Summary
Introduction
The present review serves to provide a concise overview of the current knowledge on therapeutic strategies with regard to fibrostenotic lesions in Crohn's disease.
Methods
A literature search was performed focusing on the last 5 years, and current concepts of pathophysiology, epidemiology, and treatment have been summarized.
Results
Fibrostenotic lesions in Crohn's disease are currently considered to be a consequence of the chronic inflammatory nature of the disease. Hence, therapeutic strategies are limited to the concept that early treatment of the inflammatory lesions can prevent structural changes, and to various endoscopic and surgical approaches. Direct targeting of the fibrostenotic lesion itself has not been the focus until now. This review will provide an overview of the pathophysiology and epidemiology of fibrostenotic lesions including current therapeutic approaches. Since research with regard to other organ systems and fibrosis is far more advanced, current strategies from available studies in these areas will be discussed. The results and the potential impact for Crohn's disease will be considered.
Conclusion
The vision of these approaches is to reverse structural changes and restore normal function.
Keywords: Crohn's disease, Stenosis, Fibrosis
Pathophysiologic Thoughts and Background
A primary hit is required in order to induce an accumulation of extracellular matrix subsequently leading to fibrotic lesions [1]. The chronic inflammatory condition existing in Crohn's disease suffices to induce and perpetuate the progression of fibrosis [1]; however, disease chronicity is a prerequisite since acute and even ulcerating intestinal inflammation can heal with complete restitution of the intestinal structure. While several molecules and factors have been associated with fibrosis, the question arises which of these have been found to be of functional impact.
In the animal model of trinitrobenzene sulfonic acid (TNBS)-induced colitis, inhibition of interleukin (IL)-13 signaling was sufficient to attenuate intestinal fibrosis [2]. Work from the same group indicated that IL-13 production induced transforming growth factor (TGF)-β1 production of macrophages [3]. In line with this, increased expression of IL-13 was found in intestinal smooth muscle from Crohn's disease patients [4]. However, another study did not indicate any differences in tissue IL-13 expression in Crohn's disease patients [5].
In other organs, the IL-17 subtypes A and E have been shown to exert profibrotic activity including cardiac fibroblasts, hepatic stellate cells, skin fibroblasts, and lung epithelial cells [6,7,8,9]. Accordingly, IL-17 tissue concentrations were increased in models for intestinal inflammation [10]. In fibrosing Crohn's disease, tissue IL-17A but not IL-17E has been found to be up-regulated. Furthermore, myofibroblasts from Crohn's disease strictures express the IL-17A receptor and indicate more collagen production [11]. Strikingly, in a clinical trial in which IL-17A was blocked with secukinumab, this strategy failed to improve disease activity in Crohn's disease [12], indicating the complex nature of this network.
As introduced above, the central molecule mediating fibrosis is TGF-β1 [13]. Hence the TGF-β/Smad signaling pathway is pivotal for the development of fibrosis [13,14]. Additional recent evidence provides further insight into the underlying mechanistic pathways. Several cytokines including IL-6 are released by activated mesenchymal cells resulting in an activation of the STAT3 pathway. Remarkably, a unique pattern emerged in patients with Montreal B2 Crohn's disease, namely a phospho-STAT3(S727) response. In this study, neutralization of this pathway led to normalized expression of TGF-β1. The strategy of this approach was then functionally proven in the model of TNBS colitis where fibrosis development could be decreased in mice treated with a STAT3 inhibitor [15]. Besides cytokines, the balance of the extracellular matrix in between matrix metalloproteinases and tissue inhibitors of metalloproteinases (TIMP)-1 is critical for maintaining a healthy state [16,17].
Associated with recent advances in the understanding of the intestinal microbiota, evidence indicates that ligands to receptors of the innate immune system result in the activation of the NFκB pathway in intestinal mesenchymal cells [18]. Experimental models indicate that this ultimately leads to the development of inflammation and fibrosis [19]. Hence, the direct contact with microbiota is unique for intestinal fibrosis, in particular when compared to other organ systems where fibrosis plays a role. Nevertheless, the concept of intestinal fibrosis is still lacking significant information required to develop more specific therapeutic approaches. Hence, with regard to future strategies, we will focus on approaches developed for other organ systems.
Epidemiology
The incidence of Crohn's disease has continuously increased over the past 50 years [20,21,22]. For instance, in Northern France, it has increased from 5.3/100,000 in 1988 to 7/100,000 in 1999 [22]. The understanding of the natural history of Crohn's disease has improved mainly owing to the postoperative recurrence model [23,24]. This model depicts the inflammatory nature of the disease ultimately resulting in structural damage. According to this model, as early as 8 days after surgery for Crohn's disease, a first localized inflammatory infiltrate can be found in the ileum above the anastomosis [25]. This is followed by the development of aphthous lesions that are visible in as many as 66% of patients within 3 months post surgery. In a next step, inflammation precedes the development of ulcers and strictures [24] and hence structural changes. These strictures can be associated with fistulas [26]. This model equally applies to the progression of Crohn's disease without surgery. However, the time of progression can vary from weeks to years, and can be stopped or even reversed, spontaneously or through treatment. Population-based data from Denmark indicate that after 1 year of being diagnosed, 55% of Crohn's disease patients are in remission and 15% only have mild disease [27]. However, up to one third of patients will have highly active disease. Comparably, data from Olmstedt County revealed that 64.4% of the follow-up time of patients is characterized by medical or surgical remission [28].
While superficial lesions tend to heal without structural residues, a stricture associated with prestenotic dilatation can currently not be reversed. This stricturing phenotype reflects the development of fibrostenotic lesions and tends to recur even after surgery [29,30]. The development of strictures strongly depends on the disease distribution. Here, ileal disease location results more frequently in stricturing disease than colonic disease location [31]. Strictures may be subdivided into fibrotic and inflammatory as well as mixed forms [32].
