Abstract
AIM
To compare the safety and efficacy of phacoemulsification and small incision cataract surgery (SICS) in patients with uveitic cataract.
METHODS
In a prospective, randomized multi-centric study, consecutive patients with uveitic cataract were randomized to receive phacoemulsification or manual SICS by either of two surgeons well versed with both the techniques. A minimum inflammation free period of 3mo (defined as less than 5 cells per high power field in anterior chamber) was a pre-requisite for eligibility for surgery. Superior scleral tunnel incisions were used for both techniques. Improvement in visual acuity post-operatively was the primary outcome measure and the rate of post-operative complications and surgical time were secondary outcome measures, respectively. Means of groups were compared using t-tests. One way analysis of variance (ANOVA) was used when there were more than two groups. Chi-square tests were used for proportions. Kaplan Meyer survival analysis was done and means for survival time was estimated at 95% confidence interval (CI). A P value of <0.05 was considered statistically significant.
RESULTS
One hundred and twenty-six of 139 patients (90.6%) completed the 6-month follow-up. Seven patients were lost in follow up and another six excluded due to either follow-up less than six months (n=1) or inability implant an intraocular lens (IOL) because of insufficient capsular support following posterior capsule rupture (n=5). There was significant improvement in vision after both the procedures (paired t-test; P<0.001). On first postoperative day, uncorrected distance visual acuity (UDVA) was 20/63 or better in 31 (47%) patients in Phaco group and 26 (43.3%) patients in SICS group (P=0.384). The mean surgically induced astigmatism (SIA) was 0.86±0.34 dioptres (D) in the phacoemulsification group and 1.16±0.28 D in SICS group. The difference between the groups was significant (t-test, P=0.002). At 6mo, corrected distance visual acuity (CDVA) was 20/60 or better in 60 (90.9%) patients in Phaco group and 53 (88.3%) in the manual SICS group (P=0.478). The mean surgical time was significantly shorter in the manual SICS group (10.8±2.9 versus 13.2±2.6min) (P<0.001). Oral prednisolone, 1 mg/kg body weight was given 7d prior to surgery, continued post-operatively and tapered according to the inflammatory response over 4-6wk in patients with previously documented macular edema, recurrent uveitis, chronic anterior uveitis and intermediate uveitis. Rate of complications like macular edema (Chi-square, P=0.459), persistent uveitis (Chi-square, P=0.289) and posterior capsule opacification (Chi-square, P=0.474) were comparable between both the groups.
CONCLUSION
Manual SICS and phacoemulsification do not differ significantly in complication rates and final CDVA outcomes. However, manual SICS is significantly faster. It may be the preferred technique in settings where surgical volume is high and access to phacoemulsification is limited, such as in eye camps. It may also be the appropriate technique for uveitic cataract under such circumstances.
Keywords: small incision cataract surgery, phacoemulsification, uveitis, corrected distance visual acuity, uncorrected distance visual acuity
INTRODUCTION
Cataract surgery in uveitis is different from that in age related cataract. Band shaped keratopathy and corneal deposits may decrease visibility during surgery; poor pupillary dilatation, bleeding from fragile vessels, pupillary membranes or presence of synechia pose surgical challenges. The optimal surgical procedure worldwide is not known as different uveitic syndromes may respond differently to surgery. Secondly, in cases with extensive posterior synechia and extremely dense nuclei, it may be prudent to enlarge the incision to facilitate manual nucleus extraction.
Most authors are of the opinion that with careful patient selection, diligent surgery and appropriate perioperative care, phacoemulsification with intraocular lens (IOL) implantation is safe and effective in most patients with uveitis[1],[2].
Manual small incision cataract surgery (SICS) has emerged as an alternative to phacoemulsification in settings with high surgical volume or regions with limited access, over the last decade; it is significantly faster, requires minimal instrumentation and can be performed in all settings[3],[4]. However, safety and efficacy of SICS in uveitis has not been established. An extensive review of literature did not reveal any published randomized controlled trials comparing phacoemulsification and manual SICS for uveitic cataracts (Medline search). A prospective, randomized clinical trial was done at three referral eye centres to compare risks, and postoperative outcomes of the two techniques in patients with uveitis.
