Abstract
Background
Clinical features of thyroid dysfunction vary with age, and an oligosymptomatic presentation of hyperthyroidism is frequently observed in the elderly. This suggests age modulation of thyroid hormone (TH) action, which may occur, for example, by alterations in TH production, metabolism and/or TH action in target organs. Objectives: In this paper, we address possible changes in TH transporter expression in liver tissues as a mechanism of age-dependent variation in TH action.
Methods
Chronic hyperthyroidism was induced in 4- and 20-month-old C57BL6/NTac male mice (n = 8-10) by intraperitoneal injections of 1 µg/g body weight L-thyroxine (T4) every 48 h over 7 weeks. Control animals were injected with PBS. Total RNA was isolated from liver samples for analysis of the TH transporter and TH-responsive gene expression. TH concentrations were determined in mice sera.
Results
Baseline serum free T4 (fT4) concentrations were significantly higher in euthyroid young compared to old mice. T4 treatment increased total T4, fT4 and free triiodothyronine to comparable concentrations in young and old mice. In the euthyroid state, TH transporter expression was significantly higher in old than in young mice, except for Mct8 and Oatp1a1 expression levels. Hyperthyroidism resulted in upregulation of Mct10, Lat1 and Lat2 in liver tissue, while Oatp1a1, Oatp1b2 and Oatp1a4 expression was downregulated. This effect was preserved in old animals.
Conclusion
Here, we show age-dependent differences in TH transporter mRNA expression in the euthyroid and hyperthyroid state of mice focusing on the liver as a classical TH target organ.
Key Words: Thyroid, Hyperthyroidism, Liver, Aging
Introduction
Hyperthyroidism is a pathological state characterized by excessive thyroid hormone (TH) action in the body. Classical clinical features include anxiety, nervousness, tremor, weight loss and heat intolerance, which are often obscured in elderly patients and hence diagnosis may be missed [1]. Hyperthyroidism is associated with significant morbidity and mortality, mainly from cardiovascular and cerebrovascular disease, which are conditions that are particularly relevant to an aging population [2,3,4].
It is widely accepted that circulating TH levels may not solely and comprehensively reflect TH organ status. In fact, TH action is complex and, in addition to different TH derivatives, involves distinct transmembrane transporters and nongenomic versus classical nuclear modes of TH action, all of which may be altered with age. However, their variations upon aging have not been thoroughly analyzed, neither in humans nor in mice.
Animal models serve as useful tools to dissect mechanisms of TH action. In this study, we aimed to investigate whether age may result in alterations of proposed TH transporters in liver as a classical TH target organ. To this aim, young (4 months) versus aged (20 months) C57BL/6NTac male mice were studied under conditions of euthyroidism and chronic hyperthyroidism. In addition to serum concentrations, total thyroxine (TT4), free thyroxine (fT4) and free triiodothyronine (fT3), TH transporter mRNA levels and expression of TH-responsive genes were determined in liver tissues of euthyroid and hyperthyroid young and old mice.
Materials and Methods
Animals
Male C57BL/6NTac (Taconic Europe A/S, Denmark, and Taconic Biosciences Inc., USA) mice (n = 32) aged 4 months (young) and 20 months (old) were housed in temperature-(23 ± 1°C) and light-controlled (inverse 12:12-hour light-dark cycle) conditions. Food and water were provided ad libitum. All animal experiments were performed in accordance with the German Regulations for Laboratory Animal Science (GVSOLAS) and the European Health Law of the Federation of Laboratory Animal Science Associations (FELASA). The protocols for animal studies were approved by the Landesamt für Natur, Umwelt und Verbraucherschutz Nordrhein-Westfalen (LANUV-NRW), Germany.
Treatment
For induction of a chronic hyperthyroid state, animals received freshly prepared intraperitoneal (i.p.) injections of 1 µg/g body weight (BW) T4 [Sigma-Aldrich, USA; stock-solution: 2 mg/ml in 0.01 M NaOH, 0.1% BSA (albumin from bovine serum, Sigma-Aldrich); injection solution: stock solution diluted 1:10 with PBS] every other day. Control groups received 150 µl PBS i.p. every other day. The treatment period was 7 weeks.