In Crohn's disease, about 50% of patients develop complications such as strictures, fistulas, and abscesses that frequently require surgery within 10 years following the initial diagnosis [33,34]. Data from two cohorts (from Spain and Minnesota, respectively) covering two different time periods indicate that the number of surgeries has not decreased over time. One may conclude that applying clinical symptoms as treatment targets fails to ensure that the underlying condition is adequately controlled [35,36,37]. In line with the post-surgery model, patients with an early inflammatory course tend to develop more severe disease, hence there is a substantial need for strategies aimed at disrupting the natural disease course. This requires that high-risk groups can be identified early on. Variables associated with a complicated disease course include smoking, young age, steroids at diagnosis, perianal disease, and extraintestinal manifestations [38,39,40,41,42]. Considering these aspects surrounding the natural history and evolution of the disease, one could conclude that patients at highest risk require early aggressive therapy in order to prevent structural damage. However, prospective trials proving this concept are currently lacking.
Current Therapeutic Concepts
Considering the above stated, strictures including inflammatory alterations might benefit from anti-inflammatory therapy through decreasing the inflammation-driven edema [43]. However, recent epidemiological data indicate that although early immunosuppressive therapy has been introduced in Crohn's disease patients with an increased risk for a more aggressive disease course, the frequency of stricturing disease did not change [44]. However, as already emphasized above, controlled prospective trials on this matter are lacking. When considering current medical strategies applied in Crohn's disease, steroids have been shown to decrease collagen production in vivo and to inhibit collagen activity [45]. In humans, steroid treatment exerted some antifibrotic effects in retroperitoneal fibrosis [46], systemic sclerosis [47,] and idiopathic pulmonary fibrosis [48]. While local injections of steroids showed some efficacy in Crohn's disease [49], intestinal fibroblasts responded to steroids with enhanced collagen expression [50]. In addition, long-term steroid treatment is obsolete in patients with intestinal bowel disease. Azathioprine has shown some beneficial effects in retroperitoneal fibrosis [51] and fibrotic pulmonary disease [52,53]. In Crohn's disease, there is limited evidence indicating a delay in post-surgery fibrostenotic complications [54]. In line with this, anti-TNFα strategies exerted antifibrotic effects in liver [55] and pulmonary fibrosis [56] as well as systemic sclerosis [57]. This can be explained by TNFα-mediated myofibroblast activation, increased collagen production, TIMP-1 expression, and a decrease in MMP2 activity and collagen degradation [58]. Retrospective data indicate a partial amelioration of stricture formation with anti-TNFα therapy [59] and a delay in the time to surgery [60].
Nevertheless, specific antifibrotic strategies are urgently required for the treatment of stricturing Crohn's disease. At this point, treatment strategies do not differ between inflammatory, penetrating, and stricturing disease.
Based on these data, the current treatment options are rather simplistic. Primary therapy comprises anti-inflammatory approaches as well as endoscopic dilatation and surgery [61,62]. Isolated strictures with a length below 5 cm and a lack of ulceration qualify for endoscopic dilatation [61,62,63,64]. While endoscopic dilatation can widely improve symptoms, a substantial part of these patients still require surgery in the long term [61,62]. Alternatively, dependent on the length of the stenosis, various surgical techniques are suitable. This includes classic stricturoplasty for short segments as well as more advanced techniques for longer segment strictures [65]. Endoscopy as well as surgery are associated with complications including perforation, infection, anastomotic leakage, abscess, and fistula [61,66,67]. In addition, even after surgery, there is a substantial recurrence rate [24,68,69]. This insufficient concept clearly highlights the need for more specific therapeutic approaches.
Future Options
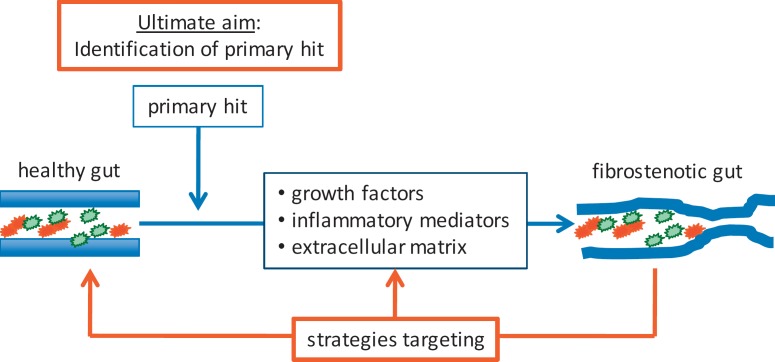
At this point, Crohn's disease patients with established strictures are considered to inevitably require surgery in the future. Nevertheless, experimental data suggest that intestinal fibrosis might disappear after elimination of the profibrotic stimulus [19]. In the following, mainly based on data from other organ systems, potential future strategies are discussed. A visual summary of the concept is provided in figure 1.
Fig. 1.
Development of fibrostenotic lesions in Crohn's disease and potential therapeutic approaches.
Based on the pathophysiological considerations discussed above, targeting the TGF-β1 pathway appears promising. Indeed, there is evidence from clinical trials outside the intestine supporting this concept [70,71]. However, TGF-β1-deficient mice early on develop a multi-organ lethal inflammatory syndrome [72,73]. In line with this, clinical trials assessing a monoclonal antibody targeting TGF-β1 in systemic sclerosis revealed relevant side effects [74]. From this, one might conclude that this strategy is not suitable as a systemic approach. An alternative option might be agents that affect the underlying pathway; this includes HMG-CoA reductase inhibitors as well as renin-angiotensin system modulators [75,76,77,78]. Similarly, PPAR-γ agonists antagonize the Smad3 pathway [79].
Another angle are the inflammatory mediators. Here IL-6 activates IL-13 followed by the induction of the TGF-β pathway via IL-13Rα [2]. As discussed above, antagonism of IL-13 is effective in preventing fibrosis development in experimental colitis [3,80]. Anti-IL-13 strategies are currently being evaluated for liver and pulmonary fibrosis [1,81,82,83]. Hence, although anti-IL-13 strategies failed in ulcerative colitis, they might represent a novel strategy in fibrostenotic Crohn's disease [84].