SUBJECTS AND METHODS
The study was performed between November 2008 and April 2011 at Laser Eye Clinic, Noida, Santosh Medical College, Ghaziabad and Rotary Eye Hospital, Palampur, India. Informed consent was obtained from all subjects (or their guardians) based on Declaration of Helsinki. The research protocol was approved by the local ethics committee.
Inclusion and Exclusion Criteria
Patients with visually significant cataract and cataract impairing adequate visualization of posterior segment were included in the study[5]. A minimum inflammation free period of 3mo [defined as less than 5 cells per high power field in anterior chamber (AC)] was a pre-requisite for eligibility for surgery. However, in cases with vitritis, cells may persist even in inactive stage and cannot be completely eliminated. Exclusion criteria were less than 6mo follow-up, sulcus or sulcus-bag implantation of the IOL, posterior capsule rent, traumatic and subluxated cataracts and diabetes mellitus. In cases with adequate fundus visualization (due to less dense cataract) and possible preoperative diagnosis, uveitis associated fundal diseases likely to affect surgical outcome like epiretinal membrane (ERM), optic atrophy, and macular hole, were excluded from the study.
Preoperative Examination
The pre-operative protocol included routine investigations such as total and differential leucocyte counts, erythrocyte sedimentation rate and blood sugar levels, Mantoux test, chest X-ray, X-rays of the cervical spine and sacroiliac joints. Special investigations included rheumatoid factor, angiotensin converting enzyme essay, anti-nuclear factor, human leucocyte antigen typing and enzyme linked immunosorbant assay for toxoplasmosis, human immunodeficiency virus and tuberculosis. B-scan ultrasonography was performed in cases where funduscopy was not possible due to dense cataract. Intraocular pressure was measured with applanation tonometry.
The standardization of uveitis nomenclature (SUN) working group has recommended that the classification of uveitis entities should be on the basis of the location of the inflammation and not on the presence of structural complications. However, there are certain ambiguities regarding the terms “pars planitis”, “neuroretinitis” and “anterior and intermediate uveitis”[6]. Therefore, for the sake of simplicity and familiarity, in the present study, aqueous flare and cells were graded with a modified Hogan's technique[7]. Vitreous cells were graded with the classification proposed by Bloch-Michel and Nussenblatt[8].
Data collected include sex, age at surgery, aetiology of uveitis, pre-operative findings such as uncorrected distance visual acuity (UDVA), corrected distance visual acuity (CDVA), corticosteroid intake, frequency and duration of quiescence of inflammation before surgery, surgical time and duration of follow-up, post-operative vision and presence of complications.
Oral prednisolone, 1 mg/kg body weight was given 7d prior to surgery, continued post-operatively and tapered according to the inflammatory response over 4-6wk in patients with previously documented macular edema, recurrent uveitis, chronic anterior uveitis and intermediate uveitis[9].
Sample Size, Randomization and Masking
Sample size to compare mean difference in logMAR visual acuity was calculated using the website www.stat.ubc.ca. A pilot study was done on 14 subjects. The mean decrease in logMAR visual acuity in SICS group was 0.82 to 0.26 (0.56 logMAR units).The mean decrease in logMAR visual acuity in phacoemulsification group was 0.81 to 0.19 (0.62 logMAR units). The common standard deviation was 0.12. Assuming 1:1 randomization, alpha was set at 0.05 and power 80%. The sample size in each group was estimated to be 63.
The procedures were performed by either of two surgeons, well versed with both procedures. The allocation codes were generated by a DOS based computer software in the department of community ophthalmology. The allocation was concealed in green coloured envelopes that were opened by health care staff not involved in patient care, 10min prior to surgery. Patients were not informed about the type of procedure assigned. There were two independent investigators (Kaur A & Ali A, not a study surgeon), an ophthalmologist and an optometrist, who assessed vision, respectively. They were masked to the identity of the operating surgeons and the type of procedure.
