Abstract
Sphingolipid research has surged in the past two decades and has produced a wide variety of evidence supporting the role of this class of molecules in mediating cellular growth, differentiation, senescence, and apoptosis. Ceramides are a subgroup of sphingolipids (SLs) that are directly involved in the process of initiation of apoptosis. We, and others, have recently shown that ceramides are capable of the formation of protein-permeable channels in mitochondrial outer membranes under physiological conditions. These pores are indeed good candidates for the pathway of release of pro-apoptotic proteins from the mitochondrial intermembrane space (IMS) into the cytosol to initiate intrinsic apoptosis. Here, we review recent findings on the regulation of ceramide channel formation and disassembly, highlighting possible implications on the initiation of the intrinsic apoptotic pathway.
Abbreviations: Bcl-2, B cell CLL/lymphoma-2; Cer, ceramide; CerS, ceramide synthase; DES, dihydroceramide desaturase; DHCer, dihydroceramide; ER, endoplasmic reticulum; IMS, intermembrane space; KSR, 3-ketosphinganine reductase; MOMP, mitochondrial outer membrane permeability; SLs, sphingolipids; So, sphingosine; SM, sphingomyelin; SPT, serine palmitoyl transferase
Keywords: Ceramide channels, Mitochondria, Apoptosis, Assembly and disassembly, Bcl-2 family proteins, de novo synthesis, Sphingolipids, Chain length
1. Introduction
Whether a cell proliferates or perishes requires a complex set of cellular decisions that depend on the environment and its physical and nutritional states. Signaling in cells is regulated optimally through a wide array of macromolecules and messengers that control the fate of the cell creating many intertwined networks of regulators and effectors. Until recently, SLs were merely considered structural components of cellular membranes. We now know of a plethora of functions these molecules perform in cell signaling, stress and death (Hannun, 1996, Saba et al., 1996, Chalfant et al., 2001, Jenkins and Hannun, 2001). Multiple novel agents that modulate SL metabolism have been studied and at least in one instance applied therapeutically for cancer treatment (Adan-Gokbulut et al., 2013). Ceramide (Cer), is responsible for several intracellular signals and is considered the parent SL molecule (Hannun, 1996, Jayadev and Hannun, 1996, Perry et al., 1996, Lee et al., 1996).
Ceramides are a family of lipids with a sphingosine (So) backbone N-acylated with a variety of fatty acids, creating a diverse group of molecules (Fig. 1). They are involved in cell signaling in various contexts (Hannun, 1996, Saba et al., 1996, Jayadev and Hannun, 1996, Lee et al., 1996, Hayakawa et al., 1996, Yoon et al., 2002, Shin et al., 2002, Obeid et al., 1993). In this review we focus on one aspect of Cer cell signaling: the regulation of cell death by Cer channel formation in the outer membranes of mitochondria (Siskind et al., 2002). In addition to this pathway of protein release, there are many proposed pathways to explain how mitochondrial IMS proteins are discharged from mitochondria: the opening of the permeability transition pore (Crompton, 1999, Crompton et al., 1999); the oligomerization of Bax monomers to achieve channel activity (Antonsson, 2001, Antonsson et al., 2000, Antonsson et al., 2001, Saito et al., 2000); the opening of the mitochondrial apoptosis-induced channels (Pavlov et al., 2001); the interactions of BH3/Bax/cardiolipin (Kuwana et al., 2002), and the interactions between Bax and Cer (Belaud-Rotureau et al., 2000). Regardless of the pathway, it is clear that Bcl-2 (B cell CLL/lymphoma) family proteins play a central role in the regulation of mitochondrial permeability. Here, we review the effects of these proteins, and other metabolites, on Cer channel formation and disassembly highlighting possible implications on the onset of intrinsic apoptosis.
Figure 1.
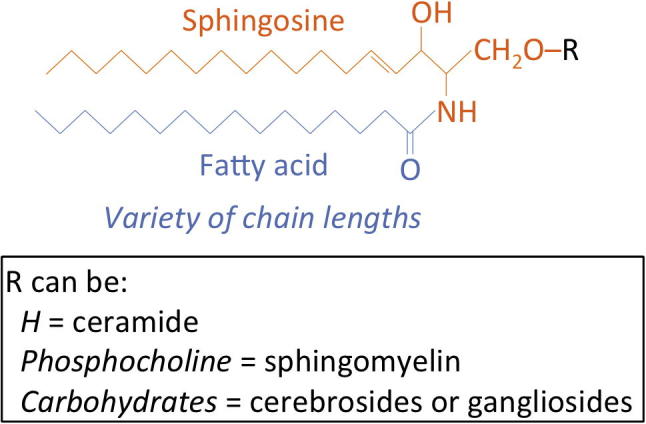
The basic structure of sphingolipids. SLs have a So backbone (orange) that is N-acylated with fatty acids of a variety of chain lengths (blue). The C-1 hydroxyl can be as simple as a hydrogen atom (in ceramides) or as complex as multiple carbohydrate subunits in cerebrosides and gangliosides. The trans double bond at C-4 is characteristic of Cer and when it is saturated the molecule is DHCer.
2. Ceramide
Cer is a condensation product of the amino alcohol So and a fatty acid in an acylation reaction. The range of acylation is wide, creating ceramides that contain fatty acids varying from 6 to 34 or even more carbons. d-erythro-N-palmitoylsphingosine (C16-Cer) is an example of one of the naturally occurring forms of Cer (Fig. 1). Another key aspect of the Cer molecule is the presence of a 4,5-trans double bond that clearly has a profound impact on the biophysical characteristics of Cer and on cell survival pathways, as will be presented later.
Of the various roles of Cer inside cells, the ability to induce apoptosis is the clearest. Ceramides have been shown to induce apoptosis directly and indirectly (Saba et al., 1996, Obeid et al., 1993, Linardic et al., 1996, Danial and Korsmeyer, 2004, Wiesner and Dawson, 1996a, Wiesner and Dawson, 1996b). MCF7 breast cancer cells experienced mitochondrial outer membrane permeabilization and apoptosis when bacterial sphingomyelinase (a Cer-generating enzyme) was targeted to mitochondria and Cer was generated specifically in mitochondria (Birbes et al., 2001). When bacterial sphingomyelinase was targeted to other organelles, apoptosis and mitochondrial permeabilization did not occur (Hannun et al., 2001). In leukemia cells, Cer levels were increased significantly upon the addition of the chemotherapeutic agent vincristine resulting in growth suppression and marked apoptosis (Zhang et al., 1996).
The mechanisms by which Cer causes mitochondrial outer membrane permeabilization which results in apoptosis are diverse (reviewed in Siskind (2005)). Remarkably, Cer is able to permeabilize mitochondrial outer membranes through the formation of channels that are large enough to allow the egress of IMS proteins into the cytosol (Siskind et al., 2002, Siskind et al., 2006, Stiban et al., 2008). Thus, channel formation by Cer is an upstream event to the induction of apoptosis (reviewed in Colombini (2010)). The permeability of the mitochondrial outer membrane to proteins including cytochrome c can be increased by the incubation of the isolated mitochondria with ceramide in a time- and dose-dependent manner (Siskind et al., 2002). This was the first indication that a lipid can form pores in a biological membrane. Different groups observed similar effects of ceramide channel formation in protein-free systems (Siskind and Colombini, 2000, Montes et al., 2002, Pajewski et al., 2005, Stiban et al., 2006). Increasing evidence (topped by the visualization of the channels by transmission electron microscopy (Samanta et al., 2011) demonstrated that a channel formed by a lipid is possible and valid (Stiban et al., 2008, Siskind et al., 2003, Siskind et al., 2005, Ganesan et al., 2010, Siskind et al., 2008). Since it is inherently different than a protein channel, Cer channel formation depends on the steady state level of Cer in the membrane. Thus, the formation of Cer channels is controlled mainly by the metabolism of Cer in the membrane.
3. Ceramide biosynthesis
Ceramides are central molecules in sphingolipid synthesis. In vivo, there are three pathways that lead to the generation of Cer (Fig. 2). The de novo Cer synthesis pathway starts in the endoplasmic reticulum (ER) with the condensation of palmitoyl-CoA with serine to form 3-ketosphinganine catalyzed by serine palmitoyl transferase (SPT). The ensuing product is then reduced by 3-ketosphinganine reductase (KSR) to sphinganine which is acylated by a family of Cer synthases (CerS) generating dihydroceramides (DHCer) with varying fatty acyl chain lengths. In the final step of this pathway DHCer desaturase (DES) facilitates the formation of ceramide inserting a double bond between C4 and C5 of the sphingoid base. A variety of evidence suggests that this pathway occurs in the endoplasmic reticulum (Hirschberg et al., 1993) however, some enzymes were localized in mitochondria of some cell types (Yu et al., 2007, Novgorodov et al., 2011) but not others (Stiban et al., 2008, Mesicek et al., 2010), indicating that this localization might be cell-type specific. Even though under normal conditions liver mitochondria were shown to be devoid of DES activity (Stiban et al., 2008), N-myristoylation targeted the recombinant enzyme to mitochondria inducing cellular apoptosis (Beauchamp et al., 2009), indicating that the localization of Cer-metabolizing enzymes to the mitochondria is dependent on cellular conditions as well.
Figure 2.
Ceramide is the hub of sphingolipid metabolism. In the cell three different pathways produce Cer: the de novo synthesis from serine and palmitoyl-CoA (top), SM hydrolysis (left) and the salvage pathway (right). Cer can be used to produce various simple and complex sphingolipids (bottom). The enzymes used in those pathways are boxed. SPT: serine palmitoyl transferase; KSR: 3-ketosphinganine reductase; CerS1-6: ceramide synthases 1–6; DES: dihydroceramide desaturase; SMS: SM synthase; SMase: sphingomyelinase.
Sphingomyelin (SM) hydrolysis in the plasma membrane also contributes to the production of ceramide by different isoforms of sphingomyelinase (Birbes et al., 2001). SM hydrolysis has been shown to increase Cer content upon daunorubicin treatment (Allouche et al., 1997). Another Cer-producing sequence is the salvage pathway whereby CerS are used to directly acylate So to form Cer without the need to generate and desaturate DHCer (Kitatani et al., 2008, Mullen et al., 2012, Mullen et al., 2011).
Thus, a variety of steps can increase Cer contents in cells. These lipids function differentially in different locations in the cell. They can form ordered microdomains in the plasma membrane contributing to signal transduction by receptor protein aggregation (Silva et al., 2009). They themselves may be considered second messengers (Becker and Hannun, 2005), and they can also be responsible for the permeabilization of mitochondrial outer membranes prior to the initiation of apoptosis (Birbes et al., 2001).