Several growth factors have been shown to be involved in the development of fibrosis. For instance, vascular endothelial growth factor (VEGF)-A has been described to be upregulated in inflammatory bowel diseases [85]]. Platelet-derived growth factor (PDGF) is known to increase proliferation and migration of fibroblasts and myofibroblasts [14]. In this context, in human intestine, PDGF facilitates extracellular matrix deposition and is upregulated in inflamed colonic tissue of Crohn's disease patients [86]. Blocking strategies from other organs suggest an antifibrotic effect in vivo [87].
Extracellular matrix modulators have been implicated in the pathogenesis of fibrostenotic diseases; however, current clinical and experimental data fail to show efficacy [88,89].
Lastly, mTOR inhibitors have shown direct antifibrotic properties by inhibition of IL-4, IL-6, IL-13, TGF-β, IL-17 as well as type I and III collagen [90,91]. Efficacy has been proven for other organ systems including lung, skin, kidney, and liver [14]. With regard to Crohn's disease, one randomized trial indicated that everolimus is as effective as azathioprine in achieving steroid-fee remission [92]. However, data specifically evaluating an antifibrotic effect in Crohn's disease are lacking.
Conclusion
Medical therapy for fibrostenotic Crohn's disease is still highly restricted and lacking innovative approaches. Several points will have to be addressed in the future to improve current therapy. Ultimately, identifying the first stimulus sufficient to activate the fibrostenotic pathway appears to be an intriguing aim, albeit at this point not for the near future. In the meantime, we should take advantage of the knowledge gained in other organ systems to facilitate the introduction of novel strategies in Crohn's disease. The vision that fibrostenotic lesions can be reversed would represent a complete change in the disease concept.
Disclosure Statement
BS has received speaker's fees from Abbvie, Ferring, Falk, MSD, Merck, and Takeda, and consultation fees from Abbvie, Falk, Hospira, Merck, MSD, Mundipharma, and Takeda.
References
- 1.Friedman SL, Sheppard D, Duffield JS, Violette S. Therapy for fibrotic diseases: nearing the starting line. Sci Transl Med. 2013;5:167sr161. doi: 10.1126/scitranslmed.3004700. [DOI] [PubMed] [Google Scholar]
- 2.Fichtner-Feigl S, Young CA, Kitani A, Geissler EK, Schlitt HJ, Strober W. IL-13 signaling via IL-13R alpha2 induces major downstream fibrogenic factors mediating fibrosis in chronic TNBS colitis. Gastroenterology. 2008;135:2003–2013. doi: 10.1053/j.gastro.2008.08.055. 2013.e2001-2007. [DOI] [PubMed] [Google Scholar]
- 3.Fichtner-Feigl S, Strober W, Kawakami K, Puri RK, Kitani A. IL-13 signaling through the IL-13alpha2 receptor is involved in induction of TGF-beta1 production and fibrosis. Nat Med. 2006;12:99–106. doi: 10.1038/nm1332. [DOI] [PubMed] [Google Scholar]
- 4.Bailey JR, Bland PW, Tarlton JF, Peters I, Moorghen M, Sylvester PA, Probert CS, Whiting CV. IL-13 promotes collagen accumulation in Crohn's disease fibrosis by down-regulation of fibroblast MMP synthesis: a role for innate lymphoid cells? PLoS One. 2012;7:e52332. doi: 10.1371/journal.pone.0052332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Biancheri P, Di Sabatino A, Ammoscato F, Facciotti F, Caprioli F, Curciarello R, Hoque SS, Ghanbari A, Joe-Njoku I, Giuffrida P, Rovedatti L, Geginat J, Corazza GR, MacDonald TT. Absence of a role for interleukin-13 in inflammatory bowel disease. Eur J Immunol. 2014;44:370–385. doi: 10.1002/eji.201343524. [DOI] [PubMed] [Google Scholar]
- 6.Valente AJ, Yoshida T, Gardner JD, Somanna N, Delafontaine P, Chandrasekar B. Interleukin-17a stimulates cardiac fibroblast proliferation and migration via negative regulation of the dual-specificity phosphatase MKP-1/DUSP-1. Cell Signal. 2012;24:560–568. doi: 10.1016/j.cellsig.2011.10.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Meng F, Wang K, Aoyama T, Grivennikov SI, Paik Y, Scholten D, Cong M, Iwaisako K, Liu X, Zhang M, Osterreicher CH, Stickel F, Ley K, Brenner DA, Kisseleva T. Interleukin-17 signaling in inflammatory, Kupffer cells, and hepatic stellate cells exacerbates liver fibrosis in mice. Gastroenterology. 2012;143:765–776. doi: 10.1053/j.gastro.2012.05.049. e1-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Okamoto Y, Hasegawa M, Matsushita T, Hamaguchi Y, Huu DL, Iwakura Y, Fujimoto M, Takehara K. Potential roles of interleukin-17a in the development of skin fibrosis in mice. Arthritis Rheum. 2012;64:3726–3735. doi: 10.1002/art.34643. [DOI] [PubMed] [Google Scholar]
- 9.Mi S, Li Z, Yang HZ, Liu H, Wang JP, Ma YG, Wang XX, Liu HZ, Sun W, Hu ZW. Blocking IL-17a promotes the resolution of pulmonary inflammation and fibrosis via TGF-beta1-dependent and -independent mechanisms. J Immunol. 2011;187:3003–3014. doi: 10.4049/jimmunol.1004081. [DOI] [PubMed] [Google Scholar]