Surgical Technique
Both surgeons used standardized and similar surgical techniques. Peribulbar anaesthesia was delivered. For SICS, a side port entry was made at the 10 o'clock position with a 20 G micro vitreo-retinal surgery (MVR) or a 15° angled knife. A 5.5-6 mm superior incision was made on the sclera, 2 mm posterior to the limbus. A self-sealing (tri-planar) sclero-corneal tunnel was made with a 2.2 mm bevel up crescent knife with adequate side pockets. AC was formed with ophthalmic viscosurgical device (2% hydroxypropyl methylcellulose). AC entry was performed and enlarged with a 2.8 mm keratome. Iris dilatation techniques such as synechiolysis, peeling of pupillary membrane or iris retractors, were used as needed due to insufficient pupil dilatation. Adjunctive Trypan blue-assisted continuous curvilinear capsulorrhexis (CCC) was created followed by hydrodissection with a 2 mL syringe attached to a 25 G cannula. The nucleus was rotated in the bag with a bent capsulotomy needle and prolapsed into the AC. The nucleus was delivered by the sandwich technique. Lens matter aspiration was performed with a Simcoe cannula. A 5.5 mm optic foldable acrylic IOL was implanted in the capsular bag. The self-sealing wound was left unsutured.
Phacoemulsification was performed with superior scleral tunnel incision using peribulbar anaesthesia. Two side-port corneal incisions were created 180 degrees apart with 20 G MVR knife. AC entry was fashioned with a 2.8 mm keratome. AC was maintained with 2% hydroxypropyl methylcellulose. Iris dilatation techniques such as synechiotomy, membrane peeling or iris hooks were used in case of non-dilating pupils. Trypan blue-assisted CCC was done as described previously. Cortical cleavage hydrodissection was done just below the anterior capsule rim and nucleus rotated in the bag. Phacoemulsification was performed with an Infinity vision system (Alcon, Inc.) using phaco-chop method. Cortical material was removed by bimanual irrigation/aspiration. A foldable square-edge acrylic IOL was implanted in the capsular bag using a cartridge. Paracentesis was hydrated.
A subconjunctival injection of gentamicin 20 mg and dexamethasone 4 mg was given at end of both the procedures.
Postoperative Care Routine
Post-operative care included topical moxifloxacin 0.5% 6 times a day, 1.0% atropine 3 times a day and 0.1% topical betamethasone hourly that was tapered over 10-12wk. Topical ketorolac tromethamine 0.4% was used selectively, 3 times a day in patients who developed cystoid macular edema; these patients also received topical corticosteroids.
Patients were followed-up on first, third and seventh postoperative days, then weekly for two weeks, monthly for two months and every three months thereafter. At each visit, UDVA/CDVA, aqueous cells and flare, and fundus details were recorded. In patients on preoperative systemic corticosteroids, dose was tapered over 4-6wk, depending on anti-inflammatory response and they further underwent monitoring of blood sugar, blood pressure and urine analysis.
Outcome Measures
The primary outcome measure was an improvement in visual acuity post-operatively. The secondary outcome measure was the rate of post-operative complications. Surgical time was recorded from initial side port entry to hydration of paracentesis.
Statistical Analysis
Statistical analysis was performed on intent to treat basis using SPSS software for windows (version 18, SPSS Inc.). One way analysis of variance (ANOVA) was used when there were more than two groups. Means of groups were compared using t-tests. The 95% confidence interval (CI) values were calculated for each mean. Chi-square tests were used for proportions. Kaplan Meyer survival analysis was done and means for survival time was estimated at 95% CI. A P value <0.05 was considered statistically significant.
