Abstract
Pertussis (whooping cough) is a respiratory disease caused by the bacterium Bordetella pertussis. Despite the implementation of immunization programs and high vaccine coverage in most jurisdictions, pertussis is still one of the most common vaccine-preventable diseases, suggesting that the current vaccines and immunization schedules have not been sufficiently effective. Several factors are thought to contribute to this. The acellular pertussis vaccine that has been used in many jurisdictions since the 1990s is less effective than the previously used whole-cell vaccine, with immunity waning over time. Both whole-cell and acellular pertussis vaccines are effective at reducing disease severity but not transmission, resulting in outbreaks in vaccinated cohorts. In this review, we discuss various limitations of the current approaches to protection from pertussis and outline various options for reducing the burden of pertussis on a population level.
Keywords: Pertussis, whooping cough, vaccine, vaccine effectiveness, transmission, immunity
This review discusses various limitations of the current approaches to protection from pertussis.
Graphical Abstract Figure.
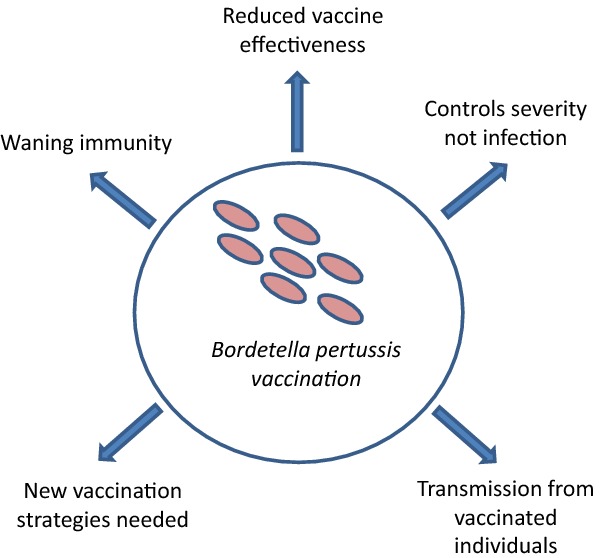
This review discusses various limitations of the current approaches to protection from pertussis.
INTRODUCTION
Recurrent pertussis outbreaks in the USA, France, UK and the Netherlands (Mooi et al. 2009; Billingsley 2012; Centres for Disease Control and Prevention 2012, 2013a; Cherry 2012a; Winter et al. 2012; Guiso 2014; Health Protection Agency 2013) as well as a persistent upward incidence trend in several jurisdictions (Guris et al. 1999; Tanaka et al. 2003) provide accumulating evidence that immunization programs using acellular pertussis (aP) vaccines have not been the success they were hoped to be. Several hypotheses have been presented to explain the resurgence of pertussis cases, including reduced vaccine effectiveness, a waning immune response that is mismatched to the pathogen, suboptimal vaccine coverage and ongoing transmission in vaccinated individuals (Burns, Meade and Messionnier 2014). It is likely that, rather than one of these being the reason for the shortcomings of the aP vaccine, several factors are responsible (Burns, Meade and Messionnier 2014). It is time to go back to basics in order to move forward in achieving the primary aim of pertussis immunization programs—to protect infants who have not completed their primary immunization series from severe disease (World Health Organization 2010). In achieving this, we may also succeed at secondary aims, like reducing morbidity and transmission in older age groups. New thinking and a new strategy is needed that is informed by a multidisciplinary approach to the challenges presented by this fascinating organism.
REDUCED EFFECTIVENESS OF THE aP VACCINE
Compared with the best whole-cell pertussis (wP) vaccines, aP vaccines are not as effective in mass immunization programs (Vickers et al. 2006; Cherry 2012b), although their success in having far fewer reactions than were associated with wP vaccines (Zhang et al. 2014) should not be discounted. Recent studies of outbreaks in highly immunized populations have shown that the duration of protection of aP vaccines is too short (Klein et al. 2012; Misegades et al. 2012), resulting in a decrease in immunity in older children and adolescents, and a corresponding increase in cases in this age group (Skowronski et al. 2002; Klein et al. 2012). This is in contrast to wP vaccines, which provide protection well into the teenaged years (Klein et al. 2012). As a result of these shortcomings, in countries that switched to the aP vaccine in the 1990s we now have a generation of children not only less well-protected against pertussis but who may also be less responsive to boosters, since the vaccine with which a child is primed may determine their immune response to later booster vaccination (Podda et al. 1995; Mascart et al. 2007; Sheridan et al. 2012; Liko, Robison and Cieslak 2013; Smits et al. 2013).
