Abstract
Lipid nanoparticles (LNPs) have attracted special interest during last few decades. Solid lipid nanoparticles (SLNs) and nanostructured lipid carriers (NLCs) are two major types of Lipid-based nanoparticles. SLNs were developed to overcome the limitations of other colloidal carriers, such as emulsions, liposomes and polymeric nanoparticles because they have advantages like good release profile and targeted drug delivery with excellent physical stability. In the next generation of the lipid nanoparticle, NLCs are modified SLNs which improve the stability and capacity loading. Three structural models of NLCs have been proposed. These LNPs have potential applications in drug delivery field, research, cosmetics, clinical medicine, etc. This article focuses on features, structure and innovation of LNPs and presents a wide discussion about preparation methods, advantages, disadvantages and applications of LNPs by focusing on SLNs and NLCs.
Keywords: Colloidal carriers, SLNs, NLCs, physical stability, preparation methods
Introduction
liposomes are traditional models of lipid-based formulations which were invented in 1965 and have been widely studied in recent decades.1 A liposome is defined as a spherical vesicle with an aqueous internal cavity enclosed by a lipid bilayer membrane. The name of liposome is resulting from two Greek words, 'lipid' meaning fat and 'soma' meaning body. They have been investigated in recent decades for dermal, pharmaceutical and cosmetics studies. Liposomes have unique advantages as a pharmaceutical carrier such as protection of drug against enzymes degradation, low toxicity, flexibility, biocompatibility, entirely biodegradablity and non-immunogenicity.2-4 However, many of its applications are limited due to some disadvantages such as short shelf life, poor stability, low encapsulation efficacy, rapid removal by reticuloendothelial system (RES), cell interactions or adsorption and intermembrane transfer.5 Despite the fact that liposomes have been the hallmark of lipid nanoparticles (LNPs) for site specific delivery of therapeutics, there is a need to develop approaches for advanced control over drug release and drug delivery, which may not potentially load into liposomes. One of the reasons for this, apart from potential technological problems, is the non-availability of a ' cheap ' pharmaceutical liposome.
SLN and NLC have been introduced as potential attractive and marketable options due to their natural components.6 SLNs and NLCs have been identified since 1990 as a substitute carrier system to liposomes, emulsions and polymeric nanoparticles.7 Historical development of colloidal carrier system is illustrated in Figure 1.These LNPs have an average size of 40 to 1000 nm and a spherical morphology8 and are composed of solid phase lipid and surfactant. Dispersed phase is solid fat, and surfactant is used as emulsifier. Lipid components of SLNs are solid at both body and ambient temperature9 and can be highly purified triglycerides, complex glyceride mixtures or even waxes.10 Surfactants are used in concentrations of about 0.5 to 5% to enhance stability. The proper selection of lipids and surfactants can affect physicochemical properties and quality of them such as particle size and drug loading.11 Compared to liposomes, they have drug stability and prolonged release and they are safer than polymeric carriers because of avoidance of organic solvents in their production. As well as they have no problems with respect to large scale production. But the common disadvantages of SLNs are unpredictable gelation tendency and inherent low incorporation rates resulting from the crystalline structure of the solid lipid.12,13
Figure 1.
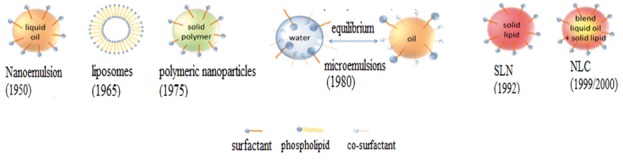
Schematic of historical development of colloidal carrier system.26
NLCs have been introduced as the next generation of the SLNs at the ending of the 1990s to dominate the possible difficulties of SLNs.14-16 NLCs improve the stability, capacity loading and prevent the drug expulsion during storage. They are detectable from SLNs by the composition of the solid matrix. lipidic phase in NLCs are contained both solid and liquid lipids at the room and ambient temperature.17 Formless, imperfect and multiple types are three forms of NLCs which are discussed in the next part of paper.18
There are several production techniques for LNPs such as high pressure homogenization (HPH), solvent emulsification /evaporation, supercritical fluid extraction of emulsions (SFEE), ultrasonication or high speed homogenization19-21 and spray drying.22 Two processes of the HPH, hot and cold processes were developed. These are two basic production methods which in both, drug is dissolved or solubilized in the lipid being melted at around 5-10 °C above its melting point.19
SLNs and NLCs have remarkably wide range of properties which make them useful for parenteral, dermal, pulmonary and topical delivery of drugs. These products have been developed in order to reduce toxic side effects of the incorporated highly potent drugs and increase the efficacy of the treatment. As well as, they have presented good potential in gene transfer, cosmetic and food industry. However, because of mentioned limitations and difficulties related to them, the total number of products on the market is still limited.23-26
Definition and description of structural properties of SLNs and NLCs
SLNs were presented since 1990 as a substitute carrier system to liposomes, emulsions and polymeric nanoparticles.17 They have an average size of 40 to 1000 nm and a spherical morphology that can be studied with TEM (Transmission electron microscopy) and SEM (scanning electron microscopy).18 SLNs are composed of approximately 0.1 – 30 (% w/w) solid fat which is dispersed in an aqueous phase. Surfactants are used in concentrations of about 0.5 to 5% to enhance stability. The proper selection of lipids and surfactants can affect the particle size, long-term stability during storage, drug loading and behaviors of release.19 Lipid components of them are solid at both body and ambient temperature.23 The lipids which are used in preparation of SLNs include fatty acids, steroids, waxes, monoglycerides, diglycerides and triglycerides. Depending on method of preparation, SLNs may be used for both hydrophilic and hydrophobic drugs.12,24 Compared with other systems, SLNs have many benefits including ease of preparation, low cost, high-scale production, excellent physical stability, good release profile, chemical versatility, preparation in the absence of organic solvent, no toxicity of lipid carrier system, biodegradability of lipids, being cheaper than polymeric carrier, being easier to get approval and reliability and biodegradability of lipids.25,27 Common disadvantages of SLNs are: Lipid particle growth, tendency to gelation, dynamics of polymorphic transitions, and their inherent low incorporation rate due to the crystalline structure of the solid lipid.12,19,25,28-30
Dependent of production method of SLNs, three models of drug incorporation into them have been reported which is described in detail in Table 1. These are including solid solution model, core-shell model (drug-enriched Shell) and core-shell model (drug-enriched core) which is shown in Figure 2.25,31,32
Table 1. Three models of drug incorporation into SLNs .
