Abstract
IMPORTANCE
Septic shock is a clinical emergency that occurs in more than 230 000 US patients each year.
OBSERVATIONS AND ADVANCES
In the setting of suspected or documented infection, septic shock is typically defined in a clinical setting by low systolic (≤90 mm Hg) or mean arterial blood pressure (≤65 mm Hg) accompanied by signs of hypoperfusion (eg, oliguria, hyperlactemia, poor peripheral perfusion, or altered mental status). Focused ultrasonography is recommended for the prompt recognition of complicating physiology (eg, hypovolemia or cardiogenic shock), while invasive hemodynamic monitoring is recommended only for select patients. In septic shock, 3 randomized clinical trials demonstrate that protocolized care offers little advantage compared with management without a protocol. Hydroxyethyl starch is no longer recommended, and debate continues about the role of various crystalloid solutions and albumin.
CONCLUSIONS AND RELEVANCE
The prompt diagnosis of septic shock begins with obtainment of medical history and performance of a physical examination for signs and symptoms of infection and may require focused ultrasonography to recognize more complex physiologic manifestations of shock. Clinicians should understand the importance of prompt administration of intravenous fluids and vasoactive medications aimed at restoring adequate circulation, and the limitations of protocol-based therapy, as guided by recent evidence.
Shock is life-threatening circulatory failure with inadequate tissue perfusion.1 The typical presentation is hypotension (low systolic ≤90 mm Hg) or mean arterial blood pressure (≤65 mm Hg) accompanied by clinical signs of hypoperfusion. Historically, shock was attributed to a neurologic response to injury, vasomotor changes to the circulation, or a problem of missing blood.2 By the mid-20th century, Blalock and Weil organized shock into distinct constructs: cardiogenic, obstructive, hypovolemic, or vasogenic.3,4 Although these categories are valuable teaching concepts, the diagnosis of shock is far more complex. We focus this review on septic shock, which is the most common cause of noncardiogenic shock and has several of the Blalock and Weil physiologic constructs at the same time.5 Septic shock occurs in more than 230 000 US patients each year, with more than 40 000 US deaths annually. A recent Burden of Diseases article found that primary risk factors for septic shock (ie, infection) is the fifth leading cause of years of productive life lost due to premature mortality.6 Given the public health burden, we review advances in diagnosis, treatment, and areas of uncertainty in septic shock from January 2010 to June 2015.
Methods
We performed a review of the MEDLINE and the Cochrane Database of Systematic Reviews from 2010 to 2015 using specific search strategies. Our primary search used the terms shock, septic shock, diagnosis, and treatment, among others. We provide search strings and Preferred Reporting Items for Systematic Reviews and Meta-Analyses diagram in eAppendix (in the Supplement). We restricted articles to adult (age ≥18 years) human data reported in the English language only. We screened articles published between January 1, 2010, and June 1, 2015, and excluded opinion articles, commentaries, case series, and cohort studies—focusing on randomized clinical trials (RCTs), meta-analyses, systematic reviews, and clinical practice guidelines. After screening 8329 titles and abstracts, more articles were identified for full-text review, after which manual review of bibliographies generated additional references. A total of 181 articles were manually reviewed, of which 35 were selected with relevant content (eFigure in the Supplement). We selected only articles deemed to provide major advances in the diagnosis or treatment of septic shock. We considered sources of bias in these articles and defined areas of uncertainty as those in which the evidence conflicted. We used the American Heart Association classification of recommendations to grade the quality of evidence (grade A, data from many large RCTs; grade B, data from fewer, smaller RCTs, careful analyses of nonrandomized studies, or observational registries; and grade C, expert consensus).
Results
Major Diagnostic Advances
A conceptual framework for the diagnosis of shock has multiple domains including an initial evaluation of the etiology and clinical features, assessment of the primary hemodynamic manifestations, and consideration of alterations in cellular biology and the degree of local tissue injury. Major advances and areas of uncertainty with in these domains (Table 1) will be discussed.
Table 1.
Major Advances in the Diagnosis and Treatment of Septic Shock
| Action | Caveat |
|---|---|
| Diagnostic | |
| Clinical diagnosis is the criterion standard: Typically, systolic blood pressure ≤90 mm Hg or mean arterial blood pressure ≤65 mm Hg or >40-mm Hg decrease from baseline; Poor peripheral perfusion, low urinary output, altered mentation, elevated lactate |
Normotensive shock with isolated hyperlactemia needs clarity; Lactate and systolic blood pressure thresholds are uncertain; Biologic phenotyping may be promising but not yet feasible in real time or tested in randomized clinical trials |
| Pulmonary artery catheterization and continuous monitoring of central venous oxygen saturation not recommended for routine diagnosis | May have a role in right ventricular dysfunction, complex cases with diagnostic uncertainty |
| Focused ultrasonography is suggested if there is concern for overlapping hemodynamic manifestations of shock | Practical, easy to use, and recommended by expert consensus7 |
| Arterial pulse contour analysis | Awaiting randomized clinical trials for patient outcomes; Requires controlled mechanical ventilation and sinus rhythm |
| Treatment | |
| Prompt fluid bolus is recommended (500–1000 mL) with appropriate safety limits | Fluid therapy with balanced crystalloids vs albumin is suggested based on meta-analyses,8,9 while specific fluid comparisons undergo additional randomized clinical trials |
| Norepinephrine is recommended as a first-line vasopressor | Vasopressin may spare norepinephrine at higher doses |
| Hydroxylethyl starch may cause harm | Increases mortality and worsens renal outcomes among survivors |
| Protocolized early goal-directed therapy is not superior to usual care in early septic shock | Tested among patients with prompt shock recognition, intravenous fluid boluses, and early antibiotics |
| Low-dose corticosteroids to be considered for vasopressor-dependent shock | Dosing regimen and timing of discontinuation remains controversial |
Initial Evaluation
At the bedside, a clinician begins by asking, “Is this patient in shock?” Consensus guidelines for septic shock agree on core diagnostic elements including suspected or documented infection accompanied by arterial hypotension and evidence of tissue hypoperfusion (eg, oliguria, altered mental status, poor peripheral perfusion, or hyperlactemia).10,11 Yet the requirement for adequate fluid resuscitation, absence of vasopressors, or thresholds for blood pressure vary across shock definitions. In fact, a recent European Society of Intensive Care Medicine (ESICM) consensus statement suggests shock may present in the absence of hypotension.12 There is no reference standard for the bedside diagnosis of shock. Rather, observational studies report how mortality varies across combinations of shock features from 29% to 46%.13
If shock is present, the clinician must determine the inciting cause by asking, “What just happened?” Such clinical risk factors will guide immediate intervention. And although severe infection may be evident, it is often more difficult to recognize. We found no changes to the typical clinical approach to the diagnosis of infection in septic shock during our review. However, many bio-markers and blood culture–independent, molecular diagnostics are undergoing study to help discriminate sterile inflammation from infection.14
The primary physiologic manifestations of shock should be assessed, although they are unlikely to fit simply into the Blalock and Weil framework. For example, patients with septic shock will develop myocardial depression in as many as 30% of cases.5 A prompt assessment of the relevant mechanisms driving the shock state is imperative because patient delay prior to care and immediate therapy will complicate the evolving presentation.
Hemodynamic Monitoring for the Diagnosis of Septic Shock
Hemodynamic monitoring devices may clarify the primary physiologic manifestations in septic shock. The clinical usefulness of these monitoring devices can result from the device, the algorithm linked to the device, or the static/dynamic target of the algorithm (eTable 1 in the Supplement). As such, there is a lack of consensus and considerable debate about the role of these devices.
