Abstract
Background:
We previously reported that the target genes in sporadic mismatch repair (MMR)-deficient colorectal carcinomas (CRCs) in the distal colon differ from those occurring elsewhere in the colon. This study aimed to compare the target gene mutational pattern in microsatellite instability (MSI) CRC from Lynch syndrome patients stratified by tumour location and germline mutation, as well as with that of sporadic disease.
Methods:
A series of CRC from Lynch syndrome patients was analysed for MSI in genes predicted to be selective MSI targets and known to be involved in several pathways of colorectal carcinogenesis.
Results:
The most frequently mutated genes belong to the TGF-β superfamily pathway, namely ACVR2A and TGFBR2. A significantly higher frequency of target gene mutations was observed in CRC from patients with germline mutations in MLH1 or MSH2 when compared with MSH6. Mutations in microsatellite sequences (A)7 of BMPR2 and (A)8 of MSH3 were significantly more frequent in the distal CRC. Additionally, we observed differences in MSH3 and TGFBR2 mutational frequency between Lynch syndrome and sporadic MSI CRC regarding tumour location.
Conclusions:
Our results indicate that the pattern of genetic changes differs in CRC depending on tumour location and between Lynch syndrome and sporadic MSI CRC, suggesting that carcinogenesis can occur by different pathways even if driven by generalised MSI.
Keywords: microsatellite instability, target genes, Lynch syndrome
Lynch syndrome is a highly penetrant, autosomal dominant disease characterised by early-onset colorectal cancer (CRC) and extracolonic tumours of the endometrium, stomach, small bowel, ureter, renal pelvis, ovary and hepatobiliary tract (Lynch and de la Chapelle, 2003). This is the most common hereditary CRC syndrome accounting for up to 4% of all CRC cases (Aaltonen et al, 1998; Hampel et al, 2008) and is commonly caused by a genetic defect affecting one of the four mismatch repair (MMR) genes MLH1, MSH2, MSH6 and PMS2 (Lagerstedt Robinson et al, 2007). The selection of families for genetic testing is mainly based on personal and family cancer history using the Amsterdam criteria or the Bethesda guidelines (Vasen et al, 1999; Umar et al, 2004).
More than 95% of the tumours arising in carriers of MMR gene mutations show microsatellite instability (MSI) (Aaltonen et al, 1994). Microsatellite instability is characterised by a widespread instability in coding and noncoding short repeat microsatellite sequences, because of MMR deficiency (Perucho, 1996). Through the MSI pathway, CRC progression is accelerated by a rapid mutation accumulation in coding repetitive sequences of target genes with growth-related functions. In Lynch syndrome MSI CRC, somatic mutations have been described in several genes with important cellular roles, such as growth factor receptors (TGFBR2 and IGF2R), genes involved in apoptosis (BAX) and DNA repair (MSH3 and MSH6) (Yamaguchi et al, 2006), together with many other microsatellite mutations that are not mechanistically responsible for the behaviour of tumour cells.
We have previously reported that, in sporadic MSI CRC, the target genes in MMR-deficient tumours of distal colon and rectum differ from tumours elsewhere in the colon (Pinheiro et al, 2010). In this study, we aimed to compare the target gene mutational pattern in MSI CRC from Lynch syndrome patients stratified by tumour location, germline-mutated gene and type of mutation (founder mutations compared with other mutations in the same gene).
Materials and methods
Patients, samples and DNA extraction
This study includes altogether 129 CRC samples from 114 patients belonging to 98 Lynch syndrome families with deleterious (class 5 or 4 according to the InSIGHT variant interpretation committee guidelines) MMR germline mutations (Table 1). The test series includes 78 CRCs, obtained by surgical resection, from 65 patients belonging to 50 Portuguese Lynch syndrome families presenting a germline MMR gene mutation. These mutations were identified by routine genetic diagnosis during the period of 1997–2011 at the Department of Genetics of the Portuguese Oncology Institute, Porto, Portugal, after genetic counselling and informed consent. Thirty-nine families were followed at the Portuguese Oncology Institute and 11 at Centro Hospitalar de S João, Porto, Portugal. Twenty-seven of the families fulfilled the Amsterdam criteria, whereas the remaining presented the Bethesda criteria for genetic testing. Twenty-seven (54%) families carried a pathogenic germline mutation in MSH2, 16 (32%) in MLH1, 6 (12%) in MSH6 and 1 (2%) in PMS2 (Table 1). Furthermore, 9 (18%) and 10 (20%) of these families carried the MLH1 c.1896+280_oLRRFIP2:c.1750-678del and the MSH2 c.388_389del Portuguese founder mutations, respectively (Pinheiro et al, 2011, 2013). Forty (51%) tumours belonged to patients carrying a germline mutation in MSH2, 31 (40%) in MLH1, 6 (8%) in MSH6 and 1 (1%) in PMS2. The 65 patients included 32 females and 33 males with a mean CRC diagnosis age of 46 years (range, 15–75 years). Clinical data were drawn from hospital records and tumour staging was performed using the American Joint Committee on Cancer (AJCC) criteria. Summary of the clinicopathological data is presented in Table 1. All large bowel regions up to the splenic flexure were considered as proximal colon and after that as distal colon. All tumour samples were paraffin embedded and were reviewed by a pathologist. Peripheral blood was collected from the same patients. DNA was isolated from paraffin-embedded tumour and from peripheral blood using standard procedures. This study was approved by the Institutional Review Board.
