Abstract
Aims
Nicotinamide adenine dinucleotide oxidases (NOXs) are important contributors to cellular oxidative stress in the cardiovascular system. The NOX2 isoform is upregulated in numerous disorders, including dystrophic cardiomyopathy, where it drives the progression of the disease. However, mechanisms underlying NOX2 overexpression are still unknown. We investigated the role of microRNAs (miRs) in the regulation of NOX2 expression.
Methods and results
Duchenne muscular dystrophy (DMD) was used as a model of cardiomyopathy. After screening with miRNA target prediction databases and following qRT-PCR analysis, we found drastic downregulation of miR-448-3p in hearts of mdx mice, an animal model of DMD. The downregulation correlated with overexpression of the Ncf1 gene, encoding the NOX2 regulatory subunit p47phox. Specificity of Ncf1 targeting by miR-448-3p was validated by luciferase reporter assay. Silencing of miR-448-3p in wild-type mice had a dramatic effect on cellular and functional properties of cardiac muscle as assessed by western blotting, qRT-PCR, confocal imaging, echocardiography, and histology. Acute treatment of mice with LNA-miR-448 inhibitors led to increased Ncf1 expression, abnormally elevated reactive oxygen species (ROS) production and exacerbated Ca2+ signalling in cardiomyocytes, reminiscent of features previously observed in dystrophic cardiac cells. In addition, chronic inhibition of miR-448-3p resulted in dilated cardiomyopathy and arrhythmia, hallmarks of dystrophic cardiomyopathy.
Conclusions
Our studies suggest that downregulation of miR-448-3p leads to the increase in the expression of Ncf1 gene and p47phox protein, as well as to the substantial increase in NOX2-derived ROS production. Cellular oxidative stress subsequently triggers events that finally culminate in cardiac tissue damage and development of cardiomyopathy.
Keywords: Cardiomyopathy, Oxidative stress, NOX, microRNA, Dystrophin
1. Introduction
Oxidative stress is implicated in the development of numerous cardiovascular pathologies, such as cardiac hypertrophy (CH), heart failure (HF), myocardial infarction (MI), hypertension, atherosclerosis, and stroke (reviewed in1–3). It is a result of excessive generation of reactive oxygen species (ROS) as well as of reduced antioxidant capacity in the vasculature and heart. ROS are produced by most of cardiovascular cell types, including endothelial and smooth muscle cells as well as cardiomyocytes. They are generated by various sources: enzymes, such as xanthine oxidize and nicotinamide adenine dinucleotide oxidase (NOX), and mitochondria. It is currently believed that ROS action highly depends on their source, cellular location, and type of species that are generated. Among different sources of ROS, the NOX family of enzymes is particularly important for redox signalling in the physiology and pathophysiology of the heart. NOX2 and NOX4 are two major isoforms expressed in cardiac tissue.4 Increased levels of NOX2 and/or its activity have been shown to be involved in ROS-dependent hypertrophic cardiac responses, myocardial inflammation and fibrosis, post-MI remodelling as well as in initiating and maintaining atrial fibrillation.2
Recent studies demonstrated that NOX2-driven oxidative stress is involved in the pathology of Duchenne muscular dystrophy (DMD).4,5 DMD is a fatal rapidly developing muscle wasting disease caused by an X-linked mutation in the dystrophin gene. Cardiac complications of the disease develop in the majority of boys with DMD. About 20% (and increasing) of adolescent patients develop ventricular dysfunctions and arrhythmias that ultimately lead to HF.2,6,7 The cellular pathophysiology of the heart of mdx mouse (an animal model of DMD) includes oxidative stress and impaired Ca2+ signalling.8–16 These pathophysiological features are observed at very early stages of the disease, well before the first sign of cardiomyopathy is apparent.17 Our recent report suggests that oxidation of the intracellular environment initiates post-translational modifications of Ca2+ release channels (ryanodine receptors, RyR) in sarcoplasmic reticulum (SR) leading to the increased sensitivity of RyR to activation by Ca2+.17 The latter results in intensified Ca2+ signalling, activation of apoptotic and necrotic pathways and consequently, the loss-of-functional myocytes and development of fibrosis.5 The major source of oxidative stress in young mdx hearts is NOX2. NOX2 expression and NOX2-derived ROS production is significantly increased in dystrophy, and this clearly precedes the first clinical manifestation of dystrophic cardiomyopathy.11,17 Therefore, oxidative stress and NOX2 are considered to be appealing targets in developing new therapies for muscular dystrophy as well as for some other cardiac diseases mentioned above.
As of today, overall outcomes of clinical studies investigating effects of antioxidants have been disappointing, despite the promising results of the same experiments with research animals. Therefore, the employment of new approaches is a necessary and important step in a therapeutic design. One of the possibilities is to regulate the expression of the gene of interest. microRNAs (miRs) is a group of endogenous, short non-coding RNAs.18 These molecules play a critical role in translational and posttranscriptional regulation of gene expression. They finely tune the output of target messenger RNAs (mRNAs) by their destabilization and/or translational inhibition.19 A number of miRs are primarily expressed in skeletal and cardiac muscles (miR-1, miR-21, miR-133, miR-208, etc.). These housekeeping molecules play important roles in muscle development, regeneration, myogenesis, electrical remodelling, progression of fibrosis, etc.20,21 Other miRs are expressed in other tissues, but can still affect distant targets, including muscle, by circulating in the blood stream.22 There are limited reports on the role of miRs in muscle dystrophy. They are either related to the skeletal muscle phenotype of the disease or restricted to the characterization of the miRs expression profile.23,24 To the best of our knowledge, no studies linking oxidative stress in dystrophy to the dysregulation of miRs were reported. Moreover, there is very little known about regulation of NOX2 expression by miRs.
