Abstract
Human γ-herpesviruses contain Epstein Barr virus (EBV), the first human tumor virus that was identified in man, and Kaposi Sarcoma associated herpesvirus (KSHV), one of the most recently identified human oncogenic pathogens. Both of these have co-evolved with humans to cause tumors only in a minority of infected individuals, despite their exquisite ability to establish persistent infections. In this review we will summarize the fine-tuned balance between immune responses, immune escape and cellular transformation by these viruses, which results in lifelong persistent, but asymptomatic infection with immune control in most virus carriers. A detailed understanding of this balance is required to immunotherapeutically reinstall it in patients that suffer from EBV and KSHV associated malignancies.
1. Innate immune control of EBV
Epstein Barr virus (EBV) is the prototypic oncogenic γ-herpesvirus. Despite its discovery in the most frequent Subsaharan childhood tumor, Burkitt’s lymphoma, 51 years ago [1], it is carried by more than 90% of the human adult population as an asymptomatic persistent infection [2]. EBV infection and tumorigenesis is kept under control by cell-mediated immunity, which targets both the transforming latent and the virus producing lytic program of the virus. This comprehensive immune control of EBV is established by successive waves of innate and adaptive immune responses.
Immune detection of the large double-stranded DNA virus EBV occurs via plasmacytoid dendritic cells (pDCs). These antigen presenting cells (APCs) readily secrete type I interferon (IFN-α/β) upon detection of unmethylated viral DNA via the toll like receptor 9 (TLR9) [3–5]. IFN-α/β restricts EBV infection of human B cells during the first 24h after inoculation [6] and might be even more important for natural killer cell activation during EBV infection [4,7,8]. Even so pDCs are the primary APCs to detect EBV, their ability to prime EBV specific T cells remains unclear. In contrast, inflammatory monocyte-derived DCs are able to cross-present EBV antigen from infected B cells [9] and conventional DCs (cDCs), especially the minor CD141+ cDC subset, recognize EBV encoded RNAs (EBERs) via TLR3 after their release from latently EBV infected B cells [10]. Therefore, both pDCs and cDCs get activated during lytic and latent EBV infection, respectively, to restrict infection initially and activate innate and adaptive lymphocytes.
Among the innate lymphocytes NK, NKT and γδ T cells might play a role in innate immune control of EBV [7,11–16]. In particular during symptomatic primary EBV infection, called infectious mononucleosis, NK cells have been found to expand [17,18]. Especially an early differentiated NK cell population accumulates and stays elevated up to six months in peripheral blood of infectious mononucleosis patients [13]. This NK cell subset is maintained at higher frequency in tonsils of EBV positive individuals [19]. These early differentiated NK cells preferentially recognize lytic EBV replicating cells, and NK cells control lytic EBV infection in mice with reconstituted human immune system components (HIS mice) [12,13]. In the absence of this NK cell-mediated immune control lytic EBV replication drives CD8+ T cell lymphocytosis, causing infectious mononucleosis symptoms [12]. Interestingly, infectious mononucleosis affects more frequently adolescents that acquire EBV later in life [20] and the frequency of early differentiated NK cells declines during the first decade after birth [1]. In addition to this recognition of lytic EBV infection by innate lymphocytes, NKT and Vγ9Vδ2 T cells can target latently infected EBV transformed B cells (LCLs) [14,16]. EBV infection activates CD1d restricted invariant NKT cells [7,21]. These are able to kill LCLs and prevent EBV associated lymphomagenesis in HIS mice [14,15]. Furthermore, activated Vγ9Vδ2 T cells also limit LCL growth in vitro and in vivo via recognition by their TCR and the NKG2D receptor and via TRAIL and Fas dependent killing [16]. This limits lymphoproliferative disease after LCL transfer in vivo [22], but it remains unclear if and how Vγ9Vδ2 T cells get activated during EBV infection. Thus, innate lymphocyte compartments control both lytic and latent EBV infection with NK and NKT as well as γδ T cells, respectively.
2. Adaptive immune control of EBV
Comprehensive immune control of latent and lytic EBV infection is long-term maintained by T cell responses, while B cell responses are used for diagnostic purposes, but can be absent in healthy virus carriers [2,23]. Both latent and lytic EBV antigens are recognized by CD4+ helper and CD8+ cytotoxic T cells [2]. However, distinct hierarchies for antigen specificity of these T cells exist with CD4+ T cells most consistently recognizing EBV nuclear antigen 1 (EBNA1) and late lytic antigens, while CD8+ T cells mainly recognize the EBNA3 proteins and immediate early as well as early lytic EBV antigens [24–26]. Both CD4+ and CD8+ T cells contribute to EBV specific immune control and primarily CD8+ T cells prevent EBV induced lymphomas in HIS mice [12,27]. Furthermore, adoptive transfer of EBV specific T cell lines is clinically used to treat some EBV associated lymphomas, primarily post-transplant lymphoproliferative disease (PTLD) [25]. In these cell lines EBV antigen specificities that mediate protection are mostly ill-defined and it is assumed that T cells against transforming latent EBV antigens primarily mediate anti-tumor effects after adoptive transfer. Interestingly, recent studies have suggested that some EBV associated malignancies might benefit from lytic EBV infection [28,29] and that lytic EBV antigen specific CD4+ and CD8+ T cells might be able to contribute to EBV associated lymphoma suppression [29,30]. In good agreement, plasma EBV viral loads, which might in part result from virus shedding, correlate with the occurrence and progression of some EBV associated malignancies, including PTLD, Hodgkin’s lymphoma and nasopharyngeal carcinoma [31–33]. Thus a comprehensive T cell mediated immune control of both latent and lytic EBV infection might not only be required to establish low viral loads in healthy EBV carriers, but also to prevent EBV associated malignancies.
In order to persist, EBV employs a variety of immune escape mechanisms against these comprehensive T cell responses, and the main strategies differ between latent and lytic EBV infection [34] (Figure 1). During latent infection EBV protein down-regulation is the main strategy. For example, EBNA1 translation is limited by its RNA structure and, therefore, barely enough T cell epitopes can be presented on MHC class I molecules for CD8+ T cell recognition [35,36]. Furthermore, EBV persists in memory B cells, in which all EBV protein expression has been down-regulated [37]. In contrast during lytic replication, when the virus needs to express a large number of proteins to build its infectious particles, it employs active immune evasion. The lytic EBV proteins BARF1 and BPLF1 block differentiation and activation of inflammatory DCs for T cell priming [38,39]. BNLF2a and BILF1, which target antigen presentation to CD8+ T cells by limiting peptide supply for MHC class I loading and internalization of the loaded complexes, compromise early and late EBV lytic antigen recognition, respectively [40]. Interestingly, the host-shutoff protein BGLF5 seemed to only minimally compromise this antigen presentation [40]. Finally, BZLF2 (gp42) and BCRF1 (vIL-10) inhibit CD4+ T cell recognition of MHC class II complexes and their priming towards anti-viral Th1 cells as well as their effector functions [41,42]. Thus, EBV limits antigen expression and actively inhibits immune responses for immune evasion during latent and lytic EBV infection, respectively.