Table 1. Clinicopathologic characteristics of Lynch syndrome patients and families.
|
Portuguese Lynch syndrome families |
Swiss Lynch syndrome families |
||||||||
|---|---|---|---|---|---|---|---|---|---|
| Total | MLH1 | MSH2 | MSH6 | PMS2 | Total | MLH1 | MSH2 | MSH6 | |
| Families | 50 | 16 (32%) | 27 (54%) | 6 (12%) | 1 (2%) | 48 | 26 (54%) | 21 (44%) | 1 (2%) |
|
Criteria | |||||||||
| Amsterdam | 27 (54%) | 9 (33%) | 17 (63%) | 1 (4%) | 0 (0%) | 33 (69%) | 22 (67%) | 10 (30%) | 1 (3%) |
| Bethesda | 23 (46%) | 7 (30%) | 10 (43%) | 5 (22%) | 1 (4%) | 15 (31%) | 4 (27%) | 11 (73%) | 0 (0%) |
| CRC patients | 65 | 25 (38%) | 33 (51%) | 6 (9%) | 1 (2%) | 49 | 27 (55%) | 21 (43 %) | 1 (2%) |
|
Gender | |||||||||
| Female | 32 (49%) | 13 (41%) | 16 (50%) | 2 (6%) | 1 (3%) | 26 (53%) | 14 (54%) | 11 (42%) | 1 (4%) |
| Male | 33 (51%) | 12 (36%) | 17 (52%) | 4 (12%) | 0 (0%) | 23 (47%) | 13 (57%) | 10 (43%) | 0 (0%) |
|
Age at diagnosis of CRC (years) | |||||||||
| Mean | 46 | 46 | 46 | 44 | 55 | 45 | 45 | 46 | 48 |
| Range | 15–75 | 27–68 | 15–75 | 21–71 | 21–79 | 30–65 | 21–79 | ||
| ⩽50 | 55 (71%) | 21 (38%) | 29 (53%) | 5 (9%) | 0 (0%) | 35 (69%) | 19 (54%) | 15 (43%) | 1 (3%) |
| >50 | 23 (29%) | 10 (43%) | 11 (48%) | 1 (4%) | 1 (4%) | 16 (31%) | 9 (56%) | 7 (44%) | 0 (0%) |
| Total CRC | 78 | 31 (40%) | 40 (51%) | 6 (8%) | 1 (1%) | 51 | 28 (55%) | 22 (43%) | 1 (2%) |
|
CRC localisation | |||||||||
| Proximal | 51 (65%) | 23 (45%) | 24 (47%) | 4 (8%) | 0 (0%) | 35 (69%) | 22 (63%) | 12 (34%) | 1 (3%) |
| Distal | 27 (35%) | 8 (30%) | 16 (59%) | 2 (7%) | 1 (4%) | 16 (31%) | 6 (38%) | 10 (63%) | 0 (0%) |
|
TNM stagea | |||||||||
| I/II | 45 (68%) | 20 (44%) | 23 (51%) | 2 (4%) | – | 32 (70%) | 20 (63%) | 12 (38%) | 0 (0%) |
| III/IV | 21 (32%) | 9 (43%) | 9 (43%) | 3 (14%) | – | 14 (30%) | 6 (43%) | 7 (50%) | 1 (7%) |
|
Differentiation gradeb | |||||||||
| Well/moderately | 53 (90%) | 20 (38%) | 28 (53%) | 5 (9%) | 0 (0%) | 25 (66%) | 15 (60 %) | 10 (40 %) | 0 (0%) |
| Poorly | 6 (10%) | 2 (33%) | 3 (50%) | 0 (0%) | 1 (17%) | 13 (34%) | 9 (69%) | 4 (31%) | 0 (0%) |
Abbreviations: CRC=colorectal carcinoma; TNM=tumor node metastasis.
Information was not available for 12 and 5 CRC from the Portuguese and Swiss Lynch syndrome families, respectively.
Information was not available for 19 and 13 CRC from the Portuguese and Swiss Lynch syndrome families, respectively.
We analysed an additional series that included 51 CRC samples from 49 Swiss patients belonging to 48 Lynch syndrome families referred to and genetically characterised by the Basel research group Human Genomics (Kovac et al, 2011). Twenty-six (54%) families carried germline mutations in MLH1, 21 (44%) in MSH2 and 1 (2%) in MSH6 (Table 1). The 49 patients included 26 females and 23 males, and summary if the clinicopathological data are shown in Table 1. Twenty-eight (55%) tumours belonged to patients carrying a germline mutation in MLH1, 22 (43%) in MSH2 and 1 (2%) in MSH6. Thirty-three of these families fulfilled the Amsterdam criteria, whereas the remaining presented the Bethesda criteria for genetic testing (Table 1).
Additionally, the data on Lynch syndrome patients were compared with that of a series of 42 sporadic MSI CRCs (22 and 20 localised in the proximal and distal colons, respectively) (Pinheiro et al, 2010).
MMR immunohistochemical and MSI analyses
Assessment of MLH1, MSH2, MSH6 and PMS2 immunoexpression was performed as described previously (Pinheiro et al, 2010) in 65 and 51 tumours belonging to the Portuguese and Swiss Lynch syndrome families, respectively. Microsatellite instability evaluation was performed using the Bethesda panel of markers (BAT25, BAT26, D2S123, D5S346 and D17S250) and the 1997 National Cancer Institute guidelines, as described previously (Pinheiro et al, 2009).
Target gene analyses
For somatic mutation analysis, we selected 17 genes belonging to pathways involved in CRC carcinogenesis, most of them known to acquire mutations in coding microsatellite sequences in tumours with MSI: TGFBR2 (A)10, ACVR2A (A)8, BMPR1A (T)6 (two sequences), BMPR2 (A)7 and (A)11, EGFR (A)13, E2F4 (CAG)13, BAX (G)8, PRDM2 (A)8 and (A)9, TCF7L2 (A)9, APC (A)6 and (A)4, AXIN1 (C)6 (two sequences) and (G)6, AXIN2 (A)6, (G)7, (C)5 (two sequences) and (C)6, PTEN (A)6 (two sequences), MSH6 (C)8, MSH3 (A)8, IGF2R (G)8 and B2M (CT)4 (Supplementary Table 1). The selected microsatellite sequences were analysed by PCR and fragment analysis involves using fluorescence-labelled primers (Supplementary Table 1). Fragment length variations were analysed on an ABI Prism 310 DNA sequencer (Applied Biosystems, Foster City, CA, USA) and allele sizes were determined using the Genemapper software (version 3.7; Applied Biosystems). The results were independently scored by two observers, and an additional round of analyses confirmed the results. Additionally, all cases that presented length variations between tumour and matching blood samples were confirmed by direct sequencing on an ABI 310 DNA sequencer using Big Dye Terminator V1.1 Chemistry (Applied Biosystems), according to the manufacturer's recommendations.
