Abstract
Nanoparticles (NPs) are considered a promising tool in both diagnosis and therapeutics. Theranostic NPs possess the combined properties of targeted imaging and drug delivery within a single entity. While the categorization of theranostic NPs is based on their structure and composition, the pharmacokinetics of NPs are significantly influenced by the physicochemical properties of theranostic NPs as well as the routes of administration. Consequently, altered pharmacokinetics modify the pharmacodynamic efficacy and toxicity of NPs. Although theranostic NPs hold great promise in nanomedicine and biomedical applications, a lack of understanding persists on the mechanisms of the biodistribution and adverse effects of NPs. To better understand the diagnostic and therapeutic functions of NPs, this review discusses the factors that influence the pharmacokinetics, pharmacodynamics and toxicology of theranostic NPs, along with several strategies for developing novel diagnostic and therapeutic modalities.
Graphical Abstract
1. Introduction
Nanoparticles (NPs) possess a relatively small size in the nanorange (1–1000 nm),1 but have a significant advantage over atoms and molecules owing to a larger surface area per unit volume. NPs also have a greater formulating flexibility for various sizes and shapes with different chemical surface traits.2 Due to their versatile nature, they have been successfully used as both diagnostic and therapeutic tools.3 “Theranostics” refers to the development of compounds, which exhibit the characteristics of diagnostics and therapeutics in a single entity.1, 4 The rapid advancement in nanotechnology has allowed the emergence of theranostic NPs, which have shown advantages of diagnosis and drug delivery as well as targeting the biomarkers of the disease at the molecular level.5 For the clinical use, however, the size of a NP has to be limited up to 220 nm because a standard 0.22μm (220 nm) filter is used routinely in the clinic before injecting theranostic agents into the body. National Nanotechnology Initiative (NNI) also defines “nanomaterials” as 1) research and technology development at the atomic, molecular or macromolecular levels, in the length scale of approximately 1–100 nm range; 2) creating and using structures, devices and systems that have novel properties and functions because of their small and/or intermediate size; and 3) ability to control or manipulate at the atomic scale.2, 6
Although theranostic NPs hold great promise in nanomedicine and biomedical applications, a lack of understanding persists on the mechanisms of the biodistribution and adverse effects of NPs. An ideal theranostic NP model should possess several important properties. For delivery, NPs should act on the target tissues and demonstrate appropriate release kinetics of the drug in optimum concentrations at the site of action, illustrating their efficient therapeutic potency. Since it also possesses diagnostic abilities, it should help determine the precise location and characteristics of the disease. Along with these properties, it is very important that the NP should be non-toxic and easily excretable or eliminated from the body.4 There have been several reviews providing an in-depth outlook on the potential of NPs and their application in several aspects, such as their usage as theranostic agents in drug delivery5 and the application of theranostic NPs in cancer therapy,7, 8 which is one of the most rapidly developing therapy involving nanosystems. Recognizing that the in vivo availability and efficacy of NPs are mainly determined by their pharmacokinetics (PK) and potential toxicity, we provide a brief review of these facets of theranostic NPs.
2. Backbone Materials of Theranostic NPs
NP-based theranosis is considered as a promising future nanomedicine because NPs can possess several unique features including targeting, imaging (diagnosis), and therapeutic potentials within a single nanoplatform. In contrast to small molecules, theranostic NPs can be tuned for optical, electrical, magnetic and biological properties and can carry large payloads along with contrast agents.3
The backbone materials can be categorized into two classes based on their compositions: organic vs. inorganic materials, and their key characteristics are summarized in Table 1. In organic nanomaterials, synthetic polymers and biopolymers including dendrimers, lipoproteins and liposomes have been often utilized for targeted drug delivery in the past decades. Organic nanomaterials have biocompatibility and allow them to functionalize with targeting moieties on their surface; however, they usually need a complexation and covalent conjugation of contrast agents. On the other hand, many inorganic nanomaterials, especially being led from the development of superparamagnetic iron oxide nanoparticles (SPIONs) and quantum dots (QDs), have been intensively studied and already developed as core imaging materials. Consequently, such inorganic nanomaterial-based theranostic NPs can be easily prepared by loading therapeutic drugs onto and/or into the NP’s surface.1 However, the inorganic and carbon-based materials are generally required to modify their surface with a biocompatible organic coating due to the poor water solubility, stability, and potential toxicity (Table 1).
Table 1.
Pre-clinically available theranostic NPs.2
| Class | NP Type | Composition | Therapeutic modality | Pros | Cons | Ref. |
|---|---|---|---|---|---|---|
| Inorganic NPs | Magnetic NPs | Iron oxide | Chemotherapy; siRNA; magnetic hyperthermia | Intrinsic MRI contrast; thermal therapeutic agent | Interference in imaging | 9–11 |
| QDs | Semiconductor | Chemotherapy; siRNA; photodynamic therapy | Broadband absorption; small size; tunable emission band | High potential toxicity | 12 | |
| Silica NPs | Mesoporous silica | Chemotherapy; siRNA | Multi functionality; facile synthesis; solubility | Stability; need contrast agents | 13 | |
| Carbon NPs | Graphene oxide | Photothermal therapy; photodynamic therapy; chemotherapy | Large surface area; thermal therapeutic agent | Size control; difficulty in purification | 14–16 | |
| Carbon nanotube | Size tunability; mechanical strength | High aspect ratio; difficulty in purification; poor solubility | 17–19 | |||
| Gold NPs | Gold nanoshell | Photothermal therapy; chemotherapy | Size tunability; Intrinsic thermal therapeutic agent; tunable in NIR region | Potential toxicity | 20 | |
| Gold nanorod | Photothermal therapy | High aspect ratio; toxicity; difficulty in therapeutic payload | 21, 22 | |||
| Others | CuS NPs | Photothermal; chemotherapy | Thermal therapeutic agent; tunable in NIR region | Potential toxicity | 23, 24 | |
| MoS2 nano-sheet | Photothermal; chemotherapy; siRNA | Large surface area; thermal therapeutic agent | Need contrast agents; difficulty in size control | 25, 26 | ||
| Organic NPs | Biological NPs | Naturally polymers and lipoprotein | Chemotherapy; siRNA | Biocompatibility; biodegradability | Need contrast agents; difficulty in size and degradability control | 27, 28 |
| Polymer NPs | Linear or branched polymer | Photodynamic therapy; chemotherapy | Biodegradability; flexibility; size tunability | Need contrast agents | 29–31 | |
| Dendrimers | Tree-like macromolecules | Chemotherapy | Size tunability; solubility | Limited synthesis; need contrast agents | 32, 33 | |
| Liposomes | Phospholipid bilayers | Chemotherapy; siRNA | Conventional drug delivery; large payload | Need contrast agents; poor stability | 34–36 |
Abbreviations used are: MRI, magnetic resonance Imaging; NIR, near-infrared; NP, nanoparticle; QD, quantum dot; siRNA, small interfering RNA.
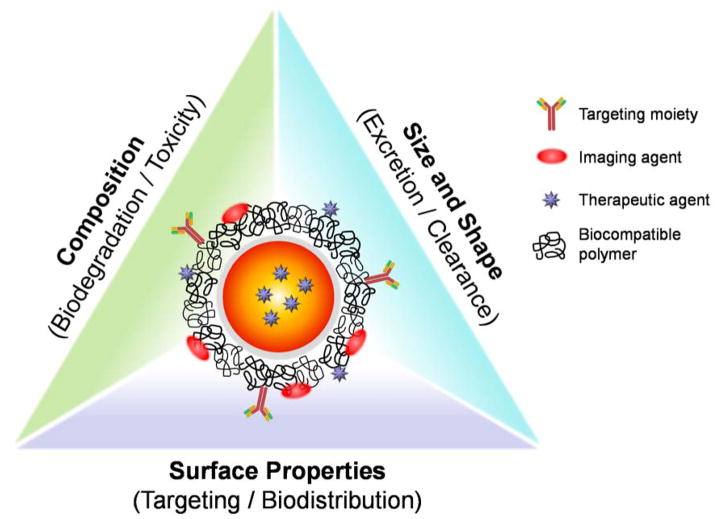
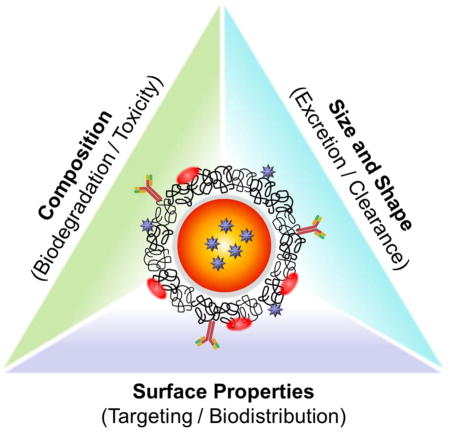
Regardless of their compositions, all theranostic NPs must be designed to have a reasonable half-life in blood, selective targetability, and effective elimination from the body after comprehensive delivery to the target site.2, 37–39 To acquire these desired pharmacokinetic behaviors of NPs for clinical use, it is necessary to modulate the hydrodynamic diameter (HD), shape, composition, and surface characteristics of NPs based on the “Choi Criteria” (Figure 1).2 For instance, the overall HD of theranostic NPs is required to be < 5.5 nm for renal clearance after complete targeting in order to achieve high signal-to-background ratio.40 In the following section, we discuss more details about the physicochemical properties of theranostic NPs in terms of size, shape, surface, composition and route of administration.