The aim of this study was to identify miR(s) that regulate NOX2 expression in cardiac dystrophy at preclinical stages of the disease. In addition, we wanted to determine if experimental manipulation of these miR(s) in control wild-type (WT) mice induces a dystrophic cardiac phenotype and leads to the deterioration of cardiac function at the molecular, cellular, and organ levels. NOX2 contains a membrane-bound core, comprising a catalytic gp91phox subunit and a p22phox subunit, as well as four regulatory subunits: p67phox, p40phox, p47phox, and Rac1.2,4 The regulatory subunits must translocate from the cytosol to the membrane to form an active enzyme. We found a 10-fold downregulation of miR-448-3p in hearts of young dystrophic mice. The latter correlated with overexpression of the Ncf1 gene, encoding the NOX2 regulatory subunit p47phox. Acute inhibition of miR-448-3p in WT mice with LNA-miR-448-3p inhibitors resulted in an increase in Ncf1 expression as well as in enhanced ROS production and exacerbated intracellular Ca2+ signalling in isolated cardiomyocytes. In addition, chronic (over 1 month) inhibition of miR-448 led to the deterioration of cardiac contractility and development of arrhythmia. Our data suggest that downregulation of miR-448-3p relieves inhibition of Ncf1 translation in dystrophic cardiomyopathy. It results in an increase in Ncf1 expression and consequently in oxidative stress and enhanced Ca2+ signalling in dystrophic heart, cellular events underlying muscle damage. The results of our studies can also be applied to many other cardiac diseases associated with NOX2-derived ROS-driven oxidative stress. Overall, our data point to miR-448-3p as a potential therapeutic target in various cardiac pathologies.
2. Materials and methods
All experiments conformed to the NIH Guide for the Care and Use of Laboratory Animals published by the US National Institute of Health (NIH publication, 8th edition, 2011) and were approved by the Institutional Animal Care and Use Committee of the New Jersey Medical School, Rutgers University, USA. Brief protocol descriptions are below, and all methods are described in details in Supplementary material online.
2.1. Cell isolation
C57BL10 mice (WT) and dystrophin-deficient mdx (C57BL/10ScSnmdx) mice at the age of 1 month (young), 3–4 months (adults), and at least 15 months old (senescent) were used in this study. Animals were purchased from the Jackson Laboratory. Ventricular myocytes from mouse heart were isolated as previously described.25
2.2. RNA purification and real-time reverse transcription PCR
Total RNA was isolated using Tri-Reagent® and Direct-zol RNA MiniPrep (Zymo Research, Irvine, CA, USA) and quantified using Nanodrop ND-1000 Spectrophotometer (Nanodrop Technologies, Wilmington, DE, USA). Real-time PCR was performed using 7500 Fast Real-Time PCR system (Applied Biosystems) and different detection assays.
2.3. Western blots
Protein extractions and western blotting were performed as previously described.17
2.4. Luciferase reporter assay
HEK293 cells were seeded in 96-well plates. Ncf1 3′-UTR, mutated Ncf1 3′-UTR, or control luciferase reporter plasmid (GeneCopoeia) was co-transfected with either mirVanaTM miR mimic hsa-miR-448 or mirVanaTM miR mimic negative control (NC; Ambion) using FugeneHD:DNA ratio of 3:1 (Promega), or Lipofectamine 3000 (Thermofisher, MA, USA). Luciferase activity was measured with Secre-PairTM Dual-Luciferase Reporter Assay (GeneCopoeia).
2.5. LNA-antimiR inhibitors
The miRCURY LNATM miR inhibitors were purchased from Exiqon (Exiqon, Woburn, MA, USA) and used as described in Supplementary material online.
2.6. Confocal imaging
Changes in cytoplasmic [Ca2+] and ROS production were monitored with fluorescent indicators fluo-4 AM and CM-H2DCFDA, respectively.
2.7. Histological analysis
Histological analysis was performed on heart samples fixed in 10% buffered formalin and embedded in paraffin as described in Supplementary material online.
2.8. Cytokine assays
Proteome profiler mouse cytokine array panel A (R&D systems, MN, USA, cat #ARY006) was used to measure the levels of selective chemokine and cytokines in samples.
2.9. Echocardiography
Echocardiography was performed on avertin (2.5% tribromethanol) anaesthetized mice (290 mg/kg, IP) with Acuson Sequoia C256 Ultrasound System.
2.10. Data analysis and statistics
Results are shown as mean ± standard error of mean, unless otherwise specified. All biochemical and functional data sets contain results from 3 to 6 mice. Cellular studies were carried out on cells isolated from at least three mice. The variable N indicates the number of animals used, and n specifies the number of cells studied. Pairwise comparisons between two groups were made by unpaired two-tailed Student’s t-test. P-value of <0.05 and less was considered to be significant. Statistical analyses were performed with the Sigma Plot 13 software (Systat Software Inc., San Jose, CA, USA). Image processing and analyses were done with ImageJ software (public domain software developed by NIH).
3. Results
3.1. Overexpression of NOX2 is associated with oxidative stress in dystrophic heart
We have already demonstrated that NOX2-derived ROS production is enhanced in dystrophic heart well before cardiac myopathy is detected.17 In particular, using fluorescence ROS-sensitive indicator CM-H2DCFDA, we have shown that the extent of ROS production in ventricular cardiomyocytes isolated from young (1 month old) mdx animals is significantly greater than in cells from WT hearts.6,17 The slope of change in DCF fluorescence (the fluorescent derivative of CM-H2DCFDA) detected with confocal imaging reflects the rate of ROS production. In agreement with our previous report, Figure 1A shows that the slope was significantly greater in mdx myocytes compared with that in WT cells. Here we also demonstrate that this difference is completely eliminated by the DPI (diphenylene iodonium, 10 µM), inhibitor of membrane-bound gp91phox subunit of NOX2 complex. Please note that ROS generation in mdx myocytes after incubation with DPI was reduced to levels even lower than that of WT cells. This suggests that NOX2 is not only the major contributor to oxidative stress in young dystrophic hearts but it also contributes to cytosolic redox potential in WT cells. In addition, we previously reported a similar reduction in ROS production by apocynin,10,17 a drug which has different pharmacological mechanism of action. Apocynin inhibits translocation of the regulatory p47phox subunit of NOX2 to the membrane.26–28
Figure 1.
