Abstract
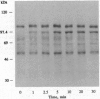
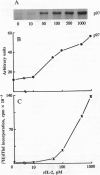
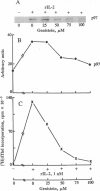
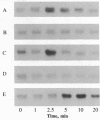
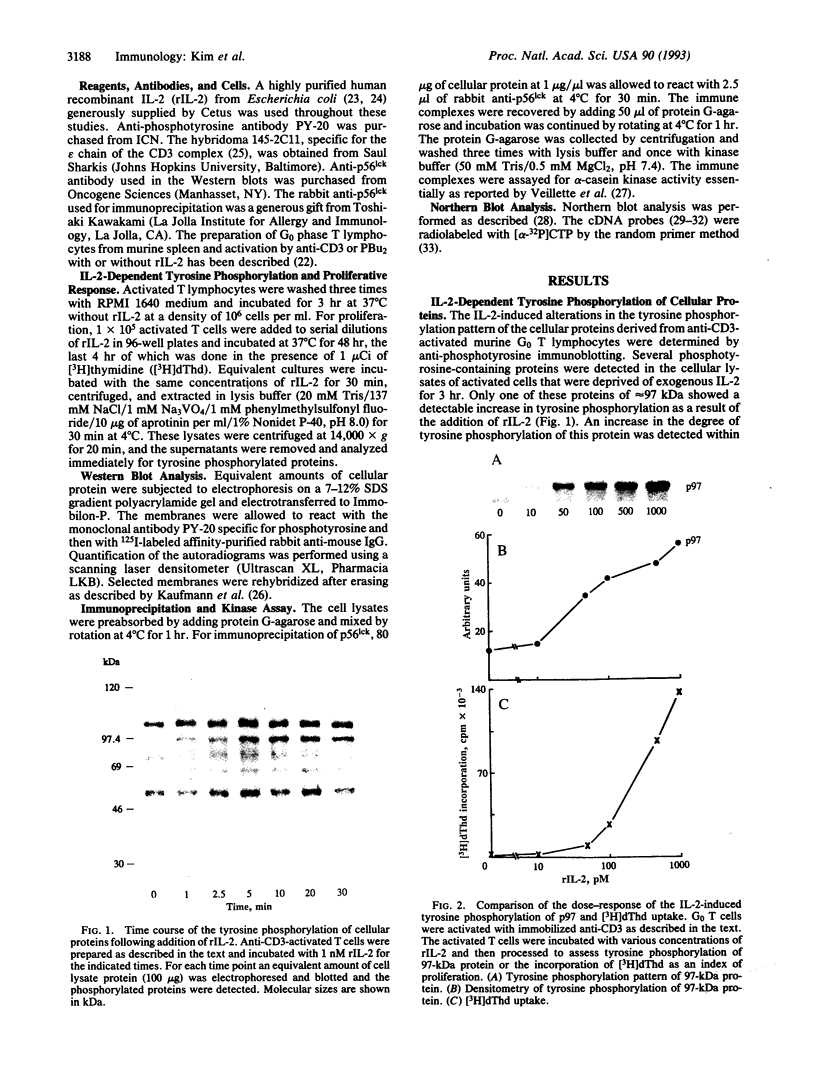
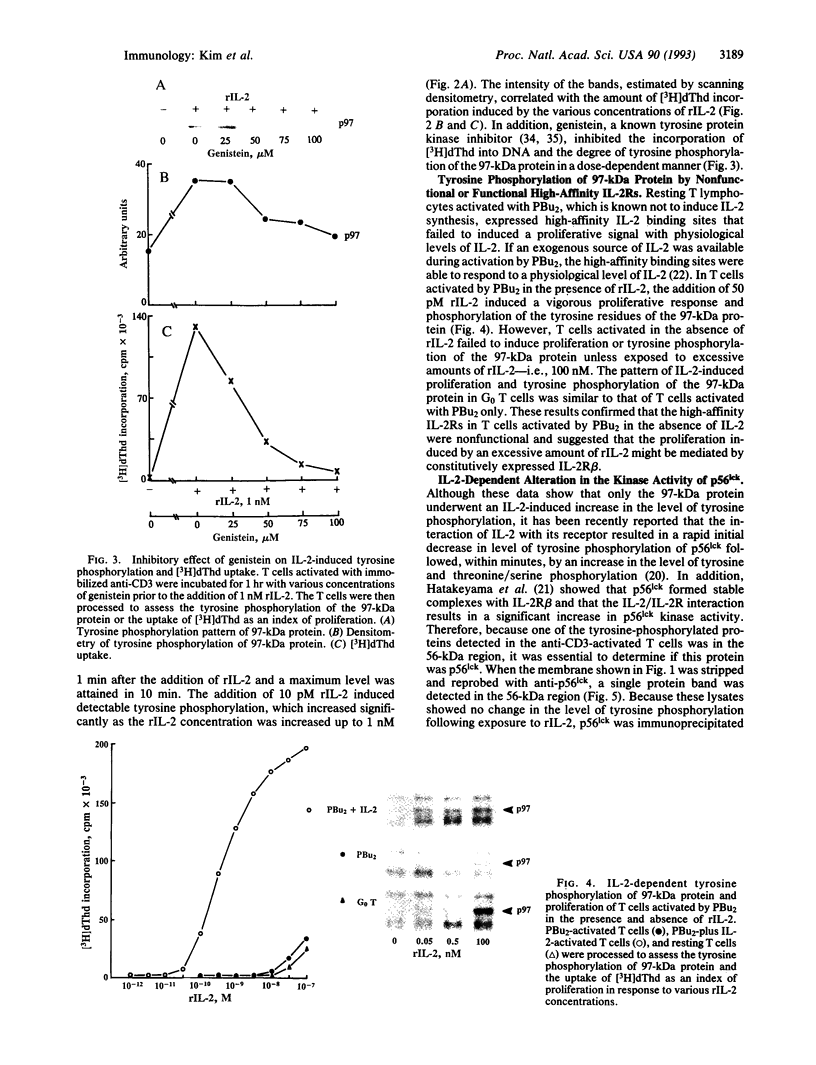
The addition of recombinant interleukin 2 (rIL-2) to anti-CD3-activated murine G0 phase T cells results in an increased level of tyrosine phosphorylation of a single 97-kDa protein. The degree of tyrosine phosphorylation paralleled the amount of rIL-2 added and correlated with the extent of DNA synthesis. IL-2 treatment resulted in a transient increase in p56lck kinase activity without detectable modification of its level of tyrosine phosphorylation and gel mobility. When G0 T cells were activated by phorbol dibutyrate in the absence of IL-2, the high-affinity IL-2 receptor (IL-2R) expressed failed to induce a proliferative signal, and neither the tyrosine phosphorylation of the 97-kDa protein nor the transient increase in p56lck kinase activity was detected. Northern analysis of the total RNA extracted from these cells showed the accumulation of IL-2R alpha chain-specific mRNA but neither c-myc nor cdc2 mRNA was expressed. The addition of 100 nM rIL-2 to T cells activated by phorbol dibutyrate was able to induce a proliferative response, and under these conditions tyrosine phosphorylation of the 97-kDa protein, the transient increase in p56lck kinase activity, and specific mRNA for IL-2R alpha chain, c-myc, and cdc2 were detected. Unstimulated G0 T cells responded to 100 nM rIL-2 in the same manner as phorbol dibutyrate-activated cells. Irrespective of the signal-transducing structures involved, the IL-2-induced proliferative response closely correlates with an increase in p56lck kinase activity along with the tyrosine phosphorylation of a 97-kDa protein.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Akiyama T., Ishida J., Nakagawa S., Ogawara H., Watanabe S., Itoh N., Shibuya M., Fukami Y. Genistein, a specific inhibitor of tyrosine-specific protein kinases. J Biol Chem. 1987 Apr 25;262(12):5592–5595. [PubMed] [Google Scholar]
- Augustine J. A., Schlager J. W., Abraham R. T. Differential effects of interleukin-2 and interleukin-4 on protein tyrosine phosphorylation in factor-dependent murine T cells. Biochim Biophys Acta. 1990 May 2;1052(2):313–322. doi: 10.1016/0167-4889(90)90227-5. [DOI] [PubMed] [Google Scholar]
