell counts. Following 96 h of culture, blastocysts were transferred to recipients to determine implantation rates and fetal and placental weights.
Results
Blastocyst development, hatching rates, developmental kinetics, and total number of cells were negatively affected by octanoic acid at concentrations commonly used in human IVF. Implantation was not affected by octanoic acid but fetal and placental weights at 800 μM octanoic acid were increased relative to control.
Conclusions
Octanoic acid, a standard additive to human protein supplements used in IVF, can have long-term negative effects on embryonic and fetal development. The use of octanoic acid for human embryo culture should be monitored and reduced.
Keywords: Octanoic acid, Mouse embryo assay, Embryo time-lapse, Embryo culture
Introduction
Optimal culture of human embryos for clinical care requires minimizing stress from both defined and undefined sources. Defined variables, such as pH and temperature, are recognized as critical components of a culture system and as a result feature prominently in a laboratory’s quality management program [1]. Undefined variables appear in products that demonstrate uneven quality, such as mineral oil [2] and protein [3–5], variability that represents a challenge to quality programs utilized by manufacturers and clinical laboratories. To minimize the impact of products with variable quality, components that negatively affect embryo development should be identified in order to apply analytical methods to screen batches for variability. Methods more sensitive than the standard bioassay currently in use should be developed to detect these detrimental components.
Protein supplements for culture medium demonstrate lot-to-lot variability that can adversely affect embryo development [4, 6–9]. Protein used in clinical assisted reproductive technology (ART) is supplied as either human serum albumin (HSA) or as a complex mixture of proteins including albumin and globulins [10–12]. Though routine screening of protein supplements for endotoxins, electrolytes, and hormones is advocated by Meintjies [13], there are many other components that have only recently been described and therefore are not routinely measured. Proteomic analysis identified 110 non-declared proteins in HSA preparations, though 94 % of protein present was albumin [3]. Additional components identified in either HSA or complex protein supplements include cytokines [14], microRNA [15], transition metals [5], and the stabilizers N-acetyl-tryptophan and octanoic acid [4]. As these additional non-declared components of protein become defined, clinical significance, potential for harm, and lot-to-lot variability should be assessed.
Octanoic acid (OA) is a stabilizer added to the preparations of HSA prior to heat inactivation of viruses [16]. Biologically, OA is a well-described intermediary metabolite in fatty acid metabolism and is known to be cytotoxic at high concentrations [17]. Studies on the effect of OA on cell function in vitro demonstrate that it disrupts energy metabolism and induces oxidative damage [18], effects that occur at concentrations of OA similar to what is present in embryo culture after addition of protein supplements. Lot-to-lot variation of OA concentrations in HSA preparations [4, 19] coupled with clinic preference for amount of protein supplementation means that the actual amount of OA present in human embryo culture is highly variable and undefined.
We previously demonstrated that OA reduces blastocyst development in a mouse embryo assay at concentrations that are clinically relevant [4]. Here, we use recombinant human serum albumin that does not contain OA to determine the effect of increasing concentrations of OA on blastocyst development, implantation, and fetal growth.
Materials and methods
Two separate sets of mouse embryo experiments were conducted. Studies on embryo morphokinetics, blastocyst development, and cell numbers were performed at the Mayo Clinic. Implantation rates and fetal growth studies which included a second set of blastocyst development data were performed at the National Foundation for Fertility Research (NFFR).
Mouse embryo assay
The Mayo Clinic Institutional Animal Care and Use Committee (IACUC) approved all procedures involving animals. Female inbred FVB mice, age 6–7 weeks (in-house breeding colony), were superovulated with 5 IU of intraperitoneal pregnant mare serum (PMS Gonadotropin; NHPP Torrance, CA) followed 48 h later by 5 IU of intraperitoneal human chorionic gonadotropin (hCG; APP Pharmaceuticals Schaumburg, IL). The females were mated with male CF1 mice (Charles River Laboratories, Wilmington, MA), and 18 h post hCG, one-cell embryos were collected from the oviducts. Cumulus cells were removed with hyaluronidase (0.05 % w/v; Sigma, St. Louis, MO).
Embryo culture
The embryos were placed into individual wells in an EmbryoSlide (Fertilitech, Aarhus, Denmark) and cultured in an EmbryoScope (Fertilitech). The EmbryoScope slides were prepared the day before embryo collection by equilibration in an incubator with 5 % O2, 6.2 % CO2 and balance nitrogen to maintain pH between 7.20 and 7.30. The individual wells contained 25 uL of Global medium (LifeGlobal Guilford, CT) supplemented with 5 mg/mL recombinant human serum albumin (AlbIX, Novozymes, Bagsvaerd, Denmark) and were covered with 1.3 mL of mineral oil (LiteOil, LifeGlobal). The one-cell embryos (n = 12 per slide) were randomly allocated to the four treatment groups, and a different EmbryoScope slide was prepared for each concentration of OA (0, 400, 800, or 1200 μM). Treatments were prepared by making a 40 % stock solution of OA (200 μL OA (Sigma) in 300 μL ethyl alcohol) which was then used to prepare a 3 mM OA stock by adding 11.9 μL 40 % stock solution to 10 mL Global medium + 5 mg/mL AlbIX. The treatments were prepared via serial dilutions using Global medium + AlbIX.
Time-lapse system
Precise developmental kinetics were captured using the time-lapse microscopy feature of the EmbryoScope incubator (Fertilitech). Once embryos were placed in the incubator, seven planes of images were obtained every 20 min for 96 h. Blastocyst development was defined as the percentage of embryos that developed to the expanded blastocyst stage by 96 h. Cell division timings were determined manually and included t2, t3, t4, t5, t6, t7, and t8 (time from collection to 2, 3, 4, 5, 6, 7, and 8 cells), time of compaction, tM, initiation of blastulation, tsB, time to formation of blastocoel, tB, and time to an expanded blastocyst, tEB. Cell cycles were analyzed [20] and included cc2a (duration of the second cell cycle: time (h) an embryo is at the two-cell stage), cc3a (duration of the third interphase: time an embryo is at the four-cell stage), s2 (synchrony of the second cell division: time an embryo is at the three-cell stage), and s3 (synchrony of the third cell division: time an embryo contains 5, 6, or 7 cells).
