Abstract
Plasmid-mediated quinolone resistance (PMQR) refers to a family of closely related genes that confer decreased susceptibility to fluoroquinolones. PMQR genes are generally associated with integrons and/or plasmids that carry additional antimicrobial resistance genes active against a range of antimicrobials. In Ho Chi Minh City (HCMC), Vietnam, we have previously shown a high frequency of PMQR genes within commensal Enterobacteriaceae. However, there are limited available sequence data detailing the genetic context in which the PMQR genes reside, and a lack of understanding of how these genes spread across the Enterobacteriaceae. Here, we aimed to determine the genetic background facilitating the spread and maintenance of qnrS1, the dominant PMQR gene circulating in HCMC. We sequenced three qnrS1-carrying plasmids in their entirety to understand the genetic context of these qnrS1-embedded plasmids and also the association of qnrS1-mediated quinolone resistance with other antimicrobial resistance phenotypes. Annotation of the three qnrS1-containing plasmids revealed a qnrS1-containing transposon with a closely related structure. We screened 112 qnrS1-positive commensal Enterobacteriaceae isolated in the community and in a hospital in HCMC to detect the common transposon structure. We found the same transposon structure to be present in 71.4 % (45/63) of qnrS1-positive hospital isolates and in 36.7 % (18/49) of qnrS1-positive isolates from the community. The resulting sequence analysis of the qnrS1 environment suggested that qnrS1 genes are widely distributed and are mobilized on elements with a common genetic background. Our data add additional insight into mechanisms that facilitate resistance to multiple antimicrobials in Gram-negative bacteria in Vietnam.
Introduction
Fluoroquinolones are among the current first line of drugs in Vietnam for treating infections ranging from pneumonia to diarrhoea to bacteraemia. Since the mid-1990s, resistance to quinolones has been increasing steadily in multiple organisms causing respiratory tract infections, diarrhoea and bacteraemia (Holt et al., 2013; Nga et al., 2012; Nhu et al., 2014). The emergence of fluoroquinolone resistance in these pathogens and other bacteria represents a clear threat to the effective treatment of common bacterial infections. Resistance to quinolones occurs commonly via mutations in the gene of the target enzyme, DNA gyrase, but can also be plasmid mediated. Plasmid-mediated quinolone resistance (PMQR), in the form of a qnr gene, was first described in 1998 in a Klebsiella pneumoniae isolate from a patient with urinary tract infection in North America (Martínez-Martínez et al., 1998). Since this first report, multiple studies have described a range of PMQR determinants found within the Enterobacteriaceae and other bacterial families (Strahilevitz et al., 2009). To date, five qnr genes have been described and are distinguished on the basis of their sequence homology: qnrA, qnrB and qnrS1, and more recently qnrC and qnrD (Cavaco et al., 2009; Hata et al., 2005; Jacoby et al., 2006; Martínez-Martínez et al., 1998; Wang et al., 2009). The qnr genes appear to be highly promiscuous, having the capacity to become rapidly disseminated among related and unrelated hosts. The transmissibility of the qnr genes makes the genomic mechanisms facilitating their movement of considerable interest and of relevance in the community and in healthcare settings. Of the five qnr genes identified, only the genetic contexts of qnrA and qnrB have been comprehensively described. These genes are commonly located within complex sul1-type class 1 integrons (Garnier et al., 2006; Poirel et al., 2006). The context of qnrS is less well described, but there are reports of the gene being located within a gene cluster flanked by IS26 transposases (Chen et al., 2006; Hu et al., 2008). However, it is currently unknown if the qnrS gene is always associated with IS26 or if qnrS can be transferred and/or maintained by other, unrelated, mobile elements.
Several qnrS1-carrying plasmids have been described in the literature and have publicly available nucleotide sequences. These plasmids range in size and belong to various incompatibility groups including IncN (Dobiasova et al., 2013; Literak et al., 2010), IncI1 (Dobiasova et al., 2013), IncX1 (Dobiasova et al., 2013; Literak et al., 2010) and IncX2 (Literak et al., 2010; Sumrall et al., 2014). These qnrS1-encoding plasmids have been identified in Asia and Europe and in a range of Gram-negative bacteria including K. pneumoniae, Escherichia coli and Enterobacter aerogenes (Dobiasova et al., 2013; Hu et al., 2008; Park et al., 2009; Sumrall et al., 2014). Two of the earliest and best-described qnrS1-encoding plasmids are pTPqnrS-1a and pK245. Plasmid pTPqnrS-1a is a 10 kb replicon, and was isolated from a multidrug-resistant (MDR) Salmonella Typhimurium DT193 in the UK (Kehrenberg et al., 2007). The second, pK245, was characterized in a clinical isolate of K. pneumoniae originating in Taiwan (Chen et al., 2006). In contrast to pTPqnrS-1a, pK245 is a large MDR plasmid of approximately 100 kb. The MDR phenotype of pK245 was demonstrated by transferring this plasmid into an antimicrobial-susceptible Escherichia coli strain by electrotransformation (Chen et al., 2006). The pK245-positive transformant showed an increase in MICs to multiple classes of antimicrobials, including aminoglycosides, β-lactams and (fluoro)quinolones and had an extended-spectrum β-lactamase (ESBL) phenotype (Chen et al., 2006). Comparative sequence analysis of available qnrS1 plasmids revealed that the genetic architecture surrounding the qnrS1 gene is identical between pTPqnrS-1a and pK245, and they additionally sharing a high sequence identity with the qnrS1 genetic region in other partial plasmid sequences, including pAH0376 from Shigella flexneri (Hata et al., 2005) and pINF5 from Salmonella Infantis (Kehrenberg et al., 2006).
In a study investigating the distribution of PMQR determinants in Enterobacteriaceae isolated from hospitalized patients and healthy volunteers from Ho Chi Minh City (HCMC), Vietnam, we found an exceptionally high prevalence of the qnrS1 genes in both hospital (63/139, 45 %) and community (49/413, 12 %) bacterial isolates (Vien et al., 2009). We therefore hypothesized that qnrS1 was embedded on a highly mobile and conserved genetic element, which was contributing to the spread and the apparent success of qnrS1 across the Enterobacteriaceae in this setting. In this current study, we aimed to characterize the dominant qnrS1-containing elements circulating in Enterobacteriaceae isolated from hospital patients and community volunteers in HCMC to understand if qnrS1 is being disseminated on one or more elements by defining their genetic context. To achieve this, we selected three qnrS1-containing plasmids, broadly representative of those found to be circulating in the hospital and community environments (Vien et al., 2012), for DNA sequencing and analysis in their entirety. The resulting sequence analysis of the qnrS1-containing mobile elements has broadened our knowledge of the genetic architecture surrounding the qnrS1 gene and added insight into MDR mechanisms that are circulating within these differing bacterial environments in Vietnam.
