Abstract
Despite evidence of the benefits of physical activity, most individuals with type 2 diabetes do not meet physical activity recommendations. The purpose of this study was to test the efficacy of a brief intervention targeting self-efficacy and self-regulation to increase physical activity in older adults with type 2 diabetes.
Older adults (Mage = 61.8 ± 6.4) with type 2 diabetes or metabolic syndrome were randomized into a titrated physical activity intervention (n = 58) or an online health education course (n = 58). The intervention included walking exercise and theory-based group workshops. Self-efficacy, self-regulation and physical activity were assessed at baseline, post-intervention, and a follow-up.
Results indicated a group by time effect for self-regulation [F(2,88) = 14.021, p < .001, η2 = .24] and self-efficacy [F(12,77) = 2.322, p < .05, η2 = .266] with increases in the intervention group. The intervention resulted in short-term increases in physical activity (d = .76, p < .01), which were partially maintained at the six-month follow-up (d = .35, p < .01).
The intervention increased short-term physical activity but was not successful at maintaining increases in physical activity. Similar intervention effects were observed in self-efficacy and self-regulation. Future research warrants adjusting intervention strategies to increase long-term change.
Keywords: diabetes, physical activity, older adults, self-regulation, self-efficacy
INTRODUCTION
The prevention and treatment of metabolic disease, such as type 2 diabetes, has become a public health priority. It is estimated that 25.8 million Americans, approximately 8% of the population, have diabetes and almost 2 million new cases are diagnosed each year (CDC, 2011). The prevalence of type 2 diabetes increases in older adults to 26.9%, as aging is associated with incremental and gradual losses of glycemic control (CDC, 2011; Resnick, Harris, Brock & Harris, 2000). As the population is increasingly represented by older adults, the long-term consequences of metabolic disease and associated comorbidities hold substantial individual, community, and societal impact.
Lifestyle modification, specifically physical activity, is crucial to controlling disease progression and is considered first-line therapy for type 2 diabetes (CDC, 2011; Grundy et al., 2005; Jeon, Looken, Hu, & van Dam, 2007). Physical activity alone, with or without weight loss, improves glucose tolerance and whole-body insulin sensitivity (Duncan et al., 2003; Lunciano et al., 2002; Matos et al., 2010; Touati et al., 2011). Despite evidence of the benefits of physical activity for health and American Diabetes Association’s position that it is a cornerstone of treatment (Sigal, Kenny, Wasserman, Castandeda-Sceppa, & White, 2006), epidemiological evidence suggests that most individuals with or at risk for type 2 diabetes do not meet physical activity recommendations (Morrato, Hill, Wyatt, Ghushchyan, & Sullivan, 2007). Between 23% and 37% of adults with type 2 diabetes meet the recommended levels of physical activity (Nwasuruba, Khan, & Egede, 2007) compared to 50% of Americans who reach the aerobic exercise recommendation of ≥150 minutes/week (CDC, 2013). Individuals affected by type 2 diabetes are less physically active than their peers even though physical inactivity is a risk factor for further disease progression.
Adherence to complex behaviors, such as regular physical activity, is difficult. Being physically active requires substantial effort and continued perseverance (McAuley & Blissmer, 2000). Adopting physical activity is a difficult task for many and attrition estimates are approximately 50% within the first three to six months (Dishman, 1982). Social cognitive theory specifies a set of psychosocial determinants and is useful for understanding, explaining, and predicting physical activity behavior (Bandura, 1986). Two core constructs include self-efficacy and self-regulation. Self-efficacy, the belief in one’s own capabilities to successfully carry out a course of action, influences individual effort expenditure, activity choice, and persistence in the face of barriers or failure (McAuley & Blissmer, 2000). Self-regulation involves goal-setting, planning, self-monitoring, and self-rewarding. Self-regulation allows individuals to influence their own health habits to bring their behavior in-line with their goals (Maes & Karoly, 2005). Personal agency relies on efficacy and self-regulation (Bandura, 1997).
Considerable research has examined the impact of lifestyle modification and specific physical activity prescription on diabetes risk reduction and disease outcomes. Results, from trials, such as the Diabetes Prevention Program, indicate that lifestyle modification is a powerful treatment to prevent diabetes (Diabetes Prevention Program Research Group, 2002). However, most previous studies have utilized structured, on-site programs, with devoted resources to ensuring program adherence. As the efficacy of physical activity as a therapy for type 2 diabetes has been established, more research examining mechanisms and determinants of individual adherence to habitual physical activity is needed. The purpose of this study was to determine the efficacy of a brief, titrated, behavioral intervention to increase physical activity levels in older adults with type 2 diabetes. It was hypothesized that individuals in the intervention group would adopt and sustain higher levels of physical activity compared to baseline levels and an education control group. It was also hypothesized that improvements in the social cognitive constructs of self-efficacy and self-regulation would be related to change in physical activity.
METHODS
Participants
Individuals between the ages of 50 to 75, diagnosed with type 2 diabetes or metabolic syndrome, were recruited to participate in a six-month physical activity research study. Additional inclusion criteria included: willingness to be randomized, physician clearance to exercise, ability to communicate in English, and a score of 21+ on the Telephone Interview for Cognitive Status (de Jager, Budge, & Clarke, 2003). Individuals who reported regular exercise of 30 or more minutes at least twice weekly for the last six months were excluded. There was no racial or gender bias in the selection of participants.
Measures
Demographics and Health History
Each participant completed a standard health history and basic demographics questionnaire. All current medications, including insulin, were recorded. Height and weight were measured and body mass index was calculated.
Physical Activity
Physical activity was measured via accelerometry. Participants were instructed to wear an activity monitor (Actigraph, Pensacola, FL, Model GT1M or GT3X) on their non-dominant hip during all waking hours, except for bathing or swimming, for seven full days. Participants kept a home log, which was used to verify wear-time. Activity data were checked for long periods of non-wear time (0’s) and were validated with the criteria of: 1) at least 10 hours of wear time per day (Masse et al., 2005), 2) at least 3 days of valid data, and 3) a 60-minute interruption period (Copeland, 2009; Mailey et al., 2014). Activity data were collected in one-minute intervals (epochs) with the total counts/day summed and divided by the number of valid days to calculate average daily activity. Freedson cut-points for older adults were used to estimate time spent in sedentary, light, and moderate to vigorous physical activity (Freedson, Melanson, & Sirand, 1998). Accelerometer data were scored using Meterplus version 4.2 (Santech, Inc., San Diego, CA).