Blood Sample Collection and TH Measurements
Blood samples were collected from the retrobulbar venous plexus with a heparinized micropipette at the start of experiment and after 5 weeks of T4 treatment (48 h after the last T4 injection) from each animal. Blood samples were stored on ice for 30 min for coagulation and serum was obtained by centrifugation at 4°C for 10 min at 10,000 g. Serum aliquots were stored at −80°C. TT4, fT3 and fT4 concentrations in serum of mice were measured using commercial ELISA kits according to the manufacturer's instructions (DRG Instruments GmbH, Germany). As standards, serum samples with known TH concentrations were used.
Collection of Mice Livers
For liver collection, mice were perfused with heparinized saline through a needle placed in the left ventricle. Livers were isolated quickly, frozen in liquid nitrogen and stored at −80°C until further processing.
Isolation of RNA
For RNA extraction, tissues were homogenized in 600 µl of RLT buffer (Qiagen, Germany) using a tissue homogenizer ULTRA-TURRAX® T25 at 4,000 rpm (Janke & Kunkel IKA, Germany). The tissue lysates were further treated with proteinase K (Qiagen) based on the manufacturers' protocol. Total RNA from liver tissue lysates was purified by using the RNeasy Mini Kit and further by on-column DNase digestion using the RNase-Free DNase Set (Qiagen). RNA quantity and quality were determined using NanoDrop 1000 (Thermo Scientific), and the integrity of RNA was further assessed via gel electrophoresis followed by ethidium bromide staining to visualize 28S and 18S rRNA.
Synthesis of cDNA and Real-Time PCR
2 µg of RNA was reverse transcribed to cDNA using random hexamers and SuperScript III First-Strand Synthesis System for real-time PCR according to the instruction manuals (Life Technologies, Germany). Exon-spanning primers for amplification of TH responsive genes (online suppl. table 1; for suppl. material, see www.karger.com/doi/10.1159/000381020) were designed using PrimerBlast (NCBI) and synthesized by Eurofins (Eurofins MWG Synthesis, Germany). Quantitative real-time PCR was performed using LightCycler® DNA Master SYBR Green I and the LightCycler®480 System (Roche, Germany). The PCR program consisted of an initial denaturation step (5 min at 95°C) and 40 amplification cycles with 15 s at 95°C, 10 s at 60°C and 20 s at 72°C. Melting curve analysis was performed after each PCR, and PCR products were verified by agarose gel electrophoresis.
For normalization of gene expression, the reference genes 18S, Ppia (peptidylprolyl isomerase A, cyclophilin A) and Rpl13a (ribosomal protein L13a) were used. The stability of the housekeeping genes was determined by calculation of the coefficient of variation on the normalized relative quantities and by calculation of the geNorm M value [5]. The geometric average of the ‘best’ three housekeeping genes (best keeper index) was calculated by repeated pair-wise correlation analysis [5]. Fold changes were calculated by the Relative Expression Software Tool (REST©, efficiency-corrected ΔΔCt method) [6,7].
Statistical Analysis
All data are shown as means ± SD or standard error of the mean (SEM), as indicated. Statistical analysis was performed using the unpaired Student's t test of GraphPad Prism 5 Software. For statistical analysis of real-time PCR results (in figures presented as logarithmic fold-change data) antilogarithmic data of T4-treated mice were compared to antilogarithmic data of PBS-treated controls (variations within the control groups are not shown). Values of * p < 0.05, ** p < 0.01, *** p < 0.001 and # p < 0.0001 were considered statistically significant.
Results
Effect of T4 Administration and Aging on TT4, fT4 and fT3 Serum Concentrations in Mice
To examine TH serum changes with T4 administration over the experimental time period, retro-orbital blood samples were collected at the beginning of and during T4 treatment.