Figure 1.
Schematic illustration of a theranostic NP and its physicochemical properties that regulate in vivo pharmacokinetics, biodistribution and toxicity.
3. Pharmacokinetics of NPs
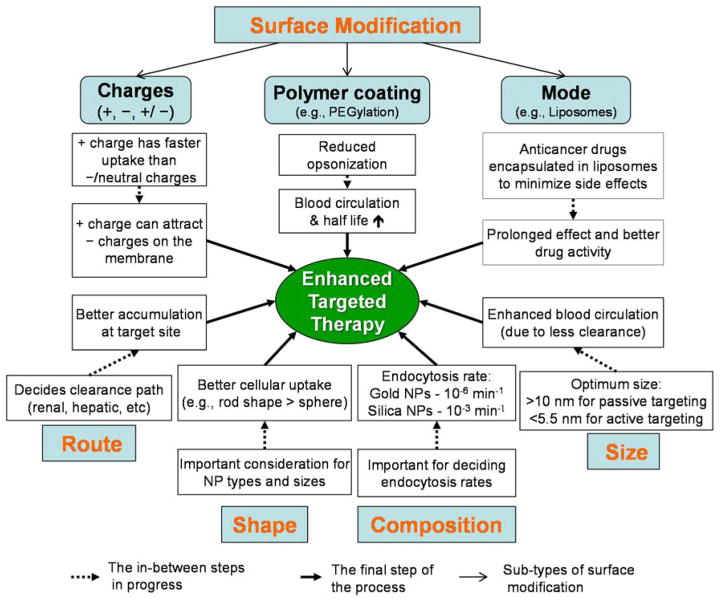
The physicochemical properties of theranostic NPs are of significant importance in modulating PK because they determine the immediate pharmacological response in the body when the NPs are administered. Drugs with low bioavailability can have better drug dissolution rates by the technique of “nanosizing” a drug formulation, which would promote increased absorption of the drug.41 Also, NPs can prolong the half-life of drugs in blood circulation, which would otherwise be rapidly cleared or degraded. Since the PK plays a major role in determining the therapeutic efficacy and toxicity of the administered NPs, several key factors influencing the PK of NPs (Figure 2) are discussed. In this section, we avoid reticuloendothelial system-mediated NP clearance and focus on smaller NPs and their theranostic aspects because larger NPs have slim chances of clinical translation. As previously reported, renal excretion is a preferred and desirable pathway for theranostic NPs compared with hepatic clearance because the NPs can be rapidly eliminated from the body while little cellular internalization/metabolism is involved, thus effectively minimizing body exposure to the NPs.40, 42–44
Figure 2.
Factors influencing the pharmacokinetics of nanoparticles (NPs). Size, shape, composition, administration route, and surface modification should be considered for the enhanced targeted therapy mediated by NPs.
3.1 Size and Shape
The ability of the NPs to enter the cell is determined by both physicochemical parameters and biological barriers. Due to the high surface area to volume ratio (small size), they are able to cross the biological barriers by penetration through the cell membrane45 and deliver the drug inside the cell. It has been found that a general size range of 10–12 nm is ideal and offers high permeation and minimal accumulation in tissues.46 Choosing a proper size in designing an NP is also essential as it directs which excretion pathway the drug would follow. For example, particles with a smaller HD of < 5.5 nm follow the route of renal excretion,2 whereas larger sized NPs are eliminated through the liver.40 Also, choosing a proper carrier is of high significance. NP systems, such as liposomes and organic/inorganic hybrid nanospheres, have been used in precise targeting of various diseases with intravenous delivery of theranostic small molecules. The drug-loaded carrier can control the efficiency of drug delivery and also protect the drug from inactivation and/or degradation, which can reduce its adverse effects.47 The cellular uptake is also influenced by the shape of the NPs; for example, elongated NPs are better absorbed than spherical ones.48
3.2 Surface property
A modification in the surface of NPs significantly affect on the physical, chemical and biological nature of the entire molecules in biological systems.
NPs can induce positive/negative charges on the surface where the interactions with cell membrane change in a different manner, which affects their absorption and distribution. NPs with positively-charged surface show greater uptake than negatively-charged NPs due to electrostatic interactions.48 Alteration of the NP surface with a neutral non-ionic polymer imparts stability to the NP by decreasing opsonisation and increasing blood circulation time, as exemplified by NPs coated with polyethylene glycol (PEG) on their surface.48 Surface property also plays an important role when the NPs are placed in biological fluids (e.g., blood). The surface of NPs is coated with a layer of proteins (protein corona) when in contact with the biological fluid.48 This layer plays a vital role to determine the attraction of the NP to the cell membrane. Different NPs form different protein corona and thus, each type of NPs has different affinity for a particular protein in a biological fluid and affects the physicochemical characteristics, which would subsequently affect the rate and extent of biodistribution.
3.3 Administration Route
The PK of a drug from a NP depends upon the route by which the drug has been administered, which modifies pharmacological efficacy of the drug. For instance, when bovine insulin was given orally by means of a pH-responsive NP system of chitosan and poly(γ-glutamic acid) to rats, it showed a greater bioavailability compared with subcutaneously-injected insulin in diabetic patients.49 These oral NPs infiltrate the mucous layer of the intestinal tract and gradually destabilize and disintegrate due to their pH sensitivity. The increased bioavailability may be attributed to the pH-sensitive insulin release from the NPs. The difference in biodistribution of insulin and prolonged reduction of glucose levels between subcutaneous insulin and oral NPs could be because insulin, via the oral route, mimics the physiological pathway of the endogenously secreted insulin, which reaches the liver and helps to control the glucose levels in the body. In contrast, the insulin given by the subcutaneous route fails to mimic this since it enters the peripheral circulation, which is not the normal route of insulin production and secretion.
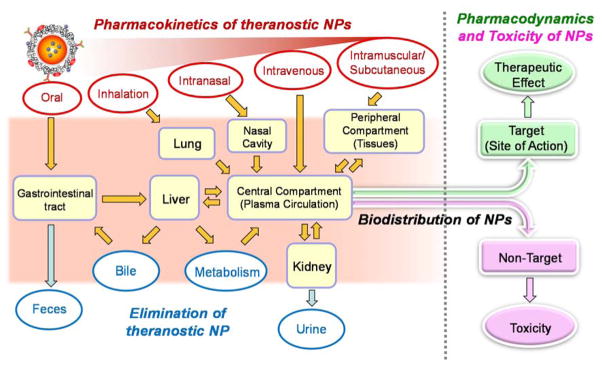
There are several publications concerning the effect of injection routes on the biodistribution and elimination of NPs.50–52 Very recently, Huang el al. reported the biodistribution, clearance and tumor uptake of renally clearable carbon dots with three different injection routes, including intravenous, intramuscular and subcutaneous administrations.53 The blood clearance and urinary accumulation rate of administered NPs followed the order of intravenous > intramuscular > subcutaneous injections. In addition, tumor uptake of carbon dots by subcutaneous and intravenous injections was higher than that by intramuscular injection. Such examples are indicative of the route-dependent therapeutic potential and clinical benefits of NP-based theranostic systems. Absorption, biodistribution, elimination and pharmacologic and toxic effects of NPs following different routes of administration are summarized in Figure 3.
Figure 3.
The pharmacokinetics, pharmacodynamics and toxicity of theranostic NPs. Shown are absorption, biodistribution, elimination and pharmacologic and toxic effects of NPs following different routes of administration. The fate of the theranostic NPs depends on physicochemical properties of NPs and the route of administration as well as altered body functions (e.g., nutrition status and disease conditions).
3.4 Composition
Many therapeutic NPs are composed of several different elements with specific geometry/conformation such as core-shell, core-satellite, linear, and hyper-branched structures. The little differences of the geometry or conformation can contribute to their in vivo performance such as absorption, biodistribution, elimination as well as targeting ability.2 In addition, the geometry/conformation changes of NPs and decomposition in in vivo environments can significantly affect toxicity.
The biodegradability of theranostic NPs relies on their chemical compositions. Polymeric NPs containing hydrolysable linkages, such as ester, ortho-ester and anhydride, in their backbones are biodegradable in the body.54 The use of biodegradable polymers can significantly increase the elimination of NPs from the body and reduce the long-term toxicity. On the contrary, most inorganic NPs are not biodegradable. Such inorganic NPs remain for a relatively long period of time in the body due to their larger size and greater hydrophobicity compared with small molecules, therefore, concerns have been raised about the potential long-term toxicity of these NPs.
Taken together, both physicochemical properties (i.e. surface charge, chemistry and size of the NP) and exposure routes are critical factors that determine the PK of NPs,41 and these factors can be modified to control (enhance or decrease) the blood circulation and tissue permeation of the drug. On the other hand, poorly designed NPs can promote an enhanced delivery of the drug molecules to certain non-target tissues non-specifically and cause undesirable side effects, which warrant the appropriate assessment of toxicity for the use of NPs.
