Abstract
Hydrogel-coated coils are truly detachable coils with a platinum core covered with hydrogel. The coils are available in 0.018- and 0.035-in systems. These coils have the ability to expand up to four times their size ∼20 minutes after deployment, thus providing a very effective mechanical vascular occlusion effect. The vessel-occlusive effect of these coils is a volume, space-occupying effect, not a thrombotic effect, as seen in fibered coils. Hydrogel-coated coils were originally developed and designed to treat brain aneurysms; however, their use has expanded to peripheral applications. Hydrogel-coated coils have been used in the management of visceral aneurysms, high-flow vascular arteriovenous fistulae, and endoleaks after endovascular thoracic and abdominal aneurysm repair. The purpose of this article is to describe the hydrogel-coated coil system, the mechanism of action, technical details for optimal deployment, and clinical applications.
Keywords: endovascular embolization, coil embolization, endoleak management, visceral aneurysms, portosystemic shunts, interventional radiology
Objectives: Upon completion of this article, the reader will be able to describe the mechanism of action of the hydrogel-coated coil system, and be able to apply this information to decide optimal applications of this coil system in their clinical practice.
Accreditation: This activity has been planned and implemented in accordance with the Essential Areas and Policies of the Accreditation Council for Continuing Medical Education (ACCME) through the joint providership of Tufts University School of Medicine (TUSM) and Thieme Medical Publishers, New York. TUSM is accredited by the ACCME to provide continuing medical education for physicians.
Credit: Tufts University School of Medicine designates this journal-based CME activity for a maximum of 1 AMA PRA Category 1 Credit™. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
Endovascular transcatheter coil embolization is an established technique used to manage a myriad of medical conditions including brain aneurysms, visceral aneurysms, endoleaks, active bleeding, and arteriovenous fistulas.1 2 3 New coil products are now available to facilitate embolization of large and complex lesions. These coil products include longer (>30 cm) and larger diameter (≥30 mm) systems, which are quite effective in the management of complex vascular lesions.
The hydrogel detachable coil system is a unique product that combines a platinum wire with an expandable hydrogel polymer (AZUR; Terumo Medical Corporation, Somerset, NJ) (Fig. 1). These coils are easily detached by connecting them to a battery-operated detachment controller system (Fig. 2). These coil systems were originally developed to treat intracranial aneurysms4 5; however, their use has now expanded to other applications such as the management of visceral aneurysms, peripheral aneurysms, traumatic pseudoaneurysms, endoleaks, and high-flow arteriovenous fistulas. The purpose of this article is to provide a description of the 0.018- and 0.035-in hydrogel-coated coil system, describe preparation and deployment techniques, as well as optimal applications.
Fig. 1.

A magnified view of the hydrogel-coated coil. This coil system is composed of a platinum wire covered by hydrogel polymer. (Courtesy of Terumo Interventional Systems, Somerset, NJ).
Fig. 2.

Battery-operated detachment controller system. The trailing end of the coil is inserted into this unit and the coil deployed by activation of the white oval button. (Courtesy of Terumo Interventional Systems, Somerset, NJ).
Product Description
The AZUR detachable coil is a platinum wire covered by a hydrogel polymer. Once the delivery catheter has been placed in optimal position, the coil is detached using a battery-operated detachment controller system. The detachment controller system is engaged to the trailing end of the coil wire and activated by the push of a button. The coils are available in 0.018- and 0.035-in systems; the 0.018-in coils are ideally advanced through microcatheters with an inner diameter of 0.021 in, and the 0.035-in coils through angiographic catheters with a 0.038-in inner diameter. The AZUR hydrogel-coated coil systems were approved by the FDA in 2009.
In the 0.018-in system, the coil diameters range between 2 and 20 mm and lengths range between 2 and 30 cm. The maximum length of the 2-mm coils is 4 cm, and the maximum length of the 20-mm coils is 30 cm. In the 0.035-in system, coil diameters range between 4 and 20 mm, and the lengths vary between 5 and 30 cm; the maximum length of the 4-mm coils is 15 cm.
