Abstract
Wild and domesticated Atlantic salmon males display large variation for sea age at sexual maturation, which varies between 1–5 years. Previous studies have uncovered a genetic predisposition for variation of age at maturity with moderate heritability, thus suggesting a polygenic or complex nature of this trait. The aim of this study was to identify associated genetic loci, genes and ultimately specific sequence variants conferring sea age at maturity in salmon. We performed a genome wide association study (GWAS) using a pool sequencing approach (20 individuals per river and phenotype) of male salmon returning to rivers as sexually mature either after one sea winter (2009) or three sea winters (2011) in six rivers in Norway. The study revealed one major selective sweep, which covered 76 significant SNPs in which 74 were found in a 370 kb region of chromosome 25. Genotyping other smolt year classes of wild and domesticated salmon confirmed this finding. Genotyping domesticated fish narrowed the haplotype region to four SNPs covering 2386 bp, containing the vgll3 gene, including two missense mutations explaining 33–36% phenotypic variation. A single locus was found to have a highly significant role in governing sea age at maturation in this species. The SNPs identified may be both used as markers to guide breeding for late maturity in salmon aquaculture and in monitoring programs of wild salmon. Interestingly, a SNP in proximity of the VGLL3 gene in humans (Homo sapiens), has previously been linked to age at puberty suggesting a conserved mechanism for timing of puberty in vertebrates.
Author Summary
For most species the factors that contribute to the genetic predisposition for age at maturity are currently unknown. In salmon aquaculture early maturation is negative for the growth, disease resistance and flesh quality. In addition, using populations of salmon selected to mature late may limit the genetic impact of aquaculture escapees, as these late maturing fish are more likely to die before they reach maturity. The aim of this study was to elucidate the genetic predisposition for salmon maturation. We determined the sequences of genomes from Atlantic salmon maturing early and late in six Norwegian rivers. This methodology enabled us to identify a short genomic region involved in determining the age at maturity in male Atlantic salmon. This region has also previously been linked to time of puberty in humans–supporting a general mechanism behind age at maturity in vertebrates. The results of this study may be used to breed salmon that are genetically predisposed to mature late which will improve welfare and production in aquaculture industry and aid in the management of escaped farmed salmon.
Introduction
Both wild and domesticated populations of Atlantic salmon (Salmo salar L.) show large phenotypic variation for sea age at sexual maturity [1]. Salmon males can stay in the sea 1–5 years before they initiate sexual maturation and return to their native river to spawn, while females usually return to the river after 1–3 years in the sea. In aquaculture the variation in age at sexual maturation is a remaining problem since precocious puberty in males results in negative effects on somatic growth, flesh quality, animal welfare and susceptibility to disease [1]. Early maturation in farmed salmon can also increase the risk of genetic introgression of escaped salmon in wild populations [2,3], as maturing fish will have a higher likelihood of migrating to a nearby river to spawn. Immature fish on the other hand will more likely migrate to sea where mortality is high before reaching maturity [4].
Salmonids in general display moderately high heritability (up to H2 p = 0.39) for age at sexual maturation [5–9] and QTLs relating to this trait have been identified previously [10]. Also three recent papers used single nucleotide polymorphism (SNP) arrays to identify markers associated with sea age at puberty in an aquaculture strain using a low density SNP array [11,12] and regions under selection in wild populations using a high density SNP array [13]. These three reports revealed association of the trait to multiple loci but gave no clear answer regarding possible mechanisms, genes and genomic regions behind age at puberty. Previous studies screened the genome for loci under selection using a limited set of SNPs, which may exclude the causative variants [14]. The recent sequencing of the Atlantic salmon genome ([15], AKGD00000000.4) provides an opportunity for large-scale mapping and comparison of parallel sequencing reads on to the published genome assembly for the species, thereby enabling genome-wide detection not only of novel SNPs, but also small indels and structural variation [16]. Hence, the use of sequencing allows prediction of how genetic variants affect regulatory regions and genes, which may link traits with new biological mechanisms and provide opportunities for subsequent functional studies.
This study aimed to elucidate the genes and genomic regions that regulate sea age at puberty in male Atlantic salmon. Males were chosen since the mechanisms of maturation may differ between the sexes, and because precocious maturation of males in aquaculture represents a significant challenge in production of this species. To investigate this trait, we performed genome resequencing of scale samples from sexually mature wild salmon from six rivers in Western Norway, returning either after one or three years at sea (Fig 1).Using this approach we identified a region on chromosome 25 (Chr 25) harboring a dense set of significant SNPs in a stretch of 370 kb. These results were also confirmed in other year classes of wild salmon and in domesticated salmon that had been reared under controlled aquaculture conditions. In conclusion, we show for the first time the importance of one single genomic region in determining age at maturity in male salmon.
Fig 1. Geographical location of salmon rivers used.
Map of Norway and a magnification of Western Norway showing rivers used in the experiment including three rivers in Sogn og Fjordane county; Eidselven, Gloppenelven and Flekkeelven, and three rivers in Rogaland county; Suldalslågen, Vormo and Årdalselven. Derivative of https://commons.wikimedia.org/wiki/File:Norway_municipalities_2012_blank.svg, licensed with CC-BY-SA-2.0.
Results and Discussion
To find SNPs associated with age at maturation in salmon males, we sequenced 20 salmon per river and sea winter age (1SW and 3SW). This number of individuals in each pool has been shown to be sufficient to identify causative SNPs for a trait in Drosophila melanogaster [17]. Mapping our data yielded a 12.32X mean coverage (0.24 SE) of unique mapped reads per river and sea age at puberty (S1 Fig). This depth of coverage is similar to what has been used in other successful genome wide association studies (GWAS) by pool sequencing in vertebrates, including pig (Sus scrofa) and chicken (Gallus gallus) [18,19]. We have mapped the salmon sequences to the most recent salmon genome assembly (AKGD00000000.4). Within this assembly, 34% of the genome has not been assigned to chromosomes, probably due to a high number of repetitive sequences and the partially tetraploid nature of the genome [20]. This probably also explains why the unassigned part of the genome harbored only 1% of our uniquely mapped reads (S1 Fig). SNP calling revealed altogether 4,326,591 SNPs in all sea ages and rivers, the data has been deposited at http://marineseq.imr.no/salmatsnp/.