4. Imaging and Therapeutic Modalities
The selection of imaging modality is another important component for theranostic NPs. The use of minimal- or non-invasive imaging modality is beneficial to characterize the PK and biodistribution as well as therapeutic efficacy of theranostic NPs.55 Current clinically available imaging modalities include nuclear imaging (positron emission tomography; PET, and single photon emission computed tomography; SPECT), magnetic resonance imaging (MRI), computed tomography (CT), ultrasound (US), optical imaging, and photoacoustic (PA) imaging (Table 2). However, for efficient molecular imaging, the surface or core of NPs should be modified with various radioisotopes, paramagnetic ion chelates, or fluorophores, except in the case of using inherent contrast NPs such as iron oxide NPs, QDs, and dyes-doped silica NPs. Based on the intrinsic sensitivity and tissue penetration ability of imaging modalities, the theranostic NPs could be visualized via non-invasive (more desired) or minimally invasive (less desired) manner in diagnostic procedures. The pros and cons of each imaging modality are summarized in Table 2. In this section, we describe therapeutic modalities of NPs.
Table 2.
| Modality | Probe(s) | Pros | Cons |
|---|---|---|---|
| Nuclear imaging: PET, SPECT | Radionuclides (e.g. F-18, In-111, Cu-64) | Quantitative analysis High sensitivity |
Radioisotope exposure Expensive procedures |
| MRI | Paramagnetic atoms (e.g. Gd, Mn) Superparamagnetic NPs (e.g. SPION) |
Able to image physiological and anatomical details Soft tissue contrast |
Limited acquisition time High cost |
| CT | Heavy elements (e.g. iodine) | Quantitative anatomical information High spatial resolution |
Radiation exposure Limited to morphological information |
| Ultrasound imaging | Gas filled microbubbles | Ease of procedure Low cost |
Low resolution Low sensitivity |
| NIR imaging | Fluorophores (e.g. fluorescence dye, QD) | High sensitivity and spatial resolution Low autofluorescence Cost efficiency and simplicity |
Limited penetration (<5 mm) |
| Photoacoustic imaging | Light absorbates (e.g. fluorophore, quencher) | High spatial resolution Functional information |
Limited penetration (<5 cm) |
Abbreviations used are: PET, positron emission tomography; SPECT, single photon emission computed tomography; MRI, magnetic resonance Imaging; NP, nanoparticle; SPION, superparamagnetic iron oxide nanoparticle; QD, quantum dot
4.1 Chemotherapy
Since cancer is one of the leading causes of death worldwide, NP-based cancer therapy has great potential for overcoming biological barriers and selective targeting to desired sites.56 Furthermore, NPs are relatively small and have greater affinity ligands, which decreases non-specific biodistribution and the for the cell membrane, thus can easily enter the cancer cells toxicity in non-target organs.57 after binding to the cell surface specifically through targeting
Polymeric NPs like liposomes and micelles have been used to solubilize hydrophobic drugs so that higher percentage of injected dose (%ID) can be achieved at the target site. Doxil, for example, is a PEGylated liposome coated on doxorubicin (DOX), where the PEG coating prevents the degradation of drugs by immune system and controls the release of drugs into the blood, resulting in a prolonged terminal half-life and higher drug efficacy.57,60 Another example is a QD-aptamer-DOX conjugate [QD-Apt(DOX)] for prostate cancer therapy. The QD-Apt(DOX) conjugate can perceive and render DOX at the target site by using the fluorescence resonance energy transfer (FRET) effect between DOX and QDs. The conjugate is composed of the following 3 parts; 1) therapeutic DOX, 2) targeted RNA aptamers, which are covalently attached onto the surface of QDs, and 3) diagnostic QDs for fluorescence imaging. This activatable system works by turning “on” the fluorescence by releasing DOX in the tumor cells, while the DOX-loaded QD-Apt is “off” in the normal cells.61
4.2 Gene Therapy
Gene therapy implies the replacement of a faulty gene in the cell with a proficient gene or by overexpression or silencing of a gene by introducing a foreign DNA and modifying the cellular signalling.62 NPs have a capability to replace viral vectors as they are small in size and therefore can communicate with many biological moieties like cytokines and proteins. Although they possess some drawbacks, such as inefficient transfecting efficiency, these can be overcome by chemical modification of the functional groups.63 Magnetic NPs have been used in gene therapy by intercalation of the functional gene with the SPION and its effective transfection into the desired cell by high gradient magnets. Morishita et al. demonstrated magnetic NPs, with cell fusion vectors hemagglutinating virus of Japan envelope with protamine sulfate magnetic NPs, showed a significantly improved transfection efficiency in presence of a magnetic source along with reduced toxicity in BHK21 cells.64,65
4.3 Thermal Therapy
NPs can be used in thermal therapies such as photothermal ablation and magnetic hyperthermia, due to unique surface plasmon resonance or magnetic susceptiblility of NPs.66 For instance, the magnetic NPs continuously emit heat via Néel and Brownian relaxation pathways upon exposure to the alternating external magnetic field.9 Noble metal nanostructures such as nanorods,21, 67, 68 multi-branched particles,69, 70 nanoshells,71, 72 and hollow-shells73, 74 have been used for photothermal therapeutic applications with non-invasive therapy. Stern et al. have recently demonstrated an NIR activated gold nanoshell as a preclinical treatment modality-which completely enabled photothermal destruction of human prostate cancer in xenograft model.75 These NPs can be utilized as promising drug carriers for thermally triggering of drugs as well as thermal therapy.
4.4 Photodynamic Therapy
Photodynamic therapy is used for selective destruction of cancer cells and tissues by utilization of photosensitizers. When an external light excites the photosensitizers in the presence of oxygen molecules, the photosensitizers produce toxic singlet oxygen species, which lead to cell death.76 Tsay et al. designed peptide-coated QD-photosensitizer conjugates using rose bengal and chlorin e6 photosensitizers.77 These photosensitizers were covalently bound to the peptides-overcoated green and red CdSe/CdS/ZnS QDs. The production of singlet oxygen was enhanced by direct or indirect activation of the photosensitizers. Upon activation, these photosensitizers could perform both imaging and therapeutic activities.
5. Theranostic Applications and Pharmacodynamics of NPs
The application of theranostic NPs has probably been most successfully implanted in cancer research. For theranostic and clinical applications, however, most NPs should have inert surface coatings with organic polymeric and/or biological materials as discussed above. In addition, selective targeting is an essential property to overcome one of the limitations of conventional therapy and to minimize potential side effects. There are two major approaches for efficient tumor targeting: In passive targeting, therapeutic NPs reach the tumor site through leaky endothelium surrounding tumor tissues (enhanced permeability and retention (EPR) effect). In contrast, active targeting is based on targeting ligands, such as antibody, aptamer and peptide, on the NP surface which allow NPs to bind to the receptors overexpressed on cancer cells. In this section, we introduce several advanced examples of theranostic nanoplatforms.
5.1 Synthetic polymer NPs
Photosensitizer-conjugated amine functionalized polyacrylamide NPs synthesized by oil-in-water microemulsion technique have been reported by Kopelman and co-wokers.54 For tumor-specific targeting, the surface of NPs was modified with cell-permeable peptide and biologically inert PEG. Once fluorophore-embedded NPs enter the tumor, the fluorescence dye lights up the tumor cells and the drug is photosensitized by irradiation, which specifically kills the cancer cells. In addition, Liu et al. reported polyelectrolyte-based polyprodrugs which possess imaging, chemotherapeutic and photodynamic properties.31 The NPs were covalently conjugated to doxorubicin through a reactive oxygen species (ROS) cleavable linker. PEGylated polyelectrolytes efficiently produce ROS under light irradiation, which then not only kill the cancerous cells by photosensitization but also can release doxorubicin for chemotherapy. Light-triggered chemotherapy and photodynamic therapy have been combined to produce better results to cure cancers with synergistic advantages such as overcoming multiple drug resistance and improved therapeutic efficacy.31
5.2 Biological NPs (Naturally Derived Polymers)
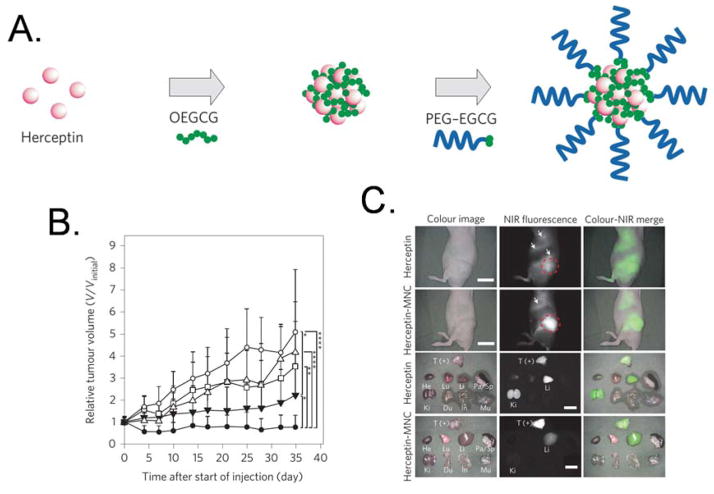
The self-assembled micellar nanocomplex (MNC) has been developed for delivery of protein drugs. Chung and Kurisawa’s group reported that simple sequential self-assembly of the epigallocatechin-3-O-gallate (EGCG) derivative, a major ingredient of green tea, with anticancer protein leads to the formation of stable micellar nanocomplex.78 The anticancer effect of Herceptin-loaded micellar nanocomplex (Herceptin–MNC) was investigated in vitro and in vivo and compared with those of bovine serum albumin (BSA)-MNC and free Herceptin. Herceptin–MNC exhibited a 2.3-fold greater accumulation in the tumor site, 29-fold longer blood half-life, and significantly higher anticancer effect in the tumor in comparison with free Herceptin (Figure 4).