WANING IMMUNITY TO PERTUSSIS OVER TIME
Progress in understanding the immunology of pertussis vaccines has been hampered by the lack of a simple humoral correlate of protection (Plotkin 2014); a standard used by industry and regulators for assessing vaccine efficacy (Olin et al. 2001). That aP vaccines are less successful in eliminating pertussis than wP vaccines illustrates the limitations arising from the lack of a relevant correlate of protection (humoral or cell mediated), or method of assessment that could be standardized. It is known that, unlike natural infection and wP vaccination, aP vaccines do little to engage cellular immunity and Th1 responses, which, in addition to Th17 responses, have been shown to be essential for clearance of Bordetella pertussis and may be the key to sustained protection (Ryan et al. 1998; Higgs et al. 2012; Ross et al. 2013). At the same time, the aP vaccine stimulates a robust Th2 response, perhaps partially due to the fact that aP vaccines are adjuvanted by alum, a Th2 stimulator (Ross et al. 2013). This is in contrast to wP vaccines, which include the Th1 inducer lipopolysaccharide (Barton and Medzhitov 2002). The Th2 response elicited by aP vaccine results in high antibody levels, which do not necessarily correlate well with immunity (Olin et al. 2001). For example, although pertussis toxin (PT) antibodies are thought to be associated with protection (Cherry et al. 1998), PT antibody levels induced by aP are much higher than those induced by wP, even though protection is lower (Miller et al. 1991; Olin et al. 2001). Compounding the issue of a skewed Th2 immune response is waning immunity following aP vaccination. Despite an initially strong response, antibody levels fall several fold between aP doses (Guerra et al. 2009). Cell-mediated immunity, including memory T cells, persists in aP recipients, but more robust lymphocyte proliferation and cytokine responses are observed in those primed with wP compared to aP (Smits et al. 2013). The emerging story appears to be that antibodies have an important role in controlling infection and disease, but they may not be sufficient, requiring other immune functions for durable protection. This could explain the year-on-year decrease in aP vaccine effectiveness that has been shown in older children and teens (Cherry 2012b; Klein et al. 2012; Witt, Katz and Witt 2012; Koepke et al. 2014; McGirr and Fisman 2015). However, without clear humoral and cellular correlates of protection, the significance of antibody decay and cell-mediated immunity in contributing to reduced effectiveness of the aP vaccine is not clear.
TRANSMISSION IN aP VACCINATED INDIVIDUALS
Pertussis outbreaks are cyclical, mediated by the balance between susceptible and immune individuals. When there are ample immune individuals (through vaccination or natural infection) in the population, there is little pertussis activity. When a susceptible cohort accumulates in the population (for example by birth or waning immunity), an epidemic threshold is reached and an outbreak occurs (Fine and Clarkson 1987). Theoretically, the introduction of vaccination should lengthen the interval between epidemics, since vaccination should keep a critical mass of immune individuals in the population. However, this has not been universally observed for pertussis. Many years ago, Paul Fine asked why the interepidemic period for pertussis in England did not seem to be changed by vaccination programs, with outbreaks occurring every three to four years both before and after vaccination was introduced (Fine and Clarkson 1982). Close examination of outbreak patterns revealed that, although the interepidemic period remained the same, the amplitude of disease cycles increased following periods of low vaccine uptake (Fine and Clarkson 1982, 1987), an epidemiological signal that the vaccine may not be controlling infection and disease transmission, but rather clinical disease severity. Conversely, following years of high vaccine uptake, cycles still followed the same pattern but morbidity was reduced.
Capturing the precise epidemiology of pertussis is challenging, because case reporting and the resulting estimation of morbidity in the population is not always accurate, and varies depending on several factors. For example, case reporting varies by age group, with underreporting being common in older children and adults (McGirr, Tuite and Fisman 2013), as well as by severity, with milder cases less likely to be reported. In addition, underreporting is more likely to occur during interepidemic periods, while more comprehensive case reporting usually occurs during outbreaks.
A recent, more nuanced, perspective has considered whether the vaccine affects transmission and to what extent (Gay and Miller 2000; Domenech de Celles et al. 2014). Although transmission is often the focus for epidemiologists, experiments by microbiologists and immunologists typically address disease. Vaccine design requires attention on both fronts, and pertussis vaccine studies in the laboratory must take this into account. A program that focuses on individual-level protection is unlikely to ever have the success of one that works through a combined individual and herd approach that targets both protection from disease as well as transmission, such as that achieved for smallpox, polio, measles and rubella.