Methods
Bacterial strains
A total of 115 qnrS1-positive Enterobacteriaceae strains (38 Escherichia coli, 69 K. pneumoniae and eight from other Enterobacteriaceae species) were selected for analysis in this study. All of the strains have been described previously and were isolated from patients admitted to the tetanus ward of the Hospital for Tropical Diseases (HTD) in HCMC, Vietnam, between May and October 2004 and between June and November 2005 or from healthy volunteers participating in a typhoid vaccine study in 2005 and 2006 (Tran et al., 2010). The presence of the qnrS1 gene in these strains has been previously confirmed and described (Vien et al., 2009).
Three qnrS1-containing plasmids identified previously (Vien et al., 2012) were selected to be broadly representative of qnrS1-containing plasmids circulating in our setting (i.e. harbouring strain, hospital/community infections and size) and were sequenced in their entirety, assembled and annotated gene by gene in comparison to sequences available in public databases. These plasmids (selected out of the 115 qnrS1-positive Enterobacteriaceae strains) were pE66An (Escherichia coli host), pK18An (K. pneumoniae host) and pK1HV (K. pneumoniae host). A summary of the basic features of these plasmids is given in Table 1.
Table 1. Features of the three sequenced qnrS1-containing plasmids.
| Characteristic | pE66An | pK18An | pK1HV |
| Original strain | Escherichia coli | K. pneumoniae | K. pneumoniae |
| Source | Hospital | Hospital | Community |
| Size (kb) | 80.105 | 51.160 | 133.191 |
| G+C (mol%) | 51.25 | 51.32 | 52.5 |
| Inc group | IncN | IncN | IncFII |
| Predicted coding sequences | 109 | 72 | 167 |
| Essential function genes | 22 | 15 | 14 |
| Conjugative system genes | 16 | 6 | 21 |
| Resistance genes | 7 | 5 | 11 |
| IS elements | IS26, IS6100, IS6100903D, ISEcp1 | IS26, IS10, IS5 | IS26, IS4, IS1414 |
| Integron | Integron 1 | Integron 1 | Integron 1 |
Plasmid sequencing and annotation
The three qnrS1-containing plasmids were sequenced at the Wellcome Trust Sanger Institute in the UK using conventional Sanger sequencing methods as described previously (Parkhill et al., 2001). The plasmid sequences were annotated and analysed using Artemis (Rutherford et al., 2000) and aligned and compared using the Artemis Comparison Tool (Carver et al., 2008). Plasmid circularization and graphical representations were performed using DNA Plotter software (Carver et al., 2009).
PCR amplification for the qnrS1-containing transposon and RFLP typing
Total genomic DNA from 112 qnrS1 PCR amplification-positive Enterobacteriaceae isolates was extracted using a Wizard Genomic DNA purification kit (Promega), according to the manufacturer's specifications. PCR amplification for the qnrS1-containing transposon from the extracted genomic DNA was performed under the following condition: 94 °C for 10 s, 55 °C for 30 s and 68 °C for 6 min for 35 cycles. Amplification was performed using the Expand Long Template PCR System (Roche) using the primers Trans-qnrS-F (5′-CAGGAAGAGGCATTGTCAAAGG-3′) and Trans-qnrS-R (5′-GGTGCTTGTCAGCGTAAA-3′). These primers were designed using Primer Express 5 software (Applied Biosystems, Life Technologies) and their specificity was assessed in silico using blastn (http://blast.ncbi.nlm.nih.gov/Blast.cgi). The resulting PCR amplicons were examined by electrophoresis and UV visualization on 2 % agarose gels containing 2 % ethidium bromide. The PCR amplicons containing the qnrS1 gene were typed using RFLP with three different enzymes: EcoRV, HindIII and PvuII (New England Biolabs). The restriction-digested PCR amplicons were analysed by gel electrophoresis for 2 h on a 1 % agarose gel, stained with 2 % ethidium bromide and examined under UV light. The restriction fragments were sized and compared for group typing using Bionumerics software (Applied Maths).
Primer-walking sequencing
The qnrS1-containing transposon from the K34N strain was sequenced using primer walking. The sequencing reaction was performed in a 20 μl reaction containing 4 μl Big Dye Terminator, 2 μl buffer, 20 ng genomic DNA and distilled water up to 20 μl. Each fragment was repeated four times using an ABI 3130XL machine (Applied Biosystems, Life Technologies). All sequences were assembled using Vector NTI software (Life Technologies).
Electrotransformation
PCR-negative isolates for the qnrS1-containing transposon were analysed for the presence of subfamilies of the known transposon. Plasmid DNA from these negative isolates was extracted using a Qiagen Midi Prep Plasmid DNA Extraction kit, as per the manufacturer's recommendations. Escherichia coli TOP10 cells (Invitrogen, Life Technologies) were transformed with isolated plasmid DNA by a Bio-Rad gene pulser, using conditions recommended by the manufacturer (Invitrogen, Life Technologies). Transformants were selected on Luria–Bertani medium supplemented with 0.03 mg ciprofloxacin l− 1. Plasmid DNA from these transformants was extracted by the method of Kado & Liu (1981), examined on an agarose gel for the presence of only one plasmid and then subjected to PCR amplification for the qnrS1 gene to ensure transformation of the appropriate plasmid.
Southern blot analysis
Isolates containing a subfamily of known qnrS1-containing transposons were detected by Southern blotting with two different probes: qnrS1 and blaLAP-2, using the primers described for amplification of the qnrS1 region. Plasmid DNA from the transformants was extracted using a Qiagen Midi Prep Plasmid DNA Extraction kit, as per manufacturer's recommendations. These plasmids were then digested with EcoRI and duplicates were run on a gel. The gel was subsequently transferred to a membrane. The membrane was cut into two pieces and each was hybridized with one of the probes, qnrS1 or blaLAP-2. If an isolate had signal with both probes binding to the same fragment, it was assigned as carrying a subfamily of a qnrS1-containing transposons.