Figure 4.
a) Schematic diagram of the self-assembly process used to form the micellar nanocomplexes, which are formed via two sequential self-assemblies in an aqueous solution: complexation of OEGCG with proteins to form the core, followed by complexation of PEG–EGCG surrounding the preformed core to form the shell. b) Anticancer effect on BT-474-xenografted nude mouse model. PBS (vehicle control, open circles), BSA–MNC (open triangles), Herceptin (2.5 mg/kg, open squares), sequential injection of BSA–MNC and Herceptin (filled inverted triangles) and Herceptin–MNC (filled circles). c) Real-time intraoperative tumor detection and NIR fluorescence image-guided resection at 24 h post-injection. Arrows indicate nonspecific uptake (liver, kidneys, intestine). The red dashed circle delineates the region of interest. Abbreviations used are: BSA, bovine serum albumin; EGCG, Epigallocatechin-3-O-gallate; MNC, micellar nanocomplex; OEGCG, oligomerized EGCG; PEG, polyethylene glycol; T (+), positive tumor. Reprinted with permission from ref. 78. Copyright 2014 Nature Publishing Group.
5.3 Mesoporous Silica NPs
Mesoporous silica NPs have been used successfully in cancer therapy, mainly because of large surface area and pore volume, and ease of surface modification.13, 79 Recently, magnetic NPs or gold NPs were embedded into mesoporous silica NPs for thermally triggered drug release. An anticancer drug was loaded into porous cavities of mesoporous silica NPs and porous structures were capped with thermally releasable molecule. When external stimuli, such as magnetic field and NIR laser, are applied to these mesoporous silica nanoplatforms, drug release can be controlled precisely. This controlled release behavior is a very important feature in target specific therapy as it can overcome the side effects of conventional drug delivery system.
5.4 Magnetic NPs
Another example of theranostic NPs in cancer is the use of magnetic NPs (MNPs). MNP-based theranostics can be divided into three ways in terms of therapeutic methods: 1) hydrophobic drug or gene delivery, 2) thermal therapy in the magnetic field, and 3) magnetic/mechanical controlling in cell signalling. Theranostic MNPs normally contain a superparamagnetic iron oxide core, which is used for MRI to detect the tumor, covered by a hydrophilic surface coat on the outside, and have been linked with an anticancer drug or siRNA to treat the tumor.80 As one of the key examples, Moore and co-workers have reported dextran-coated SPIONs for in vivo siRNA delivery.11 The amine-dextran coated SPIONs were labelled with Cy5.5 dye for simultaneous optical imaging, and covalently linked to thiolated siRNA duplex and myristoylated polyarginine peptides, which are membrane translocation modules, for intracellular delivery. This study showed advancement of siRNA delivery and silencing with imaging strategies. MNPs can be also developed by conjugating chemotherapeutic drugs on the surface of NPs to target and treat cancers. Lee et al.81 have also developed a nanocarrier containing MNP conjugated to the anticancer drug Gemcitabin. These NPs deliver the drug by receptor-mediated endocytosis to its target, urokinase plasminogen activator receptor and also allows in vivo MRI of the tumor.81
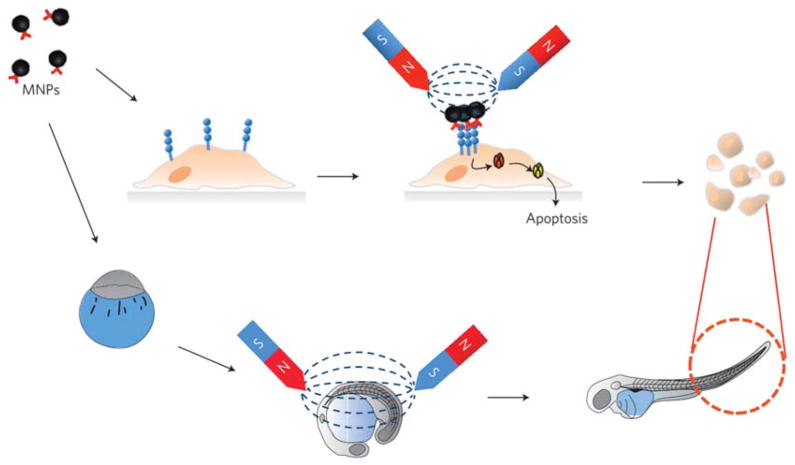
Magnetic thermal therapy utilizes heat induced from MNPs in external high frequency alternating magnetic field, which allowed us to control heat generation after specific targeting to tumor region of interest.82 Although external triggering is one of the advantages in magnetic field induced thermal therapy, the efficacy is limited even with high concentration of therapeutic MNPs. Very recently, to control cell signalling, a magnetic switch method has been developed by using zinc-doped iron oxide MNPs.83 The thiolated MNPs were conjugated with antibody for targeting death receptor 4 (DR4) of DLD-1 colon cancer cells. When a magnetic field is applied to MNP bound DR4s on DLD-1 cells, clustering of DR4s was formed and apoptosis signalling pathways were induced. For in vivo apoptosis experiment, the magnetic switch method was applied to zebrafish and apoptotic morphology changes of zebrafish could be observed in the magnetically activated group (Figure 5).
Figure 5.
A schematic representation of the magnetic switch for apoptosis signalling in in vitro cells and zebrafish. MNPs first bind to the death receptors, and subsequent aggregation on the application of a focused magnetic field triggers extrinsic apoptosis signalling. Magnetic switching of death receptor clustering results in the death of cells and also causes morphological changes in zebrafish. Reprinted with permission from ref. 83. Copyright 2012 Nature Publishing Group.
6. Toxicity of Theranostic NPs
Although the development of NPs has advanced the field of drug delivery and nanomedicine, it is relatively new to other forms of pharmaceutical formulations. Moreover, certain mechanisms by which nanosystems can lead to toxicity have not been fully characterized in a proper manner,84 likely because there are only a small number of nanomaterial drugs approved by the FDA.2 The toxicity of NPs depends on various conditions, including not only physicochemical properties of NP (e.g. HD size, shape, surface charge, and chemical composition), but physiological status (e.g. genetics, disease conditions). For example, people with coronary artery diseases are more prone to heart attack when they are subject to NP therapy.85 Although small size and large surface area are two of the unique properties making NPs popular, these properties also significantly affect toxicity.40 Since NPs are smaller in size than cells and cell organelles86 and they possess the potential to penetrate into these cellular structures in several organs by circulatory, nervous and lymphatic systems, they can disrupt physiological functions and promote tissue inflammation, abnormal cell functioning or even cell death.46
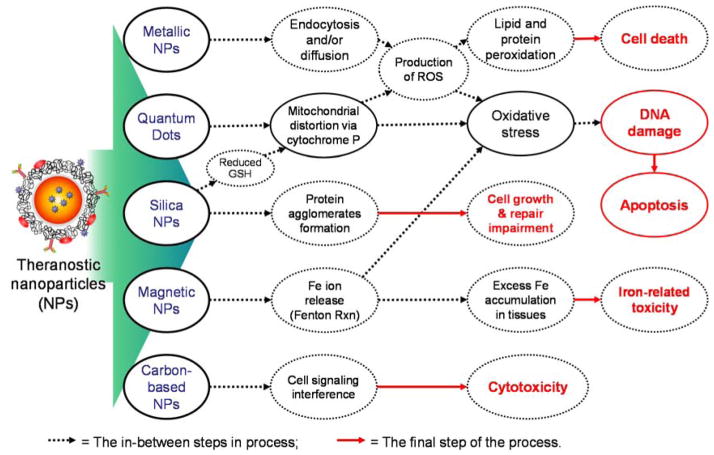
Nanomaterials, involving dendrimers, cationic polymers, QDs, magnetic and metallic NPs, and carbon based materials, have been widely used as biosensors, contrast agents, and drug carriers.87 However, the toxicity of these NPs is still a problem to in vivo applications. Metallic NPs (e.g. silver, gold) can enter the cells either by endocytosis or diffusion due to their relative small size. Upon their entry into the cell, they reach mitochondria and can impair the mitochondrial function by disturbing the electron transport chain, resulting in causing oxidative stress. Moreover, these metallic NPs can generate ROS, which enter the nucleus and cause oxidative stress, leading to DNA damage by cross-linking or formation of DNA adducts. All these damages ultimately promote cell death. The ROS can also cause protein oxidation and lipid peroxidation, which hinder cell growth ultimately. Silica NPs reduce levels of antioxidant glutathione as well as causing oxidative stress via ROS production, resulting in DNA damage. In addition, silica NPs can induce the formation of protein agglomerates and inhibit the cell growth. QDs can cause toxicity by disruption of mitochondrial function, which leads to DNA damage.88 SPIONs can release iron, which can be taken up by cells through iron transporters or SPIONs themselves can be endocytosed into the cell and then release iron by lysosomal degradation of the SPION. The intracellular iron can mediate the Fenton reaction to produce ROS, which enters the nucleus and damages DNA.89
Furthermore, iron can accumulate in tissues and cause iron-related toxicity.90–93 Carbon-based nanomaterials (e.g. carbon nanotubes and graphene) are known to interfere with the cell signaling and cause cytotoxicity.94 Toxicity mechanisms of different types of NPs are summarized in Figure 6. In this section, we describe more examples of NP toxicities and discuss the relationship between toxicity and physicochemical properties of NPs.