This perspective on the effect of immunization programs relates to the population biology of pathogens as well as host and to the centrality of herd immunity in explaining the success of immunization. It reveals a limitation of vaccine studies that are designed to test the protection conferred on the vaccinated individual but do not measure subclinical infections that can contribute to ongoing circulation. The positive examples of smallpox, polio, measles and rubella led to the logical conclusion that protecting an individual from disease necessarily resulted in an end to the chain of transmission. Pertussis demonstrates that this may not necessarily be true. Interrupting pathogen transmission is at the heart of herd immunity, since organisms only continue to survive if they continue to spread. Preventing onwards transmission may be achieved by vaccines that prevent or reduce disease symptoms (such as cough) that serve to transmit the pathogen. In the case of aP vaccines, the reduction of transmission in individuals recently vaccinated might be offset by the increased transmission that occurs because immunity wanes faster than immunity acquired through natural infection or wP vaccines. In order to conceptualize how we can intervene to prevent transmission, it is helpful to consider the series of challenges that organisms have had to overcome to avoid extinction in populations. Organisms seeking to survive must first develop mechanisms to infect their host, compete with resident bacterial flora and evade innate immunity to colonize and replicate using pathogen-specific virulence factors. To spread, organisms then induce symptoms that contribute to transmission (such as cough) to another susceptible person before the host's adaptive immunity catches up with them and without killing so many hosts that the host population (the pathogen's ecological niche) is disrupted. Pertussis remains remarkably successful despite extensive control efforts, underscoring the significance of understanding the mechanisms involved in its ability to both infect and transmit efficiently.
Prospects to elucidate the determinants of disease transmission have been improved by new animal models, which provide an experimental system in which the transmission process can be examined via a reductionist approach that can reveal the bacterial genes and host immune functions involved. Pertussis animal models have demonstrated that, although it is fairly effective at controlling disease, the aP vaccine is poor at controlling colonization, shedding and transmission. For example, the baboon model has recently been shown to mimic important aspects of symptoms and immune response to infection and, most excitingly, to demonstrate efficient transmission between animals over distance, presumably via aerosolized droplets believed to transmit B. pertussis between humans (Warfel, Zimmerman and Merkel 2014). The baboon model demonstrated that wP-vaccinated baboons rapidly cleared infection. Baboons vaccinated with aP vaccine were protected from severe diseases, but still colonized B. pertussis and transmitted it to other naïve baboons (Warfel, Zimmerman and Merkel 2014).
Unfortunately, various difficulties of baboon studies limit the number of experiments that can be performed. An experimental murine model has been developed using B. bronchiseptica, a closely related species to B. pertussis (Mattoo and Cherry 2005). These two bacteria share the ability to transmit between hosts via respiratory droplets, so the mechanisms involved are likely to be similar. In addition, since B. bronchiseptica shares four of the five antigens used in most aP vaccines (Pertacin, Fim 2 and 3, and filamentous hemagglutinin) (Smallridge et al. 2014), it is a suitable model in which to study pathogen–host interactions and vaccine effects (Goebel, Zhang and Harvill 2009). This model mimics important aspects of bacteria–immune interactions that affect transmission amongst baboons and humans. For example, when mice were vaccinated with wP vaccine, both shedding and transmission were inhibited, but the aP vaccine was unable to inhibit either shedding or transmission. These observations from biology align very well with what we observe epidemiologically (Nicholson et al. 2012). The efficiency of the murine model will allow for the analysis of many candidate bacterial genes that might be involved in the transmission process. Many tools of mouse molecular immunology will also allow aspects of the host immune response that might inhibit, or contribute to, transmission to be identified. Most exciting is the prospect for using the combination of these systems to efficiently identify bacterial and host factors in mice, confirm their roles in a recently developed aerosol transmission experimental system in pigs (Nicholson et al. 2012) and verify their relevance to B. pertussis transmission in baboons.
THE ROAD FORWARD
Until better understanding provides a pathway to improved vaccines, it would be healthy to debate whether the minority of the world that has experimented with aP vaccines should seriously be considering a return to wP, or the introduction of a mixed wP/aP vaccine schedule, since this may be the fastest avenue to better control of pertussis. From an ethics perspective, there is a question whether we are in a state of clinical equipoise, whether as a clinician counselling individual patients or parents or at a program level and if we know that wP vaccines are more effective. But this is a hard topic to debate. How would we balance the risk of local reactions against the risk of vaccine failure? If we pushed to use a more effective vaccine with a worse safety profile, what would be the implications for public confidence?
In response to the evident limitations of the aP vaccine, encouraging progress is being made with evaluating alternative aP pertussis vaccination schedules. A cocooning strategy—vaccination of post-partum women and other household members of infants—was previously recommended in some jurisdictions (Kretsinger et al. 2006; McIntyre and Wood 2009), since studies suggest that household members and particularly parents were the most common source of pertussis transmission to infants (Wendelboe et al. 2007). However, due to logistical hurdles and questions about cost and effectiveness, cocooning has not been widely implemented (Terranella et al. 2013; Lim, Deeks and Crowcroft 2014; Fernandez-Cano, Armadans Gil and Campins Marti 2015).