RESULTS
One hundred and thirty-nine patients were enrolled in the study. Out of these, 75 (53.9%) patients were randomized to the Phaco group and 64 (46.0%) patients to SICS group. Seven patients were lost in follow up and another six excluded due to either follow-up less than six months (n=1) or inability implant an IOL because of insufficient capsular support following posterior capsule rupture (n=5). One hundred and twenty six of 139 patients (90.6%) completed the 6-month follow-up. The mean age (P=0.33), sex (P=0.86), anatomical type of uveitis (P=1.00) and follow up period (P=0.82) was comparable in both the groups. Table 1 shows the baseline characteristics of patients, etiology and type of uveitis in both the groups.
Table 1. Baseline parameters of patients.
| Parameter | Phaco group | SICS group |
| Age (a) | 53.3±8.8 | 54.8±7.6 |
| Sex (n) | ||
| M | 30 | 28 |
| F | 36 | 32 |
| Follow up (mo) | 12.02±3.6 | 11.87±4.0 |
| Surgical Time(min) | 13.2±2.6 | 10.8±2.9 |
| Uveitis type (n) | ||
| Anterior | 45 | 39 |
| Intermediate | 12 | 10 |
| Posterior | 3 | 4 |
| Pan uveitis | 6 | 7 |
| Etiology of uveitis n (%) | ||
| Idiopathic | 54 (82) | 50 (83) |
| FHC | 4 (6) | 3 (5) |
| AS | 2 (3) | 3 (5) |
| VKH | 1 (1.5) | 2 (3.3) |
| Toxoplasmosis | 3 (4.5) | 1 (1.7) |
| Sarcoidosis | 2 (3) | 1 (1.7) |
SICS: Small incision cataract surgery; FHC: Fuchs heterochromic cyclitis; AS: Ankylosing spondylitis; VKH: Vogt's Koyanagi Harada Syndrome.
Intraoperative Time
Preoperatively, 23 (34.8%) and 28 (46.7%) eyes had posterior synechia, 8 (12.1%) and 6 (10%) had pupillary membranes, 21 (31.8%) and 24(40%) eyes had miotic pupils in Phaco and SICS groups respectively. Consequently, 44 (66.7%) eyes in the Phaco group and 43 (71.7%) eyes in SICS group required intraoperative membrane peeling, synechiotomy or use of iris hooks to dilate the pupil. The mean surgical time was significantly shorter in the SICS group (10.8±2.9min) than in the Phaco group (13.2±2.6min) (P<0.001).
Visual Acuity
There was significant improvement in vision after both the procedures (paired t-test; P<0.001). On the first postoperative day, the mean logMAR UDVA in Phaco group (0.32±0.11) was comparable to SICS group (0.34±0.14); the difference was not statistically significant (P=0.384). On first postoperative day, UDVA was 20/63 or better in 31 (47%) patients in Phaco group and 26(43.3%) patients in SICS group (P=0.384).
The mean CDVA at 6mo (Figure 1) in Phaco group (0.28±0.16) was comparable to SICS group (0.29±0.18); the difference was not statistically significant (P=0.478). Table 2 shows percentage of patients at each level of CDVA at 6mo postoperatively. At six months, (60/90.9%) had a CDVA of 20/60 or better as compared to (53/88.3%) in SICS group. The mean surgically induced astigmatism (SIA) was 0.86±0.34 dioptres (D) in the Phaco group and 1.16±0.28 D in SICS group. The difference between the groups was significant (t-test, P=0.002).
Figure 1. A line diagram comparing preoperative, day 1 and final vision between both techniques.
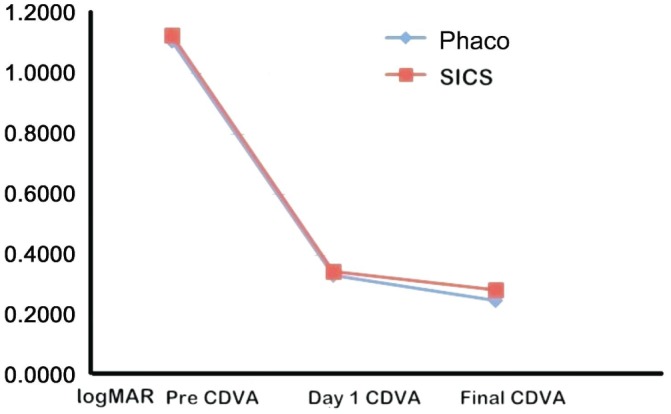
Table 2. Visual acuity 6mo postoperatively.