Self-efficacy
Several aspects of physical activity related self-efficacy were assessed. The Barriers-specific Self-Efficacy Scale, a 13-item questionnaire, measured beliefs in personal abilities to exercise in the face of barriers such as discouragement or bad weather (McAuley, 1992). Walking self-efficacy was measured relative to ability to 1) exercise (walk) for a specific duration of time (40+ minutes) for the next month, two months, three months etc., (exercise self-efficacy, McAuley, Lox, & Duncan, 1993) and 2) walk continuously at a fast pace for 5 minutes, 10 minutes, 15 minutes, etc., (walking self-efficacy; McAuley, Blissmer, Katula, Duncan, & Mihalko, 2000; McAuley et al., 2009). All items were presented on a continuum from 0 “not confident at all” to 100 “completely confident” to complete the task. The barriers, walking, and exercise self-efficacy scales all showed excellent internal reliability (α = .96, .98, .98 respectively). Self-efficacy was measured at an additional time-point two weeks into the trial due to cognitive re-evaluation of efficacy that can occur after beginning an exercise program (McAuley et al., 2011).
Self-regulation
Self-regulation specific to physical activity was assessed by the 12-item Physical Activity Self-Regulation Scale (PASR-12; Umstattd, Motl, Wilcox, Saunders & Watford, 2009). Its subscales include the following domains: self-monitoring, goal setting, eliciting social support, reinforcement, time management, and relapse prevention. Internal consistency for subscales ranged from α = .72 to .92.
Procedures
An institutional review board approved all procedures. Study (clinical trial #NCT01790724) was advertised via flyers, Internet, email blasts, and local diabetes educators and medical clinics. Advertisements targeted adults with metabolic syndrome or diabetes who were not currently active. Individuals responding to study advertisements were screened by telephone for all qualifying criteria. If individuals met all inclusionary criteria and remained interested in participation, they were scheduled for an orientation meeting and mailed a copy of the informed consent. Both physician consent to participate and confirmation of metabolic syndrome or type 2 diabetes were obtained before randomization. For an overview of the study design refer to Figure 1.
Figure 1.
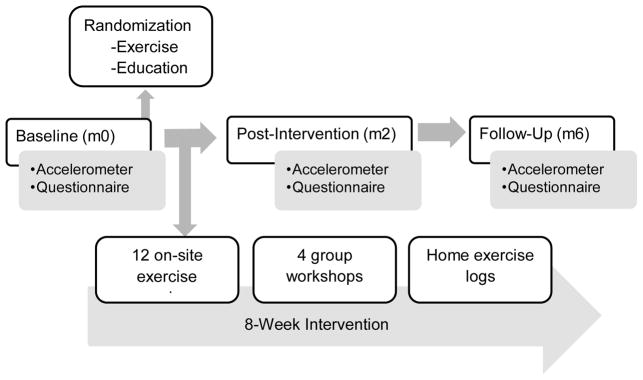
Overview of study design
After all baseline data had been collected, participants were randomized into the exercise intervention or education group; randomization, referencing a computer-generated statistical series based on random sampling, was stratified by age and gender and was conducted by the principal investigator. Study coordinators then contacted participants with their group assignment. As randomization occurred after baseline testing, participants and testers were blinded at baseline. For follow-up assessments, research personnel collecting data were blinded to randomization allocation. Participants who were randomized into the education group were offered a complimentary exercise consultation after study completion. Data collection procedures were repeated immediately post-intervention (month two) and at a four-month follow-up (month six).
Exercise Intervention
The intervention was eight weeks long and included: on-site walking, group workshops, assigned independent aerobic exercise (walking), and the completion of home logs. The intervention was titrated in nature such that participants began with on-site walking sessions three times weekly, which decreased by one session per week every other week. Simultaneously, participants were encouraged to gradually increase independent exercise. In weeks three and four, participants were assigned independent exercise 2x/week. By week 5, participants were instructed to exercise independently 4x/week. Finally, by week 7 participants were exercising completely independent of the research team.
The four group workshops were evenly spaced throughout the intervention period and taught behavior modification strategies grounded in social cognitive theory. The hour-long group workshops were guided by the study investigator and consisted of group discussions and various problem-solving activities. Group activities specifically focused on physical-activity-related self-efficacy and self-regulation. Table 1 outlines the content of the group workshops. Goal setting and goal monitoring were highlighted in each workshop to teach participants how to appropriately set and adjust personal goals. Participants were taught to self-monitor exercise through completing home exercise logs. During the on-site walking sessions, the exercise leader taught participants to complete the logs, which were reviewed at each group workshop. Table 2 illustrates how the sources of self-efficacy information and the sub-components of self-regulation were specifically targeted throughout the entire intervention.
Table 1.
Group workshop topic overview
| Group Workshop | Topics/Activities |
|---|---|
| Session 1 |
|
| Session 2 |
|
| Session 3 |
|
| Session 4 |
|
Table 2.
Specific intervention activities targeting the theoretical constructs of self-efficacy and self-regulation
| Construct | Construct Sources/Sub-components | Tasks/Activities | Timing |
|---|---|---|---|
| Self-efficacy | 1. Social Persuasion |
|
|
| 2. Social Modeling |
|
|
|
| 3. Mastery of Experience |
|
|
|
| 4. Interpretation of States |
|
|
|
| Self- regulation | 1. Self-monitoring |
|
|
| 2. Goal-setting |
|
|
|
| 3. Eliciting Social Support |
|
|
|
| 4. Reinforcement |
|
|
|
| 5. Time Management |
|
|
|
| 6. Relapse Prevention |
|
|
Educational Control
Participants randomized into the educational control group completed an eight-week online diabetes and health education course. Each week consisted of an education module with videos, readings, activities, and a discussion question, which required approximately an hour to complete. The topics were: 1) glucose and why it matters, 2) glucose and insulin, 3) weight control, 4) food and nutrition, 5) physical activity, 6) preventing diabetic complications, 7) stress management, and 8) healthy aging.
Statistical Analysis
Sample size was based on: (a) data from previous estimations of change in physical activity across time (McAuley et al., 2000) and (b) estimates of attrition. With retaining 94 of the initial 125, we calculate power in excess of .90 to detect a conservative effect in the primary outcome of minutes/day of moderate-to-vigorous physical activity. Data were checked for missing items, normality, outliers and errors. Independent t-tests were used to examine group differences at baseline. Repeated measures analysis of variance (ANOVA) was used to detect changes over time in self-efficacy, self-regulation, and physical activity. Interactions and main effects were examined and effect sizes calculated. When baseline values differed by group, repeated measures analysis of covariance (ANCOVA) was used to control for baseline values. Post-hoc tests of group mean equality at follow-up time-points were conducted within the repeated measures ANOVAs.