Figure 6.
Toxicity mechanisms of different types of NPs.
6.1 Toxicity of cationic lipid and polymer NPs
Cationic lipids and polymers as non-viral vectors have been developed for gene delivery because of their relative safety, capacity to transfer large genes, site-specificity and their low immune response.95 However, toxicities of cationic lipids and polymers in gene vectors are still an obstacle to the application of non-viral vectors to gene therapy. The cytotoxic effect is associated with the cationic nature that can interact with critical enzymes such as protein kinase C.96 To overcome toxicity problems, degradable polymers in a low pH environment have been developed. For example, the toxicity of low molecular weight polyethylenimine polymers is found to be reduced because of the degradation of acid-labile linkage.96, 97 In addition, PEGylation polycomplex shows a marked decrease in the toxicity compared with non-PEGylated cationic polymer.98
6.2 Toxicity of metallic NPs
The in vivo use of heavy metals is debatable with regard to safety concerns. Divalent metal ions even in small concentrations are toxic as they accumulate in the kidneys and become nephrotoxic. Cadmium and selenium are two metals chiefly used in making the core of QDs and these metals are related to moderate toxicities in vertebrates. It has been reported that, under oxidative stress, cadmium ions are released and bind to the sulfhydryl group in the mitochondria and cause a loss in their function, leading to cell poisoning.99 Metallic NPs (e.g. gold NPs) have been frequently used for diagnosis and therapy. They offer several advantages, such as low toxicity in comparison to QDs and better potential as a contrast agent than organic dyes and better surface and refractive index spectroscopic properties.100 Although gold NPs are thought to possess low toxicity, certain forms like the cationic gold nanospheres could exhibit moderate cellular toxicity, whereas their anionic counterparts are non-toxic. The toxicity of the cationic form could be explained by interaction with the negatively charged membrane, leading to distortion of the cell membrane.101 Silver NPs have also shown to cause oxidative stress in the brain94 and a few other tissues like liver102 by reducing the respiratory chain complexes I, II, II and IV of mitochondria.94 These particles can also induce necrosis and apoptosis.103 SPIONs are composed of hematite, magnetite or maghemite core which is coated with organic or inorganic polymers. And, SPIONs have a unique property called superparamagnetism in which each magnetic particle has a distinct magnetic moment and acts like a superparamagnetic atom and spontaneously responds to external magnetic fields.104 These NPs have been actively used as imaging tools in MRI, in targeted drug delivery, induced hyperthermia cancer therapy. However, these particles also have several concerns about toxicity because they can generate a huge amount of free radicals due to redox cycling at the surface of particle. The ROS can promote oxidative stress and interfere with the cellular functioning. Furthermore, since SPIONs contain iron, they can lead to excessive accumulation of iron at the target site when the SPIONs are given in excess, which can lead to metal toxicity related to iron overload in the target tissue.105
6.3 Toxicity of carbon based nanomaterials
Carbon nanotubes (CNTs) are tubular cylinders of carbon atoms and can either be single-walled CNT (CWCNT) or multi-walled CNT (MWCNT). Although CNTs have attracted great interest due to their remarkable tunablity, and ability to incorporate multiple functionalities, it is necessary to disperse entangled/bundled CNTs uniformly in solution without impurities.106 Studies have shown that the aggregation of MWCNTs causes toxicity in tissues by depositing there due to the presence of larger particles when exposed for long time, whereas the SWCNTs that are composed of smaller particles undergo phagocytosis and are non-toxic. Also, cationic carbon nanotubes could be more toxic than the neutral or negatively charged CNTs since they can cause platelet aggregation.107 Graphene oxide (GO) has been found to be one of the promising candidates as nanocarriers due to its unique 2D shape.108, 109 However, uncoated GO exhibits in vivo toxicity such as inducing blood clots or pulmonary edema and granuloma through accumulation in lungs after intravenous administration.110, 111 Yang et al. reviewed the behavior and the toxic effects of graphene and its derivatives in different biological organisms and suggested that their physicochemical properties majorly from the functional groups on the surface could determine the in vivo behavior and toxicity.109
6.4 Toxicity of mesoporous silica NPs
Mesoporous silica NPs have been used as theranostics in cancer therapy since they can target the tumor and release the drug in a controlled manner. However, it was found that, despite reduced toxicity compared with colloidal silica, mesoporous silica NPs could still induce cytotoxicity associated with oxidative stress by increased ROS production and decreased GSH levels, which can ultimately lead to cell death.112, 113 It has been known that oxidative stress and apoptosis are responsible for the dysfunction of endothelial cells and several cardiovascular disorders like bradycardia and pericardial toxicity.114 Silica NPs can also inhibit gene expression by forming protein agglomerates and can interfere with processes involved in the cell growth and replication.115,94 Although NPs have a huge potential for diagnosis and therapy, they also possess the capacity to cause severe toxic effects in the body and thus their formulation should be carefully designed keeping their toxic properties in mind.
7. Future Directions
Many different types of theranostic NPs have been successfully implicated in targeting and therapy of various diseases. Anti-cancer agents may be enclosed within or embedded on different NP structures for targeted imaging and therapy. In fact, most therapeutic NPs boast unique size-dependent optical or magnetic properties, multifunctionality, strong EPR effect and long-term blood circulation. However, NPs are insufficient to meet some important properties for clinical translation such as high physiological stability, efficient clearance, minimum accumulation in non-targeted tissues and organs, and rapid distribution to various organs and tissues.44 Also, clinical applications of most NPs have been hampered by lack of comprehensive knowledge of toxicities of individual NPs. More studies in the context of preclinical and predictive toxicology are warranted to address this question.
In terms of therapeutic method, the majority of applications have been related to curing cancers by killing cancerous cells by drugs, heat, and gene transfections. Very recently, a promising theranostic application of multifunctional NPs has been proposed for diagnosis and treatment of chronic neurodegenerative disorders such as Alzheimer’s disease, Parkinson’s disease and strokes.116, 117 For instance, multifunctional NPs can applied for the amelioration of brain disorders that are associated with iron overload. Oxidative stress caused by metals like iron, which accumulate in the brain in excess, is considered one of the major causes of many neurodegenerative diseases, including Alzheimer’s disease.118 Iron chelators have been extensively used with the aim to remove the excess iron from the brain to improve these disease conditions. An ideal chelator should demonstrate increased specificity to the target tissues with minimal distribution into other tissues and organs. Unfortunately, this is the major drawback of any chelators because they do not act via specific targeting mechanism and bind to several tissues which can cause different adverse effects and toxicities.119 Gang et al.120, 121 proposed a possible mechanism using NPs in iron chelation therapy for treating Alzheimer’s disease based on the fact that NPs are capable of crossing the blood-brain barrier through apolipoprotein E (ApoE) or low density lipoprotein (LDL) receptor in the brain by imitating LDL. This mechanism allows NPs to enter the brain where they chelate the excess iron and exit the brain with the chelated complex. This would reduce the toxicity of chelators since they would be directed to the brain and the therapy would prove to be more efficient since less amount of chelators would be needed for therapeutic effects with reduced side effects.120 This mechanism seems plausible and, if developed properly, the formulations could be a major breakthrough in treating not only Alzheimer’s disease, but many other neurodegenerative disorders. However, other tissues could exert receptor-mediated uptake of these NPs and deplete iron that is required for essential physiological function. While rigorous assessments of toxicity are needed, NPs with imaging modality could track the real-time PK/pharmacodynamics and help to design better formulations to avoid the distribution into non-target tissues. In addition, nanobiosensors including nanowell arrays and microfluidic chips with ultra-high signalling accuracy can be utilized for ex vivo molecular diagnosis, since it is important to validate the in vivo functionality of NPs by analysing nanoliters of biosamples (i.e., blood, urine or cerebrospinal fluid) with detecting isolating single cells, toxic moieties or secreted proteins.122 This will enhance clinical benefits of theranostic NPs and further contribute to the development of advanced, multifunctional NPs.