| logMAR CDVA | Phaco group | SICS group |
| 0.00-0.25 | 29 (43.9) | 26 (43.3) |
| 0.26-0.50 | 31 (47) | 27 (45) |
| 0.51-0.75 | 3 (4.5) | 4 (6.7) |
| 0.76-1.00 | 2 (3.0) | 3 (5.0) |
| 1.01-1.25 | 1 (1.5) | 0 (0) |
| 1.26-1.50 | 0 (0) | 0 (0) |
| Total | 66 (100) | 60 (100) |
n (%)
The cause of CDVA worse than 20/120 was macular edema in 2 cases, age-related macular degeneration in 2 cases and persistent uveitis in 2 cases.
Twenty one (31.8%) patients in Phaco group and 18 (30%) patients in SICS group received systemic corticosteroids. Patients on systemic corticosteroids (patients with previously documented macular edema, recurrent uveitis, chronic anterior uveitis and intermediate uveitis) had significantly better final visual acuity than patients who did not used pre-operative corticosteroids in both the groups ( t-test; P=0.101 and P=0.001, respectively). However, final CDVA was comparable in both groups.
The final visual outcome in Phaco and SICS groups was not significantly influenced by the anatomical location of uveitis (ANOVA; P=0.889 and P=0.671 respectively). The visual outcome was also not significantly influenced by the etiological type of uveitis (ANOVA; P=0.028 and P=0.062 respectively). Most cases of uveitis were idiopathic (Table 1).
There was a significant improvement in vision in patients requiring additional procedures like vitrectomy, ERM peeling, neodymium yttrium aluminium garnet laser capsulotomy in both the groups (t-test; P=0.005 and P=0.002, respectively).
Complications and Follow up
Both the groups did not differ significantly in the rate of postoperative complications (Table 3). On Kaplan Meyer survival analysis (Figure 2), means of survival time at 95% CI was 16.86±1.05 in Phaco group whereas in SICS group mean was 16.01±1.09, after censoring (Chi-square test; P=0.146). At any point of time, both groups were similar with respect to rate of complications.
Table 3. Rate of complications.
| Cause of reduced vision | Phaco group | SICS group | Chi-square P |
| PCO | 11 (16.7) | 9 (15) | 0.474 |
| Macular edema | 8 (12.1) | 7 (11.7) | 0.459 |
| ERM | 5 (7.6) | 3 (5) | 0.875 |
| ARMD | 4 (6) | 5 (8.3) | 0.889 |
| Persistent uveitis | 9 (13.6) | 10 (16.7) | 0.289 |
| Secondary glaucoma | 4 (6) | 3 (5) | 0.573 |
PCO: Posterior capsule opacification; ERM: Epiretinal membrane; ARMD: Age-related macular degeneration.
n (%)
Figure 2. Survival function graph showing that any point of time, both techniques were comparable in rates of complications.

Secondary Procedures
In both groups, additional procedures like Nd: YAG laser capsulotomy was done in 20 eyes (15.9%) after a quiet postoperative period of 3mo. Eight (6.3%) patients were referred to retina clinic for ERM peeling. Glaucoma filtering surgery was done in 4 (3.2%) cases.
DISCUSSION
To the best of our knowledge (Medline search), there are no randomized clinical trials comparing the surgical outcomes of manual SICS and phacoemulsification for uveitic cataract.
In most countries and settings, phacoemulsification is now the preferred technique of cataract surgery. However, the outcomes of cataract surgery are sometimes difficult to assess as eyes with different types of uveitis respond differently to surgery[10],[11]. In our experience, phacoemulsification is not always successful in fragmenting extremely dense nuclei with extensive posterior synechia; therefore, we routinely use scleral tunnel incisions for phacoemulsification of uveitic cataracts, as it may be necessary to enlarge the incision to facilitate manual nuclear extraction.