| Solid solution model | Core-shell model (drug-enriched shell) | Core-shell model (drug-enriched core) |
| Formation of this model in cold homogenization technique | Formation of this model in hot homogenization technique | Dispersion cooling leads to a supersaturation of the drug which is dissolved in the lipid. |
| Using no drug-solubilizing surfactant | Formation of lipid core at recrystallization temperature of lipid | Precipitation of drug in melted lipid |
| Drug dispersed in lipid matrix | Cooling of the obtained dispersion leads to re-partitioning of the drug to the lipid phase | Finally, further cooling lead to recrystallization of the lipid |
| There is a strong interaction between lipid and drug | Concentration of drug in surrounding membrane | Formation of drug-enriched core |
Figure 2.
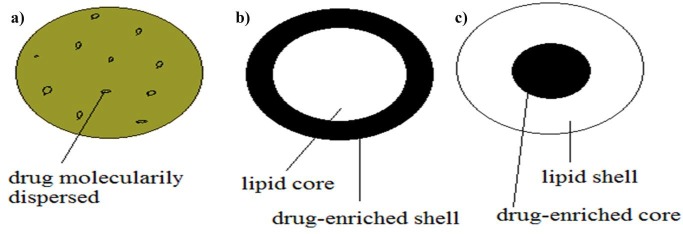
Schematic representation of drug incorporation models of SLNs. a) solid solution model, b) core-shell model (drug-enriched Shell), c) core-shell model (drug-enriched core).
In the next generation of the lipid nanoparticle, NLCs are modified SLNs in which lipidic phase is contained both solid (fat) and liquid (oil) lipids at ambient temperature.33 In fact NLCs are modified generations of SLNs that presenting a mixture of solid and liquid phase (oil) forming a formless matrix, which improves the stability and capacity loading.34 NLCs show contrasts with SLNs: more loading capacity for some drugs, some less water in the dispersion, prevent or minimize the drug expulsion during storage, but not reported significant difference between the biotoxicity of SLNs and NLCs. 17,32,33,35,36 Three forms of structure for NLCs have been presented which is shown in Figure 3. The first class is imperfect type in which solid and liquid fats (oil) are mixed in various lipid structures. Specific conditions in the crystallization procedure lead to an extremely disordered. Imperfect lipid matrix structure presenting gap between triglyceride fatty acid chains in crystal and thus increase the ability of the drugs for entering to the matrix.37-39 The second class is formless type (non-crystalline matrix), this is a type of NLCs that has no crystalline structure and thus prevents the expulsion of loaded drug which is known as amorphous type. In this form, crystals are formed during cooling and for preventing from it, must be used certain lipids mixture.39-41 The third class is multiple type: in this class drug solubility in liquid lipid is greater than in solid lipid, thus maintained from decomposition by solid lipid. This form of NLCs is similar to w/o/w emulsions.27,42
Figure 3.
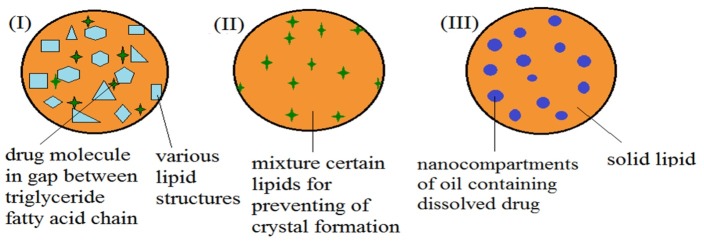
Structures of NLC. Class I (imperfect type), class II (formless type), class III (multiple type). To prevent drawbacks of SLN, the NLC should possess sufficient gap to better drug accommodation.43
Preparation of lipid nanoparticles
There are many systems for the production of LNPs. Commonly used methods for preparation of SLNs are high pressure homogenization at elevated or low temperatures (including hot homogenization and cold homogenization), solvent emulsification ,evaporation or diffusion, supercritical fluid (supercritical fluid extraction of emulsions (SFEE)), ultrasonication or high speed homogenization19-21 and spray drying.22
High pressure homogenization (HPH)
HPH technology has emerged as an established and potent technique for production of lipid nanoparticles. In contrast to other techniques, this method for large-scale production of LNs also can be used. Two processes of the homogenization, hot and cold processes were developed. The pharmaceutical compound is dissolved or dispersed in the melted lipid before to the HPH, in both processes. High pressure (100–2000 bar) moves the fluid in the narrow gap in homogenizer. Average particle size is in sub-micron region. Homogenization has several advantages including large-scale production, absence of organic solvent, improved product stability and improved loading of drugs, but specific high pressure and temperature conditions make challenges about its application.2,44
Hot homogenization
In this method, homogenization occurs at temperatures upper than melting point of lipid. Drug loaded lipid melt is dispersed in hot aqueous surfactants phase (isothermal) by mixing device (Ultra-Turrax) and leads to the formation of pre-emulsions. Because of the reduced viscosity at high temperatures, particle size becomes lesser mainly.45,46 This technique is illustrated in Figure 4. Hot homogenization has three basic problems. The first is temperature-dependent degradation of the drug, the second is the drug penetrates into the aqueous phase during homogenization and the third is complexity of the crystallization step of the nanoemulsion leading to several modifications and/or supercooled melts.14,47
Figure 4.
