Abstract
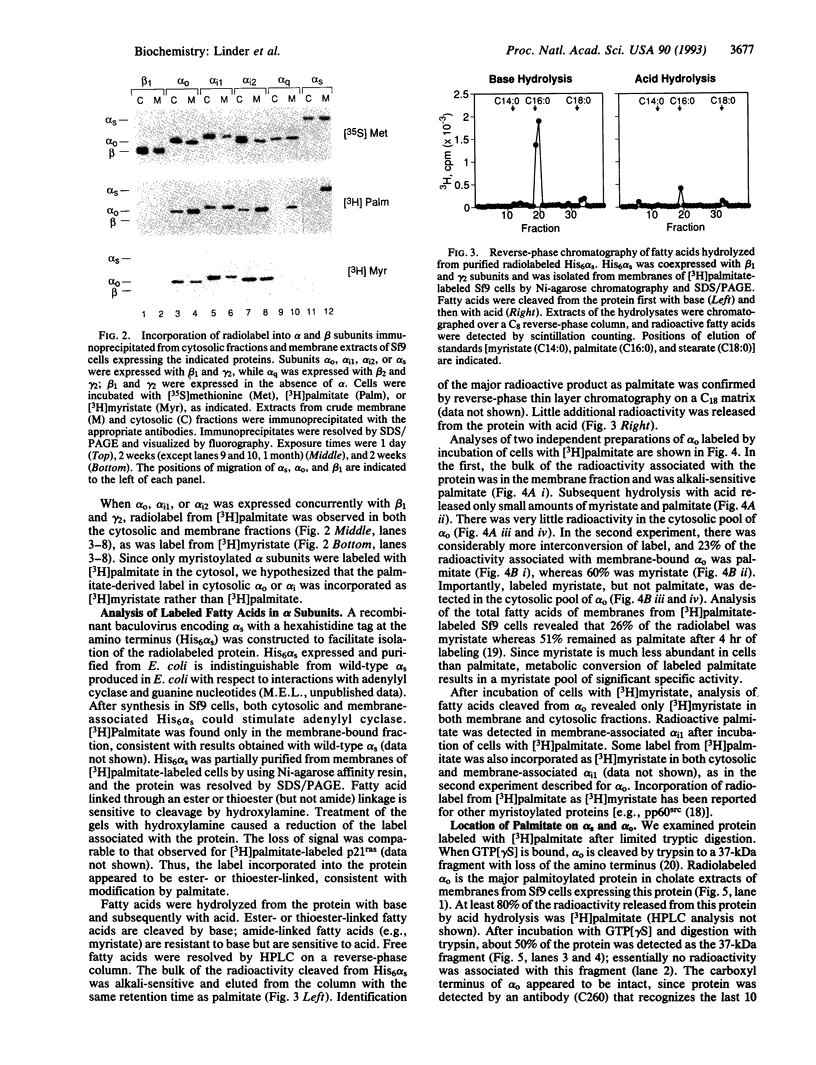
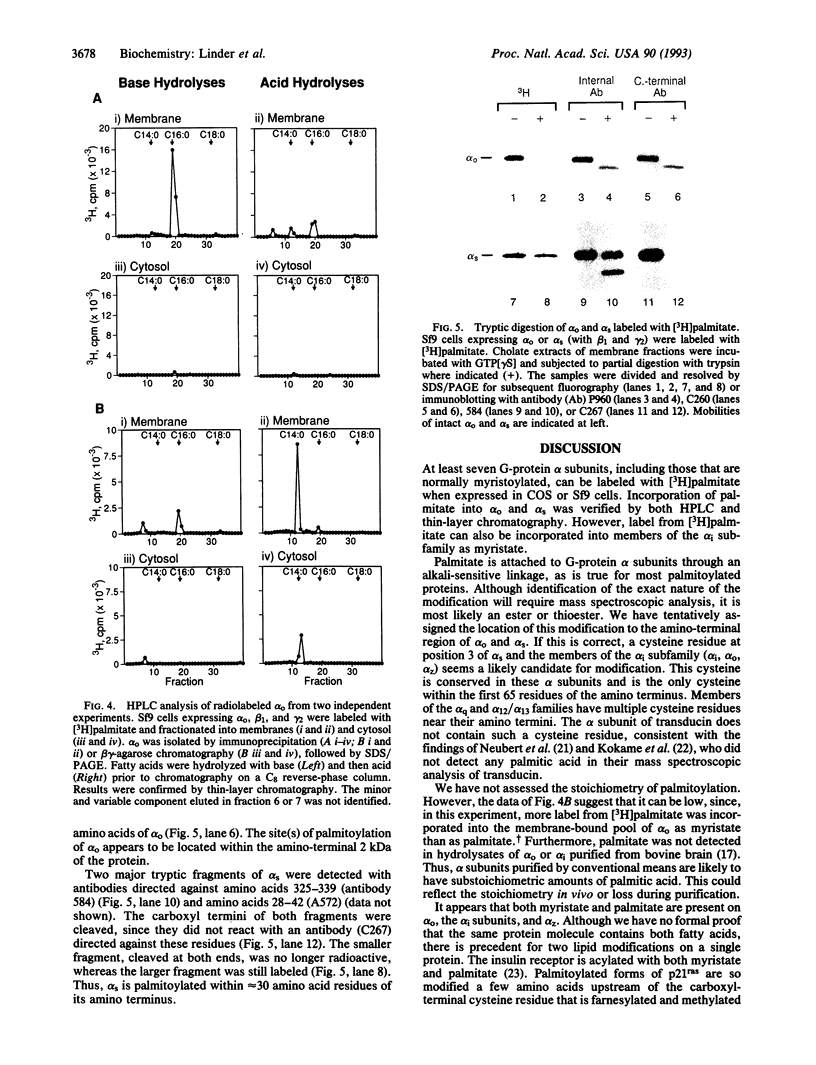
A small number of membrane-associated proteins are reversibly and covalently modified with palmitic acid. Palmitoylation of G-protein alpha and beta subunits was assessed by metabolic labeling of subunits expressed in simian COS cells or insect Sf9 cells. The fatty acid was incorporated into all of the alpha subunits examined (alpha s, alpha o, alpha i1, alpha i2, alpha i3, alpha z, and alpha q), including those that are also myristoylated (alpha o, alpha i, and alpha z). Palmitate was released by treatment with base, suggesting attachment to the protein through a thioester or ester bond. Limited tryptic digestion of activated alpha o and alpha s resulted in release of the amino-terminal portions of the proteins and radioactive palmitate. These data are consistent with palmitoylation of the proteins near their amino termini, most likely on Cys-3. Reversible acylation of G-protein alpha subunits may provide an additional mechanism for regulation of signal transduction.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Audigier Y., Journot L., Pantaloni C., Bockaert J. The carboxy-terminal domain of Gs alpha is necessary for anchorage of the activated form in the plasma membrane. J Cell Biol. 1990 Oct;111(4):1427–1435. doi: 10.1083/jcb.111.4.1427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buss J. E., Mumby S. M., Casey P. J., Gilman A. G., Sefton B. M. Myristoylated alpha subunits of guanine nucleotide-binding regulatory proteins. Proc Natl Acad Sci U S A. 1987 Nov;84(21):7493–7497. doi: 10.1073/pnas.84.21.7493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buss J. E., Sefton B. M. Myristic acid, a rare fatty acid, is the lipid attached to the transforming protein of Rous sarcoma virus and its cellular homolog. J Virol. 1985 Jan;53(1):7–12. doi: 10.1128/jvi.53.1.7-12.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Christgau S., Aanstoot H. J., Schierbeck H., Begley K., Tullin S., Hejnaes K., Baekkeskov S. Membrane anchoring of the autoantigen GAD65 to microvesicles in pancreatic beta-cells by palmitoylation in the NH2-terminal domain. J Cell Biol. 1992 Jul;118(2):309–320. doi: 10.1083/jcb.118.2.309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Graber S. G., Figler R. A., Garrison J. C. Expression and purification of functional G protein alpha subunits using a baculovirus expression system. J Biol Chem. 1992 Jan 15;267(2):1271–1278. [PubMed] [Google Scholar]
- Graziano M. P., Freissmuth M., Gilman A. G. Expression of Gs alpha in Escherichia coli. Purification and properties of two forms of the protein. J Biol Chem. 1989 Jan 5;264(1):409–418. [PubMed] [Google Scholar]