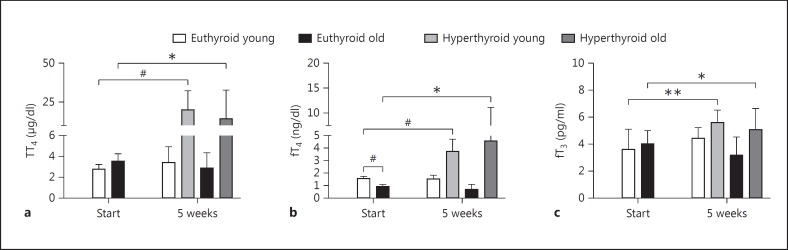
Under T4 treatment, young and old male mice displayed an up to 4- to 6-fold increase in TT4 (p < 0.0001 in young and p < 0.05 in old male mice) and a 2- to 4-fold increase in fT4 (p < 0.0001 in young and p < 0.05 in old male mice) serum concentrations, respectively. In addition, small but significant increases of serum fT3 concentrations (p < 0.01 in young and p < 0.05 in old male mice) were measured under T4 treatment (fig. 1).
Fig. 1.
Serum levels of TH in euthyroid and T4-treated young and old male mice. TT4 (a), fT4 (b) and fT3 (c) were determined in sera by ELISA at the start and after 5 weeks of T4 treatment (1 µg/g BW T4 i.p. every 48 h). A significant increase of TT4, fT4 and fT3 serum parameters was found in T4-treated young (4 months) and old (20 months) mice as compared to the euthyroid start group. The euthyroid control groups of young and old mice showed stable TH serum parameters throughout the experiments. Euthyroid old mice have significantly lower fT4 values than euthyroid young mice. Data are presented as means ± SD; n = 16 animals for start, n = 8 animals (PBS-treated control) and n = 8 animals (hyperthyroid) for end groups; t test, * p < 0.05, ** p < 0.01, # p < 0.0001.
In the control animals, age differences in TH serum concentrations were observed for fT4 values, which were 2-fold higher in young compared to old male mice (p < 0.0001; fig. 1b). In contrast, TT4 as well as fT3 concentrations remained unchanged with aging (fig. 1a, c).
Effect of Aging on TH Transporter Expression in Liver
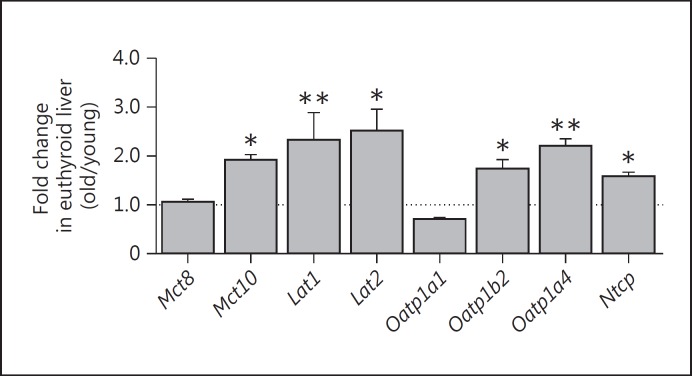
To examine the impact of aging on TH transporter expression, fold changes of TH transporter expression levels in old liver were compared to young liver tissues for euthyroid (control) animals. We found an increased expression of most TH transporters with aging in livers of euthyroid mice. Thus, old control animals showed a significantly higher expression of Mct10, Lat2, Oatp1b2 and Ntcp (p < 0.05; fig. 2) as well as of Lat1 and Oatp1a4 (p < 0.01; fig. 2) in liver tissues compared to young mice. No changes were detected for Mct8 mRNA levels, and Oatp1a1 levels were marginally decreased with aging (fig. 2).
Fig. 2.
Effect of aging on TH transporter mRNA expression in liver tissues of old compared to young euthyroid male mice. Gene expression levels were determined by quantitative real-time PCR in livers of old euthyroid mice (21.8 months) and were normalized to respective expression levels in livers of euthyroid young mice (5.8 months). Mct10, Lat1, Lat2, Oatp1b2, Oatp1a4 and Ntcp mRNA expression levels are significantly upregulated in liver tissues of old mice compared to young animals. 18S, Ppia and Rpl13a were used as reference genes. Data are presented as fold changes, means ± SEM; n = 7; efficiency-corrected ΔΔCt method; t test, * p < 0.05, ** p < 0.01.
