Abstract
To determine whether postexercise criteria for peripheral artery disease (PAD) diagnosis recommended by the American Heart Association (AHA) identifies the same group of PAD patients.
Diagnosis of PAD is performed using ankle-brachial index at rest (resting-ABI). When resting-ABI is not contributive, an AHA scientific statement recommend to use 1 of 2 following criteria: a postexercise ABI decrease of greater than 20% or a postexercise ankle pressure decrease of greater than 30 mm Hg.
Between 1996 and 2012, 31,663 consecutive patients underwent lower-extremity arterial study at Mayo Clinic. Among them, only unique patients who had exercise treadmill testing were analyzed. In this retrospective analysis, resting-ABI, postexercise ABI, and postexercise decrease of ankle pressure measured at 1-minute were measured in each patient. We conducted an analysis of agreement between postexercise criteria expressing the agreement separately for the positive and the negative ratings. Twelve thousand three hundred twelve consecutive patients were studied with a mean age of 67 ± 12 years, 61% male. According to resting-ABI, 4317 (35%) patients had PAD. In the whole population, if a clinician diagnoses “PAD” with 1 postexercise criterion, the probability that other clinicians would also diagnose “PAD” is 74.3%. If a clinician diagnoses “no PAD”, the probability that other clinicians would also diagnose “no PAD” is 82.4%. In the patients to be of potential benefit from treadmill test when the resting-ABI > 0.90, if a clinician diagnoses “PAD” with 1 postexercise criterion, the probability that other clinicians would also diagnose “PAD” is 58.4% whereas if a clinician diagnoses “no PAD,” the probability that other clinicians would also diagnose “no PAD” is 87.5%.
Postexercise criteria do not identify the same group of PAD patients. In our opinion, postexercise criteria to define PAD deserve additional study.
INTRODUCTION
Lower extremity peripheral artery disease (PAD) is common, affecting 12% to 29% of the elderly1 and as many as 8 million Americans2 and approximately 11% of the French population older than 40 years.3 PAD includes disorders that affect the leg arteries and is associated with functional impairment that adversely impacts on the quality of life.4,5 PAD is due to different physiopathologic processes that contribute to the development of stenosis of the leg arterial circulation. The most common cause of PAD is atherosclerosis.4 Intermittent claudication has been considered as the most classic symptom of PAD. However, contemporary studies showed that 20% to 50% of PAD patients are asymptomatic.6 Ankle brachial index at rest (ABI), the ratio of the systolic blood pressure measured at the ankle to the systolic blood pressure measured at the brachial artery,7,8 is the main clinical means to diagnose the presence and severity of PAD. It also serves as a marker of cardiovascular risk and atherosclerosis.9,10
PAD is diagnosed when the ABI at rest (resting-ABI) is equal to or lower than 0.90.4,9 However, when there is a high clinical suspicion of PAD a resting-ABI above 0.90 may not be reassuring. In these cases, the recent American Heart Association (AHA) scientific position paper has stated that when the resting-ABI is >0.90 but there is clinical suspicion of PAD, postexercise ABI or other noninvasive tests, which may include imaging, should be used (Class I; Level of Evidence A).11–14 There is a further recommendation that a postexercise ankle pressure decrease from resting value of >30 mm Hg or a postexercise ABI decrease of >20% from the resting values are diagnostic criteria for PAD (Class IIa; Level of Evidence A).11,13,15
Surprisingly, although 2 postexercise criteria are suggested in the guidelines, no study has shown that the same group of patients is identified with each. If both postexercise criteria detect the same population then physicians and laboratories could choose either as preferred. However, if the criteria do not identify the same population, a patient considered to have PAD in 1 laboratory could be classified as normal in another. The aim of this study was to analyze patients referred to our vascular laboratory for exercise evaluation of suspected PAD using both criteria recommended by the position paper.9
MATERIALS AND METHODS
Studied Population
Between 1996 and 2012, 31,663 consecutive patients were referred for arterial lower extremity arterial testing. In our hospital as a preset protocol, all patients who were addressed for arterial lower extremity study and who can walk have a treadmill test. We excluded patients who did not accept participation in research; did not have treadmill exercise test; were aged < 18 years old at the time of the arterial assessment. Patients’ characteristics such as age and gender were collected. The Mayo Clinic Institutional Review Board approved the study.