Results and Discussion
Global comparison of the three qnrS1-containing plasmids
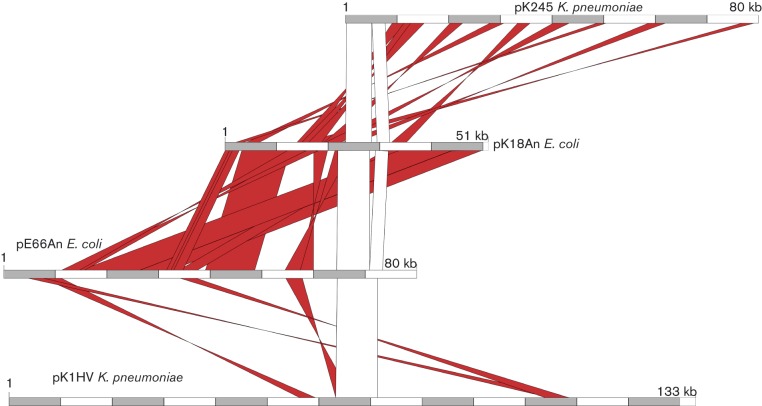
Figure 1 shows a global DNA alignment of the three sequenced plasmids and a previously sequenced qnrS1-containing plasmid (pK245) identified in a K. pneumoniae isolate from Taiwan as a comparator (Chen et al., 2006). These alignments showed that the two plasmids isolated independently from different bacterial genera within the hospital environment (pE66An and pK18An) exhibited substantial gene synteny with each other, but generally shared a lower degree of DNA homology with the plasmid identified in a community isolate (pK1HV).
Fig. 1. Linear DNA sequence alignments of pE66An, pK18An and pK1HV against plasmid pK245 centred at the qnrS1-encoding region. Regions of DNA identity of 99 % or greater are linked by red blocks. The open box is the region containing the qnrS1-harbouring transposon, which is identical in all four plasmids except for a 980 bp insert in pK18An. The sizes of the plasmids are shown, with each shaded/open block representing 10 kbp.
Plasmid pE66An
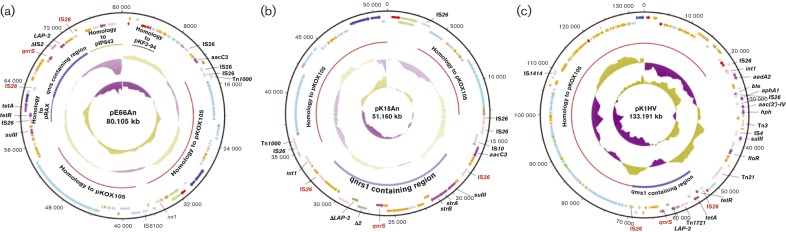
The qnrS1-encoding plasmid pE66An was extracted from an Escherichia coli strain isolated from a rectal swab taken from a patient admitted to the tetanus ward of the HTD in HCMC. Plasmid pE66An is 80 105 bp with an approximately neutral G+C content of 51.52 mol% (Fig. 2). After complete annotation of the plasmid sequence, 109 predicted coding sequences (CDSs) were identified; the protein products of seven of these CDSs were predicted to be associated with resistance to a variety of antimicrobial classes. These antimicrobial resistance genes were: aacC3 (gentamicin), sulII (sulfonamides), tetR and tetA (tetracyclines), qnrS1 (quinolones), bla LAP-2 (β-lactams) and bla CTX-M-14 (third-generation cephalosporins). An association between the qnrA gene and ESBL-encoding genes has been reported previously (Castanheira et al., 2007; Hamouda et al., 2008; Lavigne et al., 2006). The bla CTX-M-14 gene within pE66An was adjacent to the element ISEcp1. The insertion element ISEcp1 has been shown previously to mediate the transfer of bla CTX-M-14 (Bou et al., 2002). As the bla CTX-M-14 gene is in association with this ISEcp1 insertion element, the potential for dissemination of this gene to other plasmids or transposable elements is likely to be enhanced.
Fig. 2. Annotated circular plasmid maps of pE66An, pK18An and pK1HV orientated from the origin of replication. (a) pE66An; (b) pK18An; (c) pK1HV. Identified and annotated ORFs are colour coded on each of the three plasmids: red, plasmid replication; dark blue, inorganic/metal/UV resistance; sky blue, conjugal transfer; dark pink, antimicrobial resistance; light green, unknown; light blue, regulators; orange, conserved hypothetical; brown, pseudogenes or partial genes; light pink, IS elements. The arrow annotation shows the strand on which the ORF is located. The inner circle shows the GC skew ([GC]/[G+C]) and the next outer circle shows G+C (mol%) plot. Fragments with substantial DNA homology to other sequenced plasmids and the qnrS-encoding region are highlighted.
Comparative analysis showed that two regions within plasmid pE66An, a 16.7 and a 10.2 kb region, exhibited significant DNA sequence similarity to regions within the previously described Klebsiella oxytoca plasmid pKOX105 (Carattoli et al., 2010) and three smaller regions (5.9, 5.2 and 3.5 kb) within plasmids pIP843 (Cao et al., 2002), pRAx (Fricke et al., 2009) and pKF3-94 (Zhao et al., 2010) from HCMC, Madagascar and China, respectively (Fig. 2a). Plasmid pKOX105 was isolated from a K. oxytoca isolate present in the intestinal microbiota of an individual in a long-term care facility in Bolzano, Italy, in 2005. Both pE66An and pKOX105 are IncN plasmids, with each containing the highly conserved IncN plasmid backbone (Carattoli et al., 2010). Plasmid pE66An contains two regions of 10 280 and 5183 bp that encode sequences predicted to be responsible for the conjugal transfer. Indeed, our previous work has shown that pE66An has the capacity to be efficiently conjugated at high frequency into a suitable recipient strain. We therefore concluded that this conjugation system is functionally active (Vien et al., 2009).
In addition to antimicrobial resistance and conjugal transfer functions, plasmid pE66An also encodes genes that suggest that it may be able to adapt to a variety of hosts and environments. For example, we identified a CDS with 99.5 % identity to frmA, a class III alcohol dehydrogenase identified in a Pasteurella piscicida isolate (Kim & Aoki, 1994), and 98.1 % identity to a class III alcohol dehydrogenase identified in an Escherichia coli isolate (Hochhut et al., 2006). The function of the protein product encoded by frmA is involved in resistance to formaldehyde and other aldehydes. We predict that frmA provides a selective advantage for bacterial hosts in hospital environments where disinfectants containing aldehydes are used (Chen et al., 2006).
Plasmid pK18An
Plasmid pK18An was carried by a K. pneumoniae isolate taken from a rectal swab of a tetanus patient in the HTD in HCMC. The sequence of plasmid pK18An showed it was a circular replicon of 51 160 bp, with a G+C content of 51.32 mol% (Fig. 2). The annotation of pK18An identified 72 CDSs, the functions of six of which were predicted to be associated with resistance to antimicrobials, including aacC3 (gentamicin), sulII (sulfonamides), strA and strB (streptomycins), dhfrV (trimethoprim) and qnrS1 (quinolones). Plasmid pK18An was also found to harbour a bla LAP-2 gene in close proximity to qnrS1, but that carried an IS5 element insertion and so was likely to have been inactivated. The sulII–strA–strB genes were located in close proximity to each other and adjacent to an IS26 element. The strA–strB genes are often linked with the sulII sulfonamide-resistance gene, commonly encoded on broad-host-range non-conjugative plasmids in a range of Gram-negative bacteria found in humans and animals. The usage of streptomycin in clinical and animal medicine has diminished dramatically over the last 10–20 years, yet the persistence of sulII–strA–strB implies that factors other than a direct selection pressure from the antimicrobial are important for the maintenance of these genes (Sundin & Bender, 1996).