Supplementary Material
Acknowledgments
This study was supported by the following grants from NIH/NIBIB grant #R01-EB-011523 (H.S.C.), NIH/NIEHS grant #R00-ES-017781 (J.K.) and Basic Science Research Program through the National Research Foundation (NRF) of Korea funded by the Ministry of Science, ICT & Future Planning (NRF-2014-R1A6A3A03057790); the contents of this paper are solely the responsibility of the authors and do not necessarily represent the official views of the NIH.
Abbreviations
- BSA
bovine serum albumin
- CNT
carbon nanotube
- CT
computed tomography
- EPR
enhanced permeability and retention
- HD
hydrodynamic diameter
- LDL
low density lipoprotein
- MNC
micellar nanocomplex
- MRI
magnetic resonance imaging
- NNI
National Nanotechnology Initiative
- NIR
near infrared
- NP
nanoparticle
- PA
photoacoustic
- PEG
polyethylene glycol
- PET
positron emission tomography
- PK
pharmacokinetics
- QD
quantum dot
- RNA
Ribonucleic acid
- ROS
reactive oxygen species
- SPECT
single photon emission computed tomography
- SPION
superparamagnetic iron oxide nanoparticle
- US
ultrasound
Contributor Information
Hak Soo Choi, Email: hchoi@bidmc.harvard.edu.
Jonghan Kim, Email: j.kim@neu.edu.
Notes and references
- 1.Xie J, Lee S, Chen X. Adv Drug Delivery Rev. 2010;62:1064–1079. doi: 10.1016/j.addr.2010.07.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Choi HS, Frangioni JV. Molecular imaging. 2010;9:291–310. [PMC free article] [PubMed] [Google Scholar]
- 3.Barros ALBd, Soares DCF. J Mol Pharm Org Process Res. 2014;2:e113. [Google Scholar]
- 4.Chen F, Ehlerding EB, Cai W. J Nucl Med. 2014;55:1919–1922. doi: 10.2967/jnumed.114.146019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Janib SM, Moses AS, MacKay JA. Adv Drug Delivery Rev. 2010;62:1052–1063. doi: 10.1016/j.addr.2010.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Approaches to safe nanotechnology: managing the health and safety concerns associated with engineered nanomaterials. Atlanta: National Institute for Occupational Safety and Health; 2009. [Google Scholar]
- 7.Ahmed N, Fessi H, Elaissari A. Drug Discov Today. 2012;17:928–934. doi: 10.1016/j.drudis.2012.03.010. [DOI] [PubMed] [Google Scholar]
- 8.Allegra A, Penna G, Alonci A, Rizzo V, Russo S, Musolino C. Anti-Cancer Agents Med Chem. 2011;11:669–686. doi: 10.2174/187152011796817682. [DOI] [PubMed] [Google Scholar]
- 9.Yoo D, Lee JH, Shin TH, Cheon J. Acc Chem Res. 2011;44:863–874. doi: 10.1021/ar200085c. [DOI] [PubMed] [Google Scholar]
- 10.Namiki Y, Namiki T, Yoshida H, Ishii Y, Tsubota A, Koido S, Nariai K, Mitsunaga M, Yanagisawa S, Kashiwagi H, Mabashi Y, Yumoto Y, Hoshina S, Fujise K, Tada N. Nat Nanotechnol. 2009;4:598–606. doi: 10.1038/nnano.2009.202. [DOI] [PubMed] [Google Scholar]
- 11.Medarova Z, Pham W, Farrar C, Petkova V, Moore A. Nat Med. 2007;13:372–377. doi: 10.1038/nm1486. [DOI] [PubMed] [Google Scholar]
- 12.Chen AA, Derfus AM, Khetani SR, Bhatia SN. Nucleic Acids Res. 2005;33:e190. doi: 10.1093/nar/gni188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lee JE, Lee N, Kim T, Kim J, Hyeon T. Acc Chem Res. 2011;44:893–902. doi: 10.1021/ar2000259. [DOI] [PubMed] [Google Scholar]
- 14.Hu SH, Chen YW, Hung WT, Chen IW, Chen SY. Adv Mater. 2012;24:1748–1754. doi: 10.1002/adma.201104070. [DOI] [PubMed] [Google Scholar]
- 15.Tian B, Wang C, Zhang S, Feng L, Liu Z. ACS Nano. 2011;5:7000–7009. doi: 10.1021/nn201560b. [DOI] [PubMed] [Google Scholar]
- 16.Zhang L, Xia J, Zhao Q, Liu L, Zhang Z. Small. 2010;6:537–544. doi: 10.1002/smll.200901680. [DOI] [PubMed] [Google Scholar]
- 17.Bianco A, Kostarelos K, Prato M. Curr Opin Chem Biol. 2005;9:674–679. doi: 10.1016/j.cbpa.2005.10.005. [DOI] [PubMed] [Google Scholar]
- 18.Kam NWS, O’Connell M, Wisdom JA, Dai H. Proc Natl Acad Sci U S A. 2005;102:11600–11605. doi: 10.1073/pnas.0502680102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Liu Z, Chen K, Davis C, Sherlock S, Cao Q, Chen X, Dai H. Cancer Res. 2008;68:6652–6660. doi: 10.1158/0008-5472.CAN-08-1468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Bardhan R, Lal S, Joshi A, Halas NJ. Acc Chem Res. 2011;44:936–946. doi: 10.1021/ar200023x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.von Maltzahn G, Park JH, Agrawal A, Bandaru NK, Das SK, Sailor MJ, Bhatia SN. Cancer Res. 2009;69:3892–3900. doi: 10.1158/0008-5472.CAN-08-4242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Huang X, El-Sayed IH, Qian W, El-Sayed MA. J Am Chem Soc. 2006;128:2115–2120. doi: 10.1021/ja057254a. [DOI] [PubMed] [Google Scholar]
- 23.Zhou M, Zhang R, Huang M, Lu W, Song S, Melancon MP, Tian M, Liang D, Li C. J Am Chem Soc. 2010;132:15351–15358. doi: 10.1021/ja106855m. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Guo L, Yan DD, Yang D, Li Y, Wang X, Zalewski O, Yan B, Lu W. ACS Nano. 2014;8:5670–5681. doi: 10.1021/nn5002112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Liu T, Wang C, Gu X, Gong H, Cheng L, Shi X, Feng L, Sun B, Liu Z. Adv Mater. 2014;26:3433–3440. doi: 10.1002/adma.201305256. [DOI] [PubMed] [Google Scholar]
- 26.Kou Z, Wang X, Yuan R, Chen H, Zhi Q, Gao L, Wang B, Guo Z, Xue X, Cao W, Guo L. Nanoscale Res Lett. 2014;9:587. doi: 10.1186/1556-276X-9-587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ng KK, Lovell JF, Zheng G. Acc Chem Res. 2011;44:1105–1113. doi: 10.1021/ar200017e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Zhang Z, Cao W, Jin H, Lovell JF, Yang M, Ding L, Chen J, Corbin I, Luo Q, Zheng G. Angew Chem Int Ed. 2009;48:9171–9175. doi: 10.1002/anie.200903112. [DOI] [PubMed] [Google Scholar]
- 29.Koo H, Huh MS, Sun IC, Yuk SH, Choi K, Kim K, Kwon IC. Acc Chem Res. 2011;44:1018–1028. doi: 10.1021/ar2000138. [DOI] [PubMed] [Google Scholar]
- 30.Cabral H, Nishiyama N, Kataoka K. Acc Chem Res. 2011;44:999–1008. doi: 10.1021/ar200094a. [DOI] [PubMed] [Google Scholar]
- 31.Yuan Y, Liu J, Liu B. Angew Chem Int Ed. 2014;53:7163–7168. doi: 10.1002/anie.201402189. [DOI] [PubMed] [Google Scholar]
- 32.Majoros IJ, Thomas TP, Mehta CB, Baker JR. J Med Chem. 2005;48:5892–5899. doi: 10.1021/jm0401863. [DOI] [PubMed] [Google Scholar]
- 33.Wang Y, Guo R, Cao X, Shen M, Shi X. Biomaterials. 2011;32:3322–3329. doi: 10.1016/j.biomaterials.2010.12.060. [DOI] [PubMed] [Google Scholar]
- 34.Namiki Y, Fuchigami T, Tada N, Kawamura R, Matsunuma S, Kitamoto Y, Nakagawa M. Acc Chem Res. 2011;44:1080–1093. doi: 10.1021/ar200011r. [DOI] [PubMed] [Google Scholar]
- 35.PURI A, BLUMENTHAL R. Acc Chem Res. 2011;44:1071–1079. doi: 10.1021/ar2001843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Al-Jamal WT, Kostarelos K. Acc Chem Res. 2011;44:1094–1104. doi: 10.1021/ar200105p. [DOI] [PubMed] [Google Scholar]