Effect of Chronic Hyperthyroidism on TH Transporter Expression in Liver of Young and Old Mice
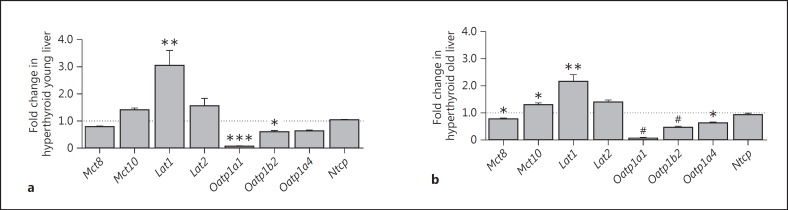
In the hyperthyroid state, similar changes in the pattern of TH transporter mRNA expression in liver were observed for young and old mice (fig. 3). T4 treatment led to marked downregulation of Oatp1a1 mRNA levels (0.06-fold; p < 0.001 in young and p < 0.0001 in old mice) and significant upregulation of Lat1 expression (3-fold in young, 2.2-fold in old mice; p < 0.01). Furthermore, upregulation was found for Mct10 (1.5-fold and significant in old mice; p < 0.05) and downregulation was observed for Mct8 (0.7-fold and significant in old mice; p < 0.05) and Oatp1a4 mRNA (0.6-fold and significant in old mice; p < 0.05) in the hyperthyroid state. Downregulation of Oatp1b2 mRNA expression was observed in both age groups after T4 treatment [0.59-fold in young (p < 0.05) and 0.46-fold in old (p < 0.0001) mice].
Fig. 3.
Impact of hyperthyroidism on TH transporter mRNA expression in liver tissues of young and old male mice. Gene expression levels were measured by quantitative real-time PCR in liver tissues of T4-treated (1 µg/g BW T4 i.p. every 48 h over 7 weeks) young (5.8 months) (a) and old (21.8 months) (b) mice, and were normalized to expression levels of euthyroid (PBS-treated) age-matched controls. a Lat1 mRNA expression was significantly upregulated, whereas Oatp1a1 and Oatp1b2 mRNA expression levels were significantly decreased in liver tissues of T4-treated young mice compared to controls. b Mct10 and Lat1 mRNA expression levels were significantly increased, whereas Mct8, Oatp1a1, Oatp1b2 and Oatp1a4 mRNA expression levels were significantly decreased in liver tissues of T4-treated old mice compared to controls. 18S, Ppia and Rpl13a were used as reference genes. Data are presented as fold changes, means ± SEM; n = 7; efficiency-corrected ΔΔCt method; t test, * p < 0.05, ** p < 0.01, *** p < 0.001, # p < 0.0001.
Effect of Chronic Hyperthyroidism on TH Responsive Gene Expression in Liver of Young and Old Mice
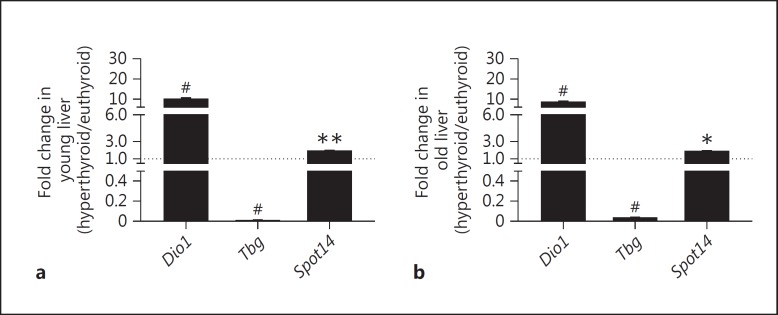
To examine TH status in liver under chronic T4 excess, mRNA transcription levels of genes proposed to respond to TH, namely Spot14 (thyroid hormone-responsive Spot14 homolog, Thrsp), Tbg (TH-binding globulin) and Dio1 (deiodinase 1), were assessed in young and old mice after T4 treatment over 7 weeks [8,9]. Spot14 showed the smallest variations with regard to mRNA levels with upregulation of 1.5-fold under T4 treatment in young (p < 0.01) and old (p < 0.05) mice. In both age groups, Dio1 was highly expressed in the hyperthyroid state (10-fold increase; p < 0.0001) while Tbg expression was downregulated to very low levels in young and old mice (p < 0.0001; fig. 4).
Fig. 4.