Like pE66An, plasmid pK18An shared two large regions, of 10.2 and 9.4 kb, with extensive DNA homology to plasmid pKOX105 (Carattoli et al., 2010) (Fig. 2b). Similarly, plasmid pK18An also contained two regions containing CDSs that are predicted to be responsible for the conjugal transfer, but these operons were disrupted by numerous IS26 elements. These sequence data probably explain why it was not possible to conjugate pK18An into a recipient Escherichia coli under laboratory conditions (Vien et al., 2009). Whilst pK18An was not conjugative, the plasmid sequence was littered with multiple IS elements, particularly surrounding antimicrobial resistance genes, suggesting that such elements may facilitate the independent transfer of these genes to other plasmids.
Plasmids pK18An and pE66An both contained restriction modification systems; pK18An contained an ecoRIIM gene and pE66An contained the ecoRIIR and ecoRIIM genes. The ecoRIIR and ecoRIIM genes share 98.8 % nucleotide identity with the ecoRII endonuclease gene (Bhagwat et al., 1990), and have 100 % nucleotide identity with the Escherichia coli modification methylase gene, ecoRII (Som et al., 1987). In addition to assisting with defence against bacteriophage infection, this restriction modification system has also been reported to contribute to the spread and maintenance of plasmids encoding these systems (Kobayashi, 2001).
Plasmid pK1HV
Plasmid pK1HV was isolated from a K. pneumoniae strain cultured from a healthy child, resident in HCMC. Plasmid pK1HV was the largest of the three sequenced plasmids at 133 191 bp with a G+C content of 52.5 mol% (Fig. 2). Plasmid pK1HV contained 167 predicted CDSs, of which the overwhelming majority were of unknown function. However, pK1HV was also found to carry 11 genes that are associated with resistance to various classes of antimicrobials, including aadA2 (streptomycin), ble (bleomycin), aphA1 (gentamicin), aac(3′)-IV (gentamicin), hph (hygromycin), sulII (sulfonamides), forR (chloramphenicol), tetR and tetA (tetracyclines), bla LAP-2 (β-lactams) and qnrS1 (quinolones). Similar to pE66An, pK1HV also carried an IS26–tetR–tetA complex, which is a common mechanism facilitating the transfer of tetracycline resistance. Plasmid pK1HV also harboured a type 1 integron containing the dfrA12–orfF–aadA2 cassette, an antimicrobial resistance region that remains common in contemporarily isolated MDR Gram-negative organisms (Gestal et al., 2005). The presence this dfrA12–orf–aadA2-containing type 1 integron in pK1HV (isolated from the community) again poses questions regarding the ongoing selection of genes encoding resistance to streptomycin.
Comparative sequence analysis of pK1HV with pKOX105 revealed a large contiguous fragment of ∼84 kb shared between both plasmids (Carattoli et al., 2010) (Fig. 2c). All of the predicted genes on this extended fragment of plasmid DNA were mostly conserved but functionally unknown, or were genes proposed to encode components required for conjugal transfer. The remaining regions of pK1HV carried the identified antimicrobial resistance genes and an array of IS elements (Fig. 2c). Whilst pK1HV did not contain the mucAB operon, like pE66An and pK18An, it did contain the imp operon, which had 85 % nucleotide identity to impA and impB on the IncI1 plasmid TP110 from a Salmonella Typhimurium strain isolated in the UK in 1968 (Lodwick et al., 1990).
Characterization of qnrS1-containing transposons
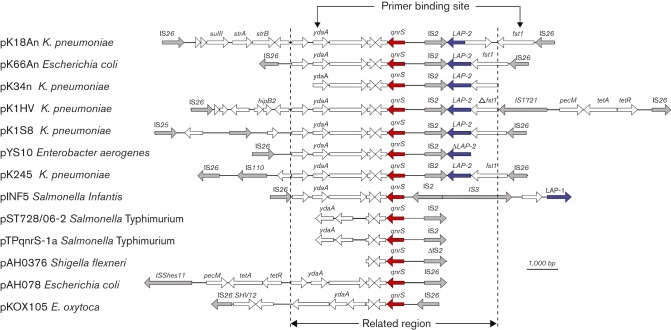
Annotation of the three sequenced qnrS1-containing transposons extracted from the plasmid sequences of pE66An, pK18An and pK1HV and their alignments with other described qnrS1-containing fragments are shown in Fig. 3. The alignment of qnrS1-containing fragments from pE66An, pK18An and pK1HV showed that they were identical, except for pK18An, which contained a 980 bp insertion. The qnrS1 gene in all of the three plasmids was located within a transposon structure composed of two identical IS26 elements at either terminal portion of the transposon. In addition to containing the qnrS1 gene, these transposons also carried the bla LAP-2 gene, which confers resistance to narrow-spectrum β-lactams. Additionally, this transposon shared a common backbone with other available qnrS sequences and contained an IS2 element, a putative resolvase (ydaA) predicted to belong to a family of stress proteins (Beliaev et al., 2002), and three other proteins of unknown function (Park et al., 2009; Wu et al., 2008) (Fig. 3). The G+C content of the qnrS-containing transposons was ∼50 mol%, which was slightly lower than the mean G+C content of the sequenced plasmids in their entirety, consistent with the notion that the qnrS1-encoding transposons have been inserted into these plasmids via horizontal gene transfer.
Fig. 3. A schematic representation of sequenced qnrS1-containing transposons. Graphical representation of the synteny between the qnrS-containing transposons between the three plasmids sequenced here (pK18An, pE66An and pK1HV) and other sequenced fragments containing the qnrS-encoding region. The plasmids and the host organism in which they were first identified are given. The region with the greatest DNA homology is identified and includes the highlighted ORFs for qnrS (red), a putative IS2 element (grey), a gene encoding a putative resolvase protein (ydaA) and three other ORFs encoding hypothetical proteins of unknown function. Additional genes are colour coded: blue, bla LAP-2; grey, IS elements; white, ORFs without a name encoding hypothetical uncharacterized proteins. The locations of the binding sites for PCR amplification of the transposon are highlighted.