- 10.Zhu MY, Lu YM, Ou YX, Zhang HZ, Chen WX. Dynamic progress of 2,4,6-trinitrobenzene sulfonic acid induced chronic colitis and fibrosis in rat model. J Dig Dis. 2012;13:421–429. doi: 10.1111/j.1751-2980.2012.00607.x. [DOI] [PubMed] [Google Scholar]
- 11.Biancheri P, Pender SL, Ammoscato F, Giuffrida P, Sampietro G, Ardizzone S, Ghanbari A, Curciarello R, Pasini A, Monteleone G, Corazza GR, Macdonald TT, Di Sabatino A. The role of interleukin 17 in Crohn's disease-associated intestinal fibrosis. Fibrogenesis Tissue Repair. 2013;6:13. doi: 10.1186/1755-1536-6-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hueber W, Sands BE, Lewitzky S, Vandemeulebroecke M, Reinisch W, Higgins PD, Wehkamp J, Feagan BG, Yao MD, Karczewski M, Karczewski J, Pezous N, Bek S, Bruin G, Mellgard B, Berger C, Londei M, Bertolino AP, Tougas G, Travis SP. Secukinumab, a human anti-IL-17a monoclonal antibody, for moderate to severe Crohn's disease: unexpected results of a randomised, double-blind placebo-controlled trial. Gut. 2012;61:1693–1700. doi: 10.1136/gutjnl-2011-301668. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hawinkels LJ, Ten Dijke P. Exploring anti-TGF-beta therapies in cancer and fibrosis. Growth Factors. 2011;29:140–152. doi: 10.3109/08977194.2011.595411. [DOI] [PubMed] [Google Scholar]
- 14.Latella G, Sferra R, Speca S, Vetuschi A, Gaudio E. Can we prevent, reduce or reverse intestinal fibrosis in IBD? Eur Rev Med Pharmacol Sci. 2013;17:1283–1304. [PubMed] [Google Scholar]
- 15.Li C, Iness A, Yoon J, Grider JR, Murthy KS, Kellum JM, Kuemmerle JF. Noncanonical STAT3 activation regulates excess TGF-β1 and collagen I expression in muscle of stricturing Crohn's disease. J Immunol. 2015;194:3422–3431. doi: 10.4049/jimmunol.1401779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.McKaig BC, McWilliams D, Watson SA, Mahida YR. Expression and regulation of tissue inhibitor of metalloproteinase-1 and matrix metalloproteinases by intestinal myofibroblasts in inflammatory bowel disease. Am J Pathol. 2003;162:1355–1360. doi: 10.1016/S0002-9440(10)63931-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Medina C, Santos-Martinez MJ, Santana A, Paz-Cabrera MC, Johnston MJ, Mourelle M, Salas A, Guarner F. Transforming growth factor-beta type 1 receptor (ALK5) and Smad proteins mediate TIMP-1 and collagen synthesis in experimental intestinal fibrosis. J Pathol. 2011;224:461–472. doi: 10.1002/path.2870. [DOI] [PubMed] [Google Scholar]
- 18.Otte JM, Rosenberg IM, Podolsky DK. Intestinal myofibroblasts in innate immune responses of the intestine. Gastroenterology. 2003;124:1866–1878. doi: 10.1016/s0016-5085(03)00403-7. [DOI] [PubMed] [Google Scholar]
- 19.Rieder F, Kessler S, Sans M, Fiocchi C. Animal models of intestinal fibrosis: new tools for the understanding of pathogenesis and therapy of human disease. Am J Physiol Gastrointest Liver Physiol. 2012;303:G786–801. doi: 10.1152/ajpgi.00059.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Vind I, Riis L, Jess T, Knudsen E, Pedersen N, Elkjaer M, Bak Andersen I, Wewer V, Norregaard P, Moesgaard F, Bendtsen F, Munkholm P. Increasing incidences of inflammatory bowel disease and decreasing surgery rates in Copenhagen City and County, 2003-2005: a population-based study from the Danish Crohn colitis database. Am J Gastroenterol. 2006;101:1274–1282. doi: 10.1111/j.1572-0241.2006.00552.x. [DOI] [PubMed] [Google Scholar]
- 21.Yapp TR, Stenson R, Thomas GA, Lawrie BW, Williams GT, Hawthorne AB. Crohn's disease incidence in Cardiff from 1930: an update for 1991-1995. Eur J Gastroenterol Hepatol. 2000;12:907–911. doi: 10.1097/00042737-200012080-00010. [DOI] [PubMed] [Google Scholar]
- 22.Jacobsen BA, Fallingborg J, Rasmussen HH, Nielsen KR, Drewes AM, Puho E, Nielsen GL, Sorensen HT. Increase in incidence and prevalence of inflammatory bowel disease in Northern Denmark: a population-based study, 1978-2002. Eur J Gastroenterol Hepatol. 2006;18:601–606. doi: 10.1097/00042737-200606000-00005. [DOI] [PubMed] [Google Scholar]
- 23.Zvidi I, Hazazi R, Birkenfeld S, Niv Y. The prevalence of Crohn's disease in Israel: a 20-year survey. Dig Dis Sci. 2009;54:848–852. doi: 10.1007/s10620-008-0429-1. [DOI] [PubMed] [Google Scholar]
- 24.Rutgeerts P, Geboes K, Vantrappen G, Beyls J, Kerremans R, Hiele M. Predictability of the postoperative course of Crohn's disease. Gastroenterology. 1990;99:956–963. doi: 10.1016/0016-5085(90)90613-6. [DOI] [PubMed] [Google Scholar]
- 25.Olaison G, Smedh K, Sjodahl R. Natural course of Crohn's disease after ileocolic resection: endoscopically visualised ileal ulcers preceding symptoms. Gut. 1992;33:331–335. doi: 10.1136/gut.33.3.331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Oberhuber G, Stangl PC, Vogelsang H, Schober E, Herbst F, Gasche C. Significant association of strictures and internal fistula formation in Crohn's disease. Virchows Arch. 2000;437:293–297. doi: 10.1007/s004280000226. [DOI] [PubMed] [Google Scholar]