Increased level of NOX2 results in oxidative stress in dystrophic heart. (A) Representative images of DCF fluorescence in cardiomyocytes isolated from young WT and mdx mice. Graph in the middle illustrates time changes in the average DCF signals. Bar graph on the right shows the rate of DCF oxidation (slope) in WT cells (N = 3, n = 9) and mdx without (N = 3, n = 12) and with (N = 3, n = 8) incubation with DPI (10 µM). (B) Quantitative real-time PCR analysis of mRNA of Cybb, Ncf1, Cyba, Ncf4, Ncf2, and Rac1 genes in cardiac tissues of young, adult, and senescent WT and mdx mice. N = 5–6. (C) Western blot analysis indicates increase in expression of gp91phox and p47phox subunits in dystrophic heart compared with WT cardiac tissue, N = 5. *P < 0.05, **P < 0.001, t-test.
Next, we quantified mRNA levels of genes encoding all six subunits of NOX2 with qPCR. Cybb gene encoding gp91phox subunit, as well as Ncf1 (p47phox), Cyba (p22phox), Ncf2 (p67phox), Ncf4 (p40phox), and Rac1 genes in young (1 month old), adult (3–4 months old), and senescent (15–16 months old) both in WT and mdx cardiac tissue were tested (Figure 1B). We found that mRNA expression of Cybb and Ncf1 genes significantly increased in mdx cardiac tissue starting from early age (1 month old), whereas levels of Cyba, Ncf2, Ncf4, and Rac1 mRNA were essentially unchanged. In addition, western blot analysis showed that gp91phox and regulatory p47phox subunits proteins are overexpressed in hearts of young mdx mice compared with WT (Figure 1C).
3.2. Upregulation of Ncf1 gene in heart tissue is mediated by miR-448
Initial screening of miR using the mouse miRNome Sanger miRBase miR Profiler Set (System Bioscience, CA, USA) revealed significant changes in miR expression in hearts of mdx mice of different ages compared with WT. In young hearts, for example, 65% of miRs were downregulated, 4% were upregulated, and 31% did not change (Supplementary material online, Figure S1). Based on this screening and results of miR target prediction databases (miRWalk, miRBase, TargetScan, and miRanda), we selected 12 miRs with predicted targeting of Cybb (miR-9, miR-92, miR-224, miR-17, and miR-138) and Ncf1 (miR-17, miR-138, and miR-448) in addition to miRs shown to be associated with cardiac dysfunctions (miR-1, miR-21, miR-23, miR-29, and miR-320). As expected, some of the cardiac-specific miRs (in particular miR-1 and miR-21) were increased, especially in adult hearts already displaying cardiac myopathy (Figure 2A).
Figure 2.
Changes in miRs expression in dystrophic heart. (A) Real-time PCR analysis of cardio-specific miRs and miRs predicted to target Cybb and Ncf1 genes. Graphs show changes in miR levels in mdx hearts relative to their levels in WT samples (indicated by dashed line). N = 5–6, *P < 0.05, t-test. (B) Genomic localization of mmu-miR-448 on intron 4 of the Htr2C gene located on X-chromosome of mouse genome. It is further processed to pre-miR-448 and later to the mature miR-448, where miR-448-3p is conserved among species. (C) Left, schematic representation of miR-448-3p binding region (‘seed region’) in mouse Ncf1 gene. On the bottom is the map of quadruple mutant (mutated residuals is in light grey). Right, luciferase activity assay shows the dose-dependent decrease of luciferase activity in cells transfected with miR-448 mimic and NCF1-3′UTR plasmid, but not with construct with the mutated seed region. For each transfection, a total of six wells for each condition were collected and measured individually, and then averaged (n = 6). A total of three separate transfections were assayed and averaged (N = 3). *P < 0.05, t-test.
We have also found that expression of miR-9 and miR-448 in dystrophic heart was significantly and constitutively decreased in all age groups (Figure 2A and Supplementary material online, Table S1), which correlates with an increased expression of their predicted target genes, Cybb and Ncf1, encoding two subunits of NOX2, as seen in Figure 1B. In the present study, we choose to focus on miR-448 and miR-9, as their levels were already significantly reduced in hearts of very young animals that did not yet develop cardiomyopathy but already showing signs of oxidative stress.8–17
In rodents (mice and rats) biogenesis of miR-448 results in two mature forms—miR-448-3p and miR-448-5p, which originate from 3′ and/or 5′ of precursor pre-miR-448 correspondingly. Analysis of qPCR showed dramatic downregulation of both of these forms in dystrophic heart (Figure 2A). However, only the mature mouse miR-448-3p, but not miR-448-5p, sequence is highly conserved among all species, including humans, where it is called miR-448. Therefore, miR-448-3p was selected for further investigation.
There are substantial limitations of in silico bioinformatics tools and broad screening techniques. Therefore, we carried out in vitro experiments to confirm that Ncf1 (p47phox), as well as Cybb (gp91phox) are indeed targets of miR-448-3p and miR-9, respectively. Using luciferase reporter assay, we found that luciferase activity is significantly decreased when HEK-293 cells were co-transfected with a luciferase construct containing mouse 3′ UTR region of Ncf1 and mimic or precursor of miR-448-3p, a.k.a. pre-miR (Figure 2C and Supplementary material online, Figure S2C), in comparison with the same experiments when scrambled mimic or precursor was used (a.k.a. NC). Moreover, the inhibition of the luciferase signal was dose dependent (additional test for target specificity, Figure 2C). In addition, when HEK-293 cells were co-transfected with a luciferase construct containing mutated mouse 3′ UTR region of Ncf1 (mutation for miR-448 seed region) and miR-448-3p mimics (yet another crucial test for targeting), no significant changes in the luciferase signal were observed.