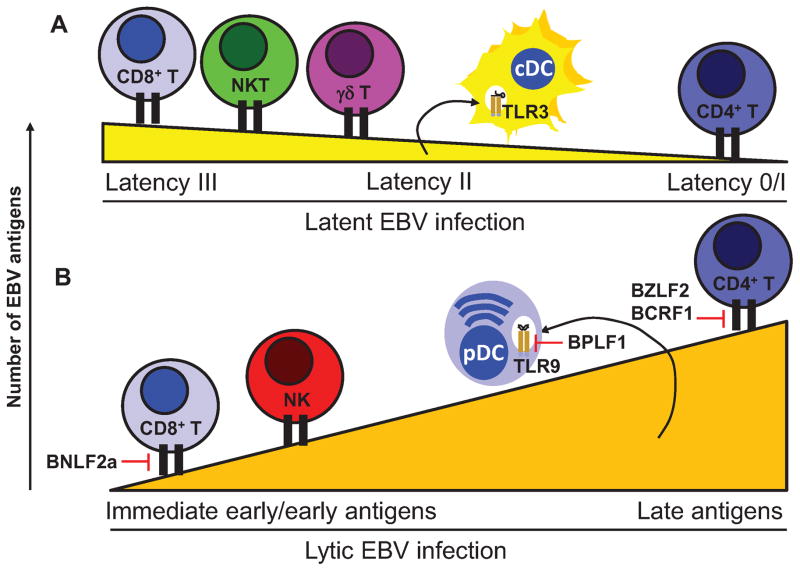
Figure 1. Comprehensive cell-mediate immune control of Epstein Barr virus infection.
A) During latent infection viral antigen expression is down-regulated with successive B cell differentiation from 8 (latency III) to 3 (latency II) and 1 (latency I) or none (latency 0). While CD8+ T cells strongly recognize the latency III antigens EBNA3s, CD4+ T cells consistently recognize the latency I antigen EBNA1. NKT and γδ T cells also target latent EBV infection. EBV encoded RNAs (EBERs) are released from latently infected cells and stimulate conventional DCs (cDCs) via TLR3. B) During lytic infection successively more EBV gene products are expressed, starting from 2 immediate early antigens to more than 80 proteins. Immediate early and early lytic EBV antigens are preferentially recognized by CD8+ T cells, while late lytic EBV antigens are mainly targeted by CD4+ T cells. NK cells also recognize lytically EBV replicating cells and unmethylated EBV DNA of virus particles is sensed by plasmacytoid DCs (pDCs) via TLR9. BNLF2a blocks MHC class I restricted early lytic EBV antigen presentation to CD8+ T cells and BZLF2 as well as BCRF1 MHC class II restricted late EBV lytic antigen presentation to CD4+ T cells as well as their activity, respectively. BPLF1 is transferred with EBV particles to pDCs and blocks TLR activation.
3. Innate immune control of KSHV
Kaposi’s sarcoma-associated herpesvirus (KSHV), also known as human herpesvirus 8 (HHV-8), is the most recently identified human herpesvirus [43] and its namesake, Kaposi’s sarcoma, is the most common cancer in untreated AIDS patients [44]. KSHV is also associated with two rare lymphoproliferative disorders: primary effusion lymphoma (PEL) and multicentric Castleman’s disease (MCD) [45]. Like other herpesviruses, KSHV establishes lifelong infections in spite of host immune defenses. Both innate and adaptive immunity coordinate the control of KSHV in infected individuals [46–49]. On the other hand, inflammatory cytokines and immune cell infiltrates play a crucial role in the early stages of KS development [50,51]. Moreover, KSHV paradoxical immune reconstitution inflammatory syndrome, a rare but life-threatening complication of KS patients, is precipitated by immune reconstitution following the initiation of HAART [52]. Thus, the interplay between KSHV and host immune system plays an important role of KSHV-associated diseases.
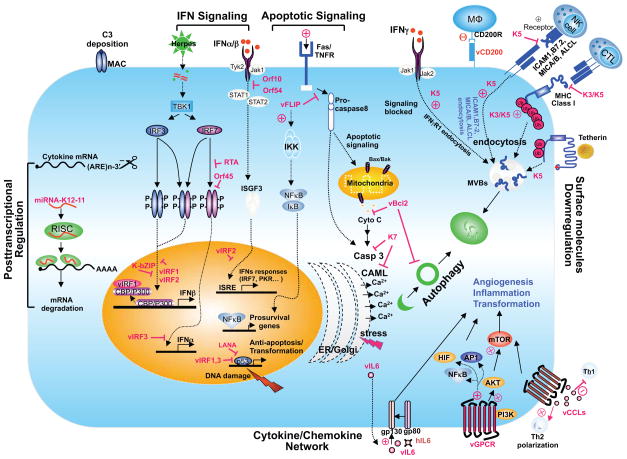
The main target cells of KSHV are B cells and endothelial cells but monocytes and dendritic cells are also susceptible to infection. The engagement of pattern recognition receptors (PRRs) in turn leads to signaling cascades, leading to the expression of IFNα/β and pro-inflammatory cytokines. Upon de novo infection of primary endothelial cells, KSHV genomic DNA is evident within IFNγ-inducible 16 (IFI16) DNA sensor-containing nuclear bodies and this phenotype is accompanied by IFI16-dependent inflammasome formation and caspase-1-mediated secretion of IL-1β [53], which may be particularly relevant to elevated levels of IL-1β and other cytokines in KS lesion [50,54]. RIG-I, a cytosolic RNA sensor, contributes to IFNβ production and suppression viral gene expression upon KSHV infection [8,55], which is, on the other hand, reduced by the ORF64 deubiquitinase activity-containing tegument protein [8]. The KSHV virion delivers the tegument protein, ORF45, which prevents the phosphorylation of IRF7 [56–61], and the RTA induces the proteasomal degradation of IRF3 and IRF7 by recruiting the RTA-associated ubiquitin E3 ligase [62,63]. KSHV encodes an orthologue of miR-155 designated miR-K12-11, which targets IKKε mRNA for degradation, resulting in the reduced IKKε-mediated IRF3 and IRF7 phosphorylation [64–66]. One of the particular features of the KSHV genome is the presence of four homologues of the IRF family of proteins. Interestingly, three of these gene products (vIRF1, 2 and 3 but not 4) are known to block transcription of type I IFN genes or ISGs (reviewed in [67–69]). Finally, several KSHV genes including vIL6 and dUTPase-related proteins (ORF10, ORF11 and ORF54), have been shown to block the second phase of the IFN response by targeting the IFNAR1-JAK-STAT-ISGF3 pathway [70,71]. Thus, the fact that KSHV devotes much of its coding potential to the inhibition of the IFN response (Figure 2) underscores the importance of this pathway in the control of its infections. In contrast, EBV does not seem to dedicate such a substantial effort to actively interfering with the type I IFN response, possibly due to the fact that latent EBV infection with its heavily methylated DNA [72] allows persistence with limited activation of the respective IFN pathways.