Differential cell counts
At 96 h of culture, blastocysts were fixed and stained for CDX2 (mouse monoclonal anti-human CDX2 cat # MU392A-UC Biogenex, Fremont, CA, USA) and SOX2 (rabbit monoclonal anti-human SOX2 cat # AN579-5M, Biogenex) [21]. CDX2 and SOX2 immunostaining was used to quantify cells of the trophectoderm (TE) and inner cell mass (ICM), respectively. Fluorescent signals were visualized using an inverted microscope at ×200 magnification (Leica DMI6000B with Leica Application Suite—Advanced Fluorescence software, Buffalo Grove, IL, USA) using UV illumination and fluorescent filters specific to each secondary antibody. Images were analyzed using Adobe Photoshop CS6.
Embryo transfer
All mouse protocols were approved by the NFFR Ethics in Research Committee and followed animal care and use guidelines, as described by the Guide for the Care and Use of Laboratory Animals [22]. A second set of embryo culture experiments were conducted to obtain additional one-cell mouse embryos to produce embryos for transfer. Female inbred FVB mice, age 5–9 weeks (Harlan, Indianapolis, IN, USA), were superovulated using 5 IU PMSG (Calbiochem®, EMD Millipore, Billerica, MA, USA) followed in 48 h by 5 IU hCG (Calbiochem®) and mating to fertile CF-1 males. Seventeen hours post hCG, one-cell embryos were collected by puncturing the ampulla. Cumulus cells were removed using hyaluronidase (0.01 %; 80–160 U/mL). The one-cell embryos were randomly allocated to the four culture treatments (0 (control), 400, 800, and 1200 μM OA) and cultured in Global medium (LifeGlobal, Canada) supplemented with 5 mg/mL recombinant human serum albumin (AlbIX, Novozymes, Bagsvaerd, Denmark) in 20-μL droplets under Ovoil at 37 °C in 7.3 % CO2 and 6.5 % O2. Gas concentrations were increased to compensate for the elevation at which these experiments were conducted (Lone Tree, CO, ~1830 m above sea level) and are approximately equal to 6.0 % CO2 and 5 % O2 at sea level [23, 24]. After 96 h of culture, the embryos were assessed for development. Those embryos that had reached at least the expanded blastocyst stage and were designated for transfer were placed into a pre-equilibrated droplet of EmbryoGlue® (Vitrolife AB, Göteborg, Sweden) for 1 h. The embryos were surgically transferred (average of six embryos per uterine horn), one treatment per uterine horn/two treatments per female, to recipient Swiss Webster females (Harlan) age 2 to 5 months [25]. The recipients had been successfully mated to vasectomized CF-1 males, as evidenced by a vaginal plug, 1 day later than embryo donors (3.5 dpc at transfer). Following transfer, when the embryos reached 16.5 dpc, the recipient females were euthanized and implantation, fetal weights, and placental weights were recorded.
Statistics
