Abstract
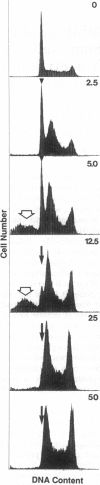
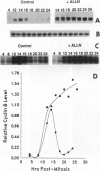
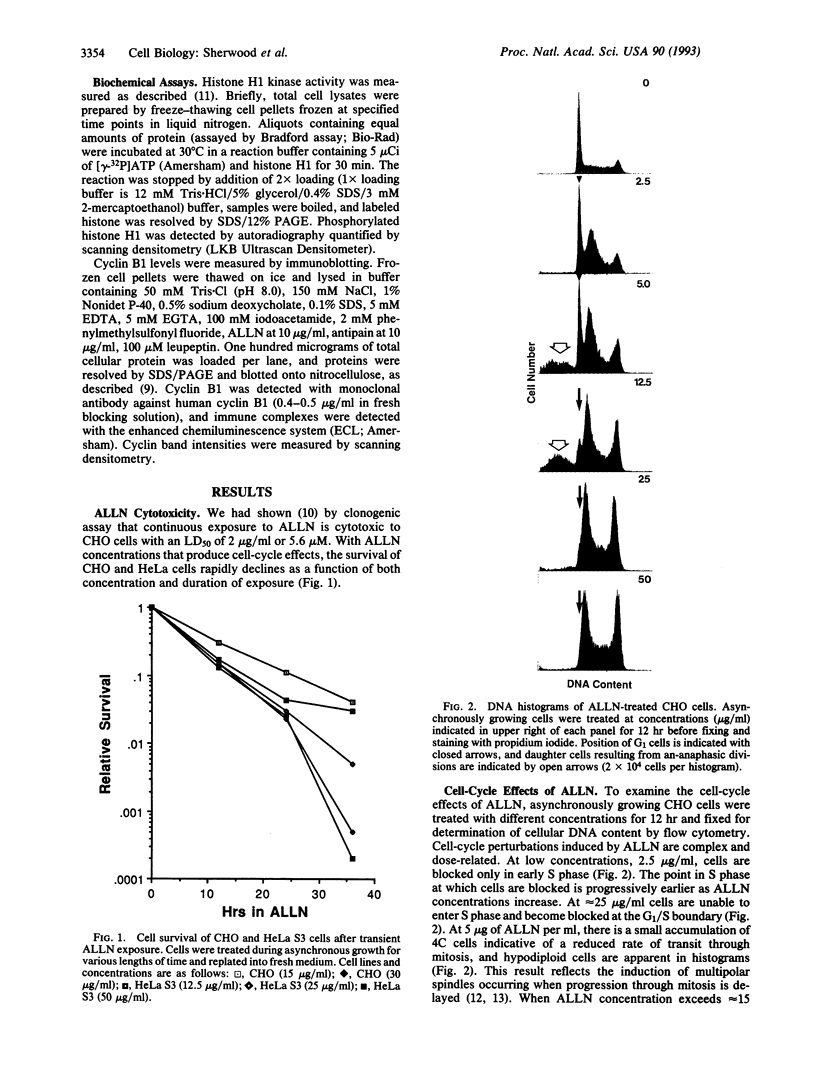
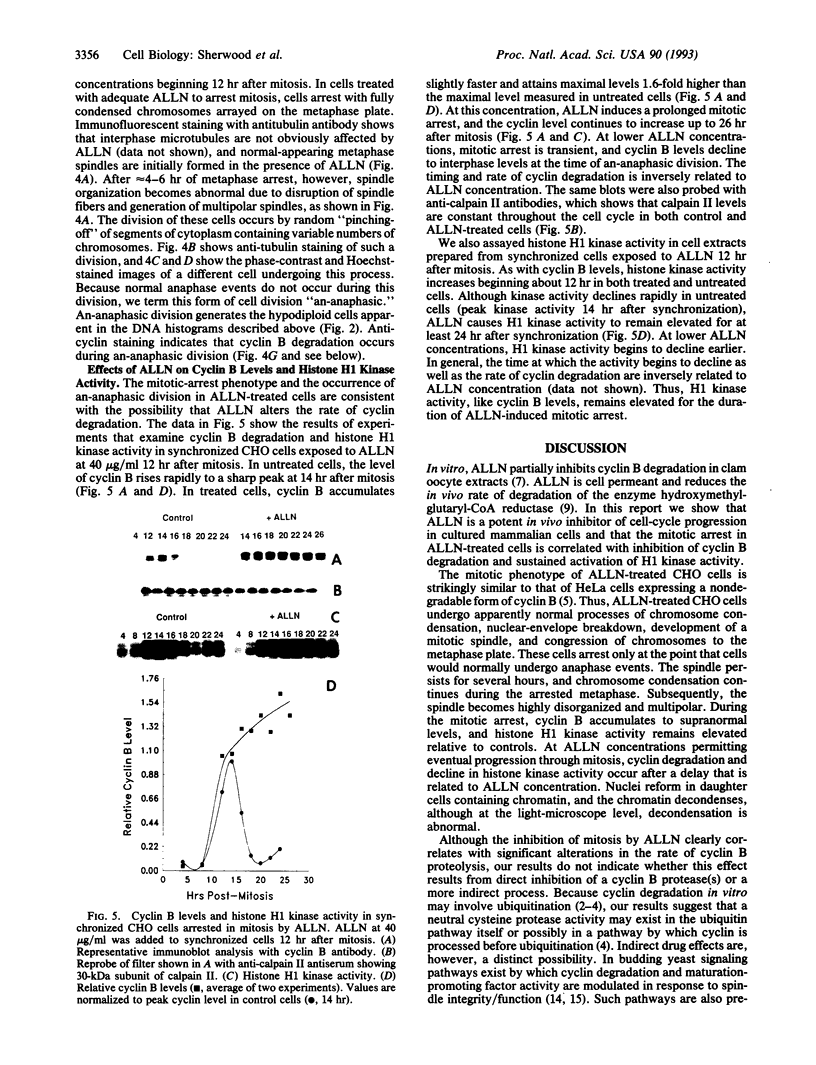
The cytotoxic neutral cysteine protease inhibitor N-acetylleucylleucylnorleucinal (ALLN) inhibits cell-cycle progression in CHO cells, affecting the G1/S and metaphase-anaphase transition points, as well as S phase. Mitotic arrest induced by ALLN is associated with the inhibition of cyclin B degradation. At mitosis-inhibiting concentrations of ALLN, cells undergo nuclear-envelope breakdown, spindle formation, chromosome condensation, and congression to the metaphase plate. However, normal anaphase events do not occur, and cells arrest in a metaphase configuration for a prolonged period. Steady-state levels of cyclin B increase to greater than normal mitotic levels, and cyclin B is not degraded for an extended period. Histone H1 kinase activity remains elevated during mitotic arrest. Duration of mitotic arrest depends on ALLN concentration; high concentrations (> 50 micrograms/ml) produce a prolonged mitotic arrest, whereas at lower concentrations, cells are transiently delayed through mitosis (up to 4-12 hr), after which they undergo aberrant cell division resulting in randomly sized daughter cells containing variable amounts of DNA. Cyclin B degradation fails to occur, and histone H1 kinase remains activated for the duration of mitotic arrest at all ALLN concentrations.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Dulić V., Lees E., Reed S. I. Association of human cyclin E with a periodic G1-S phase protein kinase. Science. 1992 Sep 25;257(5078):1958–1961. doi: 10.1126/science.1329201. [DOI] [PubMed] [Google Scholar]
- Gallant P., Nigg E. A. Cyclin B2 undergoes cell cycle-dependent nuclear translocation and, when expressed as a non-destructible mutant, causes mitotic arrest in HeLa cells. J Cell Biol. 1992 Apr;117(1):213–224. doi: 10.1083/jcb.117.1.213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Glotzer M., Murray A. W., Kirschner M. W. Cyclin is degraded by the ubiquitin pathway. Nature. 1991 Jan 10;349(6305):132–138. doi: 10.1038/349132a0. [DOI] [PubMed] [Google Scholar]
- Hershko A., Ciechanover A. The ubiquitin system for protein degradation. Annu Rev Biochem. 1992;61:761–807. doi: 10.1146/annurev.bi.61.070192.003553. [DOI] [PubMed] [Google Scholar]
- Hershko A., Ganoth D., Pehrson J., Palazzo R. E., Cohen L. H. Methylated ubiquitin inhibits cyclin degradation in clam embryo extracts. J Biol Chem. 1991 Sep 5;266(25):16376–16379. [PubMed] [Google Scholar]
- Hoyt M. A., Totis L., Roberts B. T. S. cerevisiae genes required for cell cycle arrest in response to loss of microtubule function. Cell. 1991 Aug 9;66(3):507–517. doi: 10.1016/0092-8674(81)90014-3. [DOI] [PubMed] [Google Scholar]
- Hunt T., Luca F. C., Ruderman J. V. The requirements for protein synthesis and degradation, and the control of destruction of cyclins A and B in the meiotic and mitotic cell cycles of the clam embryo. J Cell Biol. 1992 Feb;116(3):707–724. doi: 10.1083/jcb.116.3.707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Inoue S., Bar-Nun S., Roitelman J., Simoni R. D. Inhibition of degradation of 3-hydroxy-3-methylglutaryl-coenzyme A reductase in vivo by cysteine protease inhibitors. J Biol Chem. 1991 Jul 15;266(20):13311–13317. [PubMed] [Google Scholar]
- Keryer G., Ris H., Borisy G. G. Centriole distribution during tripolar mitosis in Chinese hamster ovary cells. J Cell Biol. 1984 Jun;98(6):2222–2229. doi: 10.1083/jcb.98.6.2222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kirschner M. The cell cycle then and now. Trends Biochem Sci. 1992 Aug;17(8):281–285. doi: 10.1016/0968-0004(92)90435-c. [DOI] [PubMed] [Google Scholar]
- Kung A. L., Sherwood S. W., Schimke R. T. Cell line-specific differences in the control of cell cycle progression in the absence of mitosis. Proc Natl Acad Sci U S A. 1990 Dec;87(24):9553–9557. doi: 10.1073/pnas.87.24.9553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lew D. J., Dulić V., Reed S. I. Isolation of three novel human cyclins by rescue of G1 cyclin (Cln) function in yeast. Cell. 1991 Sep 20;66(6):1197–1206. doi: 10.1016/0092-8674(91)90042-w. [DOI] [PubMed] [Google Scholar]
- Li R., Murray A. W. Feedback control of mitosis in budding yeast. Cell. 1991 Aug 9;66(3):519–531. doi: 10.1016/0092-8674(81)90015-5. [DOI] [PubMed] [Google Scholar]
- Luca F. C., Ruderman J. V. Control of programmed cyclin destruction in a cell-free system. J Cell Biol. 1989 Nov;109(5):1895–1909. doi: 10.1083/jcb.109.5.1895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Norbury C., Nurse P. Animal cell cycles and their control. Annu Rev Biochem. 1992;61:441–470. doi: 10.1146/annurev.bi.61.070192.002301. [DOI] [PubMed] [Google Scholar]
- Schollmeyer J. E. Calpain II involvement in mitosis. Science. 1988 May 13;240(4854):911–913. doi: 10.1126/science.2834825. [DOI] [PubMed] [Google Scholar]
- Sharma R. C., Inoue S., Roitelman J., Schimke R. T., Simoni R. D. Peptide transport by the multidrug resistance pump. J Biol Chem. 1992 Mar 25;267(9):5731–5734. [PubMed] [Google Scholar]
- Sluder G., Begg D. A. Experimental analysis of the reproduction of spindle poles. J Cell Sci. 1985 Jun;76:35–51. doi: 10.1242/jcs.76.1.35. [DOI] [PubMed] [Google Scholar]
- Watanabe N., Vande Woude G. F., Ikawa Y., Sagata N. Specific proteolysis of the c-mos proto-oncogene product by calpain on fertilization of Xenopus eggs. Nature. 1989 Nov 30;342(6249):505–511. doi: 10.1038/342505a0. [DOI] [PubMed] [Google Scholar]
- Whitaker M., Patel R. Calcium and cell cycle control. Development. 1990 Apr;108(4):525–542. doi: 10.1242/dev.108.4.525. [DOI] [PubMed] [Google Scholar]