ABI and Ankle Pressure Decrease Measurements
All included patients had pressure measurements at rest to calculate resting-ABI and after exercise to calculate postexercise ABI and postexercise ankle pressure decrease.
Resting-ABI
Patients were relaxed, head and heels supported, in a room with comfortable temperature (approximately 22°C). After a 10-minute supine rest period, a trained technician used a hand-held Doppler probe with a pressure cuff (with an appropriate size) to obtain systolic pressures in the right and left brachial, posterior tibial, and dorsalis pedis arteries. The resting-ABI was calculated by dividing the highest pressure of the limb (dorsalis pedis or posterior tibial pressures) by the highest arm pressure as recommended.9 For each limb we studied the highest resting-ABI, and for each patient the lowest ABI of limbs was used to classify patients as to presence or absence of PAD.
PAD: resting-ABI ≤ 0.90.
Borderline: 0.90 < resting-ABI ≤ 1.00.
Normal: 1.00 < resting-ABI < 1.40.
Noncompressible: resting-ABI ≥ 1.40.
We defined patients to be of potential benefit from treadmill test when the resting-ABI > 0.90.
Treadmill Tests
Different treadmill test protocols with continuous electrocardiographic monitoring were used depending upon the patient ability to walk. Walking speed ranged from 0.8 to 2 mph (most typically 1.0, 1.5, or 2.0 mph). The grade (incline) for all tests was 10%.16 The maximal duration for treadmill test was 5 minutes, which corresponds to a maximal walking distance of 259 m.
Postexercise ABI and Postexercise Ankle Pressure Decrease
Patients walked on the treadmill until the protocol was completed or stopped due to symptoms. Immediately (approximately within 1 minute) after the end of exercise the patient was placed on the bed in a supine position and the 1 technician remeasured systolic pressures of the highest of the resting brachial arteries while another technician simultaneously measured the leg arterial pressures. Resting-ABI, postexercise ABI were assessed on the same artery to calculate the postexercise ABI decrease. Postexercise ankle pressure decrease was calculated using the pressure used at rest to define the resting-ABI and on the same artery for the measurement of the postexercise ankle pressure. When the Doppler signal was absent postexercise, a systolic pressure of 0 mm Hg was recorded as the postexercise ankle pressure and used to calculate the postexercise ABI.
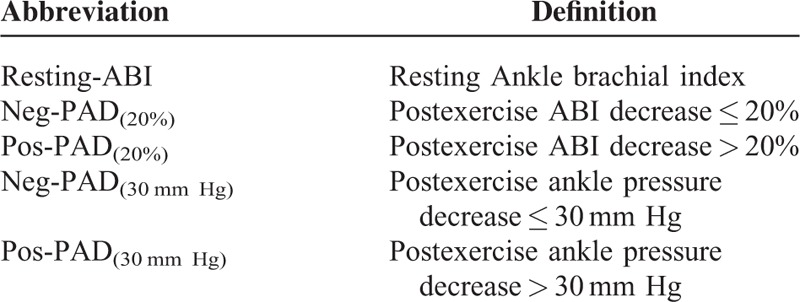
Two types of postexercise PAD were defined depending upon the criterion used.9 Pos-PAD(20%) was defined as a postexercise ABI decrease > 20%, while Neg-PAD(20%) was defined as a postexercise ABI decrease ≤ 20%. Pos-PAD(30 mm Hg) was defined as a postexercise ankle pressure decrease > 30 mm Hg following exercise, while Neg-PAD(30 mm Hg) was defined as a postexercise ankle pressure decrease ≤ 30 mm Hg (see Table 1).
TABLE 1.
List of Main Abbreviations
Statistical Analysis
Results were expressed as the mean ± SD. To compare proportion of postexercise PAD diagnosis according to resting-ABI categories, we used the McNemar test. As proposed by de Vet et al, an analysis of agreement between the 2 postexercise criteria was conducted expressing the agreement separately for the positive and the negative ratings.17 The positive agreement and negative agreement were calculated according to the literature.18 Statistical analyses were performed using MedCalc for Windows, version 12.5 (MedCalc Software, Ostend, Belgium). For all statistical tests, a 2-tailed probability level of P < 0.05 was used to indicate statistical significance.