Invasive Hemodynamic Monitoring
Decades ago, the standard care of shock patients included invasive devices like the pulmonary artery catheter (PAC) or continuous central venous oxygen saturation (ScvO2) catheterization. The PAC can estimate cardiac output and measure mixed venous oxygen saturation, among other parameters, to refine the etiology of shock and potentially affect patient outcomes. A 2013 Cochrane review of 2923 general intensive care unit (ICU) patients (proportion in shock not reported) found no difference in mortality comparing PAC vs no PAC management.15 A secondary analysis of the Fluid and Catheter Treatment Trial of 774 patients with acute respiratory distress syndrome, among whom 40% were in shock, confirmed that PAC increases hospital costs with no change in mortality.16 The continuous ScvO2 catheter is an alternative to the PAC but had no advantage over lactate clearance when included in a recent RCT testing resuscitation of septic shock (Table 2).17 Consensus recommends against the placement of PAC in routine management of shock and suggests its use only in the minority of cases with right ventricular dysfunction or severe acute respiratory distress syndrome.12 Meanwhile, the United States has largely reduced PAC use over the past 15 years.22
Table 2.
Major Diagnostic Advances in Septic Shock in Selected Trials
| Source by Category | Diagnostic Management | Type of Evidence | No. of Studies | No. of Patients (% With Shock) | Setting | Conclusion | Grade Evidencea |
|---|---|---|---|---|---|---|---|
| Invasive Hemodynamic Device | |||||||
| Rajaram et al,15 2013 | PAC vs no PAC | Systematic review/meta-analysis | 5 | 2923 (NA) | ICU | No change in mortality for PAC | B |
| Clermont et al,16 2011 | PAC vs central venous catheter | RCT | 1 | 335 (39) | ICU | Greater hospital costs with no change in mortality for PAC | B |
| Jones et al,17 2010 | ScvO2 catheter vs lactate clearance | RCT | 1 | 300 (100) | Emergency department/ICU | Equivalent hospital mortality rate for ScvO2 vs lactate | B |
| Noninvasive Hemodynamic Device | |||||||
| Labovitz et al,7 2010 | Bedside ultrasonography | Guideline | Recommended in initial assessment for all undifferentiated shock | C | |||
| Zhang et al,18 2015 | Noninvasive vs invasive device | RCT | 1 | 350 (47) | ICU | No change in 28-d mortality with management by noninvasive device | B |
| Richard et al,19 2015 | Noninvasive vs invasive device | RCT | 1 | 60 (100) | ICU | No change in time to shock resolution from noninvasive device | B |
| Biomarkers of Local Tissue Injury | |||||||
| Jansen et al,20 2010 | Lactate-guided therapy every 2 hours for 8 hours vs lactate at admission only | RCT | 1 | 348 (19) | ICU | No change in unadjusted hospital mortality but reduced ICU length of stay using lactate every 2 hours | B |
| Dellinger et al,21 2013 | Lactate as target for resuscitation | Guideline | Weak recommendation based on low-quality evidence (grade 2C) | C | |||
Abbreviations: ICU, intensive care unit; NA, not available; PAC, pulmonary artery catheter; RCT, randomized clinical trial; ScvO2, continuous central venous oxygen saturation catheter.
Grade of evidence was assessed using the American Heart Association classification of recommendations. Grade A indicates data from many large RCTs; grade B, data from fewer, smaller RCTs, careful analyses of nonrandomized studies, or observational registries; and grade C, expert consensus.
Noninvasive Hemodynamic Monitoring
The physiology underlying shock can be further clarified using minimally or noninvasive techniques such as arterial pulse contour analysis or focused echocardiography. Calibrated pulse contour analysis devices provide continuous estimations of cardiac output, beat-to-beat stroke volume, and pulse pressure variation, among other parameters. In one trial, 388 hemodynamically unstable patients in 3 ICUs were randomized to a minimally invasive hemodynamic monitoring device for 24 hours vs usual care.23 With no protocol linked to the device, the intervention groups did not differ in resolution of hemodynamic instability at 6 hours or mortality. Two small, randomized trials also found no difference in 28-day mortality and time to shock resolution comparing pulse contour analysis–guided management vs other strategies.18,19 Ongoing studies24 are testing non-invasive estimates of stroke volume variation linked to fluid resus-citation protocols in septic shock. A recent systematic review did find benefit of hemodynamic optimization by pulse contour analysis in patients undergoing high-risk surgery.25 The application of pulse contour analysis in shock patients outside the operating theater is practically limited by the requirement for controlled mechanical ventilation, adequate arterial pressure waveform, and the absence of arrhythmias.
Focused ultrasonograpy can help discern central hemodynamics and the etiology of shock in undifferentiated patients.26 It can reveal right and left cardiac chamber size and contractility, pericardial fluid, and inferior vena cava size and collapsibility suggestive of hypovolemia, among other features. At the time of this publication, our search revealed no rigorous RCTs of focused cardiac ultrasonography affecting patient-centered outcomes in septic shock. Yet, recent guidelines and consensus statements recommend focused ultrasonography as best clinical practice in the initial assessment of hemodynamically unstable patients with septic shock (Table 2).7,12
Markers of Tissue Injury
Systemic markers of local tissue injury can suggest that organs are under stress in shock, including blood lactate level, base deficit, tissue oxygen saturation by near-infrared spectroscopy, or various microcirculatory changes. These tests may refine a clinical diagnosis but also serve as targets during optimization and stabilization of shock (Table 2).26 Lactate levels are not currently included in the 2001 ESICM/SCCM (Society of Critical Care Medicine) consensus definition of septic shock, but suggested in the 2014 ESICM consensus panel on circulatory shock.10,12 Serial lactate measurements are none-the less widely used in practice,27 but the specific threshold for diagnosing shock and its role in monitoring remains unknown. One open-label randomized clinical trial in 4 ICUs tested a protocol targeting a 20% reduction in lactate every 2 hours on top of recommended resuscitation guidelines. They found a significant reduction in only a secondary outcome (ICU length of stay), but included few patients in shock (19%).20 The use of near-infrared spectroscopy or tissue oxygen saturation to either diagnosis or manage septicshockstateshasnotbeenevaluatedinclinicaltrialsofpatientcentered outcomes during our review period.
Areas of Uncertainty
From a biologic perspective, no definition or cut point for shock is perfect, and guidelines, quality improvement, and trial enrollment deserve a uniform definition that balances sensitivity and specificity (Box). Not all patients with shock have a classic presentation, and cases on the margin may be as important as those that are clinically overt. For example, patients with normal arterial pressure and hyperlactemia may have similar outcomes to overt shock,28 but hyperlactermia could be either hypoxia-induced microcirculatory hypoperfusion, high glycolytic flux from an inflammatory response, or impaired clearance.28,29 The host response to shock is also complex, with both proinflammatory and antiinflammatory reactions at the local and systemic level.30 Cases could be further identified using biologic phenotypes, although none are widely accepted. Some candidate approaches include immunophenotyping, genome-wide expression mosaics, or clinico-metabolomic profiles.31–33 Third, a consensus definition for shock is needed across different phases of care (eg, from prehospital to emergency department to ICU). Major trials in each setting use different criteria for lactate and shock,34,35 leading to uncertainty about optimal treatment.
Box. Areas of Uncertainty in the Diagnosis and Treatment of Septic Shock.