Figure 2. Overview of KSHV-mediated immune evasion.
The KSHV proteins are marked with the red color. Detailed mechanisms are described in the text.
Programmed cell death is one of the major innate defense mechanisms against viral infection and can be grouped into apoptosis, autophagic cell death, pyroptosis and necroptosis [73]. KSHV encodes several anti-apoptosis factors, including vBcl-2 [74], vFLIP [75], vIAP [74], K1 [76,77], vIRF4 [78], LANA [79] and miR-K10a [80]. Together, these viral factors target both the extrinsic and intrinsic apoptosis pathways. The sequestration of damaged organelles, protein aggregates or invading pathogens is orchestrated by a homeostatic process called autophagy. KSHV encodes three autophagy inhibitors (vBcl-2, K7 and vFLIP) that are each able to block distinct steps of autophagy. In contrast, EBV seems to benefit from autophagosomal membranes for its enveloping and efficient infectious particle production during lytic replication [81,82]. In summary, the identification and characterization of KSHV innate immune evasion genes has significantly advanced our understanding of viral persistent infection.
4. Cell-mediated immune control of KSHV
A hallmark of KS histology is abundant inflammatory infiltrates, including B cells, T cells, and monocytes. Thus, leukocyte trafficking and effector-target cell interactions are not only crucial for the innate and adaptive immune response to KSHV, but may also play a role in KS development. Several studies have examined Natural Killer (NK) cell-mediated control of KSHV infection and pathogenesis. AIDS-KS patients with ongoing symptoms have reduced NK cell-mediated immunity compared to patients with indolent classic KS or normal blood donors [46,47]. Active KS is also associated with higher serum levels of cytokines and other secreted factors that dampens NK cell cytotoxicity [47]. While KSHV-infected cells can be predisposed to NK cell recognition, the virus is genetically equipped to circumvent this threat. Two KSHV-encoded, membrane-bound E3 ligases called K3 and K5 selectively remove proteins from the cell surface by triggering ubiquitin-dependent endocytosis [83–86]. K5 expression can reduce the surface expression of activating ligands such as MICA, MICB and AICL; and costimulatory molecules such as ICAM-1 and B7-2 [21,87,88]. As a result, NK cell-mediated cytotoxicity is significantly diminished in K5-expressing target cells [21]. In addition, ORF54 can induce downregulation of NKp44L [89], while miR-K12-7 targets MICB mRNA for degradation [90]. The role of inhibitory ligands is equally important: K5 induces selective downregulation of HLA-A and HLA-B, whereas K3 broadly targets HLA-A, B, C, and E [84]. Delayed expression of K3 may be important for maintaining the expression of inhibitory ligands such as HLA-C and HLA-E during the early stages of lytic replication. Thus, K3, K5, ORF54, miR-K12-7 and other viral factors may function in concert to subvert NK cell surveillance. In contrast, lytic EBV replication renders affected B cells susceptible for NK cell recognition [12,13]. As for the type I IFN response EBV latent infection, which does not seem to render B cells susceptible for NK cell recognition, allows viral persistence without actively compromising NK cell recognition.
Interestingly, the KSHV genome encodes pirated genes with putative roles in modulating leukocyte trafficking: CC chemokines (vMIP-I, vMIP-II, and vMIP-III), a cytokine (vIL-6) and MARCH family members (K3 and K5). Apart from their role in evading cell-mediated immunity, K3 and K5 also downregulate adhesion molecules such as PECAM-1, ICAM-1, ALCAM, VCAM and VE-cadherin [21,91–93], all of which are involved in transendothelial migration [94]. Another modulator of leukocyte trafficking is viral macrophage inflammatory protein II (vMIP-II). vMIP-II antagonizes CCR3 and CCR5 and blocks RANTES-induced chemotaxis of monocytes [95] and also binds to CX3CR1 and CCR5 and occludes the binding of fractalkine and RANTES, respectively, resulting in impaired NK cell chemotaxis [96]. KSHV also has several potential ways of obstructing neutrophil recruitment: for example, vIL-6 is essential for preventing recruitment of neutrophils in KSHV infected endothelial cells [97]. vCD200 is able to block neutrophil recruitment in mice treated with carrageenan, a compound used to induce neutrophil migration [98]. Together, these viral proteins may be part of strategy to divert harmful leukocytes and facilitate viral dissemination through a skewed inflammatory response. EBV seems to also compromise granulocyte maintenance and thereby similar to KSHV prevent inflammatory infiltrates by expression of the inhibitor of Colony Stimulating Factor-1 signaling BARF1 [38].
Although studies have identified KSHV-specific T cell responses largely from HIV-infected KS patients, knowledge of T cell epitopes and their value as targets for the control of KSHV pathogenesis is very limited. The highly active antiretroviral therapy can resolve KS by enabling anti-KSHV immune reconstitution, including NK cell restoration, and by rapidly affecting KSHV replication. However, Guihot et al. [99] has revealed that individuals with KS had less frequent KSHV-specific T cell responses than asymptomatic subjects, regardless of their HIV status, CD4 count or KSHV load. Furthermore, not all KSHV-infected subjects develop associated diseases, suggesting that disease development is therefore likely dependent upon host factors, occurring through immune surveillance failure. Several studies have suggested certain HLA alleles might be associated with increased KSHV lytic replication and/or pathogenesis, but definitive associations have yet to be determined.
5. Conclusions
Herpesviruses are among the most ubiquitous and successful viruses known, and are thought to have co-evolved with their hosts during speciation. To achieve this, γ-herpesviruses devote multiple strategies and resources to manipulate key signaling pathways, ultimately promoting the survival of virus infected cell, immune evasion and tumorigenesis. However, EBV and KSHV subvert different checkpoints for survival of infected cells and their escape mechanisms from immune control are more similar during lytic than during latent infection.γ-herpesvirus-associated malignancies express varying subsets of virally-encoded antigens that potentially render them susceptible to virus-specific immunological responses. However, their effective immune evasion mechanisms present a considerable problem to immunotherapy. Thus, the ongoing study of immune modulatory activities will provide deeper understanding of the interactions between γ-herpesvirus and the immune system, and offer opportunities to overcome the obstacle for successful immunotherapy.
Highlights.
Innate lymphocyte responses determine the outcome of primary EBV infection.
Different EBV immune evasins compromise early and late lytic antigen recognition by T cells.
KSHV compromises interferon responses with several immune evasins.
Different forms of cell death are targeted by KSHV gene products.
Receptor down-regulation by KSHV blocks both innate and adaptive lymphocytes.