4. Apoptosis
The term apoptosis (pronounced aepu’tosis) come from the Greek word for falling off (Kerr et al., 1972). Apoptosis (also called programed cell death) is the process by which a cell ends its life without causing inflammation as it packages all its contents and sends them to be ingested by neighboring cells (Kerr et al., 1972, Raff et al., 1994, Hengartner, 2000). There are two major routes that converge into a common apoptotic pathway (Fig. 3). Apoptosis can be initiated from signals coming from outside the cell (extrinsic apoptosis) or from inside the cell (intrinsic/mitochondrial apoptosis). These signals eventually induce the permeability of the mitochondrial outer membrane to cytochrome c and other IMS proteins (Susin et al., 2000). When these proteins are released into the cytosol, other signaling events occur activating the cascade of caspases (cysteine-aspartic acid proteases) that cleave different nuclear and cytoplasmic substrates leading to the execution of the cell (Degterev et al., 2003).
Figure 3.
An overview of mechanisms of apoptosis. Apoptosis can be activated extrinsically or intrinsically. In extrinsic apoptosis, a macrophage carrying a Fas ligand (FasL) binds to Fas receptor (FasR) on the plasma membrane of the cell inducing the trimerization of the receptor. This induces the recruitment of Fas activated death domain (FADD) which recruits and activates procaspase-8 which ultimately activates the executioner caspase-3 that is responsible for the activities that lead to apoptosis. On the other hand, intrinsic apoptosis is centered around mitochondrial outer membrane permeabilization. Different pro-apoptotic proteins reside in the IMS of mitochondria and when the integrity of the outer membrane is compromised they leak out and cause the activation of apoptosomes, the condensation of chromatin, membrane blebbing and nuclear envelope destruction. Different proteins affect various steps in both pathways positively (green arrows) or negatively (red blunt-ended lines). IAP: inhibitor of apoptosis protein; FLIP: (FADD-like IL-1β-converting enzyme)-inhibitory protein; Smac/DIABLO: second mitochondria-derived activator of caspases/Direct IAP-binding protein with low pI; HSP 27: heat shock protein 27; Apaf-1: apoptotic protease activating factor 1; Bid and tBid: BH3 interacting domain death agonist and truncated Bid; Bax: Bcl-2-associated X protein; Bcl-xL: Bcl-2-extra large; EndoG: endonuclease G; Omi/HtrA2: high temperature requirement A serine endoprotease; AIF: apoptosis-inducing factor; mPTP: mitochondrial permeability transition pore. Note that the release of all pro-apoptotic proteins from the IMS can be through any one of the shown channels.
In mitochondrial apoptosis, the permeability of the outer membrane is enhanced by members of the Bcl-2 family proteins (Cory and Adams, 2002) (such as Bax, Bak and Bad (Green and Kroemer, 2004), whereas other members of the same family (Bcl-2, Bcl-xL and Mcl-1 (Zhang et al., 1996, Nijhawan et al., 2003, Yang et al., 1997, Minn et al., 1999) render the outer membrane more intact. Hence, the balance between anti-apoptotic family members and their pro-apoptotic counterparts can dictate whether or not apoptosis occurs (Cory and Adams, 2002).
Intrinsic apoptosis is dependent on the increased mitochondrial outer membrane permeability (MOMP). Once permeabilized, mitochondria release a number of pro-apoptotic proteins into the cytosol (mainly, cytochrome c, AIF, Smac/DIABLO and Omi/HtrA2 (Saelens et al., 2004). In a proliferating cell, a family of proteins termed IAP (inhibitor of apoptosis) functions to inhibit the activation of caspases and hence counter apoptosis. Once Smac/DIABLO is released it blocks IAP and thus indirectly activates caspases (Vucic et al., 2002). Moreover, after its release from the IMS cytochrome c binds to Apaf-1 (apoptotic protease-activating factor-1) and this binding cleaves inactive procaspase-9 into active caspases-9. Caspase-9 cleaves and hence activates the executioner caspase, caspase-3 which is the key step for the caspase cascade in intrinsic apoptosis (Yang et al., 1997, Liu et al., 1996). Through the caspase cascade, other proteins and enzymes get activated such as endonucleases, DNases, proteases and others, and they start breaking down the cell within their own capacity (e.g. the activation of caspase-activated DNase (CAD) which degrades DNA into fragmented ladder is a hallmark of apoptosis (Nagata, 2000)). Also, this cascade activates other caspases which induce several cytosolic proteins facilitating the formation of the blebs in the plasma membrane and causing the rest of the morphological changes manifesting in apoptotic cells (Hengartner, 2000). Previous studies showed that in order for the cell to die by intrinsic apoptosis, it must pass by the irreversible step of cytochrome c release (Susin et al., 2000), but also, the cell must have an active Apaf-1 in order to die after the release of cytochrome c (Sanchis et al., 2003).
Morphologically, the apoptotic process occurs in several steps (Fig. 4): it starts with the condensation of chromatin in the nucleus, followed by the fragmentation of the nuclear envelope and DNA hydrolysis by DNases. Moreover, phosphatidylserine (a phospholipid normally found exclusively at the cytosolic side of the plasma membrane bilayer) flips to the extracellular face of the bilayer (Jacobson et al., 1993, Jacobson et al., 1994a, Jacobson et al., 1994b). The cells undergoing apoptosis lose their attachment to their neighboring cells (Ruoslahti and Reed, 1994), and blebs (membrane-bound vesicles) form in the plasma membrane accompanied by the shrinkage of the cell (Jacobson et al., 1993, Jacobson et al., 1994a, Jacobson et al., 1994b). Finally, the components of the cell are packed into membrane-bound bodies called apoptotic bodies which are endocytosed by macrophages and/or neighboring cells (Hart et al., 1996).
Figure 4.
Cytology of apoptosis. The different stages of apoptotic cell death start by cellular shrinkage and chromatin condensation, concomitant with formation of membrane blebs. Organelles and nucleus fragment and the blebs begin formation of apoptotic bodies which are eventually engulfed by macrophages or neighboring cells by endocytosis/phagocytosis. The lack of release of cellular components to the extracellular fluid results in the absence of inflammation.
Despite having different routes that lead to apoptosis, the process itself is fully regulated (Perry et al., 1996, Degterev et al., 2003, Raff et al., 1993). Apoptosis often occurs as a result of a variety of factors, for instance DNA damage (Wang, 2001); cellular stress (Samali et al., 2010, Fulda et al., 2010); disruption of calcium homeostasis (Preston et al., 1997); the closure of the voltage-dependent anion channel (VDAC) which inhibits the metabolic exchange between the mitochondria and the cytosol (Lai et al., 2006), etc. Regardless of the causes, the onset of intrinsic apoptosis is accompanied by a concomitant increase in mitochondrial Cer levels (Garcia-Ruiz et al., 1997, Dai et al., 2004). Moreover, exogenous Cer addition to cells induces apoptosis and DNA fragmentation (Allouche et al., 1997). The regulation of apoptosis is multi-faceted; nevertheless, it is centered around a family of proteins, the Bcl-2 family.
5. Bcl-2 family proteins
The proliferation and death of cells are tightly regulated by the cell cycle enzymes and the Bcl-2 family proteins and Tumor necrosis factors. Some of these proteins interact with mitochondria and lead to the release of cytochrome c and apoptosis inducing factors among other proteins. There are around 20 proteins in Bcl-2 family and they function primarily as pro- or anti-apoptotic mediators. Pro-apoptotic Bcl-2 proteins (such as Bak and Bax) are activated upon cellular stresses to induce MOMP (Chipuk et al., 2010) by the formation of pores in mitochondrial outer membrane. Bax, for instance transports to and restructures at the outer membrane forming channels (Kim et al., 2009). This breach in mitochondrial integrity allows pro-apoptotic proteins to be released from the IMS and produce the apoptotic response (Green, 2005). Another subset of death-promoting family members is the BH3-only proteins such as Bid, which aids Bax and Bak in causing mitochondrial permeability (Dewson et al., 2008). While most of the family members are pro-apoptotic, six members were shown to maintain cell survival and thus exhibiting anti-apoptotic properties (e.g. Bcl-2, Bcl-xL and Mcl-1) (Sorenson, 2004). These proteins mainly interfere with the ability of pro-apoptotic proteins to interact with BH3-only proteins to be activated (Chipuk et al., 2008). It is, therefore, the ability of the anti-apoptotic proteins to interfere with channel-formation by pro-apoptotic counterparts that allows for the regulation of apoptosis induction by Bcl-2 family (Beverly, 2012). Furthermore, some BH3-only proteins (e.g. PUMA and Bad) bind and inhibit anti-apoptotic family members (Letai et al., 2002, Kuwana et al., 2005).
It is evident that Bcl-2 family proteins are central to the regulation of the initiation of mitochondrial apoptosis. Since both Bcl-2 family proteins and SLs (particularly Cer) are directly involved in apoptosis, the relationship between this family and Cer has been extensively studied (Kawatani et al., 2003, Zhang and Saghatelian, 2013, Beverly et al., 2013, Jensen et al., 2014). Their roles in Cer channel formation and disassembly are discussed below. It is remarkable that the inhibition of anti-apoptotic Bcl-2 proteins induces C16-Cer synthesis whereas the exogenous addition of the pro-apoptotic recombinant Bak potentiated CerS activity in vitro (Beverly et al., 2013) thus implying other routes of regulation of apoptosis initiation by Bcl-2 family via Cer production.