Diagnostic
No consensus definition for shock across locations of care
Thresholds for systolic blood pressure, lactate, and adequacy of fluid resuscitation lack consensus during prehospital care and in the emergency department and intensive care unit; this may hinder epidemiology, trial enrollment, and quality improvement efforts
Inconsistent definition for cryptic shock
Isolated hyperlactemia with normal systolic blood pressure may reflect tissue hypoperfusion, and little is known about the epidemiology and outcomes of cryptic shock; may also be referred to as occult or normotensive shock
Biologic phenotypes of shock
Novel biologic phenotypes are proposed using genetic, molecular, and metabolomic markers, but these lack validation and testing in clinical trials
Treatment
Choice and timing of fluid administration
Both albumin and balanced crystalloid solutions may be superior in meta-analyses, but direct comparisons in randomized clinical trials of early shock are needed
Targets for hemodynamic optimization
The ideal resuscitation target (static vs dynamic; microcirculation vs regional vs peripheral) is an urgent knowledge gap that may be different for different phases of resuscitation in shock
De-escalation and removal of fluid
The optimal method and timing of fluid removal after shock resuscitation requires further study, with options including diuretics, ultrafiltration
Major Therapeutic Advances
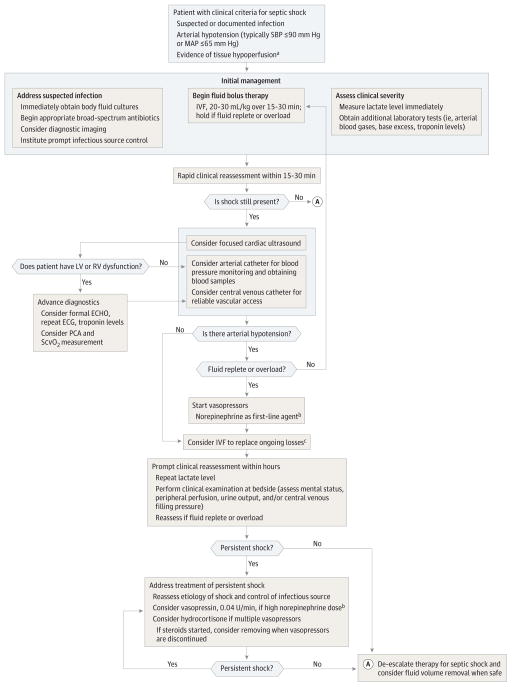
Many factors contribute to a steadily improving case-fatality rate in shock,36 including early recognition and prompt intervention. A sample treatment algorithm for septic shock typically proceeds through rescue, optimization, stabilization, and de-escalation of care (Figure).26 Although rescue steps may differ depending specifically on the inciting cause, adult patients with septic shock typically receive immediate intravenous access, fluid administration, vasopressors, and care directed at restoring adequate circulation. We briefly review major advances in these topics (Table 1).
Figure.
Proposed Algorithm for Treatment of Septic Shock
ECG indicates electrocardiogram; ECHO, echocardiogram; IVF, intravenous fluids; LV/RV, left ventricular/right ventricular; MAP, mean arterial pressure; PCA pulse contour analysis; SBP, systolic blood pressure; ScvO2, continuous central venous oxygen saturation.
aTissue hypoperfusion typically manifests as altered mentation, low urinary output, poor peripheral perfusion, and/or hyperlactemia (≥2.0 mmol/L).
bNorepinephrine may not always be the first choice in setting of tachycarrythmias or atrial fibrillation; consider adding vasopressin for norepinephrine rates that exceed 15 3g/kg/min.
cThe choice for fluid repletion and type will be refined by ongoing safety checks for pulmonary edema/fluid overload, metabolic derangements from unbalanced crystalloids, and ongoing losses.
Crystalloids
There are many choices for crystalloids in septic shock.37 Although there is variability across crystalloid solutions in tonicity and inorganic/organic anions, such that few entirely resemble plasma,38 normal saline is widely used in the United States.37 A sequential-period observational trial tested chloride-liberal vs chloride-restrictive fluids for all fluid therapy in a single ICU over 18 months, in which 10% of patients were in shock. They found an increase in injury and failure class (RIFLE-defined [Risk, Injury, Failure, Loss of kidney function, End-stage kidney disease]) acute kidney injury during the chloride-rich period.39 Others leveraged the indirect comparisons between 6 different fluids in 14 distinct RCTs in a network meta-analysis of 18 916 patients with sepsis. They report that balanced crystalloids were somewhat superior to normal saline (odds ratio [OR] = 0.78 [95% CI, 0.58–1.05]), although with low-moderate confidence and without reporting among the subset with septic shock.8 The same authors report no difference in rate of renal replacement therapy for this comparison in a separate study.9 Given the persistent equipoise, the SPLIT (Saline vs Plasma-Lyte 148 for Intensive Care Fluid Therapy) study is testing balanced crystalloids vs 0.9% normal saline.40,41
Colloids
Colloid solutions, such as albumin, dextran, gelatins, or hydroxy-ethyl starch, are the most widely used fluids in critically ill patients, although with variability across ICUs and countries.42 Clinicians’ choice among colloids is influenced by availability, cost, and desire to minimize interstitial edema. Many believe a greater intravascular volume is achieved from colloids in shock,38 but the effects are modified by their molecular weight and concentration, and endothelial changes during inflammation.43 The ALBIOS (Albumin Italian Outcomes Study) trial randomized nearly 1800 patients in 100 ICUs with severe sepsis to albumin with crystalloids vs crystalloids alone and found no difference in 28-day mortality.44 A post hoc analysis restricted to patients with septic shock suggested a 28-day mortality benefit from albumin (relative risk [RR], 0.87 [95% CI, 0.77–0.99]), without affecting safety. The CRISTAL (Colloids vs Crystalloids for the Resuscitation of the Critically Ill) trial compared crystalloids with colloids in 2857 adults in shock in 57 ICUs, finding no difference in 28-day mortality or renal outcomes.45 These studies build on data from the 6S (Scandinavian Starch for Severe Sepsis/Septic Shock) and CHEST (Crystalloid vs Hydroxyethyl Starch) trials in severe sepsis,46,47 which together randomized more than 7000 patients to reduced concentration 6% hydroxyethyl starch 130/0.4 vs crystalloids, and found no mortality benefit at 90 days to 1 year,48 but observed increases in the rate of renal replacement therapy. These studies outline no clear benefit (or harm) from albumin in septic shock, and continue to support harm from low concentration hydroxyethyl starch solutions (Table 3).
Table 3.