RESULTS
Among the 31,663 patients screened, 12,312 patients were studied according to inclusion and exclusion criteria (Figure 1). Among the studied population, the average age was 67 ± 12 years and 7518 (61%) were men. Average resting highest systolic arm pressure was 136 ± 20 mm Hg. Five thousand one hundred ninety-eight (42%) patients had a resting highest arm pressure ≥ 140 mm Hg. A resting arm pressure difference ≥ 15 mm Hg between right and left arms was found in 518 patients (4%). According to the guidelines, 4317 (35%) patients had PAD, 1554 (13%) were “borderline,” 162 (1%) had noncompressible arteries, and 6279 (51%) were normal. Numbers of treadmill tests with walking speed at between 0.8 and 1.0, between 1.1 and 1.5, and between 1.6 and 2.0 mph were 3440 (28%), 3728 (30%), and 5144 (42%), respectively. Average postexercise systolic arm pressure was 166 ± 27 mm Hg. Table 2 shows the number of patients classified as patients having PAD according to the postexercise criteria proposed by the AHA statement. In the whole population (n = 12,312), 79% of patients (n = 9739) are classified in the same group by either criterion. Twenty-one percent (n = 2573) were mismatched (ie, positive by 1 criterion and negative by the other; Figure 2). Among the population without patients with noncompressible arteries (ie, patients with resting-ABI ≤ 1.40) (n = 12,150), 2531 patients (21%) presented a mismatch. In the whole population (n = 12,312), agreements between both postexercise criteria are presented in Table 3. Positive agreement and negative agreement were 74.3% and 82.4%, respectively.
FIGURE 1.
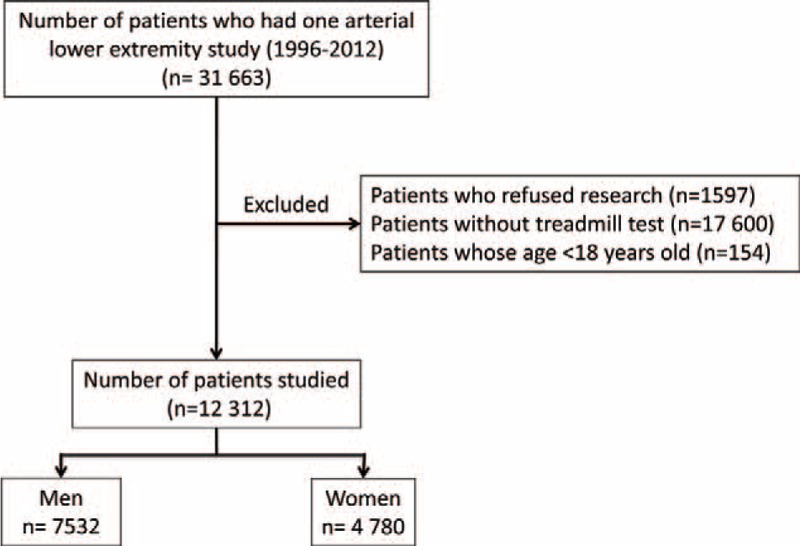
Flow diagram of the patients in this analysis.
TABLE 2.
Classification of Patients According to Their Postexercise Results and Resting-ABI
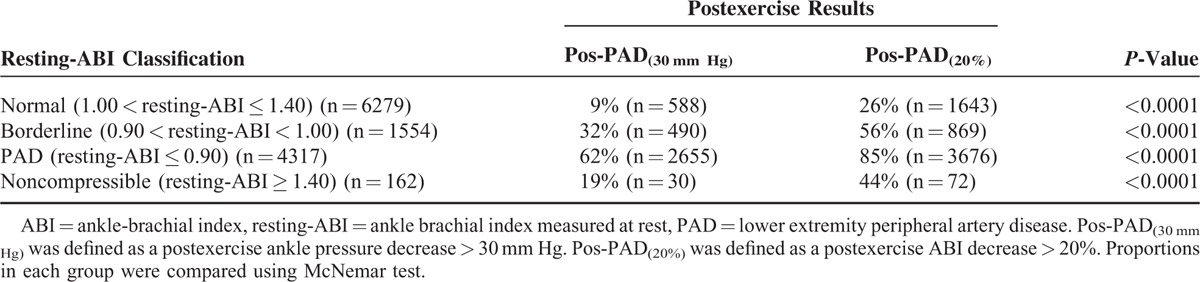
FIGURE 2.