Statistical analysis
Statistical analysis was carried out with SPSS version 22 (IBM, Armonk, NY, USA). Results were expressed in absolute frequencies and percentages. The statistical significance of association between different variables was performed using the Fisher's exact probability test. Mean comparison between two groups was performed using T-test. Analysis of variance (ANOVA) was used for mean comparison between more than two groups and the statistical significance was assessed using the Scheffe's multiple comparison test. P-values <0.05 were considered statistically significant.
Unsupervised hierarchical cluster analysis and heat maps were performed using the software Multi Experiment Viewer MeV version 4.9 (TM 4 group; Dana Farber Cancer Institute, Boston, MA, USA) using a Pearson's correlation distance metric with average linkage clustering.
Results
MMR immunohistochemical analysis
In all tumours from the Porto test series analysed by immunohistochemistry from patients carrying a mutation in the MLH1 gene, MLH1 and PMS2 protein expression was absent; patients with an MSH2 mutation did not show MSH2 and MSH6 protein expression; patients with a mutation in MSH6 gene had no MSH6 protein expression and one case also displayed loss of MSH2 protein expression; and the tumour of the patient with the PMS2 mutation showed loss of PMS2 protein expression. In the additional Swiss series, all the CRCs analysed by immunohistochemistry from patients carrying a mutation in the MLH1 gene, MLH1 and PMS2 protein expression was absent; patients with an MSH2 mutation did not show MSH2 and MSH6 protein expression; and patients with a mutation in MSH6 gene had no MSH6 protein expression.
Overall MSI
The frequency of MSI in the test series from Porto was 99% (77 out of 78) and the only CRC that did not present MSI belonged to a patient carrying a germline mutation in the MSH6 gene. In the additional Switzerland series, all 51 Lynch syndrome CRCs presented MSI.
Target gene mutation frequencies
The microsatellite sequence most frequently mutated in the test series from Porto was ACVR2A (90.9%), followed by TGFBR2 (89.6%), EGFR (88.3%) and BMPR2 (A)11 (75%) sequences. Mutations in the E2F4 (53.3%), MSH3 (49.4%), BAX (44.2%), TCF7L2 (41.6%) and BMPR2 (A)7 (35.1%) microsatellite sequences were also frequent. The remaining genes presented a mutational frequency <30% (Table 2). All the MSI CRCs presented MSI in at least one of the genes analysed and the overall mean mutation frequency was 6.7±2.4.
Table 2. Mutational frequency of the target gene microsatellite sequences according to tumour location in the Lynch syndrome MSI-H test (Porto) series.
| Gene | Total (%) | Proximal colon (%) | Distal colon (%) | P-value |
|---|---|---|---|---|
| ACVR2A | 70/77 (90.9) | 46/51 (90.2) | 24/26 (92.3) | 1 |
| TGFBR2 | 69/77 (89.6) | 46/51 (90.2) | 23/26 (88.5) | 1 |
| EGFR | 68/76a (88.3) | 44/50 (88.0) | 24/26 (92.3) | 0.708 |
| BMPR2 (A)11 | 57/76a (75.0) | 36/50 (72.0) | 21/26 (80.8) | 0.578 |
| E2F4 | 40/75b (53.3) | 30/50 (60.0) | 10/25 (40.0) | 0.141 |
| MSH3 | 38/77 (49.4) | 20/51 (39.2) | 18/26 (69.2) | 0.017 |
| BAX | 34/77 (44.2) | 21/51 (41.2) | 13/26 (50.0) | 0.478 |
| TCF7L2 | 32/77 (41.6) | 19/51 (37.3) | 13/26 (50.0) | 0.333 |
| BMPR2 (A)7 | 27/77 (35.1) | 13/51 (25.5) | 14/26 (53.8) | 0.022 |
| PRDM2 | 22/77 (28.6) | 15/51 (29.4) | 7/26 (26.9) | 1 |
| MSH6 | 19/77 (24.7) | 10/51 (19.6) | 9/26 (34.6) | 0.170 |
| IGF2R | 16/77 (20.8) | 9/51 (17.6) | 7/26 (26.9) | 0.382 |
| B2M | 7/77 (9.1) | 6/51 (11.8) | 1/26 (3.8) | 0.412 |
| APC | 6/77 (7.8) | 6/51 (11.8) | 0/26 (0.0) | 0.091 |
| PTEN | 6/77 (7.8) | 0/51 (0.0) | 6/26 (23.1) | 0.001 |
| AXIN2 | 3/77 (3.9) | 1/51 (2.0) | 2/26 (7.7) | 0.262 |
Abbreviations: CRC=colorectal carcinoma; MSI=microsatellite instability.
We did not detect mutations in BMPR1A microsatellite sequences. P<0.05 are indicated in bold.
One proximal CRC case was not analysed for this gene.
Two cases (one proximal and one distal CRC were not analysed for this gene.
In the additional Switzerland series, we only analysed the target gene sequences showing significant differences between proximal and distal CRCs in the Porto series (see below), namely the MSH3, BMPR2 (A)7 and PTEN microsatellite sequences (Table 2). The mutational frequency in this series was 47.1%, 39.2% and 11.8% for MSH3, BMPR2 and PTEN, respectively, being 48.4% for MSH3, 36.7% for BMPR2 and 9.4% for PTEN in the combined series.