Major Therapeutic Advances in Septic Shock in Selected Randomized Clinical Trials
| Source by Category |
Setting (Study Duration) |
No. of Patients (% in Septic Shock) |
Intervention | Control | Primary Outcome |
Relative Risk (95% CI) for Primary Outcome |
Conclusions | Grade of Evidencea |
|---|---|---|---|---|---|---|---|---|
| Fluidsb | ||||||||
| Caironi et al,44 2014 | 100 Mixed ICUs (2008–2012) | 1810 (63) | 20% Albumin and crystalloids | Crystalloids alone | 28-d Mortality | 1.00 (0.87–1.14) | No difference in 28-d or 90-d mortality | B |
| Perner et al,47 2012 | 26 Mixed ICUs (2009–2011) | 798 (84) | Hydroxylethyl starch 130/0.42 | Ringer acetate | 6-mo Mortality | 1.12 (0.98–1.29) | No difference in 6-mo or 1-y mortality | Ac |
| Annane et al,45 2013 | 57 Mixed ICUs (2003–2012) | 2857 (54) | Gelatins, dextrans, hydroxylethyl starch, or 4% or 20% albumin | Isotonic, hypertonic saline, Ringer lactate | 28-d Mortality | 0.96 (0.88–1.04) | No difference in 28-d mortality | Ac |
| Myburgh et al,46 2012 | 23 Mixed ICUs (2009–2012) | 7000 (13)d | 6% Hydroxylethyl starch 130/0.4 in 0.9% sodium chloride | 0.9% Sodium chloride | 90-d Mortality | 1.06 (0.96–1.18) | No difference in 90-d mortality; increased risk of renal replacement therapy with hydroxylethyl starch | Ac |
| Perner et al,47 2012 | 26 Mixed ICUs (2009–2011) | 798 (84) | Hydroxylethyl starch 130/0.42 | Ringer acetate | 90-d Mortality | 1.17 (1.01–1.36) | Greater 90-d mortality and renal replacement therapy with hydroxylethyl starch | Ac |
| Vasopressors | ||||||||
| De Backer et al,49 2010 | 8 Mixed ICUs (2003–2007) | 1679 (62) | Dopamine | Norepinephrine | 28-d Mortality | 1.17 (0.97–1.42) | No difference in mortality but more adverse events and arrhythmias with dopamine | B |
| Protocols | ||||||||
| Yealy et al,35 2014 | 31 Emergency departments (2003–2007) | 1341 (100) | EGDT vs protocolized standard care | Usual care | 60-d In-hospital mortality | 1.04 (0.82–1.31) | No difference in 28-d, 90-d, or 1-y mortality for protocol-based vs usual care or in post hoc subgroups | A |
| Mouncey et al,50 2015 | 56 Centers (2011–2014) | 1260 (100) | EGDT | Usual care | 90-d Mortality | 1.01 (0.85–1.20) | No difference in 90-d mortality, greater cost with EGDT | A |
| Peake et al,51 2014 | 51 Centers (2008–2014) | 1600 (100) | EGDT | Usual care | 90-d Mortality | 0.98 (0.80–1.21) | No difference in 90-d mortality for EGDT vs usual care or any a priori subgroup | A |
| Jones et al,17 2010 | 3 Emergency departments (2007–2009) | 300 (82) | Lactate-guided EGDT | ScvO2 catheter-guided EGDT | In-hospital mortality | Noninferiority of using lactate-guided EGDT vs ScvO2 catheter | B | |
| Asfar et al,52 2014 | 29 Centers (2007–2009) | 776 (100) | High mean arterial pressure target (80–85 mm Hg) | Low mean arterial pressure target (65–70 mm Hg) | 28-d Mortality | 1.07 (0.84–1.38) | No difference in 28-d mortality using a lower mean arterial pressure target | B |
| Andrews et al,53 2014 | 1 Center (2012) | 112 (NR) | Modified EGDT | Usual care | In-hospital mortality | 1.06 (0.79–1.41) | Trial stopped due to increased hypoxemia in intervention group | B |
| Holst et al,54 2014 | 32 ICUs (2011–2013) | 1005 (100) | Hemoglobin threshold 7.0 g/dL | Hemoglobin threshold 9.0 g/dL | 90-d Mortality | 0.94 (0.78–1.09) | No difference in 90-d mortality or secondary outcomes with lower hemoglobin threshold | B |
Abbreviations: EGDT, early, goal-directed therapy; ICU, intensive care unit; NR, not reported; RCT, randomized clinical trial; ScvO2, central venous oxygen saturation.
Grade of evidence assessed using the American Heart Association classification of recommendations. Grade A, data from many large RCTs; grade B, data from fewer, smaller RCTs, careful analyses of nonrandomized studies, or observational registries; and grade C, expert consensus.
Excluded Siegemund et al55 (BASES [Basel Starch Evaluation in Sepsis]) trial as results not yet publicly reported.
Grade A evidence for null treatment effect (or harm) for HES vs other fluids for both mortality and renal replacement therapy outcomes.
In the CHEST trial, all 7000 enrolled were critically ill patients, of whom 29% had sepsis and 45% were in shock; maximum potential septic shock accrual is estimated as 913 patients.
Several meta-analyses have found consistent results. A network meta-analysis using direct and indirect comparisons in severe sepsis found evidence of greater of mortality with hydroxyethyl starch vs crystalloid (RR, 1.13 [95% CI, 0.99–1.30]; high confidence), and no difference for albumin (RR, 0.83 [95% CI, 0.65–1.04]; moderate confidence) or gelatin vs crystalloids (RR, 1.24 [95% CI, 0.61–2.55]; very low confidence). Comparison of albumin vs hydroxyethyl starch indirectly favored albumin (RR, 0.73 [95%CI, 0.56–0.95]; moderate confidence).8 Additional meta-analyses in sepsis have confirmed a greater rate of renal failure or all-cause mortality comparing hydroxylethyl starch to other solutions, although no meta-analysis evaluated patients strictly in shock (eTable 2 in the Supplement).9
Vasopressors
For shock that is persistent despite adequate circulating volume, vasopressors are recommended to maintain perfusion of vital organs. Vasopressors such as norepinephrine, epinephrine, dopamine, and phenylephrine differ in their half-life, β- and α-adrenergic stimulation, and dosing regimens. Recent evidence comes from the SOAP II trial (Sepsis Occurrence in Acutely Ill Patients), a double-blind RCT in 8 centers testing norepinephrine vs dopamine in 1679 undifferentiated ICU patients with shock, of whom 62% had sepsis (Table 3).49 Although no difference was observed in 28-day mortality or in predefined septic shock subgroup, arrhythmias were significantly greater with dopamine. A meta-analysis of 6 trials in septic shock found a greater mortality with dopamine vs norepinephrine (RR, 1.12 [95% CI, 1.01–1.20]; eTable 3 in the Supplement).56 As a result, expert opinion26 and consensus guidelines21 recommend norepinephrine as the first vasopressor choice in septic shock. Vasodilatory shock in sepsis can also be reversed with the endogenous hormone, vasopressin. The administration of vasopressin can reduce norepinephrine dose and has been found to be safe, albeit with no mortality benefit, in subsequent meta-analyses.57,58 Consensus guidelines suggest vasopressin at a fixed dose (0.03–0.04 U/min) in patients without contraindication who are taking a norepinephrine dose of at least 0.15 μg/kg/minute. There may be select indications for alternative vasopressors such as when tachyarrythmias, limb ischemia, or other adverse effects dictate.1
Protocols
Current guidelines and an expert opinion recommend that clinicians incorporate a structured approach to resuscitation in septic shock.21,30 The principles of initial management include rapid recognition, prompt antibiotics, obtainment of cultures, and control of the infection source. After these initial steps, new evidence suggests that protocol-based, early goal-directed therapy (EGDT) may confer little survival advantage compared with clinical assessments of organ perfusion and management without a protocol (Table 3).35,50,51 The PROCESS (Protocol-Based Care for Early Septic Shock) trial found that 60-day in-hospital mortality for protocolized standard care (18.2%) was similar to usual care (18.9%) and protocolized early goal directed therapy (21%) among 1341 patients enrolled in 31 US emergency departments.35 The ARISE (Australasian Resuscitation in Sepsis Evaluation) trial confirmed this finding, reporting that among 1600 early septic shock patients in 51 centers in Australia and New Zealand that 90-day mortality was similar between EGDT and usual care.51 The PROMISE (Protocolized Management in Sepsis) trial enrolled 1260 patients in 56 hospitals in England, finding that EGDT offered no mortality benefit in early septic shock, but increased treatment intensity and cost.50 These findings were consistent for multiple a priori subgroups—including those stratified by demographics, severity of illness, time to enrollment, and lactate. Multiple subsequent meta-analyses of PROCESS, ARISE, and PROMISE trials have confirmed that EGDT offers no mortality benefit while increasing health care utilization and ICU admission in well-resourced countries.59–62 Notably, these studies enrolled patients with distinct physiology and improved preenrollment resuscitation than prior research.63 Modified versions of EGDT were also tested in lower-resourced settings with no change in outcome.53