- Bishop J. M. Viral oncogenes. Cell. 1985 Aug;42(1):23–38. doi: 10.1016/s0092-8674(85)80098-2. [DOI] [PubMed] [Google Scholar]
- Burton J., Goldman C. K., Rao P., Moos M., Waldmann T. A. Association of intercellular adhesion molecule 1 with the multichain high-affinity interleukin 2 receptor. Proc Natl Acad Sci U S A. 1990 Sep;87(18):7329–7333. doi: 10.1073/pnas.87.18.7329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chan Y. L., Gutell R., Noller H. F., Wool I. G. The nucleotide sequence of a rat 18 S ribosomal ribonucleic acid gene and a proposal for the secondary structure of 18 S ribosomal ribonucleic acid. J Biol Chem. 1984 Jan 10;259(1):224–230. [PubMed] [Google Scholar]
- Cisek L. J., Corden J. L. Phosphorylation of RNA polymerase by the murine homologue of the cell-cycle control protein cdc2. Nature. 1989 Jun 29;339(6227):679–684. doi: 10.1038/339679a0. [DOI] [PubMed] [Google Scholar]
- Colamonici O. R., Neckers L. M., Rosolen A. Putative gamma-subunit of the IL-2 receptor is detected in low, intermediate, and high affinity IL-2 receptor-bearing cells. J Immunol. 1990 Jul 1;145(1):155–160. [PubMed] [Google Scholar]
- Crabtree G. R. Contingent genetic regulatory events in T lymphocyte activation. Science. 1989 Jan 20;243(4889):355–361. doi: 10.1126/science.2783497. [DOI] [PubMed] [Google Scholar]
- Dymecki S. M., Niederhuber J. E., Desiderio S. V. Specific expression of a tyrosine kinase gene, blk, in B lymphoid cells. Science. 1990 Jan 19;247(4940):332–336. doi: 10.1126/science.2404338. [DOI] [PubMed] [Google Scholar]
- Feinberg A. P., Vogelstein B. A technique for radiolabeling DNA restriction endonuclease fragments to high specific activity. Anal Biochem. 1983 Jul 1;132(1):6–13. doi: 10.1016/0003-2697(83)90418-9. [DOI] [PubMed] [Google Scholar]
- Fung M. R., Greene W. C. The human interleukin-2 receptor: insights into subunit structure and growth signal transduction. Semin Immunol. 1990 Mar;2(2):119–128. [PubMed] [Google Scholar]
- Hatakeyama M., Kono T., Kobayashi N., Kawahara A., Levin S. D., Perlmutter R. M., Taniguchi T. Interaction of the IL-2 receptor with the src-family kinase p56lck: identification of novel intermolecular association. Science. 1991 Jun 14;252(5012):1523–1528. doi: 10.1126/science.2047859. [DOI] [PubMed] [Google Scholar]