Impact of T4 treatment on expression of TH-responsive genes in liver tissues of young and old male mice. Gene expression levels of Dio1, Spot14 and Tbg were measured by quantitative real-time PCR in liver tissues of T4-treated (1 µg/g BW T4 i.p. every 48 h over 7 weeks) young (5.8 months) (a) and old (21.8 months) (b) mice, and were normalized to expression levels of euthyroid (PBS-treated) age-matched controls. Dio1 and Spot14 mRNA expression levels were significantly upregulated, whereas Tbg mRNA expression was significantly and marked downregulated in liver tissues of T4-treated young and old mice compared to controls. 18S, Ppia and Rpl13a were used as reference genes. Data are presented as fold changes, means ± SEM; n = 7; efficiency-corrected ΔΔCt method; t test, * p < 0.05, ** p < 0.01, # p < 0.0001.
Discussion
We examined changes of TH transporter expression during aging in a euthyroid as well as a hyperthyroid state in the TH target organ liver. T4 treatment was chosen as the preferred TH for induction of hyperthyroidism since T4 has been the most widely used iodothyronine in (hyperthyroid) animal experiments, allows for the assessment of T4 to T3 conversion in a living organism and is standard in patients requiring TH substitution.
To assess the effects of chronic T4 exposure and aging on TH serum status, TH serum parameters were determined in euthyroid controls and T4-treated animals. We could confirm significantly lower fT4 concentrations in old male mice (20 months) compared to young male (4 months) mice. A decline in fT4 values has been reported in male mice during development from 2 to 20 weeks [10]. Decreasing fT4 concentrations were also noted in aging men [11]. Under T4 treatment, TT4, fT4 and fT3 concentrations increased in young and old mice to comparable values. Investigation of gene expression in liver tissue confirmed a hyperthyroid tissue state after T4 treatment with the expected changes in the expression of the positively TH-regulated genes Dio1 and Spot14[8] and the negatively regulated gene Tbg[9]. Further analysis of liver tissue with regard to TH transporters showed that hyperthyroidism led to a decrease in expression of the best-studied TH transporter Mct8 [12,13] and an upregulation of Mct10. This was seen in young and old animals, and could represent a compensatory reaction. Mct8 has been reported to have a greater impact on TH efflux than influx in liver cells, as Mct8-/y mice have higher T3 content in liver and show upregulated Dio1 expression and activity [14,15,16]. Similarly Mct10 has been proposed to mediate TH efflux in liver tissue [16]. However, in our study the extent of regulation of both transporters remained in a significant range in livers of old mice only.
Oatp1a1[17], Oatp1b2[18] and Oatp1a4[19] were downregulated in a comparable fashion in hyperthyroid young and old mice, with marked downregulation found for Oatp1a1. As secondary TH transporters, they mainly transport organic compounds, amino acids and hormones [13,20]. Therefore, their lower expression levels might result in lower levels of amino acids available for protein synthesis in hepatocytes [21]. In contrast, Lat1 and Lat2 were upregulated to high levels and in a significant manner. Because Lat1 is known to be highly expressed in hepatocytes under limited amino acid availability, these results are in line with the observed downregulation of Oatps. The Lat1 response is believed to explain how hepatocytes can still grow under limiting amino acid availability, and thus acquire a survival advantage [22,23]. While Lat1 prefers large neutral amino acids, Lat2 has a broader specificity mediating transport of small neutral amino acids as well [24]. Thus an upregulation of Lat2 might reflect the liver's need for small amino acids. NTCp as a multispecific organic anion transporter [13] remained unaltered during hyperthyroidism.
The most surprising finding of our observation is perhaps the upregulation of most investigated TH transporters with aging. This effect was seen in euthyroid and hyperthyroid old animals, suggesting that the liver may react similarly to TH excess in young and old age, at least at the TH transporter level. This is also in agreement with the observed age-independent effect of T4 on TH-responsive genes Spot14, Dio1 and Tbg in the liver of hyperthyroid old and young mice. Age-independent expression levels of Mct8 may underline its importance in maintaining TH transport across the hepatocyte plasma membrane in TH-challenging conditions. Finally, our findings suggest a response to increased needs for amino acids and hormones in aging hepatocytes through the upregulation of, for example, Lat1 and other secondary TH transporters like Lat2. Presently our findings rely on mRNA expression studies and it is unknown whether they are representative for functional protein in the liver. However, with lack of specific antibodies for many TH transporters analyzed in this study, this issue is technically difficult to resolve in a comprehensive manner at this point.