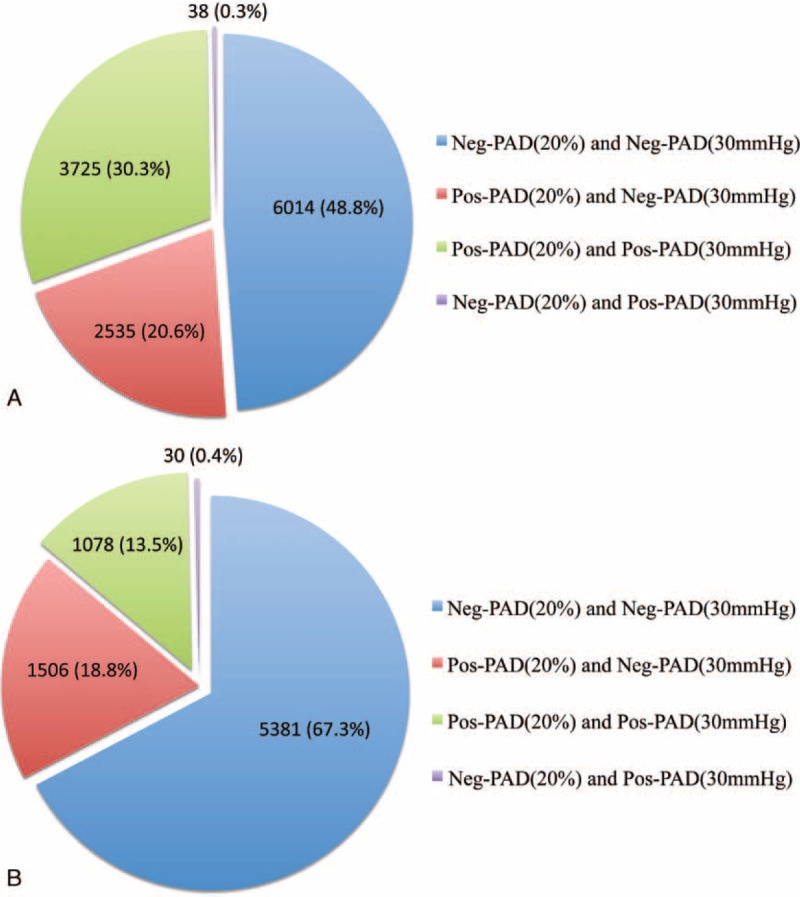
Repartition of the patients according to postexercise results in the whole population (n = 12,312) (Panel A) and in the population with resting-ABI > to 0.90 with need for exercise (n = 7995) (Panel B). PAD means lower extremity peripheral artery disease. ABI means ankle-brachial index. Resting-ABI: ankle brachial index measured at rest; Pos-PAD(20%) was defined as a postexercise ABI decrease > 20%; Neg-PAD(20%) was defined as a postexercise ABI decrease ≤ 20%; Pos-PAD(30 mm Hg) was defined as a postexercise ankle pressure decrease > 30 mm Hg; Neg-PAD(30 mm Hg) was defined as a postexercise ankle pressure decrease ≤ 30 mm Hg.
TABLE 3.