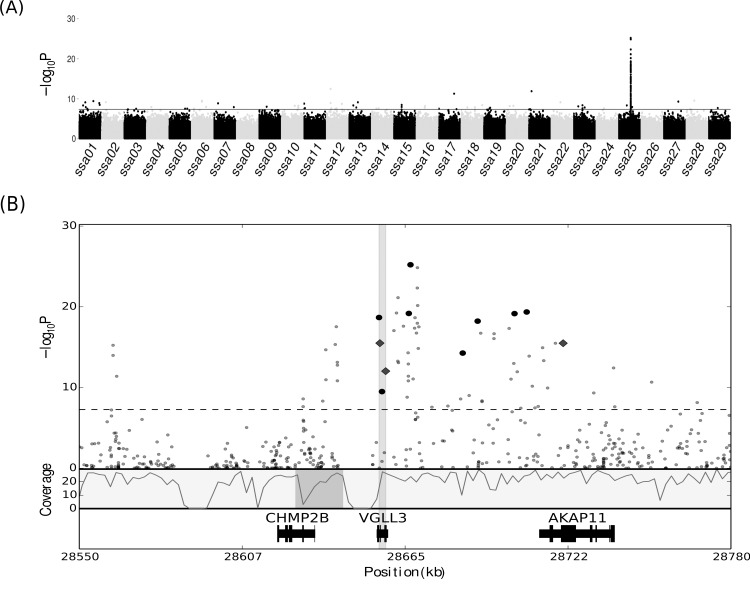
Comparing 1SW and 3SW allele frequencies using the Cochran-Mantel-Haenszel (CMH) test in 4,326,591 SNPs revealed 138 SNPs that were significantly associated (0.1% FDR) with sea age at puberty (Fig 2A and S1 Table). Several single significant SNP associations with the phenotype were detected on chromosomes 1–7, 9–24 and 27–29, although these were not found to be among the most significant SNPs (Fig 2A and S1 Table). None of the loci harboring single significant SNPs were further assessed as candidate loci since the power of sequencing pools increases with numbers of reads assessed. Although we cannot rule out true association of single SNPs with maturation we regard such signals as likely false positives. In a previous QTL study for precocious parr maturation the trait was shown to be linked to Chr 12 [21]. Chr 12 has also been associated with sea age at maturation in another study [22]. In a GWAS, using a 6.5 kb SNP chip, the trait of 1SW maturation or “grilsing” was found to be weakly linked to both Chr 12 and Chr 25 [11]. In our study, 74 of the 138 (48%) SNPs associated significantly with the trait were located in a region on Chr 25, covering ~370kb (Fig 2B). From our data we conclude that in Western Norway a single selective sweep on Chr 25 has had a large effect on sea age at maturity while other regions in the genome might contribute to a lesser degree. This is in contrast to earlier reports showing a more polygenic nature of this trait, with contributions from several genomic regions [11,12,22]. A previous theoretical based model study also suggested that age at maturity could be regulated by a stable genetic polymorphism, in accordance with our current findings [23].
Fig 2. Identification of a selective region conferring age at maturity in Atlantic salmon.
(A) Manhattan plot of SNPs associated with age at maturity. The x-axis presents genomic coordinates along chromosomes 1–29 in Atlantic salmon. On the y-axis the negative logarithm of the SNPs associated p-value is displayed. All SNPs above the solid horizontal line in the plot are significantly (p < 5.2e-8, 0.1% FDR) associated with the trait. (B) Magnification of the 370kb region showing only 230 kb of the region on Chr 25 (28550–28780 kb) including 62 significant SNPs. The SNPs are represented by black dots, where the 11 large dots indicate SNPs used in genotyping assays. The square dots indicate three missense mutations in vgll3 and akap11. On the upper y-axis the negative logarithm of the SNPs associated p-value is displayed. The dashed line within the plot indicates the significance threshold (0.1% FDR). Below the SNP illustration, the lower y-axis shows average read depth of coverage (using 2 kb windows). Genomic organization of the three genes found in the region is illustrated in the bottom track. The x-axis shows the location of the region in Chr 25 and covers 28550–28780 kb. The grey area around vgll3 demarcates the shorter region discovered in the domesticated strain. The dark grey area in the coverage track is showing the misplaced contig in the most recent salmon genome release containing exon 1 and 2 of chmp2b.
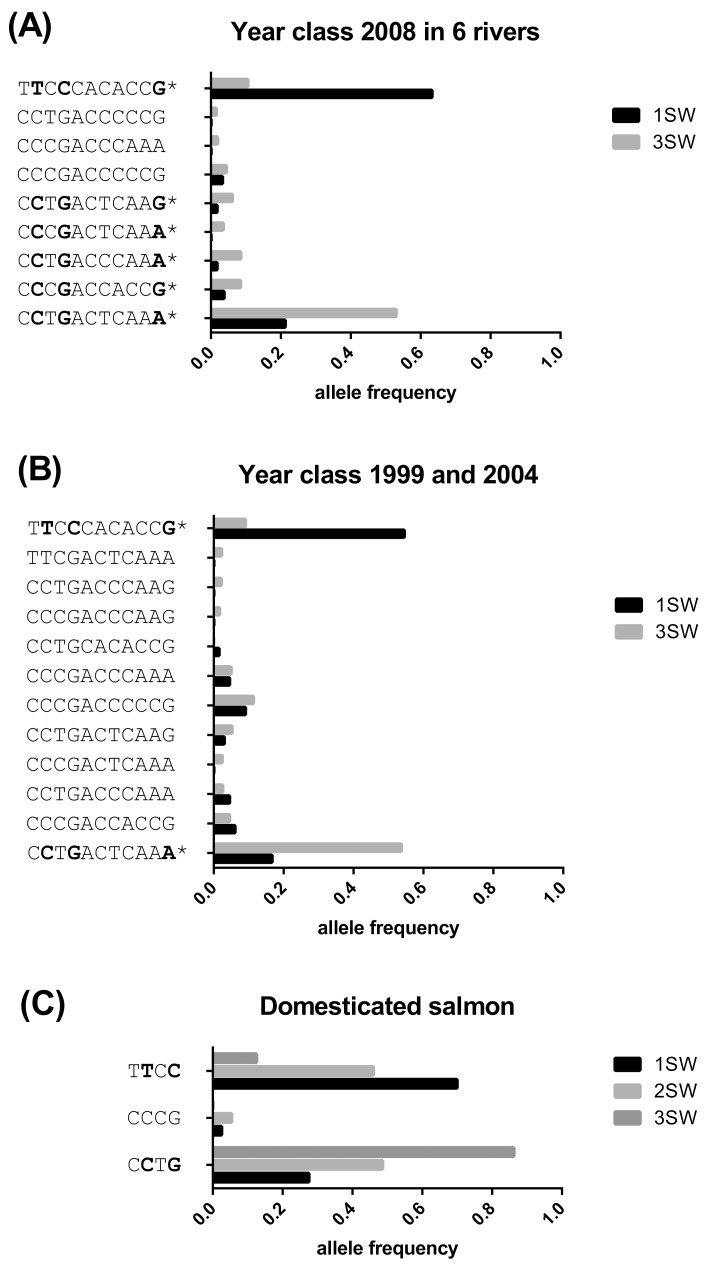
To verify the GWAS findings and to ascertain whether genotypes of single individuals for SNPs are associated with sea age at maturity we designed a Sequenom assay for 11 of the most significant SNPs in the selective sweep found in Chr 25 (S2 Table). Genotyping of all 240 individuals included in the sequenced pools used in GWAS confirmed a strong association between allele frequencies and age at maturity (S2 Fig). To characterize haplotypes using the 11 assayed SNPs, we performed a pairwise disequilibrium analysis on all samples that had been sequenced [24]. This analysis revealed two dissimilar haplogroups comprising 11 haplotypes in one block (Fig 3A). One and five of these haplotypes showed significant association with maturing early and late, respectively. The significant 1SW haplotype explained 54% (β-value -1.0, p-value = 3.88e-40) of the phenotypic variance for this trait. The most significant 3SW haplotype explained 21% (β-value 0.66, p-value = 3.98e-13) of the variation in age at maturity, the other four 3 SW haplotypes explained the 1.9, 2.2, 3.6 and 3.7%, adding up to 32.4% of the variance of the age at maturity in 3SW haplotypes. The genotyping data clearly confirmed our findings from the pool re-sequencing and further supported that this locus exerted a large effect on the trait.