- 37.Choi HS, Ashitate Y, Lee JH, Kim SH, Matsui A, Insin N, Bawendi MG, Semmler-Behnke M, Frangioni JV, Tsuda A. Nat Biotechnol. 2010;28:1300–1303. doi: 10.1038/nbt.1696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Choi HS, Ipe BI, Misra P, Lee JH, Bawendi MG, Frangioni JV. Nano Lett. 2009;9:2354–2359. doi: 10.1021/nl900872r. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Choi HS, Liu W, Liu F, Nasr K, Misra P, Bawendi MG, Frangioni JV. Nat Nanotechnol. 2010;5:42–47. doi: 10.1038/nnano.2009.314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Choi HS, Liu W, Misra P, Tanaka E, Zimmer JP, Itty Ipe B, Bawendi MG, Frangioni JV. Nat Biotechnol. 2007;25:1165–1170. doi: 10.1038/nbt1340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Li SD, Huang L. Mol Pharm. 2008;5:496–504. doi: 10.1021/mp800049w. [DOI] [PubMed] [Google Scholar]
- 42.Phillips E, Penate-Medina O, Zanzonico PB, Carvajal RD, Mohan P, Ye Y, Humm J, Gönen M, Kalaigian H, Schöder H. Science translational medicine. 2014;6:260ra149–260ra149. doi: 10.1126/scitranslmed.3009524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Liu J, Yu M, Zhou C, Yang S, Ning X, Zheng J. J Am Chem Soc. 2013;135:4978–4981. doi: 10.1021/ja401612x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Yu M, Zheng J. ACS Nano. 2015;9:6655–6674. doi: 10.1021/acsnano.5b01320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Wang Z, Malik AB. Ther Deliv. 2013;4:131–133. doi: 10.4155/tde.12.140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Wei A, Mehtala JG, Patri AK. J Controlled Release. 2012;164:236–246. doi: 10.1016/j.jconrel.2012.10.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Moghimi SM, Hunter AC, Andresen TL. Annu Rev Pharmacol Toxicol. 2012;52:481–503. doi: 10.1146/annurev-pharmtox-010611-134623. [DOI] [PubMed] [Google Scholar]
- 48.Salatin S, Dizaj SM, Khosroushahi AY. Cell Biol Int. 2015 doi: 10.1002/cbin.10459. [DOI] [Google Scholar]
- 49.Sonaje K, Lin KJ, Wey SP, Lin CK, Yeh TH, Nguyen HN, Hsu CW, Yen TC, Juang JH, Sung HW. Biomaterials. 2010;31:6849–6858. doi: 10.1016/j.biomaterials.2010.05.042. [DOI] [PubMed] [Google Scholar]
- 50.Zhang XD, Wu HY, Wu D, Wang YY, Chang JH, Zhai ZB, Meng AM, Liu PX, Zhang LA, Fan FY. Int J Nanomedicine. 2010;5:771–781. doi: 10.2147/IJN.S8428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Harivardhan Reddy L, Sharma RK, Chuttani K, Mishra AK, Murthy RS. Journal of controlled release: official journal of the Controlled Release Society. 2005;105:185–198. doi: 10.1016/j.jconrel.2005.02.028. [DOI] [PubMed] [Google Scholar]
- 52.Chertok B, David AE, Yang VC. Journal of controlled release: official journal of the Controlled Release Society. 2011;155:393–399. doi: 10.1016/j.jconrel.2011.06.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Huang X, Zhang F, Zhu L, Choi KY, Guo N, Guo J, Tackett K, Anilkumar P, Liu G, Quan Q, Choi HS, Niu G, Sun YP, Lee S, Chen X. ACS Nano. 2013;7:5684–5693. doi: 10.1021/nn401911k. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Wang S, Kim G, Lee YE, Hah HJ, Ethirajan M, Pandey RK, Kopelman R. ACS Nano. 2012;6:6843–6851. doi: 10.1021/nn301633m. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Lammers T, Aime S, Hennink WE, Storm G, Kiessling F. Acc Chem Res. 2011;44:1029–1038. doi: 10.1021/ar200019c. [DOI] [PubMed] [Google Scholar]
- 56.Blanco E, Shen H, Ferrari M. Nature biotechnology. 2015;33:941–951. doi: 10.1038/nbt.3330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Park K, Lee S, Kang E, Kim K, Choi K, Kwon IC. Advanced Functional Materials. 2009;19:1553–1566. [Google Scholar]
- 58.Willmann JK, van Bruggen N, Dinkelborg LM, Gambhir SS. Nat Rev Drug Discovery. 2008;7:591–607. doi: 10.1038/nrd2290. [DOI] [PubMed] [Google Scholar]
- 59.Wu D, Huang L, Jiang MS, Jiang H. Int J Mol Sci. 2014;15:23616–23639. doi: 10.3390/ijms151223616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Zamboni WC. Clinical cancer research: an official journal of the American Association for Cancer Research. 2005;11:8230–8234. doi: 10.1158/1078-0432.CCR-05-1895. [DOI] [PubMed] [Google Scholar]
- 61.Bagalkot V, Zhang L, Levy-Nissenbaum E, Jon S, Kantoff PW, Langer R, Farokhzad OC. Nano letters. 2007;7:3065–3070. doi: 10.1021/nl071546n. [DOI] [PubMed] [Google Scholar]
- 62.Schillinger U, Brill T, Rudolph C, Huth S, Gersting S, Krötz F, Hirschberger J, Bergemann C, Plank C. Journal of Magnetism and Magnetic Materials. 2005;293:501–508. [Google Scholar]
- 63.Perez-Martinez FC, Carrion B, Cena V. Journal of Alzheimer’s disease: JAD. 2012;31:697–710. doi: 10.3233/JAD-2012-120661. [DOI] [PubMed] [Google Scholar]
- 64.Morishita N, Nakagami H, Morishita R, Takeda S-i, Mishima F, Terazono Bungo, Nishijima S, Kaneda Y, Tanaka N. Biochemical and Biophysical Research Communications. 2005;334:1121–1126. doi: 10.1016/j.bbrc.2005.06.204. [DOI] [PubMed] [Google Scholar]
- 65.Dobson J. Gene therapy. 2006;13:283–287. doi: 10.1038/sj.gt.3302720. [DOI] [PubMed] [Google Scholar]
- 66.Shenoi MM, Shah NB, Griffin RJ, Vercellotti GM, Bischof JC. Nanomedicine. 2011;6:545–563. doi: 10.2217/nnm.10.153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Huang XH, El-Sayed IH, Qian W, El-Sayed MA. J Am Chem Soc. 2006;128:2115–2120. doi: 10.1021/ja057254a. [DOI] [PubMed] [Google Scholar]
- 68.Dembereldorj U, Choi SY, Ganbold EO, Song NW, Kim D, Choo J, Lee SY, Kim S, Joo SW. Photochem Photobiol. 2014;90:659–666. doi: 10.1111/php.12212. [DOI] [PubMed] [Google Scholar]
- 69.Yuan H, Fales AM, Vo-Dinh T. J Am Chem Soc. 2012;134:11358–11361. doi: 10.1021/ja304180y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Nergiz SZ, Gandra N, Tadepalli S, Singamaneni S. ACS Appl Mater Interfaces. 2014 doi: 10.1021/am504795d. [DOI] [PubMed] [Google Scholar]
- 71.Ayala-Orozco C, Urban C, Knight MW, Urban AS, Neumann O, Bishnoi SW, Mukherjee S, Goodman AM, Charron H, Mitchell T. ACS Nano. 2014 doi: 10.1021/nn501871d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Noh MS, Lee S, Kang H, Yang JK, Lee H, Hwang D, Lee JW, Jeong S, Jang Y, Jun BH, Jeong DH, Kim SK, Lee YS, Cho MH. Biomaterials. 2015;45:81–92. doi: 10.1016/j.biomaterials.2014.12.036. [DOI] [PubMed] [Google Scholar]
- 73.Xia Y, Li W, Cobley CM, Chen J, Xia X, Zhang Q, Yang M, Cho EC, Brown PK. Acc Chem Res. 2011;44:914–924. doi: 10.1021/ar200061q. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Jang H, Kim YK, Huh H, Min DH. ACS Nano. 2014;8:467–475. doi: 10.1021/nn404833b. [DOI] [PubMed] [Google Scholar]
- 75.Stern JM, Stanfield J, Kabbani W, Hsieh JT, Cadeddu JA. The Journal of urology. 2008;179:748–753. doi: 10.1016/j.juro.2007.09.018. [DOI] [PubMed] [Google Scholar]
- 76.Chatterjee DK, Fong LS, Zhang Y. Advanced Drug Delivery Reviews. 2008;60:1627–1637. doi: 10.1016/j.addr.2008.08.003. [DOI] [PubMed] [Google Scholar]