Given the challenge of studying multistep protocols,64 the SepsisPAM (Sepsis and Mean Arterial Pressure) trial tested a single element in shock protocols—the mean arterial pressure target. Among 776 septic shock patients in France, a high mean arterial pressure target (80–85 mm Hg) conferred no survival advantage at 28 days (hazard ratio, 1.07 [95%CI, 0.84–1.38]; P = .57) compared with a low mean arterial pressure target (65–70 mm Hg).52 Notably, the subgroup of patients with a history of hypertension had lower rates of acute kidney injury and renal replacement therapy in the high mean arterial pressure target group. Beyond this study, a meta-analysis confirmed the paucity of evidence to help guide blood pressure management in septic shock.65 The original EGDT protocol also targeted a high hemoglobin threshold of greater than 10 g/dL. The recent Scandinavian TRISS (Transfusion Requirements in Septic Shock) trial demonstrated in 1005 septic shock patients that a lower threshold (7 g/dL) resulted in similar 90-day mortality as a higher threshold (9 g/dL) and reduced transfusions by 50%.54
Adjuncts
Many adjunctive treatments in septic shock target perturbations in the innate immune response and coagulation cascade. Yet few trials demonstrate benefit, most notably those of activated protein C and the TLR4 antagonist, Eritoran.66,67 However, specific adjuncts like corticosteroids in septic shock continued to be widely used.68 A large negative clinical trial69 and a conflicting systematic review in 2009 extended the debate about whether corticosteroids improve 28-day mortality or shock reversal.70 A recent meta-analysis reported on 8 trials of approximately 1000 patients finding that hydrocortisone (≤300 mg/d) was associated with no significant change in 28-day mortality yet reduced the odds of shock over 7 and 28 days.71 Consensus guidelines recommend low-dose glucocorticoid therapy only in patients with vasopressor-dependent septic shock and removal once vasopressors are no longer needed.21 A more extensive discussion of the trade offs of corticosteroid therapy is found elsewhere.68
Areas of Uncertainty
The prompt administration of intravenous fluid is a ubiquitous therapy in septic shock, yet many aspects of this treatment are unknown. First, the timing (ie, prehospital vs emergency department) and effectiveness of fluid bolus therapy has come under question.72 Second, no trial has directly compared balanced vs unbalanced crystalloids in early septic shock. Third, the ideal resuscitation target remains an important knowledge gap, particularly since recent evidence suggests there may be a disconnect between the augmentation of systemic hemodynamics and different measures of regional perfusion.73 Additionally, the overuse of fluids in septic shock is common.74 More work is needed to understand the optimal timing and method of fluid removal.
Discussion
The typology of shock is informed by classic animal experiments in specific physiologic states, such as crushing injury, hypovolemia, or profound hemorrhage. This has led to a clinical approach to shock based on aggressive fluid resuscitation and supported by evidence from small, single-center clinical trials.63 The underlying heterogeneity of shock is now more apparent as modern pragmatic trials enroll large numbers of patients with complex physiology. Although unusual cases will always be difficult to define, the variety of enrollment criteria in recent trials highlights that experts lack consensus even about the core elements of shock.75 This issue is magnified as clinicians and researchers attempt to treat shock at its earliest presentation. Ultimately, a simple strategy to promptly diagnose shock using easy-to-measure clinical features may help lower overall mortality in the majority of cases. However, those cases with complex physiology or occult presentations may require a more precise approach informed by biologic phenotypes and advanced hemodynamic monitoring.
This review has several limitations. First, we restricted our search to the past 5 years, and excluded articles prior to 2010. Second, we addressed the prompt diagnosis and treatment of shock, and various diagnostic tools or treatments will have different benefit or harm at later stages.76 Third, guidelines for shock are infrequently updated—a process less nimble to new evidence and challenged by trials where usual care is equivalent to the intervention. Finally, many studies in this review enrolled patients without septic shock. These studies included patients with sepsis who did and did not have varying degrees of organ dysfunction. Thus, the rate of shock across studies ranged from 13% to 100% and few meta-analyses could focus entirely on septic shock due to trial heterogeneity.
Clinical Bottom Line
Diagnosis
Septic shock is an emergency event requiring prompt clinical diagnosis.
Focused ultrasonography may assist in early shock diagnosis and alert clinicians to underlying physiologic disturbance.
Invasive (eg, pulmonary artery catheter) and noninvasive hemodynamic monitoring devices (eg, pulse contour analysis) are only recommended for use in select subgroups of septic shock.
Lactate is widely used in shock assessment but deserves further evaluation of its specific role in diagnostic and treatment algorithms.
Treatment
The first step in the treatment of septic shock is promptly addressing suspected or documented infection.
Protocol-guided fluid resuscitation in septic shock is not superior to management by clinical assessment without a protocol.
A variety of crystalloid fluids or albumin are recommended in septic shock, while hydroxyethyl starch solutions may be associated with worse outcomes.
Conclusions
Septic shock is a clinical emergency. A prompt diagnosis of septic shock begins with a focused history and physical examination for signs and symptoms of infection and may require focused ultrasonographytorecognizecomplexphysiologicmanifestationsofshock. Clinicians should understand the importance of prompt administration of intravenous fluids aimed at restoring adequate circulation, vasoactive medications, and the limitations of protocol-based therapy, as guided by recent evidence.
Supplementary Material
Acknowledgments
Funding/Support: This work was supported in part by a grant from the National Institutes of Health (NIH) (K23GM104022).
Role of the Funder/Sponsor: The NIH had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Footnotes
Author Contributions: Dr Seymour had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Study concept and design: Seymour, Rosengart. Acquisition, analysis, or interpretation of data:Seymour, Rosengart.
Drafting of the manuscript: Seymour, Rosengart. Critical revision of the manuscript for important intellectual content: Seymour, Rosengart.
Statistical analysis: Seymour.
Obtained funding: Seymour.
Administrative, technical, or material support: Seymour.
Conflict of Interest Disclosures: Both authors have completed and submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest. Dr Seymour reports receipt of consultancy fees from Beckman Coulter outside the submitted work. No other disclosures were reported.
Additional Contributions: The authors would like to thank Charles B. Wessel, MLS, University of Pittsburgh Health Sciences Library System, for assistance in developing search strings. The authors also thank Michael Pinsky, MD, University of Pittsburgh School of Medicine; Derek Angus, MD, MPH, University of Pittsburgh School of Medicine and JAMA Associate Editor; and John Kellum, MD, University of Pittsburgh Department of Critical Care Medicine, for critical review of the manuscript. None of these individuals received compensation in association with their respective contributions to this article.
Correction: This article was corrected online August 27, 2015, for an incorrect title to Table 1.
Submissions: We encourage authors to submit papers for consideration as a Review. Please contact Edward Livingston, MD, at Edward.livingston@jamanetwork.org or Mary McGrae McDermott, MD, at mdm608@northwestern.edu.
References
- 1.Walley KR, Wood LDH. Shock. In: Hall J, Wood LDH, editors. Principles of Critical Care. New York, NY: McGraw Hill Co; 1998. pp. 277–301. [Google Scholar]
- 2.Millham FH. A brief history of shock. Surgery. 2010;148(5):1026–1037. doi: 10.1016/j.surg.2010.02.014. [DOI] [PubMed] [Google Scholar]
- 3.Manji RA, Wood KE, Kumar A. The history and evolution of circulatory shock. Crit Care Clin. 2009;25(1):1–29. doi: 10.1016/j.ccc.2008.12.013. [DOI] [PubMed] [Google Scholar]
- 4.Weil MH, Shubin H. Proposed reclassification of shock states with special reference to distributive defects. Adv Exp Med Biol. 1971;23(0):13–23. doi: 10.1007/978-1-4615-9014-9_3. [DOI] [PubMed] [Google Scholar]
- 5.Parker MM, Shelhamer JH, Bacharach SL, et al. Profound but reversible myocardial depression in patients with septic shock. Ann Intern Med. 1984;100(4):483–490. doi: 10.7326/0003-4819-100-4-483. [DOI] [PubMed] [Google Scholar]
- 6.Murray CJ, Atkinson C, Bhalla K, et al. US Burden of Disease Collaborators. The state of US health, 1990–2010: burden of diseases, injuries, and risk factors. JAMA. 2013;310(6):591–608. doi: 10.1001/jama.2013.13805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Labovitz AJ, Noble VE, Bierig M, et al. Focused cardiac ultrasound in the emergent setting: a consensus statement of the American Society of Echocardiography and American College of Emergency Physicians. J Am Soc Echocardiogr. 2010;23(12):1225–1230. doi: 10.1016/j.echo.2010.10.005. [DOI] [PubMed] [Google Scholar]
- 8.Rochwerg B, Alhazzani W, Sindi A, et al. Fluids in Sepsis and Septic Shock Group. Fluid resuscitation in sepsis: a systematic review and network meta-analysis. Ann Intern Med. 2014;161(5):347–355. doi: 10.7326/M14-0178. [DOI] [PubMed] [Google Scholar]
- 9.Rochwerg B, Alhazzani W, Gibson A, et al. FISSH Group (Fluids in Sepsis and Septic Shock) Fluid type and the use of renal replacement therapy in sepsis: a systematic review and network meta-analysis [published online April 23, 2015] Intensive Care Med. doi: 10.1007/s00134-015-3794-1. [DOI] [PubMed] [Google Scholar]
- 10.Levy MM, Fink MP, Marshall JC, et al. SCCM/ESICM/ACCP/ATS/SIS. 2001 SCCM/ESICM/ACCP/ATS/SIS International Sepsis Definitions Conference. Crit Care Med. 2003;31(4):1250–1256. doi: 10.1097/01.CCM.0000050454.01978.3B. [DOI] [PubMed] [Google Scholar]
- 11.Tisherman SA, Barie P, Bokhari F, et al. Clinical practice guideline: endpoints of resuscitation. J Trauma. 2004;57(4):898–912. doi: 10.1097/01.ta.0000133577.25793.e5. [DOI] [PubMed] [Google Scholar]
- 12.Cecconi M, De Backer D, Antonelli M, et al. Consensus on circulatory shock and hemodynamic monitoring: task force of the European Society of Intensive Care Medicine. Intensive Care Med. 2014;40(12):1795–1815. doi: 10.1007/s00134-014-3525-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Levy MM, Dellinger RP, Townsend SR, et al. The Surviving Sepsis Campaign: results of an international guideline-based performance improvement program targeting severe sepsis. Intensive Care Med. 2010;36(2):222–231. doi: 10.1007/s00134-009-1738-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Reinhart K, Bauer M, Riedemann NC, Hartog CS. New approaches to sepsis: molecular diagnostics and biomarkers. Clin Microbiol Rev. 2012;25(4):609–634. doi: 10.1128/CMR.00016-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Rajaram SS, Desai NK, Kalra A, et al. Pulmonary artery catheters for adult patients in intensive care. Cochrane Database Syst Rev. 2013;2:CD003408. doi: 10.1002/14651858.CD003408.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Clermont G, Kong L, Weissfeld LA, et al. NHLBI ARDS Clinical Trials Network. The effect of pulmonary artery catheter use on costs and long-term outcomes of acute lung injury. PLoS One. 2011;6(7):e22512. doi: 10.1371/journal.pone.0022512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Jones AE, Shapiro NI, Trzeciak S, Arnold RC, Claremont HA, Kline JA Emergency Medicine Shock Research Network (EMShockNet) Investigators. Lactate clearance vs central venous oxygen saturation as goals of early sepsis therapy: a randomized clinical trial. JAMA. 2010;303(8):739–746. doi: 10.1001/jama.2010.158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Zhang Z, Ni H, Qian Z. Effectiveness of treatment based on PiCCO parameters in critically ill patients with septic shock and/or acute respiratory distress syndrome: a randomized controlled trial. Intensive Care Med. 2015;41(3):444–451. doi: 10.1007/s00134-014-3638-4. [DOI] [PubMed] [Google Scholar]
- 19.Richard JC, Bayle F, Bourdin G, et al. Preload dependence indices to titrate volume expansion during septic shock: a randomized controlled trial. Crit Care. 2015;19(1):5. doi: 10.1186/s13054-014-0734-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Jansen TC, van Bommel J, Schoonderbeek FJ, et al. LACTATE study group. Early lactate-guided therapy in intensive care unit patients: a multicenter, open-label, randomized controlled trial. Am J Respir Crit Care Med. 2010;182(6):752–761. doi: 10.1164/rccm.200912-1918OC. [DOI] [PubMed] [Google Scholar]
- 21.Dellinger RP, Levy MM, Rhodes A, et al. Surviving Sepsis Campaign Guidelines Committee including the Pediatric Subgroup. Surviving sepsis campaign: international guidelines for management of severe sepsis and septic shock: 2012. Crit Care Med. 2013;41(2):580–637. doi: 10.1097/CCM.0b013e31827e83af. [DOI] [PubMed] [Google Scholar]
- 22.Wiener RS, Welch HG. Trends in the use of the pulmonary artery catheter in the United States, 1993–2004. JAMA. 2007;298(4):423–429. doi: 10.1001/jama.298.4.423. [DOI] [PubMed] [Google Scholar]
- 23.Takala J, Ruokonen E, Tenhunen JJ, Parviainen I, Jakob SM. Early non-invasive cardiac output monitoring in hemodynamically unstable intensive care patients: a multi-center randomized controlled trial. Crit Care. 2011;15(3):R148. doi: 10.1186/cc10273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Garcia X, Gruartmoner G. ClinicalTrials.gov website. [Accessed July 15, 2015];Monitoring resuscitation in severe sepsis and septic shock ( NCT01747057) https://clinicaltrials.gov/ct2/show/NCT01747057?term=nct01747057&rank=1.
- 25.Pearse RM, Harrison DA, MacDonald N, et al. OPTIMISE Study Group. Effect of a perioperative, cardiac output-guided hemodynamic therapy algorithm on outcomes following major gastrointestinal surgery: a randomized clinical trial and systematic review. JAMA. 2014;311(21):2181–2190. doi: 10.1001/jama.2014.5305. [DOI] [PubMed] [Google Scholar]
- 26.Vincent JL, De Backer D. Circulatory shock. N Engl J Med. 2013;369(18):1726–1734. doi: 10.1056/NEJMra1208943. [DOI] [PubMed] [Google Scholar]
- 27.Liu V, Morehouse JW, Soule J, Whippy A, Escobar GJ. Fluid volume, lactate values, and mortality in sepsis patients with intermediate lactate values. Ann Am Thorac Soc. 2013;10(5):466–473. doi: 10.1513/AnnalsATS.201304-099OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Puskarich MA, Trzeciak S, Shapiro NI, Heffner AC, Kline JA, Jones AE Emergency Medicine Shock Research Network (EMSHOCKNET) Outcomes of patients undergoing early sepsis resuscitation for cryptic shock compared with overt shock. Resuscitation. 2011;82(10):1289–1293. doi: 10.1016/j.resuscitation.2011.06.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Gomez H, Kellum JA. Lactate in sepsis. JAMA. 2015;313(2):194–195. doi: 10.1001/jama.2014.13811. [DOI] [PubMed] [Google Scholar]
- 30.Angus DC, van der Poll T. Severe sepsis and septic shock. N Engl J Med. 2013;369(21):2063. doi: 10.1056/NEJMc1312359. [DOI] [PubMed] [Google Scholar]
- 31.Boomer JS, To K, Chang KC, et al. Immunosuppression in patients who die of sepsis and multiple organ failure. JAMA. 2011;306(23):2594–2605. doi: 10.1001/jama.2011.1829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Wong HR, Cvijanovich NZ, Allen GL, et al. Validation of a gene expression-based subclassification strategy for pediatric septic shock. Crit Care Med. 2011;39(11):2511–2517. doi: 10.1097/CCM.0b013e3182257675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Langley RJ, Tsalik EL, van Velkinburgh JC, et al. An integrated clinico-metabolomic model improves prediction of death in sepsis. Sci Transl Med. 2013;5(195):195ra95. doi: 10.1126/scitranslmed.3005893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Bulger EM, May S, Kerby JD, et al. ROC investigators. Out-of-hospital hypertonic resuscitation after traumatic hypovolemic shock: a randomized, placebo controlled trial. Ann Surg. 2011;253(3):431–441. doi: 10.1097/SLA.0b013e3181fcdb22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Yealy DM, Kellum JA, Huang DT, et al. ProCESS Investigators. A randomized trial of protocol-based care for early septic shock. N Engl J Med. 2014;370(18):1683–1693. doi: 10.1056/NEJMoa1401602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kaukonen KM, Bailey M, Suzuki S, Pilcher D, Bellomo R. Mortality related to severe sepsis and septic shock among critically ill patients in Australia and New Zealand, 2000–2012. JAMA. 2014;311(13):1308–1316. doi: 10.1001/jama.2014.2637. [DOI] [PubMed] [Google Scholar]
- 37.Myburgh JA, Mythen MG. Resuscitation fluids. N Engl J Med. 2013;369(25):2462–2463. doi: 10.1056/NEJMc1313345. [DOI] [PubMed] [Google Scholar]
- 38.Morgan TJ. The ideal crystalloid—what is ‘balanced’? Curr Opin Crit Care. 2013;19(4):299–307. doi: 10.1097/MCC.0b013e3283632d46. [DOI] [PubMed] [Google Scholar]
- 39.Yunos NM, Bellomo R, Hegarty C, Story D, Ho L, Bailey M. Association between a chloride-liberal vs chloride-restrictive intravenous fluid administration strategy and kidney injury in critically ill adults. JAMA. 2012;308(15):1566–1572. doi: 10.1001/jama.2012.13356. [DOI] [PubMed] [Google Scholar]
- 40.Young P. Australian New Zealand Clinical Trials Registry website. [Accessed June 4, 2015];0.9% Saline vs Plasma-Lyte 148 for intensive care fluid therapy: the SPLIT study. ID: ACTRN12613001370796. https://www.anzctr.org.au/Trial/Registration/TrialReview.aspx?id=365460.
- 41.Reddy SK, Bailey MJ, Beasley RW, et al. A protocol for the 0.9% saline versus Plasma-Lyte 148 for intensive care fluid therapy (SPLIT) study. Crit Care Resusc. 2014;16(4):274–279. [PubMed] [Google Scholar]
- 42.Finfer S, Liu B, Taylor C, et al. SAFE TRIPS Investigators. Resuscitation fluid use in critically ill adults: an international cross-sectional study in 391 intensive care units. Crit Care. 2010;14(5):R185. doi: 10.1186/cc9293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Goldenberg NM, Steinberg BE, Slutsky AS, Lee WL. Broken barriers: a new take on sepsis pathogenesis. Sci Transl Med. 2011;3(88):88ps25. doi: 10.1126/scitranslmed.3002011. [DOI] [PubMed] [Google Scholar]
- 44.Caironi P, Tognoni G, Masson S, et al. ALBIOS Study Investigators. Albumin replacement in patients with severe sepsis or septic shock. N Engl J Med. 2014;370(15):1412–1421. doi: 10.1056/NEJMoa1305727. [DOI] [PubMed] [Google Scholar]
- 45.Annane D, Siami S, Jaber S, et al. CRISTAL Investigators. Effects of fluid resuscitation with colloids vs crystalloids on mortality in critically ill patients presenting with hypovolemic shock: the CRISTAL randomized trial. JAMA. 2013;310(17):1809–1817. doi: 10.1001/jama.2013.280502. [DOI] [PubMed] [Google Scholar]
- 46.Myburgh JA, Finfer S, Bellomo R, et al. CHEST Investigators; Australian and New Zealand Intensive Care Society Clinical Trials Group. Hydroxyethyl starch or saline for fluid resuscitation in intensive care. N Engl J Med. 2012;367(20):1901–1911. doi: 10.1056/NEJMoa1209759. [DOI] [PubMed] [Google Scholar]
- 47.Perner A, Haase N, Guttormsen AB, et al. 6S Trial Group; Scandinavian Critical Care Trials Group. Hydroxyethyl starch 130/0.42 versus Ringer’s acetate in severe sepsis. N Engl J Med. 2012;367(2):124–134. doi: 10.1056/NEJMoa1204242. [DOI] [PubMed] [Google Scholar]
- 48.Perner A, Haase N, Winkel P, et al. Long-term outcomes in patients with severe sepsis randomised to resuscitation with hydroxyethyl starch 130/0.42 or Ringer’s acetate. Intensive Care Med. 2014;40(7):927–934. doi: 10.1007/s00134-014-3311-y. [DOI] [PubMed] [Google Scholar]
- 49.De Backer D, Biston P, Devriendt J, et al. SOAP II Investigators. Comparison of dopamine and norepinephrine in the treatment of shock. N Engl J Med. 2010;362(9):779–789. doi: 10.1056/NEJMoa0907118. [DOI] [PubMed] [Google Scholar]
- 50.Mouncey PR, Osborn TM, Power GS, et al. PROMISE Trial Investigators. Trial of early, goal-directed resuscitation for septic shock. N Engl J Med. 2015;372(14):1301–1311. doi: 10.1056/NEJMoa1500896. [DOI] [PubMed] [Google Scholar]
- 51.Peake SL, Delaney A, Bailey M, et al. ARISE Investigators; ANZICS Clinical Trials Group. Goal-directed resuscitation for patients with early septic shock. N Engl J Med. 2014;371(16):1496–1506. doi: 10.1056/NEJMoa1404380. [DOI] [PubMed] [Google Scholar]
- 52.Asfar P, Meziani F, Hamel JF, et al. SEPSISPAM Investigators. High versus low blood-pressure target in patients with septic shock. N Engl J Med. 2014;370(17):1583–1593. doi: 10.1056/NEJMoa1312173. [DOI] [PubMed] [Google Scholar]
- 53.Andrews B, Muchemwa L, Kelly P, Lakhi S, Heimburger DC, Bernard GR. Simplified severe sepsis protocol: a randomized controlled trial of modified early goal-directed therapy in Zambia. Crit Care Med. 2014;42(11):2315–2324. doi: 10.1097/CCM.0000000000000541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Holst LB, Haase N, Wetterslev J, et al. TRISS Trial Group; Scandinavian Critical Care Trials Group. Lower versus higher hemoglobin threshold for transfusion in septic shock. N Engl J Med. 2014;371(15):1381–1391. doi: 10.1056/NEJMoa1406617. [DOI] [PubMed] [Google Scholar]
- 55.Siegemund M. Clinicaltrials.gov website. [Accessed July 16, 2015];Basel Study for Evaluation of Starch (130;0.4) Infusion in Septic Patients: BASES (130;0.4) Trial. https://clinicaltrials.gov/ct2/show/NCT00273728?term=NCT00273728&rank=1.