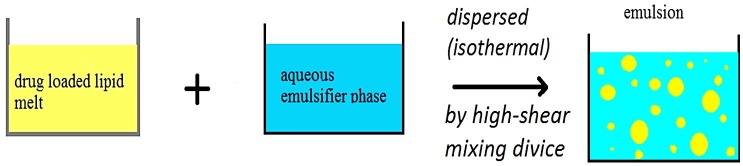
Hot homogenization technique for production of SLN48
Cold homogenization
Like the hot homogenization method, the drug is dissolved in the lipid melt, and then rapidly cooled by liquid nitrogen or dry ice. Milling leads to formation of nanoparticles in the range of 50-100 nm which are dispersible in a cold surfactant phase that form a pre-suspension. PHP is done at ambient temperature that leads to break the nanoparticles to SLNs. Cold homogenization technique has been expanded to resolve the problems of the hot homogenization technique3,49 Schematic diagram of this method is given in Figure 5.
Figure 5.
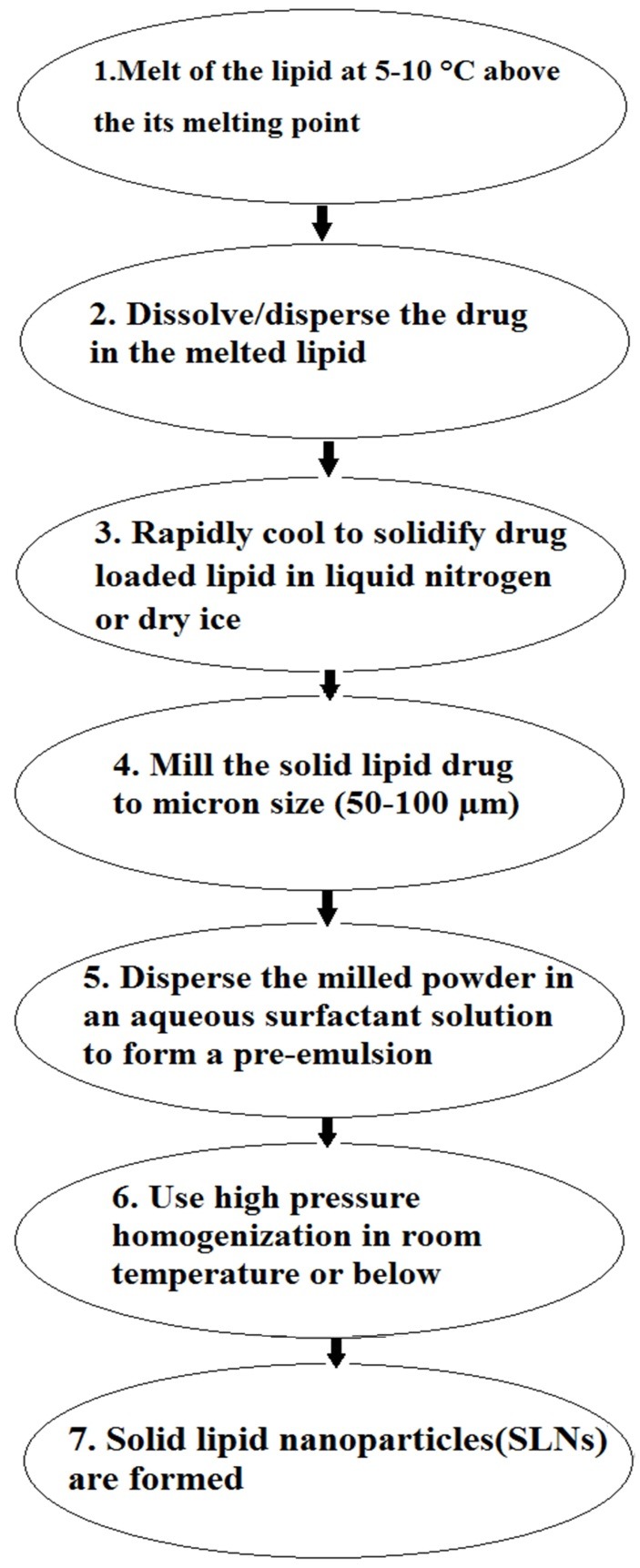
Schematic diagram of cold homogenization method for preparation of SLNs
Solvent emulsification /evaporation
In this method, the lipid is dissolved in a water-immiscible organic solvent. Next an emulsion in an aqueous phase containing surfactant is formed. To remove the solvent from the emulsion, evaporation under reduced pressure is used. Evaporation leads to the dispersion of nanoparticles in the aqueous phase (using lipid precipitation process in the aqueous phase). Unlike cold homogenization, this method will not enter to any thermal stress, but the organic solvent which is used in this method is a disadvantage. Particle size can vary according to the solid lipid and surfactant.41,50
Supercritical fluid extraction of emulsions (SFEE)
SFEE is a relatively novel approach for SLN preparation. This method uses a supercritical fluid such as carbon dioxide for solvent extraction from o/w emulsions. Carbon dioxide is a good option, but it should be noted that it can’t dissolve many of the drugs. Therefore supercritical antisolvent precipitation (SAS) can be an alternative method to SFEE.51,52
Ultrasonication or high speed homogenization
One of the methods for the production of LNPs is ultrasonication or high-shear homogenization. The aqueous phase containing a large amount of surfactant, and lipid phase is dispersed in this phase. The high amount of surfactant will be considered as a disadvantage. Another disadvantage of this method is that it doesn’t produce a narrow particle size distribution, thus leading to instability during storage. This technique uses from the simple instruments that can be found in every lab unlike hot and cold homogenization.53,54
Spray drying
This system is a substitute to lyophilization method that leads to the production of pharmaceutical product from aqueous SLN dispersion. Spray drying is cost effective method rather than lyophilization, but is not used for the production of lipids commonly. Because the high temperatures and shear forces used in this way, leads to particle aggregation. According to previous studies, lipids with melting point greater than 70 °C are suitable for spray drying.55
Applications of lipid nanoparticles
SLN and NLC have remarkably wide range of application and have shown greatly to control the skin penetration of several actives, delivery of food and drugs, cosmetics and other applications.26
Oral drug delivery applications
Oral drug administration is common and preferred route due to good patient compliance, non-invasiveness and therapeutic success, but poorly water-solubility of drugs is limiting step for the absorption of them. Thus an approach is needed to improve the bioavailability of drugs. Lipid-based delivery systems in the recent decades have shown many advances for this purpose. These systems include a wide range of formulations such as self-nanoemulsifying drug delivery system (SNEDDS), self-microemulsifying drug delivery system (SMEDDS), nanoemulsions, SLNs and NLCs. Since in these systems, drug is dissolved in the lipid thus makes the potential for improving the bioavailability of poorly soluble drugs in water, especially lipophilic drugs. In fact, these systems can increase dissolution of drug, residence time and lymphatic uptake. A good thing is that toxicity has not been observed in most cases.56-59
Pulmonary drug delivery applications