Identical experiments with a luciferase construct containing mouse 3′ UTR region of Cybb were conducted to test if miR-9 targets this gene. No changes in the level of inhibition of luciferase activity were detected with an increased concentration of miR-9 mimic (Supplementary material online, Figure S2B).
Therefore, we selected miR-448 for the rest of the studies, which, as validated by the data above, regulates the expression of Ncf1 gene.
3.3. Acute systemic inhibition of miR-448-3p increases Ncf1 expression in WT hearts
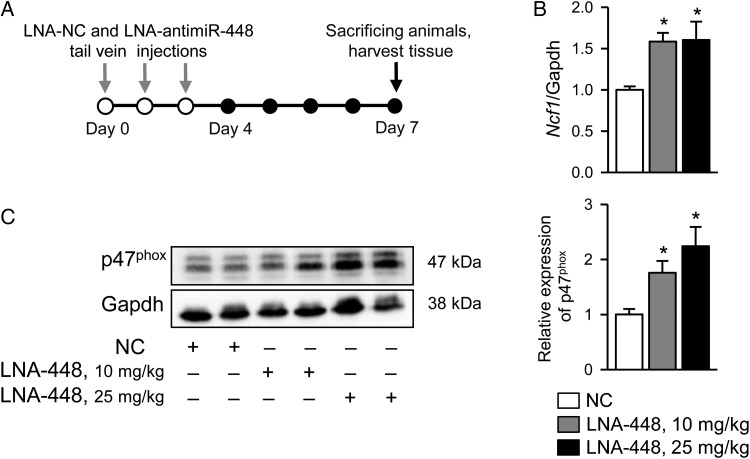
Previous studies demonstrated that individual miRs in the heart can be inhibited by anti-miRs—chemically modified antisense oligonucleotides.29 Among them are LNA-miR inhibitors, based on Locked Nucleic Acid (LNA™) Technology. To determine the role of miR-448-3p in vivo, we injected WT mice with miRCURY LNA™ miR-448-3p inhibitor (Exiqon, Woburn, MA, 10 mg/kg and 25 mg/kg) or with scrambled LNA-NC (10 mg/kg) through a tail vein according to manufacturer's protocol (Figure 3). Three days after the last injection, mice were sacrificed and qPCR and western blots were performed on heart tissue. Our results indicate that in vivo inhibition of miR-448-3p with its LNA-antimiR inhibitor significantly increased Ncf1 expression on both, gene (Figure 3B) and protein (Figure 3C) levels.
Figure 3.
Targeting of miR-448 by LNA-antimiR-448 inhibitor in WT mice results in an increase in Ncf1 gene and p47phox protein expression. (A) Protocol for LNA-antimiR-448 transfer. (B) Real-time PCR analysis of Ncf1 mRNA expression in cardiac tissue form WT mice treated with LNA-antimiR-448 inhibitor at two different concentrations. N = 5, *P < 0.05, t-test. (C) Expression of p47phox subunit of NOX2 in hearts of WT mice treated with LNA-NC and LNA-antimiR 448 inhibitors. N = 5, *P < 0.05, t-test.
3.4. Acute systemic inhibition of miR-448-3p induces cellular phenotype of dystrophic hearts
In a parallel group of experiments, we examined intracellular ROS production (with H2DCFDA) and intracellular Ca2+ stretch-response (with Fluo-4) in ventricular cardiomyocytes isolated from WT mice acutely injected either with miRCURY LNA™ miR-448-3p inhibitor (10 or 25 mg/kg) or with its NC (scrambled inhibitor, NC, 10 mg/kg), as described above. As illustrated in Figure 4A, inhibition of miR-448-3p resulted in a significant increase in intracellular ROS production as indicated by the increase in ROS-dependent DCF fluorescence signal. Incubation of myocytes with NOX2 inhibitor DPI decreased ROS level in all treated groups of animals to the same level, implying that increase of basal ROS level in cardiomyocytes after systemic miR-448-3p inhibition is an NOX2-dependent process.
Figure 4.
Systemic inhibition of miR-448 in WT mice results in an increase in NOX2-dependent ROS production and exacerbated Ca2+ responses in ventricular cardiomyocytes. (A) Representative images of DCF fluorescence in myocytes isolated from mice treated with LNA-NC and LNA-antimiR-448 inhibitor at two different concentrations. Graph in the middle illustrates changes in average DCF signals. Bar graph on the right illustrates the rate of DCF oxidation in six different groups of experiments: LNA-NC (N = 5, n = 17; N = 4, n = 22), LNA-antimiR-448, 10 mg/kg (N = 3, n = 25; N = 3, n = 13), and LNA-antimiR-448, 25 mg/kg (N = 3, n = 22; N = 3, n = 15) without and with incubation with DPI, respectively. (B) Intracellular Ca2+ responses to mild hypo-osmotic shock in ventricular cardiomyocytes from WT mice treated with LNA-NC and LNA-antimiR-448. Left panels are line-scan representations of series of images acquired from cells and converted to a two-dimensional X,t image. Middle panel represents time course of normalized fluo-4 fluorescence in WT cells treated with LNA-NC and LNA-antimiR-448. Right panel shows pooled data of mean values of normalized fluorescence during 60 s after the osmotic shock. Number of cells studied was LNA-NC (N = 4, n = 26), LNA-antimiR-448, 10 mg/kg (N = 4 n = 27; N = 3 n = 10), and LNA-antimiR-448, 25 mg/kg (N = 4 n = 27; N = 3 n = 12) without and with incubation with DPI, respectively. *P < 0.05, **P < 0.001, t-test (for ROS and Ca2+ measurements) and #P < 0.05, ##P < 0.001, t-test (for ROS and Ca2+ measurements after DPI treatment).