Fig 3. Haplotype frequencies in different year classes in wild and domesticated salmon.
(A) Haplotype frequency associated with either 1SW (black bars) or 3SW (dark grey bars) in male Atlantic salmon for six rivers in Western Norway from year class 2008. (B) Haplotype frequencies associated with either 1SW (black bars) or 3SW (dark grey bars) male Atlantic salmon in year classes 1999 from Eidselva and 2004 from Suldalslågen. (C) Haplotype frequencies linked to age at maturity in the domesticated Mowi strain maturing after 1 (black bars), 2 (grey bars) or ≥ 3 (dark grey bars) years in sea water. In all graphs the x-axis indicates frequency of that trait for the identified haplotype, while the y-axis presents the haplotype block obtained from the genotype assay. * Indicates that the haplotype was significantly linked to the trait. The bold bases in the haplotypes are indicating missense mutations.
In samples from the pool sequencing we identified haplotypes associated with sea age at maturity in the 2008 year class (year of migration to sea). These fish have possibly been exposed to similar environmental conditions during their early stay in the sea, therefore showing a selection for those conditions as postulated by several previous studies in salmon [25–27]. To investigate this we identified genotypes using the SNP assays in other year classes: 1999 for Eidselva (20 1SW, 8 3SW) and 2004 for Suldalslågen (13 1SW, 13 3SW). Allele frequencies derived from the 11 SNP assays showed correlation to the 1SW and 3SW trait also in these year classes (S3 Fig). Haplotype association analysis of these year classes again revealed two significant haplotypes also found in the 2008 year class (Fig 3B). In the 1999 and 2004 year classes 44% (β-value -0.96, p-value = 3.77e-08) and 22% (β-value 0.66, p-value = 2.85e-04) of the phenotypic variation for age at maturity was explained by the 1SW or the 3SW haplotype, respectively. We thus conclude that genotypes at a single locus strongly influence sea age at maturity independent of year class across multiple salmon populations in Norway.
Sea age at maturity can be significantly altered in salmon by modulating both light and temperature [1,28,29]. As a consequence, current aquaculture production methods include the use of constant light during the winter months to inhibit or reduce the incidence of early sexual maturation. The use of photoperiod to inhibit maturation in Norwegian farming has thereby masked the impact of this trait in commercial production. We were also interested to see how much the identified genetic trait contributed to the sea age at maturity trait in domesticated farmed salmon males, since wild salmon live in a different environment including different feed availability and water temperature that may trigger time of male puberty differently. To assay the linkage between phenotype and genotype for sea age at maturity in a domesticated strain, we utilized DNA from sexually maturing salmon from four different families of the Mowi strain. This strain has been in aquaculture for at least ten generations and has been selected for a variety of traits including growth and late maturation [30–32]. Mowi was originally obtained from a range of large wild salmon populations from Western Norway in 1969, and has later been bred using a four-year life cycle. The long life cycle breeding has thereby probably increased the allele frequency for the late maturity phenotype. In this common garden experiment using the Mowi strain, fish were grown under natural light conditions in sea cages where males were matured after 1, 2 or 3 or more years in sea. Haplotype analysis of these fish (n = 97) revealed a shorter haplotype, consisting only of four SNPs, covering only 2386 bp in the 5’ end of the region assayed (Fig 3C and S4 Fig). The observed differences between wild and domesticated fish may be due to the domestication process in this strain. These data clearly demonstrate that age at puberty can be explained by SNPs in this region also in a domesticated strain in culture for more than ten generations.
Altogether the experiments clearly show that the selective sweep on Chr 25 significantly contributes to sea age at maturity both in wild and domesticated male salmon. Gene prediction in this area revealed three genes; charged multivesicular protein 2B (chmp2B), vestigial-like protein 3 (vgll3) and a-kinase anchor protein 11 (akap11, Fig 2B). From the analysis of domesticated salmon we could decrease the area of selection to a 2.4 kb region covering only vgll3. For this locus we could identify loci containing paralogous genes. Two such loci were found tandemly repeated in Chr 21. To assay if these two identical regions had SNPs associated with sea age at maturity we had to manually inspect both regions, using ambiguously mapped reads. No SNPs associated with the trait were discovered within these two paralogous regions. From our genotyping assay on domesticated samples we could with certainty reduce the region under selection in the downstream region of the vgll3 locus since we had genotyped several SNPs in this area. We can however not exclude that the upstream region of the vgll3 locus contained SNPs contributing to the haplotype since this area was not represented in our genotyping assay due to a large gap in the genome at this region (Fig 2B). The 2.4 kb region contained two missense mutations in vgll3; at amino acid (aa) 54 and 323. The sea age at maturity trait was strongly associated to the genotype of these SNPs since 36% (nt. 28656101 Chr25, β-value -0.61, p-value = 9.80e-07) and 33% (nt. 28658151 Chr 25, β-value -0.60, p-value = 3.77e-08) of the phenotypic variation could be explained by the genotype. The haplotypes associated with the 3SW trait encode a Thr and a Lys at these positions whereas the haplotype associated with 1SW encodes a Met and an Asp. Our analysis could not conclude whether these missense mutations are causative for the sea age at maturity trait, but since they occurred consistently together in the material we cannot rule out whether both or other non-coding variants at this locus are involved in age at maturity phenotype. It is also known from other studies that co-occurring amino acid changes can confer a phenotype [33]. The Vgll3 protein functions as a cofactor for the TEA Domain (TEAD) family of transcription factors [34]. The transcription factor binding region spanning aa105-aa134 in Vgll3 does not include any of the aa changes discovered which suggests that any direct binding differences between 1SW or 3SW fish are unlikely. It is thus difficult to predict how these amino acid changes affect the protein. At this point we cannot elucidate whether it is these missense mutations or other SNPs outside coding regions, which confer the trait variation. The question about the ancestral and derived alleles remains elusive, but we surveyed sequences from other salmonids for information about the amino acid variants and found that both brown trout (Salmo trutta L.), rainbow trout (Oncorhynchus mykiss) and arctic char (Salvelinus alpinus) all have the 3SW variants of the amino acids. In addition, we ran an allelic discrimination assay on five individuals from the Swedish landlocked Atlantic salmon population, Gullspång (landlocked for 10,000 years), all carrying only the 3SW (Thr-Lys) amino acid variant. This indicates that the 3SW version of the Vgll3 is ancestral and that the 1SW (Met-Asp) is derived.