Agreement Between Postexercise Criteria
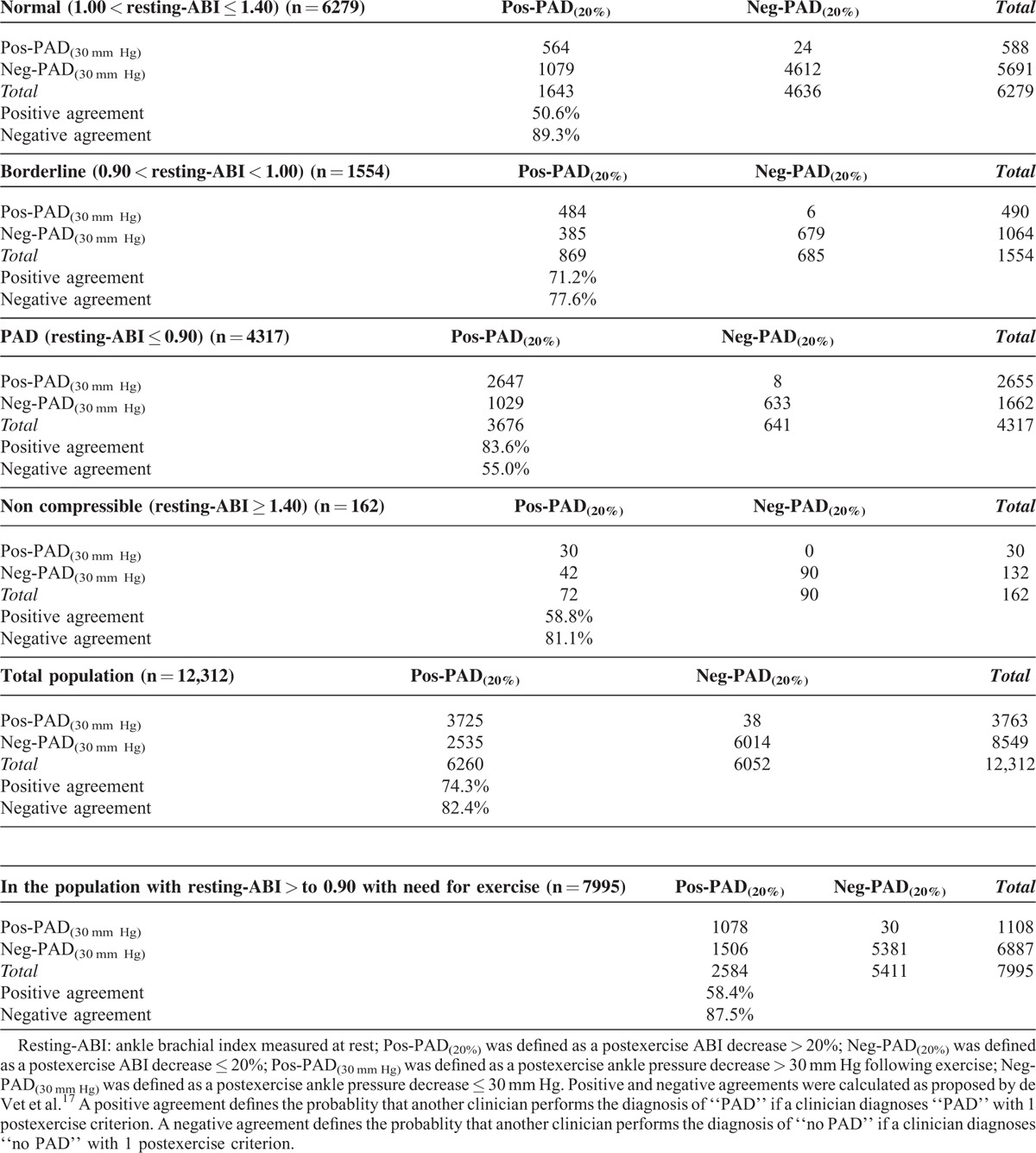
In the group defined as having the most potential benefit from treadmill testing (ie, resting-ABI > 0.90), there were 7995 patients (mean age 65 ± 13 years, 4707 men (59%)). In this group, 19% of patients (n = 1536) presented with a mismatch. In this group, agreements between both postexercise criteria are presented in Table 3. Positive agreement and negative agreement were 58.4% and 87.5%, respectively. Finally, the percentage of mismatch was 19% (n = 1494) in this group of patients without noncompressible patients (ie, patients with 0.90 < resting-ABI < 1.40) (n = 7833) who can be considered to have a treadmill test.
The same proportion of mismatch was found whatever the walking speed used on the treadmill. The mismatches were 21.57%, 21.97%, 19.67% for treadmill speeds from 0.8 to 1.0 mph, from 1.1 to 1.5 mph, and from 1.6 to 2.0 mph.