6. Ceramide channel formation in mitochondria
The production of Cer in the cell, particularly in mitochondria, is associated with MOMP permeabilization and apoptosis initiation (Birbes et al., 2001, Birbes et al., 2002, Ghafourifar et al., 1999, Richter and Ghafourifar, 1999, Di Paola et al., 2004). The method of permeabilization was a source of debate until Siskind and Colombini, in the year 2000, pioneered the discovery of the Cer channel in planar phospholipid bilayers (Siskind and Colombini, 2000) and in isolated mitochondria (Siskind et al., 2002, Siskind et al., 2006). The work was later confirmed by inhibition assays (Stiban et al., 2006, Elrick et al., 2006), molecular dynamics simulation (Anishkin et al., 2006) and the visualization of these channels by transmission electron microscopy (Samanta et al., 2011). The biophysics and biochemistry of Cer channel formation was excellently reviewed by Colombini, 2010, Colombini, 2013, Colombini, 2012, Hage-Sleiman et al., 2013. Briefly, Cer channels are formed from columns of Cer that arrange in an anti-parallel fashion making a cylindrical shape spanning the hydrophobic interior of the mitochondrial outer membrane. Each column is composed of six Cer molecules (Siskind et al., 2002, Siskind et al., 2003, Siskind and Colombini, 2000, Stiban et al., 2006). Hydrogen bonding, hydrophobic stacking and dipole–dipole interactions stabilize the columns. The stacking of Cer columns in a cylindrical barrel-stave channel is biophysically sound. The hydrogen bonds between the amide linkages of Cer and underlying carbonyl of another Cer molecule are numerous. In addition, the hydroxyls of the Cer molecule would line up in an ice-lattice-like structure in the lumen of the channel. Together with dipole–dipole interactions and hydrophobic stacking, these weak interactions accumulate to create a stable structure that transverses the hydrophobic part of the membrane in a water-filled pathway through which proteins can cross (Colombini, 2010).
Cer channels are dynamic. They grow or shrink by the addition or removal of Cer columns. The channels can vary in size depending on the local Cer concentration and the presence of different proteins that may interact with the channels. Since the formation and disassembly of these channels is dynamic, they are severely influenced by the environment. In this case, the environment of the channel traverses two phases, the aqueous phase in the lumen of the channel and the hydrophobic liquid crystal phase of the bilayer. Hence multiple molecules have been shown to influence Cer channels by changing the dynamic equilibrium in one phase or another.
7. Regulators of ceramide channels
The discovery of lipid channels is novel. The fact that these channels control a very important aspect of cellular life and death is outstanding. Since they control the initiation of apoptosis at the point of no return, the induction of MOMP, their own formation must be regulated extensively. A few inhibitors were shown to induce channel disassembly in vitro. Some were metabolites in SL pathways and others were ions and Bcl-2 family proteins (Fig. 5). Here we review inhibitors and stabilizers of Cer channels.
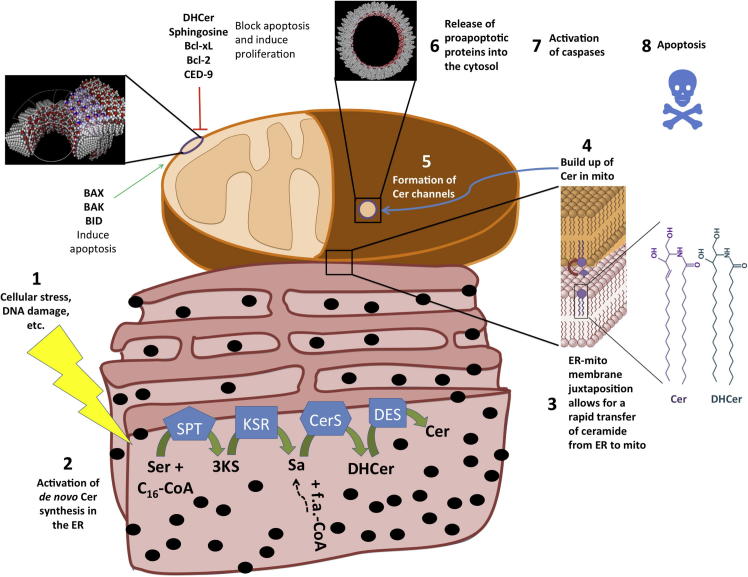
Figure 5.
A mechanistic view of the action at the mitochondrial outer membrane following the activation of intrinsic apoptosis. The sequence of events leading to the death of the cell is summarized as follows: (1) cellular stresses induce (2) the biosynthesis of Cer in the ER via the de novo synthesis pathway. (3) Cer synthesized in the ER is then readily imported into mitochondria via ER-mitochondria contact sites termed MAM leading to (4) a buildup of Cer in mitochondria. (5) The increased local Cer concentration allows those lipids to self-assemble into a barrel-stave channel through which (6) pro-apoptotic proteins are released into the cytosol. Those channels are inhibited by DHCer, So, anti-apoptotic Bcl-2 family proteins; and they are potentiated by pro-apoptotic Bcl-2 proteins (see the text for details). The release of proteins into the cytosol (7) activates caspases which ultimately (8) induce apoptosis. Note: the structures in black of Cer channels were obtained from Prof. Marco Colombini at the University of Maryland College Park.
7.1. Inhibitors
7.1.1. Lanthanum
The first inhibitor of Cer channels to be characterized was an ion: La3+ (Siskind et al., 2003). The effect of La3+ on membranes and channels has been studied for different systems. Phase transitions (Tanaka et al., 2001, Hammoudah et al., 1979) and vesicle fusion (Hammoudah et al., 1979) for instance can be attributed to La3+. Lanthanum ions are known to block several calcium channels (Grupe et al., 2010, Hoth and Penner, 1993, Ross and Cahalan, 1995). Similarly, lanthanides inhibit a wide array of membrane channels such as mechanogated channels (Gustin et al., 1988, Lee et al., 1999), VDAC (Gincel et al., 2001), the voltage-gated sodium channel (Armstrong and Cota, 1990) and the nonselective cation channel (Grosman and Reisin, 2000).
It appears that La3+ acts by affecting the ice-lattice-like hydrogen bonding of the lumen of the channel by interfering with the hydroxyl, carbonyl and/or amide functional groups that stabilize the channels (Siskind et al., 2003). Alternatively, La3+ can induce a shift in the dynamic equilibrium between conducting and non-conducting Cer structures in the membrane favoring structures with a reduced occupied membrane area (Shao et al., 2012).
7.1.2. Trehalose
Another molecule that disrupts hydrogen bonds in the lumen of the channel is the disaccharide, trehalose (α-d-glucopyranosyl-(1 → 1)-α-d-glucopyranoside). This molecule has been extensively studied and used as a protecting agent that maintains the organellar and cytoplasmic structures under environmental stresses (Wiemken, 1990) such as dehydration and freezing. It has been used to maintain membrane integrity in several taxa due to its ability to influence surrounding hydrogen bonds of water molecules (Block, 2003, Cordone et al., 2005).
Trehalose inhibits Cer channels in phospholipid membranes by inducing partial disassembly. This property of trehalose allowed Colombini and coworkers to distinguish between Cer channel permeability and that of Bax when both molecules were used together, since activated Bax conductance is insensitive to trehalose inhibition (Ganesan et al., 2010). It is noteworthy to mention that sucrose had similar, albeit less prominent, effects on Cer channels in phospholipid bilayers and in isolated mitochondria forcing us to switch the buffer used to isolate mitochondria from a sucrose to a mannitol buffer (data not shown and Stiban et al., 2006). Working with different Cer analogs to test the hydrogen-bonding requirement for Cer channel formation, Perera et al. showed that methylation of the C1-hydroxyl reduced the hydrogen-bonding capabilities of the molecule and resulted in the formation of transient channels which were unstable and unable to allow the release of cytochrome c (Perera, Ganesan et al., 2012). Similarly urea-Cer increased hydrogen-bonding and therefore formed more channels in the outer membrane (Perera, Ganesan et al., 2012).
7.1.3. Bovine serum albumin
Early experiments in planar phospholipid membranes identified albumin as a key inhibitor of C2- but not C16-Cer permeability (Siskind et al., 2002). BSA is a molecule that has the ability to carry different hydrophobic compounds such as free fatty acids, bilirubin, lipid soluble hormones, and some drugs in the blood (Farrugia, 2010). BSA is therefore able to extract short chain Cer from the membrane thus lowering the local Cer concentration in the membrane favoring channel disassembly. C16-Cer is probably harder to pull from the membrane and hence is unaffected by BSA. Mitochondria treated with C2-Cer after incubation with BSA showed reduced MOMP as did mitochondria treated with BSA after C2-Cer incubation arguing that BSA is able to disassemble Cer channels as well as to prevent them from forming (Siskind et al., 2002) by sequestering Cer molecules in its hydrophobic pocket, away from the membrane. Indeed, BSA was able to remove C2-Cer from mitochondria as mitochondria treated with C2-Cer alone had 10-fold excess of Cer in the membrane compared to when BSA was added. Permeability was increased 5-fold under these circumstances. Thus, the ability of BSA to reverse MOMP is indeed due to the depletion of Cer from the membrane (Siskind et al., 2006).
7.1.4. Dihydroceramide
Early experiments in the field of SL biology indicated that Cer is a pro-apoptotic molecule which induces cytochrome c release into the cytosol whereas DHCer is biologically silent in both categories (Obeid et al., 1993, Ghafourifar et al., 1999, Richter and Ghafourifar, 1999). DHCer is not only inactive in producing an apoptotic response, but also inhibitory to Cer MOMP and apoptosis induction, as both C2- and C16-Cer channels were inhibited by C2- and C16-DHCer, respectively (Stiban et al., 2006). As noted previously, the de novo synthesis produces DHCer by the action of different CerS which are then desaturated by different isoforms of DES to produce Cer. DES is an ER-bound enzyme under normal conditions (Stiban et al., 2008) but it can be targeted to mitochondria upon N-myristoylation (Beauchamp et al., 2009). Only the wild-type myristoylable DES1-Gly (and not the unmyristoylable mutant DES1-Ala), induced apoptosis of COS-7 cells (Beauchamp et al., 2009). MOMP was significantly enhanced upon supplementing mitochondria with DHCer and NADPH (both substrates of the mixed-function oxidase DES) compared to supplementing them with only one of the substrates. In addition, mixing microsomal membranes supplemented with DHCer and NADPH with ER mitochondrial preparations enhanced MOMP indicating that Cer produced in the ER is readily transferrable to mitochondria via contact sites termed mitochondrial associated membranes (MAM) Stiban et al., 2008. DHCer, due to its saturated C4 and C5, is able to interfere with the strict packing of Cer molecules in the columns making the channel. Thus 1 part in 10 of DHCer was able to induce Cer channel disassembly, making DHCer a strong antagonist of Cer channel dynamics. The trans double bond is therefore crucial for channel formation. The formation and stability of Cer channels were enhanced when using Cer containing an additional trans double bond in a position allowing for π resonance between the two bonds (Perera, Ganesan et al., 2012). The mode of inhibition is therefore biophysical and is due to the intercalation of DHCer molecules within Cer columns which is destabilizing.
Biologically, there are many implications for this DHCer inhibition of Cer channel formation in vivo. In cells lacking DES, Cer was not produced and the cells were larger with respect to wild-type controls and resistant to apoptosis. Moreover, strong activation of the anti-apoptotic and anabolic signaling pathway regulated by Akt/protein kinase B was observed in these cells (Siddique et al., 2013). The ablation of DES also protected cells from etoposide-induced apoptosis (Siddique et al., 2012). Furthermore, mitochondrial depolarization and late-apoptosis were reduced by DES knockdown and the pro-apoptotic effects of Cer were significantly reduced by DHCer (Breen et al., 2013). These results highlight the effects of DHCer on Cer channel formation and the onset of apoptosis and identify DES as a potential therapeutic target for a variety of diseases.
Even though DHCer does not induce MOMP or apoptosis, it is indeed a biologically active molecule with different characteristics from its Cer relatives (Rodriguez-Cuenca et al., 2015). In obesity, for instance, it was shown that pharmacological inhibition of DES prevents adipocyte differentiation; on the other hand obese patients exhibited lower expression of the gene encoding DES in their adipose tissue indicating that DHCer to Cer conversion is essential and that the accumulation of DHCer is associated with other biological functions (Barbarroja et al., 2014). Moreover, in the hypoxic heart, total Cer levels increased then decreased sharply concomitant with an increase in DHCer levels in the right ventricle (Noureddine et al., 2008) due to the repression of DES gene (Azzam et al., 2013). All these data confirm the significance of the switch from unsaturated DHCer to monounsaturated Cer in a variety of biological scenarios including Cer channel formation.
7.1.5. Sphingosine
Another precursor of Cer is the amino alcohol So. Different CerS can N-acylate So to form Cer in the salvage pathway. Unlike DHCer, So is another lipid that can form channels in membranes (Siskind et al., 2005). However, like DHCer, it has the capability to disassemble Cer channels (Elrick et al., 2006). The intercalation of So with Cer columns is probably destabilizing since those lipids may be incompatible. As previously noted, Cer channels are dynamic: there is a constant insertion on more columns and/or removal of other columns resulting in the enlargement or contraction of the channel. There is a dynamic equilibrium between Cer columns in the channel and non-conducting structures in the membrane. In the case of So, similar to DHCer, the interaction between this molecule and Cer columns seems to be destabilizing the structure of the channel leading to disassembly. Despite the fact that both lipids form channels with some order in the membrane, their combination may result in instability due to poorly organized hybrid structures (Colombini, 2013).
7.1.6. Very long chain ceramides
Cer with different fatty acyl chains permeabilize mitochondria differentially. They all form channels in mitochondrial outer membranes as well as in artificial membranes. Recently we showed that similar results were obtained with different Cer species inhibiting each other’s ability to form channels. Indeed C24-Cer inhibited C16-Cer channel formation and vice versa in liposomes and mitochondria (Stiban and Perera, 2015). The notion of instability of the combination of molecules is valid in this case as well. Using several biophysical techniques, it was shown by Silva and colleagues that Cer with different fatty acyl chain lengths affect properties of phospholipid membranes differently (Pinto et al., 2011, Pinto et al., 2014), in agreement with previous results exhibiting the biophysical properties of membranes formed from microsomes lacking very long chain sphingolipids (Silva et al., 2012). In vitro experiments with protein-free liposomes showed a biphasic effect of permeability when titrating C16-Cer with C22-Cer. As C16-Cer was decreased and C22-Cer was increased, liposomal permeability was first inhibited then returned to the starting level indicating that different channels were formed and that both Cer species interfered with the channels formed by the other. This was also observed in isolated mitochondria (Stiban and Perera, 2015).
This interplay between Cer species with different acyl chains indeed has severe implications in SL biology. There are 6 mammalian isoforms of CerS each of which catalyzing the acylation of the sphingoid base with specific subsets of fatty acyl chains (Stiban et al., 2010). Different CerS enzymes undergo homo- or heterodimerization as a regulatory mechanism (Laviad et al., 2012) affecting the products of these enzymes. This biophysical inhibition of Cer channel formation by different Cer species adds to the complexity of when and where permeabilization may occur. In colon cancer cells, co-overexpression of CerS2 (responsible for producing very long chain ceramides) with CerS4 (C18-Cer) or CerS6 (C16-Cer) increased the total Cer levels but did not result in cell death indicating that the ability to instruct cells to start apoptosis depends on the equilibrium between the various Cer species in biological membranes (Hartmann et al., 2013). Moreover, in a variety of neurodegenerative diseases, different subsets of Cer and different CerS enzymes are activated or inhibited, further showing that the chain length is an extremely important functional feature of Cer (Ben-David and Futerman, 2010).
Very long chain Cer species have key roles in cells. CerS2 null mice are unable to synthesize very long chain Cer and SLs and exhibit glucose intolerance as insulin signaling is altered in the liver. In hepatocytes of CerS2 null mice, insulin receptor is unable to translocate to lipid microdomains to initiate insulin response, as those microdomains differ significantly from wild-type mice due to their lack of very long chain Cer (Park et al., 2013). The disequilibrium of Cer carrying different chain lengths was proposed to be important for cancer progression whereas normal cells have equilibrium between several Cer species (Hartmann et al., 2012).
7.1.7. Anti-apoptotic Bcl-2 proteins
Apoptosis is regulated mainly by Bcl-2 family proteins (Chipuk et al., 2010, Chipuk et al., 2008, Sorenson, 2004). The effects of anti-apoptotic Bcl-2 proteins (e.g. the mammalian Bcl-xL and Caenorhabditis elegans CED-9) on Cer-induced MOMP were thoroughly investigated. Both anti-apoptotic proteins inhibited Cer channel formation in isolated mitochondria and were able to reverse the permeabilization, presumably by favoring channel disassembly (Siskind et al., 2008). Even though other pro-apoptotic proteins are present in isolated mitochondria and there is a possibility that Bcl-xL interferes with them and hence influences Cer indirectly, almost identical results were obtained using mitochondria isolated from the yeast Saccharomyces cerevisiae. This particular yeast strain is devoid of proteins homologous to those of the Bcl-2 family. Thus both Bcl-xL and CED-9 are interfering directly with the ability of Cer to cause MOMP. Moreover, overexpression of Bcl-2 in yeast resulted in mitochondria that were resistant to Cer permeabilization (Siskind et al., 2008). Furthermore, in the protein-free system of planar membranes formed from pure phospholipids and cholesterol, both CED-9 and Bcl-xL were shown to disassemble Cer-induced conductances arguing that the action of Bcl-xL is directly on the Cer channel (Siskind et al., 2008).
Using a variety of Cer analogs, it was demonstrated that Bcl-xL preferentially binds the aliphatic hydrocarbon chains in Cer (Perera, Lin et al., 2012). Bcl-xL interferes with Cer channel formation optimally when the Cer used has C16- or C18- fatty acyl side chain. This interference is markedly reduced when using Cer with longer or shorter chains (Perera, Lin et al., 2012). Mutant Bcl-xL protein which is not anti-apoptotic did not reduce the MOMP by Cer (Siskind et al., 2008) hence the effect of Bcl-xL is protein- and lipid-specific. Furthermore, neither SM hydrolysis nor Cer generation was affected by Bcl-2 overexpression in leukemia cells even though this overexpression protected the cells from undergoing apoptosis; therefore, the protection by Bcl-2 of apoptosis downstream to Cer generation (Allouche et al., 1997) in agreement with a direct effect of anti-apoptotic Bcl-2 family proteins of Cer channel itself.
7.2. Stabilizers: pro-apoptotic Bcl-2 proteins
Naturally, the effects of pro-apoptotic Bcl-2 family proteins are the opposite to those of their anti-apoptotic family members. Some pro-apoptotic Bcl-2 family proteins work by permeabilizing the mitochondrial outer membrane. Activated Bax is able to permeabilize mitochondria without the need of Cer. On the other hand, Cer is able to cause MOMP in cells deficient of Bax or Bak as well as in protein-free membranes. However, treating isolated mitochondria with both activated Bax and Cer, for instance, induced a higher level of permeabilization than either treatment alone, in synergy (Ganesan et al., 2010). Bax and Cer were also reported to act synergistically in irradiated HeLa cells to facilitate MOMP and cytochrome c release (Lee et al., 2011). Synergy was also reported between Bax and Cer in inducing mtPTP (Pastorino et al., 1999). As opposed to Bcl-xL, Bax binds at a different region of the Cer channel as it preferentially binds Cer at the amide nitrogen. When this nitrogen is blocked by methylation the interaction is lost (Perera, Lin et al., 2012). Activated Bax is thought to be a scaffolding protein on which Cer channel is driven to a specific radius of curvature (Colombini, 2013). In this sense, Bax is functioning as a Cer channel stabilizer and the synergy is contingent on Cer domains aiding in Bax oligomerization and Bax stabilizing Cer channel.
So far, Bax is the sole molecule that has been reported to stabilize Cer channels in vitro. We expect more proteins to follow suit, as stabilization of the channel is required for a sustained response to initiate apoptosis. Another pro-apoptotic Bcl-2 protein, Bak, was recently shown to enhance Cer production upon UV-C irradiation, cisplatin or growth factor withdrawal. This enhancement was due to the activation of CerS by Bak and not Bax (Siskind et al., 2010). Thus it is striking to observe that Bax and Bak (which are considered to be redundant proteins) operate via two very different mechanisms pertaining interaction with Cer metabolism in vivo.
8. Conclusion and future directions
In this brief review the role of Cer in the initiation of apoptosis was probed, particularly its ability to form channels in mitochondrial outer membranes. The relationship between Cer, Cer metabolism, Cer channels, and Bcl-2 family proteins was also discussed in light of new and exciting data. Apoptosis is an elaborate process that requires intricate networks of molecules working together to produce a timely response. Mitochondrial apoptosis is contingent on MOMP. There are multiple proposed mechanisms for the pathway through the outer membrane that allows IMS proteins to be released to initiate apoptosis. Due to the complex nature of apoptosis, it is unlikely that the method of IMS proteins release is solitary. In vivo there are a plethora of signaling molecules, signaling events and cascades that need to be coordinated in order to achieve the ultimate goal of killing the cell without harming the surrounding neighbors. Pro-apoptotic Bcl-2 family proteins form a water-filled pathway in the outer membrane, but so does Cer. Both molecules are therefore involved in MOMP directly. The regulation of Cer channel formation and disassembly is an established field now. It is therefore a challenge lying ahead to identify different inhibitors or Cer channels in vitro as well as in vivo whether or not these molecules are members of Bcl-2 family. Furthermore, the coordination of Cer channels with other outer membrane channels may be probed to identify other synergistic effects of membrane Cer.