- 77.Tsay JM, Trzoss M, Shi L, Kong X, Selke M, Jung ME, Weiss S. Journal of the American Chemical Society. 2007;129:6865–6871. doi: 10.1021/ja070713i. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Chung JE, Tan S, Gao SJ, Yongvongsoontorn N, Kim SH, Lee JH, Choi HS, Yano H, Zhuo L, Kurisawa M, Ying JY. Nat Nanotechnol. 2014;9:907–912. doi: 10.1038/nnano.2014.208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Chen NT, Cheng SH, Souris JS, Chen CT, Mou CY, Lo LW. J Mater Chem B. 2013;1:3128–3135. doi: 10.1039/c3tb20249f. [DOI] [PubMed] [Google Scholar]
- 80.Berezin MY. Nanotechnology for Biomedical imaging and diagnosis: from nanoparticle design to clinical applications. John Wiley & Sons; Hobokin, New Jersey: 2015. [Google Scholar]
- 81.Lee GY, Qian WP, Wang L, Wang YA, Staley CA, Satpathy M, Nie S, Mao H, Yang L. ACS Nano. 2013;7:2078–2089. doi: 10.1021/nn3043463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Lee JH, Kim JW, Cheon J. Mol Cells. 2013;35:274–284. doi: 10.1007/s10059-013-0103-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Cho MH, Lee EJ, Son M, Lee JH, Yoo D, Kim JW, Park SW, Shin JS, Cheon J. Nat Mater. 2012;11:1038–1043. doi: 10.1038/nmat3430. [DOI] [PubMed] [Google Scholar]
- 84.Love SA, Maurer-Jones MA, Thompson JW, Lin YS, Haynes CL. Annu Rev Anal Chem. 2012;5:181–205. doi: 10.1146/annurev-anchem-062011-143134. [DOI] [PubMed] [Google Scholar]
- 85.Seaton A, Donaldson K. The Lancet. 2005;365:923–924. doi: 10.1016/S0140-6736(05)71061-8. [DOI] [PubMed] [Google Scholar]
- 86.Medina C, Santos-Martinez MJ, Radomski A, Corrigan OI, Radomski MW. Br J Pharmacol. 2007;150:552–558. doi: 10.1038/sj.bjp.0707130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Chen X. Nanoplatform-based molecular imaging. John Wiley & Sons, Inc; Hoboken, New Jersey: 2011. [Google Scholar]
- 88.Nguyen KC, Rippstein P, Tayabali AF, Willmore WG. Toxicological sciences: an official journal of the Society of Toxicology. 2015;146:31–42. doi: 10.1093/toxsci/kfv068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Singh N, Jenkins GJ, Asadi R, Doak SH. Nano reviews. 2010;1 doi: 10.3402/nano.v1i0.5358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Pietrangelo A. N Engl J Med. 2004;350:2383–2397. doi: 10.1056/NEJMra031573. [DOI] [PubMed] [Google Scholar]
- 91.Ringo JM, Dowse HB, Barton KM. Behavior genetics. 1987;17:141–154. doi: 10.1007/BF01065993. [DOI] [PubMed] [Google Scholar]
- 92.Gaiter A, Nebiolo P, Torazza M, Gabella P, Bottacchi E, Alloatti S. Minerva urologica e nefrologica= The Italian journal of urology and nephrology. 1987;40:309–312. [PubMed] [Google Scholar]
- 93.Chaudhury C, Kim J, Mehnaz S, Wani MA, Oberyszyn TM, Bronson C, Mohanty S, Hayton WL, Robinson JM, Anderson CL. The Journal of nutrition. 2006;136:2993–2998. doi: 10.1093/jn/136.12.2993. [DOI] [PubMed] [Google Scholar]
- 94.Yildirimer L, Thanh NTK, Loizidou M, Seifalian AM. Nano Today. 2011;6:585–607. doi: 10.1016/j.nantod.2011.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Boulaiz H, Marchal JA, Prados J, Melguizo C, Aranega A. Cell Mol Biol. 2004;51:3–22. [PubMed] [Google Scholar]
- 96.Lv H, Zhang S, Wang B, Cui S, Yan J. J Controlled Release. 2006;114:100–109. doi: 10.1016/j.jconrel.2006.04.014. [DOI] [PubMed] [Google Scholar]
- 97.Kim YH, Park JH, Lee M, Kim Y-H, Park TG, Kim SW. J Controlled Release. 2005;103:209–219. doi: 10.1016/j.jconrel.2004.11.008. [DOI] [PubMed] [Google Scholar]
- 98.Kircheis R, Schüller S, Brunner S, Ogris M, Heider KH, Zauner W, Wagner E. J Gene Med. 1999;1:111–120. doi: 10.1002/(SICI)1521-2254(199903/04)1:2<111::AID-JGM22>3.0.CO;2-Y. [DOI] [PubMed] [Google Scholar]
- 99.Aillon KL, Xie Y, El-Gendy N, Berkland CJ, Forrest ML. Adv Drug Delivery Rev. 2009;61:457–466. doi: 10.1016/j.addr.2009.03.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Boisselier E, Astruc D. Chem Soc Rev. 2009;38:1759–1782. doi: 10.1039/b806051g. [DOI] [PubMed] [Google Scholar]
- 101.Alkilany AM, Murphy CJ. J Nanopart Res. 2010;12:2313–2333. doi: 10.1007/s11051-010-9911-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Takenaka S, Karg E, Roth C, Schulz H, Ziesenis A, Heinzmann U, Schramel P, Heyder J. Environmental Health Perspectives. 2001;109:547–551. doi: 10.1289/ehp.01109s4547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.El-Ansary A, Al-Daihan S. Journal of Toxicology. 2009;2009 doi: 10.1155/2009/754810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Valdiglesias V, Kilic G, Costa C, Fernandez-Bertolez N, Pasaro E, Teixeira JP, Laffon B. Environmental and molecular mutagenesis. 2015;56:125–148. doi: 10.1002/em.21909. [DOI] [PubMed] [Google Scholar]
- 105.Li L, Jiang L-L, Zeng Y, Liu G. Chinese Physics B. 2013;22:127503. [Google Scholar]
- 106.Kim JE, Kang SH, Moon Y, Chae JJ, Lee AY, Lee JH, Yu KN, Jeong DH, Choi M, Cho MH. Chem Res Toxicol. 2014;27:290–303. doi: 10.1021/tx400397g. [DOI] [PubMed] [Google Scholar]
- 107.Du J, Wang S, You H, Zhao X. Environ Toxicol Pharmacol. 2013;36:451–462. doi: 10.1016/j.etap.2013.05.007. [DOI] [PubMed] [Google Scholar]
- 108.Bao H, Pan Y, Ping Y, Sahoo NG, Wu T, Li L, Li J, Gan LH. Small. 2011;7:1569–1578. doi: 10.1002/smll.201100191. [DOI] [PubMed] [Google Scholar]
- 109.Yang K, Li Y, Tan X, Peng R, Liu Z. Small. 2013;9:1492–1503. doi: 10.1002/smll.201201417. [DOI] [PubMed] [Google Scholar]
- 110.Yang K, Wan J, Zhang S, Zhang Y, Lee ST, Liu Z. ACS Nano. 2011;5:516–522. doi: 10.1021/nn1024303. [DOI] [PubMed] [Google Scholar]
- 111.Zhang X, Yin J, Peng C, Hu W, Zhu Z, Li W, Fan C, Huang Q. Carbon. 2011;49:986–995. [Google Scholar]
- 112.Lee S, Yun HS, Kim SH. Biomaterials. 2011;32:9434–9443. doi: 10.1016/j.biomaterials.2011.08.042. [DOI] [PubMed] [Google Scholar]
- 113.Yang H, Liu C, Yang D, Zhang H, Xi Z. Journal of Applied Toxicology. 2009;29:69–78. doi: 10.1002/jat.1385. [DOI] [PubMed] [Google Scholar]
- 114.Duan J, Yu Y, Li Y, Yu Y, Sun Z. Biomaterials. 2013;34:5853–5862. doi: 10.1016/j.biomaterials.2013.04.032. [DOI] [PubMed] [Google Scholar]
- 115.Chen M, von Mikecz A. Experimental cell research. 2005;305:51–62. doi: 10.1016/j.yexcr.2004.12.021. [DOI] [PubMed] [Google Scholar]
- 116.Kabanov A, Gendelman H. Prog Polym Sci. 2007;32:1054–1082. doi: 10.1016/j.progpolymsci.2007.05.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Modi G, Pillay V, Choonara YE. Ann N Y Acad Sci. 2010;1184:154–172. doi: 10.1111/j.1749-6632.2009.05108.x. [DOI] [PubMed] [Google Scholar]
- 118.Todorich BM, Connor JR. Ann N Y Acad Sci. 2004;1012:171–178. doi: 10.1196/annals.1306.014. [DOI] [PubMed] [Google Scholar]
- 119.Ward RJ, Dexter DT, Crichton RR. J Trace Elem Med Biol. 2015;31:267–273. doi: 10.1016/j.jtemb.2014.12.012. [DOI] [PubMed] [Google Scholar]
- 120.Liu G, Men P, Perry G, Smith MA. Methods Mol Biol. 2010;610:123–144. doi: 10.1007/978-1-60327-029-8_8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Liu G, Men P, Kudo W, Perry G, Smith MA. Neurosci Lett. 2009;455:187–190. doi: 10.1016/j.neulet.2009.03.064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Kim P, Lee BK, Lee HY, Kawai T, Suh KY. Adv Mater. 2008;20:31. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.