Screening for qnrSI transposons in commensal Enterobacteriaceae isolated in HCMC
Using the newly generated DNA sequences of the qnrS1-containing transposons from the three sequenced plasmids (Fig. 3), we designed PCR primers to amplify and compare qnrS1-containing transposons from DNA extracted from 112 qnrS1-positive hospital- and community-acquired Enterobacteriaceae that have been described previously (Vien et al., 2011, 2012; see Methods). The locations of the primer-binding sites are shown in Fig. 3 and the predicted sizes of PCR amplicons were 6859 bp (pE66An qnrS1 transposon, type A) and 8059 bp (pK18An qnrS1 transposon, type B). Seventy-one of the 112 isolates (63.4 %) were PCR amplicon-positive for the qnrS1-containing transposon and were of sizes consistent with those described in pE66An and pK18An. The 71 PCR amplicons with known qnrS1-containing transposons were subjected to RFLP analysis with EcoRV, HindIII and PvuII. In addition to the two described qnrS1-containing transposons that were identified in the three sequenced plasmids, the RFLP mapping patterns from these 71 amplicons also revealed a transposon with a third structure (K34N strain, type C). Using a primer-walking sequencing method, we found that the qnrS1-containing transposon in K34N strain was 6652 bp and identical to the qnrS1-containing transposon from E66An strain, except for a 200 bp deletion in a gene of unknown function (Fig. 3). We were therefore able to distinguish three related yet distinct qnrS1-containing transposons of 6859, 8059 and 6652 bp, which we arbitrarily named types A, B and C, respectively.
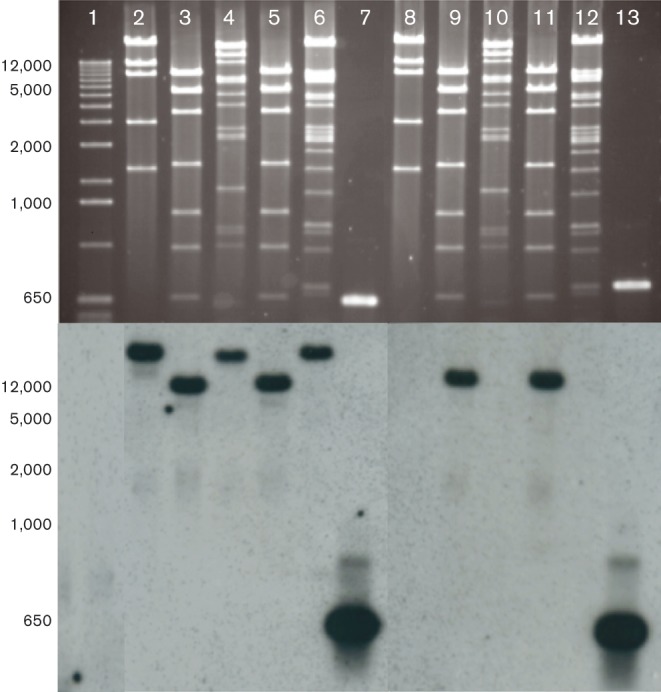
The 41 isolates with an undetermined qnrS1-containing mobile element were further investigated for the presence of a subfamily of the known transposon. There have been multiple reports regarding the association between qnrS1 and blaLAP-2 genes (Cano et al., 2009; Dahmen et al., 2010; Huang et al., 2008; Park et al., 2009; Poirel et al., 2006, 2007). Yet, due to a lack of a PCR amplicon, we hypothesized that some isolates contain both the qnrS1 and the bla LAP-2 sequence, but the adjacent regions demonstrate a different structure. A laboratory Escherichia coli strain was successfully transformed with a qnrS1-encoding plasmid extracted from 27 of the 41 isolates with an undefined qnrS1-containing transposon structure. Plasmid DNA from these 27 transformants was extracted and digested with EcoRI and individually probed by Southern blotting targeting the qnrS1 and bla LAP-2 genes. With the resulting plasmid hybridization of the plasmid DNA extracted from the 27 isolates, only two produced a detectable signal with both qnrS1 and bla LAP-2 probes on the same digestion fragment, implying that these two plasmids (and their corresponding hosts) also carry a qnrS1-containing transposon of the same subfamily as described in the sequenced plasmids (Fig. 4).
Fig. 4. EcoRI digestion of qnrS-encoding plasmids hybridized with the qnrS1 and bla LAP-2 probes. Upper panel: agarose gel electrophoresis of qnrS-encoding plasmids after digestion with EcoRI. The resulting patterns are duplicate digestions from five plasmids after conjugation. Lanes: 2 and 8, isolate LTMV18; 3 and 9, isolate LTMV33; 4 and 10, isolate LTMV6; 5 and 11, isolate LTMV30; 6 and 12, isolate LTMV1. The ladder (lane 1) is 1 kb Plus (Invitrogen) with sizes shown in kb. Lanes 7 and 13 contain the PCR amplicons of qnrS and bla LAP-2 as positive controls. Bottom panel (left): hybridization against the qnrS1 probe after Southern blotting. All five plasmids contained fragments corresponding to probe qnrS1. Bottom panel (right): hybridization against the bla LAP-2 probe after Southern blotting.
We stratified the proportion of the three qnrS1-containing transposons across all the hospital- and community-acquired isolates that were compared (Table 2). Forty-five of the 63 hospital isolates (71.4 %) harboured a qnrS1-containing transposon that was identical to pE66An (type A), one isolate (1.6 %) carried a qnrS1-containing transposon identical to pK18An (type B) and three isolates (4.8 %) carried a qnrS1-containing transposon identical to K34N (type C). Similarly, in the 49 community isolates, there were 18 (36.7 %), one (2 %) and three (6.1 %) isolates carrying type A, B and C qnrS1-containing transposons, respectively. However, 14 isolates (22.2 %) from the hospital and 27 isolates (55.1 %) from the community qnrS1 gene-positive isolates were negative by PCR targeting the qnrS1-containing transposon, suggesting that the qnrS1 gene in these strains is embedded on a different genetic element, which was undeterminable by the described PCR amplification methods used here.