Neonatal immunization has also been explored, and although it resulted in adequate antibody titres, there was evidence of reduced titres for several pertussis antigens by the end of the primary vaccination series (Halasa et al. 2008). Additionally, there was some concern about interference with immune responses to other antigens/vaccines (Knuf et al. 2010; Wood et al. 2010; Amirthalingam et al. 2014), suggesting that, should this intervention be applied, a single antigen pertussis vaccine would need to be developed (Burns, Meade and Messionnier 2014). A new consideration in pertussis immunology is the growing evidence that antenatal immunization protects infants, whether through indirect protection because mothers do not develop pertussis, or through direct protection of infants via antibodies in cord blood (Amirthalingam et al. 2014). Immunization in pregnant women is currently recommended in several countries (Centers for Disease Control and Prevention 2013b; Olley 2014) due to transplacentral antibody transfer in utero (Gall, Myers and Pichichero 2011) and increased IgA in breastmilk (Halperin et al. 2011). This approach was shown to be highly effective when high coverage was achieved during a national outbreak in the UK (Amirthalingam et al. 2014). However, some concern remains about maternal antibodies potentially inhibiting pertussis antibody production in infants following vaccination (Amirthalingam 2013). In addition, in some jurisdictions maternal reimmunization has been suggested at every pregnancy due to waning immunity, regardless of the interval between pregnancies or the last adult booster (Healy, Rench and Baker 2013; Abu Raya et al. 2015; McGirr and Fisman 2015). Finally, many countries offer a single adult pertussis-containing booster vaccine, which could be extended to regular boosters throughout adult life, anchored to our need for regular tetanus immunization. It has been suggested that a decennial adult booster may be required to significantly reduce the burden of pertussis (Cherry 2010; McGirr and Fisman 2015), although some are skeptical (Fine 1997), studies have demonstrated that waning immunity results in reduced protection well before the 10-year mark (Healy, Rench and Baker 2013; Abu Raya et al. 2015). This approach would be potentially cost-effective (Lee, Riffelmann and Wirsing von Konig 2008), but may not be practicable. In the context of already suboptimal vaccination rates in many jurisdictions or in periods of low pertussis activity, the feasibility and acceptability of adult boosters and reimmunization in every pregnancy is unclear (Cherry 2010).
It should be recognized that, without clear correlates of protection, the long-term effectiveness of each of the above interventions is uncertain. Furthermore, each of the approaches outlined above can be conceptualized as either targeting high risk (neonates) or low coverage (adults) groups and therefore unlikely to have the impact on the population biology of pertussis that could maximize benefits to health. They are not ideal approaches. We also need a long-term perspective on what will happen when the generation of aP primed individuals become parents. How will this affect this cohort's future experience of pertussis as adults, and what are the implications of this for their own children?
CONCLUSIONS
Research directed at developing effective vaccines that uncovers the mechanisms whereby natural infection and wP vaccine may yield better duration of immunity and prevent transmission should be an immediate priority. Essential to this is the determination of correlates of protection for pertussis, including both humoral and cellular correlates. A simple model of disease that works so well for other toxin-mediated diseases such as tetanus and diphtheria is too simple for pertussis. But a complex model may still be amenable to control by a simple and effective intervention that might emerge from a better understanding of the biology at individual and population levels. The results of this work would help elucidate the way forward.
Options under discussion include altering the current aP vaccination schedule, using a live-attenuated vaccine (Fedele et al. 2011), modifying the aP vaccine with new antigens and/or adjuvants, developing a completely new vaccine or returning to the wP vaccine. In considering these strategies, a focus on prevention of transmission in addition to preventing disease may help in innovating responses (Burns, Meade and Messionnier 2014). Improving effectiveness at a program level can be achieved by administering more boosters, including in pregnancy, or by altering the vaccines to increase the duration of protection. The latter is not necessarily in the commercial best interests of industry (Burns, Meade and Messionnier 2014), and may require some public stimulus.
Developing a new direction for pertussis vaccines requires innovation at the interface between different disciplines. It takes time for applied problems to be translated into answerable scientific questions. Every three years an international multidisciplinary Bordetella Symposium brings together people who work on every aspect of pertussis from immunological studies in animals, through clinicians, to public health practitioners. This is a unique forum in which diverse groups can try to understand each other and share questions and ideas. At the 2004 meeting in Hinxton, Cambridge, UK, the question of studying how to interrupt the transmission of pertussis was raised. Nine years later, at the 2013 meeting in Dublin, Ireland, animal models in mice and baboons were presented that are starting to answer the question (8th International Symposium on Bordetella 2006; 9th International Symposium on Bordetella 2010). It may take time to discover how a better vaccine might be constructed, although science is not a linear process, and progress unpredictable. Developing a new and better vaccine may be expensive, but the costs of managing cases and outbreaks are also high. Outbreaks, disease and death are a burden and an expense borne by public health and the public, while vaccine development costs are borne by the academic sector and industry. A long-term perspective and intersectoral partnership approach combining the perspectives of all disciplines would benefit everyone.