Acknowledgments
This work was partly supported by CA82057, CA31363, CA115284, CA180779, DE023926, HL110609, AI073099, AI116585, Hastings Foundation, and Fletcher Jones Foundation (JUJ). Moreover, CM receives research support from Cancer Research Switzerland (KFS-3234-08-2013), Worldwide Cancer Research (14-1033), the clinical research priority programs KFSPMS and KFSPHHLD of the University of Zurich, the Sobek Foundation, Fondation Acteria, the Swiss Vaccine Research Institute and the SNF (310030_143979 and CRSII3_136241)
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
• of special interest
•• of outstanding interest
- 1.Epstein MA, Hummeler K, Berkaloff A. The Entry and Distribution of Herpes Virus and Colloidal Gold in Hela Cells after Contact in Suspension. J Exp Med. 1964;119:291–302. doi: 10.1084/jem.119.2.291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Rickinson AB, Long HM, Palendira U, Münz C, Hislop AD. Cellular immune controls over Epstein-Barr virus infection: new lessons from the clinic and the laboratory. Trends Immunol. 2014;35(4):159–169. doi: 10.1016/j.it.2014.01.003. [DOI] [PubMed] [Google Scholar]
- 3.Fiola S, Gosselin D, Takada K, Gosselin J. TLR9 contributes to the recognition of EBV by primary monocytes and plasmacytoid dendritic cells. J Immunol. 2010;185(6):3620–3631. doi: 10.4049/jimmunol.0903736. [DOI] [PubMed] [Google Scholar]
- 4.Lim WH, Kireta S, Russ GR, Coates PT. Human plasmacytoid dendritic cells regulate immune responses to Epstein-Barr virus (EBV) infection and delay EBV-related mortality in humanized NOD-SCID mice. Blood. 2007;109(3):1043–1050. doi: 10.1182/blood-2005-12-024802. [DOI] [PubMed] [Google Scholar]
- 5.Severa M, Giacomini E, Gafa V, Anastasiadou E, Rizzo F, Corazzari M, Romagnoli A, Trivedi P, Fimia GM, Coccia EM. EBV stimulates TLR- and autophagy-dependent pathways and impairs maturation in plasmacytoid dendritic cells: implications for viral immune escape. Eur J Immunol. 2013;43(1):147–158. doi: 10.1002/eji.201242552. [DOI] [PubMed] [Google Scholar]
- 6.Lotz M, Tsoukas CD, Fong S, Carson DA, Vaughan JH. Regulation of Epstein-Barr virus infection by recombinant interferons. Selected sensitivity to interferon-gamma. Eur J Immunol. 1985;15(5):520–525. doi: 10.1002/eji.1830150518. [DOI] [PubMed] [Google Scholar]
- 7.Strowig T, Brilot F, Arrey F, Bougras G, Thomas D, Muller WA, Munz C. Tonsilar NK cells restrict B cell transformation by the Epstein-Barr virus via IFN-gamma. PLoS Pathog. 2008;4(2):e27. doi: 10.1371/journal.ppat.0040027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Inn KS, Lee SH, Rathbun JY, Wong LY, Toth Z, Machida K, Ou JH, Jung JU. Inhibition of RIG-I-mediated signaling by Kaposi’s sarcoma-associated herpesvirus-encoded deubiquitinase ORF64. J Virol. 2011;85(20):10899–10904. doi: 10.1128/JVI.00690-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bickham K, Goodman K, Paludan C, Nikiforow S, Tsang ML, Steinman RM, Münz C. Dendritic cells initiate immune control of Epstein-Barr virus transformation of B lymphocytes in vitro. J Exp Med. 2003;198(11):1653–1663. doi: 10.1084/jem.20030646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Iwakiri D, Zhou L, Samanta M, Matsumoto M, Ebihara T, Seya T, Imai S, Fujieda M, Kawa K, Takada K. Epstein-Barr virus (EBV)-encoded small RNA is released from EBV-infected cells and activates signaling from Toll-like receptor 3. J Exp Med. 2009;206(10):2091–2099. doi: 10.1084/jem.20081761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Pappworth IY, Wang EC, Rowe M. The switch from latent to productive infection in epstein-barr virus-infected B cells is associated with sensitization to NK cell killing. J Virol. 2007;81(2):474–482. doi: 10.1128/JVI.01777-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Chijioke O, Muller A, Feederle R, Barros MH, Krieg C, Emmel V, Marcenaro E, Leung CS, Antsiferova O, Landtwing V, Bossart W, et al. Human natural killer cells prevent infectious mononucleosis features by targeting lytic Epstein-Barr virus infection. Cell Rep. 2013;5(6):1489–1498. doi: 10.1016/j.celrep.2013.11.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13•.Azzi T, Lünemann A, Murer A, Ueda S, Beziat V, Malmberg KJ, Staubli G, Gysin C, Berger C, Münz C, Chijioke O, et al. Role for early-differentiated natural killer cells in infectious mononucleosis. Blood. 2014;124(16):2533–2543. doi: 10.1182/blood-2014-01-553024. This study documents expansion of early differentiated natural killer (NK) cells during symptomatic primary EBV infection and targeting of lytically EBV replicating B cells by this NK cell population. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14•.Chung BK, Tsai K, Allan LL, Zheng DJ, Nie JC, Biggs CM, Hasan MR, Kozak FK, van den Elzen P, Priatel JJ, Tan R. Innate immune control of EBV-infected B cells by invariant natural killer T cells. Blood. 2013;122(15):2600–2608. doi: 10.1182/blood-2013-01-480665. This study documents EBV specific immune control by invariant NKT cells. [DOI] [PubMed] [Google Scholar]
- 15.Yuling H, Ruijing X, Li L, Xiang J, Rui Z, Yujuan W, Lijun Z, Chunxian D, Xinti T, Wei X, Lang C, et al. EBV-induced human CD8+ NKT cells suppress tumorigenesis by EBV-associated malignancies. Cancer Res. 2009;69(20):7935–7944. doi: 10.1158/0008-5472.CAN-09-0828. [DOI] [PubMed] [Google Scholar]
- 16•.Xiang Z, Liu Y, Zheng J, Liu M, Lv A, Gao Y, Hu H, Lam KT, Chan GC, Yang Y, Chen H, et al. Targeted activation of human Vgamma9Vdelta2-T cells controls Epstein-Barr virus-induced B cell lymphoproliferative disease. Cancer Cell. 2014;26(4):565–576. doi: 10.1016/j.ccr.2014.07.026. This study documents that gamma delta T cells can be harnessed against EBV infection. [DOI] [PubMed] [Google Scholar]
- 17.Williams H, McAulay K, Macsween KF, Gallacher NJ, Higgins CD, Harrison N, Swerdlow AJ, Crawford DH. The immune response to primary EBV infection: a role for natural killer cells. Br J Haematol. 2005;129(2):266–274. doi: 10.1111/j.1365-2141.2005.05452.x. [DOI] [PubMed] [Google Scholar]