Table 2. Distribution of the various qnrS1-containing transposons in 122 Enterobacteriaceae isolated in HCMC.
| Strain | Type A | Type B | Type C | Subtype | Unknown |
| Hospital strains (n = 63) | |||||
| Escherichia coli (n = 5) | 3 (60 %) | 0 | 0 | 0 | 2 (40 %) |
| K. pneumoniae (n = 52) | 40 (77 %) | 1 (2 %) | 3 (5.7 %) | 0 | 8 (15.3 %) |
| Others (n = 6) | 2 (33.3 %) | 0 | 0 | 0 | 4 (66.7 %) |
| Community strains (n = 49) | |||||
| Escherichia coli (n = 32) | 13 (40.6 %) | 0 | 0 | 1 (3.1 %) | 18 (56.3 %) |
| K. pneumoniae (n = 15) | 5 (33.3 %) | 1 (6.7 %) | 3 (20 %) | 1 (6.7 %) | 5 (33.3 %) |
| Others (n = 2) | 0 | 0 | 0 | 0 | 2 (100 %) |
Conclusions
Hospital-acquired infections with antimicrobial-resistant organisms can be dangerous, and we have recently shown the problems that can be associated with highly virulent clones of K. pneumoniae (Chung The et al., 2015). Here, we have shown that there is a dominant qnrS1-containing transposon circulating in qnrS1-positive Enterobacteriaceae in HCMC, Vietnam, which we found to be present in 71.4 % of qnrS1-positive isolates from a hospital and in 36.7 % of qnrS1-positive isolates from the community. Moreover, we determined the complete nucleotide sequences of three qnrS1-encoding plasmids. These sequences permitted a description of the circulating genes contributing to an MDR phenotype in three bacterial isolates from a hospital and the community, and also provided insights into the means of adaptation of these plasmids within a variety of hosts and environments. Notably, the DNA sequences of the two plasmids isolated from two different bacterial genes in a hospital setting exhibited substantial homology, thus presenting evidence of genetic transfer among nosocomial commensal bacteria. Finally, the annotation of plasmid pE66An provides further evidence for the association of the PMQR gene qnrS and the ESBL gene bla CTX-M-14.
Acknowledgements
The authors wish to thank all the staff of the intensive care unit at the HTD for assisting in sample and data collection and patient care, and Ms Song Chau for her ongoing efforts. This work was supported by the Wellcome Trust of the UK, through core funding (089276/2/09/2). S. B. is a Sir Henry Dale Fellow, jointly funded by the Wellcome Trust and the Royal Society (100087/Z/12/Z). The authors declare they have no conflicts of interest.
Abbreviations:
- CDS
coding sequence
- ESBL
extended-spectrum β-lactamase
- HCMC
Ho Chi Minh City
- HTD
Hospital for Tropical Diseases
- MDR
multidrug-resistant
- PMQR
plasmid-mediated quinolone resistance
References
- Beliaev et al., 2002. Beliaev A.S., Thompson D.K., Fields M.W., Wu L., Lies D.P., Nealson K.H., Zhou J. (2002). Microarray transcription profiling of a Shewanella oneidensis etrA mutant J Bacteriol 184 4612–4616 10.1128/JB.184.16.4612-4616.2002 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhagwat et al., 1990. Bhagwat A.S., Johnson B., Weule K., Roberts R.J. (1990). Primary sequence of the EcoRII endonuclease and properties of its fusions with β-galactosidase J Biol Chem 265 767–773 . [PubMed] [Google Scholar]
- Bou et al., 2002. Bou G., Cartelle M., Tomas M., Canle D., Molina F., Moure R., Eiros J.M., Guerrero A. (2002). Identification and broad dissemination of the CTX-M-14 β-lactamase in different Escherichia coli strains in the northwest area of Spain J Clin Microbiol 40 4030–4036 10.1128/JCM.40.11.4030-4036.2002 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cano et al., 2009. Cano M.E., Rodríguez-Martínez J.M., Agüero J., Pascual A., Calvo J., García-Lobo J.M., Velasco C., Francia M.V., Martínez-Martínez L. (2009). Detection of plasmid-mediated quinolone resistance genes in clinical isolates of Enterobacter spp. in Spain J Clin Microbiol 47 2033–2039 10.1128/JCM.02229-08 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cao et al., 2002. Cao V., Lambert T., Courvalin P. (2002). ColE1-like plasmid pIP843 of Klebsiella pneumoniae encoding extended-spectrum β-lactamase CTX-M-17 Antimicrob Agents Chemother 46 1212–1217 10.1128/AAC.46.5.1212-1217.2002 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carattoli et al., 2010. Carattoli A., Aschbacher R., March A., Larcher C., Livermore D.M., Woodford N. (2010). Complete nucleotide sequence of the IncN plasmid pKOX105 encoding VIM-1, QnrS1 and SHV-12 proteins in Enterobacteriaceae from Bolzano, Italy compared with IncN plasmids encoding KPC enzymes in the USA J Antimicrob Chemother 65 2070–2075 10.1093/jac/dkq269 . [DOI] [PubMed] [Google Scholar]
- Carver et al., 2008. Carver T., Berriman M., Tivey A., Patel C., Böhme U., Barrell B.G., Parkhill J., Rajandream M.-A. (2008). Artemis and ACT: viewing, annotating and comparing sequences stored in a relational database Bioinformatics 24 2672–2676 10.1093/bioinformatics/btn529 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carver et al., 2009. Carver T., Thomson N., Bleasby A., Berriman M., Parkhill J. (2009). DNAPlotter: circular and linear interactive genome visualization Bioinformatics 25 119–120 10.1093/bioinformatics/btn578 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Castanheira et al., 2007. Castanheira M., Pereira A.S., Nicoletti A.G., Pignatari A.C.C., Barth A.L., Gales A.C. (2007). First report of plasmid-mediated qnrA1 in a ciprofloxacin-resistant Escherichia coli strain in Latin America Antimicrob Agents Chemother 51 1527–1529 10.1128/AAC.00780-06 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cavaco et al., 2009. Cavaco L.M., Hasman H., Xia S., Aarestrup F.M. (2009). qnrD, a novel gene conferring transferable quinolone resistance in Salmonella enterica serovar Kentucky and Bovis morbificans strains of human origin Antimicrob Agents Chemother 53 603–608 10.1128/AAC.00997-08 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen et al., 2006. Chen Y.-T., Shu H.-Y., Li L.-H., Liao T.-L., Wu K.-M., Shiau Y.-R., Yan J.-J., Su I.-J., Tsai S.-F., Lauderdale T.-L. (2006). Complete nucleotide sequence of pK245, a 98-kilobase plasmid conferring quinolone resistance and extended-spectrum-β-lactamase activity in a clinical Klebsiella pneumoniae isolate Antimicrob Agents Chemother 50 3861–3866 10.1128/AAC.00456-06 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chung The et al., 2015. Chung The H., Karkey A., Pham Thanh D., Boinett C.J., Cain A.K., Ellington M., Baker K.S., Dongol S., Thompson C., other authors (2015). A high-resolution genomic analysis of multidrug-resistant hospital outbreaks of Klebsiella pneumoniae EMBO Mol Med 7 227–239 10.15252/emmm.201404767 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dahmen et al., 2010. Dahmen S., Poirel L., Mansour W., Bouallègue O., Nordmann P. (2010). Prevalence of plasmid-mediated quinolone resistance determinants in Enterobacteriaceae from Tunisia Clin Microbiol Infect 16 1019–1023 10.1111/j.1469-0691.2009.03010.x . [DOI] [PubMed] [Google Scholar]