- Horak I. D., Gress R. E., Lucas P. J., Horak E. M., Waldmann T. A., Bolen J. B. T-lymphocyte interleukin 2-dependent tyrosine protein kinase signal transduction involves the activation of p56lck. Proc Natl Acad Sci U S A. 1991 Mar 1;88(5):1996–2000. doi: 10.1073/pnas.88.5.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaufmann S. H., Ewing C. M., Shaper J. H. The erasable Western blot. Anal Biochem. 1987 Feb 15;161(1):89–95. doi: 10.1016/0003-2697(87)90656-7. [DOI] [PubMed] [Google Scholar]
- Kim Y. H., Proust J. J., Buchholz M. J., Chrest F. J., Nordin A. A. Expression of the murine homologue of the cell cycle control protein p34cdc2 in T lymphocytes. J Immunol. 1992 Jul 1;149(1):17–23. [PubMed] [Google Scholar]
- Land H., Parada L. F., Weinberg R. A. Tumorigenic conversion of primary embryo fibroblasts requires at least two cooperating oncogenes. Nature. 1983 Aug 18;304(5927):596–602. doi: 10.1038/304596a0. [DOI] [PubMed] [Google Scholar]
- Leo O., Foo M., Sachs D. H., Samelson L. E., Bluestone J. A. Identification of a monoclonal antibody specific for a murine T3 polypeptide. Proc Natl Acad Sci U S A. 1987 Mar;84(5):1374–1378. doi: 10.1073/pnas.84.5.1374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Linnekin D., Evans G., Michiel D., Farrar W. L. Characterization of a 97-kDa phosphotyrosylprotein regulated by multiple cytokines. J Biol Chem. 1992 Nov 25;267(33):23993–23998. [PubMed] [Google Scholar]
- Miller J., Malek T. R., Leonard W. J., Greene W. C., Shevach E. M., Germain R. N. Nucleotide sequence and expression of a mouse interleukin 2 receptor cDNA. J Immunol. 1985 Jun;134(6):4212–4217. [PubMed] [Google Scholar]
- Mills G. B., Zhang N., Schmandt R., Fung M., Greene W., Mellors A., Hogg D. Transmembrane signalling by interleukin 2. Biochem Soc Trans. 1991 Apr;19(2):277–287. doi: 10.1042/bst0190277. [DOI] [PubMed] [Google Scholar]
- Neckers L. M., Cossman J. Transferrin receptor induction in mitogen-stimulated human T lymphocytes is required for DNA synthesis and cell division and is regulated by interleukin 2. Proc Natl Acad Sci U S A. 1983 Jun;80(11):3494–3498. doi: 10.1073/pnas.80.11.3494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perlmutter R. M., Marth J. D., Ziegler S. F., Garvin A. M., Pawar S., Cooke M. P., Abraham K. M. Specialized protein tyrosine kinase proto-oncogenes in hematopoietic cells. Biochim Biophys Acta. 1989 Feb;948(3):245–262. doi: 10.1016/0304-419x(89)90001-2. [DOI] [PubMed] [Google Scholar]
