Abstract
The dNab2 polyadenosine RNA binding protein is the D. melanogaster ortholog of the vertebrate ZC3H14 protein, which is lost in a form of inherited intellectual disability (ID). Human ZC3H14 can rescue D. melanogaster dNab2 mutant phenotypes when expressed in all neurons of the developing nervous system, suggesting that dNab2/ZC3H14 performs well-conserved roles in neurons. However, the cellular and molecular requirements for dNab2/ZC3H14 in the developing nervous system have not been defined in any organism. Here we show that dNab2 is autonomously required within neurons to pattern axon projection from Kenyon neurons into the mushroom bodies, which are required for associative olfactory learning and memory in insects. Mushroom body axons lacking dNab2 project aberrantly across the brain midline and also show evidence of defective branching. Coupled with the prior finding that ZC3H14 is highly expressed in rodent hippocampal neurons, this requirement for dNab2 in mushroom body neurons suggests that dNab2/ZC3H14 has a conserved role in supporting axon projection and branching. Consistent with this idea, loss of dNab2 impairs short-term memory in a courtship conditioning assay. Taken together these results reveal a cell-autonomous requirement for the dNab2 RNA binding protein in mushroom body development and provide a window into potential neurodevelopmental functions of the human ZC3H14 protein.
Introduction
Nascent RNA transcripts in eukaryotic cells associate with RNA binding proteins that regulate gene expression via effects on splicing, 3’-end cleavage, polyadenylation, export, trafficking, translation and ultimately destruction (McKee and Silver, 2007). The importance of these interactions is underscored by the prevalence of diseases linked to defects in RNA binding proteins (Cooper et al., 2009). The frequency of neurological impairments among these diseases suggests that post-transcriptional regulatory mechanisms are particularly important in neurons. For example, the most common form of inherited intellectual disability, Fragile-X Syndrome, is caused by loss of the Fragile X Mental Retardation Protein (FMRP) RNA binding protein (Santoro et al., 2011).
One key family of proteins that contributes to post-transcriptional regulation interacts with the 3’-polyadenosine (poly(A)) tails of mRNAs in both the nucleus and cytoplasm. These polyadenosine RNA binding proteins (Pabs) modulate transcript export from the nucleus as well as stability and translation in the cytoplasm (Goss and Kleiman, 2013; Wigington et al., 2014). The human PABPN1 protein (called Pabp2 inDrosophila) localizes to the nucleus, enhances poly(A) polymerase activity, and modulates alternative polyadenylation (Banerjee et al., 2013). The most abundant human Pab, cytoplasmic PABPC1 (called Pab1 in Drosophila), enhances translation by bridging an interaction between 3’ poly(A) tails and initiation factors at the 5’-mG cap (Charlesworth et al., 2013; Goss and Kleiman, 2013). While these Pab proteins recognize polyadenosine RNA via RNA Recognition Motif (RRM) domains, a recently described Pab protein termed ZC3H14 (Zinc finger CysCysCysHis domain-containing protein 14) does so via zinc finger motifs and thus constitutes a new class of Pab proteins (Kelly et al., 2007; Kelly et al., 2014).
The biological importance of ZC3H14 is highlighted by the recent finding that mutation of the gene encoding ZC3H14 causes an autosomal recessive form of intellectual disability (Pak et al., 2011). Patients homozygous for ZC3H14 loss-of-function mutations have very low IQs ranging from 30–50 compared to the average of 100 (Ropers, 2010), indicating that ZC3H14 plays a critical role in the brain (Pak et al., 2011; Kelly et al., 2012). ZC3H14 [also termed mSut2; (Guthrie et al., 2011)] is expressed highly in the brain and co-localizes with poly(A) mRNA speckles in hippocampal neurons (Pak et al., 2011). Consistent with a critical role for ZC3H14 in the brain, adult Drosophila lacking the ZC3H14 homolog, dNab2, have extended poly(A) tails on a subset of brain mRNAs, impaired motor response in a negative geotaxis assay, and reduced survival that is specifically rescued by neuronal expression of dNab2 or human ZC3H14 (Pak et al., 2011; Kelly, 2013; Kelly et al., 2014).
We have previously found that expression of human ZC3H14 in the developing nervous system rescues multiple defects in dNab2 null flies, including extended poly(A) tails, reduced adult eclosion, and impaired locomotor responses in a negative geotaxis assay (Kelly et al., 2014). These effects of ZC3H14 expression is indicative of a significant level of functional overlap between the Nab2 and ZC3H14 proteins in developing neurons, providing strong justification for the use of Drosophila to model ZC3H14 function. The role of Nab2/ZC3H14 in the developing nervous system has not yet been explored. However, work on Nab2/ZC3H14 in neuronal disease is accelerating and has linked Nab2 homologs to a Tau-induced model of Alzheimer’s pathology in C. elegans and cultured vertebrate cells (Guthrie et al, 2009, 2011). Thus there is significant interest in uncovering the role of the Nab2/ZC3H14 family in both neurodevelopment and disease.
Here we exploit the Drosophila model to assess requirements for dNab2 in the developing brain. We find that dNab2 loss disrupts development of the mushroom bodies (MBs), twin neuropil structures required for learning and memory (Heisenberg, 2003). dNab2-deficient MBs develop two highly penetrant defects in axonal development: the first is overgrowth of axons across the brain midline, and the second is a distinct defect in which these same axons apparently fail to branch along developmentally stereotyped paths. These neuronal defects in dNab2 zygotic mutants imply a role for the dNab2 protein in regulating neuronal RNAs within MB neurons. Consistent with this hypothesis, we find dNab2 to be expressed highly in the cell bodies of MB neurons. Complementary RNAi-depletion and single-cell tracing experiments confirm that dNab2 acts within brain neurons to control axon morphology and short-term memory. In sum, these genetic and cellular data reveal a key role for the dNab2 RNA binding protein in supporting developmental axonogenesis in MB neurons. Given the conserved nature of many neurodevelopmental mechanisms, our data provide a potential paradigm for understanding roles of dNab2/ZC3H14 in development and disease.
MATERIALS AND METHODS
Drosophila stocks and genetics
All crosses and stocks were maintained in standard conditions unless otherwise noted. The dNab2ex3 strong loss-of-function mutant and dNab2pex41 precise excision isogenic control stocks were described previously (Pak et al., 2011). The following Gal4 stocks were used to drive expression in various subsets of mushroom body neurons: c739-Gal4 (α/β lobes; BL #7362), NP7175-Gal4 (core α/β lobes; DGRC #114120), 201Y-Gal4 (enriched in γ-lobes; BL #4440), OK107-Gal4 (all MB lobes; BL #854), c305a-Gal4 (α’/β’-lobes; BL #30829). C155-Gal4 (all neurons commencing at embryonic stage 12; BL #458) was used to drive expression in all neurons. The following transgenic stocks were used in this study: UAS-CD8-GFP (Lee and Luo, 1999) and UAS-dNab2:Flag (Pak et al., 2011). Single-cell marking of MB neurons was achieved using the 3R MARCM stock (BL #44408) in combination with OK107-Gal4 and dNab2ex3.
Brain dissections and immunohistochemistry
Brain dissections and staining were performed as described previously (Wu and Luo, 2006). Briefly, brains of anesthetized animals were dissected in PTN buffer (0.1M NaPO4, 0.1% Triton X-100), fixed in 4% paraformaldehyde (Electron Microscopy Sciences), and then stained overnight with primary antibodies diluted in PTN. Following several washes, brains were incubated with the appropriate fluorescently conjugated secondary antibody (1:250) in PTN for 3 hours at room temperature, washed in PTN, and then mounted in Vectashield (Vector Labs). The polyclonal antibody recognizing dNab2 has been described previously (Pak et al., 2011) and was preabsorbed on fixed Drosophila embryos. The 1D4 anti-FasII hybridoma (1:20) developed by C. Goodman (Grenningloh et al., 1991) was obtained from the Developmental Studies Hybridoma Bank (DSHB). GFP polyclonal antibody (1:500) was obtained from Aves Labs. Quantitation of MB phenotypes was performed as described in Michel et al., 2004 (see text for detailed explanation).
Conditioned Courtship Behavior
For courtship behavioral training, virgin males of the appropriate genotype were collected between 0 and 6 hours after eclosion and transferred to individual food vials (Ejima and Griffith, 2011). All flies were maintained at 25°C in a 12:12 light:dark cycle at 50% humidity. Behavioral tests were performed in a separate room maintained at 25°C and 50% humidity and illuminated under a constant 130 V white light Kodak Adjustable Safelight Lamp mounted above the courtship chambers. Behavior was digitally recorded using a Sony DCR-SR47 Handycam with Zeiss optics. Subsequent digital video analysis of male courtship behavior was quantified using iMovie software (Apple). The male Courtship Index (CI) was calculated as the total time each male was observed performing courting behavior divided by the total time assayed, as described in (Siegel and Hall, 1979).
Virgin female wildtype (Canton S) flies were collected and kept in vials in groups of 10. Male flies were aged for 3 days prior to behavioral training and testing. All tests were performed during the relative light phase. Five-day old mated Canton S females were used for training. Virgin female Canton S targets used were 4 days old. Male flies were assigned to random groups the day of training, and assays were set up and scored blind. Male flies were transferred without anesthesia to one half of a 15 mm partitioned mating chamber from Aktogen (aktogen.com) that contained a previously mated Canton S female in the other partitioned half. Males were allowed to acclimate for 1 minute, and then the partition between the male and female was removed. Male flies were then trained for 60 minutes. After 60 minutes, male flies were transferred within 2 minutes without anesthesia to one half of a clean partitioned mating chamber that contained a virgin Canton S female in the other partitioned half. The partition was removed and the flies were recorded for 10 minutes. A total of 20 flies were scored for each genotype, both trained and sham. To determine significance among individuals of the same genotype for the learning phase of this assay, a two-tailed paired t-test was performed. A two-tailed unpaired t-test was performed to determine significant differences between sham and untrained male flies of the same genotype.
Statistics
All statistical analyses were performed in IBM SPSS statistics, version 22.0.0.0. To determine whether there was a significant difference in the number of brains displaying β-lobe fusion defects in the rescue experiment shown in Figure 5, all categories of fusion were collapsed in a single ‘fusion’ category and the total number of brains in this fusion category was compared to the number of brains showing no fusion using the non-parametric Mann-Whitney U test.
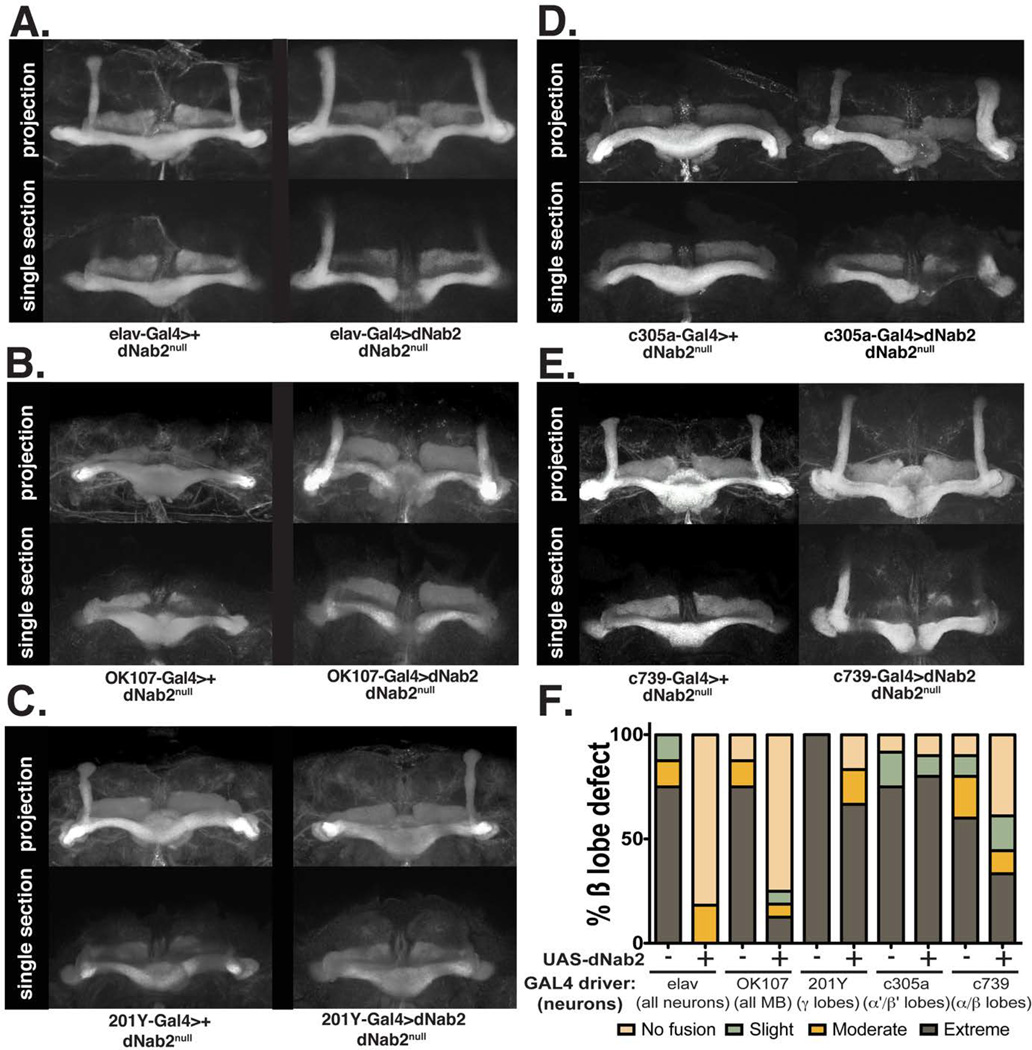
Figure 5. Mushroom body-specific expression of dNab2 rescues the β-lobe morphology defects in dNab2ex3 homozygous null mushroom bodies.
Representative images of anti-FasII staining of dNab2ex3 homozygous null brains expressing a suite of Gal4 drivers with or without a UAS-dNab2 transgene. The following Gal4 drivers were used: (A) the pan-neuronal driver elav-Gal4 (genotype: elav-Gal4/+;;dNab2ex3/dNab2ex3, n=8, or elav-Gal4/UAS-dNab2;; dNab2ex3/dNab2ex3, n=11); (B) the pan-MB driver OK107-Gal4 (genotype: dNab2ex3/dNab2ex3;OK107-Gal4/+, n=8, or UAS-dNab2/+;;dNab2ex3/dNab2ex3;OK107-Gal4/+, n=16); (C) the γ-lobe driver 201Y-Gal4 (genotype: 201Y-Gal4/+; dNab2ex3/dNab2ex3, n=4, or UAS-dNab2/+;201Y-Gal4/+; dNab2ex3/dNab2ex3, n=6); (D) the α’/β’-lobe driver c305a-Gal4 (genotype: c305a-Gal4/+; dNab2ex3/dNab2ex3, n=12, or UAS-dNab2/+;c305a-Gal4/+; dNab2ex3/dNab2ex3, n=10); or (E) the α/β-lobe c739-Gal4 driver (genotype: c739-Gal4/+; dNab2ex3/dNab2ex3, n=10, or UAS-dNab2/+;c739-Gal4/+; dNab2ex3/dNab2ex3, n=18). (F) Quantification of the frequency of β-lobe fusion defects according to the scoring system used in Michel et al. (2004). Note that re-expression of dNab2 in all neurons (Mann-Whitney U=80, p<0.05) or in all mushroom body neurons (Mann-Whitney U=104, p<0.05) significantly rescued the dNab2ex3 homozygous null β-neuron midline fusion phenotype.
Results
dNab2 loss disrupts structure of the mushroom bodies
To begin to assess the neurodevelopmental functions of proteins in the ZCH14 family, we exploited our Drosophila dNab2 model (Pak et al., 2011; Kelly et al., 2014). As ZC3H14 is highly expressed in hippocampal neurons and patients with loss of function mutations in this gene show cognitive defects (Pak et al., 2011), these analyses focused on the mushroom bodies (MBs) due to their roles in specific forms of learning and memory (Heisenberg, 2003). As shown in Fig. 1A, each mushroom body (MB) is divided into α/α’, β/β’ and γ-lobes composed of bundled axons that project from a dorsally located group of ~2000 Kenyon cells, with later growing axons following a path through the central core of earlier pioneer axons (Armstrong et al., 1998; Lee et al., 1999). The β/β’ branches and γ-lobes project medially toward the ellipsoid and fan-shaped bodies of the midbrain, while the α/α’ branches project dorsally. The neuronal adhesion protein Fasciclin-II (FasII) is enriched on α and β axon branches and to a lesser extent on the γ-lobes and regions of the Drosophila central complex (Crittenden et al., 1998; Fushima and Tsujimura, 2007) and was thus used as a marker to assess the effect of dNab2 loss on MB structure. This analysis used dNab2ex3 homozygous null flies, which contain an RNA and protein null deletion of the dNab2 gene caused by imprecise excision of a nearby P-element that removes the transcriptional start site and approximately half of the coding sequence (Pak et al., 2011), and control precise excision dNab2pex41 homozygotes (Fig. 1A,B). The pex41 allele was generated by a precise excision of the original P-element found in stock EY08422 that was used to generate the dNab2ex3 allele (Pak et al., 2011).
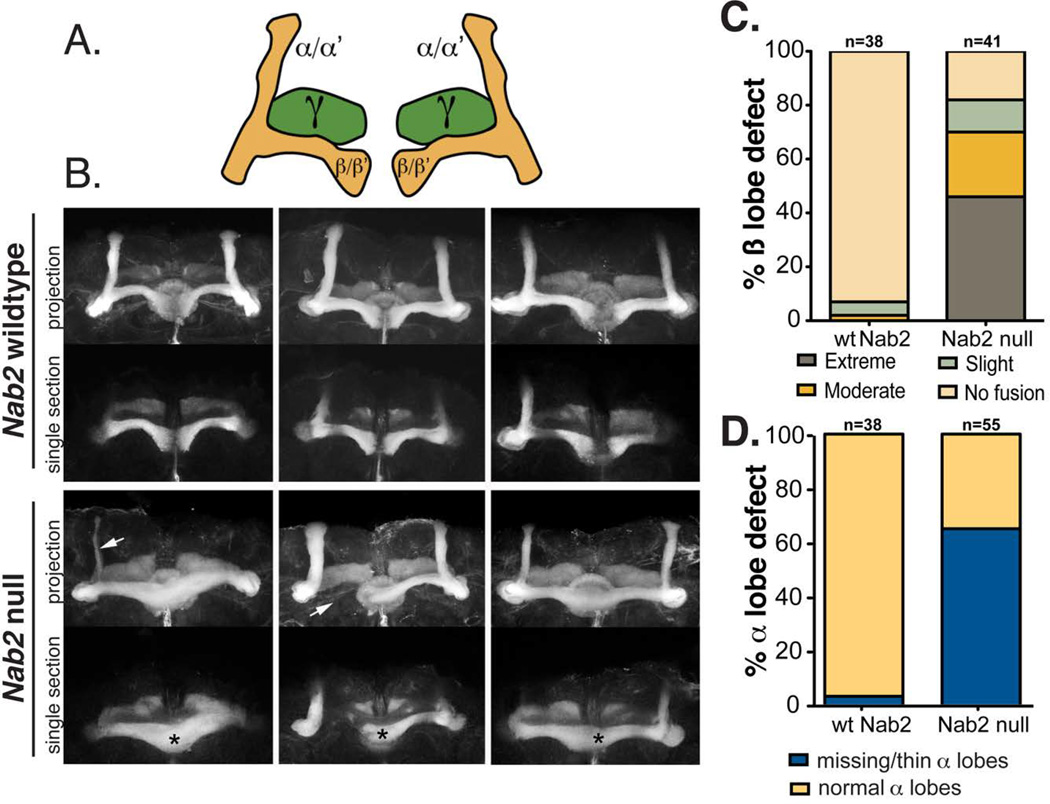
Figure 1. dNab2 is required for proper development of the Drosophila mushroom body neurons.
A, Diagram of the adult Drosophila mushroom body lobes depicting the axons of the medially projecting gamma (γ) neurons (green), the vertical alpha (α) and alpha prime (α’) neurons, and the medially projecting beta (β) and beta-prime (β’) neurons. B, Fasciclin II (FasII) antibody staining of three control (dNab2pex41) brains or three dNab2 null (dNab2ex3) brains. Maximum intensity Z-stack projections (projection) as well as single 1.5 µm sections in the transverse plane of the β-lobes are shown. β-lobes of control brains rarely cross the mid-line and these brains have well-formed α-lobes, while dNab2ex3 null brains have β-lobes that often project to the contralateral hemisphere and appear to fuse (black asterisks) or have missing α or β lobes (white arrows). C, Quantification of the frequency of the dNab2ex3 null β-lobe defect according to the scoring system described in the text (defined by Michel et al. (2004). D, Quantification of the frequency the dNab2ex3 null α-lobe defect for missing or thinned lobes.
Serial optical sectioning of FasII-stained dNab2ex3 brains reveals thinned or missing MB α and β lobes (arrows in 1B) and overgrowth of β lobes across the midline of the brain (asterisks in 1B). By contrast, loss of dNab2 has no discernable effect on the structure of the MB γ-lobes. We also noted subtle defects in the pattern of FasII staining of the ellipsoid body (EB) in dNab2ex3 homozygous null brains and those with dNab2 depleted from neurons using an RNAi transgene co-expressed with the Dicer2 protein (elav-Gal4/Y;UAS-dNab2RNAi) (Supplemental Fig. S1). Ellipsoid body neurons normally project axons medially towards the midline and eventually form a closed ring structure (Zheng et al., 2006). dNab2ex3 homozygous null brains and dNab2-RNAi depleted neurons lack this completely closed ring structure and leave an opening along the ventral side of the ring (Fig. S1). Thus, dNab2 loss affects the morphological development of a subset of structures within the brain, with a particularly discernable effect on the MB α and β lobes.
The effect of dNab2 loss on β-lobe development was quantitated according to the parameters used by Michel et al (2004) for analysis of MB defects in adult brains lacking the Drosophila Fragile-X protein, dFmr1. Briefly, extreme fusion was defined as a FasII-positive neuron bundle crossing the midline that was of equal or greater thickness than the adjacent β lobes; moderate fusion was defined as a substantial FasII-positive fiber bundle crossing the midline that was less than the width of the β-lobes; mild fusion was defined as a thin strand of FasII-positive fibers crossing the midline. β-lobe crossing in dNab2ex3 homozygous null brains is often symmetric, but in some cases only one β-lobe crosses the midline; this phenotype is more apparent when the contralateral lobe is missing (e.g. the dNab2 mutant brain in middle panel Fig. 1B). By these scoring criteria, β-lobe defects occur in slightly more than 80% of dNab2ex3 homozygous brains while α-lobe defects occur in approximately 65% of dNab2ex3 homozygous brains. Each of these lobe defects occurs in less that 10% of control (i.e. dNab2pex41) brains (Fig. 1C,D). More than half of dNab2ex3 homozygous null brains with β-lobe fusion fall into the ‘severe’ class (approximately 45% of all brains), and in all cases tested, single transverse optical sections confirm that opposing β-lobes in this group are merged at the midline (Fig. 1B, midline).
The FasII antigen marks bundled groups of axons and thus does not provide sufficient resolution to track the path of single axons or subsets of axons within the MBs. To more directly examine axon projection patterns, specific subsets of dNab2ex3 homozygous null or control dNab2pex41 MB neurons were marked by Gal4/UAS-driven expression (Brand and Perrimon, 1993) of a membrane-tethered fluorescent protein CD8-GFP (Fig. 2). This technique illuminates the axon projection paths of the marked neurons. Two Gal4 transgenes were used for this analysis: c739-Gal4, which marks MB α/β neurons, and NP7175-Gal4, which is expressed in adults in approximately 45–70 late-born Kenyon neurons whose axons track through the central core of the α/β lobes (Aso et al., 2009). In control brains, GFP-positive α and β axons (c739>CD8-GFP) substantially overlap with the region of FasII expression in the dorsally projecting α-lobes and medially projecting β-lobes. Importantly, all control β-lobes examined (c739>CD8-GFP; n=4) terminate appropriately short of the brain midline (Fig. 2A and Fig. 2C). By contrast, one-third of dNab2ex3 homozygous null brains examined (n=3) contain GFP-positive β-lobe neurons (c739>CD8-GFP) that project contralaterally across the brain midline (arrows, Fig. 2A and Fig. 2C). Similarly, all control (Nab2pex41) brains examined (n=8) contain GFP-positive core β-lobe axons (NP7175>CD8-GFP) that terminate at the midline while ~80% of homozygous dNab2ex3 brains (n=6) contain core β-lobe axons that cross the midline (Fig. 2B and Fig. 2C). dNab2ex3 null flies expressing CD8-GFP in all α/β mushroom body neurons (c739>CD8-GFP) or in just the core α/β mushroom body neurons (NP7175>CD8-GFP) also have a dramatically increased rate of missing α or β lobes (75% and 100%, respectively) (Fig. 2C). Thinned FasII-positive α-lobes in dNab2ex3 homozygous null brains often lack NP7175-expressing core axons, as revealed by the lack of CD8-GFP fluorescence in merged projections of FasII-positive α-lobes in the NP7175>CD8-GFP background (white asterisk, Fig. 2B). As NP7175 is expressed in late-born Kenyon neurons (Aso et al., 2009; Shin and DiAntonio, 2011), these data suggest that dNab2 loss could impair both projection and branching of developing MB axons.