- Dobiasova et al., 2013. Dobiasova H., Dolejska M., Jamborova I., Brhelova E., Blazkova L., Papousek I., Kozlova M., Klimes J., Cizek A., Literak I. (2013). Extended spectrum β-lactamase and fluoroquinolone resistance genes and plasmids among Escherichia coli isolates from zoo animals, Czech Republic FEMS Microbiol Ecol 85 604–611 10.1111/1574-6941.12149 . [DOI] [PubMed] [Google Scholar]
- Fricke et al., 2009. Fricke W.F., Welch T.J., McDermott P.F., Mammel M.K., LeClerc J.E., White D.G., Cebula T.A., Ravel J. (2009). Comparative genomics of the IncA/C multidrug resistance plasmid family J Bacteriol 191 4750–4757 10.1128/JB.00189-09 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garnier et al., 2006. Garnier F., Raked N., Gassama A., Denis F., Ploy M.-C. (2006). Genetic environment of quinolone resistance gene qnrB2 in a complex sul1-type integron in the newly described Salmonella enterica serovar Keurmassar Antimicrob Agents Chemother 50 3200–3202 10.1128/AAC.00293-06 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gestal et al., 2005. Gestal A.M., Stokes H.W., Partridge S.R., Hall R.M. (2005). Recombination between the dfrA12-orfF-aadA2 cassette array and an aadA1 gene cassette creates a hybrid cassette, aadA8b Antimicrob Agents Chemother 49 4771–4774 10.1128/AAC.49.11.4771-4774.2005 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hamouda et al., 2008. Hamouda A., Vali L., Alsultan A., Dancer S., Amyes S.G.B. (2008). First report of ciprofloxacin resistance among Klebsiella pneumoniae harbouring the qnrA1 gene and producing SHV-5 extended-spectrum β-lactamase in Scotland J Chemother 20 753–755 10.1179/joc.2008.20.6.753 . [DOI] [PubMed] [Google Scholar]
- Hata et al., 2005. Hata M., Suzuki M., Matsumoto M., Takahashi M., Sato K., Ibe S., Sakae K. (2005). Cloning of a novel gene for quinolone resistance from a transferable plasmid in Shigella flexneri 2b Antimicrob Agents Chemother 49 801–803 10.1128/AAC.49.2.801-803.2005 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hochhut et al., 2006. Hochhut B., Wilde C., Balling G., Middendorf B., Dobrindt U., Brzuszkiewicz E., Gottschalk G., Carniel E., Hacker J. (2006). Role of pathogenicity island-associated integrases in the genome plasticity of uropathogenic Escherichia coli strain 536 Mol Microbiol 61 584–595 10.1111/j.1365-2958.2006.05255.x . [DOI] [PubMed] [Google Scholar]
- Holt et al., 2013. Holt K.E., Thieu Nga T.V., Thanh D.P., Vinh H., Kim D.W., Vu Tra M.P., Campbell J.I., Hoang N.V.M., Vinh N.T., other authors (2013). Tracking the establishment of local endemic populations of an emergent enteric pathogen Proc Natl Acad Sci U S A 110 17522–17527 10.1073/pnas.1308632110 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu et al., 2008. Hu F.P., Xu X.G., Zhu D.M., Wang M.G. (2008). Coexistence of qnrB4 qnrS1 in a clinical strain of Klebsiella pneumoniae Acta Pharmacol Sin 29 320–324 10.1111/j.1745-7254.2008.00757.x . [DOI] [PubMed] [Google Scholar]
- Huang et al., 2008. Huang Z., Mi Z., Wang C. (2008). A novel β-lactamase gene, LAP-2, produced by an Enterobacter cloacae clinical isolate in China J Hosp Infect 70 95–96 10.1016/j.jhin.2008.04.012 . [DOI] [PubMed] [Google Scholar]
- Jacoby et al., 2006. Jacoby G.A., Walsh K.E., Mills D.M., Walker V.J., Oh H., Robicsek A., Hooper D.C. (2006). qnrB, another plasmid-mediated gene for quinolone resistance Antimicrob Agents Chemother 50 1178–1182 10.1128/AAC.50.4.1178-1182.2006 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kado C.I., Liu S.T. (1981). Rapid procedure for detection and isolation of large and small plasmids J Bacteriol 145 1365–1373 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kehrenberg et al., 2006. Kehrenberg C., Friederichs S., de Jong A., Michael G.B., Schwarz S. (2006). Identification of the plasmid-borne quinolone resistance gene qnrS in Salmonella enterica serovar Infantis J Antimicrob Chemother 58 18–22 10.1093/jac/dkl213 . [DOI] [PubMed] [Google Scholar]
- Kehrenberg et al., 2007. Kehrenberg C., Hopkins K.L., Threlfall E.J., Schwarz S. (2007). Complete nucleotide sequence of a small qnrS1-carrying plasmid from Salmonella enterica subsp. enterica Typhimurium DT193 J Antimicrob Chemother 60 903–905 10.1093/jac/dkm283 . [DOI] [PubMed] [Google Scholar]
- Kim E.H., Aoki T. (1994). The transposon-like structure of IS26-tetracycline, and kanamycin resistance determinant derived from transferable R plasmid of fish pathogen, Pasteurella piscicida Microbiol Immunol 38 31–38 10.1111/j.1348-0421.1994.tb01741.x . [DOI] [PubMed] [Google Scholar]
- Kobayashi I. (2001). Behavior of restriction-modification systems as selfish mobile elements and their impact on genome evolution Nucleic Acids Res 29 3742–3756 10.1093/nar/29.18.3742 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lavigne et al., 2006. Lavigne J-P., Marchandin H., Delmas J., Bouziges N., Lecaillon E., Cavalie L., Jean-Pierre H., Bonnet R., Sotto A. (2006). qnrA in CTX-M-producing Escherichia coli isolates from France Antimicrob Agents Chemother 50 4224–4228 10.1128/AAC.00904-06 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Literak et al., 2010. Literak I., Dolejska M., Janoszowska D., Hrusakova J., Meissner W., Rzyska H., Bzoma S., Cizek A. (2010). Antibiotic-resistant Escherichia coli bacteria, including strains with genes encoding the extended-spectrum β-lactamase and QnrS, in waterbirds on the Baltic Sea Coast of Poland Appl Environ Microbiol 76 8126–8134 10.1128/AEM.01446-10 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lodwick et al., 1990. Lodwick D., Owen D., Strike P. (1990). DNA sequence analysis of the imp UV protection and mutation operon of the plasmid TP110: identification of a third gene Nucleic Acids Res 18 5045–5050 10.1093/nar/18.17.5045 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martínez-Martínez L., Pascual A., Jacoby G.A. (1998). Quinolone resistance from a transferable plasmid Lancet 351 797–799 10.1016/S0140-6736(97)07322-4 . [DOI] [PubMed] [Google Scholar]