In rural and semi-urban settings in the sub-continent, patients with uveitis are often referred to tertiary care centres for further management, due to limited access to phacoemulsification. SICS requires minimal instrumentation and can be performed in all setups specially eye-care camps, obviating the need for referral to tertiary care centres.
Surgical speed and efficiency are of paramount importance in developing nations having a high burden of cataract. In the present study, SICS proved to be a much faster (10.8±2.9 versus 13.2±2.6min) surgical technique than phacoemulsification in patients with uveitis. Bhargava et al[12] recently conducted a retrospective study on 54 eyes in patients with uveitis. The authors found that SICS with posterior chamber IOL implantation is safe in most cataracts due to uveitis provided inflammation is well-controlled pre-operatively for at least 3mo. The mean surgical time for SICS in uveitic cataract was 10.2±3.8min. This was comparable to that of present study. In a randomized controlled trial by Cook et al[13], in a series of 200 patients in African subcontinent, there was no difference in the incidence of intraocular complications and uncorrected visual acuity on first postoperative day between phacoemulsification and SICS. In another prospective randomized study, SICS was significantly faster (8.8±3.4min) as compared to phacoemulsification (12.2±4.6min) for advanced white cataracts[14]. In Nepal, a study by Ruit et al[15] found excellent visual outcomes and low complication rates following phacoemulsification and manual SICS for advanced age related cataracts. The procedure time and costs of manual SICS was significantly less as compared to phacoemulsification. However, mean surgical time was significantly higher in both the groups as compared to age related cataract. Additional procedures like synechiotomy, iris hooks or pupillary membrane peeling, which are often required in patients with uveitis, accounts for the difference in procedure time.
In the present study, CDVA at 6mo was excellent with both the techniques; however, slightly higher number of patients in the Phaco group achieved a UDVA of 20/63 or better. This could be explained by the higher SIA in the SICS group due to larger incision. However, the percentage of patients who achieved CDVA of 20/60 or better was slightly lower in both the groups as compared to other studies. Table 4 shows the results in other studies that evaluated the safety and efficacy of phacoemulsification and SICS in patients with uveitis[11],[12],[16]–[19]. However, most of these studies were not prospective randomized comparisons and may have been influenced by selection bias; that is, a tendency towards scheduling affluent patients with immature or early sub-capsular cataracts towards either of the techniques.
Table 4. Surgical outcomes in reported series of patients with uveitis.
| Author | n | Year | Type | P | CDVA (%) | Complications |
| Bhargava et al[12] | 54 | 2014 | R | SICS | 93 | PCO (16.7), CME (14.8), persistent uveitis (9.2), ERM (7.4) |
| Kosker et al[11] | 55 | 2013 | P | Phaco | 94.5 | PCO, CME, recurrent uveitis (12.7), raised IOP (15.4). |
| Ram et al[16] | 108 | 2010 | R | Phaco | 92 | PCO (28.7), CME (21.3), recurrent uveitis (5.5), ERM (4.6), synechia (25). |
| Kawakuchi et al[17] | 131 | 2007 | R | Phaco | 85 | PCO(23.7), CME (6.1), IOP elevation (8.4), posterior synechia ( 6.1) |
| Hazari and Sangwan[18] | 106 | 2002 | P | P+ECCE | 87 | PCO (14.9), CME (20.9), persistent uveitis (23.9%) |
| Estafanous et al[19] | 39 | 2001 | R | Phaco | 95 | PCO (62), CME (33), recurrent uveitis (41), ERM (15). |
| Present study | 66&60 | 2014 | P | P+SICS | 90.9&88.3 | PCO (16.7 & 15), CME (12.1 & 15), persistent uveitis (13.6 & 16.7), glaucoma (6 &5), ERM (6 & 5) |
CDVA: Corrected distance visual acuity; SICS: Small incision cataract surgery; R: Retrospective; P: Prospective; P: Procedure; Phaco: Phacoemulsification; PCO: Posterior capsule opacification; CME: Cystoid macular edema; ERM: Epiretinal membrane; ECCE: Extracapsular cataract extraction.