As we previously reported, mechanical stress applied as osmotic shock triggered intensified Ca2+ signals (sparks and waves) in mdx but not in WT cardiomyocytes.10,17 This observation was later confirmed with more physiological longitudinal stress.16 Recently, we found that the enhancement in Ca2+ responses to mechanical stress is due to post-translational modifications of RyRs resulting in the increase of their Ca2+ sensitivity because of oxidation of the intracellular environment.11,17 Here we applied the same protocol to evaluate sensitivity of RyRs in ventricular cardiomyocytes obtained from WT mice treated with LNA-miR-448-3p and LNA-NC (Figure 4B). Cells were loaded with Fluo-4 AM and then imaged with confocal microscopy during osmotic challenge (protocol is shown on the top of left panel in Figure 4B). In 81.5% (22 of 27 cells) of cardiomyocytes treated with 10 mg/kg of LNA-miR-448-3p, the return to isotonic solution produced extreme cytosolic Ca2+ signals. Treatment of animals with 25 mg/kg of LNA-miR-448-3p resulted in 89% (24 of 27) cells with abnormally enhanced Ca2+ responses. In contrast, in only 42% (11 of 26 cells) of the LNA-NC-treated cells, osmotic shock induced Ca2+ signals and these were mostly mild and short lived. On average, the stress-induced increase in Ca2+-dependent fluorescence, determined over 60 s after osmotic challenge, was larger in cardiomyocytes from animals treated with LNA-miR-448-3p (1.7 and 2.3 times larger compared with NC, for 10 and 25 mg/ml dose, respectively). Overall, cardiomyocytes from WT animals treated with anti-miRs exhibited a cellular phenotype, similar to that observed in mdx cells.
3.5. Chronic inhibition of miR-448p stimulates cardiac remodelling and cardiomyopathy
To examine the consequences of downregulation of miR-448-3p on cardiac function, we designed experiments aimed to chronically inhibit miR-448-3p in WT mice (Figure 5A). For this, we performed three initial injections of 10 mg/kg miRCURY LNA™ miR-448-3p inhibitor or scrambled LNA-NC followed by three single injections with 7 days interval and then evaluated functional and morphological properties of the heart. As in case of acute inhibition, this intervention significantly increased the expression of p47phox subunit of NOX2 at gene and protein levels (Figure 5B). Cardiac function was monitored in sedated mice using ultrasound system. Chronic treatment of WT mice with LNA-miR-448-3p reduced contractile properties of the hearts when compared with hearts of animals treated with LNA-NC. Fractional shortening (FS) of left ventricles decreased from 41.3 ± 2.92 (N = 7) to 27.9 ± 5.22 (N = 4) in mice treated with inhibitors, compared with LNA-NC (Figure 5C, left graph). Left ventricular ejection fraction was also significantly reduced following miR-448-3p inhibition [from 78.9 ± 2.96 (N = 7) to 60.8 ± 7.53 (N = 4)] without significant change in heart rate (Figure 5C right graph, Supplementary material online, Table S2). Some animals treated with LNA-448-3p also exhibited arrhythmia (as shown in Figure 5C, echogram on the bottom left).
Figure 5.
Chronic inhibition of miR-448 in WT mice impairs cardiac functions. (A) Protocol for LNA-antimiR-448 injection. (B) Left, real-time PCR analysis of Ncf1 mRNA expression in heart of WT mice injected with LNA-antimiR inhibitor of miR-448 (10 mg/kg) compared with the corresponding NC (N = 5). Middle, immunoblot and right, summary of p47phox subunit expression under the same experimental conditions. N = 5, *P < 0.05, t-test. (C) Left, M-mode echocardiographic images of left ventricles (LVs) of LNA-treated adult WT mice. Right, bar graphs illustrate changes in FS and ventricle ejection fraction in animals treated with LNA-NC (N = 7) and LNA-antimiR-448 (N = 4), respectively. *P < 0.05, t-test.
In addition to functional changes, some morphological and histological alterations of heart tissue were also detected after a prolonged LNA-miR-448-3p regime. Figure 6A depicts hearts of animals treated with either LNA-miR-448-3p or with LNA-NC. Note that LNA-miR-448-3p-treated hearts were already somewhat hypertrophied. The body/heart weight ratio increased from 6.98 ± 0.18 (N = 3) to 7.23 ± 0.15 (N = 3). Besides, echocardiography revealed substantial decrease in septal and posterior wall thickness of the left ventricle (Supplementary material online, Table S2), suggesting the development of a dilated cardiomyopathy. A significant increase in cross-sectional area of ventricular myocytes from LNA-miR-448-3p-treated hearts was also detected (Figure 6B).
Figure 6.
Chronic inhibition of miR-448 in WT mice induces cardiac remodelling. (A) Morphological changes in hearts following chronic LNA treatment. (B) Morphological changes in cardiac myocytes. Left, representative images of LV myocardium of LNA-treated mice stained with haematoxylin-eosin. Right, averaged cross-sectional area of the myocytes in hearts of animals treated with either LNA-NC (N = 3) or LNA-antimiR-448 (N = 3). *P < 0.05, t-test. (C and D) Left, patterns of perivascular and cardiac fibrosis in LV remote myocardium stained with Masson trichrome (C) or picric acid sirius red (D). Right, relative amount of fibrotic patches in hearts of animals treated with either LNA-NC (N = 3) or LNA-antimiR-448 (N = 3). **P < 0.001, t-test. (E) Real-time PCR analysis shows overexpression of pro-fibrotic genes mRNA in cardiac tissues after acute (grey bars) and chronic (black bars) inhibition of miR-448. Dashed line indicates gene expression in LNA-NC samples. N = 4–6. *P < 0.05, **P < 0.001, t-test.