Differences in blastocyst rate, cell cycle timings, and cell numbers were determined using JMP (SAS, Cary, NC) statistical software. Differences in percent blastocyst development, implantation, and fetal development were determined using chi-square. Differences in fetal and placental weights were determined using t test. Significance was determined at P < 0.05.
Results
Embryo development
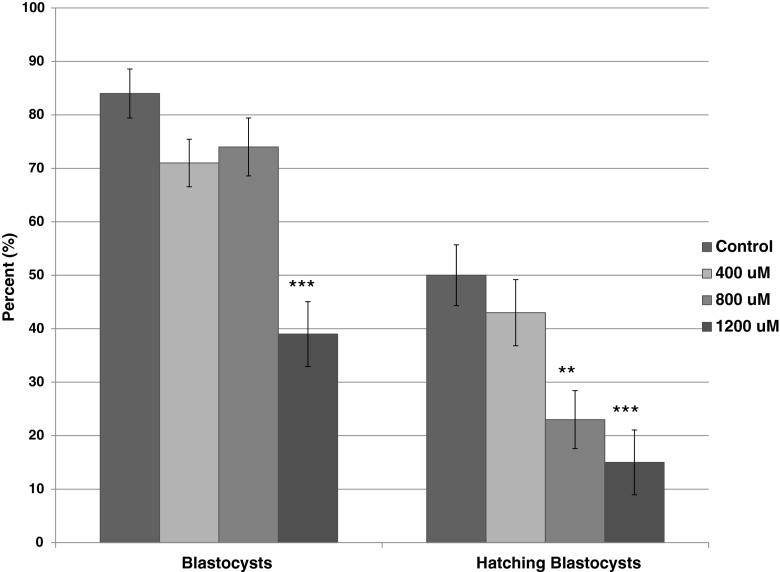
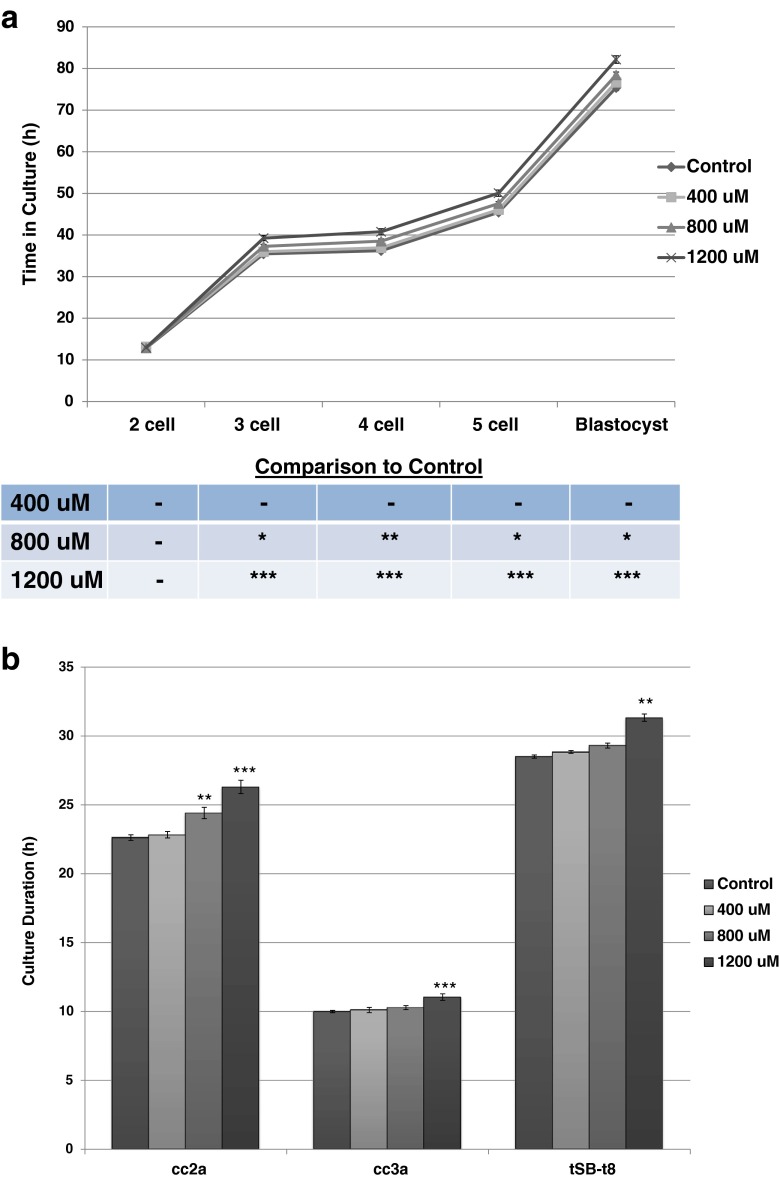
The industry standard endpoint for the mouse embryo assay is blastocyst development at 96 h which was our primary endpoint in this study (Fig. 1). In the studies conducted at the Mayo Clinic, the culture with 1200 μM OA resulted in significantly fewer blastocysts at 96 h than the control group (p < 0.0001). While the number of blastocysts was not affected by lower concentrations of OA, blastocyst hatching was lower at both 800 μM (p <0.01) and 1200 μM (p <0.0001) when compared with the controls (Fig. 1). As we have previously shown that specific timings of cell cycles using time-lapse imaging are a sensitive measure of oil toxicity [2], we compared the morphokinetics of embryo development among the treatment groups. The effects of OA at 800 and 1200 uM were detected as early as division to the three-cell stage, where division was delayed, persisted, and increased in difference through the formation of the blastocoel cavity (tSB; Fig. 2a).
Fig. 1.
Development of one-cell mouse embryos to the expanded blastocyst stage after 96 h in Global medium containing 5 mg/mL AlbIX and supplemented with 0, 400, 800, or 1200 μM octanoic acid (OA). N = 65 embryos per treatment. Significantly different than the control (0 μM OA) within category, **p < 0.01, ***p < 0.001 (Dunnett’s comparison)
Fig. 2.
a Kinetics of cell division during 96 h of culture in Global medium containing 5 mg/mL AlbIX supplemented with 0, 400, 800, and 1200 μM octanoic acid (OA). N = 65 mouse embryos per treatment. Significantly different than the control (OA) within category,*p < 0.05, **p < 0.01, ***p < 0.001 (Dunnett’s comparison). b Cell cycle length during development of one-cell mouse embryos cultured in Global medium containing 5 mg/mL AlbIX with increasing concentrations of octanoic acid (OA). cc2a duration of two-cell stage (t3–t2); cc3a duration of four-cell stage (t5–t4); tsB–t8 time from eight-cell to blastocyst. Significantly different than the control (0 μM OA) within category, **p < 0.01, ***p < 0.001 (Dunnett’s comparison)
Cell number and allocation
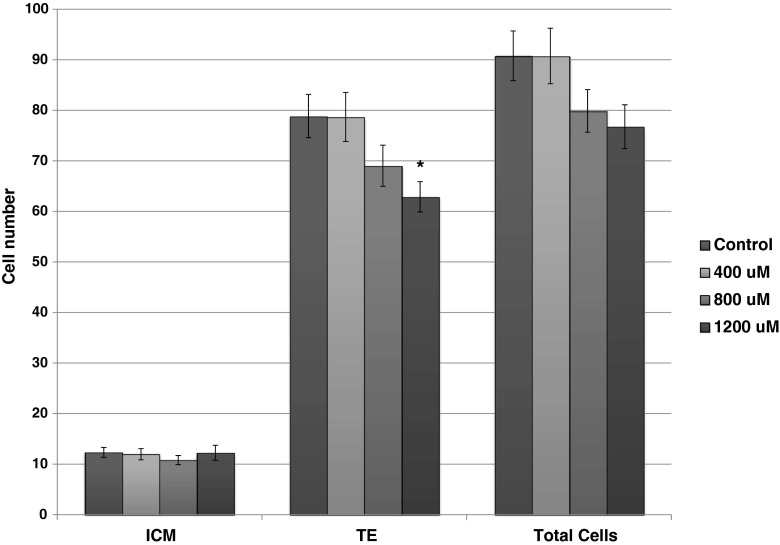
Blastocysts contained more trophectoderm cells when cultured in control medium than in medium supplemented with 1200 μM OA (p < 0.05) (Fig. 3). There was a trend for a decrease in total number of cells as the concentration of OA increased.
Fig. 3.
Cell number and allocations in mouse blastocysts following 96 h of culture in Global medium containing 5 mg/mL AlbIX supplemented with 0, 400, 800, and 1200 μM octanoic acid (OA). N = 17–28 embryos per treatment. ICM inner cell mass cells, TE trophectoderm cells, Total cells ICM plus TE. Significantly different than control (0 μM OA) within category, *p < 0.05 (Dunnett’s comparison)
Embryo transfer
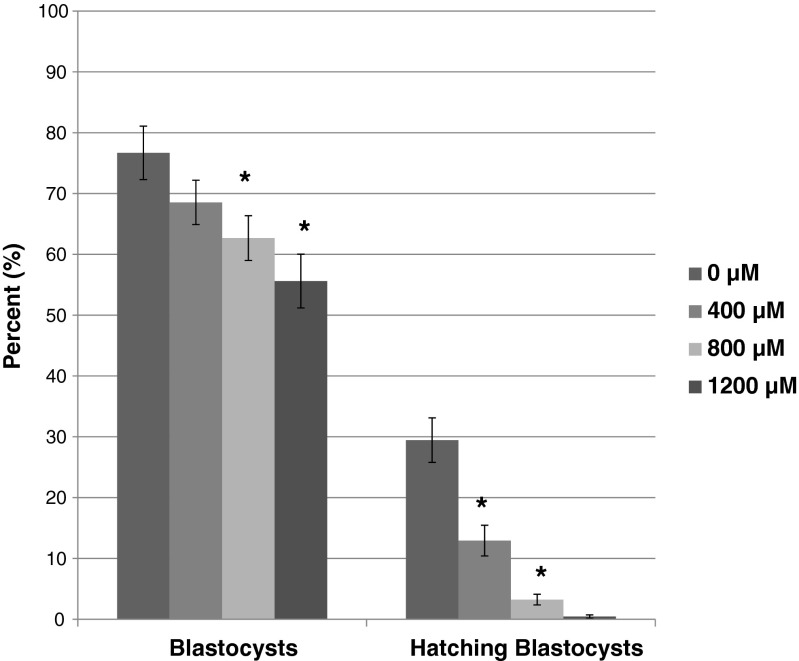
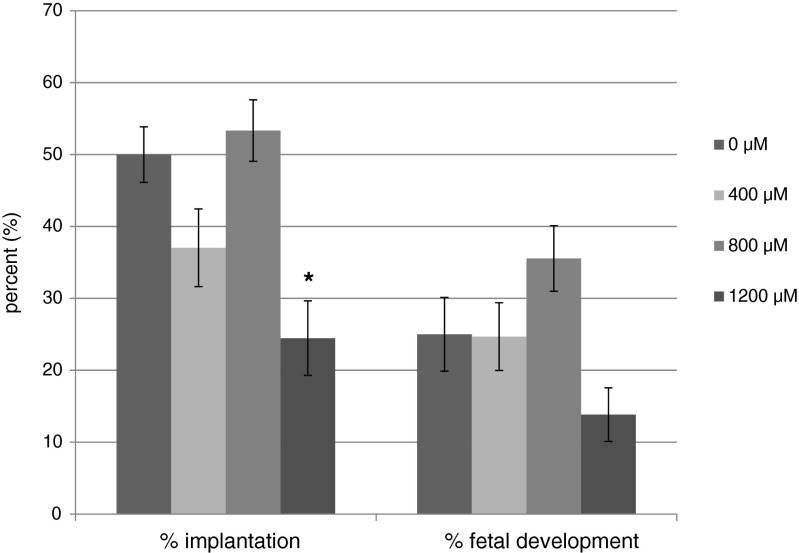
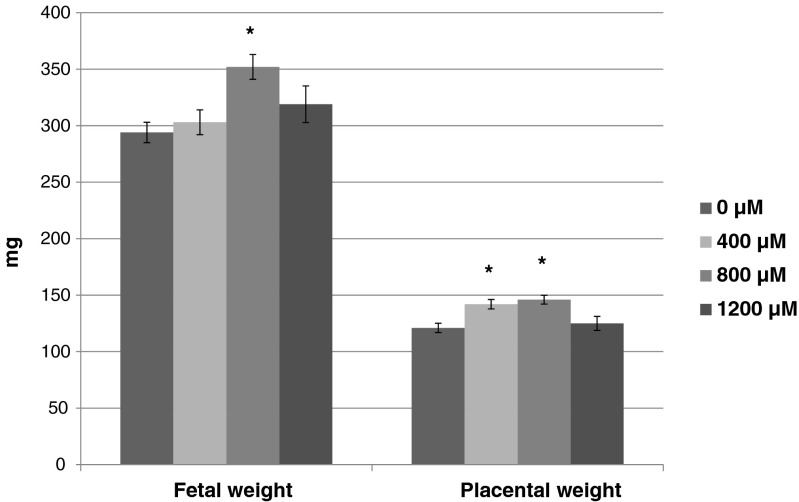
Following the blastocyst development and cell number studies, a separate set of experiments were conducted at NFFR where development of embryos produced for embryo transfer was assessed after 96 h of culture, prior to transfer to recipients. In this separate set of experiments, more blastocysts were developed in the control media than in the media supplemented with 800 μM (p < 0.05) and 1200 μM (p < 0.05) OA; whereas blastocyst development was not different for 400 μM OA (Fig. 4). In addition, OA at all concentrations significantly reduced the number of advanced blastocysts (expanded, hatching, or hatched) and hatching/hatched blastocysts compared to the control (Fig. 4). The mean number of embryos transferred per uterine horn was 5.60 ± 0.49 (0 μM), 6.23 ± 0.46 (400 μM), 5.63 ± 0.46 (800 μM), and 5.88 ± 0.51 (1200 μM). There were no differences in the pregnancy rate of any treatment compared to the control (0 μM, 53 %; 400 μM, 46 %; 800 μM, 69 %; 1200 μM, 31 %), although significantly fewer recipients became pregnant in 1200 uM compared to 800 uM OA. The implantation rate (number of implantations/number of embryos transferred) on 16.5 dpc for embryos cultured in 1200 μM (p < 0.05) OA was significantly reduced compared to the control (Fig. 5). However, fetal development (number of fetuses/number of embryos transferred) did not differ between treatments (Fig. 5). On D16.5, the weight of the fetuses derived from the embryos cultured in 800 μM was greater than the control fetuses (Fig. 6). Placentas developing from embryos cultured to the blastocyst stage in 400 and 800 μM OA were significantly heavier than the control (Fig. 6).
Fig. 4.
In a second series of experiments, development of additional one-cell mouse embryos was assessed prior to transfer to recipient females after 96 h of culture following supplementation of Global culture medium containing 5 mg/mL AlbIX with 0, 400, 800, and 1200 μM octanoic acid (OA) . N = 163–232 embryos per treatment. Advanced blastocyst; expanded, hatching, or hatched blastocyst stages. Significantly different than control (0 μM OA) within category, *p < 0.05
Fig. 5.
Implantation and fetal development after transfer of blastocysts produced in Global medium containing 5 mg/mL AlbIX supplemented with 0, 400, 800, or 1200 μM octanoic acid (OA). N = 13–16 transfers and 81–94 embryos transferred per treatment. Implantation, # implantations/# embryos transferred. Fetal development, # fetuses/# embryos transferred. Significantly different than the control (0 μM OA) within category, *p < 0.05
Fig. 6.
Fetal and placental weight following transfer of embryos produced in Global medium containing 5 mg/ml AlbIX supplemented with 0, 400, 800, or 1200 μM octanoic acid (OA). N = 16, 16, 24, and 10 fetuses from 0, 400, 800, and 1200 μM, respectively. Significantly different than the control (0 μM OA) within category, *p < 0.05
Discussion
In order to provide optimal conditions for human embryo development in vitro, the culture environment should be defined and any non-physiologic components should be absent, or at least present at minimal concentrations. We have previously demonstrated that concentrations of octanoic acid, a fatty acid used to stabilize albumin in products for human embryo culture, can vary from lot to lot and that OA inhibits mouse embryo development at concentrations commonly present during human embryo culture [4]. In this report, we use an OA-free recombinant albumin to demonstrate that OA, when added to the culture medium in defined quantities, reduces the growth rate of cleavage stage embryos by extending the time when embryos are at the 2- and 4-cell stages, resulting in lower blastocyst and hatching rates, reduced numbers of trophectoderm cells, decreased implantation, and increased fetal and placental weight.
Octanoic acid, an 8-carbon saturated fatty acid used to stabilize albumin during the process of viral heat inactivation, is present in serum at 0.2 μM [26]. Only during cases of enzyme deficiency, such as medium-chain acyl-CoA dehydrogenase deficiency (MCADD), do serum concentrations in plasma increase to levels (~0.7 mM) equal to that in embryo culture media supplemented with these protein products [18]. The Code of Regulations [27] indicates that OA should be added at concentrations of 0.08 mmol per gram of protein. For supplementation at 5 mg/mL HSA in culture media, this equates to an OA concentration of 400 or 1200 uM when HSA is 15 mg/mL. There is no industry standard for HSA concentration, though the most common concentrations are 5 and 10 mg/mL, corresponding to 400 and 800 uM OA, respectively. Our previous work demonstrated that OA concentrations vary among lots by as much as 50 % [4]. Thus, for IVF laboratories or media companies that supplement medium with 10 mg/mL HSA (10 % v/v), the normal variation in OA concentration would be 400–1200 uM, concentrations equivalent to the highest treatment in this study that induced the most marked effect.
The mechanism whereby OA altered cell cycle duration, blastocyst formation, implantation rates, and fetal weights is unknown. The adverse effects of OA on skeletal and hepatic cells in vitro are due in part to perturbed cellular metabolism, with decreased mitochondrial respiratory chain enzyme activities at concentrations of 500 μM [18]. Oxidative damage was also a feature of OA treatment in both cell types. Metabolic perturbation, one likely effect of OA, may induce changes at the pre-compaction stage that are expressed latently at the blastocyst or fetal stage (reviewed by [28]). For example, exposure to ammonium during the pre-compaction stage altered cell numbers and fetal development whereas exposure during the post-compaction stage was without effect [29]. Given the many adverse effects of OA on cellular function in vitro, the presence of OA in human embryo culture medium [4], which commonly occurs at 400–800 μM in HSA-supplemented culture medium but can be as high as 1200 uM, merits further scrutiny.