In summary, we report changes in TH transporter mRNA expression in liver as a classical TH target organ in response to TH excess but also aging, whereby the first effect pertained in young and old age.
Disclosure Statement
The authors state that no conflict of interest exists.
Supplementary Material
Supplementary data
Acknowledgements
The authors are grateful to A. Jaeger, S. Rehn and M. Rehders for their dedicated technical support. This work was supported by DFG FU 356/7-1, BR 1308/11-1, MO 1018/2-1.
References
- 1.Cooper DS. Hyperthyroidism. Lancet. 2003;362:459–468. doi: 10.1016/S0140-6736(03)14073-1. [DOI] [PubMed] [Google Scholar]
- 2.Franklyn JA, Sheppard MC, Maisonneuve P. Thyroid function and mortality in patients treated for hyperthyroidism. JAMA. 2005;294:71–80. doi: 10.1001/jama.294.1.71. [DOI] [PubMed] [Google Scholar]
- 3.Klein I, Danzi S. Thyroid disease and the heart. Circulation. 2007;116:1725–1735. doi: 10.1161/CIRCULATIONAHA.106.678326. [DOI] [PubMed] [Google Scholar]
- 4.Biondi B, Palmieri EA, Fazio S, Cosco C, Nocera M, Saccà L, Filetti S, Lombardi G, Perticone F. Endogenous subclinical hyperthyroidism affects quality of life and cardiac morphology and function in young and middle-aged patients. J Clin Endocrinol Metab. 2000;85:4701–4705. doi: 10.1210/jcem.85.12.7085. [DOI] [PubMed] [Google Scholar]
- 5.Pfaffl MW, Tichopad A, Prgomet C, Neuvians TP. Determination of stable housekeeping genes, differentially regulated target genes and sample integrity: BestKeeper – Excel-based tool using pair-wise correlations. Biotechnol Lett. 2004;26:509–515. doi: 10.1023/b:bile.0000019559.84305.47. [DOI] [PubMed] [Google Scholar]
- 6.Pfaffl MW, Horgan GW, Dempfle L. Relative expression software tool (REST) for group-wise comparison and statistical analysis of relative expression results in real-time PCR. Nucleic Acids Res. 2002;30:e36. doi: 10.1093/nar/30.9.e36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Pfaffl MW. A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res. 2001;29:2002–2007. doi: 10.1093/nar/29.9.e45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bianco AC, Anderson G, Forrest D, Galton VA, Gereben B, Kim BW, Kopp PA, Liao XH, Obregon MJ, Peeters RP, Refetoff S, Sharlin DS, Simonides WS, Weiss RE, Williams GR. American Thyroid Association guide to investigating thyroid hormone economy and action in rodent and cell models. Thyroid. 2014;24:88–168. doi: 10.1089/thy.2013.0109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Crowe TC, Cowen NL, Loidl NM, Topliss DJ, Stockigt JR, Barlow JW. Down-regulation of thyroxine-binding globulin messenger ribonucleic acid by 3,5,3′-triiodothyronine in human hepatoblastoma cells. J Clin Endocrinol Metab. 1995;80:2233–2237. doi: 10.1210/jcem.80.7.7608285. [DOI] [PubMed] [Google Scholar]
- 10.Weiss RE, Chassande O, Koo EK, Macchia PE, Cua K, Samarut J, Refetoff S. Thyroid function and effect of aging in combined hetero/homozygous mice deficient in thyroid hormone receptors alpha and beta genes. J Endocrinol. 2002;172:177–185. doi: 10.1677/joe.0.1720177. [DOI] [PubMed] [Google Scholar]
- 11.Fontes R, Coeli C, Aguiar F, Vaisman M. Reference interval of thyroid stimulating hormone and free thyroxine in a reference population over 60 years old and in very old subjects (over 80 years): comparison to young subjects. Thyroid Res. 2013;6:13. doi: 10.1186/1756-6614-6-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Friesema EC, Ganguly S, Abdalla A, Manning Fox JE, Halestrap AP, Visser TJ. Identification of monocarboxylate transporter 8 as a specific thyroid hormone transporter. J Biol Chem. 2003;278:40128–40135. doi: 10.1074/jbc.M300909200. [DOI] [PubMed] [Google Scholar]