Target gene analysis by germline MMR mutation
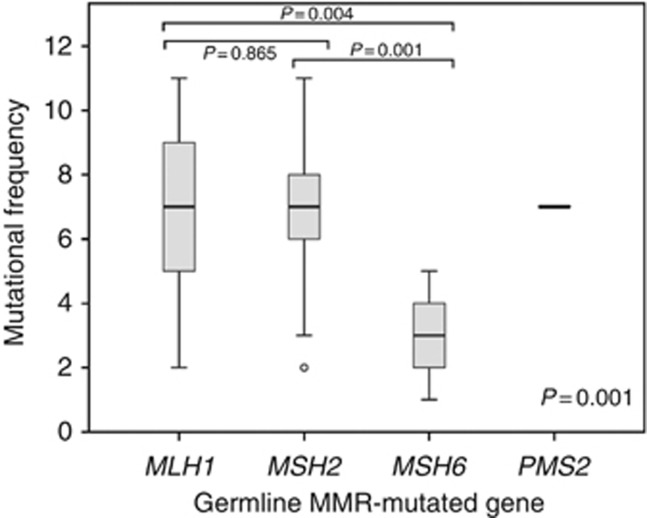
The mean frequency of target gene mutations was 7.1±2.1 in the tumours from patients presenting a germline mutation in MSH2, 6.8±2.5 in MLH1 carriers and 3.0±1.6 in MSH6 carriers, and the single tumour from a PMS2 carrier had seven mutations. To examine whether there were differences among these groups, one-way ANOVA was conducted and statistically significant differences among the groups were found (P=0.001). Post hoc Scheffe's tests revealed statistically significant differences between MLH1 and MSH6 (P=0.004) and MSH2 and MSH6 (P=0.001), but not between MLH1 and MSH2 (P=0.865) (Figure 1).
Figure 1.
Box-plot analyses of the frequency of target gene mutations (Y axis) in CRC samples from the test series categorised by MMR germline mutation (X axis). The mean comparison was calculated using the one-way ANOVA test. Statistical significance among the samples was assessed using the Scheffe's multiple comparison test.
No association was observed between the target gene mutational frequencies and pattern with the germline-mutated gene or the type of mutation, nor when comparing founder mutations to other mutations in the same gene. Unsupervised hierarchical cluster analysis of target gene mutations and MMR gene is shown in Supplementary Figure 1A.
Target gene mutation analysis by tumour location
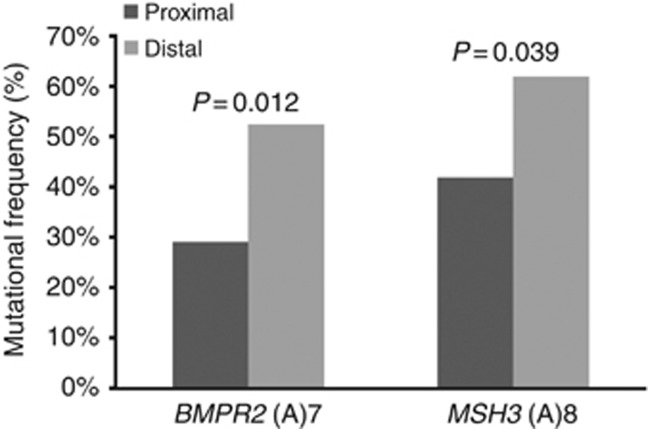
The mean frequency of target gene mutations in proximal and distal colon tumours was 6.3±2.4 and 7.5±2.5, respectively (P=0.06). Three microsatellite sequences were preferentially mutated in distal tumours (Table 2). In the CRC test series from Porto, PTEN, BMPR2 (A)7 and MSH3 mutations were present in 23.1%, 53.8% and 69.2% of the distal MSI carcinomas, whereas in proximal MSI carcinomas mutations were present in 0%, 25.5% and 39.2% of the tumours, respectively (P=0.001 for PTEN, P=0.022 for BMPR2 and P=0.017 for MSH3) (Table 2). In the smaller Lynch syndrome CRC series from Switzerland alone, none of these genes were significantly associated with distal MSI carcinomas, but for the combined series mutations in MSH3 and BMPR2 (A)7, microsatellite sequences remained significantly associated with distal tumour location (P=0.039 for MSH3 and P=0.012 for BMPR2) (Figure 2 and Table 3). Other genes showed different mutation frequencies according to the large bowel site of origin (Table 2), but their lower mutation frequencies require much larger tumour series to evaluate the statistical significance. For instance, APC microsatellite mutations were only detected in the proximal colon, whereas AXIN1 and AXIN2 microsatellite mutations only rarely were found in the proximal CRC but were recurrently detected in the distal CRC. No association was observed between target gene mutational pattern or frequency with other clinicopathologic features, namely gender, mean age of CRC diagnosis, tumour staging or differentiation grade. Unsupervised hierarchical cluster analysis of target gene mutations and tumour location is shown in Supplementary Figure 1B.
Figure 2.
Mutational frequency of the microsatellite sequences (A)7 of BMPR2 and (A)8 of MSH3 according to tumour location in tumours from both series of Lynch syndrome patients.
Table 3. Mutational frequency of MSH3, BMPR2 (A)7 and PTEN microsatellite sequences according to tumour location in the Lynch syndrome MSI-H series of Porto, Switzerland and combined.
| Cases | Gene | Total (%) | Proximal colon (%) | Distal colon (%) | P-value |
|---|---|---|---|---|---|
| Porto | MSH3 | 38/77 (49.4) | 20/51 (39.2) | 18/26 (69.2) | 0.017 |
| BMPR2 (A)7 | 27/77 (35.1) | 13/51 (25.5) | 14/26 (53.8) | 0.022 | |
| PTEN | 6/77 (7.8) | 0/51 (0) | 6/26 (23.1) | 0.001 | |
| Switzerland | MSH3 | 24/51 (47.1) | 16/35 (45.7) | 8/16 (50) | 1.000 |
| BMPR2 (A)7 | 20/51 (39.2) | 12/35 (34.3) | 8/16 (50) | 0.360 | |
| PTEN | 6/51 (11.8) | 6/35 (17.1) | 0/16 (0) | 0.159 | |
| Total | MSH3 | 62/128 (48.4) | 36/86 (41.9) | 26/42 (61.9) | 0.039 |
| BMPR2 (A)7 | 47/128 (36.7) | 25/86 (29.1) | 22/42 (52.4) | 0.012 | |
| PTEN | 12/128 (9.4) | 6/86 (7) | 6/42 (14.3) | 0.206 |
Abbreviation: MSI=microsatellite instability. P<0.05 are indicated in bold.