Fatty acids are an important energy source during pre-implantation development in most species [30, 31]. In fact, OA at low concentrations (100 μM) can be used as an energy source by mouse embryos via conversion to acetyl-CoA and beta-oxidation in medium lacking pyruvate and glucose [32]. The support of fatty acid metabolism via the addition of carnitine, critical for transport of lipids into mitochondria, is beneficial to mammalian embryo development [33–35]. However, evidence suggesting that embryos may benefit from exogenous fatty acid supplementation during culture in medium containing carbohydrates and amino acids is lacking as endogenous stores of fatty acids appear to be sufficient [36]. Unsaturated fatty acids tend to have beneficial effects whereas saturated fatty acids tend to have more deleterious effects at elevated concentrations [37–42]. Furthermore, human embryos with a higher ratio of unsaturated to saturated fatty acids are more likely to progress beyond the four-cell stage [43]. The ideal in vivo environment has an evolving amount of saturated, unsaturated, and polyunsaturated fatty acids, and the maintenance of these ratios is critical to the proper development of embryos [30]. Although it is added to protein as a stabilizer, OA may affect the delicate ratios of fatty acids leading to detrimental consequences for the early embryo such as aberrant metabolism, increased apoptosis, and long-term metabolic perturbations that carry through to fetal growth [30, 38, 44]. Altered fetal and placental weight suggests that culture with high concentrations of OA can have long-term consequences. In vitro culture of embryos potentially leads to aberrant epigenetic modifications in the genome causing alterations that would be maintained somatically and might affect gene expression at later stages of development [45, 46].
One limitation of these studies is that our control medium contained high levels of phosphate in the culture medium, which in some studies has been shown to be detrimental to mouse embryo development [47]. Since the phosphate concentration remained constant across treatments and development for the controls was within limits, this is likely not the case. We cannot exclude the possibility that the effects of OA observed are affected differently with high phosphate compared to standard phosphate concentrations.
Ultimately, OA should be removed from human embryo culture, or at least reduced to physiologic concentrations. Alternative stabilizers for current protein supplements or new protein products altogether need to be developed in order to achieve this goal. The recombinant product used in this study, AlbIX, is stabilized with high concentrations of phosphate rather than OA (4.1 mM at a working albumin concentration of 5 mg/mL [5]). The impact of a higher concentration of phosphate during human embryo culture requires further investigation. Alternatively, the recombinant albumin currently available for human embryo culture [48], which contains high concentrations of OA to keep the protein in solution, could be reformulated with a combination of reduced OA and increased phosphate, or other inert stabilizer(s). Complex protein supplements are used in the culture of human embryos because they are more closely able to mimic in vivo conditions by providing growth factors and other unidentified protein products that cannot currently be replicated. Research is needed to identify characteristics that are found in products purified from human blood so that supplements can be manufactured to replace human derived products.
In this work, we demonstrate that OA at concentrations used clinically have long-term negative effects on embryo and fetal development. These findings illustrate that undefined components of embryo culture media have the potential to adversely affect embryo and fetal development and warrant additional screening and analysis. In this regard, the suitability of OA for human embryo culture is questioned, and at the very least, its use in practice should be monitored, documented, and reduced. In light of this data, alternative stabilizers and/or protein sources should be explored that support embryo development without the safety concerns inherent in using OA.
Acknowledgments
Conflict of interest
This study was supported by Fertilitech.
Footnotes
Capsule
Octanoic acid, a standard additive to human protein supplements used in IVF, has long-term negative effects on murine embryonic and fetal development. The use of octanoic acid for human embryo culture should be monitored and reduced.
References
- 1.Mortimer D, Mortimer S. Quality and risk management in the IVF laboratory. Cambridge, UK: Cambridge University Press; 2005. [Google Scholar]
- 2.Wolff HS, Fredrickson JR, Walker DL, Morbeck DE. Advances in quality control: mouse embryo morphokinetics are sensitive markers of in vitro stress. Hum Reprod. 2013;28:1776–82. doi: 10.1093/humrep/det102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Dyrlund TF, Kirkegaard K, Poulsen ET, Sanggaard KW, Hindkjaer JJ, Kjems J, et al. Unconditioned commercial embryo culture media contain a large variety of non-declared proteins: a comprehensive proteomics analysis. Hum Reprod. 2014;29:2421–30. doi: 10.1093/humrep/deu220. [DOI] [PubMed] [Google Scholar]
- 4.Leonard PH, Charlesworth MC, Benson L, Walker DL, Fredrickson JR, Morbeck DE. Variability in protein quality used for embryo culture: embryotoxicity of the stabilizer octanoic acid. Fertil Steril. 2013;100:544–9. doi: 10.1016/j.fertnstert.2013.03.034. [DOI] [PubMed] [Google Scholar]