- 13.Friesema EC, Jansen J, Milici C, Visser TJ. Thyroid hormone transporters. Vitam Horm. 2005;70:137–167. doi: 10.1016/S0083-6729(05)70005-4. [DOI] [PubMed] [Google Scholar]
- 14.Malik R, Hodgson H. The relationship between the thyroid gland and the liver. QJM. 2002;95:559–569. doi: 10.1093/qjmed/95.9.559. [DOI] [PubMed] [Google Scholar]
- 15.Dumitrescu AM, Liao X-H, Weiss RE, Millen K, Refetoff S. Tissue-specific thyroid hormone deprivation and excess in monocarboxylate transporter (mct) 8-deficient mice. Endocrinology. 2006;147:4036–4043. doi: 10.1210/en.2006-0390. [DOI] [PubMed] [Google Scholar]
- 16.Müller J, Mayerl S, Visser T J, Darras V M, Boelen A, Frappart L, Mariotta L, Verrey F, Heuer H. Tissue-specific alterations in thyroid hormone homeostasis in combined Mct10 and Mct8 deficiency. Endocrinology. 2014;155:315–325. doi: 10.1210/en.2013-1800. [DOI] [PubMed] [Google Scholar]
- 17.Friesema EC, Docter R, Moerings EP, Stieger B, Hagenbuch B, Meier PJ, Krenning EP, Hennemann G, Visser TJ. Identification of thyroid hormone transporters. Biochem Biophys Res Commun. 1999;254:497–501. doi: 10.1006/bbrc.1998.9974. [DOI] [PubMed] [Google Scholar]
- 18.Cattori V, Hagenbuch B, Hagenbuch N, Stieger B, Ha R, Winterhalter KE, Meier PJ. Identification of organic anion transporting polypeptide 4 (Oatp4) as a major full-length isoform of the liver-specific transporter-1 (rlst-1) in rat liver. FEBS Lett. 2000;474:242–245. doi: 10.1016/s0014-5793(00)01596-9. [DOI] [PubMed] [Google Scholar]
- 19.Abe T, Kakyo M, Sakagami H, Tokui T, Nishio T, Tanemoto M, Nomura H, Hebert SC, Matsuno S, Kondo H, Yawo H. Molecular characterization and tissue distribution of a new organic anion transporter subtype (oatp3) that transports thyroid hormones and taurocholate and comparison with oatp2. J Biol Chem. 1998;273:22395–22401. doi: 10.1074/jbc.273.35.22395. [DOI] [PubMed] [Google Scholar]
- 20.Kinne A, Schülein R, Krause G. Primary and secondary thyroid hormone transporters. Thyroid Res. 2011;4(suppl 1):S7. doi: 10.1186/1756-6614-4-S1-S7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Gong L, Aranibar N, Han YH, Zhang Y, Lecureux L, Bhaskaran V, Khandelwal P, Klaassen CD, Lehman-McKeeman LD. Characterization of organic anion-transporting polypeptide (Oatp) 1a1 and 1a4 null mice reveals altered transport function and urinary metabolomic profiles. Toxicol Sci. 2011;122:587–597. doi: 10.1093/toxsci/kfr114. [DOI] [PubMed] [Google Scholar]
- 22.Chrostowski MK, McGonnigal BG, Stabila JP, Padbury JF. Role of the L-amino acid transporter-1 (LAT-1) in mouse trophoblast cell invasion. Placenta. 2010;31:528–534. doi: 10.1016/j.placenta.2009.12.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Campbell WA, Thompson NL. Overexpression of LAT1/CD98 light chain is sufficient to increase system L-amino acid transport activity in mouse hepatocytes but not fibroblasts. J Biol Chem. 2001;276:16877–16884. doi: 10.1074/jbc.M008248200. [DOI] [PubMed] [Google Scholar]
- 24.Pochini L, Scalise M, Galluccio M, Indiveri C. Membrane transporters for the special amino acid glutamine: structure/function relationships and relevance to human health. Front Chem. 2014;2:1–23. doi: 10.3389/fchem.2014.00061. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary data