Abstract
Late-onset hypogonadism is defined as a combination of low testosterone (T) levels and typical symptoms and signs. A major area of uncertainty is whether T concentrations are always really sufficient to fully reflect Leydig cell (dys)function. Mild testicular alteration could be diagnosed only by additional biochemical markers, such as luteinizing hormone (LH) and 25-hydroxyvitamin D levels. These markers help in identifying the so-called “subclinical” hypogonadism (normal T, high LH levels). Patients with hypogonadism have frequently low levels of 25-hydroxyvitamin D due to impairment of the hydroxylating enzyme CYP2R1 in the testis. However, no data have been published dealing with the best treatment option (cholecalciferol – the Vitamin D precursor, or calcidiol - 25-hydroxylated form of Vitamin D) in these patients. We studied 66 patients with classic hypogonadism (total T [TT] <12 nmol l−1, LH ≥ 8 IU l−1) (n = 26) and subclinical hypogonadism (TT ≥ 12 nmol l−1, LH ≥ 8 IU l−1) (n = 40) and low 25-hydroxyvitamin D (<50 nmol l−1). Subjects received cholecalciferol (5000 IU per week) (n = 20) or calcidiol (4000 IU per week) (n = 46), and 25-hydroxyvitamin D and parathyroid hormone (PTH) were evaluated after 3 months of therapy. Supplementation with calcidiol significantly increased 25-hydroxyvitamin D and significantly decreased PTH levels in both groups of men with hypogonadism (primary, n = 16 and subclinical, n = 30), whereas supplementation with cholecalciferol did not modify their levels. This study shows for the first time that the administration of the 25-hydroxylated form of Vitamin D (calcidiol), and not the administration of the precursor cholecalciferol, restores 25-hydroxyvitamin D levels in subjects with hypogonadism.
Keywords: insulin-like factor 3, late-onset hypogonadism, male hypogonadism, testosterone, Vitamin D
INTRODUCTION
Differently form other endocrine glands, in which the classification and diagnosis of hypofunction are based on well-defined biochemical criteria, late-onset hypogonadism (LOH) is defined by as a combination of low testosterone (T) levels and typical symptoms and signs.1 Based on this definition, LOH is a highly prevalent condition, with a frequency of approximately 6% in the population of middle-aged to older men, and the prevalence increases with age and some frequent medical conditions.1
A major area of uncertainty is whether total T (TT) concentrations are always really sufficient to fully reflect (dys)function of the Leydig cells,2 the cells that produce T. In most cases reduced T levels are sufficient to diagnose hypogonadism. However, mild testicular dysfunction could be diagnosed only by additional biochemical markers. Although it is well known that T and luteinizing hormone (LH) levels allow distinguishing primary (low T, low LH) and secondary (low T, high LH) hypogonadism,3 guidelines did not exhaustively discussed the relevance of the so-called “compensated” or “subclinical” hypogonadism (normal T, high LH),1 also known as “Leydig cell impairment.”4,5 This condition indeed represents a frequent finding in men above age 40 (10% in the European Male Ageing Study)3 and merits more attention. Actually, it indicates a dysfunction in the hypothalamus-pituitary-testicular axis, it might already be associated with clinical manifestations, it might be a precursor of overt hypogonadism, and it predicts cardiovascular disease (CVD) events4 and all-causes and CVD mortality similarly to overt hypogonadism.5 Therefore, elevated LH with normal or low-normal T levels cannot be ignored, merits follow-up of patients to monitor progression and probably should be treated before T levels further decline. For these reasons, this condition is neither “subclinical” nor “compensated,” but probably could be defined as “incipient” hypogonadism.