- 5.Morbeck DE, Paczkowski M, Fredrickson JR, Krisher RL, Hoff HS, Baumann NA, et al. Composition of protein supplements used for human embryo culture. J Assist Reprod Genet. 2014;31:1703–11. doi: 10.1007/s10815-014-0349-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kane MT. Variability in different lots of commercial bovine serum albumin affects cell multiplication and hatching of rabbit blastocysts in culture. J Reprod Fertil. 1983;69:555–8. doi: 10.1530/jrf.0.0690555. [DOI] [PubMed] [Google Scholar]
- 7.Batt PA, Gardner DK, Cameron AW. Oxygen concentration and protein source affect the development of preimplantation goat embryos in vitro. Reprod Fertil Dev. 1991;3:601–7. doi: 10.1071/RD9910601. [DOI] [PubMed] [Google Scholar]
- 8.Leveille MC, Carnegie J, Tanphaichitr N. Effects of human sera and human serum albumin on mouse embryo culture. J Assist Reprod Genet. 1992;9:45–52. doi: 10.1007/BF01204114. [DOI] [PubMed] [Google Scholar]
- 9.McKiernan SH, Clayton MK, Bavister BD. Analysis of stimulatory and inhibitory amino acids for development of hamster one-cell embryos in vitro. Mol Reprod Dev. 1995;42:188–99. doi: 10.1002/mrd.1080420208. [DOI] [PubMed] [Google Scholar]
- 10.Adler A, Reing AM, Bedford JM, Alikani M, Cohen J. Plasmanate as a medium supplement for in vitro fertilization. J Assist Reprod Genet. 1993;10:67–71. doi: 10.1007/BF01204443. [DOI] [PubMed] [Google Scholar]
- 11.Pool TB, Martin JE. High continuing pregnancy rates after in vitro fertilization-embryo transfer using medium supplemented with a plasma protein fraction containing alpha- and beta-globulins. Fertil Steril. 1994;61:714–9. doi: 10.1016/s0015-0282(16)56651-5. [DOI] [PubMed] [Google Scholar]
- 12.Weathersbee PS, Pool TB, Ord T. Synthetic serum substitute (SSS): a globulin-enriched protein supplement for human embryo culture. J Assist Reprod Genet. 1995;12:354–60. doi: 10.1007/BF02215726. [DOI] [PubMed] [Google Scholar]
- 13.Meintjes M. Media composition: macromolecules and embryo growth. Methods Mol Biol. 2012;912:107–27. doi: 10.1007/978-1-61779-971-6_8. [DOI] [PubMed] [Google Scholar]
- 14.Menezo Y, Khatchadourian C. Peptides bound to albumin. Life Sci. 1986;39:1751–3. doi: 10.1016/0024-3205(86)90094-9. [DOI] [PubMed] [Google Scholar]
- 15.Rosenbluth EM, Shelton DN, Wells LM, Sparks AE, Van Voorhis BJ. Human embryos secrete microRNAs into culture media—a potential biomarker for implantation. Fertil Steril. 2014;101:1493–500. doi: 10.1016/j.fertnstert.2014.01.058. [DOI] [PubMed] [Google Scholar]
- 16.Anraku M, Tsurusaki Y, Watanabe H, Maruyama T, Kragh-Hansen U, Otagiri M. Stabilizing mechanisms in commercial albumin preparations: octanoate and N-acetyl-L-tryptophanate protect human serum albumin against heat and oxidative stress. Biochim Biophys Acta. 2004;1702:9–17. doi: 10.1016/j.bbapap.2004.07.002. [DOI] [PubMed] [Google Scholar]
- 17.Rinaldo P, Matern D, Bennett MJ. Fatty acid oxidation disorders. Annu Rev Plant Physiol Plant Mol Biol. 2002;64:477–502. doi: 10.1146/annurev.physiol.64.082201.154705. [DOI] [PubMed] [Google Scholar]
- 18.Scaini G, Simon KR, Tonin AM, Busanello EN, Moura AP, Ferreira GC, et al. Toxicity of octanoate and decanoate in rat peripheral tissues: evidence of bioenergetic dysfunction and oxidative damage induction in liver and skeletal muscle. Mol Cell Biochem. 2012;361:329–35. doi: 10.1007/s11010-011-1119-4. [DOI] [PubMed] [Google Scholar]
- 19.Yu MW, Finlayson JS. Quantitative determination of the stabilizers octanoic acid and N-acetyl-DL-tryptophan in human albumin products. J Pharm Sci. 1984;73:82–6. doi: 10.1002/jps.2600730122. [DOI] [PubMed] [Google Scholar]
- 20.Ciray HN, Campbell A, Agerholm IE, Aguilar J, Chamayou S, Esbert M, et al. Proposed guidelines on the nomenclature and annotation of dynamic human embryo monitoring by a time-lapse user group. Hum Reprod. 2014;29:2650–60. doi: 10.1093/humrep/deu278. [DOI] [PubMed] [Google Scholar]
- 21.Bakhtari A, Ross PJ. DPPA3 prevents cytosine hydroxymethylation of the maternal pronucleus and is required for normal development in bovine embryos. Epigenetics. 2014;9:1271–9. doi: 10.4161/epi.32087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Council NR. Guide for the care and use of laboratory animals. 8. Washington, D.C.: National Academies Press; 2011. [PubMed] [Google Scholar]
- 23.Paczkowski M, Schoolcraft WB, Krisher RL. Fatty acid metabolism during maturation affects glucose uptake and is essential to oocyte competence. Reproduction. 2014;148:429–39. doi: 10.1530/REP-14-0015. [DOI] [PubMed] [Google Scholar]
- 24.Silva E, Greene AF, Strauss K, Herrick JR, Schoolcraft WB, Krisher RL. Antioxidant supplementation during in vitro culture improves mitochondrial function and development of embryos from aged female mice. Reprod Fertil Dev 2015. [DOI] [PubMed]
- 25.Nagy A, Gertsentein M, Vintersten K, Behringer R. Manipulating the mouse embryo: a laboratory manual. 3. New York: Cold Spring Harbor Laboratory Press; 2003. pp. 268–71. [Google Scholar]
- 26.Rinaldo P. Organic acids. In: Blau N, Duran M, Gibson KM, editors. Laboratory guide to the methods in biochemical genetics. Berlin-Heidelberg: Springer; 2008. pp. 137–70. [Google Scholar]
- 27.USFaD. Code of federal regulations title 21. In: FDA, editors. 21. Vol. CFR 640.8 Subpart H--Albumin. Silver Springs, MD: FDA; 2012.