- 18.Balfour HH, Jr, Odumade OA, Schmeling DO, Mullan BD, Ed JA, Knight JA, Vezina HE, Thomas W, Hogquist KA. Behavioral, virologic, and immunologic factors associated with acquisition and severity of primary Epstein-Barr virus infection in university students. J Infect Dis. 2013;207(1):80–88. doi: 10.1093/infdis/jis646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lünemann A, Vanoaica LD, Azzi T, Nadal D, Münz C. A distinct subpopulation of human NK cells restricts B cell transformation by EBV. J Immunol. 2013;191(10):4989–4995. doi: 10.4049/jimmunol.1301046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Luzuriaga K, Sullivan JL. Infectious mononucleosis. N Engl J Med. 2010;362(21):1993–2000. doi: 10.1056/NEJMcp1001116. [DOI] [PubMed] [Google Scholar]
- 21.Ishido S, Choi JK, Lee BS, Wang C, DeMaria M, Johnson RP, Cohen GB, Jung JU. Inhibition of natural killer cell-mediated cytotoxicity by Kaposi’s sarcoma-associated herpesvirus K5 protein. Immunity. 2000;13(3):365–374. doi: 10.1016/s1074-7613(00)00036-4. [DOI] [PubMed] [Google Scholar]
- 22.Lee HR, Choi WC, Lee S, Hwang J, Hwang E, Guchhait K, Haas J, Toth Z, Jeon YH, Oh TK, Kim MH, et al. Bilateral inhibition of HAUSP deubiquitinase by a viral interferon regulatory factor protein. Nat Struct Mol Biol. 2011;18(12):1336–1344. doi: 10.1038/nsmb.2142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Savoldo B, Huls MH, Liu Z, Okamura T, Volk HD, Reinke P, Sabat R, Babel N, Jones JF, Webster-Cyriaque J, Gee AP, et al. Autologous Epstein-Barr virus (EBV)-specific cytotoxic T cells for the treatment of persistent active EBV infection. Blood. 2002;100(12):4059–4066. doi: 10.1182/blood-2002-01-0039. [DOI] [PubMed] [Google Scholar]
- 24.Münz C, Bickham KL, Subklewe M, Tsang ML, Chahroudi A, Kurilla MG, Zhang D, O’Donnell M, Steinman RM. Human CD4+ T lymphocytes consistently respond to the latent Epstein-Barr virus nuclear antigen EBNA1. J Exp Med. 2000;191(10):1649–1660. doi: 10.1084/jem.191.10.1649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Leen A, Meij P, Redchenko I, Middeldorp J, Bloemena E, Rickinson A, Blake N. Differential immunogenicity of Epstein-Barr virus latent-cycle proteins for human CD4+ T-helper 1 responses. J Virol. 2001;75(18):8649–8659. doi: 10.1128/JVI.75.18.8649-8659.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hislop AD, Ressing ME, van Leeuwen D, Pudney VA, Horst D, Koppers-Lalic D, Croft NP, Neefjes JJ, Rickinson AB, Wiertz EJ. A CD8+ T cell immune evasion protein specific to Epstein-Barr virus and its close relatives in Old World primates. J Exp Med. 2007;204(8):1863–1873. doi: 10.1084/jem.20070256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Yajima M, Imadome K, Nakagawa A, Watanabe S, Terashima K, Nakamura H, Ito M, Shimizu N, Yamamoto N, Fujiwara S. T cell-mediated control of Epstein-Barr virus infection in humanized mice. J Infect Dis. 2009;200(10):1611–1615. doi: 10.1086/644644. [DOI] [PubMed] [Google Scholar]
- 28.Ma X, Yang L, Xiao L, Tang M, Liu L, Li Z, Deng M, Sun L, Cao Y. Down-regulation of EBV-LMP1 radio-sensitizes nasal pharyngeal carcinoma cells via NF-kappaB regulated ATM expression. PLoS One. 2011;6(11):e24647. doi: 10.1371/journal.pone.0024647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29•.Antsiferova O, Muller A, Ramer PC, Chijioke O, Chatterjee B, Raykova A, Planas R, Sospedra M, Shumilov A, Tsai MH, Delecluse HJ, et al. Adoptive transfer of EBV specific CD8+ T cell clones can transiently control EBV infection in humanized mice. PLoS Pathog. 2014;10(8):e1004333. doi: 10.1371/journal.ppat.1004333. This study documents for the first time a protective role for lytic EBV antigen specific CD8+ T cells. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30•.Linnerbauer S, Behrends U, Adhikary D, Witter K, Bornkamm GW, Mautner J. Virus and autoantigen-specific CD4+ T cells are key effectors in a SCID mouse model of EBV-associated post-transplant lymphoproliferative disorders. PLoS Pathog. 2014;10(5):e1004068. doi: 10.1371/journal.ppat.1004068. This study documents for the first time a protective role of CD4+ T cells during EBV infection in vivo. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Tsai DE, Douglas L, Andreadis C, Vogl DT, Arnoldi S, Kotloff R, Svoboda J, Bloom RD, Olthoff KM, Brozena SC, Schuster SJ, et al. EBV PCR in the diagnosis and monitoring of posttransplant lymphoproliferative disorder: results of a two-arm prospective trial. Am J Transplant. 2008;8(5):1016–1024. doi: 10.1111/j.1600-6143.2008.02183.x. [DOI] [PubMed] [Google Scholar]
- 32.Hohaus S, Santangelo R, Giachelia M, Vannata B, Massini G, Cuccaro A, Martini M, Cesarini V, Cenci T, D’Alo F, Voso MT, et al. The viral load of Epstein-Barr virus (EBV) DNA in peripheral blood predicts for biological and clinical characteristics in Hodgkin lymphoma. Clin Cancer Res. 2011;17(9):2885–2892. doi: 10.1158/1078-0432.CCR-10-3327. [DOI] [PubMed] [Google Scholar]
- 33.Shao JY, Li YH, Gao HY, Wu QL, Cui NJ, Zhang L, Cheng G, Hu LF, Ernberg I, Zeng YX. Comparison of plasma Epstein-Barr virus (EBV) DNA levels and serum EBV immunoglobulin A/virus capsid antigen antibody titers in patients with nasopharyngeal carcinoma. Cancer. 2004;100(6):1162–1170. doi: 10.1002/cncr.20099. [DOI] [PubMed] [Google Scholar]
- 34.Horst D, Burrows SR, Gatherer D, van Wilgenburg B, Bell MJ, Boer IG, Ressing ME, Wiertz EJ. Epstein-Barr virus isolates retain their capacity to evade T cell immunity through BNLF2a despite extensive sequence variation. J Virol. 2012;86(1):572–577. doi: 10.1128/JVI.05151-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Murat P, Zhong J, Lekieffre L, Cowieson NP, Clancy JL, Preiss T, Balasubramanian S, Khanna R, Tellam J. G-quadruplexes regulate Epstein-Barr virus-encoded nuclear antigen 1 mRNA translation. Nat Chem Biol. 2014;10(5):358–364. doi: 10.1038/nchembio.1479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Tellam JT, Zhong J, Lekieffre L, Bhat P, Martinez M, Croft NP, Kaplan W, Tellam RL, Khanna R. mRNA Structural constraints on EBNA1 synthesis impact on in vivo antigen presentation and early priming of CD8+ T cells. PLoS Pathog. 2014;10(10):e1004423. doi: 10.1371/journal.ppat.1004423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Babcock GJ, Decker LL, Volk M, Thorley-Lawson DA. EBV persistence in memory B cells in vivo. Immunity. 1998;9(3):395–404. doi: 10.1016/s1074-7613(00)80622-6. [DOI] [PubMed] [Google Scholar]