Target gene mutation comparison between Lynch syndrome and sporadic MSI CRC
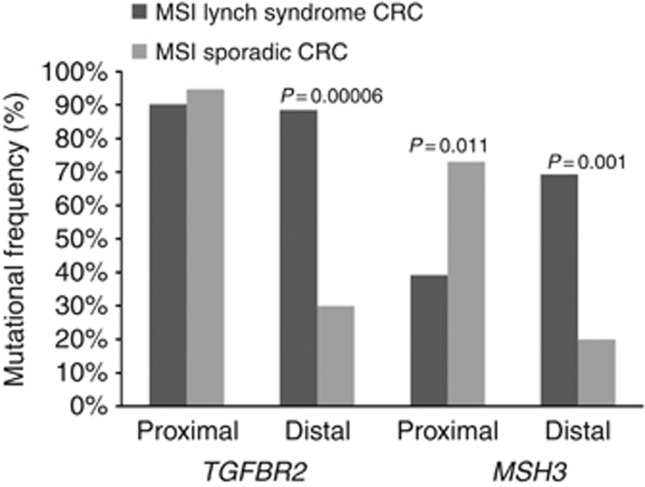
In a previous work, we observed that alterations in MSH3 and TGFBR2 were less frequent in the distal sporadic MSI CRCs (20.0% and 30.0%, respectively) when compared with the proximal tumours (72.7% and 95.5%, respectively) (Pinheiro et al, 2010). Comparing these results with Lynch syndrome MSI CRC, we observed that MSH3 alterations were significantly more frequent in the proximal sporadic MSI CRCs compared with that in proximal Lynch syndrome tumours (72.7% and 39.2%, respectively, P=0.011), with the opposite occurring in the distal CRCs (20.0% and 69.2%, respectively, P=0.001) (Figure 3). TGFBR2 alterations were significantly more frequent in distal Lynch syndrome tumours compared with that in the distal sporadic tumours (88.5% and 30.0%, respectively, P=0.00006), with no difference being observed in the proximal CRCs (Table 4 and Figure 3).
Figure 3.
Mutational frequency in TGFBR2 (A)10 and MSH3 (A)8 microsatellite sequences categorised by tumour location in sporadic (Pinheiro et al, 2010) and Lynch syndrome MSI CRC (present report).
Table 4. Mutational frequency of TGFBR2 and MSH3 microsatellite sequences according to tumour location in Lynch syndrome and sporadic MSI-H carcinomas (Pinheiro et al, 2010).
| Gene | Cases | Total (%) | Proximal colon (%) | P-value | Distal colon (%) | P-value |
|---|---|---|---|---|---|---|
| TGFBR2 | Lynch | 69/77 (89.6) | 46/51 (90.2) | 0.661 | 23/26 (88.5) | 0.00006 |
| Sporadic | 27/42 (64.3) | 21/22 (95.5) | 6/20 (30) | |||
| MSH3 | Lynch | 38/77 (49.4) | 20/51 (39.2) | 0.011 | 18/26 (69.2) | 0.001 |
| Sporadic | 20/42 (47.6) | 16/22 (72.7) | 4/20 (20) |
Abbreviation: MSI=microsatellite instability.
Discussion
Microsatellite instability is a hallmark of CRC in Lynch syndrome patients, occurring in >95% of the tumours (Aaltonen et al, 1994). The frequency of MSI detected in our study indicates that both series are representative of Lynch syndrome CRC. There is a well-established association between an ineffective MMR system and mutations in genes with key cellular roles in CRC of Lynch syndrome patients. With the purpose of comparing the mutation pattern of target genes in MSI CRC from Lynch syndrome patients stratified by tumour location and germline mutation type, we analysed several genes predicted to be selective target genes of MSI and known to be involved in several pathways of colorectal carcinogenesis. All candidate genes except BMPR1A presented frameshift mutations in the microsatellite sequences analysed. Colorectal carcinoma from patients with a germline mutation in MSH6 presented a significantly lower frequency of target gene mutations when compared with the groups with MLH1 or MSH2 germline mutations, whereas no differences were observed regarding target gene mutation frequency or pattern between CRC from carriers of MLH1 or MSH2 founder mutations and other mutations in these genes. Wu et al (1999) had already suggested that MSH6 may be involved in a proportion of Lynch syndrome patients presenting MSI-low tumours. More recently, Laghi et al (2012) also observed that the mutational rate in specific target genes was significantly lower in MSH6 compared with that in MLH1- and MSH2-deficient tumours.
The most frequently mutated genes, TGFBR2 and ACVR2A, encode for proteins belonging to the TGF-β superfamily, supporting the idea that alterations in these genes are important for the development of MSI CRC in the context of Lynch syndrome (Fujiwara et al, 1998; Miyaki et al, 2001). Additionally, we also observed a high frequency of mutations in the microsatellite sequences of BMPR2 (75% and 35.1% in (A)11 and (A)7, respectively). Kodach et al (2008) also found a high mutational frequency (81.4%) in the (A)11 microsatellite sequence in sporadic MMR-deficient tumours and indicated that loss of BMPR2 expression is associated with MSI in this sequence. Regarding the (A)7 microsatellite sequence, they detected instability in two MSI cell lines and none in the MMR-deficient tumours, whereas none of the MSS cell lines and MMR-proficient tumours analysed presented mutations in either microsatellite sequences (Kodach et al, 2008). Furthermore, Park et al (2010) detected mutations in BMPR2 (A)7 sequence in 13.2% of sporadic CRCs associated with loss of BMPR2 expression. Further studies are needed to clarify the role of the (A)7 tract mutations in BMPR2 function and expression, given that almost all cases presented in concomitance alterations in the more commonly mutated (A)11 tract. Kodach et al (2008) also observed that alterations in BMPR2 occurred in concomitance with TGFBR2 and ACVR2A mutations, suggesting that loss of one of these receptors is insufficient for complete pathway disruption. Some studies indicate that alterations in the WNT pathway may have an important role in the tumorigenesis of MMR deficiency tumours (Miyaki et al, 1999; Thorstensen et al, 2005). We detected a low frequency of mutations in APC, AXIN1, AXIN2 and PTEN, but a high frequency in TCF7L2. Mutations in TCF7L2 have been reported with a high frequency (33–58%) in Lynch syndrome CRC (Duval et al 1999; Ruckert et al, 2002; Yamaguchi et al, 2006). Mutations in a polymorphic (A)13 out of 14 repeat within the 3′-UTR of EGFR were also described in a high percentage (59–81%) of sporadic MSI colon cancers, but there is no consensus if these are oncogenic mutations (Deqin et al, 2012; Sarafan-Vasseur et al, 2013). This is the first report showing a high frequency of this type of EGFR mutations (88.3%) in Lynch syndrome CRC. The high mutation frequency observed in BMPR2, TCF7L2 and EGFR could be an indicator of selective advantage, but some of these mutations may be bystander events that do not have a causal role in carcinogenesis. Additional functional studies are needed to clarify if they are true target genes in the MSI pathway of colorectal carcinogenesis, especially in the context of Lynch syndrome.