Once the coils are deployed within a vessel, contact with blood activates the hydrogel, which starts a process of volume expansion. The hydrogel coating expands by 80% within 3 minutes and reaches maximal expansion approximately 20 minutes after deployment. Hydrogel expansion results in an increase in coil size to approximately three to four times its original size.
The hydrogel-coated coils are considered to be “MR (magnetic resonance) conditional,” which means that a patient can be scanned safely immediately after coil deployment if the static magnetic field used is 3 Tesla or less and the maximum spatial gradient field is 720 Gauss/cm or less.
Mechanism of Action and Animal Experiments
Hydrogel-coated coils were originally developed to treat intracranial aneurysms.6 Initial experimental studies with aneurysm models in rabbits demonstrated that hydrogel coils were very effective in occluding aneurysmal cavities.4 6 Histological analysis demonstrated that aneurysms embolized with hydrogel coils showed complete occlusion of the lesion by replacement of the space with coils, hydrogel, neointima, and a minimal amount of thrombus.4 6 This is an important feature that explains the persistent occlusion rates observed in clinical scenarios where these coils are employed.5
Technical Aspects: Coil Preparation and Deployment
The package insert of the product recommends coil preparation in warm saline before deployment (Fig. 3). This process may take anywhere between 1 and 5 minutes, depending on the temperature of the saline; the warmer the saline, the faster the process. Some operators elect to skip the “preparation” step. In the author's personal experience, the extra time spent in coil preparation is justified because the preparation process softens the coils resulting in an easier, faster, and smoother deployment.
Fig. 3.

Preparation of the hydrogel-coated coil system in warm saline. The coil is immersed within the warm saline for 1–5 minutes to soften the coil before deployment. (Courtesy of Terumo Interventional Systems, Somerset, NJ).
Coil insertion into either the microcatheter or angiographic catheter should be smooth and in a straight line, being particularly careful not to bend or kink the delivery wire as this may result in coil deployment failure. Coils are delivered to the desired target site by slowly and carefully pushing the coil through the delivery catheter. The coils are detached only when the device has reached an optimal position and configuration; if the optimal position cannot be reached, the coil can be withdrawn in its entirety. The recommended maximum reposition time for these coils is approximately 3 minutes, with a reported maximum of 5 minutes.6
If for any reason deployment proves to be difficult (e.g., the coil does not form optimally within the desired target within the time limit or if there is a kink in the wire that makes coil release impossible), then the entire system can be withdrawn before coil detachment precluding an unintended coil deployment or a nontarget embolization.
Clinical Applications
In the author's experience, the hydrogel-coated coil system is ideal for embolization of visceral aneurysms, high-flow vascular shunts, and embolization of endoleaks. The author has gained experience in embolization of visceral aneurysms and endoleaks with this hydrogel-coated coil system, and has just recently started using the 0.035-in system in the embolization of large, high-flow spontaneous portosystemic shunts for the management of hepatic encephalopathy.
Type I-A Endoleak Management
Endoleaks type I-A are present in 1 to 3% of cases after endovascular aneurysm repair. These endoleaks are secondary to poor apposition of the body of the stent graft to the wall of the aorta, resulting in a high-pressure leak of pulsatile blood into the aortic sac.7 8 The options for management of these endoleaks include placement of an aortic cuff cephalad to the existing main body of the stent graft; placement of a bare Palmaz stent to improve the seal; placement of sealing coils to attach the wall of the graft to the wall of the aorta; coil embolization of the leak; and open surgical repair.9
The most challenging problem in the management of type I-A endoleaks is the presence of visceral arteries closely adjacent to the graft attachment site (renal arteries and superior mesenteric artery). Selective catheterization of the type I-A endoleak sac with a microcatheter (Straight Renegade STC; Boston Scientific, Natick, MA) followed by embolization of the endoleak sac with hydrogel-coated coils has been described as a therapeutic alternative for type I-A endoleaks (Fig. 4a–d).8 9 The technique requires state-of-the-art fluoroscopic equipment, and advanced catheter and microcatheter skills.
Fig. 4.
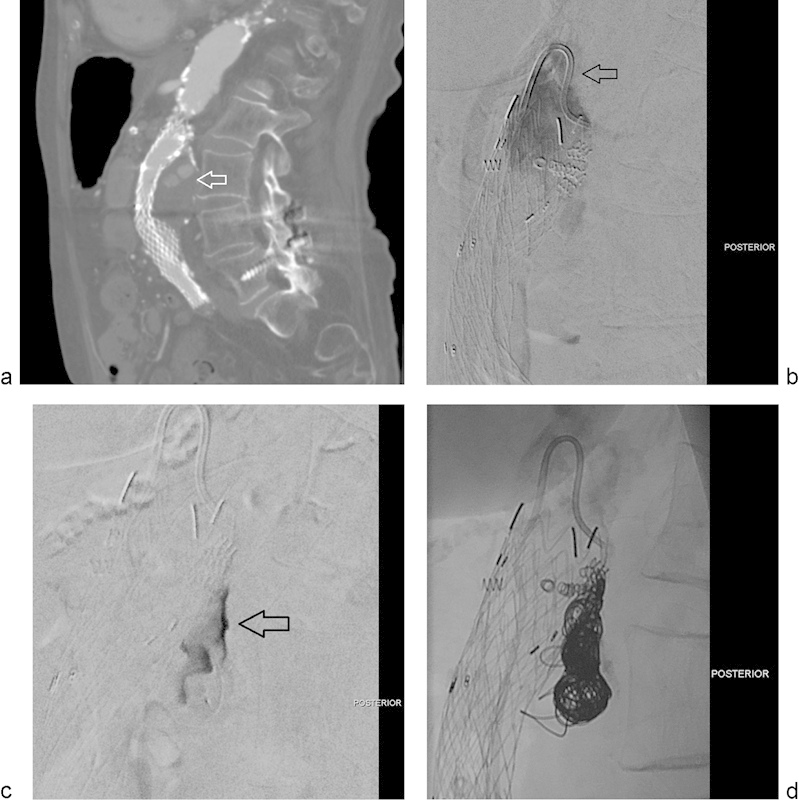
(a) Sagittal reconstruction from a CTA performed in a patient with history of endovascular repair of an abdominal aortic aneurysm. The image shows a type-I endoleak that fills the posterior aspect of the aneurysmal sac (arrow). (b) Selected image of a digital subtraction arteriogram performed in a true lateral view shows contrast injection through a reverse-curve catheter (arrow) aimed posteriorly, toward the expected site of the endoleak. The endoleak is clearly demonstrated. (c) Selected image of a digital subtraction arteriogram performed through a microcatheter within the endoleak sac. The endoleak sac is clearly opacified (arrow). No feeding collaterals are identified. (d) Selected spot film obtained after hydrogel-coated coil packing of the endoleak cavity.
Fig. 4 illustrates a case of an 85-year-old woman who underwent abdominal aortic aneurysm repair with an endograft. She was known to have a large, posterior type I-A endoleak. She was not a good candidate for open repair and had undergone additional aortic cuff placement and placement of multiple APTUS Endostaples (APTUS Endosystem, Sunnyvale, CA); however, the endoleak persisted despite these efforts. The case was discussed with the vascular surgery team and it was agreed that there was no landing zone for an additional cuff. Embolization of the endoleak with hydrogel coils was offered and agreed to be the most suitable option for this patient. Key to success in this procedure was the use of a reverse-curve catheter to engage the endoleak. A microcatheter was advanced into the endoleak sac, followed by embolization of the sac with hydrogel-coated coils. As shown in the images, this patient underwent successful occlusion of the endoleak after selective embolization with hydrogel coils.
The author has successfully used hydrogel-coated coils for this purpose. Key to this success is the unique ability of this coil system to enlarge after deployment, avoiding the potential problem of coil “compaction” that may lead to recanalization of the embolized segment.2 Long-term studies evaluating this treatment modality are not available but would be strongly desired. Probably, the main reason is the fact that this technique is used as a “last-ditch effort” to solve a persistent clinical problem.9 10
Embolization of Visceral Aneurysms
Hydrogel-coated coils are particularly well suited for embolization of visceral aneurysms, including renal, splenic, hepatic, and mesenteric aneurysms. The author's experience has specifically centered in the use of 0.018-in hydrogel-coated coils for this purpose (Fig. 5a–e). Techniques to achieve a complete embolization of the aneurysm may vary, and include exclusion by placing coils distal and proximal to the aneurysm (sandwich technique) or exclusion by packing coils within the aneurysm by super-selective catheterization with a microcatheter.11
Fig. 5.
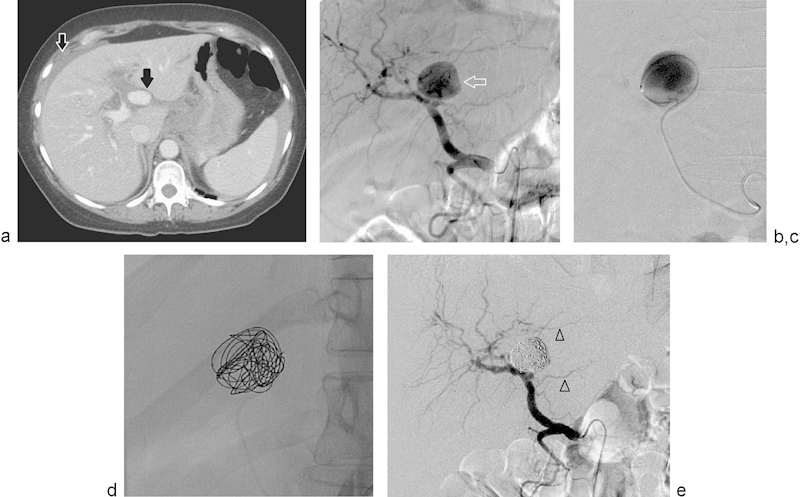
(a) Selected axial image of a triple-phase CT scan of the abdomen shows a large, ovoid, vascular structure located medial to the main portal vein (solid black arrow). There is a small amount of perihepatic fluid (open white arrow). (b) Selected image of a selective injection of the celiac trunk shows a large, saccular arterial vascular lesion arising from the left hepatic artery (arrow). (c) Selected image of a super-selective injection within the saccular vascular structure using a microcatheter. No collateral branches are identified. (d) Selected spot film during coil embolization shows a framing coil and coil packing of the vascular lesion with hydrogel-coated coils. (e) Selected image of a selective injection of the celiac trunk after coils embolization of the left hepatic artery vascular lesion shows complete obliteration of flow to this lesion with coils. Flow is preserved to the branches of the left hepatic artery (arrowheads).
Fig. 5 illustrates the case of a 70-year-old woman who presented to the emergency department with acute abdominal pain and hemodynamic instability. A computed tomographic scan revealed blood in the perihepatic area and a large aneurysm or pseudoaneurysm of the left hepatic artery. She did not have any history of trauma or previous percutaneous interventions. Angiography demonstrated a large aneurysm of the left hepatic artery, and superselective catheterization of the lesion was achieved using a microcatheter. In these cases, the author used one or two “framing” (space occupying) coils, and then filled the vascular cavity with hydrogel-coated coils. As mentioned previously, the most important characteristic of the hydrogel-coated coil system and the reason why it may be preferable in such cases is their ability to enlarge after deployment. This enlargement increases the area of embolization, and prevents the coil compaction identified and described with the use of other coil systems.2 12 Long-term follow-up studies specifically addressing this issue with this specific coil system are necessary.12
Embolization of Spontaneous Portosystemic Shunts
Standard management of hepatic encephalopathy in patients with liver cirrhosis includes the use of lactulose and rifaximin.13 The combination of these two drugs has proven to be more effective than the use of lactulose alone.13 However, certain patients will not respond to such standard treatments and are classified as “resistant” or “refractory” to medical management. In these patients, it is useful to evaluate for spontaneous portosystemic shunts, as these shunts may worsen the patient's encephalopathy. Embolization of spontaneous portosystemic shunts has recently been described as an effective technique in the management of patients with intractable encephalopathy.14 15
In these patients, access into the portal venous system is achieved either through a transhepatic or trans-splenic access route. Once reliable vascular access into the portal venous system is achieved, a vascular sheath is placed and selective catheterization of the shunts is performed using standard angiographic catheter technique. In these cases, the author has used the 0.035-in hydrogel-coated coil and embolized the spontaneous portosystemic shunts to assist in the management of hepatic encephalopathy (Fig. 6a–e). Fig. 6 demonstrates the case of an 80-year-old woman with intractable encephalopathy. This patient had two large portosystemic shunts, one connecting into large esophageal varices and the second forming a large splenorenal shunt; the case illustrates embolization of both shunts. Preliminary results have been very positive with a significant reduction of the symptoms related to the baseline problem.
Fig. 6.
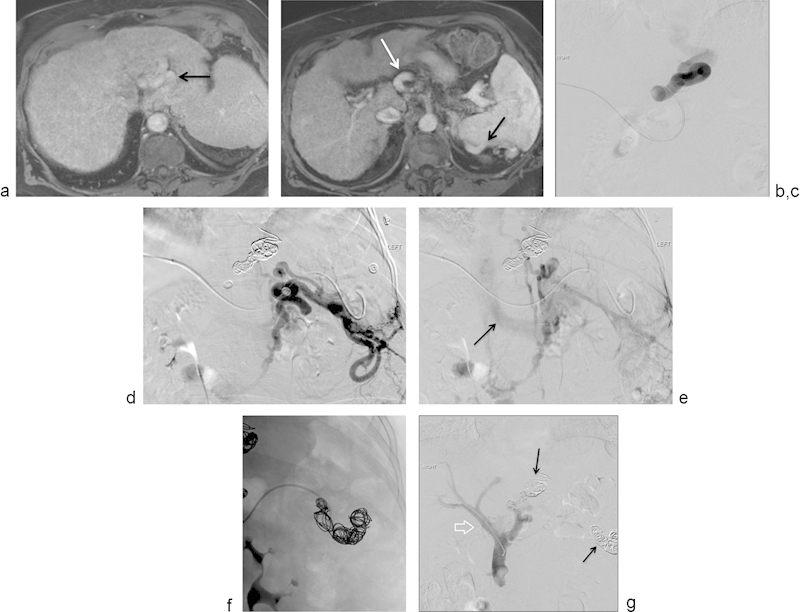
(a) Selected axial image of a contrast-enhanced MRI showing large vascular structures posterior to the left lobe of the liver, corresponding to large esophageal varices (arrow). (b) Selected axial image of a contrast-enhanced MRI showing large vascular structures anterior to the celiac trunk (white arrow) and posterior to the spleen (black arrow). These venous structures were thought to be part of a large splenorenal shunt. (c) Selected image from a digital subtraction angiogram showing injection into a pocket of large esophageal varices. No flow into the main portal vein is demonstrated. (d) Selected image from a digital subtraction angiogram showing injection into a large, complex, splenorenal shunt. (e) Selected delayed image from a digital subtraction angiogram after injection of contrast into a large, complex, splenorenal shunt demonstrates opacification of the left renal vein (arrow), draining into the inferior vena cava. (f) Spot film that shows the process of embolization of the large splenorenal shunt with hydrogel-coated coils. (g) Selected image from a digital subtraction direct portogram showing successful coil embolization of the portosystemic shunts from the left gastric vein and from the splenorenal shunt. There is now antegrade flow into the main portal vein (arrow) and intrahepatic branches. Patient's encephalopathy improved significantly after this intervention.
Conclusion
Hydrogel-coated coils are reliable vessel-occlusion devices. This coil embolization system has the unique property of progressive enlargement after deployment and occlusion by volume occupation and tissue replacement with neo-intima rather than by a superimposed vessel thrombosis mechanism. Initial experience using hydrogel-coated coils for certain complex clinical problems described in this article has been very positive.
References
- 1.Saddekni S, Abdel Aal A K, Gaddikeri S, Hadi S, Singh S P. Percutaneous transcatheter embolization of pulmonary artery pseudoaneurysms causing massive hemoptysis: two different case scenarios. Vasc Endovascular Surg. 2014;48(2):171–175. doi: 10.1177/1538574413513339. [DOI] [PubMed] [Google Scholar]
- 2.Yasumoto T, Osuga K, Yamamoto H. et al. Long-term outcomes of coil packing for visceral aneurysms: correlation between packing density and incidence of coil compaction or recanalization. J Vasc Interv Radiol. 2013;24(12):1798–1807. doi: 10.1016/j.jvir.2013.04.030. [DOI] [PubMed] [Google Scholar]
- 3.López-Benítez R, Hallscheidt P, Kratochwil C. et al. Protective embolization of the gastroduodenal artery with a one-HydroCoil technique in radioembolization procedures. Cardiovasc Intervent Radiol. 2013;36(1):105–110. doi: 10.1007/s00270-012-0361-9. [DOI] [PubMed] [Google Scholar]
- 4.Reinges M H, Krings T, Drexler A Y. et al. Bare, bio-active and hydrogel-coated coils for endovascular treatment of experimentally induced aneurysms. Long-term histological and scanning electron microscopy results. Interv Neuroradiol. 2010;16(2):139–150. doi: 10.1177/159101991001600205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.White P M, Lewis S C, Gholkar A. et al. Hydrogel-coated coils versus bare platinum coils for the endovascular treatment of intracranial aneurysms (HELPS): a randomised controlled trial. Lancet. 2011;377(9778):1655–1662. doi: 10.1016/S0140-6736(11)60408-X. [DOI] [PubMed] [Google Scholar]
- 6.Kallmes D F, Fujiwara N H. New expandable hydrogel-platinum coil hybrid device for aneurysm embolization. AJNR Am J Neuroradiol. 2002;23(9):1580–1588. [PMC free article] [PubMed] [Google Scholar]
- 7.Bashir M R, Ferral H, Jacobs C, McCarthy W, Goldin M. Endoleaks after endovascular abdominal aortic aneurysm repair: management strategies according to CT findings. AJR Am J Roentgenol. 2009;192(4):W178–W186. doi: 10.2214/AJR.08.1593. [DOI] [PubMed] [Google Scholar]
- 8.Kim S M, Ra H D, Min S I, Jae H J, Ha J, Min S K. Clinical significance of type I endoleak on completion angiography. Ann Surg Treat Res. 2014;86(2):95–99. doi: 10.4174/astr.2014.86.2.95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Törnqvist P, Resch T. Endoleaks after EVAR and TEVAR: indications for treatment and techniques. J Cardiovasc Surg (Torino) 2014;55(2) 01:105–114. [PubMed] [Google Scholar]
- 10.White S B, Stavropoulos S W. Management of endoleaks following endovascular aneurysm repair. Semin Intervent Radiol. 2009;26(1):33–38. doi: 10.1055/s-0029-1208381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hemp J H, Sabri S S. Endovascular management of visceral arterial aneurysms. Tech Vasc Interv Radiol. 2015;18(1):14–23. doi: 10.1053/j.tvir.2014.12.003. [DOI] [PubMed] [Google Scholar]
- 12.Koganemaru M, Abe T, Nonoshita M. et al. Follow-up of true visceral artery aneurysm after coil embolization by three-dimensional contrast-enhanced MR angiography. Diagn Interv Radiol. 2014;20(2):129–135. doi: 10.5152/dir.2013.13236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sharma B C, Sharma P, Lunia M K, Srivastava S, Goyal R, Sarin S K. A randomized, double-blind, controlled trial comparing rifaximin plus lactulose with lactulose alone in treatment of overt hepatic encephalopathy. Am J Gastroenterol. 2013;108(9):1458–1463. doi: 10.1038/ajg.2013.219. [DOI] [PubMed] [Google Scholar]
- 14.Gopalakrishna R, Hurkadli P S, Puthukudy N K, Nair H R. Embolization of portosystemic shunt for treatment of recurrent hepatic encephalopathy. J Clin Exp Hepatol. 2014;4(1):60–62. doi: 10.1016/j.jceh.2013.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Franzoni LdeC, de Carvalho F C, Garzon R G. et al. Embolization of splenorenal shunt associated to portal vein thrombosis and hepatic encephalopathy. World J Gastroenterol. 2014;20(42):15910–15915. doi: 10.3748/wjg.v20.i42.15910. [DOI] [PMC free article] [PubMed] [Google Scholar]