Correlational statistics and regression analysis were used to explore baseline (m0) predictors and determinants of physical activity post-intervention (m2) and at follow-up (m6). Change scores were calculated (e.g. m2 – m0) and standardized [e.g. (m2 – m0)/SD(m2 – m0)]. Partial correlations—controlling for age, gender, income, baseline glycosylated hemoglobin, and baseline physical activity—between 1) self-efficacy, self-regulation, and physical activity and 2) intervention-related changes in self-efficacy and self-regulation (m2 – m0) and change in physical activity during the follow-up period (m6 – m2) were calculated.
Then, regression analysis was used to examine contribution of changes in self-efficacy and self-regulation to physical activity immediately post intervention, at follow-up, and change in physical activity during the follow-up period (m6 – m2). All analyses employed an intent-to-treat model, comparing the intervention to education control group. Data analyses were conducted in 2014–2015.
RESULTS
Participant Characteristics and Retention
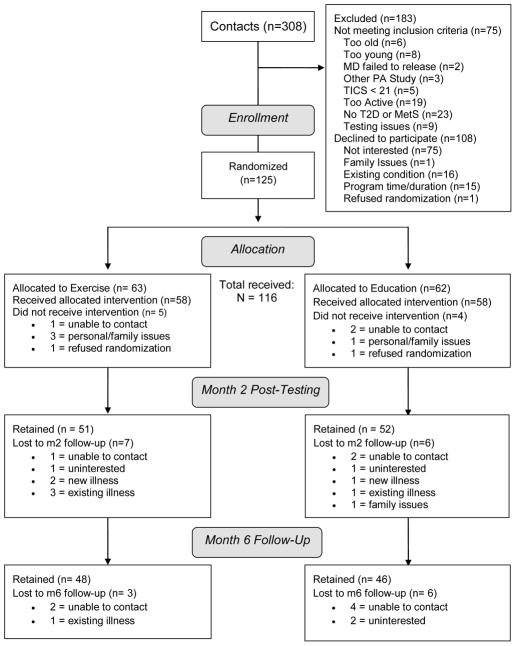
Baseline characteristics of the 116 participants who started the trial are displayed in Table 3. About two-thirds of the sample were taking oral medications for diabetes, such as Metformin or Glyburide, and approximately a quarter of the sample was taking injectable insulin, such as Humalog or Lantus. There were no significant differences between groups on any of the baseline demographic or disease variables. However, for body mass index and glycosylated hemoglobin the exercise group trended towards higher values (p = .08). Most (n = 99, 85.3%) participants had been diagnosed with type 2 diabetes; a small proportion (n = 17, 14.6%) had been diagnosed with metabolic syndrome. During study communication with participants, three adverse events were discovered and documented: one participant sustained a fall unrelated to research activities and two participants were hospitalized due to existing conditions. The CONSORT diagram illustrates participant flow through the trial (see Figure 2).
Table 3.
Baseline sample demographic and health status descriptives
| Exercise Intervention (n = 58) | Educational Control (n = 58) | Total (n = 116) | |
|---|---|---|---|
| Age, y | 61.3 ± 5.8 | 62.4 ± 6.9 | 61.8 ± 6.4 |
| Sex, No. (%) | |||
| Female | 37 (63.8) | 38 (65.5) | 75 (64.7) |
| Male | 21 (36.2) | 20 (34.5) | 41 (35.3) |
| Race, No. (%) | |||
| African American | 9 (15.5) | 7 (12.1) | 16 (13.8) |
| Asian | 2 (3.4) | 1 (1.7) | 3 (2.6) |
| White | 45 (77.6) | 49 (84.5) | 94 (81.0) |
| Multi-Racial | 2 (3.4) | 1 (1.7) | 3 (2.6) |
| Ethnicity, No. (%) | |||
| Hispanic or Latino | 2 (3.4) | None | 2 (1.7) |
| Not Hispanic or Latino | 56 (96.6) | 58 (100) | 114 (98.3) |
| Educationa, No. (%) | |||
| Not High School Graduate | 1 (1.8) | 2 (3.4) | 3 (2.6) |
| High School Diploma | 7 (12.3) | 10 (17.2) | 17 (14.8) |
| Some College | 17 (29.8) | 15 (25.9) | 32 (27.8) |
| College Graduate | 15 (26.3) | 21 (36.2) | 36 (31.3) |
| Graduate Degree | 17 (29.8) | 10 (17.3) | 27 (23.5) |
| Employmenta, No. (%) | |||
| Working Full Time | 19 (33.3) | 21 (36.2) | 40 (34.8) |
| Working Part Time | 14 (24.6) | 14 (24.1) | 28 (24.3) |
| Retired | 20 (35.1) | 17 (29.3) | 37 (32.2) |
| Unemployed/Disability | 3 (5.3) | 6 (10.3) | 10 (8.7) |
| Annual Incomea, No. (%) | |||
| < $20,000 | 6 (10.8) | 9 (16.4) | 15 (13.5) |
| ≤ $40,000 | 12 (21.5) | 11 (20.0) | 23 (20.7) |
| > $40,000 | 38 (67.9) | 35 (63.6) | 73 (65.8) |
| Taking oral medication, No. (%) | 41 (70.7) | 37 (63.8) | 78 (67.2) |
| Taking insulin, No. (%) | 16 (27.6) | 10 (17.2) | 26 (22.4) |
| Body Mass Index | 34.8 ± 5.5 | 36.8 ± 7.1 | 35.8 ± 6.4 |
| HbA1c (%) | 7.30 ± 1.5 | 6.90 ± 1.3 | 7.11 ± 1.4 |
Frequencies may not sum to group totals due to unreported data No, Number; HbA1c, glycosylated hemoglobin.
Figure 2.
Participant flow through the study: The CONSORT diagram
Changes in Self-Efficacy and Self-Regulation
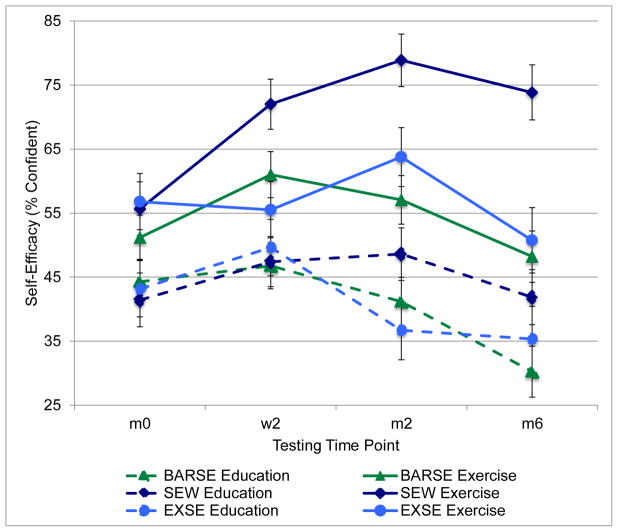
Trajectories of self-efficacy showed significant group by time differences. An overall repeated measures MANOVA (2x3x3) with walking, barriers, and exercise self-efficacy revealed significant time [F(9,80) = 8.446, p < .001, η2 = .49] and group by time effects [F(9,80) = 2.758, p < .01, η2 = .24]. Subsequently, each efficacy measure was analyzed separately using repeated measures analysis of variance or covariance (Figure 4). As walking self-efficacy baseline values were higher in the intervention group (p < .05), repeated measures analysis of covariance, controlling for baseline efficacy values, results indicated significant effects of group [F(1,81) = 32.666, p < .01, η2 = .29] with higher efficacy values in the exercise group at week two [β = 22.6, p < .01], month two [β = 25.5, p < .01], and month six [β = 23.2, p < .01]. For barriers efficacy, a time by group [F(3,81) = 2.691, p < .05, η2 = .09] interaction emerged. Further examination of these results revealed similar patterns in both groups with a between-subjects group effect [F(1,83) = 9.899, p < .01, η2 = .11] where efficacy increased in the intervention exercise group at week two [β = 15.3, p < .01]. Both groups showed similar declining efficacy trajectories thereafter with exercise group efficacy remaining higher compared to the education group at month two [β = 16.5, p < .01] and month six [β = 18.0, p < .01].
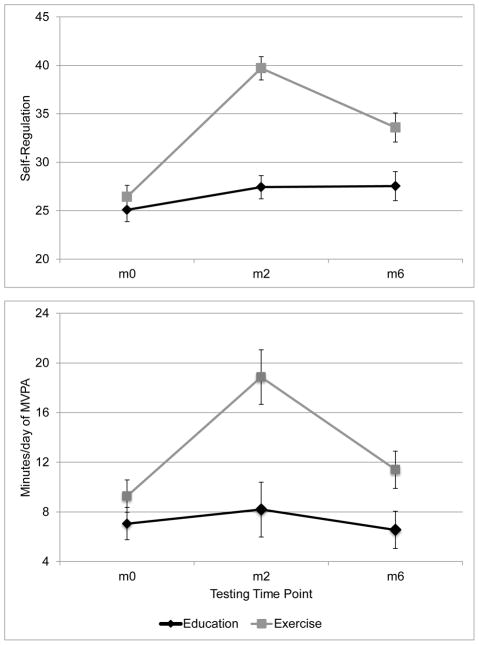
Figure 4.
Trajectories of self-regulation and physical activity by group
Within-subjects analysis on exercise self-efficacy revealed a quadratic time effect [F(1,88) = 14.60, p < .001, η2 = .14] and a cubic time by group effect [F(1,88) = 3.748, p = .06, η2 = .04]. Repeated measures ANCOVA conducted indicated an overall between-subjects group effect [F(1,82) = 7.869, p < .01, η2 = .09] with group differences at week two [β = 10.2, p < .05] and month two [β = 19.2, p < .01] indicating higher exercise efficacy in the exercise group. A series of within-group paired t-tests was used to determine short- and long-term intervention effects and calculate effect sizes for each efficacy measure (see Table 4).
Table 4.
Short- and long-term intervention effects on self-regulation, self-efficacy, and physical activity compared to baseline
| M0 M(SE) |
W2 M(SE) |
d | p | M2 M(SE) |
d | p | M6 M(SE) |
d | p | ||
|---|---|---|---|---|---|---|---|---|---|---|---|
| SEW | Exercise | 54.8(4.1) | 74.0(3.5) | .68 | <.001 | 80.6(3.3) | .96 | <.001 | 75.3(43.6) | .74 | <.001 |
| Education | 41.6(4.0) | 46.4(3.9) | .16 | .204 | 45.9(4.6) | .14 | .107 | 42.1(4.9) | .02 | .678 | |
| BARSE | Exercise | 52.8(3.3) | 62.9(3.3) | .42 | <.001 | 57.8(3.9) | .20 | .190 | 48.8(4.2) | −.15 | .413 |
| Education | 45.1(3.5) | 48.4(3.6) | .25 | .182 | 39.7(3.7) | −.20 | .155 | 29.6(3.5) | −.40 | <.001 | |
| EXSE | Exercise | 58.3(4.1) | 66.2(4.1) | .26 | .031 | 67.8(4.2) | .32 | .028 | 57.2(4.7) | −.04 | .974 |
| Education | 41.0(4.4) | 48.1(4.3) | .22 | .067 | 39.2(4.7) | −.05 | .420 | 38.2(5.5) | −.08 | .356 | |
| Self- Regulation | Exercise | 26.4(1.2) | -- | -- | -- | 40.1(1.5) | 1.48 | <.001 | 33.6(1.7) | .73 | <.001 |
| Education | 25.7(1.2) | 27.0 (1.3) | .15 | .309 | 27.5 (1.4) | .21 | .093 | ||||
| Minutes of MVPA per day | Exercise | 9.2(1.4) | -- | -- | -- | 20.2(2.5) | .76 | <.001 | 13.2(1.8) | .35 | .046 |
| Education | 7.4(1.3) | 8.3(1.3) | .10 | .517 | 7.6(1.3) | .02 | .755 |
SEW = Walking Self-Efficacy; BARSE = Barriers Self-Efficacy; EXSE = Exercise Self-Efficacy; MVPA = Moderate-to-Vigorous Physical Activity
Repeated measures analysis of variance in self-regulation revealed an overall both time group by time [F(2,83) = 10.396, p < .01, η2 = .21] effect that was quadratic in nature [F(1,84) = 16.823, p < .01, η2 = .17]. Self-regulation increased substantially in the exercise group at month two compared to the education group [β = 11.3, p < .01] and then declined slightly at month six while still maintaining group differences [β = 6.6, p < .01]. Self-regulation in the education group trended towards a slight increase at month six (p = .09). Figure 3 illustrates trajectories of self-regulation by group. Table 4 displays short- and long-term intervention effects on self-regulation.
Figure 3.
Trajectories of self-efficacy by group
Trajectories of Physical Activity
The hypothesis that increases in physical activity would be observed post-intervention and sustained at follow-up in the intervention group compared to the education control was partially supported. The results from the repeated measures (2x3) ANOVA predicting trajectory of minutes of moderate-to-vigorous physical activity per day measured by accelerometer indicated an overall time by condition interaction [F(2,83) = 5.777, p < .05, η2 = .12] while controlling for age, gender, and income. Intra-individual analysis revealed similar results [F(1.644,138.08) = 7.428, p < .01, η2 = .08] (using Greenhouse-Geisser estimation, as the dependent variable was not normally distributed). Further evaluation of these results revealed a quadratic trajectory for the accelerometer by group interaction [F(1,84) = 9.206, p < .01, η2 = .10] where a large increase was observed at month two, in the exercise group, followed by a decline at month six. On average, intervention participants increased from 9 minutes/day of moderate-to-vigorous physical activity at baseline to 20 minutes/day at month two (d = .76; p <.01) declining to approximately 13 minutes/day at month six (d = .35; p <.01). On average, control participants maintained activity level around 7 minutes/day across time (see Figure 4). Activity level by group was significantly different at month two [β = 10.7, p < .01] and month six [β = 4.8, p < .05] (see Table 4).
Baseline, week two, and month two self-efficacy and baseline and month two self-regulation were associated with physical activity at follow-up controlling for age, gender, income, baseline glycosylated hemoglobin, and baseline physical activity (see Table 5). Changes in exercise and barriers self-efficacy as well as self-regulation from baseline to post-intervention (m2 – m0/SDm2 – m0) were associated with physical activity immediately post-intervention (r = .243, .263, .314, respectively; p < .05). Changes in exercise efficacy, barriers efficacy, and self-regulation over the eight-week intervention period were associated with change in physical activity during the four-month follow-up (r = −.248, −.269, −.316, respectively; p < .05) such that those who increased more in efficacy and self-regulation during the intervention period experienced less decline in physical activity during the follow-up period.
Table 5.
Partial correlations among mid- and post-intervention self-efficacy, post-intervention self-regulation, and physical activity immediately post-intervention and at a four-month follow-up
| 1. | 2. | 3. | 4. | 5. | 6. | 7. | 8. | 9. | |
|---|---|---|---|---|---|---|---|---|---|
| 1. EXSE w2 | -- | ||||||||
| 2. SEW w2 | .587** | -- | |||||||
| 3. BARSE w2 | .695** | .504** | -- | ||||||
| 4. EXSE m2 | .585** | .633** | .549** | -- | |||||
| 5. SEW m2 | .423** | .776** | .405** | .656** | -- | ||||
| 6. BARSE m2 | .519** | .404** | .605** | .653** | .359** | -- | |||
| 7. Self- regulation m2 | .398** | .479** | .384** | .629** | .472** | .622** | -- | ||
| 8. Physical activity (MVPA) m2 | .350** | .359** | .345** | .455** | .314** | .365** | .415** | -- | |
| 9. Physical activity (MVPA) m6 | .273* | .349** | .242* | .282* | .236* | .248* | .126 | .376** | -- |
Note: partial correlations controlled for age, gender, baseline HbA1c, income, and baseline physical activity; MVPA = minutes of Moderate-to-Vigorous Physical Activity;
p < .05,
p < .01
Regression analysis indicated that standardized change in self-regulation (m2 – m0/SDm2 –m0) was a significant predictor of physical activity at month two [F(9,94) = 6.791, p < .001; adjusted R2 = .229; βm2 = .262, p < .01], physical activity at month six [F(9,87) = 9.416, p < .001; adjusted R2 = .469; βm6 = .233, p < .01] and change in physical activity between month two and month six [F(9,87) = 2.198, p < .05; adjusted R2 = .202; βm6-m2 = −.236, p < .05]. Baseline physical activity was an additional predictor of physical activity at month two and month six (βm2 = .572, βm6 = .709, p < .01). The regression model that best fit the data indicated. Neither self-efficacy nor any demographic factors emerged as significant predictors.
DISCUSSION
The purpose of this study was to test a whether a brief intervention focusing on self-efficacy and self-regulation would be successful in increasing physical activity in older adults with metabolic disease. Overall, the results provide some preliminary evidence that a short, titrated 8-week physical activity intervention leads to small improvements in physical activity levels in older adults with type 2 diabetes both post-intervention and four months later, compared to an education control group.
Intervention Effects on Physical Activity
Our hypothesis that the intervention would be effective at increasing physical activity levels was partially supported by the data. Intervention participants more than doubled minutes/day spent in moderate-to-vigorous physical activity by month two. However, intervention group physical activity declined post-intervention resulting in a mere few minutes/day more than baseline levels at month six, indicating that the intervention was not effective in maintaining physical activity. However, the intervention resulted in more physical activity compared to the education control at months two and six. These results are not surprising given that improving adherence to self-administered therapies has proven very difficult (Lerman, 2005; Wing et al., 2001). Adherence to short-term therapies appears easier compared to long-term therapies and adherence inversely relates to behavioral complexity of the behavior (Lerman, 2005). Regular physical activity engagement constitutes a complex health behavior, which requires sustained effort and motivation in the face of a multitude of barriers and few immediate rewards (Brawley, Rejeski, & King, 2003).
Given the difficulty in increasing physical activity adherence in this population (Nwasuruba et al., 2007), the modest improvements observed in this pilot trial are encouraging. Previous research in type 2 diabetes has largely focused on the efficacy of physical activity as a therapeutic treatment. Long-term adherence to physical activity has been examined in other special populations, including older adults with functional impairment and cancer survivors. Follow-up data from the LIFE-P study suggest that two-years after completing a 12-month intensive lifestyle intervention, participants still reported more moderate-intensity physical activity, enough to meet public health recommendations, though significantly lower than reported values immediately post-intervention (Rejeski et al., 2009). Rogers and colleagues (2009a) conducted a brief (3-month) physical activity adherence intervention in breast cancer survivors where participants exhibited sustained physical activity levels three months after study completion (Rogers et al., 2009b). While six months is not considered long-term adherence, Rogers and colleagues’ intervention elicited effects lasting as long as the intervention period. Both of these studies structured the intervention period into three phases: adoption, transition, and maintenance. In the LIFE-P study, the adoption, transition, and maintenance phases were eight, 15, and 27 weeks, respectively, compared to two, four, and six weeks in Rogers and colleagues’ intervention (The LIFE Study Investigators, 2006; Rogers et al., 2009a, 2009b). With the shorter intervention period, Rogers and colleagues structured adoption and transition to occur somewhat simultaneously, which may better allow participants to establish independent exercise.
These studies provide support that targeted interventions can be effective in increasing physical activity adherence in populations for whom physical activity adoption is crucial, yet difficult. It is important to note that both of these interventions, despite their distinctly different intervention lengths, were longer than intervention period presented here. In comparison, our intervention design consisted of an overlapped six weeks for adoption and transition and two weeks for maintenance. The intervention period was too short for long-lasting behavior change to occur. Adoption of new complex behaviors, such as physical activity, appear to require at least two months (Lally, Van Jaarsveld, Potts, & Wardle, 2010) and can take as long as six months before more habitual, automatic mechanisms sustain the behavior (Dishman, 1982). Additionally, the withdrawal of communication with the research team, by week eight, may have been too fast for some participants.
However, these data suggest that a brief, titrated intervention can increase short-term physical activity adherence in older adults with type 2 diabetes, extending physical activity adherence trials to a new population. As the intervention period of eight weeks was too short to elicit sustained results, further research is needed to test whether effects can be enhanced with minor intervention changes, such as extending the maintenance phase.
Intervention Effects on Self-Efficacy and Self-Regulation
The intervention had its strongest effects on self-efficacy. The intervention group experienced higher self-efficacy compared to the education group in all three types of efficacy assessed, with walking self-efficacy showing the largest group effect. Trajectories of self-efficacy increased during the intervention and had decreased by the follow-up. At the start of a program, participants can experience a boost of encouragement, motivation, and confidence. Previous research has shown that physical-activity-related efficacy tends to decline after completion of a formal program, once participants are left to continue exercising independently (McAuley et al., 2011).
Examining the efficacy results separately, we observed that the walking self-efficacy results mirror previous findings where walking self-efficacy of individuals participating in an exercise program increased at the beginning of a physical activity program and then exhibited slight declines six months later (McAuley et al., 2011). Considering the contribution of past experience to self-efficacy ideation, the on-site walking component of the behavioral intervention was likely crucial to the increase in walking self-efficacy. Interestingly, barrier efficacy beliefs peaked at week two, when the intervention participants were still benefiting from social support and social modeling from their cohort and research staff and had yet to attempt exercising independently. Barriers self-efficacy declined thereafter, presumably due to participants experiencing more barriers as they started integrating physical activity into their lives without the structured on-site program. When required to exercise independently, and then subsequently losing contact with the research team, efficacy decreased. Despite declines in self-efficacy by month six, efficacy levels in the intervention group were higher than those in the education group at month six. Overall, these data suggest that being in the intervention group had significant, positive effects on self-efficacy.
Participating in physical activity interventions does not guarantee improvements in exercise self-efficacy (McAuley, Jerome, Marquez, Elavsky, & Blissmer, 2003; Moore et al., 2006). Indeed, McAuley and Mihalko (1998) suggest that sedentary older adults may not have enough salient experience to accurately formulate beliefs of exercise self-efficacy which may explain declines in self-efficacy during program participation. McAuley and colleagues (2011) found support for this hypothesis such that older adults appeared to overestimate exercise efficacy before starting a physical activity program, which was followed by a lower efficacy adjustment three weeks later after actual exposure to regular physical activity. Contrary to those findings, these data indicate an increase in efficacy in the physical activity group at week two. It is possible that assessing efficacy at two weeks into the trial was too early for the excitement of a new program to have faded. The emphasis on learning self-regulatory strategies and boosting self-efficacy in the intervention group workshops may have been factors related to observed increases in self-efficacy.
As hypothesized, self-regulation increased in the intervention group at month two and, despite some declines at month six, remained higher compared to the education group. Self-regulation is crucial to goal-directed behavior. Behavior change, such as starting physical activity, is inherently goal-directed and cannot rely on habitual responses but instead must involve the development and use of self-regulatory skills (Bandura, 2005).
Self-Efficacy, Self-Regulation, and Physical Activity
As hypothesized, change in self-efficacy and self-regulation were associated with physical activity post-intervention. The literature suggests that self-efficacy and self-regulation are determinants of physical activity behavior and clearly both are essential constructs for successful behavior change (Bandura, 1986, 2005; McAuley et al., 2011; McAuley & Blissmer, 2000; McAuley & Mihalko, 1998). However, only self-regulation was correlated with physical activity at follow-up four months later. Specifically, the subscale of relapse prevention, which involves a priori identification and trouble-shooting of potential obstacles, was associated with physical activity at follow-up. It could be that self-regulatory strategies are more involved in longer-term maintenance of behavior change. Indeed, health habits are not changed through willpower alone, but through self-management requiring motivational and self-regulatory ability and skills (Bandura, 2005). There may be differential temporal influences of self-efficacy and self-regulation in the transition of behavioral adoption to habitual maintenance. Dishman and colleagues (2005) have proposed that self-regulatory strategies mediate the relationship between self-efficacy and physical activity. Further research is warranted to specifically test the temporal influence of social cognitive constructs.
Study Strengths & Limitations
Several study strengths and limitations are worth noting. The sample was fairly homogenous in terms of education, income, and ethnicity. However, the sample’s African American racial representation (13.8%) was higher compared to the local population (12.7%). The percentage of Asians in the sample was low, partially due to low local representation and partially due to low rates of type 2 diabetes in Asians compared to Whites or African Americans (McBean, Gilbertson, Li, & Collins, 2004). English fluently as an inclusion criterion for study participation resulted in lower reach to Latino and Hispanic individuals. The education group provided a better comparison than standard “usual care” control groups, but was not a true attention control. Therefore, caution in interpreting comparative results is warranted. The use of accelerometry, compared to subjective physical activity questionnaires, allows objective conclusions concerning intervention efficacy. Participant retention was satisfactory, with 88.8% completing post-intervention testing and 81.0% completing the six-month follow-up testing. Attendance to group workshops and on-site exercise was high in those participants who completed the intervention period. Low attrition is likely related to the relatively short length of the study. It is important to note that almost 30% (n = 90) of individuals screened for eligibility declined to participate due to lack of interest. The small number of exclusionary criteria for this trial made more individuals eligible. Uninterested individuals mentioned transportation difficulties, unwillingness to come on campus, upcoming extended travel plans (e.g. winter months in warmer climates), or not being interested in the exercise times offered (often mornings). However, most individuals who were marked as “not interested” simply never returned calls/emails from the research team after the initial contact which makes it impossible to truly know why they chose not to participate, information that would be useful for translation of this research.
Conclusions & Future Directions
The development and implementation of effective strategies to enhance adherence to physical activity recommendations is crucial for disease control and well-being in older adults with type 2 diabetes. However, physical activity adherence remains both an individual and public health challenge. This study’s brief, titrated intervention design may be practical and feasible for public health, medical, or community health professionals to implement. Results indicate that the intervention was successful in eliciting small increases in physical activity four months later. The small improvement that remained at month six, approximately two minutes/day in magnitude, is clearly insufficient to elicit significant health benefits. While intervention brevity increases feasibility, interventions lacking long-term efficacy are unhelpful no matter how feasible. Future research testing a slightly longer intervention period with follow-up support could establish an intervention protocol with longer-term results while retaining high feasibility. Strategies to promote maintenance such as additional workshops, e-health communications, or follow-up visits or phone with exercise leader should be explored. This study provides a foundation to build future interventions with the ultimate goal of improving long-term physical activity adherence while remaining logistically feasible for implementation in medical or community programming.
Acknowledgments
Research was funded by: National Institute on Aging: F31 AG042232, R01 AG0200118, 5T32AG023480-10; and by the Shahid Khan and Ann Carlson Khan Endowed Professorship.
Footnotes
Authors declare no conflict of interest.
Authors have no financial disclosures.
Note: Research was conducted at the University of Illinois at Urbana-Champaign.
INFORMED CONSENT
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2000. Informed consent was obtained from all participants for being included in the study.
Funding sources had no involvement in research design; data collection, analysis or interpretation; manuscript writing; or decision to submit for publication.
References
- Bandura A. Social foundations of thought and action: A social cognitive theory. New Jersey: Prentice Hall; 1986. [Google Scholar]
- Bandura A. Self-Efficacy: The exercise of control. New York: Freeman; 1997. [Google Scholar]
- Bandura A. The primacy of self-regulation in health promotion. Applied Psychology. 2005;54(2):245–54. [Google Scholar]
- Brawley LR, Rejeski WJ, King AC. Promoting physical activity for older adults: The challenges for changing behavior. American Journal of Preventive Medicine. 2003;25(3Sii):172–83. doi: 10.1016/s0749-3797(03)00182-x. [DOI] [PubMed] [Google Scholar]
- Centers for Disease Control and Prevention. National diabetes fact sheet: national estimates and general information on diabetes and prediabetes in the United States, 2011. 2011 Retrieved from http://www.cdc.gov/diabetes/pubs/pdf/ndfs_2011.pdf.
- Centers for Disease Control and Prevention. Press Release: One in five adults meets overall physical activity guidelines. 2013 Retrieved from http://www.cdc.gov/media/releases/2013/p0502-physical-activity.html.
- Copeland JL. Accelerometer assessment of physical activity in active, healthy older adults. Journal of Aging and Physical Activity. 2009;17:17–30. doi: 10.1123/japa.17.1.17. [DOI] [PubMed] [Google Scholar]
- de Jager CA, Budge MM, Clarke R. Utility of TICS-M for the assessment of cognitive function in older adults. International Journal of Geriatric Psychiatry. 2003;18:318–24. doi: 10.1002/gps.830. [DOI] [PubMed] [Google Scholar]
- Diabetes Prevention Program Research Group. Reduction of the incidence of type 2 diabetes with lifestyle intervention or metformin. New England Journal of Medicine. 2002;346(6):393–403. doi: 10.1056/NEJMoa012512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dishman RK. Compliance/adherence in health-related exercise. Health Psychology. 1982;1:237–67. [Google Scholar]
- Dishman RK, Motl RW, Sallis JF, Dunn AL, Birnbaum AS, Welk GJ, Jobe JB. Self-management strategies mediate self-efficacy and physical activity. American Journal of Preventive Medicine. 2005;29(1):10–8. doi: 10.1016/j.amepre.2005.03.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duncan GE, Perri MG, Theriaque DW, Hutson AD, Eckel RH, Stacpoole PW. Exercise training, without weight loss, increases insulin sensitivity and postherparin plasma lipase activity in previously sedentary adults. Diabetes Care. 2003;26:557–62. doi: 10.2337/diacare.26.3.557. [DOI] [PubMed] [Google Scholar]
- Freedson PS, Melanson E, Sirand J. Calibration of the computer science and applications, inc. accelerometer. Medicine & Science in Sports & Exercise. 1998;30:777–81. doi: 10.1097/00005768-199805000-00021. [DOI] [PubMed] [Google Scholar]
- Grundy SM, Cleeman JI, Daniels SR, Donato KA, Eckel RH, Franklin BA, Costa F. Diagnosis and management of the metabolic syndrome: An American Heart Association/National Heart, Lung, and Blood Institute scientific statement. Circulation. 2005;112:2735–52. doi: 10.1161/CIRCULATIONAHA.105.169404. [DOI] [PubMed] [Google Scholar]
- Jeon CY, Looken RP, Hu FB, van Dam RM. Physical activity of moderate intensity and risk of type 2 diabetes: A systematic review. Diabetes Care. 2007;30:744–52. doi: 10.2337/dc06-1842. [DOI] [PubMed] [Google Scholar]
- Lally P, Van Jaarsveld CHM, Potts HWW, Wardle J. How are habits formed: Modeling habit formation in the real world. European Journal of Social Psychology. 2010;40:998–1009. [Google Scholar]
- Lerman I. Adherence to treatment: The key for avoiding long-term complications of diabetes. Archives of Medical Research. 2005;36:300–6. doi: 10.1016/j.arcmed.2004.12.001. [DOI] [PubMed] [Google Scholar]
- Luciano E, Carneiro EM, Carvalho CR, Carvalheira JB, Peres SB, Reis MA, Velloso LA. Endurance training improves responsiveness to insulin and modulates insulin signaling transduction through the phosphatidylinositol 3-kinase/Akt-1 pathway. European Journal of Endocrinology. 2002;147:149–57. doi: 10.1530/eje.0.1470149. [DOI] [PubMed] [Google Scholar]
- Maes S, Karoly P. Self-regulation assessment and intervention in physical health and illness: A review. Applied Psychology. 2005;54(2):267–99. [Google Scholar]
- Mailey EL, Gothe NP, Wójcicki TR, Szabo AN, Olson EA, Mullen SP, McAuley E. Influence of allowable interruption period on estimates of accelerometer wear time and sedentary time in older adults. Journal of Aging and Physical Activity. 2014;22(2):255–60. doi: 10.1123/japa.2013-0021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Masse L, Fummeler B, Anderson C, Mathews C, Trost S, Catellier D, Treuth M. Accelerometer data reduction: A comparison of four reduction algorithms on select outcome variables. Medicine & Science in Sports & Exercise. 2005;37(11):S544–54. doi: 10.1249/01.mss.0000185674.09066.8a. [DOI] [PubMed] [Google Scholar]
- Matos A, Ropelle ER, Pauli JR, Frederico MJS, de Pinho RA, Velloso LA, De Souza CT. Acute exercise reverses TRB3 expression in the skeletal muscle and ameliorates whole body insulin sensitivity in diabetic mice. Acta Physiologica. 2010;198:61–9. doi: 10.1111/j.1748-1716.2009.02031.x. [DOI] [PubMed] [Google Scholar]
- McAuley E. The role of efficacy cognitions in the prediction of exercise behavior in middle-aged adults. Journal of Behavorial Medicine. 1992;15(1):65–88. doi: 10.1007/BF00848378. [DOI] [PubMed] [Google Scholar]
- McAuley E, Blissmer B. Self-efficacy determinants and consequences of physical activity. Exercise and Sport Science Reviews. 2000;28(2):85–88. [PubMed] [Google Scholar]
- McAuley E, Blissmer B, Katula J, Duncan TE, Mihalko SL. Physical activity, self-esteem, and self-efficacy relationships in older adults: A randomized controlled trial. Annals of Behavioral Medicine. 2000;22:131–139. doi: 10.1007/BF02895777. [DOI] [PubMed] [Google Scholar]
- McAuley E, Hall KS, Motl RW, White SM, Wójcicki TR, Hu L, Doerksen SE. Trajectory of declines in physical activity in community-dwelling older women: Social cognitive influences. Journals of Gerontology, Series B: Psychological Sciences. 2009;64B(5):543–50. doi: 10.1093/geronb/gbp049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McAuley E, Jerome GJ, Marquez DX, Elavsky S, Blissmer B. Exercise self-efficacy in older adults: Social, affective and behavioral influences. Annals of Behavioral Medicine. 2003;25:1–7. doi: 10.1207/S15324796ABM2501_01. [DOI] [PubMed] [Google Scholar]
- McAuley E, Lox C, Duncan TE. Long-term maintenance of exercise, self-efficacy, and physiologic change in older adults. Journals of Gerontology, Series B: Psychological Sciences. 1993;48(4):218–24. doi: 10.1093/geronj/48.4.p218. [DOI] [PubMed] [Google Scholar]
- McAuley E, Mailey EL, Mullen SP, Szabo AN, Wójckicki TR, White SM, Kramer AF. Growth trajectories of exercise self-efficacy in older adults: Influence of measures and initial status. Health Psychology. 2011;30(1):75–83. doi: 10.1037/a0021567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McAuley E, Mihalko SL. Measuring exercise-related self-efficacy. In: Duda JL, editor. Advances in sport and exercise psychology measurement. West Virginia: Fitness Information Technology; 1998. [Google Scholar]
- McBean AM, Gilbertson DT, Li S, Collins AJ. Differences in diabetes prevalence, incidence, and mortality among the elderly of four racial/ethnic groups: White, Blacks, Hispanics, and Asians. Diabetes Care. 2004;27:2317–24. doi: 10.2337/diacare.27.10.2317. [DOI] [PubMed] [Google Scholar]
- Moore SM, Charvat JM, Gordon NH, Pashkow F, Ribisl P, Roberts BL, Rocco M. Effects of a CHANGE intervention to increase exercise maintenance following cardiac events. Annals of Behavioral Medicine. 2006;31:53–62. doi: 10.1207/s15324796abm3101_9. [DOI] [PubMed] [Google Scholar]
- Morrato EH, Hill JO, Wyatt HR, Ghushchyan V, Sullivan PW. Physical activity in the U.S. adults with diabetes and at risk for developing diabetes, 2003. Diabetes Care. 2007;30(2):203–9. doi: 10.2337/dc06-1128. [DOI] [PubMed] [Google Scholar]
- Nwasuruba CK, Khan M, Egede LE. Racial/ethnic differences in multiples self-care behaviors in adults with diabetes. Journal of General Internal Medicine. 2007;22:115–20. doi: 10.1007/s11606-007-0120-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rejeski WJ, Marsh AP, Chmelo E, Prescott AJ, Dobrosielski M, Walkup MP, Kritchevsky S. The lifestyle interventions and independence for elders pilot (LIFE-P): 2-year follow-up. Journals of Gerontology, Series A: Biological Sciences. 2009;64A(4):462–7. doi: 10.1093/gerona/gln041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Resnick H, Harris M, Brock D, Harris T. American Diabetes Association diabetes diagnostic criteria, advancing age and cardiovascular disease risk profiles. Diabetes Care. 2000;23:176–80. doi: 10.2337/diacare.23.2.176. [DOI] [PubMed] [Google Scholar]
- Rogers LQ, Hopkins-Price P, Vicari S, Markwell S, Pamenter R, Courneya KS, Verhulst S. Physical activity and health outcomes three months after completing a physical activity behavior change intervention: Persistent and delayed effects. Cancer Epidemiology, Biomarkers, & Prevention. 2009a;18:1410–8. doi: 10.1158/1055-9965.EPI-08-1045. [DOI] [PubMed] [Google Scholar]
- Rogers LQ, Hopkins-Price P, Vicari S, Pamenter R, Courneya KS, Markwell S, Lowy M. A randomized trial to increase physical activity in breast cancer survivors. Medicine & Science in Sports & Exercise. 2009b;41(4):935–6. doi: 10.1249/MSS.0b013e31818e0e1b. [DOI] [PubMed] [Google Scholar]
- Sigal RJ, Kenny GP, Wasserman DH, Castaneda-Sceppa C, White RD. Physical activity/exercise and type 2 diabetes: A consensus statement from the American Diabetes Association. Diabetes Care. 2006;29(6):1433–8. doi: 10.2337/dc06-9910. [DOI] [PubMed] [Google Scholar]
- The LIFE Study Investigators. Effects of a physical activity intervention on measures of physical performance: Results of the lifestyle interventions and independence for elders pilot (LIFE-P) study. Journals of Gerontology, Series A: Biological Sciences. 2006;61A(11):1157–65. doi: 10.1093/gerona/61.11.1157. [DOI] [PubMed] [Google Scholar]
- Touati S, Meziri F, Devaux S, Berthelot A, Touyz R, Laurent P. Exercise reverses metabolic syndrome in high-fat diet-induced obese rats. Medicine & Science in Sports & Exercise. 2011;43(3):398–407. doi: 10.1249/MSS.0b013e3181eeb12d. [DOI] [PubMed] [Google Scholar]
- Umstattd MR, Motl RW, Wilcox S, Saunders R, Watford M. Measuring physical activity self-regulation strategies in older adults. Journal of Physical Activity and Health. 2009;6:S105–12. doi: 10.1123/jpah.6.s1.s105. [DOI] [PubMed] [Google Scholar]
- Wing RR, Goldstein MG, Acton KJ, Birch LL, Jakicic JM, Sallis JF, Surwit RS. Behavioral science research in diabetes: Lifestyle changes related to obesity, eating behavior, and physical activity. Diabetes Care. 2001;24:117–23. doi: 10.2337/diacare.24.1.117. [DOI] [PubMed] [Google Scholar]