Decrease in cardiac contractility is often associated with development of fibrosis in myocardium. Therefore, we next examined histological properties of the hearts with Masson trichrome and picric acid sirius red. Two hundred and nine sections (N = 3) of right and left ventricles as well as of intersectional septum of hearts removed from animals treated with LNA-miR-448-3p were compared with similar 166 sections (N = 3) of hearts treated with LNA-NC. A significant increase in perivascular fibrosis was detected in hearts exposed to chronic inhibition of miR-448 (Figure 6C and D).
Furthermore, we found that multiple mRNAs associated with genes involved in cardiac remodelling were overexpressed (Figure 6E). Most impressive was >2-fold increase in mRNA for Ctgf (connective tissue growth factor), Tgfb1 (transforming growth factor beta 1), and Mmp8 and Mmp9 (matrix metalloproteinases—enzymes responsible for degradation of myocardial extracellular proteins). Levels of mRNA for Fgf12 (fibroblast growth factor 12) and Col1a2 (collagen type 1, alpha 2) were also elevated although to a lesser extent. Some increase was also seen in mRNA for Nppb (natriuretic peptide precursor B). Overall, the results show a significant deterioration of cardiac performance after chronic treatment of mice with LNA-miR-448-3p inhibitor.
It should be mentioned that NOX2 is expressed in many tissues. Therefore, systemic inhibition of miR-448 can potentially increase NOX2 activity in tissues other than heart, including inflammatory cells where it is the most active.4 A subsequent boost in inflammation could also, though indirectly, contribute to a deterioration of cardiac function in mice after LNA-miR-448 treatment. In addition, the release of pro-inflammatory cytokines can either prime or even increase NOX2 activation everywhere. Supplementary material online, Figure S3 illustrates the levels of selected cytokines in cardiac samples from control and LNA-miR-448-treated animals. It seems that acute inhibition of miR-448 did not induce dramatic changes in cytokines levels. Therefore, changes in intracellular Ca2+ signalling and ROS production (Figure 4) most likely were not influenced by the activation of inflammatory processes. Nevertheless, prolonged inhibition of miR-448 resulted in a robust increase in cytokines, which indeed may contribute to the development of fibrosis and the deterioration of cardiac function in treated mice (Figures 5 and 6).
4. Discussion
The results of these studies reveal an important role of miR-448-3p in development of cardiac disease driven by oxidative stress. Animals systematically treated with LNA-miR-448-3p inhibitor exhibit significant decline of electrical and contractile properties of the heart. Cardiac dysfunctions were at least partially associated with deterioration of cardiac muscle tissue, in particular development of patches of fibrosis and morphological remodelling of cardiomyocytes. On the cellular level, inhibition of miR-448-3p results in significant increase in expression of the regulatory p47phox subunit of NOX2 enzyme, enhanced NOX2-dependent ROS production, and amplified intracellular Ca2+ signalling. The latter may in turn culminate in activation of Ca2+-dependent apoptotic and/or necrotic mechanisms, appearance of fibrotic patches and decrease in cardiac contractility, features that are often observed in pathological tissues, including dystrophic cardiac muscle. Our results extend recent reports of the role of miRs in cardiac disease and provide a novel insight into how expression of ROS-generating moieties, in particular NOX2, can be regulated at posttranscriptional level. To our knowledge these data provide the first evidence that systemic delivery of miR can directly control NOX2 expression in heart and impact cardiac functions. Because increased levels of NOX2 and/or its activity have been shown to be involved in several cardiac dysfunctions other than DMD (such as ROS-dependent hypertrophic cardiac responses, myocardial inflammation and fibrosis, post-MI remodelling as well as atrial fibrillation) our findings are important for a wide range of cardiac research.
4.1. miRs and cardiovascular disease
miRs are important regulator of gene expression. It is currently believed that they control, at least in part, many physiological and pathophysiological events in cardiovascular tissue. Dozens of miRs have already been identified to be involved in the regulation of cardiac muscle proliferation, regeneration and degeneration, development of cardiac arrhythmias, CH and HF.18–21,30 Multiple molecular targets of miRs have been identified. Some miRs directly regulate intracellular Ca2+ handling/signalling and excitation–contraction coupling by targeting specific proteins involved in these processes.30 Others, adjust the expression of various transcription factors and proteins during myocardial remodelling in cardiac disease.18 Development of specific inhibitors and mimetics of miRs that can be locally or systemically delivered to the organisms, prompted an enthusiasm for miRs to become novel therapeutic targets.21 The recent success in treatment of hepatitis C with antimiR treatment in a human clinical trial, as well as correction of cardiovascular problems in various animal models, made miR-based therapy very appealing.
4.2. Specificity of targeting, tissue distribution, and biosynthesis of miR-448-3p
There are tens to hundreds of protein-coding genes regulated by a single miR. Because miRs identify certain ‘seed sequences’ in sections of genes, any gene containing a sequence complementary to the seed region is potentially regulated by the respective miR. Some of these genes can encode proteins involved in redox and Ca2+ signalling pathways. Therefore, we used several bioinformatics portals to predict alternative targets for miR-448-3p. Supplementary material online, Figure S4 shows that the expression of some potential target genes is changed in dystrophic hearts. Of specific interest is the Sri gene that encodes sorcin, a protein that is involved in the regulation of intracellular Ca2+ signalling in cardiomyocytes. In particular, it has been demonstrated that sorcin reduces intracellular Ca2+ responses by directly inhibiting RyR channels.31,32 Sri expression is slightly, but significantly, reduced in dystrophic hearts. This, in general, can result in partial removal of RyRs inhibition by sorcin and add to the enlarged Ca2+ signalling observed in dystrophic cardiomyocytes. However, it is unlikely that sorcin is directly involved in regulation of the redox potential in young mdx hearts, as the contribution of Ca2+-dependent ROS-producing moieties, such as mitochondria, is limited at early stages of the disease.