- 38.Ohashi M, Fogg MH, Orlova N, Quink C, Wang F. An Epstein-Barr virus encoded inhibitor of Colony Stimulating Factor-1 signaling is an important determinant for acute and persistent EBV infection. PLoS Pathog. 2012;8(12):e1003095. doi: 10.1371/journal.ppat.1003095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.van Gent M, Braem SG, de Jong A, Delagic N, Peeters JG, Boer IG, Moynagh PN, Kremmer E, Wiertz EJ, Ovaa H, Griffin BD, et al. Epstein-Barr virus large tegument protein BPLF1 contributes to innate immune evasion through interference with toll-like receptor signaling. PLoS Pathog. 2014;10(2):e1003960. doi: 10.1371/journal.ppat.1003960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40••.Quinn LL, Zuo J, Abbott RJ, Shannon-Lowe C, Tierney RJ, Hislop AD, Rowe M. Cooperation between Epstein-Barr virus immune evasion proteins spreads protection from CD8+ T cell recognition across all three phases of the lytic cycle. PLoS Pathog. 2014;10(8):e1004322. doi: 10.1371/journal.ppat.1004322. This study establishes the importance of EBV immune evasins during the different phases of the viral lytic cycle. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Ressing ME, van Leeuwen D, Verreck FA, Gomez R, Heemskerk B, Toebes M, Mullen MM, Jardetzky TS, Longnecker R, Schilham MW, Ottenhoff TH, et al. Interference with T cell receptor-HLA-DR interactions by Epstein-Barr virus gp42 results in reduced T helper cell recognition. Proc Natl Acad Sci U S A. 2003;100(20):11583–11588. doi: 10.1073/pnas.2034960100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Jochum S, Moosmann A, Lang S, Hammerschmidt W, Zeidler R. The EBV immunoevasins vIL-10 and BNLF2a protect newly infected B cells from immune recognition and elimination. PLoS Pathog. 2012;8(5):e1002704. doi: 10.1371/journal.ppat.1002704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Chang Y, Cesarman E, Pessin MS, Lee F, Culpepper J, Knowles DM, Moore PS. Identification of herpesvirus-like DNA sequences in AIDS-associated Kaposi’s sarcoma. Science. 1994;266(5192):1865–1869. doi: 10.1126/science.7997879. [DOI] [PubMed] [Google Scholar]
- 44.Mesri EA, Cesarman E, Boshoff C. Kaposi’s sarcoma and its associated herpesvirus. Nat Rev Cancer. 2010;10(10):707–719. doi: 10.1038/nrc2888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Cesarman E, Chang Y, Moore PS, Said JW, Knowles DM. Kaposi’s sarcoma-associated herpesvirus-like DNA sequences in AIDS-related body-cavity-based lymphomas. N Engl J Med. 1995;332(18):1186–1191. doi: 10.1056/NEJM199505043321802. [DOI] [PubMed] [Google Scholar]
- 46.Sirianni MC, Vincenzi L, Topino S, Giovannetti A, Mazzetta F, Libi F, Scaramuzzi D, Andreoni M, Pinter E, Baccarini S, Rezza G, et al. NK cell activity controls human herpesvirus 8 latent infection and is restored upon highly active antiretroviral therapy in AIDS patients with regressing Kaposi’s sarcoma. European Journal of Immunology. 2002;32(10):2711–2720. doi: 10.1002/1521-4141(2002010)32:10<2711::AID-IMMU2711>3.0.CO;2-3. [DOI] [PubMed] [Google Scholar]
- 47.Dupuy S, Lambert M, Zucman D, Choukem SP, Tognarelli S, Pages C, Lebbe C, Caillat-Zucman S. Human Herpesvirus 8 (HHV8) Sequentially Shapes the NK Cell Repertoire during the Course of Asymptomatic Infection and Kaposi Sarcoma. Plos Pathogens. 2012;8(1):e1002486. doi: 10.1371/journal.ppat.1002486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Robey RC, Mletzko S, Gotch FM. The T-Cell Immune Response against Kaposi’s Sarcoma-Associated Herpesvirus. Adv Virol. 2010;2010:340356. doi: 10.1155/2010/340356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Hislop AD, Sabbah S. CD8+ T cell immunity to Epstein-Barr virus and Kaposi’s sarcoma-associated herpes virus. Semin Cancer Biol. 2008;18(6):416–422. doi: 10.1016/j.semcancer.2008.10.005. [DOI] [PubMed] [Google Scholar]
- 50.Ganem D. KSHV and the pathogenesis of Kaposi sarcoma: listening to human biology and medicine. J Clin Invest. 2010;120(4):939–949. doi: 10.1172/JCI40567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Mesri EA, Cesarman E, Boshoff C. Kaposi’s sarcoma and its associated herpesvirus. Nat Rev Cancer. 2011;10(10):707–719. doi: 10.1038/nrc2888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Letang E, Lewis JJ, Bower M, Mosam A, Borok M, Campbell TB, Naniche D, Newsom-Davis T, Shaik F, Fiorillo S, Miro JM, et al. Immune reconstitution inflammatory syndrome associated with Kaposi sarcoma: higher incidence and mortality in Africa than in the UK. AIDS. 2013;27(10):1603–1613. doi: 10.1097/QAD.0b013e328360a5a1. [DOI] [PubMed] [Google Scholar]
- 53.Kerur N, Veettil MV, Sharma-Walia N, Bottero V, Sadagopan S, Otageri P, Chandran B. IFI16 acts as a nuclear pathogen sensor to induce the inflammasome in response to Kaposi Sarcoma-associated herpesvirus infection. Cell Host Microbe. 2011;9(5):363–375. doi: 10.1016/j.chom.2011.04.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Samaniego F, Markham PD, Gendelman R, Gallo RC, Ensoli B. Inflammatory cytokines induce endothelial cells to produce and release basic fibroblast growth factor and to promote Kaposi’s sarcoma-like lesions in nude mice. J Immunol. 1997;158(4):1887–1894. [PubMed] [Google Scholar]