There was no significant difference in the rate of complications with both the techniques. Survival function graph showing that any point of time, both techniques were comparable in rates of complications (Figure 2). Patients in the Phaco group had a slightly lower incidence of cystoid macular edema and persistent uveitis whereas the incidence of posterior capsule opacification was marginally lower in SICS group. In a retrospective analysis of a large series of patients, Haripriya et al[20] report excellent visual outcomes with low complication rates following phacoemulsification and manual SICS by surgeons well versed with both the techniques. In a meta-analysis of six randomized controlled trials comparing phacoemulsification and SICS for age-related cataract, Zhang et al[21] did not find any significant difference in terms of visual rehabilitation between both the techniques.
Limitations of our study are that endothelial cell counts were not compared between both the techniques due to unavailability of necessary equipment.
In conclusion, we found SICS to be a safe and effective alternative to phacoemulsification for patients with uveitis, with no significant difference in complications and final CDVA outcomes. SICS can be safely performed in settings with high surgical volume and access to phacoemulsification is limited such as in eye-camps. SICS may be the more appropriate technique of cataract surgery in patients with uveitis in such circumstances.
Acknowledgments
Conflicts of Interest: Bhargava R, None; Kumar P, None; Sharma SK, None; Kumar M, None; Kaur A, None.
REFERENCES
- 1.Murthy SI, Pappuru RR, Latha KM, Kamat S, Sangwan VS. Surgical management in patients with uveitis. Indian J Ophthalmol. 2013;61(6):284–290. doi: 10.4103/0301-4738.114103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Alió JL, Chipont E, BenEzra D, Fakhry MA, International Ocular Inflammation Society, Study Group of Uveitic Cataract Surgery Comparative performance of intraocular lenses in eyes with cataract and uveitis. J Cataract Refract Surg. 2002;28(12):2096–2108. doi: 10.1016/s0886-3350(02)01452-9. [DOI] [PubMed] [Google Scholar]
- 3.Muralikrishnan R, Venkatesh R, Prajna NV, Frick KD. Economic cost of cataract surgery procedures in an established eye care centre in Southern India. Ophthalmic Epidemiol. 2004;11(5):369–380. doi: 10.1080/09286580490888762. [DOI] [PubMed] [Google Scholar]
- 4.Bhargava R, Kumar P, Prakash A, Chaudhary KP. Estimation of mean ND: Yag laser capsulotomy energy levels for membranous and fibrous posterior capsular opacification. Nepal J Ophthalmol. 2012;4(1):108–113. doi: 10.3126/nepjoph.v4i1.5861. [DOI] [PubMed] [Google Scholar]
- 5.Foster CS, Rashid S. Management of coincident cataract and uveitis. Curr Opin Ophthalmol. 2003;14(1):1–6. doi: 10.1097/00055735-200302000-00001. [DOI] [PubMed] [Google Scholar]
- 6.Khairallah M. Are the Standardization of the Uveitis Nomenclature (SUN) Working Group criteria for codifying the site of inflammation appropriate for all uveitis problems? Limitations of the SUN Working Group classification. Ocul Immunol Inflamm. 2010;18(1):2–4. doi: 10.3109/09273940903348835. [DOI] [PubMed] [Google Scholar]
- 7.Hogan MJ, Kimura SJ, Thygeson P. Signs and symptoms of uveitis. I. Anterior uveitis. Am J Ophthalmol. 1959;47(5 Pt 2):155–170. doi: 10.1016/s0002-9394(14)78239-x. [DOI] [PubMed] [Google Scholar]