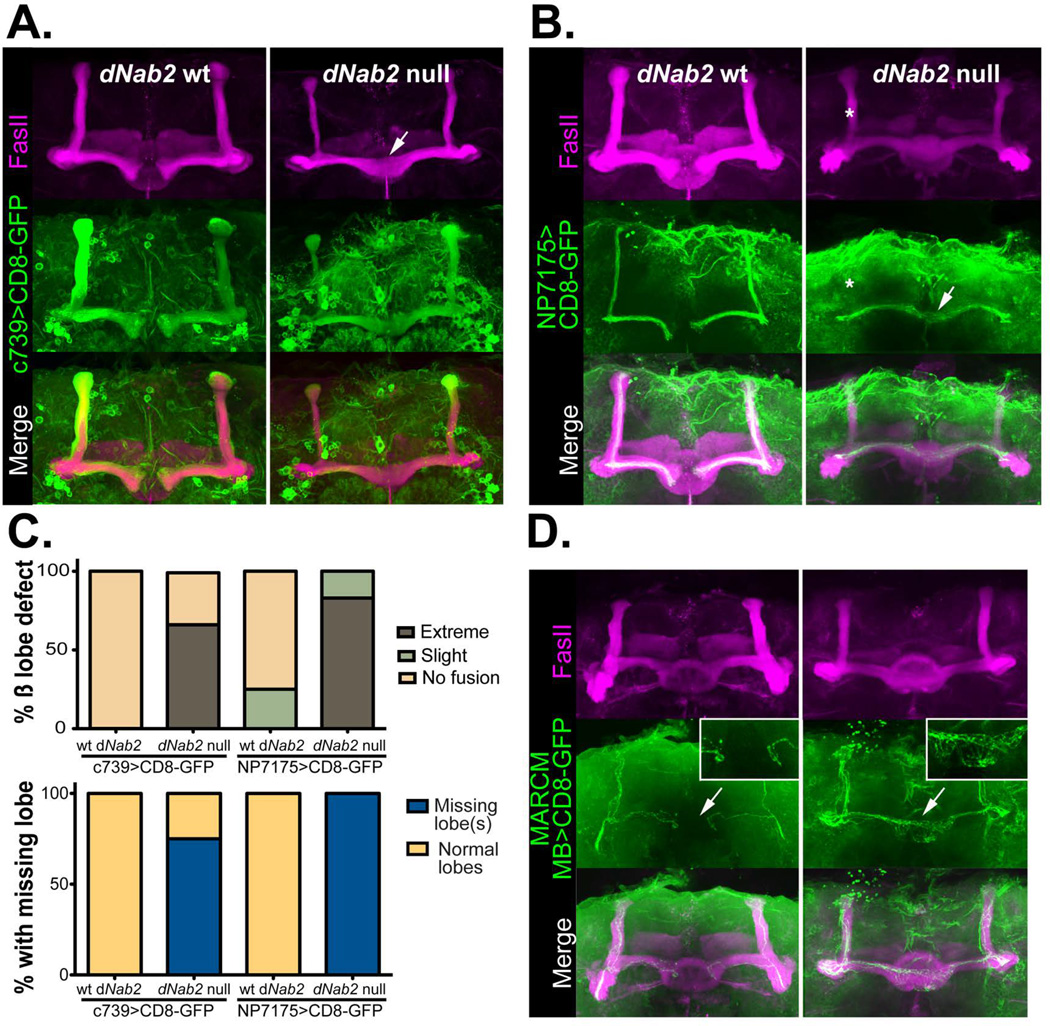
Figure 2. Loss of dNab2 disrupts the morphology of Drosophila mushroom body neurons.
UAS-CD8-GFP (green) was used in combination with cell-type specific Gal4 drivers to mark either all α/β neurons (A; c739) or “core” α/β neurons (B; NP7175) of the MBs in dNab2pex41 control or dNab2ex3 null brains. Brains are co-stained with FasII (magenta) to mark of the α/β lobes. Control β-lobes terminate at the brain midline but lobes from dNab2ex3 null brains overextend into the contralateral hemisphere (white arrows). α-lobe core axons are often absent in dNab2ex3 null brains (white asterisk). Genotypes shown are: (A) C739-Gal4/UAS-CD8-GFP;+/+ or C739-Gal4/UAS-CD8-GFP;dNab2ex3/dNab2ex3 and (B) NP7175-Gal4/Y;UAS-CD8-GFP/+;+/+ or NP7175-Gal4/Y;UAS-CD8-GFP/+; dNab2ex3/dNab2ex3. C, Quantification of the frequency of the β-lobe midline fusion defects (top) and the missing α- or β-lobe defects observed in A and B. Phenotypes were scored according to the criteria outlined in (Michel et al., 2004). Mushroom body β lobes show ‘extreme fusion’ when lobes cross the midline with similar or increased thickness to the rest of the β-lobe (white arrows in A and B). Brains showing thin β-lobes crossing the midline in comparison to the thickness of β-lobes of either side of the midline are scored as having ‘slight fusion.’ D, MARCM analysis of individual neurons (green) in dNab2 control and dNab2ex3 null brains co-stained with FasII (magenta). Wildtype β-axons terminate at the midline but Nab2 mutant axons project to the contralateral side (white arrows). Insets show a magnified regions of the midline in each genotype. Control genotype: hsFLP/+;FRTG13,UAS-CD8-GFP/FRTG13, tubulin-GAL80;dNab2pex41/dNab2pex41;OK107-Gal4/+. Mutant genotype: hsFLP/+; FRTG13,UAS-CD8-GFP/FRTG13,tubulin-GAL80;dNab2ex3/dNab2ex3;OK107-Gal4/+.
The FasII and Gal4 techniques label groups of cells but do not provide conclusive, single-cell resolution of the effects of Nab2 loss on axonal morphology. To carry out fine-scale analysis of individual Kenyon cells in either control dNab2pex41 brains or dNab2ex3 null brains, a membrane bound CD8-GFP marker was used in combination with a MB-specific MARCM system (using the MB driver OK107-GAL4) which allows visualization of one or a few axons within intact MB lobes (Lee and Luo, 2001; Yu et al., 2009; Shin and DiAntonio, 2011). Individual β-lobe axons in dNab2pex4 control brains project medially but arrest short of the brain midline (Fig. 2D, arrow in left panel). By contrast, individual β-lobe axons in dNab2ex3 homozygous null brains project medially but fail to stop, forming a meshed network of axons across the brain midline (Fig. 2D, arrow in right panel). This MARCM data is consistent with the α/β lobe core driver (c739 and NP7175 respectively) data and strongly support a model in which dNab2 is required for proper projection and branching patterns among developing MB axons.
dNab2 is a cell-autonomous regulator of axon branching and growth
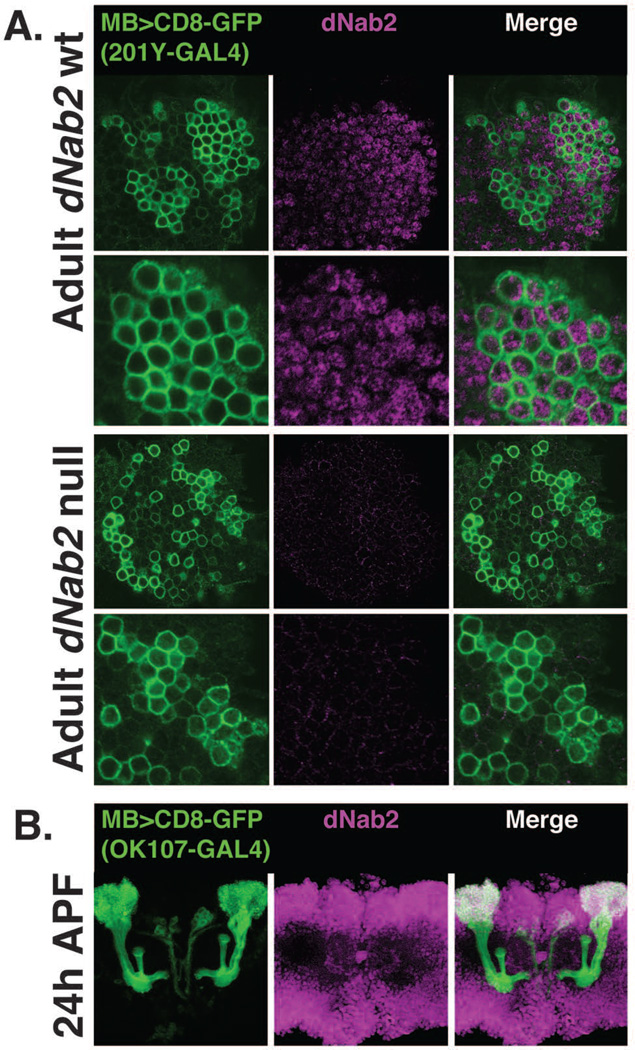
Although the dNab2 protein is expressed in all cell types examined throughout the organism, the viability, locomotor, and wing-posture phenotypes that occur in dNab2ex3 homozygotes can be rescued by pan-neuronal re-expression of wildtype dNab2 protein (Pak et al., 2011; Kelly et al., 2014). The MB defects in dNab2ex3 homozygous null adults suggest a similar autonomous requirement for dNab2 within Kenyon cells. To examine dNab2 expression in the cell bodies of adult Kenyon cells, the plasma membranes of control dNab2pex41 and mutant dNab2ex3 homozygous MB neurons were marked by expression of a membrane-tethered GFP driven by the 201Y-Gal4 driver (201Y>CD8-GFP) and co-stained with the anti-dNab2 polyclonal antibody (Pak et al., 2011). In the adult, the 201Y driver labels γ neurons and core neurons of the α and β lobes (Aso et al., 2009). dNab2 protein is readily detected in both 201Y-positive and 201Y-negative cells within cell bodies of control dNab2pex41 Kenyon cell clusters (Fig. 3A, top panels; see asterisk). The intensity of the dNab2 signal in these cells is not uniform across the plane of section, suggesting that there may be differences in dNab2 protein levels between adult Kenyon cell subgroups. Within each Kenyon cell, dNab2 localizes primarily to the nucleus in foci reminiscent of nuclear speckles (Fig. 3A, zoom), which resembles the localization of the dNab2 ortholog ZC3H14 in adult mouse hippocampal neurons and cultured mammalian cells (Leung et al., 2009; Guthrie et al., 2011; Pak et al., 2011). Importantly, anti-dNab2 fluorescence signal is largely eliminated in dNab2ex3 homozygous adult Kenyon cells (Fig. 3A, bottom panels). The low level of residual staining in dNab2ex3 MB cells is likely due to background reactivity of the anti-dNab2 sera (Pak et al., 2011). These fluorescence data confirm that dNab2 is highly expressed in Kenyon neurons and is absent in dNab2ex3 null adult α/β neurons. However, as α/β lobes begin to develop in the pupal stage (~12 hours after pupal formation, APF), we also sought to test whether dNab2 is expressed in pupal Kenyon cells as they extend α/β axons towards the midline. Brains from 24 hour pupae in which MB cells were marked by CD8-GFP driven by the OK107-Gal4 driver (Lee et al., 1999) were stained with the dNab2 antiserum (Pak et al., 2011). As shown in Figure 3B, dNab2 is expressed widely in the 24 hour pupal brain, including in the cell bodies of CD8-GFP+ developing mushroom body neurons, which is consistent with dNab2 playing a role in MB development.
Figure 3. dNab2 is expressed in the cell bodies of adult and pupa mushroom body neurons.
A, Anti-dNab2 staining (magenta) in Kenyon cells labeled by 201Y-driven expression of UAS-CD8-GFP (green) in wildtype or dNab2ex3 homozygous null flies. dNab2 is readily detected in wildtype Kenyon cell bodies (top panels) but absent in Kenyon cells from dNab2ex3 homozygous null flies (bottom panels). B, dNab2 staining in pupal brains 24 hours after pupal formation (24APF). All MB lobes (γ, α’/β’, and α/β) are present at this time point (Lee et al., 1999) and are labeled by OK107-GAL4 driven expression of UAS-CD8-GFP. dNab2 is present (white overlap in the merged image) in the cell bodies of mushroom body neurons during pupal development.
The broad expression of dNab2 in the pupal brain suggests that dNab2 could act in other cell types to non-autonomously control aspects of Kenyon cell pathfinding. To assess the role of dNab2 within specific groups of mushroom body neurons, we exploited the Gal4/UAS system in combination with a dNab2 RNA-interference transgene (UAS-dNab2RNAi) (Pak et al., 2011) to knockdown dNab2 expression in the developing brain. To assess the efficiency of dNab2 knockdown, dNab2 mRNA transcript levels were examined by quantitative real-time PCR on samples isolated from heads of adult flies with dNab2 knocked down in all neurons (elav-Gal4,UAS-dNab2RNAi) (Fig. 4C). Levels of dNab2 mRNA in these samples were decreased relative to dNab2pex41 controls but less so than in dNab2ex3 homozygous null heads, consistent with the RNA-null nature of the ex3 allele (Pak et al., 2011). Inclusion of a UAS-dicer2 transgene in combination with the UAS-dNab2-RNAi transgene (elav-Gal4,UAS-dNab2RNAi, UAS-dcr2) significantly enhanced knockdown efficiency, but did not lower dNab2 expression to the level observed in dNab2ex3 homozygous null heads, which is consistent with dNab2 being widely expressed in neuronal and non-neuronal cells (Pak et al., 2011). Neuronal expression of Dcr2 alone (elav-Gal4,UAS-dcr2) also significantly lowered levels of dNab2 mRNA in adult heads (Fig. 4C), although not to the level achieved with the dNab2 RNAi transgene, suggesting that Nab2 levels may be regulated by a Dcr2-dependent mechanism in vivo.
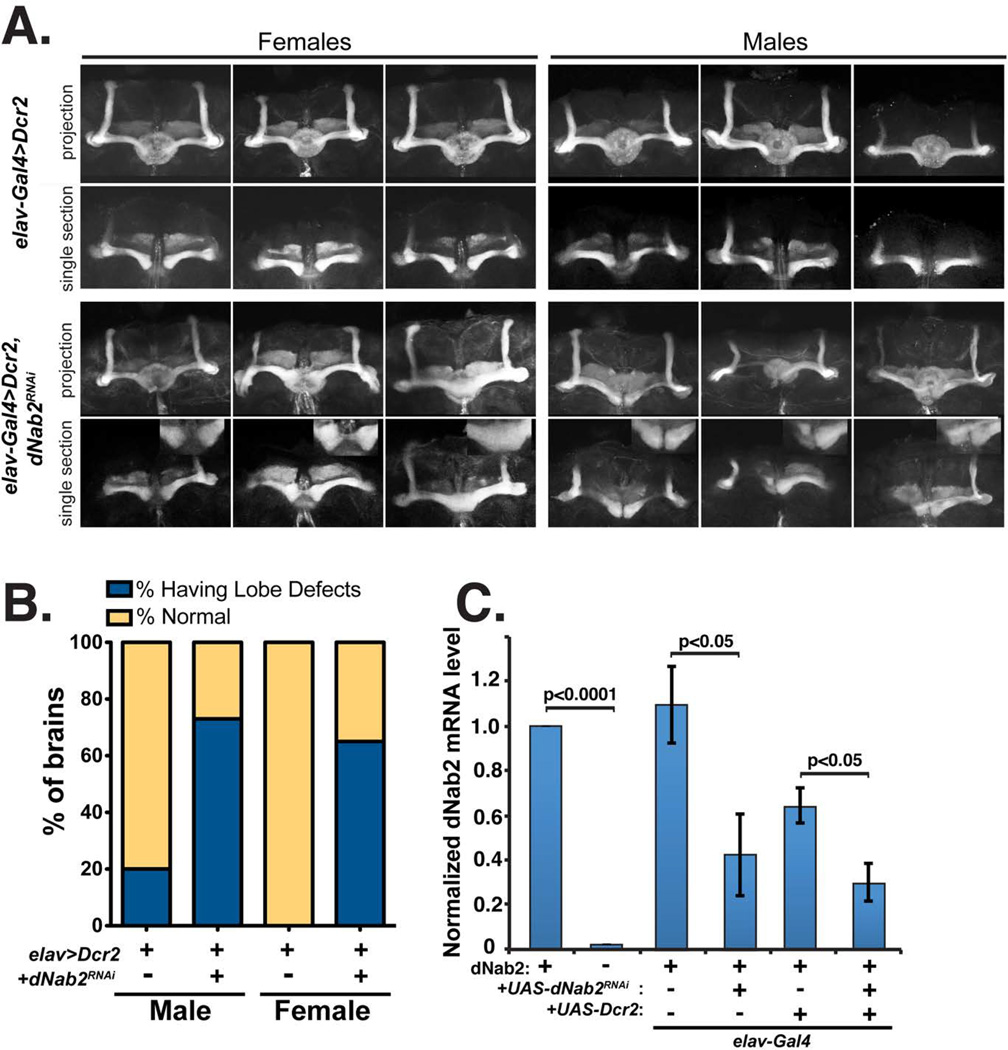
Figure 4. dNab2 is required in neurons for mushroom body development.
A, Merged projections (projection) and single 1.5 µm transverse slices (midline) of FasII-positive α/β-lobes of Dcr2 control flies (top, genotype: elav-Gal4/+ or Y; UAS-dcr2/+) or flies with RNAi knockdown of dNab2 in all neurons (bottom, genotype: elav-Gal4/+ or Y; UAS-dcr2/UAS-dNab2RNAi). dNab2-depleted neurons show evidence of mushroom body fusion (arrow). B, Quantification of missing/thin α-lobes or missing/over-extending β-lobes as observed in A (Drc2 alone: males n=20, females n=14; Dcr2+dNab2-RNAi: males n=15, females n=14). All flies contain elav-Gal4>UAS-Dcr2. C, qPCR analysis of dNab2 levels in the indicated genotypes (as in A). Data represents an average of three biological replicates of ~50 adult fly heads for each genotype. Precise excision control (dNab2pex41/dNab2pex41) flies and dNab2ex3 homozygous null flies (dNab2ex3/dNab2ex3) were used as positive and negative controls, respectively. Statistical significance values via two-tailed independent t-test are shown between indicated samples.
Pan-neuronal depletion of dNab2 led to an increase in MB defects relative to the control pan-neuronal expression of Dcr2 alone (elav-Gal4, UAS-dcr2, UAS-dNab2RNAi, vs. elav-Gal4,UAS-dcr2) (Figs. 4A,B), which resemble those seen in dNab2ex3 null brains. These defects include thinned or absent α and β-lobes, and variable degrees of β-lobe fusion (see magnified insets in Fig. 4A). In females, elav-Gal4, UAS-dcr2, UAS-dNab2RNAi β-lobe phenotypes vary from unaffected, to ‘mild’ (white arrow in bottom inset of Fig. 4A) or ‘severe’ fusion (Michel et al., 2004), while males show a mixture of complete fusion and an intermediate defect in which the β-lobes are very closely apposed over the brain midline (Fig. 4A). Overall, the percentage of elav-Gal4, UAS-dcr2, UAS-dNab2RNAi males and females showing fused β lobes, thinned α/β lobes, or completely missing lobes is 73% and 65%, respectively (Fig. 4B). The enhanced frequency of MB defects in dNab2-depleted males could be due to sex-specific differences in transgene expression (e.g. dosage compensation) and consistent with this hypothesis, approximately 20% of males overexpressing Dcr2 alone (elav-dcr2) show α/β lobe defects while no females show this effect (Fig. 4B). These data are consistent with enhanced transgene expression in males and further suggest that a Dcr2-dependent micro-RNA might play a role in MB development [e.g. let-7; (Wu et al., 2012)] that is independent of dNab2.
To more precisely characterize the spatial and temporal requirement for dNab2 within subsets of developing MB neurons, a UAS-dNab2 transgene was used in combination with Gal4 drivers to rescue the α/β-lobe axon defects in dNab2ex3 null brains. Representative examples of each genotype are shown in Figure 5. Re-expression of dNab2 in all neurons (elav-Gal4) (Fig. 5A, right panel) significantly rescued β-lobe fusion defects in a majority of dNab2ex3 null brains (Mann-Whitney test, p<0.05); the elav-Gal4 driver alone had no effect on β-lobe defects in dNab2ex3 null brains (Fig. 5A, left panel; penetrance ~75%, n=8). Importantly, dNab2 re-expression from the pan-MB driver OK107 (Aso et al., 2009) also significantly rescued the dNab2ex3 null β-lobe fusion defect; while 87.5% of dNab2ex3 homozygous null flies containing just the OK107-Gal4 driver alone (n=8) showed extreme, moderate, or slight fusion defects, only 25% of flies re-expressing dNab2 (n=16) in all mushroom body neurons (OK107>dNab2) showed some level of β-lobe fusion (Fig. 5B, right panel; Mann-Whitney test, p<0.05). Gal4 transgenes with enriched expression in either the adult γ-lobe (201Y) or the α’/β’-lobes (c305a) (Aso et al., 2009) were also used in conjunction with the UAS-dNab2 transgene. Neither of these genotypes conferred significant rescue of the dNab2 mutant MB structural defects as assessed by FasII staining (Mann-Whitney test, p>0.05), Fig. 5C,D). The c739-Gal4 driver, which is expressed mainly in the α/β-lobe neurons (Aso et al., 2009), produced an observable but not significant degree of rescue to the β-lobe fusion defect (Fig. 5E) when in combination with the UAS-dNab2 transgene (Mann-Whitney test, p=0.226, c739>dNab2 [n=18] vs. c739 alone [n=10]). In sum, these RNAi and Gal4 rescue experiments support a model in which the dNab2 RNA binding protein is required cell-autonomously within Kenyon cells to control the branching and projection of their axons into the α and β lobes.
dNab2 loss impairs short-term memory
dNab2 is the fly ortholog of a protein, ZC3H14, which is lost in an inherited form of human intellectual disability (Pak et al., 2011; Kelly et al., 2014). To extend our analysis of dNab2 function, we tested whether dNab2 is required for proper cognitive function in adult flies. To assess the pan-neuronal requirement for dNab2 in supporting cognitive behavior, a courtship-conditioning assay (Ejima and Griffith, 2011) was used to test the effect of pan-neuronal dNab2 loss on learning and short-term memory (Fig. 6). Briefly, in this assay individual adult male flies are placed in courtship chambers with single mated wildtype adult females for one hour. The amount of time each male engages in courtship activity during the first and last ten minutes of this “training session” is recorded and analyzed in order to derive a courtship index (CI). Previously mated females reject courtship advances of males, whose consequent decrease in CI between the first and last 10 minutes indicates learning. The durability of this CI suppression is indicative of memory.
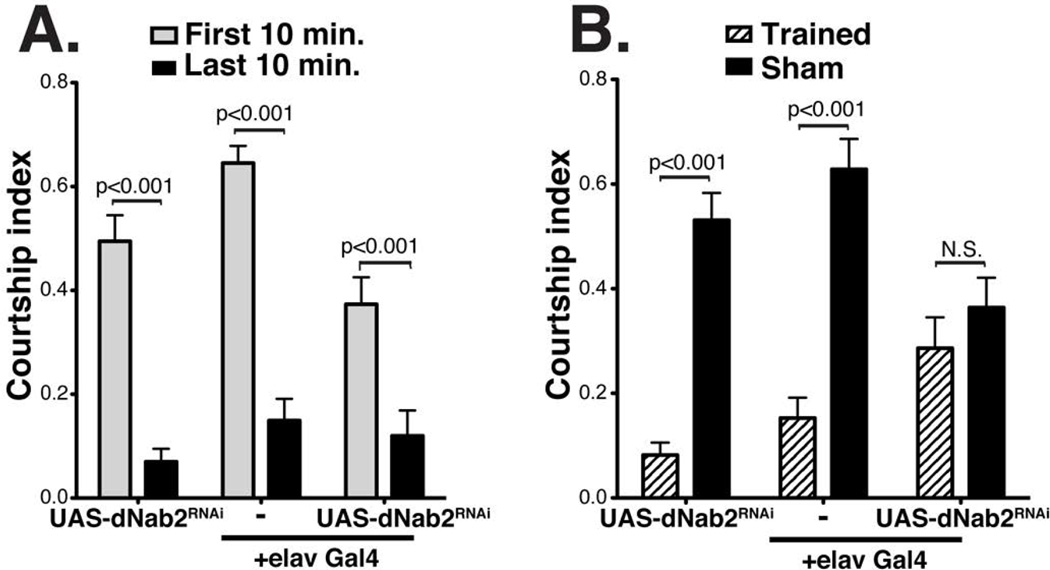
Figure 6. dNab2 is required for short-term memory.
Courtship indices among two-day old males of the indicated genotypes during the training/learning phase (A) or the retesting/memory phase (B) of the courtship conditioning assay. While Trained males carrying the Gal4 driver or RNAi cassette alone show robust CI suppression in the retesting phase, flies with dNab2 knockdown specifically in neurons (UAS-dNab2RNAi+neuronal-Gal4) showed no significant CI suppression upon retesting and were indistinguishable from mock treated controls. For each genotype, n=20. Statistical significance determined by two-tailed t-test.
Three separate genotypes were tested in the courtship conditioning assay: the pan-neuronal driver alone (elav-Gal4/Y), the dNab2 RNAi transgene alone (+/Y;UAS-dNab2RNAi/+), or combined driver and RNAi (elav-Gal4/Y;UAS-dNab2RNAi/+). We omitted the UAS-dcr2 transgene from these experiments due to its independent effects on dNab2 levels (see Fig. 4C). All three test groups show statistically significant suppression of CI across the training period (Fig. 6A), indicating that they are able to learn and adapt their behaviors to a continuous training regimen. Following the conditioning period, trained males (Trained) were transferred to a new courtship chamber containing a single virgin female to measure the durability of the learned CI suppression. Untrained flies (Sham) were included in this assay as negative controls. While Sham males display robust courtship of virgin females, control Trained males (elav-Gal4 or UAS-dNab2RNAi alone) maintained suppressed CI values, indicative of intact short-term memory. By contrast, the CI value of Trained males with pan-neuronal depletion of dNab2 (elav-Gal4>dNab2RNAi) was comparable to that of Sham trained flies (Fig. 6B). This difference is not due to a difference in motor activity as Nab2-depleted (elav-Gal4>dNab2RNAi) and control (elav-Gal4) adult males show similar frequencies of beam breaks per waking minute over the course of a 24-hour period in a Trikinetics Drosophila activity monitor (independent t-test, p=0.682; Fig. S2). These behavioral data are consistent with the hypothesis that the dNab2 polyadenosine RNA binding protein regulates pathways involved in short-term memory in addition to its role in axon branching and guidance.
Discussion
Here we present an analysis of the cellular and cognitive requirements for the Drosophila polyadenosine RNA binding protein dNab2, an ortholog of the ZC3H14 protein lost in an inherited intellectual disability (Pak et al., 2011), in brain development. The data show that dNab2 is required for proper development of the mushroom bodies, a brain region that supports learning and memory (Armstrong et al., 1998), and that this defect is due to a cell-autonomous role for dNab2 within subsets of mushroom body neurons. Furthermore, evidence also shows that pan-neuronal knockdown of dNab2 results in a defect in short-term memory, possibly paralleling cognitive defects produced by ZC3H14 loss in humans.
The analysis of dNab2 reveals a number of parallels to another RNA binding protein, dFmr1, which is also an ortholog of a protein lost in heritable intellectual disability, FMRP. As with ZC3H14, FMRP is a ubiquitously expressed protein whose loss leads to defects in brain function (reviewed in Santoro et al., 2011). Strikingly, dFmr1 mutant flies show adult MB defects very similar to those described here for dNab2 mutant flies, including thinned/missing α lobes and fused β lobes (Michel et al., 2004). Human and Drosophila FMRP/dFmr1 are well-established translational repressors (Laggerbauer et al., 2001; Zhang et al., 2001; Ishizuka et al., 2002; Chen et al., 2014), and while the precise molecular role of ZC3H14 and dNab2 have yet to be determined, the role of these proteins in limiting poly(A) tail length (Pak et al., 2011; Kelly et al., 2014) suggests that they could impact the fate of mRNAs in the cytoplasm, perhaps via effects upstream of translation. Finally, the dNab2 ortholog ZC3H14 is highly expressed in hippocampal neurons (Pak et al., 2011), which are also an important site of FMRP action (reviewed in Santoro et al., 2011). These similarities between dNab2/ZC3H14 and dFmr1/FMRP are suggestive of potential links between these RNA binding proteins that warrant further investigation.
Given its proposed molecular role as a Pab, dNab2 is likely to support neurodevelopment and memory via effects on the stability and/or translation of neuronal mRNAs. These roles could be linked such that defects in regulation of RNAs supporting axon projection lead to corresponding defects in memory circuits. Alternatively these phenotypes could reflect a requirement for dNab2 in regulating distinct pools of RNAs involved in each process. Our observations that neuronal RNAi-mediated depletion of dNab2 elicits penetrant effects on locomotor behavior (Pak et al., 2011) and short-term memory (this study), but comparatively mild effects on α/β-lobe structure (approximately 65% of brains affected), suggests these two phenotypes could stem from effects in different cells and perhaps different target RNAs. Indeed some proteins required for courtship memory act in γ-lobe neurons (Keleman et al., 2012) whose structure is unaffected by dNab2 loss, while other proteins are only required in the α/β-lobes (Ishimoto et al., 2013). Future studies will need to define dNab2 target RNAs in groups of brain neurons and assess their roles in axon projection and STM phenotypes that arise upon dNab2 loss.
The RNAs responsible for axonal defects in dNab2 mutant Kenyon cells whose projections the α/β MB lobes are as yet undefined. Although dNab2 is localized to the nucleus at steady-state, the budding yeast Nab2 protein shuttles between the cytoplasm and nucleus, presenting the possibility that dNab2 could impact RNA regulatory processes beyond nuclear processing. Studies have implicated a diverse set of molecules in MB development, including the cell-cell adhesion proteins N-cadherin (Zhu and Luo, 2004), Down-syndrome cell adhesion molecule (Dscam) (Wang et al., 2002; Wang et al., 2004; Zhan et al., 2004), and L1CAM (Goossens et al., 2011), as well as signaling cascades from Ephrin (Boyle et al., 2006) and Wingless/Wnt signals (Grillenzoni et al., 2007; Shimizu et al., 2011; Ng, 2012), providing a number of candidate pathways. Coordinated control of these signals during axon outgrowth, bifurcation, and synapse formation likely requires precise temporal and spatial control of mRNA stability, transport, and translation. The dNab2/ZC3H14 Pab restricts poly(A) tail length in vivo (Pak et al., 2011; Kelly et al., 2014). Thus, the required role in MB axon development could stem from effects on one or more transcript(s) involved in axonal projection and branching. Identifying these target RNAs will require functional assays that define dNab2-regulated transcripts in neurons and physical interaction screens that recover transcripts bound by dNab2. The identity of these transcripts will provide important clues as to how dNab2 influences cellular processes in the fly brain. However, equally important will be determining the fate of these RNAs once bound by dNab2, and testing whether dNab2 primarily influences neuronal gene expression by controlling the nuclear export, stability, transport, or translation of cytoplasmic RNAs, even if its role is primarily restricted to controlling poly(A) tail length in the nucleus. This combined analysis of dNab2 targets and how each is regulated by dNab2 will likely shed considerable light on the role of the dNab2/ZC3H14 protein family in brain development and function.
Supplementary Material
Acknowledgements
Financial support as follows - NIH 5K12 GM000680-12 (SMK), NIH F31 HD079226 (RB), and NIH MH107305 and GM058728 (AHC and KHM). We thank the Bloomington Drosophila Stock Center (Indiana, USA) and Developmental Studies Hybridoma Bank (Iowa, USA) for providing stocks and antibodies, and members of the Moberg, Kelly, and Corbett laboratories and S. Sanyal for helpful discussions and advice.
Footnotes
The authors declare no competing financial interests
References
- Armstrong JD, de Belle JS, Wang Z, Kaiser K. Metamorphosis of the mushroom bodies; large-scale rearrangements of the neural substrates for associative learning and memory in Drosophila. Learn Mem. 1998;5:102–114. [PMC free article] [PubMed] [Google Scholar]
- Aso Y, Grubel K, Busch S, Friedrich AB, Siwanowicz I, Tanimoto H. The mushroom body of adult Drosophila characterized by GAL4 drivers. J Neurogenet. 2009;23:156–172. doi: 10.1080/01677060802471718. [DOI] [PubMed] [Google Scholar]
- Banerjee A, Apponi LH, Pavlath GK, Corbett AH. PABPN1: molecular function and muscle disease. FEBS J. 2013;280:4230–4250. doi: 10.1111/febs.12294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boyle M, Nighorn A, Thomas JB. Drosophila Eph receptor guides specific axon branches of mushroom body neurons. Development. 2006;133:1845–1854. doi: 10.1242/dev.02353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brand AH, Perrimon N. Targeted gene expression as a means of altering cell fates and generating dominant phenotypes. Development. 1993;118:401–415. doi: 10.1242/dev.118.2.401. [DOI] [PubMed] [Google Scholar]
- Charlesworth A, Meijer HA, de Moor CH. Specificity factors in cytoplasmic polyadenylation. Wiley Interdiscip Rev RNA. 2013;4:437–461. doi: 10.1002/wrna.1171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen E, Sharma MR, Shi X, Agrawal RK, Joseph S. Fragile X mental retardation protein regulates translation by binding directly to the ribosome. Mol Cell. 2014;54:407–417. doi: 10.1016/j.molcel.2014.03.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cooper TA, Wan L, Dreyfuss G. RNA and disease. Cell. 2009;136:777–793. doi: 10.1016/j.cell.2009.02.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crittenden JR, Skoulakis EM, Han KA, Kalderon D, Davis RL. Tripartite mushroom body architecture revealed by antigenic markers. Learn Mem. 1998;5:38–51. [PMC free article] [PubMed] [Google Scholar]
- Ejima A, Griffith LC. Assay for courtship suppression in Drosophila. Cold Spring Harb Protoc. 2011 doi: 10.1101/pdb.prot5575. 2011:pdb prot5575. [DOI] [PubMed] [Google Scholar]
- Fushima K, Tsujimura H. Precise control of fasciclin II expression is required for adult mushroom body development in Drosophila. Dev Growth Differ. 2007;49:215–227. doi: 10.1111/j.1440-169X.2007.00922.x. [DOI] [PubMed] [Google Scholar]
- Goossens T, Kang YY, Wuytens G, Zimmermann P, Callaerts-Vegh Z, Pollarolo G, Islam R, Hortsch M, Callaerts P. The Drosophila L1CAM homolog Neuroglian signals through distinct pathways to control different aspects of mushroom body axon development. Development. 2011;138:1595–1605. doi: 10.1242/dev.052787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goss DJ, Kleiman FE. Poly(A) binding proteins: are they all created equal? Wiley Interdiscip Rev RNA. 2013;4:167–179. doi: 10.1002/wrna.1151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grenningloh G, Rehm EJ, Goodman CS. Genetic analysis of growth cone guidance in Drosophila: fasciclin II functions as a neuronal recognition molecule. Cell. 1991;67:45–57. doi: 10.1016/0092-8674(91)90571-f. [DOI] [PubMed] [Google Scholar]
- Grillenzoni N, Flandre A, Lasbleiz C, Dura JM. Respective roles of the DRL receptor and its ligand WNT5 in Drosophila mushroom body development. Development. 2007;134:3089–3097. doi: 10.1242/dev.02876. [DOI] [PubMed] [Google Scholar]
- Guthrie CR, Greenup L, Leverenz JB, Kraemer BC. MSUT2 is a determinant of susceptibility to tau neurotoxicity. Hum Mol Genet. 2011;20:1989–1999. doi: 10.1093/hmg/ddr079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heisenberg M. Mushroom body memoir: from maps to models. Nat Rev Neurosci. 2003;4:266–275. doi: 10.1038/nrn1074. [DOI] [PubMed] [Google Scholar]
- Ishimoto H, Wang Z, Rao Y, Wu CF, Kitamoto T. A novel role for ecdysone in Drosophila conditioned behavior: linking GPCR-mediated non-canonical steroid action to cAMP signaling in the adult brain. PLoS Genet. 2013;9:e1003843. doi: 10.1371/journal.pgen.1003843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ishizuka A, Siomi MC, Siomi H. A Drosophila fragile X protein interacts with components of RNAi and ribosomal proteins. Genes Dev. 2002;16:2497–2508. doi: 10.1101/gad.1022002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keleman K, Vrontou E, Kruttner S, Yu JY, Kurtovic-Kozaric A, Dickson BJ. Dopamine neurons modulate pheromone responses in Drosophila courtship learning. Nature. 2012;489:145–149. doi: 10.1038/nature11345. [DOI] [PubMed] [Google Scholar]
- Kelly S, Pak C, Garshasbi M, Kuss A, Corbett AH, Moberg K. New kid on the ID block: Neural functions of the Nab2/ZC3H14 class of Cys3His tandem zinc-finger polyadenosine RNA binding proteins. RNA Biol. 2012;9 doi: 10.4161/rna.20187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kelly SM, Leung SW, Pak C, Banerjee A, Moberg KH, Corbett AH. A conserved role for the zinc finger polyadenosine RNA binding protein, ZC3H14, in control of poly(A) tail length. RNA. 2014;20:681–688. doi: 10.1261/rna.043984.113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kelly SM, Pabit SA, Kitchen CM, Guo P, Marfatia KA, Murphy TJ, Corbett AH, Berland KM. Recognition of polyadenosine RNA by zinc finger proteins. Proc Natl Acad Sci U S A. 2007;104:12306–12311. doi: 10.1073/pnas.0701244104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laggerbauer B, Ostareck D, Keidel EM, Ostareck-Lederer A, Fischer U. Evidence that fragile X mental retardation protein is a negative regulator of translation. Hum Mol Genet. 2001;10:329–338. doi: 10.1093/hmg/10.4.329. [DOI] [PubMed] [Google Scholar]
- Lee T, Lee A, Luo L. Development of the Drosophila mushroom bodies: sequential generation of three distinct types of neurons from a neuroblast. Development. 1999;126:4065–4076. doi: 10.1242/dev.126.18.4065. [DOI] [PubMed] [Google Scholar]
- Lee T, Luo L. Mosaic analysis with a repressible cell marker for studies of gene function in neuronal morphogenesis. Neuron. 1999;22:451–461. doi: 10.1016/s0896-6273(00)80701-1. [DOI] [PubMed] [Google Scholar]
- Lee T, Luo L. Mosaic analysis with a repressible cell marker (MARCM) for Drosophila neural development. Trends Neurosci. 2001;24:251–254. doi: 10.1016/s0166-2236(00)01791-4. [DOI] [PubMed] [Google Scholar]
- Leung SW, Apponi LH, Cornejo OE, Kitchen CM, Valentini SR, Pavlath GK, Dunham CM, Corbett AH. Splice variants of the human ZC3H14 gene generate multiple isoforms of a zinc finger polyadenosine RNA binding protein. Gene. 2009;439:71–78. doi: 10.1016/j.gene.2009.02.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McKee AE, Silver PA. Systems perspectives on mRNA processing. Cell Res. 2007;17:581–590. doi: 10.1038/cr.2007.54. [DOI] [PubMed] [Google Scholar]
- Michel CI, Kraft R, Restifo LL. Defective neuronal development in the mushroom bodies of Drosophila fragile X mental retardation 1 mutants. J Neurosci. 2004;24:5798–5809. doi: 10.1523/JNEUROSCI.1102-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ng J. Wnt/PCP proteins regulate stereotyped axon branch extension in Drosophila. Development. 2012;139:165–177. doi: 10.1242/dev.068668. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pak C, Garshasbi M, Kahrizi K, Gross C, Apponi LH, Noto JJ, Kelly SM, Leung SW, Tzschach A, Behjati F, Abedini SS, Mohseni M, Jensen LR, Hu H, Huang B, Stahley SN, Liu G, Williams KR, Burdick S, Feng Y, Sanyal S, Bassell GJ, Ropers HH, Najmabadi H, Corbett AH, Moberg KH, Kuss AW. Mutation of the conserved polyadenosine RNA binding protein, ZC3H14/dNab2, impairs neural function in Drosophila and humans. Proc Natl Acad Sci U S A. 2011;108:12390–12395. doi: 10.1073/pnas.1107103108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ropers HH. Genetics of early onset cognitive impairment. Annu Rev Genomics Hum Genet. 2010;11:161–187. doi: 10.1146/annurev-genom-082509-141640. [DOI] [PubMed] [Google Scholar]
- Santoro MR, Bray SM, Warren ST. Molecular Mechanisms of Fragile X Syndrome: A Twenty-Year Perspective. Annu Rev Pathol. 2011 doi: 10.1146/annurev-pathol-011811-132457. [DOI] [PubMed] [Google Scholar]
- Shimizu K, Sato M, Tabata T. The Wnt5/planar cell polarity pathway regulates axonal development of the Drosophila mushroom body neuron. J Neurosci. 2011;31:4944–4954. doi: 10.1523/JNEUROSCI.0154-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shin JE, DiAntonio A. Highwire regulates guidance of sister axons in the Drosophila mushroom body. J Neurosci. 2011;31:17689–17700. doi: 10.1523/JNEUROSCI.3902-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siegel RW, Hall JC. Conditioned responses in courtship behavior of normal and mutant Drosophila. Proc Natl Acad Sci U S A. 1979;76:3430–3434. doi: 10.1073/pnas.76.7.3430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang J, Ma X, Yang JS, Zheng X, Zugates CT, Lee CH, Lee T. Transmembrane/juxtamembrane domain-dependent Dscam distribution and function during mushroom body neuronal morphogenesis. Neuron. 2004;43:663–672. doi: 10.1016/j.neuron.2004.06.033. [DOI] [PubMed] [Google Scholar]
- Wang J, Zugates CT, Liang IH, Lee CH, Lee T. Drosophila Dscam is required for divergent segregation of sister branches and suppresses ectopic bifurcation of axons. Neuron. 2002;33:559–571. doi: 10.1016/s0896-6273(02)00570-6. [DOI] [PubMed] [Google Scholar]
- Wigington CP, Williams KR, Meers MP, Bassell GJ, Corbett AH. Poly(A) RNA-binding proteins and polyadenosine RNA: new members and novel functions. Wiley Interdiscip Rev RNA. 2014 doi: 10.1002/wrna.1233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu JS, Luo L. A protocol for dissecting Drosophila melanogaster brains for live imaging or immunostaining. Nat Protoc. 2006;1:2110–2115. doi: 10.1038/nprot.2006.336. [DOI] [PubMed] [Google Scholar]
- Wu YC, Chen CH, Mercer A, Sokol NS. Let-7-complex microRNAs regulate the temporal identity of Drosophila mushroom body neurons via chinmo. Dev Cell. 2012;23:202–209. doi: 10.1016/j.devcel.2012.05.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu HH, Chen CH, Shi L, Huang Y, Lee T. Twin-spot MARCM to reveal the developmental origin and identity of neurons. Nat Neurosci. 2009;12:947–953. doi: 10.1038/nn.2345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhan XL, Clemens JC, Neves G, Hattori D, Flanagan JJ, Hummel T, Vasconcelos ML, Chess A, Zipursky SL. Analysis of Dscam diversity in regulating axon guidance in Drosophila mushroom bodies. Neuron. 2004;43:673–686. doi: 10.1016/j.neuron.2004.07.020. [DOI] [PubMed] [Google Scholar]
- Zhang YQ, Bailey AM, Matthies HJ, Renden RB, Smith MA, Speese SD, Rubin GM, Broadie K. Drosophila fragile X-related gene regulates the MAP1B homolog Futsch to control synaptic structure and function. Cell. 2001;107:591–603. doi: 10.1016/s0092-8674(01)00589-x. [DOI] [PubMed] [Google Scholar]
- Zheng X, Zugates CT, Lu Z, Shi L, Bai JM, Lee T. Baboon/dSmad2 TGF-beta signaling is required during late larval stage for development of adult-specific neurons. EMBO J. 2006;25:615–627. doi: 10.1038/sj.emboj.7600962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu H, Luo L. Diverse functions of N-cadherin in dendritic and axonal terminal arborization of olfactory projection neurons. Neuron. 2004;42:63–75. doi: 10.1016/s0896-6273(04)00142-4. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.