Acknowledgements
This work has been possible by the receipt of Grant No. 240151 from the office of the dean of graduate studies at Birzeit University for J.S. on which M.A.G. worked as a research assistant. The authors would like to thank Dr. Emilia Rappocciolo from the Biology and Biochemistry department at Birzeit University for her critical proofreading of this review.
Footnotes
Peer review under responsibility of King Saud University.
References
- Adan-Gokbulut A., Kartal-Yandim M., Iskender G., Baran Y. Novel agents targeting bioactive sphingolipids for the treatment of cancer. Curr. Med. Chem. 2013;20:108–122. [PubMed] [Google Scholar]
- Allouche M., Bettaieb A., Vindis C., Rousse A., Grignon C., Laurent G. Influence of Bcl-2 overexpression on the ceramide pathway in daunorubicin-induced apoptosis of leukemic cells. Oncogene. 1997;14:1837–1845. doi: 10.1038/sj.onc.1201023. [DOI] [PubMed] [Google Scholar]
- Anishkin A., Sukharev S., Colombini M. Searching for the molecular arrangement of transmembrane ceramide channels. Biophys. J. 2006;90:2414–2426. doi: 10.1529/biophysj.105.071977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Antonsson B. Bax and other pro-apoptotic Bcl-2 family “killer-proteins” and their victim the mitochondrion. Cell Tissue Res. 2001;306:347–361. doi: 10.1007/s00441-001-0472-0. [DOI] [PubMed] [Google Scholar]
- Antonsson B., Montessuit S., Lauper S., Eskes R., Martinou J.C. Bax oligomerization is required for channel-forming activity in liposomes and to trigger cytochrome c release from mitochondria. Biochem. J. 2000;345(Pt 2):271–278. [PMC free article] [PubMed] [Google Scholar]
- Antonsson B., Montessuit S., Sanchez B., Martinou J.C. Bax is present as a high molecular weight oligomer/complex in the mitochondrial membrane of apoptotic cells. J. Biol. Chem. 2001;276:11615–11623. doi: 10.1074/jbc.M010810200. [DOI] [PubMed] [Google Scholar]
- Armstrong C.M., Cota G. Modification of sodium channel gating by lanthanum. Some effects that cannot be explained by surface charge theory. J. Gen. Physiol. 1990;96:1129–1140. doi: 10.1085/jgp.96.6.1129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Azzam R., Hariri F., El-Hachem N., Kamar A., Dbaibo G., Nemer G., Bitar F. Regulation of de novo ceramide synthesis: the role of dihydroceramide desaturase and transcriptional factors NFATC and Hand2 in the hypoxic mouse heart. DNA Cell Biol. 2013;32:310–319. doi: 10.1089/dna.2013.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barbarroja N., Rodriguez-Cuenca S., Nygren H., Camargo A., Pirraco A., Relat J., Cuadrado I., Pellegrinelli V., Medina-Gomez G., Lopez-Pedrera C., Tinahones F.J., Symons J.D., Summers S.A., Oresic M., Vidal-Puig A. Increased dihydroceramide/ceramide ratio mediated by defective expression of degs1 impairs adipocyte differentiation and function. Diabetes. 2014;64:1180–1192. doi: 10.2337/db14-0359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beauchamp E., Tekpli X., Marteil G., Lagadic-Gossmann D., Legrand P., Rioux V. N-Myristoylation targets dihydroceramide Delta4-desaturase 1 to mitochondria: partial involvement in the apoptotic effect of myristic acid. Biochimie. 2009;91:1411–1419. doi: 10.1016/j.biochi.2009.07.014. [DOI] [PubMed] [Google Scholar]
- Becker K.P., Hannun Y.A. Protein kinase C and phospholipase D: intimate interactions in intracellular signaling. Cell. Mol. Life Sci. 2005;62:1448–1461. doi: 10.1007/s00018-005-4531-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Belaud-Rotureau M.A., Leducq N., Macouillard Poulletier de Gannes F., Diolez P., Lacoste L., Lacombe F., Bernard P., Belloc F. Early transitory rise in intracellular pH leads to Bax conformation change during ceramide-induced apoptosis. Apoptosis. 2000;5:551–560. doi: 10.1023/a:1009693630664. [DOI] [PubMed] [Google Scholar]
- Ben-David O., Futerman A.H. The role of the ceramide acyl chain length in neurodegeneration: involvement of ceramide synthases. NeuroMol. Med. 2010;12:341–350. doi: 10.1007/s12017-010-8114-x. [DOI] [PubMed] [Google Scholar]
- Beverly L.J. Regulation of anti-apoptotic BCL2-proteins by non-canonical interactions: the next step forward or two steps back? J. Cell. Biochem. 2012;113:3–12. doi: 10.1002/jcb.23335. [DOI] [PubMed] [Google Scholar]
- Beverly L.J., Howell L.A., Hernandez-Corbacho M., Casson L., Chipuk J.E., Siskind L.J. BAK activation is necessary and sufficient to drive ceramide synthase-dependent ceramide accumulation following inhibition of BCL2-like proteins. Biochem. J. 2013;452:111–119. doi: 10.1042/BJ20130147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Birbes H., El Bawab S., Hannun Y.A., Obeid L.M. Selective hydrolysis of a mitochondrial pool of sphingomyelin induces apoptosis. FASEB J. 2001;15:2669–2679. doi: 10.1096/fj.01-0539com. [DOI] [PubMed] [Google Scholar]
- Birbes H., El Bawab S., Obeid L.M., Hannun Y.A. Mitochondria and ceramide: intertwined roles in regulation of apoptosis. Adv. Enzyme Regul. 2002;42:113–129. doi: 10.1016/s0065-2571(01)00026-7. [DOI] [PubMed] [Google Scholar]
- Block W. Water or ice? – the challenge for invertebrate cold survival. Sci. Prog. 2003;86:77–101. doi: 10.3184/003685003783238680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Breen P., Joseph N., Thompson K., Kraveka J.M., Gudz T.I., Li L., Rahmaniyan M., Bielawski J., Pierce J.S., Van Buren E., Bhatti G., Separovic D. Dihydroceramide desaturase knockdown impacts sphingolipids and apoptosis after photodamage in human head and neck squamous carcinoma cells. Anticancer Res. 2013;33:77–84. [PMC free article] [PubMed] [Google Scholar]
- Chalfant C.E., Ogretmen B., Galadari S., Kroesen B.J., Pettus B.J., Hannun Y.A. FAS activation induces dephosphorylation of SR proteins; dependence on the de novo generation of ceramide and activation of protein phosphatase 1. J. Biol. Chem. 2001;276:44848–44855. doi: 10.1074/jbc.M106291200. [DOI] [PubMed] [Google Scholar]
- Chipuk J.E., Fisher J.C., Dillon C.P., Kriwacki R.W., Kuwana T., Green D.R. Mechanism of apoptosis induction by inhibition of the anti-apoptotic BCL-2 proteins. Proc. Natl. Acad. Sci. U.S.A. 2008;105:20327–20332. doi: 10.1073/pnas.0808036105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chipuk J.E., Moldoveanu T., Llambi F., Parsons M.J., Green D.R. The BCL-2 family reunion. Mol. Cell. 2010;37:299–310. doi: 10.1016/j.molcel.2010.01.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Colombini M. Ceramide channels and their role in mitochondria-mediated apoptosis. Biochim. Biophys. Acta. 2010;1797:1239–1244. doi: 10.1016/j.bbabio.2010.01.021. [DOI] [PubMed] [Google Scholar]
- Colombini M. Mitochondrial outer membrane channels. Chem. Rev. 2012;112:6373–6387. doi: 10.1021/cr3002033. [DOI] [PubMed] [Google Scholar]
- Colombini M. Membrane channels formed by ceramide. Handb. Exp. Pharmacol. 2013:109–126. doi: 10.1007/978-3-7091-1368-4_6. [DOI] [PubMed] [Google Scholar]
- Cordone L., Cottone G., Giuffrida S., Palazzo G., Venturoli G., Viappiani C. Internal dynamics and protein-matrix coupling in trehalose-coated proteins. Biochim. Biophys. Acta. 2005;1749:252–281. doi: 10.1016/j.bbapap.2005.03.004. [DOI] [PubMed] [Google Scholar]
- Cory S., Adams J.M. The Bcl2 family: regulators of the cellular life-or-death switch. Nat. Rev. Cancer. 2002;2:647–656. doi: 10.1038/nrc883. [DOI] [PubMed] [Google Scholar]
- Crompton M. The mitochondrial permeability transition pore and its role in cell death. Biochem. J. 1999;341(Pt 2):233–249. [PMC free article] [PubMed] [Google Scholar]
- Crompton M., Virji S., Doyle V., Johnson N., Ward J.M. The mitochondrial permeability transition pore. Biochem. Soc. Symp. 1999;66:167–179. doi: 10.1042/bss0660167. [DOI] [PubMed] [Google Scholar]
- Dai Q., Liu J., Chen J., Durrant D., McIntyre T.M., Lee R.M. Mitochondrial ceramide increases in UV-irradiated HeLa cells and is mainly derived from hydrolysis of sphingomyelin. Oncogene. 2004;23:3650–3658. doi: 10.1038/sj.onc.1207430. [DOI] [PubMed] [Google Scholar]
- Danial N.N., Korsmeyer S.J. Cell death: critical control points. Cell. 2004;116:205–219. doi: 10.1016/s0092-8674(04)00046-7. [DOI] [PubMed] [Google Scholar]
- Degterev A., Boyce M., Yuan J. A decade of caspases. Oncogene. 2003;22:8543–8567. doi: 10.1038/sj.onc.1207107. [DOI] [PubMed] [Google Scholar]
- Dewson G., Kratina T., Sim H.W., Puthalakath H., Adams J.M., Colman P.M., Kluck R.M. To trigger apoptosis, Bak exposes its BH3 domain and homodimerizes via BH3:groove interactions. Mol. Cell. 2008;30:369–380. doi: 10.1016/j.molcel.2008.04.005. [DOI] [PubMed] [Google Scholar]
- Di Paola M., Zaccagnino P., Montedoro G., Cocco T., Lorusso M. Ceramide induces release of pro-apoptotic proteins from mitochondria by either a Ca2+ -dependent or a Ca2+ -independent mechanism. J. Bioenerg. Biomembr. 2004;36:165–170. doi: 10.1023/b:jobb.0000023619.97392.0c. [DOI] [PubMed] [Google Scholar]