- 56.De Backer D, Aldecoa C, Njimi H, Vincent JL. Dopamine versus norepinephrine in the treatment of septic shock: a meta-analysis. Crit Care Med. 2012;40(3):725–730. doi: 10.1097/CCM.0b013e31823778ee. [DOI] [PubMed] [Google Scholar]
- 57.Polito A, Parisini E, Ricci Z, Picardo S, Annane D. Vasopressin for treatment of vasodilatory shock: an ESICM systematic review and meta-analysis. Intensive Care Med. 2012;38(1):9–19. doi: 10.1007/s00134-011-2407-x. [DOI] [PubMed] [Google Scholar]
- 58.Serpa Neto A, Nassar AP, Cardoso SO, et al. Vasopressin and terlipressin in adult vasodilatory shock: a systematic review and meta-analysis of nine randomized controlled trials. Crit Care. 2012;16(4):R154. doi: 10.1186/cc11469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Huang DT, Angus DC, Barnato A, et al. ProCESS/ARISE/ProMISe Methodology Writing Committee. Harmonizing international trials of early goal-directed resuscitation for severe sepsis and septic shock: methodology of PROCESS, ARISE, and PROMISE. Intensive Care Med. 2013;39(10):1760–1775. doi: 10.1007/s00134-013-3024-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Angus DC, Barnato AE, Bell D, et al. A systematic review and meta-analysis of early goal-directed therapy for septic shock: the ARISE, PROCESS and PROMISE Investigators [published online May 8, 2015] Intensive Care Med. doi: 10.1007/s00134-015-3822-1. [DOI] [PubMed] [Google Scholar]
- 61.Zhang L, Zhu G, Han L, Fu P. Early goal-directed therapy in the management of severe sepsis or septic shock in adults: a meta-analysis of randomized controlled trials. BMC Med. 2015;13:71. doi: 10.1186/s12916-015-0312-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Rusconi AM, Bossi I, Lampard JG, Szava-Kovats M, Bellone A, Lang E. Early goal-directed therapy vs usual care in the treatment of severe sepsis and septic shock: a systematic review and meta-analysis [published online May 16, 2015] Intern Emerg Med. doi: 10.1007/s11739-015-1248-y. [DOI] [PubMed] [Google Scholar]
- 63.Rivers E, Nguyen B, Havstad S, et al. Early Goal-Directed Therapy Collaborative Group. Early goal-directed therapy in the treatment of severe sepsis and septic shock. N Engl J Med. 2001;345(19):1368–1377. doi: 10.1056/NEJMoa010307. [DOI] [PubMed] [Google Scholar]
- 64.Lewis RJ. Disassembling goal-directed therapy for sepsis: a first step. JAMA. 2010;303(8):777–779. doi: 10.1001/jama.2010.203. [DOI] [PubMed] [Google Scholar]
- 65.D’Aragon F, Belley-Cote EP, Meade MO, et al. Canadian Critical Care Trials Group. Blood pressure targets for vasopressor therapy: a systematic review. Shock. 2015;43(6):530–539. doi: 10.1097/SHK.0000000000000348. [DOI] [PubMed] [Google Scholar]
- 66.Opal SM, Laterre PF, Francois B, et al. ACCESS Study Group. Effect of eritoran, an antagonist of MD2-TLR4, on mortality in patients with severe sepsis: the ACCESS randomized trial. JAMA. 2013;309(11):1154–1162. doi: 10.1001/jama.2013.2194. [DOI] [PubMed] [Google Scholar]
- 67.Ranieri VM, Thompson BT, Barie PS, et al. PROWESS-SHOCK Study Group. Drotrecogin alfa (activated) in adults with septic shock. N Engl J Med. 2012;366(22):2055–2064. doi: 10.1056/NEJMoa1202290. [DOI] [PubMed] [Google Scholar]
- 68.Patel GP, Balk RA. Systemic steroids in severe sepsis and septic shock. Am J Respir Crit Care Med. 2012;185(2):133–139. doi: 10.1164/rccm.201011-1897CI. [DOI] [PubMed] [Google Scholar]
- 69.Sprung CL, Annane D, Keh D, et al. CORTICUS Study Group. Hydrocortisone therapy for patients with septic shock. N Engl J Med. 2008;358(2):111–124. doi: 10.1056/NEJMoa071366. [DOI] [PubMed] [Google Scholar]
- 70.Annane D, Bellissant E, Bollaert PE, et al. Corticosteroids in the treatment of severe sepsis and septic shock in adults: a systematic review. JAMA. 2009;301(22):2362–2375. doi: 10.1001/jama.2009.815. [DOI] [PubMed] [Google Scholar]
- 71.Wang C, Sun J, Zheng J, et al. Low-dose hydrocortisone therapy attenuates septic shock in adult patients but does not reduce 28-day mortality: a meta-analysis of randomized controlled trials. Anesth Analg. 2014;118(2):346–357. doi: 10.1213/ANE.0000000000000050. [DOI] [PubMed] [Google Scholar]
- 72.Lipcsey M, Chiong J, Subiakto I, Kaufman MA, Schneider AG, Bellomo R. Primary fluid bolus therapy for infection-associated hypotension in the emergency department. Crit Care Resusc. 2015;17(1):6–11. [PubMed] [Google Scholar]
- 73.Hernandez G, Bruhn A, Luengo C, et al. Effects of dobutamine on systemic, regional and microcirculatory perfusion parameters in septic shock: a randomized, placebo-controlled, double-blind, crossover study. Intensive Care Med. 2013;39(8):1435–1443. doi: 10.1007/s00134-013-2982-0. [DOI] [PubMed] [Google Scholar]
- 74.Boyd JH, Forbes J, Nakada TA, Walley KR, Russell JA. Fluid resuscitation in septic shock: a positive fluid balance and elevated central venous pressure are associated with increased mortality. Crit Care Med. 2011;39(2):259–265. doi: 10.1097/CCM.0b013e3181feeb15. [DOI] [PubMed] [Google Scholar]
- 75.Shankar-Hari M, Deutschman CS, Singer M. Do we need a new definition of sepsis? Intensive Care Med. 2015;41(5):909–911. doi: 10.1007/s00134-015-3680-x. [DOI] [PubMed] [Google Scholar]
- 76.Hoste EA, Maitland K, Brudney CS, et al. ADQI XII Investigators Group. Four phases of intravenous fluid therapy: a conceptual model. Br J Anaesth. 2014;113(5):740–747. doi: 10.1093/bja/aeu300. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.