FUNDING
E.T.H is the recipient of National Institutes of Health Grants GM083113, AI107016, AI116186, GM113681.
Conflict of interest. None declared.
REFERENCES
- 8th International Symposium on Bordetella. Paris: Institute Pasteur; 2006. [Google Scholar]
- 9th International Symposium on Bordetella. Baltimore, MA: University of Maryland; 2010. [Google Scholar]
- Abu Raya B, Srugo I, Kessel A, et al. The decline of pertussis-specific antibodies after tetanus, diphtheria, and acellular pertussis immunization in late pregnancy. J Infect Dis. 2015 doi: 10.1093/infdis/jiv324. pii: jiv324. [DOI] [PubMed] [Google Scholar]
- Amirthalingam G. Strategies to control pertussis in infants. Arch Dis Child. 2013;98:552–5. doi: 10.1136/archdischild-2012-302968. [DOI] [PubMed] [Google Scholar]
- Amirthalingam G, Andrews N, Campbell H, et al. Effectiveness of maternal pertussis vaccination in England: an observational study. Lancet. 2014;384:1521–8. doi: 10.1016/S0140-6736(14)60686-3. [DOI] [PubMed] [Google Scholar]
- Barton GM, Medzhitov R. Control of adaptive immune responses by Toll-like receptors. Curr Opin Immunol. 2002;14:380–3. doi: 10.1016/s0952-7915(02)00343-6. [DOI] [PubMed] [Google Scholar]
- Billingsley M. Pregnant women in UK are offered whooping cough vaccine to protect newborns. BMJ. 2012;345:e6594. doi: 10.1136/bmj.e6594. [DOI] [PubMed] [Google Scholar]
- Burns DL, Meade BD, Messionnier NE. Pertussis resurgence: perspectives from the Working Group Meeting on pertussis on the causes, possible paths forward, and gaps in our knowledge. J Infect Dis. 2014;209(Suppl 1):S32–5. doi: 10.1093/infdis/jit491. [DOI] [PubMed] [Google Scholar]
- Centers for Disease Control and Prevention. Pertussis (Whooping Cough): Surveillance and Reporting. Atlanta: Centers for Disease Prevention and Control; 2013a. http://www.cdc.gov/pertussis/surv-reporting.html (4 August 2015, date last accessed) [Google Scholar]
- Centers for Disease Control and Prevention. Updated recommendations for use of tetanus toxoid, reduced diphtheria toxoid, and acellular pertussis vaccine (Tdap) in pregnant women–Advisory Committee on Immunization Practices (ACIP), 2012. MMWR Morb Mortal Wkly Rep. 2013b;62:131–5. [PMC free article] [PubMed] [Google Scholar]
- Centres for Disease Control and Prevention. Pertussis epidemic - Washington 2012. MMWR-Morb Mort Wkly Rep. 2012;61:517–22. [PubMed] [Google Scholar]
- Cherry JD. The present and future control of pertussis. Clin Infect Dis. 2010;51:663–7. doi: 10.1086/655826. [DOI] [PubMed] [Google Scholar]
- Cherry JD. Epidemic pertussis in 2012–the resurgence of a vaccine-preventable disease. New Engl J Med. 2012a;367:785–7. doi: 10.1056/NEJMp1209051. [DOI] [PubMed] [Google Scholar]
- Cherry JD. Why do pertussis vaccines fail? Pediatrics. 2012b;129:968–70. doi: 10.1542/peds.2011-2594. [DOI] [PubMed] [Google Scholar]
- Cherry JD, Gornbein J, Heininger U, et al. A search for serologic correlates of immunity to Bordetella pertussis cough illnesses. Vaccine. 1998;16:1901–6. doi: 10.1016/s0264-410x(98)00226-6. [DOI] [PubMed] [Google Scholar]
- Domenech de Celles M, Riolo MA, Magpantay FM, et al. Epidemiological evidence for herd immunity induced by acellular pertussis vaccines. Proc Natl Acad Sci USA. 2014;111:E716–7. doi: 10.1073/pnas.1323795111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fedele G, Bianco M, Debrie AS, et al. Attenuated Bordetella pertussis vaccine candidate BPZE1 promotes human dendritic cell CCL21-induced migration and drives a Th1/Th17 response. J Immunol. 2011;186:5388–96. doi: 10.4049/jimmunol.1003765. [DOI] [PubMed] [Google Scholar]
- Fernandez-Cano MI, Armadans Gil L, Campins Marti M. Cost-benefit of the introduction of new strategies for vaccination against pertussis in Spain: cocooning and pregnant vaccination strategies. Vaccine. 2015;33:2213–20. doi: 10.1016/j.vaccine.2015.03.045. [DOI] [PubMed] [Google Scholar]
- Fine PE. Adult pertussis: a salesman's dream–and an epidemiologist's nightmare. Biologicals. 1997;25:195–8. doi: 10.1006/biol.1997.0083. [DOI] [PubMed] [Google Scholar]