- 8.Bloch-Michel E, Nussenblatt RB. International Uveitis Study Group recommendations for the evaluation of intraocular inflammatory disease. Am J Ophthalmol. 1987;103(2):234–235. doi: 10.1016/s0002-9394(14)74235-7. [DOI] [PubMed] [Google Scholar]
- 9.Okhravi N, Lightman SL, Towler HM. Assessment of visual outcome after cataract surgery in patients with uveitis. Ophthalmology. 1999;106(4):710–722. doi: 10.1016/S0161-6420(99)90155-0. [DOI] [PubMed] [Google Scholar]
- 10.Jancevski M, Foster CS. Cataracts and uveitis. Curr Opin Ophthalmol. 2010;21(1):10–14. doi: 10.1097/ICU.0b013e328332f575. [DOI] [PubMed] [Google Scholar]
- 11.Kosker M, Sungur G, Celik T, Unlu N, Simsek S. Phacoemulsification with intraocular lens implantation in patients with anterior uveitis. J Cataract Refract Surg. 2013;39(7):1002–1007. doi: 10.1016/j.jcrs.2013.02.053. [DOI] [PubMed] [Google Scholar]
- 12.Bhargava R, Kumar P, Bashir H, Sharma SK, Mishra A. Manual suture less small incision cataract surgery (SICS) in patients with uveitic cataract. Middle East Afr J Ophthalmol. 2014;21(1):77–82. doi: 10.4103/0974-9233.124110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Cook C, Carrara H, Myer L. Phaco-emulsification versus manual small-incision cataract surgery in South Africa. S Afr Med J. 2012;102(6):537–540. doi: 10.7196/samj.5393. [DOI] [PubMed] [Google Scholar]
- 14.Venkatesh R, Tan CS, Sengupta S, Ravindran RD, Krishnan KT, Chang DF. Phacoemulsification versus manual small-incision cataract surgery for white cataract. J Cataract Refract Surg. 2010;36(11):1849–1854. doi: 10.1016/j.jcrs.2010.05.025. [DOI] [PubMed] [Google Scholar]
- 15.Ruit S, Tabin G, Chang D, Bajracharya L, Kline DC, Richheimer W, Shrestha M, Paudyal G. A prospective randomized clinical trial of phacoemulsification vs manual suture less small-incision extra capsular cataract surgery in Nepal. Am J Ophthalmol. 2007;143(1):32–38. doi: 10.1016/j.ajo.2006.07.023. [DOI] [PubMed] [Google Scholar]
- 16.Ram J, Gupta A, Kumar S, Kaushik S, Gupta N, Severia S. Phacoemulsification with intraocular lens implantation in patients with uveitis. J Cataract Refract Surg. 2010;36(8):1283–1288. doi: 10.1016/j.jcrs.2010.02.019. [DOI] [PubMed] [Google Scholar]
- 17.Kawaguchi T, Mochizuki M, Miyata K, Miyata N. Phacoemulsification cataract extraction and intraocular lens implantation in patients with uveitis. J Cataract Refract Surg. 2007;33(2):305–309. doi: 10.1016/j.jcrs.2006.10.038. [DOI] [PubMed] [Google Scholar]
- 18.Hazari A, Sangwan VS. Cataract surgery in uveitis. Indian J Ophthalmol. 2002;50(2):103–107. [PubMed] [Google Scholar]
- 19.Estafanous MF, Lowder CY, Meisler DM, Chauhan R. Phacoemulsification cataract extraction and posterior chamber lens implantation inpatients with uveitis. Am J Ophthalmol. 2001;131(5):620–625. doi: 10.1016/s0002-9394(00)00909-0. [DOI] [PubMed] [Google Scholar]
- 20.Haripriya A, Chang DF, Reena M, Shekhar M. Complication rates of phacoemulsification and manual small-incision cataract surgery at Aravind Eye Hospital. J Cataract Refract Surg. 2012;38(8):1360–1369. doi: 10.1016/j.jcrs.2012.04.025. [DOI] [PubMed] [Google Scholar]
- 21.Zhang JY, Feng YF, Cai JQ. Phacoemulsification versus manual small-incision cataract surgery for age-related cataract: meta-analysis of randomized controlled trials. Clin Experiment Ophthalmol. 2013;41(4):379–386. doi: 10.1111/j.1442-9071.2012.02868.x. [DOI] [PubMed] [Google Scholar]