LNPs easily incorporated into carriers which inhaled to the lungs, therefore able to provide a deep lung deposition, good adhesion and elongated retention in the lung. Also due to improved and prolonged therapeutic effects, SLNs and NLCs have a longer dosing interval and better compliance for patients. They are typically particulate systems for various drug delivery applications. Advantages of drug release of fat in the lungs including: control of the release profile, prolonged release, faster in vivo degradation and better tolerability compared to particles made from some polymeric materials such as PLA or PLGA. Pulmonary delivery of SLNs is not widely accepted because of toxicity issues but when the physiologic lipids are used, is estimated to be safer than polymer-based systems. Dry powder formulations or aqueous suspensions of SLNs can be used for pulmonary drug delivery. Many studies are available about SLNs as local delivery carriers or as systemic delivery carriers for small molecules and for macromolecules respectively by pulmonary administration.60-63
Gene transfer applications
LNPs penetrate to biological membranes effectively through receptor-mediated pathway because lipids are the most important components of cell membranes. Thus enhance the uptake of genetic compounds.64,65 The delivery of some bioactive to particular sites in the body and their release behavior is directly dependent to particle size.66 The achievement of gene therapy (with DNA and RNA transfer) depends on the new bioactive delivery techniques. While 1980; more than 400 clinical studies in gene therapy have been reported. Delivery vectors are used in gene transfer due to restricted ability of naked DNA transfer to cells owing to propensity to enzymatic degradation.67-71 Cationic SLNs are interesting and proper nonviral gene delivery vector for systemic delivery. SLNs directly bond with DNA and can be used for gene transfection. Genospheres (such as cationic SLNs) have large potential for targeted gene delivery. Genospheres generally carry materials such as plasmid DNA, DNA and other nucleic acids. Three issues are important about them: composition of cationic SLN, their ability to condense DNA and transferring of nucleic acid to cells.59,66 NLCs can be effectively used as novel nonviral gene transfer vector that offers a promising approach for gene therapy.72
Cosmetic applications
LNPs such as SLNs and NLCs are one of the excellent vehicles for cosmetic and dermatological application. They have some characteristics which make them talented carriers for cosmetic applications for instance protection of sensitive compounds against chemical degradation15 and enhancement the water content of the skin.13 The use of LNPs as carriers for sunscreens, anti-acne and anti-ageing actives has been investigated. In fact, due to the high control behaviors of LNPs on skin penetration of active substances, they have UV-blocking and skin hydration behavior. In cosmetic products, reduction of the desire to scratch and skin damage is important. Since these formulations bear a resemblance to skin structure, there is no disruption and toxic effect when used topically.17
Food applications
LNPs are of particular interest to food manufacturers as novel delivery systems for encapsulation of bioactive compounds. LNPs are excellent potential carriers for sensitive compounds in food industry because they improve the industrial and the nutritional quality of a lipid containing food. Quality of them is affected by the lipid oxidation at storage and processing steps. Therefore must use antioxidants to prevent this process. Examples of active substances enclosed in LNPs for food industry are Beta-carotene, Lutein and Lycopene.11,73-76
Conclusion
In summary, SLNs are very complex system with obvious advantages and disadvantages compared to other colloidal carriers. Due to the stability and drug expulsion problems of SLNs, the NLCs were emerged. Based on the composition and organization of lipids and drugs in the particles, a wide collection of structural forms have been illustrated for SLNs and NLCs. The extremely unordered lipid matrix structured of NLC, enhanced drug encapsulation and stability, and also presenting good release profile made them popular in nano pharmaceutical research field and other applications. They are produced by different advanced methods. The preparation of these systems as well as of other lipid nanoparticle is possible and practicable in laboratory and on large scale. Additional efforts are needed to know the dynamics of LNPs on a molecular phase in vivo and in vitro.