In humans the VGLL3 locus has been linked to age at maturity or puberty by a SNP in close proximity of the gene [35], strengthening our notion that the salmon Vgll3 protein is involved in age at puberty in fish. Regarding the function of this protein in controlling age at maturity, it is known that Vgll3 is involved in the inhibition of adipocyte differentiation in mouse (Mus musculus) [36]. Changes in fat metabolism may be partially causative for changes in age at maturity, since increased adiposity has previously been linked to maturation in salmon [37–40]. In studies in rodent testis, vgll3 transcripts have been associated with differential expression during the early stages of steroidogenesis in the embryonic testis [41], suggesting a role in testis maturation. Further functional studies of this protein and adjacent regulatory regions will confirm if the previous study in humans and our study have actually revealed a universal regulator of age at maturity in vertebrates.
The most significant SNPs were located in the vgll3 locus but two neighboring genes, chmp2B and akap11, also contain several significant SNPs (Fig 2B). One of these, a missense mutation in akap11 translates to a Val in 1SW and a Met in 3SW at aa 214. AKAP11 is involved in compartmentalization of cyclic AMP-dependent protein kinase (PKA). This aa AKAP11 is not located in any of the known functional domains related to PKA [42]. AKAP 11 is highly expressed in elongating spermatocytes and mature sperm in human testis and is believed to contribute to cell cycle control in both germ cells and somatic cells. There are no reports clearly linking this protein to age at maturity but future functional studies may reveal if this is the case. Chmp2B did not contain any missense mutations but upon manual review of this region we detected a misplaced 16,885 bp region in the Chr 25 containing exon 1 and 2 of chmp2B. This region also carried many significant SNPs which were probably associated with the selective sweep. When this region was placed in proximity of the gene (dark grey box in Fig 2B) it became clear that many significant SNPs were near the chmp2b gene. This gene encodes a protein belonging to a protein complex which is involved in protein endocytosis [43] (Fig 2B). In humans CHMP2B is known to be essential for the survival of nerve cells and is linked to both dementia and Amyotrophic lateral sclerosis (ALS) [44–46]. It is well known that the neural system works as a gatekeeper in controlling age of puberty, also in fish [47] but whether Chmp2B is involved in the regulation of puberty remains to be elucidated.
In this study we performed a GWAS by genome re-sequencing with the aim to screen the genome of Atlantic salmon for loci regulating age at maturity in males. By investigating late and early maturing male fish from six rivers in Western Norway we demonstrated that the sea age at maturity trait was strongly associated with sequence variation at one locus on Chr 25. The haplotype associated with late maturity can be used for selective breeding on individuals predisposed for this trait, thereby possibly reducing the incidence of negative phenotypes associated with early maturation of males in salmon aquaculture. However, using only late maturing fish in aquaculture breeding will increase generation times in culture, thereby decreasing the speed at which other traits such as growth can be selected for. This potential conflict of interest could be solved by using photoperiod manipulation to shorten generation time of fish with a genetic predisposition for high age at maturity. In this study we did not investigate how female maturation is affected by vgll3 genotypes. Future studies will reveal if female puberty is also influenced by this locus. This study also shows that certain haplotypes significantly contribute to the sea age at puberty, and may therefore be implemented as markers in the management of wild salmon populations in the face of changing environmental conditions such as increased sea temperatures. Significantly, this study and a previous study in humans [35], suggests a conserved role of the Vgll3 protein in timing of puberty in vertebrates.
Materials and Methods
Samples and sampling
The samples of wild salmon upon which this study is based were collected by Rådgivende Biologer AS, Bergen, Norway (http://www.radgivende-biologer.no). Scales were taken from dead salmon fish that had been captured by anglers during the fishing season. In this manner, samples of wild salmon were acquired from six rivers in Western Norway; Eidselva, Gloppenelven, Flekkeelven, Årdalselva, Suldalslågen and Vormo (Fig 1). In order to minimize the potential influence of environmental variation on the sea age at maturity we used fish from the same smolt year class sampled as 1SW fish (returning to river 2009) and 3SW fish (returning to river 2011). Each river was represented by 20 1SW and 20 3SW males. For the genotyping assay we also included two other year classes from Eidselva and Suldalslågen. From Eidselva we retrieved scales from 20 1SW males from year 2000 and 8 3SW males from 2002. From Suldalslågen we obtained scales from 14 1SW and 14 3SW from years 2005 and 2007, respectively.
In addition to samples of wild salmon, we investigated age at maturity in four full sibling families of domesticated salmon from the Norwegian Mowi strain maturing at 1SW, 2SW or older. These fish were obtained from an ongoing study at the Matre Aquaculture Research station where they were reared in a common garden design in sea cages without the use of continuous light, i.e. under ambient light only. Before transfer to sea cages, fish were sedated (0.07 gL-1, Finquel, ScanAqua), adipose fin clipped and PIT (passive integrated transponder) tagged. Fin clips, preserved on 95% ethanol, from a total of 97 fish maturing at different sea ages were included in this study. The four families consisted of 36, 24, 13 and 24 sibling fish per family. We used the parental information to avoid within family Mendelian errors and to phase decendent’s haplotypes.
DNA extraction and PCR-based sdY test
Total DNA from selected individuals was purified from 2 to 3 scales using Qiagen DNeasy Blood & Tissue Kit (Qiagen, Hilden, Germany) according to the manufacturer´s recommendations. Sex of all samples used herein was validated by a PCR-based methodology aimed to detect the presence of the sdY gene [48,49]. Individuals showing amplicons of exon 2 and 4 were designated as males. As a positive PCR control and for species determination we used the presence of the 5S rRNA gene [50]. PCR amplifications were performed using reaction mixtures containing approximately 50 ng of extracted Atlantic salmon DNA, 10 nM Tris–HCl pH 8.8, 1.5 mM MgCl2, 50 mM KCl, 0.1% Triton X-100, 0.35 μM of each primers, 0.5 Units of DNA Taq Polymerase (Promega, Madison, WI, USA) and 250 μM of each dNTP in a final volume of 20 μL. PCR products were visualized in 3% agarose gels.