A substantial frequency of somatic mutations in MSH3 and MSH6 microsatellite sequences have been described in CRC from patients with Lynch syndrome (Akiyama et al, 1997; Yamamoto et al, 1998). These mutations are considered secondary events resulting from a germline MMR gene deficiency and are defined as ‘secondary' mutators in a ‘mutator that mutates another mutator' model (Akiyama et al, 1997; Yamamoto et al, 1998). However, the pathogenetic consequence of these somatic mutations in MMR genes is unclear and may be just a marker of generalised MSI. In fact, in addition to the necessity of biallelic mutations for a biological effect, it is well established that CRC with germline MSH2 mutations are usually associated with loss of expression of MSH6, thus somatic MSH6 mutations would not confer an additional selective advantage. Genes involved in cell cycle control and apoptosis were also found to be frequently mutated in Lynch syndrome CRC, namely E2F4, BAX and PRDM2, indicating that these are target genes of genetic instability in Lynch syndrome CRC (Miyaki et al, 2001; Moriyama et al, 2002; Yamaguchi et al, 2006).
Regarding tumour site, we observed that mutations in microsatellite sequences (A)7 of BMPR2, (A)8 of MSH3 and PTEN were significantly more frequent in the distal CRC in the Porto series. Although these associations were not clear when only the Switzerland series is considered, which could be because of the lower number of CRC samples in that series or to other factors related to a different genetic background or environmental causes, the analysis of the combined series confirmed those findings for MSH3 and BMPR2 (A)7, indicating that these genes are preferentially involved in the development or progression of distal colon cancer in Lynch syndrome patients. It is unclear why the tumour site difference was observed for BMPR2 (A7) but not BMPR2 (A11), especially in light of their frequent co-occurrence, being possible that mutations in the (A)11 microsatellite sequence could be just a marker of generalised instability without a functional consequence to the BMPR2 gene. Interestingly, the results concerning MSH3 are different from the ones we observed in a previous study of sporadic MSI tumours, where alterations in MSH3 were less frequent in tumours of the distal colon (Pinheiro et al, 2010). Furthermore, the significantly lower frequency of TGFBR2 mutations we previously observed in the distal sporadic MSI CRCs (Pinheiro et al, 2010) was not seen in the present study with Lynch syndrome MSI CRCs. Qualitative (type of target gene) and quantitative (number and frequency of altered target genes) differences have been observed regarding MSI target genes in different types of cancers. For instance, significant differences have been described between the MSI profiles of endometrial and colorectal cancers from Lynch syndrome patients presenting the same germline mutation. For example, somatic mutations in TCF7L2 were observed in ∼47% of Lynch syndrome CRC, but not in endometrial cancers (Planck et al, 2000). Similarly, TGFBR2 mutational frequency is higher in MSI colon carcinomas (70–90%) compared with that in MSI endometrial carcinoma (17–19%), whereas PTEN instability was more frequently observed (∼20%) in endometrial carcinomas compared with MSI CRCs (∼5%), suggesting that biological features and functional roles of target genes may differ depending on the tissue of origin (Lu et al, 1995; Myeroff et al, 1995; Kuismanen et al, 2002). Our data suggest that target genes differ in colorectal carcinomas depending on large bowel site of origin and between Lynch syndrome and sporadic MSI CRC, suggesting that carcinogenesis can occur by different routes even if driven by generalised MSI (Dierssen et al, 2007). Significant differences in the spectrum of molecular alterations between MSI Lynch syndrome and sporadic CRC have been observed previously. BRAF mutations, namely p.Val600Glu, have been identified in sporadic MSI CRC associated with MLH1 promoter hypermethylation but not in Lynch syndrome CRC (Domingo et al, 2004). Conversely, CTNNB1 gene mutations are frequent (43%) in Lynch syndrome tumours but not in sporadic MSI CRCs (Miyaki et al, 1999). The differences in the molecular profiles of the two pathways are consistent with the differing carcinogenesis routes of hereditary vs sporadic MSI CRCs, specifically the traditional adenoma–carcinoma sequence in the former and a serrated pathway in the latter (Jass, 2007). Furthermore, the different embryological origin of proximal and distal bowel may contribute to the molecular heterogeneity of MSI CRC, similarly to what happens during carcinogenesis of different organs in Lynch syndrome patients.
Acknowledgments
This study was supported by CHIBCHA and Projecto Mobilizador No. 13853 – DO IT. MP is a research fellow of Fundação para a Ciência e a Tecnologia (FCT) (SFRH/BD/65326/2009).
The authors declare no conflict of interest.
Footnotes
Supplementary Information accompanies this paper on British Journal of Cancer website (http://www.nature.com/bjc)
This work is published under the standard license to publish agreement. After 12 months the work will become freely available and the license terms will switch to a Creative Commons Attribution-NonCommercial-Share Alike 4.0 Unported License.