- 55.West JA, Wicks M, Gregory SM, Chugh P, Jacobs SR, Zhang Z, Host KM, Dittmer DP, Damania B. An Important Role for Mitochondrial Antiviral Signaling Protein in the Kaposi’s Sarcoma-Associated Herpesvirus Life Cycle. J Virol. 2014;88(10):5778–5787. doi: 10.1128/JVI.03226-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Zhu FX, King SM, Smith EJ, Levy DE, Yuan Y. A Kaposi’s sarcoma-associated herpesviral protein inhibits virus-mediated induction of type I interferon by blocking IRF-7 phosphorylation and nuclear accumulation. Proc Natl Acad Sci U S A. 2002;99(8):5573–5578. doi: 10.1073/pnas.082420599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Zhu FX, Yuan Y. The ORF45 protein of Kaposi’s sarcoma-associated herpesvirus is associated with purified virions. J Virol. 2003;77(7):4221–4230. doi: 10.1128/JVI.77.7.4221-4230.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Zhu FX, Li X, Zhou F, Gao SJ, Yuan Y. Functional characterization of Kaposi’s sarcoma-associated herpesvirus ORF45 by bacterial artificial chromosome-based mutagenesis. J Virol. 2006;80(24):12187–12196. doi: 10.1128/JVI.01275-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Zhu FX, Sathish N, Yuan Y. Antagonism of host antiviral responses by Kaposi’s sarcoma-associated herpesvirus tegument protein ORF45. PLoS One. 2010;5(5):e10573. doi: 10.1371/journal.pone.0010573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Sathish N, Zhu FX, Golub EE, Liang Q, Yuan Y. Mechanisms of autoinhibition of IRF-7 and a probable model for inactivation of IRF-7 by Kaposi’s sarcoma-associated herpesvirus protein ORF45. J Biol Chem. 2011;286(1):746–756. doi: 10.1074/jbc.M110.150920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Liang Q, Fu B, Wu F, Li X, Yuan Y, Zhu F. ORF45 of Kaposi’s sarcoma-associated herpesvirus inhibits phosphorylation of interferon regulatory factor 7 by IKKepsilon and TBK1 as an alternative substrate. J Virol. 2012;86(18):10162–10172. doi: 10.1128/JVI.05224-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Yu Y, Hayward GS. The ubiquitin E3 ligase RAUL negatively regulates type i interferon through ubiquitination of the transcription factors IRF7 and IRF3. Immunity. 2010;33(6):863–877. doi: 10.1016/j.immuni.2010.11.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Yu Y, Wang SE, Hayward GS. The KSHV immediate-early transcription factor RTA encodes ubiquitin E3 ligase activity that targets IRF7 for proteosome-mediated degradation. Immunity. 2005;22(1):59–70. doi: 10.1016/j.immuni.2004.11.011. [DOI] [PubMed] [Google Scholar]
- 64.Skalsky RL, Samols MA, Plaisance KB, Boss IW, Riva A, Lopez MC, Baker HV, Renne R. Kaposi’s sarcoma-associated herpesvirus encodes an ortholog of miR-155. J Virol. 2007;81(23):12836–12845. doi: 10.1128/JVI.01804-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Gottwein E, Mukherjee N, Sachse C, Frenzel C, Majoros WH, Chi JTA, Braich R, Manoharan M, Soutschek J, Ohler U, Cullen BR. A viral microRNA functions as an orthologue of cellular miR-155. Nature. 2007;450(7172):1096–U1017. doi: 10.1038/nature05992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Liang D, Gao Y, Lin X, He Z, Zhao Q, Deng Q, Lan K. A human herpesvirus miRNA attenuates interferon signaling and contributes to maintenance of viral latency by targeting IKKepsilon. Cell Res. 2011;21(5):793–806. doi: 10.1038/cr.2011.5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Lee HR, Kim MH, Lee JS, Liang C, Jung JU. Viral interferon regulatory factors. J Interferon Cytokine Res. 2009;29(9):621–627. doi: 10.1089/jir.2009.0067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Jacobs SR, Damania B. The viral interferon regulatory factors of KSHV: immunosuppressors or oncogenes? Frontiers in immunology. 2011;2:19. doi: 10.3389/fimmu.2011.00019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Baresova P, Pitha PM, Lubyova B. Distinct roles of Kaposi’s sarcoma-associated herpesvirus-encoded viral interferon regulatory factors in inflammatory response and cancer. J Virol. 2013;87(17):9398–9410. doi: 10.1128/JVI.03315-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Davison AJ, Stow ND. New genes from old: redeployment of dUTPase by herpesviruses. J Virol. 2005;79(20):12880–12892. doi: 10.1128/JVI.79.20.12880-12892.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Leang RS, Wu TT, Hwang S, Liang LT, Tong L, Truong JT, Sun R. The anti-interferon activity of conserved viral dUTPase ORF54 is essential for an effective MHV-68 infection. PLoS Pathog. 2011;7(10):e1002292. doi: 10.1371/journal.ppat.1002292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Woellmer A, Arteaga-Salas JM, Hammerschmidt W. BZLF1 Governs CpG-Methylated Chromatin of Epstein-Barr Virus Reversing Epigenetic Repression. PLoS Pathog. 2012;8(9):e1002902. doi: 10.1371/journal.ppat.1002902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Upton JW, Chan FK. Staying Alive: Cell Death in Antiviral Immunity. Mol Cell. 2014;54(2):273–280. doi: 10.1016/j.molcel.2014.01.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Cheng EH, Nicholas J, Bellows DS, Hayward GS, Guo HG, Reitz MS, Hardwick JM. A Bcl-2 homolog encoded by Kaposi sarcoma-associated virus, human herpesvirus 8, inhibits apoptosis but does not heterodimerize with Bax or Bak. Proc Natl Acad Sci U S A. 1997;94(2):690–694. doi: 10.1073/pnas.94.2.690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Tolani B, Matta H, Gopalakrishnan R, Punj V, Chaudhary PM. NEMO Is Essential for Kaposi’s Sarcoma-Associated Herpesvirus-Encoded vFLIP K13-Induced Gene Expression and Protection against Death Receptor-Induced Cell Death, and Its N-Terminal 251 Residues Are Sufficient for This Process. J Virol. 2014;88(11):6345–6354. doi: 10.1128/JVI.00028-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Wen KW, Damania B. Hsp90 and Hsp40/Erdj3 are required for the expression and anti-apoptotic function of KSHV K1. Oncogene. 2010;29(24):3532–3544. doi: 10.1038/onc.2010.124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Wang S, Wang S, Maeng H, Young DP, Prakash O, Fayad LE, Younes A, Samaniego F. K1 protein of human herpesvirus 8 suppresses lymphoma cell Fas-mediated apoptosis. Blood. 2007;109(5):2174–2182. doi: 10.1182/blood-2006-02-003178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Lee HR, Doganay S, Chung B, Toth Z, Brulois K, Lee S, Kanketayeva Z, Feng P, Ha T, Jung JU. Kaposi’s sarcoma-associated herpesvirus viral interferon regulatory factor 4 (vIRF4) targets expression of cellular IRF4 and the Myc gene to facilitate lytic replication. J Virol. 2014;88(4):2183–2194. doi: 10.1128/JVI.02106-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79••.Chudasama P, Konrad A, Jochmann R, Lausen B, Holz P, Naschberger E, Neipel F, Britzen-Laurent N, Sturzl M. Structural proteins of Kaposi’s sarcoma-associated herpesvirus antagonize p53-mediated apoptosis. Oncogene. 2015;34(5):639–4. doi: 10.1038/onc.2013.595. This study documents KSHV’s ability to inhibit the important apoptosis pathway via p53. [DOI] [PubMed] [Google Scholar]