Among factors that influence the efficiency of miR targeting to a specific gene is tissue expression profile of miRs and the level of its expression. Tissue-specific or housekeeping miRs as well as highly expressed miRs usually have more robust effects on a target. Supplementary material online, Figure S5 illustrates the tissue expression profiles of miR-448-3p and its corresponding Htr2c gene. Htr2c is located at X chromosome and encodes serotonin receptor 2c. Thus, it was not a surprise to see that Htr2c expression was the highest in brain tissue. The Htr2c gene expression in heart was below the detection level (Supplementary material online, Figure S5A). However, the significant level of miR-448-3p was detected in cardiac tissue and even in isolated cardiomyocytes (Supplementary material online, Figure S5B). Therefore, although it is not a dominant miR in cardiac muscle, miR-448-3p is expressed there and is able to directly target cardiac NOX2 subunit.
It has long been believed that transcription of miR precursors depends on transcription of their host genes. However, recently published data suggest that at least one-third of intronic miRs has transcription initiation site that is independent from host gene promoter.33,34 Moreover, Gao et al.35 has demonstrated that miR precursors can be ‘self-transcribed’, in the absence of exogenous promoters. Relevant to our studies, Zhang et al.36 recently showed that the 5′ untranslated region (intron 2) mRNA of the Htr2c (intron 4 of Htr2c contains miR-448) is widely expressed in various non-neuronal cell lines, despite the fact that the mRNA of the protein-coding region of Htr2c was not detected in these cell lines. Several miRs, encoded in intron 2, were also expressed there. However, whereas intron 2 expression was similar in all cell lines studied, levels of miR were different, indicating cell-type-specific processing or stability of pre-miRs. In addition, Li et al.37 found several predicted binding sites for transcription factors (such as NF-κB, AP1, c-Myc, and AP4) located within intron 4 of Htr2c gene upstream of pre-miR-448 stemloop sequence. They also showed that NF-κB (which expression is upregulated after systemic miR-448 inhibition, as shown in Figure 6D) directly binds the promoter of miR-448, suppressing its transcription at least in some cell lines. Therefore, we can suggest that pre-miR448 transcription in the heart may be regulated independently of host Htr2c gene expression.
Overall, our findings reveal a pivotal role of miR-448-3p in development of NOX2-ROS-driven cardiomyopathy in mice. Although we are only just beginning to gain some insights into therapeutic application of miRs, it is clear that they are important players in the development of cardiovascular diseases. Our data suggest that miRs-regulating expression of NOX2 subunits could potentially become strategic therapeutic targets that would help to slow down deterioration of cardiac function not only in DMD patients but also in patients with other cardiac pathologies associated with oxidative stress.
Supplementary material
Supplementary material is available at Cardiovascular Research online.
Conflict of interest: none declared.
Funding
This work was supported by NIH (HL093342 to N.Sh. and AG023039 to J.S.). We are grateful to Drs. I. Borbiro, C. Kang, and K. Tobon, for their help and advice.
References
- 1.Paravicini TM, Touyz RM. NADPH oxidases, reactive oxygen species, and hypertension: clinical implications and therapeutic possibilities. Diabetes Care 2008;31:S170–S180. [DOI] [PubMed] [Google Scholar]
- 2.Nabeebaccus A, Zhang M, Shah AM. NADPH oxidases and cardiac remodelling. Heart Fail Rev 2010;16:5–12. [DOI] [PubMed] [Google Scholar]
- 3.Zhang M, Perino A, Ghigo A, Hirsch E, Shah AM. NADPH oxidases in heart failure: poachers or gamekeepers? Antioxid Redox Signal 2013;18:1024–1041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bedard K, Krause K-H. The NOX family of ROS-generating NADPH oxidases: physiology and pathophysiology. Physiol Rev 2007;87:245–313. [DOI] [PubMed] [Google Scholar]
- 5.Shirokova N, Niggli E. Cardiac phenotype of Duchenne muscular dystrophy: insights from cellular studies. J Mol Cell Cardiol 2013;58:217–224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Finsterer J, Stöllberger C. The heart in human dystrophinopathies. Cardiology 2003;99:1–19. [DOI] [PubMed] [Google Scholar]
- 7.Kieny P, Chollet S, Delalande P, Le Fort M, Magot A, Pereon Y, Perrouin Verbe B. Evolution of life expectancy of patients with Duchenne muscular dystrophy at AFM Yolaine de Kepper centre between 1981 and 2011. Ann Phys Rehabil Med 2013;56:443–454. [DOI] [PubMed] [Google Scholar]
- 8.Yasuda S, Townsend D, Michele DE, Favre EG, Day SM, Metzger JM. Dystrophic heart failure blocked by membrane sealant poloxamer. Nat Cell Biol 2005;436:1025–1029. [DOI] [PubMed] [Google Scholar]
- 9.Williams IA, Allen DG. Intracellular calcium handling in ventricular myocytes from mdx mice. Am J Physiol Heart Circ Physiol 2006;292:H846–H855. [DOI] [PubMed] [Google Scholar]
- 10.Jung C, Martins AS, Niggli E, Shirokova N. Dystrophic cardiomyopathy: amplification of cellular damage by Ca2+ signalling and reactive oxygen species-generating pathways. Cardiovasc Res 2007;77:766–773. [DOI] [PubMed] [Google Scholar]
- 11.Williams IA, Allen DG. The role of reactive oxygen species in the hearts of dystrophin-deficient mdx mice. Am J Physiol Heart Circ Physiol 2007;293:H1969–H1977. [DOI] [PubMed] [Google Scholar]