- Elrick M.J., Fluss S., Colombini M. Sphingosine, a product of ceramide hydrolysis, influences the formation of ceramide channels. Biophys. J. 2006;91:1749–1756. doi: 10.1529/biophysj.106.088443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Farrugia A. Albumin usage in clinical medicine: tradition or therapeutic? Transfus. Med. Rev. 2010;24:53–63. doi: 10.1016/j.tmrv.2009.09.005. [DOI] [PubMed] [Google Scholar]
- Fulda S., Gorman A.M., Hori O., Samali A. Cellular stress responses: cell survival and cell death. Int. J. Cell Biol. 2010;2010:214074. doi: 10.1155/2010/214074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ganesan V., Perera M.N., Colombini D., Datskovskiy D., Chadha K., Colombini M. Ceramide and activated Bax act synergistically to permeabilize the mitochondrial outer membrane. Apoptosis. 2010;15:553–562. doi: 10.1007/s10495-009-0449-0. [DOI] [PubMed] [Google Scholar]
- Garcia-Ruiz C., Colell A., Mari M., Morales A., Fernandez-Checa J.C. Direct effect of ceramide on the mitochondrial electron transport chain leads to generation of reactive oxygen species. Role of mitochondrial glutathione. J. Biol. Chem. 1997;272:11369–11377. doi: 10.1074/jbc.272.17.11369. [DOI] [PubMed] [Google Scholar]
- Ghafourifar P., Klein S.D., Schucht O., Schenk U., Pruschy M., Rocha S., Richter C. Ceramide induces cytochrome c release from isolated mitochondria. Importance of mitochondrial redox state. J. Biol. Chem. 1999;274:6080–6084. doi: 10.1074/jbc.274.10.6080. [DOI] [PubMed] [Google Scholar]
- Gincel D., Zaid H., Shoshan-Barmatz V. Calcium binding and translocation by the voltage-dependent anion channel: a possible regulatory mechanism in mitochondrial function. Biochem. J. 2001;358:147–155. doi: 10.1042/0264-6021:3580147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Green D.R. Apoptotic pathways: ten minutes to dead. Cell. 2005;121:671–674. doi: 10.1016/j.cell.2005.05.019. [DOI] [PubMed] [Google Scholar]
- Green D.R., Kroemer G. The pathophysiology of mitochondrial cell death. Science. 2004;305:626–629. doi: 10.1126/science.1099320. [DOI] [PubMed] [Google Scholar]
- Grosman C., Reisin I.L. Single-channel characterization of a nonselective cation channel from human placental microvillus membranes. Large conductance, multiplicity of conductance states, and inhibition by lanthanides. J. Membr. Biol. 2000;174:59–70. doi: 10.1007/s002320001032. [DOI] [PubMed] [Google Scholar]
- Grupe M., Myers G., Penner R., Fleig A. Activation of store-operated I(CRAC) by hydrogen peroxide. Cell Calcium. 2010;48:1–9. doi: 10.1016/j.ceca.2010.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gustin M.C., Zhou X.L., Martinac B., Kung C. A mechanosensitive ion channel in the yeast plasma membrane. Science. 1988;242:762–765. doi: 10.1126/science.2460920. [DOI] [PubMed] [Google Scholar]
- Hage-Sleiman R., Esmerian M.O., Kobeissy H., Dbaibo G. P53 and ceramide as collaborators in the stress response. Int. J. Mol. Sci. 2013;14:4982–5012. doi: 10.3390/ijms14034982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hammoudah M.M., Nir S., Isac T., Kornhauser R., Stewart T.P., Hui S.W., Vaz W.L. Interactions of La3+ with phosphatidylserine vesicles. Binding, phase transition, leakage and fusion. Biochim. Biophys. Acta. 1979;558:338–343. doi: 10.1016/0005-2736(79)90270-0. [DOI] [PubMed] [Google Scholar]
- Hannun Y.A. Functions of ceramide in coordinating cellular responses to stress. Science. 1996;274:1855–1859. doi: 10.1126/science.274.5294.1855. [DOI] [PubMed] [Google Scholar]
- Hannun Y.A., Luberto C., Argraves K.M. Enzymes of sphingolipid metabolism: from modular to integrative signaling. Biochemistry. 2001;40:4893–4903. doi: 10.1021/bi002836k. [DOI] [PubMed] [Google Scholar]
- Hart S.P., Haslett C., Dransfield I. Recognition of apoptotic cells by phagocytes. Experientia. 1996;52:950–956. doi: 10.1007/BF01920103. [DOI] [PubMed] [Google Scholar]
- Hartmann D., Lucks J., Fuchs S., Schiffmann S., Schreiber Y., Ferreiros N., Merkens J., Marschalek R., Geisslinger G., Grosch S. Long chain ceramides and very long chain ceramides have opposite effects on human breast and colon cancer cell growth. Int. J. Biochem. Cell Biol. 2012;44:620–628. doi: 10.1016/j.biocel.2011.12.019. [DOI] [PubMed] [Google Scholar]
- Hartmann D., Wegner M.S., Wanger R.A., Ferreiros N., Schreiber Y., Lucks J., Schiffmann S., Geisslinger G., Grosch S. The equilibrium between long and very long chain ceramides is important for the fate of the cell and can be influenced by co-expression of CerS. Int. J. Biochem. Cell Biol. 2013;45:1195–1203. doi: 10.1016/j.biocel.2013.03.012. [DOI] [PubMed] [Google Scholar]
- Hayakawa M., Jayadev S., Tsujimoto M., Hannun Y.A., Ito F. Role of ceramide in stimulation of the transcription of cytosolic phospholipase A2 and cyclooxygenase 2. Biochem. Biophys. Res. Commun. 1996;220:681–686. doi: 10.1006/bbrc.1996.0464. [DOI] [PubMed] [Google Scholar]
- Hengartner M.O. The biochemistry of apoptosis. Nature. 2000;407:770–776. doi: 10.1038/35037710. [DOI] [PubMed] [Google Scholar]
- Hirschberg K., Rodger J., Futerman A.H. The long-chain sphingoid base of sphingolipids is acylated at the cytosolic surface of the endoplasmic reticulum in rat liver. Biochem. J. 1993;290(Pt 3):751–757. doi: 10.1042/bj2900751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoth M., Penner R. Calcium release-activated calcium current in rat mast cells. J. Physiol. 1993;465:359–386. doi: 10.1113/jphysiol.1993.sp019681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacobson M.D., Burne J.F., King M.P., Miyashita T., Reed J.C., Raff M.C. Bcl-2 blocks apoptosis in cells lacking mitochondrial DNA. Nature. 1993;361:365–369. doi: 10.1038/361365a0. [DOI] [PubMed] [Google Scholar]
- Jacobson M.D., Burne J.F., Raff M.C. Mechanisms of programmed cell death and Bcl-2 protection. Biochem. Soc. Trans. 1994;22:600–602. doi: 10.1042/bst0220600. [DOI] [PubMed] [Google Scholar]
- Jacobson M.D., Burne J.F., Raff M.C. Programmed cell death and Bcl-2 protection in the absence of a nucleus. EMBO J. 1994;13:1899–1910. doi: 10.1002/j.1460-2075.1994.tb06459.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jayadev S., Hannun Y.A. Ceramide: role in growth inhibitory cascades. J. Lipid Mediators Cell Signal. 1996;14:295–301. doi: 10.1016/0929-7855(96)00538-x. [DOI] [PubMed] [Google Scholar]
- Jenkins G.M., Hannun Y.A. Role for de novo sphingoid base biosynthesis in the heat-induced transient cell cycle arrest of Saccharomyces cerevisiae. J. Biol. Chem. 2001;276:8574–8581. doi: 10.1074/jbc.M007425200. [DOI] [PubMed] [Google Scholar]
- Jensen S.A., Calvert A.E., Volpert G., Kouri F.M., Hurley L.A., Luciano J.P., Wu Y., Chalastanis A., Futerman A.H., Stegh A.H. Bcl2L13 is a ceramide synthase inhibitor in glioblastoma. Proc. Natl. Acad. Sci. U.S.A. 2014;111:5682–5687. doi: 10.1073/pnas.1316700111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kawatani M., Uchi M., Simizu S., Osada H., Imoto M. Transmembrane domain of Bcl-2 is required for inhibition of ceramide synthesis, but not cytochrome c release in the pathway of inostamycin-induced apoptosis. Exp. Cell Res. 2003;286:57–66. doi: 10.1016/s0014-4827(03)00098-3. [DOI] [PubMed] [Google Scholar]
- Kerr J.F., Wyllie A.H., Currie A.R. Apoptosis: a basic biological phenomenon with wide-ranging implications in tissue kinetics. Br. J. Cancer. 1972;26:239–257. doi: 10.1038/bjc.1972.33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim H., Tu H.C., Ren D., Takeuchi O., Jeffers J.R., Zambetti G.P., Hsieh J.J., Cheng E.H. Stepwise activation of BAX and BAK by tBID, BIM, and PUMA initiates mitochondrial apoptosis. Mol. Cell. 2009;36:487–499. doi: 10.1016/j.molcel.2009.09.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kitatani K., Idkowiak-Baldys J., Hannun Y.A. The sphingolipid salvage pathway in ceramide metabolism and signaling. Cell. Signal. 2008;20:1010–1018. doi: 10.1016/j.cellsig.2007.12.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuwana T., Mackey M.R., Perkins G., Ellisman M.H., Latterich M., Schneiter R., Green D.R., Newmeyer D.D. Bid, Bax, and lipids cooperate to form supramolecular openings in the outer mitochondrial membrane. Cell. 2002;111:331–342. doi: 10.1016/s0092-8674(02)01036-x. [DOI] [PubMed] [Google Scholar]
- Kuwana T., Bouchier-Hayes L., Chipuk J.E., Bonzon C., Sullivan B.A., Green D.R., Newmeyer D.D. BH3 domains of BH3-only proteins differentially regulate Bax-mediated mitochondrial membrane permeabilization both directly and indirectly. Mol. Cell. 2005;17:525–535. doi: 10.1016/j.molcel.2005.02.003. [DOI] [PubMed] [Google Scholar]
- Lai J.C., Tan W., Benimetskaya L., Miller P., Colombini M., Stein C.A. A pharmacologic target of G3139 in melanoma cells may be the mitochondrial VDAC. Proc. Natl. Acad. Sci. U.S.A. 2006;103:7494–7499. doi: 10.1073/pnas.0602217103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laviad E.L., Kelly S., Merrill A.H., Jr., Futerman A.H. Modulation of ceramide synthase activity via dimerization. J. Biol. Chem. 2012;287:21025–21033. doi: 10.1074/jbc.M112.363580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee J.Y., Hannun Y.A., Obeid L.M. Ceramide inactivates cellular protein kinase Calpha. J. Biol. Chem. 1996;271:13169–13174. doi: 10.1074/jbc.271.22.13169. [DOI] [PubMed] [Google Scholar]