- 80.Abend JR, Uldrick T, Ziegelbauer JM. Regulation of tumor necrosis factor-like weak inducer of apoptosis receptor protein (TWEAKR) expression by Kaposi’s sarcoma-associated herpesvirus microRNA prevents TWEAK-induced apoptosis and inflammatory cytokine expression. J Virol. 2010;84(23):12139–12151. doi: 10.1128/JVI.00884-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81•.Nowag H, Guhl B, Thriene K, Romao S, Ziegler U, Dengjel J, Münz C. Macroautopphagy proteins assist Epstein Barr virus production and get incorporated into the virus particles. EBioMedicine. 2014;1(2–3):116–125. doi: 10.1016/j.ebiom.2014.11.007. This study documents for the first time that a virus uses autophagic membranes for its envelope. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Granato M, Santarelli R, Farina A, Gonnella R, Lotti LV, Faggioni A, Cirone M. EBV blocks the autophagic flux and appropriates the autophagic machinery to enhance viral replication. J Virol. 2014;88(21):12715–26. doi: 10.1128/JVI.02199-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Coscoy L, Ganem D. Kaposi’s sarcoma-associated herpesvirus encodes two proteins that block cell surface display of MHC class I chains by enhancing their endocytosis. Proc Natl Acad Sci U S A. 2000;97(14):8051–8056. doi: 10.1073/pnas.140129797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Ishido S, Wang C, Lee BS, Cohen GB, Jung JU. Downregulation of major histocompatibility complex class I molecules by Kaposi’s sarcoma-associated herpesvirus K3 and K5 proteins. J Virol. 2000;74(11):5300–5309. doi: 10.1128/jvi.74.11.5300-5309.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Stevenson PG, Efstathiou S, Doherty PC, Lehner PJ. Inhibition of MHC class I-restricted antigen presentation by gamma 2-herpesviruses. Proc Natl Acad Sci U S A. 2000;97(15):8455–8460. doi: 10.1073/pnas.150240097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Duncan LM, Piper S, Dodd RB, Saville MK, Sanderson CM, Luzio JP, Lehner PJ. Lysine-63-linked ubiquitination is required for endolysosomal degradation of class I molecules. EMBO J. 2006;25(8):1635–1645. doi: 10.1038/sj.emboj.7601056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Thomas M, Boname JM, Field S, Nejentsev S, Salio M, Cerundolo V, Wills M, Lehner PJ. Down-regulation of NKG2D and NKp80 ligands by Kaposi’s sarcoma-associated herpesvirus K5 protects against NK cell cytotoxicity. Proc Natl Acad Sci U S A. 2008;105(5):1656–1661. doi: 10.1073/pnas.0707883105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Coscoy L, Ganem D. A viral protein that selectively downregulates ICAM-1 and B7-2 and modulates T cell costimulation. Journal of Clinical Investigation. 2001;107(12):1599–1606. doi: 10.1172/JCI12432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Madrid AS, Ganem D. Kaposi’s sarcoma-associated herpesvirus ORF54/dUTPase downregulates a ligand for the NK activating receptor NKp44. J Virol. 2012;86(16):8693–8704. doi: 10.1128/JVI.00252-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Nachmani D, Stern-Ginossar N, Sarid R, Mandelboim O. Diverse herpesvirus microRNAs target the stress-induced immune ligand MICB to escape recognition by natural killer cells. Cell Host Microbe. 2009;5(4):376–385. doi: 10.1016/j.chom.2009.03.003. [DOI] [PubMed] [Google Scholar]
- 91.Mansouri M, Douglas J, Rose PP, Gouveia K, Thomas G, Means RE, Moses AV, Fruh K. Kaposi sarcoma herpesvirus K5 removes CD31/PECAM from endothelial cells. Blood. 2006;108(6):1932–1940. doi: 10.1182/blood-2005-11-4404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Mansouri M, Rose PP, Moses AV, Fruh K. Remodeling of endothelial adherens junctions by Kaposi’s sarcoma-associated herpesvirus. Journal of Virology. 2008;82(19):9615–9628. doi: 10.1128/JVI.02633-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Manes TD, Hoer S, Muller WA, Lehner PJ, Pober JS. Kaposi’s sarcoma-associated herpesvirus K3 and K5 proteins block distinct steps in transendothelial migration of effector memory CD4+ T cells by targeting different endothelial proteins. J Immunol. 2010;184(9):5186–5192. doi: 10.4049/jimmunol.0902938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Muller WA. Mechanisms of transendothelial migration of leukocytes. Circ Res. 2009;105(3):223–230. doi: 10.1161/CIRCRESAHA.109.200717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Kledal TN, Rosenkilde MM, Coulin F, Simmons G, Johnsen AH, Alouani S, Power CA, Luttichau HR, Gerstoft J, Clapham PR, Clark-Lewis I, et al. A broad-spectrum chemokine antagonist encoded by Kaposi’s sarcoma-associated herpesvirus. Science. 1997;277(5332):1656–1659. doi: 10.1126/science.277.5332.1656. [DOI] [PubMed] [Google Scholar]
- 96.Yamin R, Kaynan NS, Glasner A, Vitenshtein A, Tsukerman P, Bauman Y, Ophir Y, Elias S, Bar-On Y, Gur C, Mandelboim O. The viral KSHV chemokine vMIP-II inhibits the migration of Naive and activated human NK cells by antagonizing two distinct chemokine receptors. PLoS Pathog. 2013;9(8):e1003568. doi: 10.1371/journal.ppat.1003568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Butler LM, Jeffery HC, Wheat RL, Rae PC, Townsend K, Alkharsah KR, Schulz TF, Nash GB, Blackbourn DJ. Kaposi’s sarcoma-associated herpesvirus infection of endothelial cells inhibits neutrophil recruitment through an interleukin-6-dependent mechanism: a new paradigm for viral immune evasion. Journal of Virology. 2011;85(14):7321–7332. doi: 10.1128/JVI.00021-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Rezaee SAR, Gracie JA, McInnes IB, Blackbourn DJ. Inhibition of neutrophil function by the Kaposi’s sarcoma-associated herpesvirus vOX2 protein. Aids. 2005;19(16):1907–1910. doi: 10.1097/01.aids.0000189849.75699.46. [DOI] [PubMed] [Google Scholar]
- 99.Guihot A, Dupin N, Marcelin AG, Gorin I, Bedin AS, Bossi P, Galicier L, Oksenhendler E, Autran B, Carcelain G. Low T cell responses to human herpesvirus 8 in patients with AIDS-related and classic Kaposi sarcoma. J Infect Dis. 2006;194(8):1078–1088. doi: 10.1086/507648. [DOI] [PubMed] [Google Scholar]