- 12.Fanchaouy M, Polakova E, Jung C, Ogrodnik J, Shirokova N, Niggli E. Pathways of abnormal stress-induced Ca2+ influx into dystrophic mdx cardiomyocytes. Cell Calcium 2009;46:114–121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ullrich ND, Fanchaouy M, Gusev K, Shirokova N, Niggli E. Hypersensitivity of excitation-contraction coupling in dystrophic cardiomyocytes. Am J Physiol Heart Circ Physiol 2009;297:H1992–H2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sarma S, Li N, van Oort RJ, Reynolds C, Skapura DG, Wehrens XHT. Genetic inhibition of PKA phosphorylation of RyR2 prevents dystrophic cardiomyopathy. Proc Natl Acad Sci USA 2010;107:13165–13170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Fauconnier J, Thireau J, Reiken S, Cassan C, Richard S, Matecki S, Marks AR, Lacampagne A. Leaky RyR2 trigger ventricular arrhythmias in Duchenne muscular dystrophy. Proc Natl Acad Sci USA 2010;107:1559–1564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Prosser BL, Ward CW, Lederer WJ. X-ROS signaling: rapid mechano-chemo transduction in heart. Science 2011;333:1440–1445. [DOI] [PubMed] [Google Scholar]
- 17.Kyrychenko S, Polakova E, Kang C, Pocsai K, Ullrich ND, Niggli E, Shirokova N. Hierarchical accumulation of RyR post-translational modifications drives disease progression in dystrophic cardiomyopathy. Cardiovasc Res 2013;97:666–675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Small EM, Frost RJA, Olson EN. MicroRNAs add a new dimension to cardiovascular disease. Circulation 2010;121:1022–1032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hata A. Functions of microRNAs in cardiovascular biology and disease. Annu Rev Physiol 2013;75:69–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Abdellatif M. Differential expression of microRNAs in different disease states. Circ Res 2012;110:638–650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.van Rooij E, Olson EN. MicroRNA therapeutics for cardiovascular disease: opportunities and obstacles. Nat Rev Drug Discov 2012;11:860–872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Creemers EE, Tijsen AJ, Pinto YM. Circulating microRNAs: novel biomarkers and extracellular communicators in cardiovascular disease? Circ Res 2012;110:483–495. [DOI] [PubMed] [Google Scholar]
- 23.Cacchiarelli D, Incitti T, Martone J, Cesana M, Cazzella V, Santini T, Sthandier O, Bozzoni I. miR-31 modulates dystrophin expression: new implications for Duchenne muscular dystrophy therapy. EMBO Rep 2011;12:136–141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Liu N, Williams AH, Maxeiner JM, Bezprozvannaya S, Shelton JM, Richardson JA, Bassel-Duby R, Olson EN. microRNA-206 promotes skeletal muscle regeneration and delays progression of Duchenne muscular dystrophy in mice. J Clin Invest 2012;122:2054–2065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kyrychenko V, Poláková E, Janíček R, Shirokova N. Mitochondrial dysfunctions during progression of dystrophic cardiomyopathy. Cell Calcium 2015;58:186–195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Stolk J, Hiltermann TJ, Dijkman JH, Verhoeven AJ. Characteristics of the inhibition of NADPH oxidase activation in neutrophils by apocynin, a methoxy-substituted catechol. Am J Respir Cell Mol Biol 1994;11:95–102. [DOI] [PubMed] [Google Scholar]
- 27.Kinkade K, Streeter J, Miller FJ. Inhibition of NADPH oxidase by apocynin attenuates progression of atherosclerosis. IJMS 2013;14:17017–17028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Brandes RP, Weissmann N, Schröder K. Nox family NADPH oxidases: Molecular mechanisms of activation. Free Radic Biol Med 2014;76:208–226. [DOI] [PubMed] [Google Scholar]
- 29.Esau CC. Inhibition of microRNA with antisense oligonucleotides. Methods 2008;44:55–60. [DOI] [PubMed] [Google Scholar]
- 30.Harada M, Luo X, Murohara T, Yang B, Dobrev D, Nattel S. MicroRNA regulation and cardiac calcium signaling: role in cardiac disease and therapeutic potential. Circ Res 2014;114:689–705. [DOI] [PubMed] [Google Scholar]
- 31.Lokuta AJ, Meyers MB, Sander PR, Fishman GI, Valdivia HH. Modulation of cardiac ryanodine receptors by sorcin. J Biol Chem 1997;272:25333–25338. [DOI] [PubMed] [Google Scholar]
- 32.Farrell EF, Antaramian A, Rueda A, Gómez AM, Valdivia HH. Sorcin inhibits calcium release and modulates excitation-contraction coupling in the heart. J Biol Chem 2003;278:34660–34666. [DOI] [PubMed] [Google Scholar]
- 33.Ozsolak F, Poling LL, Wang Z, Liu H, Liu XS, Roeder RG, Zhang X, Song JS, Fisher DE. Chromatin structure analyses identify miRNA promoters. Genes Dev 2008;22:3172–3183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Bhattacharyya M, Das M, Bandyopadhyay S. miRT: a database of validated transcription start sites of human microRNAs. Genomics Proteomics Bioinformatics 2012;10:310–316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Song Gao J, Zhang Y, Li M, Tucker LD, Machan JT, Quesenberry P, Rigoutsos I, Ramratnam B. Atypical transcription of microRNA gene fragments. Nucleic Acids Res 2010;38:2775–2787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Zhang Z, Falaleeva M, Agranat-Tamir L, Pages A, Eyras E, Sperling J, Sperling R, Stamm S. The 5′ untranslated region of the serotonin receptor 2C pre-mRNA generates miRNAs and is expressed in non-neuronal cells. Exp Brain Res 2013;230:387–394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Li Q-Q, Chen Z-Q, Cao X-X, Xu J-D, Xu J-W, Chen Y-Y, Wang W-J, Chen Q, Tang F, Liu X-P, Xu Z-D. Involvement of NF-κB/miR-448 regulatory feedback loop in chemotherapy-induced epithelial-mesenchymal transition of breast cancer cells. Cell Death Differ 2011;18:16–25. [DOI] [PMC free article] [PubMed] [Google Scholar]