- Nga et al., 2012. Nga T.V.T., Parry C.M., Le T., Lan N.P.H., Diep T.S., Campbell J.I., Hoang N.V.M., Dung T., Wain J., other authors (2012). The decline of typhoid and the rise of non-typhoid salmonellae and fungal infections in a changing HIV landscape: bloodstream infection trends over 15 years in southern Vietnam Trans R Soc Trop Med Hyg 106 26–34 10.1016/j.trstmh.2011.10.004 . [DOI] [PubMed] [Google Scholar]
- Nhu et al., 2014. Nhu N.T.K., Lan N.P.H., Campbell J.I., Parry C.M., Thompson C., Tuyen H.T., Hoang N.V.M., Tam P.T.T., Le V.M., other authors (2014). Emergence of carbapenem-resistant Acinetobacter baumannii as the major cause of ventilator-associated pneumonia in intensive care unit patients at an infectious disease hospital in southern Vietnam J Med Microbiol 63 1386–1394 10.1099/jmm.0.076646-0 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park et al., 2009. Park Y.-J., Yu J.K., Kim S.-I., Lee K., Arakawa Y. (2009). Accumulation of plasmid-mediated fluoroquinolone resistance genes, qepA qnrS1, in Enterobacter aerogenes co-producing RmtB and class A β-lactamase LAP-1 Ann Clin Lab Sci 39 55–59 . [PubMed] [Google Scholar]
- Parkhill et al., 2001. Parkhill J., Wren B.W., Thomson N.R., Titball R.W., Holden M.T., Prentice M.B., Sebaihia M., James K.D., Churcher C., other authors (2001). Genome sequence of Yersinia pestis, the causative agent of plague Nature 413 523–527 10.1038/35097083 . [DOI] [PubMed] [Google Scholar]
- Poirel et al., 2006. Poirel L., Leviandier C., Nordmann P. (2006). Prevalence and genetic analysis of plasmid-mediated quinolone resistance determinants QnrA and QnrS in Enterobacteriaceae isolates from a French university hospital Antimicrob Agents Chemother 50 3992–3997 10.1128/AAC.00597-06 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poirel et al., 2007. Poirel L., Cattoir V., Soares A., Soussy C.-J., Nordmann P. (2007). Novel Ambler class A β-lactamase LAP-1 and its association with the plasmid-mediated quinolone resistance determinant QnrS1 Antimicrob Agents Chemother 51 631–637 10.1128/AAC.01082-06 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rutherford et al., 2000. Rutherford K., Parkhill J., Crook J., Horsnell T., Rice P., Rajandream M.A., Barrell B. (2000). Artemis: sequence visualization and annotation Bioinformatics 16 944–945 10.1093/bioinformatics/16.10.944 . [DOI] [PubMed] [Google Scholar]
- Som et al., 1987. Som S., Bhagwat A.S., Friedman S. (1987). Nucleotide sequence and expression of the gene encoding the EcoRII modification enzyme Nucleic Acids Res 15 313–332 10.1093/nar/15.1.313 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strahilevitz et al., 2009. Strahilevitz J., Jacoby G.A., Hooper D.C., Robicsek A. (2009). Plasmid-mediated quinolone resistance: a multifaceted threat Clin Microbiol Rev 22 664–689 10.1128/CMR.00016-09 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sumrall et al., 2014. Sumrall E.T., Gallo E.B., Aboderin A.O., Lamikanra A., Okeke I.N. (2014). Dissemination of the transmissible quinolone-resistance gene qnrS1 by IncX plasmids in Nigeria PLoS One 9 e110279 10.1371/journal.pone.0110279 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sundin G.W., Bender C.L. (1996). Dissemination of the strA-strB streptomycin-resistance genes among commensal and pathogenic bacteria from humans, animals, and plants Mol Ecol 5 133–143 10.1111/j.1365-294X.1996.tb00299.x . [DOI] [PubMed] [Google Scholar]
- Tran et al., 2010. Tran T.H., Nguyen T.D., Nguyen T.T., Ninh T.T., Tran N.B.C., Nguyen V.M.H., Tran T.T.N., Cao T.T., Pham V.M., other authors (2010). A randomised trial evaluating the safety and immunogenicity of the novel single oral dose typhoid vaccine M01ZH09 in healthy Vietnamese children PLoS One 5 e11778 10.1371/journal.pone.0011778 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vien et al., 2009. Vien T.M., Baker S., Le Thi T.P., Le Thi T.P., Cao T.T., Tran T.T.N., Nguyen V.M.H., Campbell J.I., Lam M.Y., other authors (2009). High prevalence of plasmid-mediated quinolone resistance determinants in commensal members of the Enterobacteriaceae in Ho Chi Minh City, Vietnam J Med Microbiol 58 1585–1592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vien et al., 2011. Vien T.M., Abuoun M., Morrison V., Thomson N., Campbell J.I., Woodward M.J., Van Vinh Chau N., Farrar J., Schultsz C., Baker S. (2011). Differential phenotypic and genotypic characteristics of qnrS1-harboring plasmids carried by hospital and community commensal Enterobacteria Antimicrob Agents Chemother 55 1798–1802 10.1128/AAC.01200-10 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vien et al., 2012. Vien T.M., Minh N.N.Q., Thuong T.C., Khuong H.D., Nga T.V.T., Thompson C., Campbell J.I., de Jong M., Farrar J.J., other authors (2012). The co-selection of fluoroquinolone resistance genes in the gut flora of Vietnamese children PLoS One 7 e42919 10.1371/journal.pone.0042919 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang et al., 2009. Wang M., Guo Q., Xu X., Wang X., Ye X., Wu S., Hooper D.C., Wang M. (2009). New plasmid-mediated quinolone resistance gene, qnrC, found in a clinical isolate of Proteus mirabilis Antimicrob Agents Chemother 53 1892–1897 10.1128/AAC.01400-08 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu et al., 2008. Wu J.-J., Ko W.-C., Chiou C.-S., Chen H.-M., Wang L.-R., Yan J.-J. (2008). Emergence of Qnr determinants in human Salmonella isolates in Taiwan J Antimicrob Chemother 62 1269–1272 10.1093/jac/dkn426 . [DOI] [PubMed] [Google Scholar]
- Zhao et al., 2010. Zhao F., Bai J., Wu J., Liu J., Zhou M., Xia S., Wang S., Yao X., Yi H., other authors (2010). Sequencing and genetic variation of multidrug resistance plasmids in Klebsiella pneumoniae PLoS One 5 e10141 10.1371/journal.pone.0010141 . [DOI] [PMC free article] [PubMed] [Google Scholar]