Ethical Issues
Not applicable.
Conflict of Interest
The authors report no conflicts of interest.
References
- 1.Mukherjee S, Ray S, Thakur RS. Solid lipid nanoparticles: A modern formulation approach in drug delivery system. Indian J Pharm Sci. 2009;71(4):349–58. doi: 10.4103/0250-474X.57282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ekambaram P, Sathali A, Priyanka K. Solid lipid nanoparticles: a review. Sci Rev Chem Commun. 2012;2(1):80–102. [Google Scholar]
- 3.Kamble MS, Vaidya KK, Bhosale AV, Chaudhari PD. Solid lipid nanoparticles and nanostructured lipid carriers–an overview. Int J Pharm chem Biol Sci. 2012;2(4):681–91. [Google Scholar]
- 4.Kumar A, Badde S, Kamble R, Pokharkar VB. Development and characterization of liposomal drug delivery system for nimesulide. Int J Pharm Pharm Sci. 2010;2(4):87–9. [Google Scholar]
- 5.Dwivedi C, Sahu R, Tiwari SP, Satapathy T, Roy A. Role of liposome in novel drug delivery system. J Drug Deliv Therap. 2014;4(2):116–29. [Google Scholar]
- 6.Madan JR, Khude PA, Dua K. Development and evaluation of solid lipid nanoparticles of mometasone furoate for topical delivery. Int J Pharm Investig. 2014;4(2):60–4. doi: 10.4103/2230-973X.133047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Patidar A, Thakur DS, Kumar P, Verma J. A review on novel lipid based nanocarriers. Int J Pharm Pharm Sci. 2010;2(4):30–5. [Google Scholar]
- 8. Attama AA, Momoh MA, Builders PF. Lipid nanoparticulate drug delivery systems: a revolution in dosage form design and development. In: Sezer AD, editor. Pharmacology, Toxicology and Pharmaceutical Science. Croatia: InTech Open Access Publisher; 2012.
- 9.Elnaggar YS, El-Massik MA, Abdallah OY. Fabrication, appraisal, and transdermal permeation of sildenafil citrate-loaded nanostructured lipid carriers versus solid lipid nanoparticles. Int J Nanomedicine. 2011;6:3195–205. doi: 10.2147/IJN.S25825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Kaur J, Singh G, Saini S, Rana A. Innivative growth in developing new methods for formulating solid lipid nanoparticles and microparticles. J Drug Deliv Therap 2012;2(5).
- 11.Waghmare A, Grampurohit N, Gadhave M, Gaikwad D, Jadhav S. Solid lipid nanoparticles: a promising drug delivery system. Int Res J Pharm. 2012;3(4):100–7. [Google Scholar]
- 12.Das S, Chaudhury A. Recent advances in lipid nanoparticle formulations with solid matrix for oral drug delivery. AAPS PharmSciTech. 2011;12(1):62–76. doi: 10.1208/s12249-010-9563-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Sinha VR, Srivastava S, Goel H, Jindal V. Solid lipid nanoparticles (SLNs)–trends and implications in drug targeting. Int J Adv Pharm Sci 2011;1(3).
- 14.Mehnert W, Mader K. Solid lipid nanoparticles: Production, characterization and applications. Adv Drug Deliv Rev. 2001;47(2-3):165–96. doi: 10.1016/s0169-409x(01)00105-3. [DOI] [PubMed] [Google Scholar]
- 15. Guimarães KL, Ré MI. Lipid nanoparticles as carriers for cosmetic ingredients: The first (SLN) and the second generation (NLC). In: Beck R, Guterres S, Pohlmann A, editors. Nanocosmetics and Nanomedicines: New approaches for skin care. Germany: Springer; 2011.