DISCUSSION
This analysis, in a very large population, demonstrates 2 major findings. First, in patients with a borderline or normal ABI at rest, the postexercise ABI was abnormal in one-third of patients by one or both criteria. This underscores the importance of exercise testing or other imaging in patients with claudication symptoms as recommended by the AHA statement.9
Second, the 2 criteria proposed by the AHA task force result in discrepant results or a “mismatch.” In the whole population, if a clinician diagnoses “PAD” with 1 postexercise criterion, the probability that other clinicians would also diagnose “PAD” is 74.3% whereas if a clinician diagnoses “no PAD,” the probability that other clinicians would also diagnose “no PAD” is 82.4%. In the patients to be of potential benefit from treadmill test when the resting-ABI > 0.90, if a clinician diagnoses “PAD” with 1 postexercise criterion, the probability that other clinicians would also diagnose “PAD” is only 58.4% whereas If a clinician diagnoses “no PAD,” the probability that other clinicians would also diagnose “no PAD” is 87.5%. Indeed, even patients with PAD according to resting-ABI criteria (≤0.90) can be classified as negative PAD patient after exercise when criteria proposed by the scientific paper statement are used.9
This study demonstrates an inconsistency between 2 “standard” postexercise criteria for identifying patients with PAD. The same patient will be classified as having PAD using 1 criterion while being classified as negative using the other. Approximately 1 in 5 patients were classified differently by the 2 criteria. Regardless of the resting-ABI, a decrease in postexercise ABI of 20% was more often positive than a fall in postexercise ankle pressure of 30 mm Hg.
If we focus on the group most likely, and indeed expected to have a positive exercise study (resting-ABI ≤ 0.90) the discovery that some of these patients will have normal postexercise results is quite puzzling. Indeed, among resting PAD patients only 62% of patients had a decrease of ankle pressure >30 mm Hg and 85% of patients had a decrease of postexercise ABI > 20%. We expected that all of these patients would have abnormal postexercise results because, from a pathophysiologic perspective, PAD symptoms are due to a deficit in arterial blood supply to meet increase demand during exercise. Abnormal results found at rest would have been amplified by the exercise in our patients who were able to walk. These discrepancies might come from the fact that a resting-ABI, which is performed at rest, is used to define the disease whereas the mismatch between blood supplies and demands mainly appears during exercise. In this case some patients who are considered to have PAD using resting-ABI could be false positive patients even if their cardiovascular risk is increased.10 A second possibility is that we did not record the resting and postexercise values correctly. This latter point is unlikely in that the protocol followed has not changed and was in place well before any guidelines or recommendation statements.7 More importantly, we have a small and highly trained group of technicians with an average longevity in the laboratory of over a decade. Technician to technician quality assessment have been performed annually for almost 20 years and a culture of confirmation on difficult studies has been routine. The third possibility is that the postexercise criteria used are not adequate. This merits further discussion.
The postexercise ankle pressure cut-off of >30 mm Hg was initially proposed by Laing and Greenhalgh in 198313 and used recently by Hoogeveen et al15 Laing et al decided to use this cut-off because they showed that ankle pressure change in 67 healthy subjects (age range 20–34 years old) following exercise (4 km/hour, 10% slope for 1 minute) ranged from −27 to +35 mm Hg, 95% of the postexercise results were between −25 and +22 mm Hg, and thus a cut-off value of greater than −30 mm Hg would seem reasonable. However, the use of these young control subjects to define normal response is questionable because the prevalence of PAD increases with age and becomes higher than 5% after 60 years old.19 Furthermore, ageing induces changes in the arterial wall20 and this can modify the pressure and the ABI.21 Ideally, healthy control subjects should be age matched. Moreover, the 1-minute duration of exercise used by Laing et al is markedly different than current protocols in most laboratories, and extrapolation to a 5-minute walk is difficult.
The second cut-off (postexercise ABI decrease >20%) suggested by the AHA position paper is based on Ouriel et al9,11 This paper must also be looked at critically.9 In figure 4 of the Ouriel et al it appears that the percentage of postexercise ABI fall was nearly 7 ± 7% in control limbs versus approximately 40 ± 25% in PAD patients. The position paper identifies the postexercise decrease in ABI at 20% instead of 40%, which is certainly a less stringent criterion for the diagnosis of PAD. Ouriel et al used a treadmill protocol with 1.5 mph and 7% grade.11 If we used the criterion from Ouriel and studied the mismatch between both criteria, 1034 (8%) patients were mismatched in the whole population (data not shown) which is less than using the >20% criterion but which is not entirely satisfactory. If we only considered patients who need to have a treadmill test, 488 (6%) patients were mismatched (data not shown). Further, Ouriel et al used arteriograms of the limbs as gold standard and receiver operating characteristics (ROC) curves to study the sensitivity and specificity of resting-ABI and post treadmill exercise ABI.11 They found that resting-ABI and posttreadmill exercise ABI were equivalent in defining normal and arteriographically diseased extremities.11 Of interest, there was no quantification of stenosis on arteriograms. Although studies of Laing et al and Ouriel et al are of interest,11,13 they have the previously mentioned biases. Therefore can we keep their results to define postexercise criteria to define PAD in a position paper? Probably not. The AHA position paper is an excellent reference for the use of resting ABI in clinical practice. However, our results and the analysis of the literature used to define postexercise criteria bring into question which postexercise criteria should be routinely used in clinical decision making.