- Gutowski S., Smrcka A., Nowak L., Wu D. G., Simon M., Sternweis P. C. Antibodies to the alpha q subfamily of guanine nucleotide-binding regulatory protein alpha subunits attenuate activation of phosphatidylinositol 4,5-bisphosphate hydrolysis by hormones. J Biol Chem. 1991 Oct 25;266(30):20519–20524. [PubMed] [Google Scholar]
- Hancock J. F., Magee A. I., Childs J. E., Marshall C. J. All ras proteins are polyisoprenylated but only some are palmitoylated. Cell. 1989 Jun 30;57(7):1167–1177. doi: 10.1016/0092-8674(89)90054-8. [DOI] [PubMed] [Google Scholar]
- Hedo J. A., Collier E., Watkinson A. Myristyl and palmityl acylation of the insulin receptor. J Biol Chem. 1987 Jan 25;262(3):954–957. [PubMed] [Google Scholar]
- Hurley J. B., Simon M. I., Teplow D. B., Robishaw J. D., Gilman A. G. Homologies between signal transducing G proteins and ras gene products. Science. 1984 Nov 16;226(4676):860–862. doi: 10.1126/science.6436980. [DOI] [PubMed] [Google Scholar]
- Iñiguez-Lluhi J. A., Simon M. I., Robishaw J. D., Gilman A. G. G protein beta gamma subunits synthesized in Sf9 cells. Functional characterization and the significance of prenylation of gamma. J Biol Chem. 1992 Nov 15;267(32):23409–23417. [PubMed] [Google Scholar]
- James G., Olson E. N. Fatty acylated proteins as components of intracellular signaling pathways. Biochemistry. 1990 Mar 20;29(11):2623–2634. doi: 10.1021/bi00463a001. [DOI] [PubMed] [Google Scholar]
- Journot L., Pantaloni C., Poul M. A., Mazarguil H., Bockaert J., Audigier Y. Amino acids 367-376 of the Gs alpha subunit induce membrane association when fused to soluble amino-terminal deleted Gi1 alpha subunit. Proc Natl Acad Sci U S A. 1991 Nov 15;88(22):10054–10058. doi: 10.1073/pnas.88.22.10054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Juhnn Y. S., Jones T. L., Spiegel A. M. Amino- and carboxy-terminal deletion mutants of Gs alpha are localized to the particulate fraction of transfected COS cells. J Cell Biol. 1992 Nov;119(3):523–530. doi: 10.1083/jcb.119.3.523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kokame K., Fukada Y., Yoshizawa T., Takao T., Shimonishi Y. Lipid modification at the N terminus of photoreceptor G-protein alpha-subunit. Nature. 1992 Oct 22;359(6397):749–752. doi: 10.1038/359749a0. [DOI] [PubMed] [Google Scholar]
- Lee E., Taussig R., Gilman A. G. The G226A mutant of Gs alpha highlights the requirement for dissociation of G protein subunits. J Biol Chem. 1992 Jan 15;267(2):1212–1218. [PubMed] [Google Scholar]