- 16.Müller RH, Staufenbiel S, Keck CM. Lipid Nanoparticles (SLN, NLC) for innovative consumer care & household products. Househ Personal Care. 2014;9(2):18–25. [Google Scholar]
- 17.Pardeike J, Hommoss A, Muller RH. Lipid nanoparticles (sln, nlc) in cosmetic and pharmaceutical dermal products. Int J Pharm. 2009;366(1-2):170–84. doi: 10.1016/j.ijpharm.2008.10.003. [DOI] [PubMed] [Google Scholar]
- 18.Thatipamula R, Palem C, Gannu R, Mudragada S, Yamsani M. Formulation and in vitro characterization of domperidone loaded solid lipid nanoparticles and nanostructured lipid carriers. Daru. 2011;19(1):23–32. [PMC free article] [PubMed] [Google Scholar]
- 19.Blasi P, Giovagnoli S, Schoubben A, Ricci M, Rossi C. Solid lipid nanoparticles for targeted brain drug delivery. Adv Drug Deliv Rev. 2007;59(6):454–77. doi: 10.1016/j.addr.2007.04.011. [DOI] [PubMed] [Google Scholar]
- 20.Muchow M, Maincent P, Muller RH. Lipid nanoparticles with a solid matrix (sln, nlc, ldc) for oral drug delivery. Drug Dev Ind Pharm. 2008;34(12):1394–405. doi: 10.1080/03639040802130061. [DOI] [PubMed] [Google Scholar]
- 21.Yuan H, Huang LF, Du YZ, Ying XY, You J, Hu FQ. et al. Solid lipid nanoparticles prepared by solvent diffusion method in a nanoreactor system. Colloids Surf B Biointerfaces. 2008;61(2):132–7. doi: 10.1016/j.colsurfb.2007.07.015. [DOI] [PubMed] [Google Scholar]
- 22.Yoon G, Park JW, Yoon IS. Solid lipid nanoparticles (SLNs) and nanostructured lipid carriers (NLCs): recent advances in drug delivery. Int J Pharm Investig. 2013;43(5):353–62. [Google Scholar]
- 23.Lima AM, Pizzol CD, Monteiro FB, Creczynski-Pasa TB, Andrade GP, Ribeiro AO. et al. Hypericin encapsulated in solid lipid nanoparticles: Phototoxicity and photodynamic efficiency. J Photochem Photobiol B. 2013;125:146–54. doi: 10.1016/j.jphotobiol.2013.05.010. [DOI] [PubMed] [Google Scholar]
- 24.Kim BD, Na K, Choi HK. Preparation and characterization of solid lipid nanoparticles (sln) made of cacao butter and curdlan. Eur J Pharm Sci. 2005;24(2-3):199–205. doi: 10.1016/j.ejps.2004.10.008. [DOI] [PubMed] [Google Scholar]
- 25.Pardeshi C, Rajput P, Belgamwar V, Tekade A, Patil G, Chaudhary K. et al. Solid lipid based nanocarriers: An overview. Acta Pharm. 2012;62(4):433–72. doi: 10.2478/v10007-012-0040-z. [DOI] [PubMed] [Google Scholar]
- 26.Wen Z, Liu B, Zheng Z, You X, Pu Y, Li Q. Preparation of liposomes entrapping essential oil from Atractylodes macrocephala Koidz by modified RESS technique. Chem Eng Res Des. 2010;88(8):1102–7. [Google Scholar]
- 27.Feng L, Mumper RJ. A critical review of lipid-based nanoparticles for taxane delivery. Cancer Lett. 2013;334(2):157–75. doi: 10.1016/j.canlet.2012.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Wang S, Chen T, Chen R, Hu Y, Chen M, Wang Y. Emodin loaded solid lipid nanoparticles: Preparation, characterization and antitumor activity studies. Int J Pharm. 2012;430(1-2):238–46. doi: 10.1016/j.ijpharm.2012.03.027. [DOI] [PubMed] [Google Scholar]
- 29.Mendes AI, Silva AC, Catita JA, Cerqueira F, Gabriel C, Lopes CM. Miconazole-loaded nanostructured lipid carriers (nlc) for local delivery to the oral mucosa: Improving antifungal activity. Colloids Surf B Biointerfaces. 2013;111:755–63. doi: 10.1016/j.colsurfb.2013.05.041. [DOI] [PubMed] [Google Scholar]
- 30.Wang S, Su R, Nie S, Sun M, Zhang J, Wu D. et al. Application of nanotechnology in improving bioavailability and bioactivity of diet-derived phytochemicals. J Nutr Biochem. 2014;25(4):363–76. doi: 10.1016/j.jnutbio.2013.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Muller RH, Radtke M, Wissing SA. Solid lipid nanoparticles (sln) and nanostructured lipid carriers (nlc) in cosmetic and dermatological preparations. Adv Drug Deliv Rev. 2002;54 Suppl 1:S131–55. doi: 10.1016/s0169-409x(02)00118-7. [DOI] [PubMed] [Google Scholar]
- 32.Uner M, Yener G. Importance of solid lipid nanoparticles (sln) in various administration routes and future perspectives. Int J Nanomedicine. 2007;2(3):289–300. [PMC free article] [PubMed] [Google Scholar]