Library preparation, sequencing and mapping
Following fluorometric quantification, equal amounts of DNA from ten males were pooled to generate paired-end libraries using the Genomic DNA Sample Preparation Kit (Illumina, CA, USA) according to manufacturer’s instructions. Libraries were sequenced on the Illumina HiSeq2000 platform (Illumina, CA, USA) at the Norwegian Sequencing center (https://www.sequencing.uio.no/, Oslo, Norway). In each sequencing lane we used pools of 10 fish from each sea age and river which made a total of 24 lanes sequenced in the whole experiment (6 rivers, 2 replicates per sea age). Raw sequence data has been deposited at SRA with BioProject Accession number PRJNA293012. Library quality control was conducted to ensure that all the samples fulfilled the quality standards (FastQC—http://www.bioinformatics.babraham.ac.uk/projects/fastqc/). Adapter and quality trimming of FastQ format reads were carried out using Cutadapt [51]. All 24 libraries containing on average 361821757 (± 4956053) paired end reads were approved for further analysis and aligned to the most recent salmon genome release (Acc. No. AGKD0000000.4) using Bowtie2 (v.2.1.0) [52]. Entire read alignment with no soft clipping was required by setting Bowtie 2 to the end-to-end mode. Seed length during alignment was set to 18, allowing only 1 mismatch. Interval function between seed substring during multiseed alignment was defined by the following variables: S,1,1.5 controlling the sensitivity of the mapping (interval function f(x) = 1+1.5*sqrt(x), x being the length of the read). Maximum number of ambiguous characters was set by the following function parameters: L,0,0.1 (f(x) = 0+0.1*x, x being read length). Minimum alignment score was governed by the function parameters L, -0.6,-0.4. (f(x) = -0.6 + -0.4*x, x being read length). Only unambiguously mapped reads (mapping quality score greater than 20) were retained for downstream analysis
SNP calling, annotation and statistical analysis
To improve the sensitivity to detect rare alleles, biological replicates in the dataset were bioinformatically fused using SAMtools merge, producing a single BAM file for each river and maturation stage [53]. SNPs were called using the Mpileup command in SAMtools. The resulting file was then recoded for use in the PoPoolation2 pipeline (v.1.2.2) [54]. A minimum base quality threshold of 20 was established in order to remove ambiguously mapped reads and low quality bases. The Cochran-Mantel-Haenszel test for repeated tests of independence for every SNP was performed using the PoPoolation 2 package (cmh-test.pl and R based custom script) in order to detect significant differences (0.1% FDR) in allele frequencies between 1SW and 3SW pools [55]. For each merged sample of 20 fish, the parameters min-count was set to a value of 10 whereas the min-coverage and max-coverage were set to 7 and 42, respectively (5–95% percentile). To annotate the salmon genome (AKGD00000000.4), Augustus gene prediction software was trained using PASA gene candidates by mapping salmon ESTs from NCBI to the salmon genome assembly with PASA [56,57]. The Augustus de novo gene prediction contained coding sequences without UTRs. The genes were validated by RNASeq from both Atlantic salmon [58] and rainbow trout [59] and annotated with Swissprot. Significant SNPs were functionally annotated to predict variant effect (custom R and Python scripts). Bioinformatical analysis identified a 16,885 bp region in Chr 25 (position 28907421–28924305) which contained the first two exons of the gene chmp2b in addition to several significant SNPs. In a previous version of the genome assembly (AKGD00000000.3) this region existed as a single contig, and has presumably been inserted into the wrong chromosomal region in the most recent genome assembly. We corrected this by reverse-complementing the region and inserting it in the gap between the third exon of chmp2b and vgll3 in Chr 25 (position 28626249–28643134) placing the exons of chmp2b in coherent order. Genotype/phenotype association analysis was performed using Plink v1.8 [24]. Selected SNPs were tested for association using a standard linear regression of phenotype on allele dosage (Wald test 1%FDR [56]). Whenever possible, asymptotic haplotype-specific association tests were performed in order to establish the percentage of the phenotypic variation that could be explained by the detected haplotypes.
Sequenom assays
The salmon genome has undergone a recent whole genome duplication and is partly tetraploid giving rise to many highly similar duplicated regions [20]. We have checked that all genotyping assays used in this study only targeted unique sequences in the genome. Eleven of the most significant SNPs (S1 Table) identified in the putative selective sweep (Fig 2B) were used to design a Sequenom assay. This was performed to be able to verify the GWAS findings for the 240 individuals used for pooled sequencing. In addition, we validated the associated SNPs in year classes 1999 and 2004 belonging to Eidselva and Suldalslågen rivers respectively as well as in 97 fish belonging to the 4 families from the domesticated Mowi strain. Genotyping was conducted on a Sequenom MassARRAY analyser (San Diego, CA, USA). A complete list of Primers and extension primers used are found in the S2 Table.
Data reporting
Sequenced pool material submitted to SRA (Bioproject number PRJNA293012). SNP data obtained has been deposited at http://marineseq.imr.no/salmatsnp/.
Ethics statement
Scale samples from wild salmon were collected by local anglers during the fishing season, thus no permits/licenses regarding the collection of these samples were required by the research team. Samples from domesticated fish were retrieved from an ongoing study at Matre Aquaculture Research station (IMR), where the experimental protocol (permit number 4268) had been approved by the Norwegian Animal Research Authority (NARA). Welfare and use of these experimental animals was performed in strict accordance with the Norwegian Animal Welfare Act of 19th of June 2009, in force from 1st of January 2010. All personnel involved in the experiment had undergone training approved by the Norwegian Food Safety Authority. This training is mandatory for all personnel running experiments involving animals included in the Animal Welfare Act.
Supporting Information
(A) Average read depth in all pooled samples (x-axis). The y-axis is showing average percent of the genome covered, with error bars. (B) Average number of uniquely mapped sequences (y-axis) on each chromosome (x-axis), with error bars. “Scaffolds” refers to unplaced contigs in the current genome assembly.
(TIF)
Frequencies of the late maturation allele in year class 2008 are shown for 1SW (green bars) and 3SW (blue bars) fish. The position of each SNP in Chr 25 is shown in the leftmost part. Above each bar the number genotyped fish is indicated. The y-axis shows the allele frequency between 0 and 1. Abbreviations; Ard—Årdalselven, Eid—Eidselven, Fle—Flekkeelven, Glo—Gloppenelven, Sul—Suldalslågen and Vor—Vormo.