- 27.Munkholm P, Langholz E, Davidsen M, Binder V. Disease activity courses in a regional cohort of Crohn's disease patients. Scand J Gastroenterol. 1995;30:699–706. doi: 10.3109/00365529509096316. [DOI] [PubMed] [Google Scholar]
- 28.Silverstein MD, Loftus EV, Sandborn WJ, Tremaine WJ, Feagan BG, Nietert PJ, Harmsen WS, Zinsmeister AR. Clinical course and costs of care for Crohn's disease: Markov model analysis of a population-based cohort. Gastroenterology. 1999;117:49–57. doi: 10.1016/s0016-5085(99)70549-4. [DOI] [PubMed] [Google Scholar]
- 29.Louis E, Collard A, Oger AF, Degroote E, Aboul Nasr El Yafi FA, Belaiche J. Behaviour of Crohn's disease according to the Vienna classification: changing pattern over the course of the disease. Gut. 2001;49:777–782. doi: 10.1136/gut.49.6.777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Cosnes J, Cattan S, Blain A, Beaugerie L, Carbonnel F, Parc R, Gendre JP. Long-term evolution of disease behavior of Crohn's disease. Inflamm Bowel Dis. 2002;8:244–250. doi: 10.1097/00054725-200207000-00002. [DOI] [PubMed] [Google Scholar]
- 31.Cosnes J. Crohn's disease phenotype, prognosis, and long-term complications: what to expect? Acta Gastroenterol Belg. 2008;71:303–307. [PubMed] [Google Scholar]
- 32.Lenze F, Wessling J, Bremer J, Ullerich H, Spieker T, Weckesser M, Gonschorrek S, Kannengiesser K, Rijcken E, Heidemann J, Luegering A, Schober O, Domschke W, Kucharzik T, Maaser C. Detection and differentiation of inflammatory versus fibromatous Crohn's disease strictures: prospective comparison of 18f- FDG-PET/CT, MR-enteroclysis, and transabdominal ultrasound versus endoscopic/histologic evaluation. Inflamm Bowel Dis. 2012;18:2252–2260. doi: 10.1002/ibd.22930. [DOI] [PubMed] [Google Scholar]
- 33.Peyrin-Biroulet L, Loftus EV, Jr, Colombel JF, Sandborn WJ. The natural history of adult Crohn's disease in population-based cohorts. Am J Gastroenterol. 2010;105:289–297. doi: 10.1038/ajg.2009.579. [DOI] [PubMed] [Google Scholar]
- 34.Solberg IC, Vatn MH, Hoie O, Stray N, Sauar J, Jahnsen J, Moum B, Lygren I. Clinical course in Crohn's disease: results of a Norwegian population-based ten-year follow-up study. Clin Gastroenterol Hepatol. 2007;5:1430–1438. doi: 10.1016/j.cgh.2007.09.002. [DOI] [PubMed] [Google Scholar]
- 35.Cannom RR, Kaiser AM, Ault GT, Beart RW, Jr, Etzioni DA. Inflammatory bowel disease in the United States from 1998 to 2005: has infliximab affected surgical rates? Am Surg. 2009;75:976–980. [PubMed] [Google Scholar]
- 36.Domenech E, Zabana Y, Garcia-Planella E, Lopez San Roman A, Nos P, Ginard D, Gordillo J, Martinez-Silva F, Beltran B, Manosa M, Cabre E, Gassull MA. Clinical outcome of newly diagnosed Crohn's disease: a comparative, retrospective study before and after infliximab availability. Aliment Pharmacol Ther. 2010;31:233–239. doi: 10.1111/j.1365-2036.2009.04170.x. [DOI] [PubMed] [Google Scholar]
- 37.Jones DW, Finlayson SR. Trends in surgery for Crohn's disease in the era of infliximab. Ann Surg. 2010;252:307–312. doi: 10.1097/SLA.0b013e3181e61df5. [DOI] [PubMed] [Google Scholar]
- 38.Beaugerie L, Seksik P, Nion-Larmurier I, Gendre JP, Cosnes J. Predictors of Crohn's disease. Gastroenterology. 2006;130:650–656. doi: 10.1053/j.gastro.2005.12.019. [DOI] [PubMed] [Google Scholar]
- 39.Wolters FL, Russel MG, Sijbrandij J, Schouten LJ, Odes S, Riis L, Munkholm P, Langholz E, Bodini P, O'Morain C, Katsanos K, Tsianos E, Vermeire S, Van Zeijl G, Limonard C, Hoie O, Vatn M, Moum B, Stockbrugger RW. Disease outcome of inflammatory bowel disease patients: general outline of a Europe-wide population-based 10-year clinical follow-up study. Scand J Gastroenterol Suppl. 2006;243:46–54. doi: 10.1080/00365520600664250. [DOI] [PubMed] [Google Scholar]
- 40.Lichtenstein GR, Olson A, Travers S, Diamond RH, Chen DM, Pritchard ML, Feagan BG, Cohen RD, Salzberg BA, Hanauer SB, Sandborn WJ. Factors associated with the development of intestinal strictures or obstructions in patients with Crohn's disease. Am J Gastroenterol. 2006;101:1030–1038. doi: 10.1111/j.1572-0241.2006.00463.x. [DOI] [PubMed] [Google Scholar]
- 41.Buning C, Genschel J, Buhner S, Kruger S, Kling K, Dignass A, Baier P, Bochow B, Ockenga J, Schmidt HH, Lochs H. Mutations in the NOD2/CARD15 gene in Crohn's disease are associated with ileocecal resection and are a risk factor for reoperation. Aliment Pharmacol Ther. 2004;19:1073–1078. doi: 10.1111/j.1365-2036.2004.01967.x. [DOI] [PubMed] [Google Scholar]
- 42.Franchimont DP, Louis E, Croes F, Belaiche J. Clinical pattern of corticosteroid dependent Crohn's disease. Eur J Gastroenterol Hepatol. 1998;10:821–825. doi: 10.1097/00042737-199810000-00001. [DOI] [PubMed] [Google Scholar]