- 33.Tamjidi F, Shahedi M, Varshosaz J, Nasirpour A. Nanostructured lipid carriers (NLC): A potential delivery system for bioactive food molecules. Innov Food Sci Emerg Tech. 2013;19(0):29–43. [Google Scholar]
- 34.Beloqui A, Solinis MA, des Rieux A, Preat V, Rodriguez-Gascon A. Dextran-protamine coated nanostructured lipid carriers as mucus-penetrating nanoparticles for lipophilic drugs. Int J Pharm. 2014;468(1-2):105–11. doi: 10.1016/j.ijpharm.2014.04.027. [DOI] [PubMed] [Google Scholar]
- 35.Doktorovova S, Souto EB, Silva AM. Nanotoxicology applied to solid lipid nanoparticles and nanostructured lipid carriers - a systematic review of in vitro data. Eur J Pharm Biopharm. 2014;87(1):1–18. doi: 10.1016/j.ejpb.2014.02.005. [DOI] [PubMed] [Google Scholar]
- 36.Araujo J, Gonzalez-Mira E, Egea MA, Garcia ML, Souto EB. Optimization and physicochemical characterization of a triamcinolone acetonide-loaded nlc for ocular antiangiogenic applications. Int J Pharm. 2010;393(1-2):167–75. doi: 10.1016/j.ijpharm.2010.03.034. [DOI] [PubMed] [Google Scholar]
- 37.Sarathchandiran I. A Review on Nanotechnology in Solid Lipid Nanoparticles. Int J Pharm Deve Tech. 2012;2(1):45–61. [Google Scholar]
- 38.Cipolla D, Shekunov B, Blanchard J, Hickey A. Lipid-based carriers for pulmonary products: Preclinical development and case studies in humans. Adv Drug Deliv Rev. 2014;75:53–80. doi: 10.1016/j.addr.2014.05.001. [DOI] [PubMed] [Google Scholar]
- 39. Jaiswal P, Gidwani B, Vyas A. Nanostructured lipid carriers and their current application in targeted drug delivery. Artif Cells Nanomed Biotechnol 2014. [DOI] [PubMed]
- 40.Kumar S, Dilbaghi N, Saharan R, Bhanjana G. Nanotechnology as Emerging Tool for Enhancing Solubility of Poorly Water-Soluble Drugs. Bio Nano Sci. 2012;2(4):227–50. [Google Scholar]
- 41.Jagdevappa P, Prashant G, Ravindra K, Sachin J, Satish M, Meghanath Meghanath S. Meghanath Applications of Solid Lipid Nanoparticle in Novel Drug Delivery System. Br Biomed Bull. 2013;1(2):103–18. [Google Scholar]
- 42.Muller RH, Mader K, Gohla S. Solid lipid nanoparticles (sln) for controlled drug delivery - a review of the state of the art. Eur J Pharm Biopharm. 2000;50(1):161–77. doi: 10.1016/s0939-6411(00)00087-4. [DOI] [PubMed] [Google Scholar]
- 43.Selvamuthukumar S, Velmurugan R. Nanostructured lipid carriers: A potential drug carrier for cancer chemotherapy. Lipids Health Dis. 2012;11:159. doi: 10.1186/1476-511X-11-159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Alhaj N, Abdullah R, Ibrahim S, Bustamam A. Tamoxifen drug loading solid lipid nanoparticles prepared by hot high pressure homogenization techniques. Am J Pharmacol Toxicol. 2008;3(3):219. [Google Scholar]
- 45.Sunil PC, Vimal K. Production Techniques of Lipid Nanoparticles: A Review. Res J Pharm Bio Chem Sci. 2012;3(3):525. [Google Scholar]
- 46.Silva AC, Gonzalez-Mira E, Garcia ML, Egea MA, Fonseca J, Silva R. et al. Preparation, characterization and biocompatibility studies on risperidone-loaded solid lipid nanoparticles (sln): High pressure homogenization versus ultrasound. Colloids Surf B Biointerfaces. 2011;86(1):158–65. doi: 10.1016/j.colsurfb.2011.03.035. [DOI] [PubMed] [Google Scholar]
- 47.Parhi R, Suresh P. Preparation and characterization of solid lipid nanoparticles-a review. Curr Drug Discov Technol. 2012;9(1):2–16. doi: 10.2174/157016312799304552. [DOI] [PubMed] [Google Scholar]
- 48.Yadav V, AlokMahor S, Alok S, AmitaVerma A, Kumar N, Kumar S. Solid lipid nanoparticles (sln): formulation by high pressure homogenization. World J Pharm Pharm Sci. 2014;3(11):1200–13. [Google Scholar]
- 49.Parhi R, Suresh P. Production of Solid Lipid Nanoparticles-Drug Loading and Release Mechanism. J Chem Pharm Res. 2010;2(1):211–27. [Google Scholar]
- 50.Kamboj S, Bala S, Nair A. Solid lipid nanoparticles: an effective lipid based technology for poorly water soluble drugs. Int J Pharm Sci Rev Res. 2010;5(2):78–90. [Google Scholar]
- 51. Kaur T, Slavcev R. Solid Lipid Nanoparticles: Tuneable Anti-Cancer Gene/Drug Delivery Systems. In: Wei M, Good D, editors. Novel Gene Therapy Approches. Intech Publisher; 2013.