(TIF)
Frequencies of the late maturation allele in Eidselva year class 1999 and Suldalslågen yearclass 2004 are shown for 1SW (green bars) and 3SW (blue bars) fish. The position of each SNP in Chr 25 is shown in the leftmost part. Above each bar the number genotyped fish is indicated. The y-axis shows the allele frequency between 0 and 1. Abbreviations; Eid—Eidselven, and Sul—Suldalslågen.
(TIF)
Frequencies of the late maturation allele in farmed salmon (Mowi strain) strains, maturing either after 1SW (green bars), 2SW (yellow bars) or after 3SW or more in sea water (blue bars). The position of each SNP in Chr 25 is shown in the leftmost part. Above each bar the number genotyped fish is indicated. The y-axis shows the allele frequency between 0 and 1.
(TIF)
Table of SNPs significantly associated to age at maturity. The table includes scaffold names, chromosome position, reference/alternative (Ref/Alt) Alleles, Average Coverage (+/-SE) and P-values.
(PDF)
(PDF)
Acknowledgments
We would like to acknowledge the expert advice of Eva Andersson, Rüdiger W Schulz, Jan Bogerd, Tom Hansen, Ketil Malde and Per Gunnar Fjelldal during the project. We are also grateful to Anne-Grethe Sørvik, Stig Mæhle, Sara Olausson, Sven Leininger, Amèlie Juanchich, and Lene Kleppe for providing laboratory support. Special thanks to Ivar Helge Matre, Jan Olav Fosse and Lise Dyrhovden for providing excellent assistance in taking care of the salmon experiment performed at Matre aquaculture station. We would like to thank Rådgivende Biologer Bergen A/S for selling us the wild salmon samples.
Data Availability
All of the relevant data are within the paper and its Supporting Information files, except for the sequencing data which has been deposited at SRA (Bioproject number PRJNA293012). SNP data obtained has been deposited at http://marineseq.imr.no/salmatsnp/.
Funding Statement
This project was financed by the Norwegian research council (NFR) and their HAVBRUK-BIOTEK 2021 program (project number 226221- SALMAT). The farmed salmon strains analyzed here were produced under the NFR financed project INTERACT. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. Taranger GL, Carrillo M, Schulz RW, Fontaine P, Zanuy S, et al. (2010) Control of puberty in farmed fish. Gen Comp Endocrinol 165: 483–515. 10.1016/j.ygcen.2009.05.004 [DOI] [PubMed] [Google Scholar]
- 2. Glover KA, Quintela M, Wennevik V, Besnier F, Sorvik AGE, et al. (2012) Three Decades of Farmed Escapees in the Wild: A Spatio-Temporal Analysis of Atlantic Salmon Population Genetic Structure throughout Norway. Plos One 7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Glover KA, Pertoldi C, Besnier F, Wennevik V, Kent M, et al. (2013) Atlantic salmon populations invaded by farmed escapees: quantifying genetic introgression with a Bayesian approach and SNPs. BMC Genetics 14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Olsen RE, Skilbrei OT (2010) Feeding preference of recaptured Atlantic salmon Salmo salar following simulated escape from fish pens during autumn. Aquaculture Environment Interactions 1: 167–174. [Google Scholar]
- 5. Wild V, Simianer H, Gjoen HM, Gjerde B (1994) Genetic-Parameters and Genotype X Environment Interaction for Early Sexual Maturity in Atlantic Salmon (Salmo-Salar). Aquaculture 128: 51–65. [Google Scholar]
- 6. Gjerde B, Simianer H, Refstie T (1994) Estimates of Genetic and Phenotypic Parameters for Body-Weight, Growth-Rate and Sexual Maturity in Atlantic Salmon. Livestock Production Science 38: 133–143. [Google Scholar]
- 7. Gjerde B, Gjedrem T (1984) Estimates of Phenotypic and Genetic-Parameters for Carcass Traits in Atlantic Salmon and Rainbow-Trout. Aquaculture 36: 97–110. [Google Scholar]
- 8. Gjerde B (1984) Response to Individual Selection for Age at Sexual Maturity in Atlantic Salmon. Aquaculture 38: 229–240. [Google Scholar]
- 9. Gjedrem T (2000) Genetic improvement of cold-water fish species. Aquaculture Research 31: 25–33. [Google Scholar]
- 10. Moghadam HK, Poissant J, Fotherby H, Haidle L, Ferguson MM, et al. (2007) Quantitative trait loci for body weight, condition factor and age at sexual maturation in Arctic charr (Salvelinus alpinus): comparative analysis with rainbow trout (Oncorhynchus mykiss) and Atlantic salmon (Salmo salar). Mol Genet Genomics 277: 647–661. [DOI] [PubMed] [Google Scholar]
- 11. Gutierrez AP, Yanez JM, Fukui S, Swift B, Davidson WS (2015) Genome-Wide Association Study (GWAS) for Growth Rate and Age at Sexual Maturation in Atlantic Salmon (Salmo salar). PLoS One 10: e0119730 10.1371/journal.pone.0119730 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Gutierrez AP, Lubieniecki KP, Fukui S, Withler RE, Swift B, et al. (2014) Detection of quantitative trait loci (QTL) related to grilsing and late sexual maturation in Atlantic salmon (Salmo salar). Mar Biotechnol (NY) 16: 103–110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Johnston SE, Orell P, Pritchard VL, Kent MP, Lien S, et al. (2014) Genome-wide SNP analysis reveals a genetic basis for sea-age variation in a wild population of Atlantic salmon (Salmo salar). Mol Ecol 23: 3452–3468. 10.1111/mec.12832 [DOI] [PubMed] [Google Scholar]
- 14. Lien S, Gidskehaug L, Moen T, Hayes BJ, Berg PR, et al. (2011) A dense SNP-based linkage map for Atlantic salmon (Salmo salar) reveals extended chromosome homeologies and striking differences in sex-specific recombination patterns. BMC Genomics 12: 615 10.1186/1471-2164-12-615 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Davidson WS, Koop BF, Jones SJ, Iturra P, Vidal R, et al. (2010) Sequencing the genome of the Atlantic salmon (Salmo salar). Genome Biol 11: 403 10.1186/gb-2010-11-9-403 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Rubin CJ, Zody MC, Eriksson J, Meadows JR, Sherwood E, et al. (2010) Whole-genome resequencing reveals loci under selection during chicken domestication. Nature 464: 587–591. 10.1038/nature08832 [DOI] [PubMed] [Google Scholar]
- 17. Ferretti L, Ramos-Onsins SE, Perez-Enciso M (2013) Population genomics from pool sequencing. Mol Ecol 22: 5561–5576. 10.1111/mec.12522 [DOI] [PubMed] [Google Scholar]