- Lee J., Ishihara A., Oxford G., Johnson B., Jacobson K. Regulation of cell movement is mediated by stretch-activated calcium channels. Nature. 1999;400:382–386. doi: 10.1038/22578. [DOI] [PubMed] [Google Scholar]
- Lee H., Rotolo J.A., Mesicek J., Penate-Medina T., Rimner A., Liao W.C., Yin X., Ragupathi G., Ehleiter D., Gulbins E., Zhai D., Reed J.C., Haimovitz-Friedman A., Fuks Z., Kolesnick R. Mitochondrial ceramide-rich macrodomains functionalize Bax upon irradiation. PLoS One. 2011;6:e19783. doi: 10.1371/journal.pone.0019783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Letai A., Bassik M.C., Walensky L.D., Sorcinelli M.D., Weiler S., Korsmeyer S.J. Distinct BH3 domains either sensitize or activate mitochondrial apoptosis, serving as prototype cancer therapeutics. Cancer Cell. 2002;2:183–192. doi: 10.1016/s1535-6108(02)00127-7. [DOI] [PubMed] [Google Scholar]
- Linardic C.M., Jayadev S., Hannun Y.A. Activation of the sphingomyelin cycle by brefeldin A: effects of brefeldin A on differentiation and implications for a role for ceramide in regulation of protein trafficking. Cell Growth Differ. 1996;7:765–774. [PubMed] [Google Scholar]
- Liu X., Kim C.N., Yang J., Jemmerson R., Wang X. Induction of apoptotic program in cell-free extracts: requirement for dATP and cytochrome c. Cell. 1996;86:147–157. doi: 10.1016/s0092-8674(00)80085-9. [DOI] [PubMed] [Google Scholar]
- Mesicek J., Lee H., Feldman T., Jiang X., Skobeleva A., Berdyshev E.V., Haimovitz-Friedman A., Fuks Z., Kolesnick R. Ceramide synthases 2, 5, and 6 confer distinct roles in radiation-induced apoptosis in HeLa cells. Cell. Signal. 2010;22:1300–1307. doi: 10.1016/j.cellsig.2010.04.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Minn A.J., Kettlun C.S., Liang H., Kelekar A., Vander Heiden M.G., Chang B.S., Fesik S.W., Fill M., Thompson C.B. Bcl-xL regulates apoptosis by heterodimerization-dependent and -independent mechanisms. EMBO J. 1999;18:632–643. doi: 10.1093/emboj/18.3.632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Montes L.R., Ruiz-Arguello M.B., Goni F.M., Alonso A. Membrane restructuring via ceramide results in enhanced solute efflux. J. Biol. Chem. 2002;277:11788–11794. doi: 10.1074/jbc.M111568200. [DOI] [PubMed] [Google Scholar]
- Mullen T.D., Jenkins R.W., Clarke C.J., Bielawski J., Hannun Y.A., Obeid L.M. Ceramide synthase-dependent ceramide generation and programmed cell death: involvement of salvage pathway in regulating postmitochondrial events. J. Biol. Chem. 2011;286:15929–15942. doi: 10.1074/jbc.M111.230870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mullen T.D., Hannun Y.A., Obeid L.M. Ceramide synthases at the centre of sphingolipid metabolism and biology. Biochem. J. 2012;441:789–802. doi: 10.1042/BJ20111626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagata S. Apoptotic DNA fragmentation. Exp. Cell Res. 2000;256:12–18. doi: 10.1006/excr.2000.4834. [DOI] [PubMed] [Google Scholar]
- Nijhawan D., Fang M., Traer E., Zhong Q., Gao W., Du F., Wang X. Elimination of Mcl-1 is required for the initiation of apoptosis following ultraviolet irradiation. Genes Dev. 2003;17:1475–1486. doi: 10.1101/gad.1093903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Noureddine L., Azzam R., Nemer G., Bielawski J., Nasser M., Bitar F., Dbaibo G.S. Modulation of total ceramide and constituent ceramide species in the acutely and chronically hypoxic mouse heart at different ages. Prostaglandins Other Lipid Mediat. 2008;86:49–55. doi: 10.1016/j.prostaglandins.2008.02.003. [DOI] [PubMed] [Google Scholar]
- Novgorodov S.A., Chudakova D.A., Wheeler B.W., Bielawski J., Kindy M.S., Obeid L.M., Gudz T.I. Developmentally regulated ceramide synthase 6 increases mitochondrial Ca2+ loading capacity and promotes apoptosis. J. Biol. Chem. 2011;286:4644–4658. doi: 10.1074/jbc.M110.164392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Obeid L.M., Linardic C.M., Karolak L.A., Hannun Y.A. Programmed cell death induced by ceramide. Science. 1993;259:1769–1771. doi: 10.1126/science.8456305. [DOI] [PubMed] [Google Scholar]
- Pajewski R., Djedovic N., Harder E., Ferdani R., Schlesinger P.H., Gokel G.W. Pore formation in and enlargement of phospholipid liposomes by synthetic models of ceramides and sphingomyelin. Bioorg. Med. Chem. 2005;13:29–37. doi: 10.1016/j.bmc.2004.10.001. [DOI] [PubMed] [Google Scholar]
- Park J.W., Park W.J., Kuperman Y., Boura-Halfon S., Pewzner-Jung Y., Futerman A.H. Ablation of very long acyl chain sphingolipids causes hepatic insulin resistance in mice due to altered detergent-resistant membranes. Hepatology. 2013;57:525–532. doi: 10.1002/hep.26015. [DOI] [PubMed] [Google Scholar]
- Pastorino J.G., Tafani M., Rothman R.J., Marcinkeviciute A., Hoek J.B., Farber J.L. Functional consequences of the sustained or transient activation by Bax of the mitochondrial permeability transition pore. J. Biol. Chem. 1999;274:31734–31739. doi: 10.1074/jbc.274.44.31734. [DOI] [PubMed] [Google Scholar]
- Pavlov E.V., Priault M., Pietkiewicz D., Cheng E.H., Antonsson B., Manon S., Korsmeyer S.J., Mannella C.A., Kinnally K.W. A novel, high conductance channel of mitochondria linked to apoptosis in mammalian cells and Bax expression in yeast. J. Cell Biol. 2001;155:725–731. doi: 10.1083/jcb.200107057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perera M.N., Ganesan V., Siskind L.J., Szulc Z.M., Bielawski J., Bielawska A., Bittman R., Colombini M. Ceramide channels: influence of molecular structure on channel formation in membranes. Biochim. Biophys. Acta. 2012;1818:1291–1301. doi: 10.1016/j.bbamem.2012.02.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perera M.N., Lin S.H., Peterson Y.K., Bielawska A., Szulc Z.M., Bittman R., Colombini M. Bax and Bcl-xL exert their regulation on different sites of the ceramide channel. Biochem. J. 2012;445:81–91. doi: 10.1042/BJ20112103. [DOI] [PubMed] [Google Scholar]
- Perry D.K., Obeid L.M., Hannun Y.A. Ceramide and the regulation of apoptosis and the stress response. Trends Cardiovasc. Med. 1996;6:158–162. doi: 10.1016/1050-1738(96)00044-8. [DOI] [PubMed] [Google Scholar]
- Pinto S.N., Silva L.C., Futerman A.H., Prieto M. Effect of ceramide structure on membrane biophysical properties: the role of acyl chain length and unsaturation. Biochim. Biophys. Acta. 2011;1808:2753–2760. doi: 10.1016/j.bbamem.2011.07.023. [DOI] [PubMed] [Google Scholar]
- Pinto S.N., Laviad E.L., Stiban J., Kelly S.L., Merrill A.H., Jr., Prieto M., Futerman A.H., Silva L.C. Changes in membrane biophysical properties induced by sphingomyelinase depend on the sphingolipid N-acyl chain. J. Lipid Res. 2014;55:53–61. doi: 10.1194/jlr.M042002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Preston G.A., Barrett J.C., Biermann J.A., Murphy E. Effects of alterations in calcium homeostasis on apoptosis during neoplastic progression. Cancer Res. 1997;57:537–542. [PubMed] [Google Scholar]
- Raff M.C., Barres B.A., Burne J.F., Coles H.S., Ishizaki Y., Jacobson M.D. Programmed cell death and the control of cell survival: lessons from the nervous system. Science. 1993;262:695–700. doi: 10.1126/science.8235590. [DOI] [PubMed] [Google Scholar]
- Raff M.C., Barres B.A., Burne J.F., Coles H.S., Ishizaki Y., Jacobson M.D. Programmed cell death and the control of cell survival. Philos. Trans. R. Soc. London B Biol. Sci. 1994;345:265–268. doi: 10.1098/rstb.1994.0104. [DOI] [PubMed] [Google Scholar]
- Richter C., Ghafourifar P. Ceramide induces cytochrome c release from isolated mitochondria. Biochem. Soc. Symp. 1999;66:27–31. doi: 10.1042/bss0660027. [DOI] [PubMed] [Google Scholar]
- Rodriguez-Cuenca S., Barbarroja N., Vidal-Puig A. Dihydroceramide desaturase 1, the gatekeeper of ceramide induced lipotoxicity. Biochim. Biophys. Acta. 2015;1851:40–50. doi: 10.1016/j.bbalip.2014.09.021. [DOI] [PubMed] [Google Scholar]
- Ross P.E., Cahalan M.D. Ca2+ influx pathways mediated by swelling or stores depletion in mouse thymocytes. J. Gen. Physiol. 1995;106:415–444. doi: 10.1085/jgp.106.3.415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruoslahti E., Reed J.C. Anchorage dependence, integrins, and apoptosis. Cell. 1994;77:477–478. doi: 10.1016/0092-8674(94)90209-7. [DOI] [PubMed] [Google Scholar]
- Saba J.D., Obeid L.M., Hannun Y.A. Ceramide: an intracellular mediator of apoptosis and growth suppression. Philos. Trans. R. Soc. Lond. B Biol. Sci. 1996;351:233–240. doi: 10.1098/rstb.1996.0021. (discussion 240-231) [DOI] [PubMed] [Google Scholar]