Last point, we found that the proportion of discordant results using the postexercise AHA guidelines is nearly the same whatever the walking speed protocol used. This corroborates the results of previous studies that postexercise ABI is not modified by the workload but the recovery time is.22,23
LIMITATIONS
Our study has some limitations. We did not record the demographics of the patients but in this study each patient was its own control since the rest and postexercise measurements were obtained in each patient. We cannot exclude that some risk factors could modify these discrepancies between postexercise criteria. However, we would like to underscore that our aim was to determine whether or not discrepancies exist in a “real life” population addressed for a suspected PAD and not to determine which factors can modify or not the discrepancies. The impact of such risk factors deserves additional study. Our study has the advantage to include consecutive patients and represent the real life when clinicians do not always know which risk factors the patient has. Furthermore we did not give any instructions (ie, abstained from smoking) before arm and leg pressures measurements.9 However, this study was done in clinical routine and therefore represents what we are expected to find in clinical practice. The fact that each patient was its own control also minimizes the effect of potential inappropriate conditions before the measurements. Finally, we did not have any gold standard (angiograms) to study which postcriterion was the most accurate to diagnose PAD after exercise. However, the aim of this study was not to define new criterion but to study the concordance or discordance of the postexercise criteria suggested by the AHA. The results of our study demonstrate that postexercise criteria that can be used to define PAD deserve additional study.
In conclusion, postexercise criteria suggested by the position paper do not identify the same group of patients (ie, nearly 1 in 5 patients has discordant results). However, after critical analysis of the papers used to define these criteria, we think that new postexercise criteria should be developed and validated.
Footnotes
Abbreviations: ABI = ankle brachial index, AHA = American Heart Association, Pos-PAD(20%) = was defined as a postexercise ABI decrease > 20%, Neg-PAD(20%) = was defined as a postexercise ABI decrease ≤ 20%, Pos-PAD(30 mm Hg) = was defined as a postexercise ankle pressure decrease > 30 mm Hg following exercise, Neg-PAD(30 mm Hg) = was defined as a postexercise ankle pressure decrease ≤ 30 mm Hg, PAD = lower extremity peripheral artery disease, resting-ABI = ankle brachial index measured at rest.
GM received grants from the “Société Française de Médecine Vasculaire” and from the “Institut Servier” for his postdoctoral fellowship at the Mayo Clinic, Rochester, MN, USA.
The authors have no conflicts of interest to disclose.
REFERENCES
- 1.Hiatt WR. Medical treatment of peripheral arterial disease and claudication. N Engl J Med 2001; 344:1608–1621. [DOI] [PubMed] [Google Scholar]
- 2.Selvin E, Erlinger TP. Prevalence of and risk factors for peripheral arterial disease in the United States: results from the National Health and Nutrition Examination Survey, 1999–2000. Circulation 2004; 110:738–743. [DOI] [PubMed] [Google Scholar]
- 3.Boccalon H, Lehert P, Mosnier M. Assessment of the prevalence of atherosclerotic lower limb arteriopathy in France as a systolic index in a vascular risk population. J Mal Vasc 2000; 25:38–46. [PubMed] [Google Scholar]
- 4.Hirsch AT, Haskal ZJ, Hertzer NR, et al. ACC/AHA 2005 Practice Guidelines for the management of patients with peripheral arterial disease (lower extremity, renal, mesenteric, and abdominal aortic): a collaborative report from the American Association for Vascular Surgery/Society for Vascular Surgery, Society for Cardiovascular Angiography and Interventions, Society for Vascular Medicine and Biology, Society of Interventional Radiology, and the ACC/AHA Task Force on Practice Guidelines (Writing Committee to Develop Guidelines for the Management of Patients With Peripheral Arterial Disease): endorsed by the American Association of Cardiovascular and Pulmonary Rehabilitation; National Heart, Lung, and Blood Institute; Society for Vascular Nursing; TransAtlantic Inter-Society Consensus; and Vascular Disease Foundation. Circulation 2006; 113:e463–e654. [DOI] [PubMed] [Google Scholar]