Supplementary Material
References
- Aaltonen LA, Peltomäki P, Mecklin JP, Järvinen H, Jass JR, Green JS, Lynch HT, Watson P, Tallqvist G, Juhola M, Sistonen P, Hamilton SR, Kinzler KW, Vogelstein B, de la Chapelle A (1994) Replication errors in benign and malignant tumors from hereditary nonpolyposis colorectal cancer patients. Cancer Res 54: 1645–1648. [PubMed] [Google Scholar]
- Aaltonen LA, Salovaara R, Kristo P, Canzian F, Hemminki A, Peltomäki P, Chadwick RB, Kääriäinen H, Eskelinen M, Järvinen H, Mecklin JP, de la Chapelle A (1998) Incidence of hereditary nonpolyposis colorectal cancer and the feasibility of molecular screening for the disease. N Engl J Med 338: 1481–1487. [DOI] [PubMed] [Google Scholar]
- Akiyama Y, Tsubouchi N, Yuasa Y (1997) Frequent somatic mutations of hMSH3 with reference to microsatellite instability in hereditary nonpolyposis colorectal cancers. Biochem Biophys Res Commun 236: 248–252. [DOI] [PubMed] [Google Scholar]
- Deqin M, Chen Z, Nero C, Patel KP, Daoud EM, Cheng H, Djordjevic B, Broaddus RR, Medeiros LJ, Rashid A, Luthra R (2012) Somatic deletions of the polyA tract in the 3' untranslated region of epidermal growth factor receptor are common in microsatellite instability-high endometrial and colorectal carcinomas. Arch Pathol Lab Med 136: 510–516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dierssen JW, de Miranda NF, Ferrone S, van Puijenbroek M, Cornelisse CJ, Fleuren GJ, van Wezel T, Morreau H (2007) HNPCC vs sporadic microsatellite-unstable colon cancers follow different routes toward loss of HLA class I expression. BMC Cancer 7: 33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Domingo E, Espín E, Armengol M, Oliveira C, Pinto M, Duval A, Brennetot C, Seruca R, Hamelin R, Yamamoto H, Schwartz S Jr (2004) Activated BRAF targets proximal colon tumors with mismatch repair deficiency and MLH1 inactivation. Genes Chromosomes Cancer 39: 138–142. [DOI] [PubMed] [Google Scholar]
- Duval A, Gayet J, Zhou XP, Iacopetta B, Thomas G, Hamelin R (1999) Frequent frameshift mutations of the TCF-4 gene in colorectal cancers with microsatellite instability. Cancer Res 59(17): 4213–4215. [PubMed] [Google Scholar]
- Fujiwara T, Stolker JM, Watanabe T, Rashid A, Longo P, Eshleman JR, Booker S, Lynch HT, Jass JR, Green JS, Kim H, Jen J, Vogelstein B, Hamilton SR (1998) Accumulated clonal genetic alterations in familial and sporadic colorectal carcinomas with widespread instability in microsatellite sequences. Am J Pathol 153: 1063–1078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hampel H, Frankel WL, Martin E, Arnold M, Khanduja K, Kuebler P, Clendenning M, Sotamaa K, Prior T, Westman JA, Panescu J, Fix D, Lockman J, LaJeunesse J, Comeras I, de la Chapelle A (2008) Feasibility of screening for Lynch syndrome among patients with colorectal cancer. J Clin Oncol 26: 5783–5788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jass JR (2007) Classification of colorectal cancer based on correlation of clinical, morphological and molecular features. Histopathology 50: 113–130. [DOI] [PubMed] [Google Scholar]
- Kodach LL, Wiercinska E, de Miranda NF, Bleuming SA, Musler AR, Peppelenbosch MP, Dekker E, van den Brink GR, van Noesel CJ, Morreau H, Hommes DW, Ten Dijke P, Offerhaus GJ, Hardwick JC (2008) The bone morphogenetic protein pathway is inactivated in the majority of sporadic colorectal cancers. Gastroenterology 134: 1332–1341. [DOI] [PubMed] [Google Scholar]
- Kovac M, Laczko E, Haider R, Jiricny J, Mueller H, Heinimann K, Marra G (2011) Familial colorectal cancer: eleven years of data from a registry program in Switzerland. Fam Cancer 10: 605–616. [DOI] [PubMed] [Google Scholar]
- Kuismanen SA, Moisio AL, Schweizer P, Truninger K, Salovaara R, Arola J, Butzow R, Jiricny J, Nyström-Lahti M, Peltomäki P (2002) Endometrial and colorectal tumors from patients with hereditary nonpolyposis colon cancer display different patterns of microsatellite instability. Am J Pathol 160: 1953–1958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lagerstedt Robinson K, Liu T, Vandrovcova J, Halvarsson B, Clendenning M, Frebourg T, Papadopoulos N, Kinzler KW, Vogelstein B, Peltomäki P, Kolodner RD, Nilbert M, Lindblom A (2007) Lynch syndrome (hereditary nonpolyposis colorectal cancer) diagnostics. J Natl Cancer Inst 99: 291–299. [DOI] [PubMed] [Google Scholar]
- Laghi L, Bianchi P, Delconte G, Celesti G, Di Caro G, Pedroni M, Chiaravalli AM, Jung B, Capella C, de Leon MP, Malesci A (2012) MSH3 protein expression and nodal status in MLH1-deficient colorectal cancers. Clin Cancer Res 18: 3142–3353. [DOI] [PubMed] [Google Scholar]
- Lu SL, Akiyama Y, Nagasaki H, Saitoh K, Yuasa Y (1995) Mutations of the transforming growth factor-beta type II receptor gene and genomic instability in hereditary nonpolyposis colorectal cancer. Biochem Biophys Res Commun 216: 452–457. [DOI] [PubMed] [Google Scholar]
- Lynch HT, de la Chapelle A (2003) Hereditary colorectal cancer. N Engl J Med 348: 919–932. [DOI] [PubMed] [Google Scholar]
- Miyaki M, Iijima T, Kimura J, Yasuno M, Mori T, Hayashi Y, Koike M, Shitara N, Iwama T, Kuroki T (1999) Frequent mutation of beta-catenin and APC genes in primary colorectal tumors from patients with hereditary nonpolyposis colorectal cancer. Cancer Res 59: 4506–4509. [PubMed] [Google Scholar]