- Saelens X., Festjens N., Vande Walle L., van Gurp M., van Loo G., Vandenabeele P. Toxic proteins released from mitochondria in cell death. Oncogene. 2004;23:2861–2874. doi: 10.1038/sj.onc.1207523. [DOI] [PubMed] [Google Scholar]
- Saito M., Korsmeyer S.J., Schlesinger P.H. BAX-dependent transport of cytochrome c reconstituted in pure liposomes. Nat. Cell Biol. 2000;2:553–555. doi: 10.1038/35019596. [DOI] [PubMed] [Google Scholar]
- Samali A., Fulda S., Gorman A.M., Hori O., Srinivasula S.M. Cell stress and cell death. Int. J. Cell Biol. 2010;2010:245803. doi: 10.1155/2010/245803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Samanta S., Stiban J., Maugel T.K., Colombini M. Visualization of ceramide channels by transmission electron microscopy. Biochim. Biophys. Acta. 2011;1808:1196–1201. doi: 10.1016/j.bbamem.2011.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanchis D., Mayorga M., Ballester M., Comella J.X. Lack of Apaf-1 expression confers resistance to cytochrome c-driven apoptosis in cardiomyocytes. Cell Death Differ. 2003;10:977–986. doi: 10.1038/sj.cdd.4401267. [DOI] [PubMed] [Google Scholar]
- Shao C., Sun B., DeVoe D.L., Colombini M. Dynamics of ceramide channels detected using a microfluidic system. PLoS One. 2012;7:e43513. doi: 10.1371/journal.pone.0043513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shin C.Y., Lee Y.P., Lee T.S., Song H.J., Sohn U.D. C(2)-ceramide-induced circular smooth muscle cell contraction involves PKC-epsilon and p44/p42 MAPK activation in cat oesophagus. Mitogen-activated protein kinase. Cell. Signal. 2002;14:925–932. doi: 10.1016/s0898-6568(02)00038-4. [DOI] [PubMed] [Google Scholar]
- Siddique M.M., Bikman B.T., Wang L., Ying L., Reinhardt E., Shui G., Wenk M.R., Summers S.A. Ablation of dihydroceramide desaturase confers resistance to etoposide-induced apoptosis in vitro. PLoS One. 2012;7:e44042. doi: 10.1371/journal.pone.0044042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siddique M.M., Li Y., Wang L., Ching J., Mal M., Ilkayeva O., Wu Y.J., Bay B.H., Summers S.A. Ablation of dihydroceramide desaturase 1, a therapeutic target for the treatment of metabolic diseases, simultaneously stimulates anabolic and catabolic signaling. Mol. Cell. Biol. 2013;33:2353–2369. doi: 10.1128/MCB.00226-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Silva L.C., Ben David O., Pewzner-Jung Y., Laviad E.L., Stiban J., Bandyopadhyay S., Merrill A.H., Jr., Prieto M., Futerman A.H. Ablation of ceramide synthase 2 strongly affects biophysical properties of membranes. J. Lipid Res. 2012;53:430–436. doi: 10.1194/jlr.M022715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Silva L.C., Futerman A.H., Prieto M. Lipid raft composition modulates sphingomyelinase activity and ceramide-induced membrane physical alterations. Biophys. J. 2009;96:3210–3222. doi: 10.1016/j.bpj.2008.12.3923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siskind L.J. Mitochondrial ceramide and the induction of apoptosis. J. Bioenerg. Biomembr. 2005;37:143–153. doi: 10.1007/s10863-005-6567-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siskind L.J., Colombini M. The lipids C2- and C16-ceramide form large stable channels. Implications for apoptosis. J. Biol. Chem. 2000;275:38640–38644. doi: 10.1074/jbc.C000587200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siskind L.J., Kolesnick R.N., Colombini M. Ceramide channels increase the permeability of the mitochondrial outer membrane to small proteins. J. Biol. Chem. 2002;277:26796–26803. doi: 10.1074/jbc.M200754200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siskind L.J., Davoody A., Lewin N., Marshall S., Colombini M. Enlargement and contracture of C2-ceramide channels. Biophys. J. 2003;85:1560–1575. doi: 10.1016/S0006-3495(03)74588-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siskind L.J., Fluss S., Bui M., Colombini M. Sphingosine forms channels in membranes that differ greatly from those formed by ceramide. J. Bioenerg. Biomembr. 2005;37:227–236. doi: 10.1007/s10863-005-6632-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siskind L.J., Kolesnick R.N., Colombini M. Ceramide forms channels in mitochondrial outer membranes at physiologically relevant concentrations. Mitochondrion. 2006;6:118–125. doi: 10.1016/j.mito.2006.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siskind L.J., Feinstein L., Yu T., Davis J.S., Jones D., Choi J., Zuckerman J.E., Tan W., Hill R.B., Hardwick J.M., Colombini M. Anti-apoptotic Bcl-2 family proteins disassemble ceramide channels. J. Biol. Chem. 2008;283:6622–6630. doi: 10.1074/jbc.M706115200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siskind L.J., Mullen T.D., Romero Rosales K., Clarke C.J., Hernandez-Corbacho M.J., Edinger A.L., Obeid L.M. The BCL-2 protein BAK is required for long-chain ceramide generation during apoptosis. J. Biol. Chem. 2010;285:11818–11826. doi: 10.1074/jbc.M109.078121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sorenson C.M. Bcl-2 family members and disease. Biochim. Biophys. Acta. 2004;1644:169–177. doi: 10.1016/j.bbamcr.2003.08.010. [DOI] [PubMed] [Google Scholar]
- Stiban J., Perera M. Very long chain ceramides interfere with C16-ceramide-induced channel formation: a plausible mechanism for regulating the initiation of intrinsic apoptosis. Biochim. Biophys. Acta. 2015;1848:561–567. doi: 10.1016/j.bbamem.2014.11.018. [DOI] [PubMed] [Google Scholar]
- Stiban J., Fistere D., Colombini M. Dihydroceramide hinders ceramide channel formation: implications on apoptosis. Apoptosis. 2006;11:773–780. doi: 10.1007/s10495-006-5882-8. [DOI] [PubMed] [Google Scholar]
- Stiban J., Caputo L., Colombini M. Ceramide synthesis in the endoplasmic reticulum can permeabilize mitochondria to proapoptotic proteins. J. Lipid Res. 2008;49:625–634. doi: 10.1194/jlr.M700480-JLR200. [DOI] [PubMed] [Google Scholar]
- Stiban J., Tidhar R., Futerman A.H. Ceramide synthases: roles in cell physiology and signaling. Adv. Exp. Med. Biol. 2010;688:60–71. doi: 10.1007/978-1-4419-6741-1_4. [DOI] [PubMed] [Google Scholar]
- Susin S.A., Daugas E., Ravagnan L., Samejima K., Zamzami N., Loeffler M., Costantini P., Ferri K.F., Irinopoulou T., Prevost M.C., Brothers G., Mak T.W., Penninger J., Earnshaw W.C., Kroemer G. Two distinct pathways leading to nuclear apoptosis. J. Exp. Med. 2000;192:571–580. doi: 10.1084/jem.192.4.571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tanaka T., Li S.J., Kinoshita K., Yamazaki M. La(3+) stabilizes the hexagonal II (H(II)) phase in phosphatidylethanolamine membranes. Biochim. Biophys. Acta. 2001;1515:189–201. doi: 10.1016/s0005-2736(01)00413-8. [DOI] [PubMed] [Google Scholar]
- Vucic D., Deshayes K., Ackerly H., Pisabarro M.T., Kadkhodayan S., Fairbrother W.J., Dixit V.M. SMAC negatively regulates the anti-apoptotic activity of melanoma inhibitor of apoptosis (ML-IAP) J. Biol. Chem. 2002;277:12275–12279. doi: 10.1074/jbc.M112045200. [DOI] [PubMed] [Google Scholar]
- Wang J.Y. DNA damage and apoptosis. Cell Death Differ. 2001;8:1047–1048. doi: 10.1038/sj.cdd.4400938. [DOI] [PubMed] [Google Scholar]
- Wiemken A. Trehalose in yeast, stress protectant rather than reserve carbohydrate. Antonie Van Leeuwenhoek. 1990;58:209–217. doi: 10.1007/BF00548935. [DOI] [PubMed] [Google Scholar]
- Wiesner D.A., Dawson G. Programmed cell death in neurotumour cells involves the generation of ceramide. Glycoconjugate J. 1996;13:327–333. doi: 10.1007/BF00731508. [DOI] [PubMed] [Google Scholar]
- Wiesner D.A., Dawson G. Staurosporine induces programmed cell death in embryonic neurons and activation of the ceramide pathway. J. Neurochem. 1996;66:1418–1425. doi: 10.1046/j.1471-4159.1996.66041418.x. [DOI] [PubMed] [Google Scholar]
- Yang J., Liu X., Bhalla K., Kim C.N., Ibrado A.M., Cai J., Peng T.I., Jones D.P., Wang X. Prevention of apoptosis by Bcl-2: release of cytochrome c from mitochondria blocked. Science. 1997;275:1129–1132. doi: 10.1126/science.275.5303.1129. [DOI] [PubMed] [Google Scholar]
- Yoon G., Kim K.O., Lee J., Kwon D., Shin J.S., Kim S.J., Choi I.H. Ceramide increases Fas-mediated apoptosis in glioblastoma cells through FLIP down-regulation. J. Neurooncol. 2002;60:135–141. doi: 10.1023/a:1020604705831. [DOI] [PubMed] [Google Scholar]
- Yu J., Novgorodov S.A., Chudakova D., Zhu H., Bielawska A., Bielawski J., Obeid L.M., Kindy M.S., Gudz T.I. JNK3 signaling pathway activates ceramide synthase leading to mitochondrial dysfunction. J. Biol. Chem. 2007;282:25940–25949. doi: 10.1074/jbc.M701812200. [DOI] [PubMed] [Google Scholar]
- Zhang T., Saghatelian A. Emerging roles of lipids in BCL-2 family-regulated apoptosis. Biochim. Biophys. Acta. 2013;1831:1542–1554. doi: 10.1016/j.bbalip.2013.03.001. [DOI] [PubMed] [Google Scholar]
- Zhang J., Alter N., Reed J.C., Borner C., Obeid L.M., Hannun Y.A. Bcl-2 interrupts the ceramide-mediated pathway of cell death. Proc. Natl. Acad. Sci. U.S.A. 1996;93:5325–5328. doi: 10.1073/pnas.93.11.5325. [DOI] [PMC free article] [PubMed] [Google Scholar]