- Levis M. J., Bourne H. R. Activation of the alpha subunit of Gs in intact cells alters its abundance, rate of degradation, and membrane avidity. J Cell Biol. 1992 Dec;119(5):1297–1307. doi: 10.1083/jcb.119.5.1297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mattera R., Graziano M. P., Yatani A., Zhou Z., Graf R., Codina J., Birnbaumer L., Gilman A. G., Brown A. M. Splice variants of the alpha subunit of the G protein Gs activate both adenylyl cyclase and calcium channels. Science. 1989 Feb 10;243(4892):804–807. doi: 10.1126/science.2536957. [DOI] [PubMed] [Google Scholar]
- Mumby S. M., Gilman A. G. Synthetic peptide antisera with determined specificity for G protein alpha or beta subunits. Methods Enzymol. 1991;195:215–233. doi: 10.1016/0076-6879(91)95168-j. [DOI] [PubMed] [Google Scholar]
- Mumby S. M., Heukeroth R. O., Gordon J. I., Gilman A. G. G-protein alpha-subunit expression, myristoylation, and membrane association in COS cells. Proc Natl Acad Sci U S A. 1990 Jan;87(2):728–732. doi: 10.1073/pnas.87.2.728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Negishi M., Hashimoto H., Ichikawa A. Translocation of alpha subunits of stimulatory guanine nucleotide-binding proteins through stimulation of the prostacyclin receptor in mouse mastocytoma cells. J Biol Chem. 1992 Feb 5;267(4):2364–2369. [PubMed] [Google Scholar]
- Neubert T. A., Johnson R. S., Hurley J. B., Walsh K. A. The rod transducin alpha subunit amino terminus is heterogeneously fatty acylated. J Biol Chem. 1992 Sep 15;267(26):18274–18277. [PubMed] [Google Scholar]
- Pang I. H., Sternweis P. C. Isolation of the alpha subunits of GTP-binding regulatory proteins by affinity chromatography with immobilized beta gamma subunits. Proc Natl Acad Sci U S A. 1989 Oct;86(20):7814–7818. doi: 10.1073/pnas.86.20.7814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ransnäs L. A., Insel P. A. Subunit dissociation is the mechanism for hormonal activation of the Gs protein in native membranes. J Biol Chem. 1988 Nov 25;263(33):17239–17242. [PubMed] [Google Scholar]
- Simon M. I., Strathmann M. P., Gautam N. Diversity of G proteins in signal transduction. Science. 1991 May 10;252(5007):802–808. doi: 10.1126/science.1902986. [DOI] [PubMed] [Google Scholar]
- Skene J. H., Virág I. Posttranslational membrane attachment and dynamic fatty acylation of a neuronal growth cone protein, GAP-43. J Cell Biol. 1989 Feb;108(2):613–624. doi: 10.1083/jcb.108.2.613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spiegel A. M., Backlund P. S., Jr, Butrynski J. E., Jones T. L., Simonds W. F. The G protein connection: molecular basis of membrane association. Trends Biochem Sci. 1991 Sep;16(9):338–341. doi: 10.1016/0968-0004(91)90139-m. [DOI] [PubMed] [Google Scholar]
- Zuber M. X., Strittmatter S. M., Fishman M. C. A membrane-targeting signal in the amino terminus of the neuronal protein GAP-43. Nature. 1989 Sep 28;341(6240):345–348. doi: 10.1038/341345a0. [DOI] [PubMed] [Google Scholar]