- 5.McDermott MM, Greenland P, Liu K, et al. Leg symptoms in peripheral arterial disease: associated clinical characteristics and functional impairment. JAMA 2001; 286:1599–1606. [DOI] [PubMed] [Google Scholar]
- 6.Norgren L, Hiatt WR, Dormandy JA, et al. Inter-Society Consensus for the Management of Peripheral Arterial Disease (TASC II). J Vasc Surg 2007; 45 Suppl S:S5–S67. [DOI] [PubMed] [Google Scholar]
- 7.Grenon SM, Gagnon J, Hsiang Y. Video in clinical medicine. Ankle-brachial index for assessment of peripheral arterial disease. N Engl J Med 2009; 361:e40. [DOI] [PubMed] [Google Scholar]
- 8.Winsor T. Influence of arterial disease on the systolic blood pressure gradients of the extremity. Am J Med Sci 1950; 220:117–126. [DOI] [PubMed] [Google Scholar]
- 9.Aboyans V, Criqui MH, Abraham P, et al. Measurement and interpretation of the ankle-brachial index: a scientific statement from the American Heart Association. Circulation 2012; 126:2890–2909. [DOI] [PubMed] [Google Scholar]
- 10.Fowkes FG, Murray GD, Butcher I, et al. Ankle brachial index combined with Framingham Risk Score to predict cardiovascular events and mortality: a meta-analysis. JAMA 2008; 300:197–208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ouriel K, McDonnell AE, Metz CE, et al. Critical evaluation of stress testing in the diagnosis of peripheral vascular disease. Surgery 1982; 91:686–693. [PubMed] [Google Scholar]
- 12.Carter SA. Indirect systolic pressures and pulse waves in arterial occlusive diseases of the lower extremities. Circulation 1968; 37:624–637. [DOI] [PubMed] [Google Scholar]
- 13.Laing S, Greenhalgh RM. The detection and progression of asymptomatic peripheral arterial disease. Br J Surg 1983; 70:628–630. [DOI] [PubMed] [Google Scholar]
- 14.Fowler B, Jamrozik K, Norman P, et al. Prevalence of peripheral arterial disease: persistence of excess risk in former smokers. Aust N Z J Public Health 2002; 26:219–224. [DOI] [PubMed] [Google Scholar]
- 15.Hoogeveen EK, Mackaay AJ, Beks PJ, et al. Evaluation of the one-minute exercise test to detect peripheral arterial disease. Eur J Clin Invest 2008; 38:290–295. [DOI] [PubMed] [Google Scholar]
- 16.Nicolai SP, Viechtbauer W, Kruidenier LM, et al. Reliability of treadmill testing in peripheral arterial disease: a meta-regression analysis. J Vasc Surg 2009; 50:322–329. [DOI] [PubMed] [Google Scholar]
- 17.de Vet HC, Mokkink LB, Terwee CB, et al. Clinicians are right not to like Cohen's kappa. BMJ 2013; 346:f2125. [DOI] [PubMed] [Google Scholar]
- 18.Dice L. Measures of the amount of ecologic association between species. Ecology 1945; 26:297–302. [Google Scholar]
- 19.Criqui MH, Fronek A, Barrett-Connor E, et al. The prevalence of peripheral arterial disease in a defined population. Circulation 1985; 71:510–515. [DOI] [PubMed] [Google Scholar]
- 20.Safar ME. Arterial aging—hemodynamic changes and therapeutic options. Nat Rev Cardiol 2010; 7:442–449. [DOI] [PubMed] [Google Scholar]
- 21.Le Faucheur A, Desvaux BN, Bouye P, et al. The physiological response of ankle systolic blood pressure and ankle to brachial index after maximal exercise in athletes is dependent on age. Eur J Appl Physiol 2006; 96:505–510. [DOI] [PubMed] [Google Scholar]
- 22.Laing SP, Greenhalgh RM. Standard exercise test to assess peripheral arterial disease. Br Med J 1980; 280:13–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Sakurai T, Matsushita M, Nishikimi N, et al. Effect of walking distance on the change in ankle-brachial pressure index in patients with intermittent claudication. Eur J Vasc Endovasc Surg 1997; 13:486–490. [DOI] [PubMed] [Google Scholar]