In the context of better characterization of hypogonadism, at least two other functions of the Leydig cells are important: they produce insulin-like factor 3 (INSL3) and they express CYP2R1 gene, which encodes the major enzyme involved in 25-hydroxylation of Vitamin D.2 Impaired INSL3 production and CYP2R1 expression (which leads to low levels of 25-hydroxyvitamin D) is found not only in cases of overt hypogonadism but also in cases of “incipient” hypogonadism.2 Although for many reasons INSL3 is considered an ideal biomarker of Leydig cell function,2 at present the role of its measurement in the clinical setting is still questionable. On the contrary, population studies showed a clear association between the levels of T and 25-hydroxyvitamin D and highlighted that men with primary and secondary hypogonadism, as well as those with “incipient” hypogonadism, are frequently deficient in Vitamin D.2
Although Vitamin D insufficiency is frequently found in hypogonadal men, no data have been published dealing with the best treatment option in these patients. In general, Vitamin D supplementation is usually done either with cholecalciferol (the Vitamin D precursor) or with calcidiol (the 25-hydroxylated form of Vitamin D),6 but no study comparing the effects of these two drugs in LOH have been published. This is particularly intriguing taken into account that the testis participate in 25-hydroxylation of cholecalciferol and this function is reduced in hypogonadal men.7
MATERIALS AND METHODS
The study has been approved by the Ethics Committee of the University-Hospital of Padova and each participant gave their written informed consent. The study has been conducted in accordance with the principles expressed in the Declaration of Helsinki.
From January to September 2013, among otherwise healthy subjects referred for infertility evaluation we selected 66 patients (mean age ± s.d., 34.5 ± 6.8 years) with classic hypogonadism (TT < 12 nmol l−1, LH ≥ 8 IU l−1) (n = 26) or subclinical hypogonadism (TT ≥ 12 nmol l−1, LH ≥ 8 IU l−1) (n = 40) and low 25-hydroxyvitamin D (<50 nmol l−1). Subjects were randomized by random number generator to receive cholecalciferol (5000 IU per week) (n = 20) or calcidiol (4000 IU per week) (n = 46) supplementation, and 25-hydroxyvitamin D and parathyroid hormone (PTH) were evaluated after 3 months of therapy.
Data are reported as mean ± s.d. of the mean. The statistical significance of the difference before and after therapy was analyzed with paired Student's t-test. Statistical analyses were performed with SAS version 9.2 (SAS Institute, Cary, NC, USA) for Windows. We performed 18 tests (2 outcomes × 9 groups), which translates into a statistically significant P value after Bonferroni correction of 0.05/18 ≈ 0.003.
RESULTS
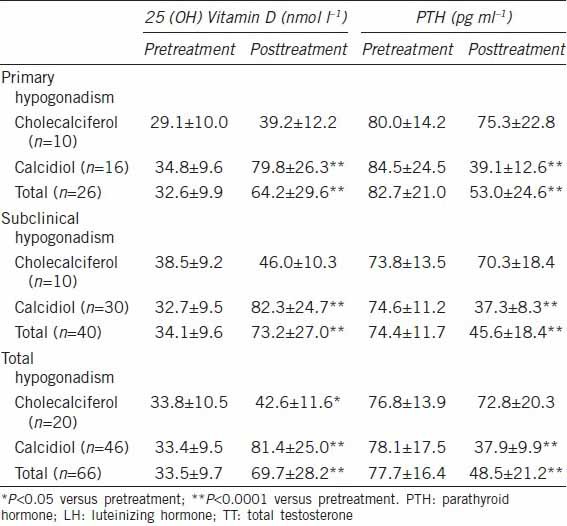
Table 1 shows the data for 25-hydroxyvitamin D and PTH levels before and after Vitamin D supplementation (cholecalciferol, n = 20 or calcidiol, n = 46). Supplementation with calcidiol significantly increased 25-hydroxyvitamin D and significantly decreased PTH levels in both groups of men with hypogonadism (primary, n = 16 and subclinical, n = 30), whereas supplementation with cholecalciferol did not modify their levels. No effect on T levels was observed after Vitamin D supplementation (data not shown).
Table 1.