- Proust J. J., Shaper N. L., Buchholz M. A., Nordin A. A. T cell activation in the absence of interleukin 2 (IL 2) results in the induction of high-affinity IL 2 receptor unable to transmit a proliferative signal. Eur J Immunol. 1991 Feb;21(2):335–341. doi: 10.1002/eji.1830210214. [DOI] [PubMed] [Google Scholar]
- Reed J. C., Alpers J. D., Nowell P. C., Hoover R. G. Sequential expression of protooncogenes during lectin-stimulated mitogenesis of normal human lymphocytes. Proc Natl Acad Sci U S A. 1986 Jun;83(11):3982–3986. doi: 10.1073/pnas.83.11.3982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reed J. C., Sabath D. E., Hoover R. G., Prystowsky M. B. Recombinant interleukin 2 regulates levels of c-myc mRNA in a cloned murine T lymphocyte. Mol Cell Biol. 1985 Dec;5(12):3361–3368. doi: 10.1128/mcb.5.12.3361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robb R. J., Greene W. C. Internalization of interleukin 2 is mediated by the beta chain of the high-affinity interleukin 2 receptor. J Exp Med. 1987 Apr 1;165(4):1201–1206. doi: 10.1084/jem.165.4.1201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosenberg S. A., Grimm E. A., McGrogan M., Doyle M., Kawasaki E., Koths K., Mark D. F. Biological activity of recombinant human interleukin-2 produced in Escherichia coli. Science. 1984 Mar 30;223(4643):1412–1414. doi: 10.1126/science.6367046. [DOI] [PubMed] [Google Scholar]
- Saito Y., Tada H., Sabe H., Honjo T. Biochemical evidence for a third chain of the interleukin-2 receptor. J Biol Chem. 1991 Nov 25;266(33):22186–22191. [PubMed] [Google Scholar]
- Saltzman E. M., Thom R. R., Casnellie J. E. Activation of a tyrosine protein kinase is an early event in the stimulation of T lymphocytes by interleukin-2. J Biol Chem. 1988 May 25;263(15):6956–6959. [PubMed] [Google Scholar]
- Saragovi H., Malek T. R. Evidence for additional subunits associated to the mouse interleukin 2 receptor p55/p75 complex. Proc Natl Acad Sci U S A. 1990 Jan;87(1):11–15. doi: 10.1073/pnas.87.1.11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sharon M., Gnarra J. R., Leonard W. J. A 100-kilodalton protein is associated with the murine interleukin 2 receptor: biochemical evidence that p100 is distinct from the alpha and beta chains. Proc Natl Acad Sci U S A. 1990 Jun;87(12):4869–4873. doi: 10.1073/pnas.87.12.4869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siegel J. P., Sharon M., Smith P. L., Leonard W. J. The IL-2 receptor beta chain (p70): role in mediating signals for LAK, NK, and proliferative activities. Science. 1987 Oct 2;238(4823):75–78. doi: 10.1126/science.3116668. [DOI] [PubMed] [Google Scholar]