- Miyaki M, Iijima T, Shiba K, Aki T, Kita Y, Yasuno M, Mori T, Kuroki T, Iwama T (2001) Alterations of repeated sequences in 5' upstream and coding regions in colorectal tumors from patients with hereditary nonpolyposis colorectal cancer and Turcot syndrome. Oncogene 20: 5215–5218. [DOI] [PubMed] [Google Scholar]
- Moriyama H, Sasamoto H, Kambara T, Matsubara N, Ikeda M, Baba S, Meltzer SJ, Lynch HT, Shimizu K, Tanaka N (2002) E2F-4 mutation in hereditary non-polyposis colorectal cancer. J Exp Clin Cancer Res 21: 185–189. [PubMed] [Google Scholar]
- Myeroff LL, Parsons R, Kim SJ, Hedrick L, Cho KR, Orth K, Mathis M, Kinzler KW, Lutterbaugh J, Park K, Bang Y, Lee HY, Park JG, Lynch HT, Roberts AB, Vogelstein B, Markowitz SD (1995) A transforming growth factor beta receptor type II gene mutation common in colon and gastric but rare in endometrial cancers with microsatellite instability. Cancer Res 55: 5545–5547. [PubMed] [Google Scholar]
- Park SW, Hur SY, Yoo NJ, Lee SH (2010) Somatic frameshift mutations of bone morphogenic protein receptor 2 gene in gastric and colorectal cancers with microsatellite instability. APMIS 118: 824–829. [DOI] [PubMed] [Google Scholar]
- Perucho M (1996) Microsatellite instability: the mutator that mutates the other mutator. Nat Med 2: 630–631. [DOI] [PubMed] [Google Scholar]
- Pinheiro M, Ahlquist T, Danielsen SA, Lind GE, Veiga I, Pinto C, Costa V, Afonso L, Sousa O, Fragoso M, Santos L, Henrique R, Lopes P, Lopes C, Lothe RA, Teixeira MR (2010) Colorectal carcinomas with microsatellite instability display a different pattern of target gene mutations according to large bowel site of origin. BMC Cancer 10: 587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pinheiro M, Pinto C, Peixoto A, Veiga I, Mesquita B, Henrique R, Baptista M, Fragoso M, Sousa O, Pereira H, Marinho C, Moreira Dias L, Teixeira MR (2011) A novel exonic rearrangement affecting MLH1 and the contiguous LRRFIP2 is a founder mutation in Portuguese Lynch syndrome families. Genet Med 13: 895–902. [DOI] [PubMed] [Google Scholar]
- Pinheiro M, Pinto C, Peixoto A, Veiga I, Mesquita B, Henrique R, Lopes P, Sousa O, Fragoso M, Dias LM, Baptista M, Marinho C, Mangold E, Vaccaro C, Evans DG, Farrington S, Dunlop MG, Teixeira MR (2013) The MSH2 c.388_389del mutation shows a founder effect in Portuguese Lynch syndrome families. Clin Genet 84: 244–250. [DOI] [PubMed] [Google Scholar]
- Pinheiro M, Veiga I, Pinto C, Afonso L, Sousa O, Fragoso M, Santos L, Lopes P, Pais I, Lopes C, Teixeira MR (2009) Mitochondrial genome alterations in rectal and sigmoid carcinomas. Cancer Lett 280: 38–43. [DOI] [PubMed] [Google Scholar]
- Planck M, Wenngren E, Borg A, Olsson H, Nilbert M (2000) Somatic frameshift alterations in mononucleotide repeat-containing genes in different tumor types from an HNPCC family with germline MSH2 mutation. Genes Chromosomes Cancer 29: 33–39. [DOI] [PubMed] [Google Scholar]
- Ruckert S, Hiendlmeyer E, Brueckl WM, Oswald U, Beyser K, Dietmaier W, Haynl A, Koch C, Rüschoff J, Brabletz T, Kirchner T, Jung A (2002) T-cell factor-4 frameshift mutations occur frequently in human microsatellite instability-high colorectal carcinomas but do not contribute to carcinogenesis. Cancer Res 62: 3009–3013. [PubMed] [Google Scholar]
- Sarafan-Vasseur N, Sefrioui D, Tougeron D, Lamy A, Blanchard F, Le Pessot F, Di Fiore F, Michel P, Bézieau S, Latouche JB, Frebourg T, Sesboüé R (2013) Genetic variations of the A13/A14 repeat located within the EGFR 3' untranslated region have no oncogenic effect in patients with colorectal cancer. BMC Cancer 13: 183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thorstensen L, Lind GE, Løvig T, Diep CB, Meling GI, Rognum TO, Lothe RA (2005) Genetic and epigenetic changes of components affecting the WNT pathway in colorectal carcinomas stratified by microsatellite instability. Neoplasia 7: 99–108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Umar A, Boland CR, Terdiman JP, Syngal S, de la Chapelle A, Rüschoff J, Fishel R, Lindor NM, Burgart LJ, Hamelin R, Hamilton SR, Hiatt RA, Jass J, Lindblom A, Lynch HT, Peltomaki P, Ramsey SD, Rodriguez-Bigas MA, Vasen HF, Hawk ET, Barrett JC, Freedman AN, Srivastava S (2004) Revised Bethesda Guidelines for hereditary nonpolyposis colorectal cancer (Lynch syndrome) and microsatellite instability. J Natl Cancer Inst 96: 261–268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vasen HF, Watson P, Mecklin JP, Lynch HT (1999) New clinical criteria for hereditary nonpolyposis colorectal cancer (HNPCC, Lynch syndrome) proposed by the International Collaborative group on HNPCC. Gastroenterology 116: 1453–1456. [DOI] [PubMed] [Google Scholar]
- Wu Y, Berends MJ, Mensink RG, Kempinga C, Sijmons RH, van Der Zee AG, Hollema H, Kleibeuker JH, Buys CH, Hofstra RM (1999) Association of hereditary nonpolyposis colorectal cancer-related tumors displaying low microsatellite instability with MSH6 germline mutations. Am J Hum Genet 65: 1291–1298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamaguchi T, Iijima T, Mori T, Takahashi K, Matsumoto H, Miyamoto H, Hishima T, Miyaki M (2006) Accumulation profile of frameshift mutations during development and progression of colorectal cancer from patients with hereditary nonpolyposis colorectal cancer. Dis Colon Rectum 49: 399–406. [DOI] [PubMed] [Google Scholar]
- Yamamoto H, Sawai H, Weber TK, Rodriguez-Bigas MA, Perucho M (1998) Somatic frameshift mutations in DNA mismatch repair and proapoptosis genes in hereditary nonpolyposis colorectal cancer. Cancer Res 58: 997–1003. [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.