- Fine PE, Clarkson JA. The recurrence of whooping cough: possible implications for assessment of vaccine efficacy. Lancet. 1982;1:666–9. doi: 10.1016/s0140-6736(82)92214-0. [DOI] [PubMed] [Google Scholar]
- Fine PE, Clarkson JA. Reflections on the efficacy of pertussis vaccines. Rev Infect Dis. 1987;9:866–83. doi: 10.1093/clinids/9.5.866. [DOI] [PubMed] [Google Scholar]
- Gall SA, Myers J, Pichichero M. Maternal immunization with tetanus-diphtheria-pertussis vaccine: effect on maternal and neonatal serum antibody levels. Am J Obstet Gynecol. 2011;204:334.e1–5. doi: 10.1016/j.ajog.2010.11.024. [DOI] [PubMed] [Google Scholar]
- Gay NJ, Miller E. Pertussis transmission in England and Wales. Lancet. 2000;355:1553–4. doi: 10.1016/S0140-6736(05)74603-1. [DOI] [PubMed] [Google Scholar]
- Goebel EM, Zhang X, Harvill ET. Bordetella pertussis infection or vaccination substantially protects mice against B. bronchiseptica infection. PLoS One. 2009;4:e6778. doi: 10.1371/journal.pone.0006778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guerra FA, Blatter MM, Greenberg DP, et al. Safety and immunogenicity of a pentavalent vaccine compared with separate administration of licensed equivalent vaccines in US infants and toddlers and persistence of antibodies before a preschool booster dose: a randomized, clinical trial. Pediatrics. 2009;123:301–12. doi: 10.1542/peds.2007-3317. [DOI] [PubMed] [Google Scholar]
- Guiso N. Bordetella pertussis: why is it still circulating? J Infect. 2014;68:S119–24. doi: 10.1016/j.jinf.2013.09.022. [DOI] [PubMed] [Google Scholar]
- Guris D, Strebel PM, Bardenheier B, et al. Changing epidemiology of pertussis in the United States: increasing reported incidence among adolescents and adults, 1990–1996. Clin Infect Dis. 1999;28:1230–7. doi: 10.1086/514776. [DOI] [PubMed] [Google Scholar]
- Halasa NB, O'Shea A, Shi JR, et al. Poor immune responses to a birth dose of diphtheria, tetanus, and acellular pertussis vaccine. J Pediatr. 2008;153:327–32. doi: 10.1016/j.jpeds.2008.03.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Halperin BA, Morris A, Mackinnon-Cameron D, et al. Kinetics of the antibody response to tetanus-diphtheria-acellular pertussis vaccine in women of childbearing age and postpartum women. Clin Infect Dis. 2011;53:885–92. doi: 10.1093/cid/cir538. [DOI] [PubMed] [Google Scholar]
- Health Protection Agency. Health Protection Report. 2013. Confirmed pertussis (England and Wales), data to end-March 2013; p. 7. http://webarchive.nationalarchives.gov.uk/20140714084352/ http://www.hpa.org.uk/hpr/archives/2013/news1913.htm (4 August 2015, date last accessed) [Google Scholar]
- Healy CM, Rench MA, Baker CJ. Importance of timing of maternal combined tetanus, diphtheria, and acellular pertussis (Tdap) immunization and protection of young infants. Clin Infect Dis. 2013;56:539–44. doi: 10.1093/cid/cis923. [DOI] [PubMed] [Google Scholar]
- Higgs R, Higgins SC, Ross PJ, et al. Immunity to the respiratory pathogen Bordetella pertussis. Mucosal Immunol. 2012;5:485–500. doi: 10.1038/mi.2012.54. [DOI] [PubMed] [Google Scholar]
- Klein NP, Bartlett J, Rowhani-Rahbar A, et al. Waning protection after fifth dose of acellular pertussis vaccine in children. New Engl J Med. 2012;367:1012–9. doi: 10.1056/NEJMoa1200850. [DOI] [PubMed] [Google Scholar]
- Knuf M, Schmitt HJ, Jacquet JM, et al. Booster vaccination after neonatal priming with acellular pertussis vaccine. J Pediatr. 2010;156:675–8. doi: 10.1016/j.jpeds.2009.12.019. [DOI] [PubMed] [Google Scholar]
- Koepke R, Eickhoff JC, Ayele RA, et al. Estimating the effectiveness of tetanus-diphtheria-acellular pertussis vaccine (Tdap) for preventing pertussis: evidence of rapidly waning immunity and difference in effectiveness by Tdap brand. J Infect Dis. 2014;210:942–53. doi: 10.1093/infdis/jiu322. [DOI] [PubMed] [Google Scholar]