- 43.Schoepfer AM, Safroneeva E, Vavricka SR, Peyrin-Biroulet L, Mottet C. Treatment of fibrostenotic and fistulizing Crohn's disease. Digestion. 2012;86(suppl 1):23–27. doi: 10.1159/000341961. [DOI] [PubMed] [Google Scholar]
- 44.Cosnes J, Nion-Larmurier I, Beaugerie L, Afchain P, Tiret E, Gendre JP. Impact of the increasing use of immunosuppressants in Crohn's disease on the need for intestinal surgery. Gut. 2005;54:237–241. doi: 10.1136/gut.2004.045294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Oikarinen AI, Vuorio EI, Zaragoza EJ, Palotie A, Chu ML, Uitto J. Modulation of collagen metabolism by glucocorticoids. Receptor-mediated effects of dexamethasone on collagen biosynthesis in chick embryo fibroblasts and chondrocytes. Biochem Pharmacol. 1988;37:1451–1462. doi: 10.1016/0006-2952(88)90006-8. [DOI] [PubMed] [Google Scholar]
- 46.Vaglio A, Palmisano A, Alberici F, Maggiore U, Ferretti S, Cobelli R, Ferrozzi F, Corradi D, Salvarani C, Buzio C. Prednisone versus tamoxifen in patients with idiopathic retroperitoneal fibrosis: an open-label randomised controlled trial. Lancet. 2011;378:338–346. doi: 10.1016/S0140-6736(11)60934-3. [DOI] [PubMed] [Google Scholar]
- 47.Badea I, Taylor M, Rosenberg A, Foldvari M. Pathogenesis and therapeutic approaches for improved topical treatment in localized scleroderma and systemic sclerosis. Rheumatology (Oxford) 2009;48:213–221. doi: 10.1093/rheumatology/ken405. [DOI] [PubMed] [Google Scholar]
- 48.Peikert T, Daniels CE, Beebe TJ, Meyer KC, Ryu JH. Assessment of current practice in the diagnosis and therapy of idiopathic pulmonary fibrosis. Respir Med. 2008;102:1342–1348. doi: 10.1016/j.rmed.2008.03.018. [DOI] [PubMed] [Google Scholar]
- 49.Kochar R, Dupont AW. Primary and secondary prophylaxis of gastric variceal bleeding. F1000 Med Rep. 2010;2 doi: 10.3410/M2-26. pii:26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Graham MF, Willey A, Adams J, Diegelmann RF. Corticosteroids increase procollagen gene expression, synthesis, and secretion by human intestinal smooth muscle cells. Gastroenterology. 1995;109:1454–1461. doi: 10.1016/0016-5085(95)90630-4. [DOI] [PubMed] [Google Scholar]
- 51.Warnatz K, Keskin AG, Uhl M, Scholz C, Katzenwadel A, Vaith P, Peter HH, Walker UA. Immunosuppressive treatment of chronic periaortitis: a retrospective study of 20 patients with chronic periaortitis and a review of the literature. Ann Rheum Dis. 2005;64:828–833. doi: 10.1136/ard.2004.029793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Rogliani P, Mura M, Assunta Porretta M, Saltini C. New perspectives in the treatment of idiopathic pulmonary fibrosis. Ther Adv Respir Dis. 2008;2:75–93. doi: 10.1177/1753465808089363. [DOI] [PubMed] [Google Scholar]
- 53.Dheda K, Lalloo UG, Cassim B, Mody GM. Experience with azathioprine in systemic sclerosis associated with interstitial lung disease. Clin Rheumatol. 2004;23:306–309. doi: 10.1007/s10067-004-0906-7. [DOI] [PubMed] [Google Scholar]
- 54.Peyrin-Biroulet L, Deltenre P, Ardizzone S, D'Haens G, Hanauer SB, Herfarth H, Lemann M, Colombel JF. Azathioprine and 6-mercaptopurine for the prevention of postoperative recurrence in Crohn's disease: a meta-analysis. Am J Gastroenterol. 2009;104:2089–2096. doi: 10.1038/ajg.2009.301. [DOI] [PubMed] [Google Scholar]
- 55.Bahcecioglu IH, Koca SS, Poyrazoglu OK, Yalniz M, Ozercan IH, Ustundag B, Sahin K, Dagli AF, Isik A. Hepatoprotective effect of infliximab, an anti-TNF-alpha agent, on carbon tetrachloride-induced hepatic fibrosis. Inflammation. 2008;31:215–221. doi: 10.1007/s10753-008-9067-1. [DOI] [PubMed] [Google Scholar]
- 56.Antoniou KM, Mamoulaki M, Malagari K, Kritikos HD, Bouros D, Siafakas NM, Boumpas DT. Infliximab therapy in pulmonary fibrosis associated with collagen vascular disease. Clin Exp Rheumatol. 2007;25:23–28. [PubMed] [Google Scholar]
- 57.Bargagli E, Galeazzi M, Bellisai F, Volterrani L, Rottoli P. Infliximab treatment in a patient with systemic sclerosis associated with lung fibrosis and pulmonary hypertension. Respiration. 2008;75:346–349. doi: 10.1159/000090248. [DOI] [PubMed] [Google Scholar]
- 58.Theiss AL, Simmons JG, Jobin C, Lund PK. Tumor necrosis factor (TNF) alpha increases collagen accumulation and proliferation in intestinal myofibroblasts via TNF receptor 2. J Biol Chem. 2005;280:36099–36109. doi: 10.1074/jbc.M505291200. [DOI] [PubMed] [Google Scholar]
- 59.Pelletier AL, Kalisazan B, Wienckiewicz J, Bouarioua N, Soule JC. Infliximab treatment for symptomatic Crohn's disease strictures. Aliment Pharmacol Ther. 2009;29:279–285. doi: 10.1111/j.1365-2036.2008.03887.x. [DOI] [PubMed] [Google Scholar]
- 60.Schnitzler F, Fidder H, Ferrante M, Noman M, Arijs I, Van Assche G, Hoffman I, Van Steen K, Vermeire S, Rutgeerts P. Long-term outcome of treatment with infliximab in 614 patients with Crohn's disease: results from a single-centre cohort. Gut. 2009;58:492–500. doi: 10.1136/gut.2008.155812. [DOI] [PubMed] [Google Scholar]
- 61.Hassan C, Zullo A, De Francesco V, Ierardi E, Giustini M, Pitidis A, Taggi F, Winn S, Morini S. Systematic review: endoscopic dilatation in Crohn's disease. Aliment Pharmacol Ther. 2007;26:1457–1464. doi: 10.1111/j.1365-2036.2007.03532.x. [DOI] [PubMed] [Google Scholar]
- 62.Thienpont C, D'Hoore A, Vermeire S, Demedts I, Bisschops R, Coremans G, Rutgeerts P, Van Assche G. Long-term outcome of endoscopic dilatation in patients with Crohn's disease is not affected by disease activity or medical therapy. Gut. 2010;59:320–324. doi: 10.1136/gut.2009.180182. [DOI] [PubMed] [Google Scholar]
- 63.Froehlich F, Juillerat P, Pittet V, Felley C, Mottet C, Vader JP, Michetti P, Gonvers JJ. Maintenance of surgically induced remission of Crohn's disease. Digestion. 2007;76:130–135. doi: 10.1159/000111027. [DOI] [PubMed] [Google Scholar]
- 64.Hoffmann JC, Heller F, Faiss S, von Lampe B, Kroesen AJ, Wahnschaffe U, Schulzke JD, Zeitz M, Bojarski C. Through the endoscope balloon dilation of ileocolonic strictures: prognostic factors, complications, and effectiveness. Int J Colorectal Dis. 2008;23:689–696. doi: 10.1007/s00384-008-0461-9. [DOI] [PubMed] [Google Scholar]
- 65.Rieder F, Zimmermann EM, Remzi FH, Sandborn WJ. Crohn's disease complicated by strictures: a systematic review. Gut. 2013;62:1072–1084. doi: 10.1136/gutjnl-2012-304353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Gustavsson A, Magnuson A, Blomberg B, Andersson M, Halfvarson J, Tysk C. Endoscopic dilation is an efficacious and safe treatment of intestinal strictures in Crohn's disease. Aliment Pharmacol Ther. 2012;36:151–158. doi: 10.1111/j.1365-2036.2012.05146.x. [DOI] [PubMed] [Google Scholar]
- 67.Wibmer AG, Kroesen AJ, Grone J, Buhr HJ, Ritz JP. Comparison of strictureplasty and endoscopic balloon dilatation for stricturing Crohn's disease – review of the literature. Int J Colorectal Dis. 2010;25:1149–1157. doi: 10.1007/s00384-010-1010-x. [DOI] [PubMed] [Google Scholar]
- 68.Baba S, Nakai K. Strictureplasty for Crohn's disease in Japan. J Gastroenterol. 1995;30(suppl 8):135–138. [PubMed] [Google Scholar]
- 69.Ambe R, Campbell L, Cagir B. A comprehensive review of strictureplasty techniques in Crohn's disease: types, indications, comparisons, and safety. J Gastrointest Surg. 2012;16:209–217. doi: 10.1007/s11605-011-1651-2. [DOI] [PubMed] [Google Scholar]
- 70.Azuma A. Pirfenidone treatment of idiopathic pulmonary fibrosis. Ther Adv Respir Dis. 2012;6:107–114. doi: 10.1177/1753465812436663. [DOI] [PubMed] [Google Scholar]
- 71.Noble PW, Albera C, Bradford WZ, Costabel U, Glassberg MK, Kardatzke D, King TE, Jr, Lancaster L, Sahn SA, Szwarcberg J, Valeyre D, du Bois RM. Pirfenidone in patients with idiopathic pulmonary fibrosis (capacity): two randomised trials. Lancet. 2011;377:1760–1769. doi: 10.1016/S0140-6736(11)60405-4. [DOI] [PubMed] [Google Scholar]
- 72.Diebold RJ, Eis MJ, Yin M, Ormsby I, Boivin GP, Darrow BJ, Saffitz JE, Doetschman T. Early-onset multifocal inflammation in the transforming growth factor beta 1-null mouse is lymphocyte mediated. Proc Natl Acad Sci U S A. 1995;92:12215–12219. doi: 10.1073/pnas.92.26.12215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Kulkarni AB, Ward JM, Yaswen L, Mackall CL, Bauer SR, Huh CG, Gress RE, Karlsson S. Transforming growth factor-beta 1 null mice. An animal model for inflammatory disorders. Am J Pathol. 1995;146:264–275. [PMC free article] [PubMed] [Google Scholar]
- 74.Denton CP, Merkel PA, Furst DE, Khanna D, Emery P, Hsu VM, Silliman N, Streisand J, Powell J, Akesson A, Coppock J, Hoogen F, Herrick A, Mayes MD, Veale D, Haas J, Ledbetter S, Korn JH, Black CM, Seibold JR. Recombinant human anti-transforming growth factor beta1 antibody therapy in systemic sclerosis: a multicenter, randomized, placebo-controlled phase I/II trial of CAT-192. Arthritis Rheum. 2007;56:323–333. doi: 10.1002/art.22289. [DOI] [PubMed] [Google Scholar]
- 75.Abe Y, Murano M, Murano N, Morita E, Inoue T, Kawakami K, Ishida K, Kuramoto T, Kakimoto K, Okada T, Narabayashi K, Umegaki E, Higuchi K. Simvastatin attenuates intestinal fibrosis independent of the anti-inflammatory effect by promoting fibroblast/myofibroblast apoptosis in the regeneration/healing process from TNBS-induced colitis. Dig Dis Sci. 2012;57:335–344. doi: 10.1007/s10620-011-1879-4. [DOI] [PubMed] [Google Scholar]
- 76.Haydont V, Bourgier C, Pocard M, Lusinchi A, Aigueperse J, Mathe D, Bourhis J, Vozenin-Brotons MC. Pravastatin inhibits the Rho/CCN2/extracellular matrix cascade in human fibrosis explants and improves radiation-induced intestinal fibrosis in rats. Clin Cancer Res. 2007;13:5331–5340. doi: 10.1158/1078-0432.CCR-07-0625. [DOI] [PubMed] [Google Scholar]