- 18. Rubin CJ, Megens HJ, Barrio AM, Maqbool K, Sayyab S, et al. (2012) Strong signatures of selection in the domestic pig genome. Proceedings of the National Academy of Sciences of the United States of America 109: 19529–19536. 10.1073/pnas.1217149109 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Rubin CJ, Zody MC, Eriksson J, Meadows JRS, Sherwood E, et al. (2010) Whole-genome resequencing reveals loci under selection during chicken domestication. Nature 464: 587–U145. 10.1038/nature08832 [DOI] [PubMed] [Google Scholar]
- 20. Gidskehaug L, Kent M, Hayes BJ, Lien S (2011) Genotype calling and mapping of multisite variants using an Atlantic salmon iSelect SNP array. Bioinformatics 27: 303–310. 10.1093/bioinformatics/btq673 [DOI] [PubMed] [Google Scholar]
- 21. Pedersen S, Berg PR, Culling M, Danzmann RG, Glebe B, et al. (2013) Quantitative trait loci for precocious parr maturation, early smoltification, and adult maturation in double-backcrossed trans-Atlantic salmon (Salmo salar). Aquaculture 410: 164–171. [Google Scholar]
- 22. Johnston SE, Orell P, Pritchard VL, Kent MP, Lien S, et al. (2014) Genome-wide SNP analysis reveals a genetic basis for sea-age variation in a wild population of Atlantic salmon (Salmo salar). Mol Ecol. [DOI] [PubMed] [Google Scholar]
- 23. Gurney WS, Bacon PJ, Speirs DC, McGinnity P, Verspoor E (2012) Sea-age variation in maiden Atlantic salmon spawners: phenotypic plasticity or genetic polymorphism? Bull Math Biol 74: 615–640. 10.1007/s11538-011-9679-8 [DOI] [PubMed] [Google Scholar]
- 24. Purcell S, Neale B, Todd-Brown K, Thomas L, Ferreira MA, et al. (2007) PLINK: a tool set for whole-genome association and population-based linkage analyses. Am J Hum Genet 81: 559–575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Miller AS, Sheehan TF, Renkawitz MD, Meister AL, Miller TJ (2012) Revisiting the marine migration of US Atlantic salmon using historical Carlin tag data. Ices Journal of Marine Science 69: 1609–1615. [Google Scholar]
- 26. Bacon PJ, Palmer SCF, MacLean JC, Smith GW, Whyte BDM, et al. (2009) Empirical analyses of the length, weight, and condition of adult Atlantic salmon on return to the Scottish coast between 1963 and 2006. Ices Journal of Marine Science 66: 844–859. [Google Scholar]
- 27. Hansen LP, Quinn TR (1998) The marine phase of the Atlantic salmon (Salmo salar) life cycle, with comparisons to Pacific salmon. Canadian Journal of Fisheries and Aquatic Sciences 55: 104–118. [Google Scholar]
- 28. Taranger GL, Haux C, Hansen T, Stefansson SO, Bjornsson BT, et al. (1999) Mechanisms underlying photoperiodic effects on age at sexual maturity in Atlantic salmon, Salmo salar . Aquaculture 177: 47–60. [Google Scholar]
- 29. Taranger GL, Vikingstad E, Klenke U, Mayer I, Stefansson SO, et al. (2003) Effects of photoperiod, temperature and GnRHa treatment on the reproductive physiology of Atlantic salmon (Salmo salar L.) broodstock. Fish Physiology and Biochemistry 28: 403–406. [DOI] [PubMed] [Google Scholar]
- 30. Glover KA, Ottera H, Olsen RE, Slinde E, Taranger GL, et al. (2009) A comparison of farmed, wild and hybrid Atlantic salmon (Salmo salar L.) reared under farming conditions. Aquaculture 286: 203–210. [Google Scholar]
- 31. Solberg MF, Zhang ZW, Nilsen F, Glover KA (2013) Growth reaction norms of domesticated, wild and hybrid Atlantic salmon families in response to differing social and physical environments. BMC Evolutionary Biology 13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Solberg MF, Skaala O, Nilsen F, Glover KA (2013) Does Domestication Cause Changes in Growth Reaction Norms? A Study of Farmed, Wild and Hybrid Atlantic Salmon Families Exposed to Environmental Stress. Plos One 8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. de Juan D, Pazos F, Valencia A (2013) Emerging methods in protein co-evolution. Nat Rev Genet 14: 249–261. 10.1038/nrg3414 [DOI] [PubMed] [Google Scholar]
- 34. Helias-Rodzewicz Z, Perot G, Chibon F, Ferreira C, Lagarde P, et al. (2010) YAP1 and VGLL3, encoding two cofactors of TEAD transcription factors, are amplified and overexpressed in a subset of soft tissue sarcomas. Genes Chromosomes Cancer 49: 1161–1171. 10.1002/gcc.20825 [DOI] [PubMed] [Google Scholar]
- 35. Cousminer DL, Berry DJ, Timpson NJ, Ang W, Thiering E, et al. (2013) Genome-wide association and longitudinal analyses reveal genetic loci linking pubertal height growth, pubertal timing and childhood adiposity. Hum Mol Genet 22: 2735–2747. 10.1093/hmg/ddt104 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Halperin DS, Pan C, Lusis AJ, Tontonoz P (2013) Vestigial-like 3 is an inhibitor of adipocyte differentiation. J Lipid Res 54: 473–481. 10.1194/jlr.M032755 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Trombley S, Mustafa A, Schmitz M (2014) Regulation of the seasonal leptin and leptin receptor expression profile during early sexual maturation and feed restriction in male Atlantic salmon, Salmo salar L., parr. General and Comparative Endocrinology 204: 60–70. 10.1016/j.ygcen.2014.04.033 [DOI] [PubMed] [Google Scholar]
- 38. Larsen DA, Beckman BR, Strom CR, Parkins PJ, Cooper KA, et al. (2006) Growth modulation alters the incidence of early male maturation and physiological development of hatchery-reared spring Chinook salmon: A comparison with wild fish. Transactions of the American Fisheries Society 135: 1017–1032. [Google Scholar]
- 39. Silverstein JT, Shearer KD, Dickhoff WW, Plisetskaya EM (1999) Regulation of nutrient intake and energy balance in salmon. Aquaculture 177: 161–169. [Google Scholar]
- 40. Silverstein JT, Shearer KD, Dickhoff WW, Plisetskaya EM (1998) Effects of growth and fatness on sexual development of chinook salmon (Oncorhynchus tshawytscha) parr. Canadian Journal of Fisheries and Aquatic Sciences 55: 2376–2382. [Google Scholar]
- 41. McDowell EN, Kisielewski AE, Pike JW, Franco HL, Yao HHC, et al. (2012) A Transcriptome-Wide Screen for mRNAs Enriched in Fetal Leydig Cells: CRHR1 Agonism Stimulates Rat and Mouse Fetal Testis Steroidogenesis. Plos One 7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Reinton N, Collas P, Haugen TB, Skalhegg BS, Hansson V, et al. (2000) Localization of a novel human A-kinase-anchoring protein, hAKAP220, during spermatogenesis. Dev Biol 223: 194–204. [DOI] [PubMed] [Google Scholar]