- Kretsinger K, Broder KR, Cortese MM, et al. Preventing tetanus, diphtheria, and pertussis among adults: use of tetanus toxoid, reduced diphtheria toxoid and acellular pertussis vaccine recommendations of the Advisory Committee on Immunization Practices (ACIP) and recommendation of ACIP, supported by the Healthcare Infection Control Practices Advisory Committee (HICPAC), for use of Tdap among health-care personnel. MMWR Recomm Rep. 2006;55:1–37. [PubMed] [Google Scholar]
- Lee GM, Riffelmann M, Wirsing von Konig CH. Cost-effectiveness of adult pertussis vaccination in Germany. Vaccine. 2008;26:3673–9. doi: 10.1016/j.vaccine.2008.04.068. [DOI] [PubMed] [Google Scholar]
- Liko J, Robison SG, Cieslak PR. Priming with whole-cell versus acellular pertussis vaccine. New Engl J Med. 2013;368:581–2. doi: 10.1056/NEJMc1212006. [DOI] [PubMed] [Google Scholar]
- Lim GH, Deeks SL, Crowcroft NS. A cocoon immunisation strategy against pertussis for infants: does it make sense for Ontario? Euro Surveill. 2014;19:20688. doi: 10.2807/1560-7917.es2014.19.5.20688. [DOI] [PubMed] [Google Scholar]
- McGirr A, Fisman DN. Duration of pertussis immunity after DTaP immunization: a meta-analysis. Pediatrics. 2015;135:331–43. doi: 10.1542/peds.2014-1729. [DOI] [PubMed] [Google Scholar]
- McGirr AA, Tuite AR, Fisman DN. Estimation of the underlying burden of pertussis in adolescents and adults in Southern Ontario, Canada. PLoS One. 2013;8:e83850. doi: 10.1371/journal.pone.0083850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McIntyre P, Wood N. Pertussis in early infancy: disease burden and preventive strategies. Curr Opin Infect Dis. 2009;22:215–23. doi: 10.1097/QCO.0b013e32832b3540. [DOI] [PubMed] [Google Scholar]
- Mascart F, Hainaut M, Peltier A, et al. Modulation of the infant immune responses by the first pertussis vaccine administrations. Vaccine. 2007;25:391–8. doi: 10.1016/j.vaccine.2006.06.046. [DOI] [PubMed] [Google Scholar]
- Mattoo S, Cherry JD. Molecular pathogenesis, epidemiology, and clinical manifestations of respiratory infections due to Bordetella pertussis and other Bordetella subspecies. Clin Microbiol Rev. 2005;18:326–82. doi: 10.1128/CMR.18.2.326-382.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller E, Ashworth LA, Robinson A, et al. Phase II trial of whole-cell pertussis vaccine vs an acellular vaccine containing agglutinogens. Lancet. 1991;337:70–3. doi: 10.1016/0140-6736(91)90735-8. [DOI] [PubMed] [Google Scholar]
- Misegades LK, Winter K, Harriman K, et al. Association of childhood pertussis with receipt of 5 doses of pertussis vaccine by time since last vaccine dose, California, 2010. JAMA. 2012;308:2126–32. doi: 10.1001/jama.2012.14939. [DOI] [PubMed] [Google Scholar]
- Mooi FR, van Loo IH, van Gent M, et al. Bordetella pertussis strains with increased toxin production associated with pertussis resurgence. Emerg Infect Dis. 2009;15:1206–13. doi: 10.3201/eid1508.081511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nicholson TL, Brockmeier SL, Loving CL, et al. Phenotypic modulation of the virulent Bvg phase is not required for pathogenesis and transmission of Bordetella bronchiseptica in swine. Infect Immun. 2012;80:1025–36. doi: 10.1128/IAI.06016-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olin P, Hallander HO, Gustafsson L, et al. How to make sense of pertussis immunogenicity data. Clin Infect Dis. 2001;33(Suppl 4):S288–91. doi: 10.1086/322564. [DOI] [PubMed] [Google Scholar]
- Olley M. Vaccination Against Pertussis (Whooping Cough) for Pregnant Women 2014: Information for Healthcare Professionals. London: Public Health England; 2014. https://www.gov.uk/government/uploads/system/uploads/attachment_data/file/338567/PHE_pertussis_in_pregnancy_information_for_HP_2014_doc_V3.pdf (4 August 2015, date last accessed) [Google Scholar]
- Plotkin SA. The pertussis problem. Clin Infect Dis. 2014;58:830–3. doi: 10.1093/cid/cit934. [DOI] [PubMed] [Google Scholar]