- 77.Wengrower D, Zanninelli G, Pappo O, Latella G, Sestieri M, Villanova A, Faitelson Y, Pines M, Goldin E. Prevention of fibrosis in experimental colitis by captopril: the role of TGF-beta1. Inflamm Bowel Dis. 2004;10:536–545. doi: 10.1097/00054725-200409000-00007. [DOI] [PubMed] [Google Scholar]
- 78.Wengrower D, Zanninelli G, Latella G, Necozione S, Metanes I, Israeli E, Lysy J, Pines M, Papo O, Goldin E. Losartan reduces trinitrobenzene sulphonic acid-induced colorectal fibrosis in rats. Can J Gastroenterol. 2012;26:33–39. doi: 10.1155/2012/628268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Rousseaux C, Desreumaux P. The peroxisome-proliferator-activated gamma receptor and chronic inflammatory bowel disease (PPARgamma and IBD) (article in French) J Soc Biol. 2006;200:121–131. doi: 10.1051/jbio:2006015. [DOI] [PubMed] [Google Scholar]
- 80.Fichtner-Feigl S, Strober W, Geissler EK, Schlitt HJ. Cytokines mediating the induction of chronic colitis and colitis-associated fibrosis. Mucosal Immunol. 2008;1(suppl 1):S24–27. doi: 10.1038/mi.2008.41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Raghu G, Brown KK, Costabel U, Cottin V, du Bois RM, Lasky JA, Thomeer M, Utz JP, Khandker RK, McDermott L, Fatenejad S. Treatment of idiopathic pulmonary fibrosis with etanercept: an exploratory, placebo-controlled trial. Am J Respir Crit Care Med. 2008;178:948–955. doi: 10.1164/rccm.200709-1446OC. [DOI] [PubMed] [Google Scholar]
- 82.Corren J, Lemanske RF, Hanania NA, Korenblat PE, Parsey MV, Arron JR, Harris JM, Scheerens H, Wu LC, Su Z, Mosesova S, Eisner MD, Bohen SP, Matthews JG. Lebrikizumab treatment in adults with asthma. N Engl J Med. 2011;365:1088–1098. doi: 10.1056/NEJMoa1106469. [DOI] [PubMed] [Google Scholar]
- 83.Horton MR, Santopietro V, Mathew L, Horton KM, Polito AJ, Liu MC, Danoff SK, Lechtzin N. Thalidomide for the treatment of cough in idiopathic pulmonary fibrosis: a randomized trial. Ann Intern Med. 2012;157:398–406. doi: 10.7326/0003-4819-157-6-201209180-00003. [DOI] [PubMed] [Google Scholar]
- 84.Danese S, Rudzinski J, Brandt W, Dupas JL, Peyrin-Biroulet L, Bouhnik Y, Kleczkowski D, Uebel P, Lukas M, Knutsson M, Erlandsson F, Hansen MB, Keshav S. Tralokinumab for moderate-to-severe UC: a randomised, double-blind, placebo-controlled, phase IIa study. Gut. 2014;64:243–249. doi: 10.1136/gutjnl-2014-308004. [DOI] [PubMed] [Google Scholar]
- 85.Scaldaferri F, Vetrano S, Sans M, Arena V, Straface G, Stigliano E, Repici A, Sturm A, Malesci A, Panes J, Yla-Herttuala S, Fiocchi C, Danese S. VEGF-a links angiogenesis and inflammation in inflammatory bowel disease pathogenesis. Gastroenterology. 2009;136:585–595. doi: 10.1053/j.gastro.2008.09.064. e5. [DOI] [PubMed] [Google Scholar]
- 86.Kumagai S, Ohtani H, Nagai T, Funa K, Hiwatashi NO, Shimosegawa, Nagura H. Platelet-derived growth factor and its receptors are expressed in areas of both active inflammation and active fibrosis in inflammatory bowel disease. Tohoku J Exp Med. 2001;195:21–33. doi: 10.1620/tjem.195.21. [DOI] [PubMed] [Google Scholar]
- 87.Richeldi L, Costabel U, Selman M, Kim DS, Hansell DM, Nicholson AG, Brown KK, Flaherty KR, Noble PW, Raghu G, Brun M, Gupta A, Juhel N, Kluglich M, du Bois RM. Efficacy of a tyrosine kinase inhibitor in idiopathic pulmonary fibrosis. N Engl J Med. 2011;365:1079–1087. doi: 10.1056/NEJMoa1103690. [DOI] [PubMed] [Google Scholar]
- 88.Catania JM, Chen G, Parrish AR. Role of matrix metalloproteinases in renal pathophysiologies. Am J Physiol Renal Physiol. 2007;292:F905–911. doi: 10.1152/ajprenal.00421.2006. [DOI] [PubMed] [Google Scholar]
- 89.Kim H, Oda T, Lopez-Guisa J, Wing D, Edwards DR, Soloway PD, Eddy AA. TIMP-1 deficiency does not attenuate interstitial fibrosis in obstructive nephropathy. J Am Soc Nephrol. 2001;12:736–748. doi: 10.1681/ASN.V124736. [DOI] [PubMed] [Google Scholar]
- 90.Osman B, Akool el S, Doller A, Muller R, Pfeilschifter J, Eberhardt W. Differential modulation of the cytokine-induced MMP-9/TIMP-1 protease-antiprotease system by the mTOR inhibitor rapamycin. Biochem Pharmacol. 2011;81:134–143. doi: 10.1016/j.bcp.2010.09.011. [DOI] [PubMed] [Google Scholar]
- 91.Wang S, Wilkes MC, Leof EB, Hirschberg R. Noncanonical TGF-beta pathways, mTORC1 and Abl, in renal interstitial fibrogenesis. Am J Physiol Renal Physiol. 2010;298:F142–149. doi: 10.1152/ajprenal.00320.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Reinisch W, Panes J, Lemann M, Schreiber S, Feagan B, Schmidt S, Sturniolo GC, Mikhailova T, Alexeeva O, Sanna L, Haas T, Korom S, Mayer H. A multicenter, randomized, double-blind trial of everolimus versus azathioprine and placebo to maintain steroid-induced remission in patients with moderate-to-severe active Crohn's disease. Am J Gastroenterol. 2008;103:2284–2292. doi: 10.1111/j.1572-0241.2008.02024.x. [DOI] [PubMed] [Google Scholar]