- 43. Bodon G, Chassefeyre R, Pernet-Gallay K, Martinelli N, Effantin G, et al. (2011) Charged multivesicular body protein 2B (CHMP2B) of the endosomal sorting complex required for transport-III (ESCRT-III) polymerizes into helical structures deforming the plasma membrane. J Biol Chem 286: 40276–40286. 10.1074/jbc.M111.283671 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Ferrari R, Kapogiannis D, Huey ED, Grafman J, Hardy J, et al. (2010) Novel missense mutation in charged multivesicular body protein 2B in a patient with frontotemporal dementia. Alzheimer Dis Assoc Disord 24: 397–401. 10.1097/WAD.0b013e3181df20c7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Ghazi-Noori S, Froud KE, Mizielinska S, Powell C, Smidak M, et al. (2012) Progressive neuronal inclusion formation and axonal degeneration in CHMP2B mutant transgenic mice. Brain 135: 819–832. 10.1093/brain/aws006 [DOI] [PubMed] [Google Scholar]
- 46. Parkinson N, Ince PG, Smith MO, Highley R, Skibinski G, et al. (2006) ALS phenotypes with mutations in CHMP2B (charged multivesicular body protein 2B). Neurology 67: 1074–1077. [DOI] [PubMed] [Google Scholar]
- 47. Zohar Y, Munoz-Cueto JA, Elizur A, Kah O (2010) Neuroendocrinology of reproduction in teleost fish. Gen Comp Endocrinol 165: 438–455. 10.1016/j.ygcen.2009.04.017 [DOI] [PubMed] [Google Scholar]
- 48. Eisbrenner WS, Botwright N, Cook M, Davidson EA, Dominik S, et al. (2014) Evidence for multiple sex-determining loci in Tasmanian Atlantic salmon (Salmo salar). Heredity 113: 86–92. 10.1038/hdy.2013.55 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Yano A, Guyomard R, Nicol B, Jouanno E, Quillet E, et al. (2012) An immune-related gene evolved into the master sex-determining gene in rainbow trout, Oncorhynchus mykiss . Curr Biol 22: 1423–1428. 10.1016/j.cub.2012.05.045 [DOI] [PubMed] [Google Scholar]
- 50. Pendas AM, Moran P, Martinez JL, Garcia-Vazquez E (1995) Applications of 5s-Rdna in Atlantic Salmon, Brown Trout, and in Atlantic Salmon X Brown Trout Hybrid Identification. Molecular Ecology 4: 275–276. [DOI] [PubMed] [Google Scholar]
- 51. Martin M (2011) Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet journal 17: pp. 10–12. [Google Scholar]
- 52. Langmead B, Salzberg SL (2012) Fast gapped-read alignment with Bowtie 2. Nat Methods 9: 357–359. 10.1038/nmeth.1923 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Li H, Handsaker B, Wysoker A, Fennell T, Ruan J, et al. (2009) The Sequence Alignment/Map format and SAMtools. Bioinformatics 25: 2078–2079. 10.1093/bioinformatics/btp352 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Kofler R, Pandey RV, Schlotterer C (2011) PoPoolation2: identifying differentiation between populations using sequencing of pooled DNA samples (Pool-Seq). Bioinformatics 27: 3435–3436. 10.1093/bioinformatics/btr589 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Benjamini Y, Hochberg Y (1995) Controlling the False Discovery Rate—a Practical and Powerful Approach to Multiple Testing. Journal of the Royal Statistical Society Series B-Methodological 57: 289–300. [Google Scholar]
- 56. Stanke M, Morgenstern B (2005) AUGUSTUS: a web server for gene prediction in eukaryotes that allows user-defined constraints. Nucleic Acids Res 33: W465–467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Haas BJ, Delcher AL, Mount SM, Wortman JR, Smith RK Jr., et al. (2003) Improving the Arabidopsis genome annotation using maximal transcript alignment assemblies. Nucleic Acids Res 31: 5654–5666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Wang S, Furmanek T, Kryvi H, Krossoy C, Totland GK, et al. (2014) Transcriptome sequencing of Atlantic salmon (Salmo salar L.) notochord prior to development of the vertebrae provides clues to regulation of positional fate, chordoblast lineage and mineralisation. BMC Genomics 15: 141 10.1186/1471-2164-15-141 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Berthelot C, Brunet F, Chalopin D, Juanchich A, Bernard M, et al. (2014) The rainbow trout genome provides novel insights into evolution after whole-genome duplication in vertebrates. Nat Commun 5: 3657 10.1038/ncomms4657 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(A) Average read depth in all pooled samples (x-axis). The y-axis is showing average percent of the genome covered, with error bars. (B) Average number of uniquely mapped sequences (y-axis) on each chromosome (x-axis), with error bars. “Scaffolds” refers to unplaced contigs in the current genome assembly.
(TIF)
Frequencies of the late maturation allele in year class 2008 are shown for 1SW (green bars) and 3SW (blue bars) fish. The position of each SNP in Chr 25 is shown in the leftmost part. Above each bar the number genotyped fish is indicated. The y-axis shows the allele frequency between 0 and 1. Abbreviations; Ard—Årdalselven, Eid—Eidselven, Fle—Flekkeelven, Glo—Gloppenelven, Sul—Suldalslågen and Vor—Vormo.
(TIF)
Frequencies of the late maturation allele in Eidselva year class 1999 and Suldalslågen yearclass 2004 are shown for 1SW (green bars) and 3SW (blue bars) fish. The position of each SNP in Chr 25 is shown in the leftmost part. Above each bar the number genotyped fish is indicated. The y-axis shows the allele frequency between 0 and 1. Abbreviations; Eid—Eidselven, and Sul—Suldalslågen.
(TIF)
Frequencies of the late maturation allele in farmed salmon (Mowi strain) strains, maturing either after 1SW (green bars), 2SW (yellow bars) or after 3SW or more in sea water (blue bars). The position of each SNP in Chr 25 is shown in the leftmost part. Above each bar the number genotyped fish is indicated. The y-axis shows the allele frequency between 0 and 1.
(TIF)
Table of SNPs significantly associated to age at maturity. The table includes scaffold names, chromosome position, reference/alternative (Ref/Alt) Alleles, Average Coverage (+/-SE) and P-values.
(PDF)
(PDF)
Data Availability Statement
All of the relevant data are within the paper and its Supporting Information files, except for the sequencing data which has been deposited at SRA (Bioproject number PRJNA293012). SNP data obtained has been deposited at http://marineseq.imr.no/salmatsnp/.