- Smith K. A. Interleukin-2: inception, impact, and implications. Science. 1988 May 27;240(4856):1169–1176. doi: 10.1126/science.3131876. [DOI] [PubMed] [Google Scholar]
- Stern J. B., Smith K. A. Interleukin-2 induction of T-cell G1 progression and c-myb expression. Science. 1986 Jul 11;233(4760):203–206. doi: 10.1126/science.3523754. [DOI] [PubMed] [Google Scholar]
- Takeshita T., Ohtani K., Asao H., Kumaki S., Nakamura M., Sugamura K. An associated molecule, p64, with IL-2 receptor beta chain. Its possible involvement in the formation of the functional intermediate-affinity IL-2 receptor complex. J Immunol. 1992 Apr 1;148(7):2154–2158. [PubMed] [Google Scholar]
- Trevillyan J. M., Lu Y. L., Atluru D., Phillips C. A., Bjorndahl J. M. Differential inhibition of T cell receptor signal transduction and early activation events by a selective inhibitor of protein-tyrosine kinase. J Immunol. 1990 Nov 15;145(10):3223–3230. [PubMed] [Google Scholar]
- Ullman K. S., Northrop J. P., Verweij C. L., Crabtree G. R. Transmission of signals from the T lymphocyte antigen receptor to the genes responsible for cell proliferation and immune function: the missing link. Annu Rev Immunol. 1990;8:421–452. doi: 10.1146/annurev.iy.08.040190.002225. [DOI] [PubMed] [Google Scholar]
- Veillette A., Horak I. D., Bolen J. B. Post-translational alterations of the tyrosine kinase p56lck in response to activators of protein kinase C. Oncogene Res. 1988 May;2(4):385–401. [PubMed] [Google Scholar]
- Waldmann T. A. The multi-subunit interleukin-2 receptor. Annu Rev Biochem. 1989;58:875–911. doi: 10.1146/annurev.bi.58.070189.004303. [DOI] [PubMed] [Google Scholar]
- Wang A., Lu S. D., Mark D. F. Site-specific mutagenesis of the human interleukin-2 gene: structure-function analysis of the cysteine residues. Science. 1984 Jun 29;224(4656):1431–1433. doi: 10.1126/science.6427925. [DOI] [PubMed] [Google Scholar]
- Wang H. M., Smith K. A. The interleukin 2 receptor. Functional consequences of its bimolecular structure. J Exp Med. 1987 Oct 1;166(4):1055–1069. doi: 10.1084/jem.166.4.1055. [DOI] [PMC free article] [PubMed] [Google Scholar]