- Podda A, Bona G, Canciani G, et al. Effect of priming with diphtheria and tetanus toxoids combined with whole-cell pertussis vaccine or with acellular pertussis vaccine on the safety and immunogenicity of a booster dose of an acellular pertussis vaccine containing a genetically inactivated pertussis toxin in fifteen- to twenty-one-month-old children. Italian Multicenter Group for the Study of Recombinant Acellular Pertussis Vaccine. J Pediatr. 1995;127:238–43. doi: 10.1016/s0022-3476(95)70301-2. [DOI] [PubMed] [Google Scholar]
- Ross PJ, Sutton CE, Higgins S, et al. Relative contribution of Th1 and Th17 cells in adaptive immunity to Bordetella pertussis: towards the rational design of an improved acellular pertussis vaccine. PLoS Pathog. 2013;9:e1003264. doi: 10.1371/journal.ppat.1003264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ryan M, Murphy G, Ryan E, et al. Distinct T-cell subtypes induced with whole cell and acellular pertussis vaccines in children. Immunology. 1998;93:1–10. doi: 10.1046/j.1365-2567.1998.00401.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sheridan SL, Ware RS, Grimwood K, et al. Number and order of whole cell pertussis vaccines in infancy and disease protection. JAMA. 2012;308:454–6. doi: 10.1001/jama.2012.6364. [DOI] [PubMed] [Google Scholar]
- Skowronski DM, De Serres G, MacDonald D, et al. The changing age and seasonal profile of pertussis in Canada. J Infect Dis. 2002;185:1448–53. doi: 10.1086/340280. [DOI] [PubMed] [Google Scholar]
- Smallridge WE, Rolin OY, Jacobs NT, et al. Different effects of whole-cell and acellular vaccines on Bordetella transmission. J Infect Dis. 2014;209:1981–8. doi: 10.1093/infdis/jiu030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smits K, Pottier G, Smet J, et al. Different T cell memory in preadolescents after whole-cell or acellular pertussis vaccination. Vaccine. 2013;32:111–8. doi: 10.1016/j.vaccine.2013.10.056. [DOI] [PubMed] [Google Scholar]
- Tanaka M, Vitek CR, Pascual FB, et al. Trends in pertussis among infants in the United States, 1980–1999. JAMA. 2003;290:2968–75. doi: 10.1001/jama.290.22.2968. [DOI] [PubMed] [Google Scholar]
- Terranella A, Asay GR, Messonnier ML, et al. Pregnancy dose Tdap and postpartum cocooning to prevent infant pertussis: a decision analysis. Pediatrics. 2013;131:e1748–56. doi: 10.1542/peds.2012-3144. [DOI] [PubMed] [Google Scholar]
- Vickers D, Ross AG, Mainar-Jaime RC, et al. Whole-cell and acellular pertussis vaccination programs and rates of pertussis among infants and young children. CMAJ. 2006;175:1213–7. doi: 10.1503/cmaj.051637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Warfel JM, Zimmerman LI, Merkel TJ. Acellular pertussis vaccines protect against disease but fail to prevent infection and transmission in a nonhuman primate model. P Natl Acad Sci USA. 2014;111:787–92. doi: 10.1073/pnas.1314688110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wendelboe AM, Njamkepo E, Bourillon A, et al. Transmission of Bordetella pertussis to young infants. Pediatr Infect Dis J. 2007;26:293–9. doi: 10.1097/01.inf.0000258699.64164.6d. [DOI] [PubMed] [Google Scholar]
- Winter K, Harriman K, Zipprich J, et al. California pertussis epidemic, 2010. J Pediatr. 2012;161:1091–6. doi: 10.1016/j.jpeds.2012.05.041. [DOI] [PubMed] [Google Scholar]
- Witt MA, Katz PH, Witt DJ. Unexpectedly limited durability of immunity following acellular pertussis vaccination in preadolescents in a North American outbreak. Clin Infect Dis. 2012;54:1730–5. doi: 10.1093/cid/cis287. [DOI] [PubMed] [Google Scholar]
- Wood N, McIntyre P, Marshall H, et al. Acellular pertussis vaccine at birth and one month induces antibody responses by two months of age. Pediatr Infect Dis J. 2010;29:209–15. doi: 10.1097/INF.0b013e3181bc98d5. [DOI] [PubMed] [Google Scholar]
- World Health Organization (WHO) Pertussis vaccines: WHO position paper. Wkly Epidemiol Rec. 2010;85:385–400. [PubMed] [Google Scholar]
- Zhang L, Prietsch SO, Axelsson I, et al. Acellular vaccines for preventing whooping cough in children. Cochrane Db Syst Rev. 2014;9:CD001478. doi: 10.1002/14651858.CD001478.pub6. [DOI] [PMC free article] [PubMed] [Google Scholar]