- 52.Chattopadhyay P, Shekunov BY, Yim D, Cipolla D, Boyd B, Farr S. Production of solid lipid nanoparticle suspensions using supercritical fluid extraction of emulsions (sfee) for pulmonary delivery using the aerx system. Adv Drug Deliv Rev. 2007;59(6):444–53. doi: 10.1016/j.addr.2007.04.010. [DOI] [PubMed] [Google Scholar]
- 53.Manjunath K, Reddy JS, Venkateswarlu V. Solid lipid nanoparticles as drug delivery systems. Methods Find Exp Clin Pharmacol. 2005;27(2):127–44. doi: 10.1358/mf.2005.27.2.876286. [DOI] [PubMed] [Google Scholar]
- 54.Patwekar S, Gattani S, Giri R, Bade A, Sangewar B, Raut V. Review on nanoparticles used in cosmetics and dermal products. World J Pharm Pharm Sci. 2014;3(8):1407–21. [Google Scholar]
- 55.Mukherjee S, Ray S, Thakur RS. Solid lipid nanoparticles: A modern formulation approach in drug delivery system. Indian J Pharm Sci. 2009;71(4):349–58. doi: 10.4103/0250-474X.57282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Silva AC, Kumar A, Wild W, Ferreira D, Santos D, Forbes B. Long-term stability, biocompatibility and oral delivery potential of risperidone-loaded solid lipid nanoparticles. Int J Pharm. 2012;436(1-2):798–805. doi: 10.1016/j.ijpharm.2012.07.058. [DOI] [PubMed] [Google Scholar]
- 57.Padhye SG, Nagarsenker MS. Simvastatin solid lipid nanoparticles for oral delivery: Formulation development and in vivo evaluation. Indian J Pharm Sci. 2013;75(5):591–8. [PMC free article] [PubMed] [Google Scholar]
- 58.Mu H, Holm R, Mullertz A. Lipid-based formulations for oral administration of poorly water-soluble drugs. Int J Pharm. 2013;453(1):215–24. doi: 10.1016/j.ijpharm.2013.03.054. [DOI] [PubMed] [Google Scholar]
- 59.Jawahar N, Meyyanathan SN, Reddy G, Sood S. Solid lipid nanoparticles for oral delivery of poorly soluble drugs. J Pharm Sci Res. 2012;4(7):1848–55. [Google Scholar]
- 60.Jawahar N, Reddy G. Nanoparticles: A novel pulmonary drug delivery system for tuberculosis. J Pharm Sci Res. 2012;4:1901–6. [Google Scholar]
- 61. Souto EB, Muller RH. Lipid nanoparticles: Effect on bioavailability and pharmacokinetic changes. Handb Exp Pharmacol 2010(197):115-41. [DOI] [PubMed]
- 62.Paranjpe M, Muller-Goymann CC. Nanoparticle-mediated pulmonary drug delivery: A review. Int J Mol Sci. 2014;15(4):5852–73. doi: 10.3390/ijms15045852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Weber S, Zimmer A, Pardeike J. Solid Lipid Nanoparticles (SLN) and Nanostructured Lipid Carriers (NLC) for pulmonary application: A review of the state of the art. Eur J Pharm Biopharm. 2014;86(1):7–22. doi: 10.1016/j.ejpb.2013.08.013. [DOI] [PubMed] [Google Scholar]
- 64.Zhang Z, Sha X, Shen A, Wang Y, Sun Z, Gu Z. et al. Polycation nanostructured lipid carrier, a novel nonviral vector constructed with triolein for efficient gene delivery. Biochem Biophys Res Commun. 2008;370(3):478–82. doi: 10.1016/j.bbrc.2008.03.127. [DOI] [PubMed] [Google Scholar]
- 65.Shao Z, Shao J, Tan B, Guan S, Liu Z, Zhao Z. et al. Targeted lung cancer therapy: Preparation and optimization of transferrin-decorated nanostructured lipid carriers as novel nanomedicine for co-delivery of anticancer drugs and DNA. Int J Nanomedicine. 2015;10:1223–33. doi: 10.2147/IJN.S77837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Charan Teja VR, Harini Chowdary V, Prasanna Raju Y, Surendra N, Vishnu Vardhan R, Kiran Kumar Reddy B. A glimpse on solid lipid nanoparticles as drug delivery systems. J Glob Tren Pharm Sci. 2014;5(2):1649–57. [Google Scholar]
- 67.Nayerossadat N, Maedeh T, Ali PA. Viral and nonviral delivery systems for gene delivery. Adv Biomed Res. 2012;1:27. doi: 10.4103/2277-9175.98152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Balazs DA, Godbey WT. Liposomes for Use in Gene Delivery. J Drug Deliv. 2011;2011:326–497. doi: 10.1155/2011/326497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Tros de Ilarduya C, Sun Y, Duzgunes N. Gene delivery by lipoplexes and polyplexes. Eur J Pharm Sci. 2010;40(3):159–70. doi: 10.1016/j.ejps.2010.03.019. [DOI] [PubMed] [Google Scholar]
- 70.Stone D. Novel viral vector systems for gene therapy. Viruses. 2010;2(4):1002–7. doi: 10.3390/v2041002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Khosravi-Darani K, Mozafari M. Nanoliposome potentials in nanotherapy: A concise overview. Int J Nanosci Nanotech. 2010;6:3–13. [Google Scholar]
- 72. Han Y, Li Y, Zhang P, Sun J, Li X, Sun X, et al. Nanostructured lipid carriers as novel drug delivery system for lung cancer gene therapy. Pharm Dev Technol 2015:1-5. [DOI] [PubMed]
- 73.Lai F, Wissing SA, Muller RH, Fadda AM. Artemisia arborescens l essential oil-loaded solid lipid nanoparticles for potential agricultural application: Preparation and characterization. AAPS PharmSciTech. 2006;7(1):E2. doi: 10.1208/pt070102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Weiss J, Decker EA, McClements DJ, Kristbergsson K, Helgason T, Awad T. Solid lipid nanoparticles as delivery systems for bioactive food components. Food Biophys. 2008;3(2):146–54. [Google Scholar]
- 75.Lason E, Ogonowski J. Solid Lipid Nanoparticles – characteristics, application and obtaining. Chemik. 2011;65(10):960–67. [Google Scholar]
- 76. Oehlke K, Walz E, Graef V, Greiner R. Technological and nutritional aspects of solid lipid nanoparticles added to o/w emulsions. Available from: http://www.icef11.org/content/papers/fms/FMS663.pdf.