- 28.Lane M, Gardner DK. Embryo culture medium: which is the best? Best Pract Res Clin Obstet Gynaecol. 2007;21:83–100. doi: 10.1016/j.bpobgyn.2006.09.009. [DOI] [PubMed] [Google Scholar]
- 29.Zander DL, Thompson JG, Lane M. Perturbations in mouse embryo development and viability caused by ammonium are more severe after exposure at the cleavage stages. Biol Reprod. 2006;74:288–94. doi: 10.1095/biolreprod.105.046235. [DOI] [PubMed] [Google Scholar]
- 30.McKeegan PJ, Sturmey RG. The role of fatty acids in oocyte and early embryo development. Reprod Fertil Dev. 2011;24:59–67. doi: 10.1071/RD11907. [DOI] [PubMed] [Google Scholar]
- 31.Ferguson EM, Leese HJ. A potential role for triglyceride as an energy source during bovine oocyte maturation and early embryo development. Mol Reprod Dev. 2006;73:1195–201. doi: 10.1002/mrd.20494. [DOI] [PubMed] [Google Scholar]
- 32.Yamada M, Takanashi K, Hamatani T, Hirayama A, Akutsu H, Fukunaga T, et al. A medium-chain fatty acid as an alternative energy source in mouse preimplantation development. Sci Rep. 2012;2:930. doi: 10.1038/srep00930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Sutton-McDowall ML, Yelland R, MacMillan KL, Robker RL, Thompson JG. A study relating the composition of follicular fluid and blood plasma from individual Holstein dairy cows to the in vitro developmental competence of pooled abattoir-derived oocytes. Theriogenology. 2014;82:95–103. doi: 10.1016/j.theriogenology.2014.03.011. [DOI] [PubMed] [Google Scholar]
- 34.Abdelrazik H, Sharma R, Mahfouz R, Agarwal A. L-carnitine decreases DNA damage and improves the in vitro blastocyst development rate in mouse embryos. Fertil Steril. 2009;91:589–96. doi: 10.1016/j.fertnstert.2007.11.067. [DOI] [PubMed] [Google Scholar]
- 35.Takahashi T, Inaba Y, Somfai T, Kaneda M, Geshi M, Nagai T, et al. Supplementation of culture medium with L-carnitine improves development and cryotolerance of bovine embryos produced in vitro. Reprod Fertil Dev. 2013;25:589–99. doi: 10.1071/RD11262. [DOI] [PubMed] [Google Scholar]
- 36.Sturmey RG, Reis A, Leese HJ, McEvoy TG. Role of fatty acids in energy provision during oocyte maturation and early embryo development. Reprod Domest Anim. 2009;44(Suppl 3):50–8. doi: 10.1111/j.1439-0531.2009.01402.x. [DOI] [PubMed] [Google Scholar]
- 37.Dunning KR, Russell DL, Robker RL. Lipids and oocyte developmental competence: the role of fatty acids and beta-oxidation. Reproduction. 2014;148:R15–27. doi: 10.1530/REP-13-0251. [DOI] [PubMed] [Google Scholar]
- 38.Leroy JL, Sturmey RG, Van Hoeck V, De Bie J, McKeegan PJ, Bols PE. Dietary fat supplementation and the consequences for oocyte and embryo quality: hype or significant benefit for dairy cow reproduction? Reprod Domest Anim. 2014;49:353–61. doi: 10.1111/rda.12308. [DOI] [PubMed] [Google Scholar]
- 39.Van Hoeck V, Bols PE, Binelli M, Leroy JL. Reduced oocyte and embryo quality in response to elevated non-esterified fatty acid concentrations: a possible pathway to subfertility? Anim Reprod Sci. 2014;149:19–29. doi: 10.1016/j.anireprosci.2014.07.015. [DOI] [PubMed] [Google Scholar]
- 40.Aardema H, Vos PL, Lolicato F, Roelen BA, Knijn HM, Vaandrager AB, et al. Oleic acid prevents detrimental effects of saturated fatty acids on bovine oocyte developmental competence. Biol Reprod. 2011;85:62–9. doi: 10.1095/biolreprod.110.088815. [DOI] [PubMed] [Google Scholar]
- 41.Leroy JL, Vanholder T, Mateusen B, Christophe A, Opsomer G, de Kruif A, et al. Non-esterified fatty acids in follicular fluid of dairy cows and their effect on developmental capacity of bovine oocytes in vitro. Reproduction. 2005;130:485–95. doi: 10.1530/rep.1.00735. [DOI] [PubMed] [Google Scholar]
- 42.Van Hoeck V, Sturmey RG, Bermejo-Alvarez P, Rizos D, Gutierrez-Adan A, Leese HJ, et al. Elevated non-esterified fatty acid concentrations during bovine oocyte maturation compromise early embryo physiology. PLoS One. 2011;6:e23183. doi: 10.1371/journal.pone.0023183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Haggarty P, Wood M, Ferguson E, Hoad G, Srikantharajah A, Milne E, et al. Fatty acid metabolism in human preimplantation embryos. Hum Reprod. 2006;21:766–73. doi: 10.1093/humrep/dei385. [DOI] [PubMed] [Google Scholar]
- 44.Jungheim ES, Louden ED, Chi MM, Frolova AI, Riley JK, Moley KH. Preimplantation exposure of mouse embryos to palmitic acid results in fetal growth restriction followed by catch-up growth in the offspring. Biol Reprod. 2011;85:678–83. doi: 10.1095/biolreprod.111.092148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Dean W, Bowden L, Aitchison A, Klose J, Moore T, Meneses JJ, et al. Altered imprinted gene methylation and expression in completely ES cell-derived mouse fetuses: association with aberrant phenotypes. Development. 1998;125:2273–82. doi: 10.1242/dev.125.12.2273. [DOI] [PubMed] [Google Scholar]
- 46.Reik W, Romer I, Barton SC, Surani MA, Howlett SK, Klose J. Adult phenotype in the mouse can be affected by epigenetic events in the early embryo. Development. 1993;119:933–42. doi: 10.1242/dev.119.3.933. [DOI] [PubMed] [Google Scholar]
- 47.Biggers JD, McGinnis LK. Evidence that glucose is not always an inhibitor of mouse preimplantation development in vitro. Hum Reprod. 2001;16:153–63. doi: 10.1093/humrep/16.1.153. [DOI] [PubMed] [Google Scholar]
- 48.Bungum M, Humaidan P, Bungum L. Recombinant human albumin as protein source in culture media used for IVF: a prospective randomized study. Reprod Biomed Online. 2002;4:233–6. doi: 10.1016/S1472-6483(10)61811-1. [DOI] [PubMed] [Google Scholar]