Serum levels of 25 (OH) Vitamin D and PTH in 66 infertile patients with primary hypogonadism (LH≥8 IU l−1, TT<12 nmol l−1) or subclinical hypogonadism (LH≥8 IU l−1, TT≥12 nmol l−1) before and after a 3 months period of supplementation with cholecalciferol (5000 IU per week) or calcidiol (4000 IU per week)
DISCUSSION
Patients with hypogonadism and testicular impairment have frequently low levels of 25-hydroxyvitamin D due to impairment of the hydroxylating enzyme CYP2R1 in the testis.2,7,8 In fact, this enzyme participates in the activation of the Vitamin D precursor cholecalciferol and it is expressed by the Leydig cells of the testis under the influence of hCG/LH.2,7,8 Low expression of CYP2R1 in patients with Leydig cell dysfunction leads to low serum levels of 25-hydroxyvitamin D and low bone mineral density (BMD).2,7,8 This study shows for the first time that the administration of the 25-hydroxylated form of Vitamin D (calcidiol), and not the administration of the precursor cholecalciferol, restores 25-hydroxyvitamin D levels in subjects with Leydig cell dysfunction (elevated LH). Furthermore, it reduces PTH levels, one of the best marker in assessing the effectiveness of supplementation. Therefore, calcidiol supplementation overcomes the decline in testis function and represents the ideal treatment in these cases, likewise in cases with decline in hepatic function.6
Current guidelines9 suggest that all adults who are Vitamin D deficient (25-hydroxyvitamin D levels below 50 nmol l−1) be treated with Vitamin D to achieve a blood level above 75 nmol l−1. Following these guidelines, in our cohort of Vitamin D deficient men, we achieved sufficient blood levels of 25-hydroxyvitamin D with calcidiol, but not with cholecalciferol treatment, further confirming that this latter therapeutic option is not optimal in these cases. Furthermore, reduced PTH levels after Vitamin D treatment, other than be used as surrogate marker of Vitamin D status,10 might have clinical implications. Vitamin D deficiency causes a decrease in intestinal calcium absorption and therefore an increase in PTH levels. This secondary hyperparathyroidism maintains serum calcium in the normal range at the expense of mobilizing calcium from the skeleton through increase in osteoclastic activity that creates a decrease in BMD, resulting in osteopenia and osteoporosis. Therefore, correction of the secondary hyperparathyroidism by calcidiol treatment is another benefit for these patients.
This study further supports the notion that male hypogonadism cannot be longer diagnosed only on the basis of T levels, and at least LH and 25-hydroxyvitamin D levels should be considered. In fact, patients with mild Leydig cell dysfunction would be missed if only T levels are assessed, because LH and 25-hydroxyvitamin D levels are more sensitive markers of Leydig cell impairment than T. Importantly, men with low 25-hydroxyvitamin D levels are at risk of clinical sequelae (such as low BMD and osteoporosis) even if they have normal T levels.2,7,8
We would like to highlight some more areas of uncertainty in the field of LOH. First, in the aging male the relationship between T levels and clinical manifestations is complex and vague, and even the symptoms suggested representing the most specific manifestations of low T (poor morning erection, low sexual desire, erectile dysfunction)11 might have different etiologies and cannot clearly define a man as hypogonadal. Low T levels are indeed associated with different co-morbidities (such as obesity, metabolic syndrome, Type 2 diabetes mellitus, hyperlipidemia, osteoporosis), predict the development of important health problems (such as metabolic syndrome, diabetes, ischemic heart disease),4 and increase mortality from CVDs.5,12 Therefore, limiting the clinical practice of T levels determination only in men with sexual symptoms might be misleading. It is also interesting to note that a combined deficiency of T and Vitamin D is more associated with mortality that deficiency of one of these hormone alone.13
Second, not only reference and cut-off values of T for defining hypotestosteronemia at different ages are still not available, but also additional features of T levels during time in single individual and additional markers of testicular function2 should be taken into consideration. Given the high inter-individual variations in T levels during aging and the vague association between them and symptoms, it appears that, in the clinical setting of personalized medicine, the most important determinants in the development of symptoms and manifestation of clinical signs seems to be the duration of hypotestosteronemia, the magnitude of the decrease in T concentrations and consequently, the T levels from which a man starts to loose Leydig cell function. In fact, longitudinal studies showed that men who develop symptoms had a higher loss of T than men who had unchanged or fewer symptoms 5 years before, and therefore the magnitude of the decrease in concentrations is a better predictor of LOH than are the actual concentrations of T.14
Furthermore, the health status affects T level. Apparent good health adds 12.6% to the level of T.15 More importantly, an aging man who incurs a significant change in health status would lose T at a greater rate than if he had maintained his good health. In fact, the entity and the degree in T decline are strongly dependent on the presence or development of co-morbidities. Two large longitudinal studies15,16 clearly showed that incident poor health accelerates the age-related decline in androgen levels11 and that maintenance of a low CVD risk mitigates the age-related decline of T.16 Furthermore, not only low T is associated with different metabolic alterations (as impaired fasting glucose, insulin resistance, Type 2 diabetes mellitus, metabolic syndrome and its individual components), but also prospective studies demonstrate that hypogonadism predicts the development of Type 2 diabetes mellitus, high blood pressure, and metabolic syndrome, and conversely Type 2 diabetes mellitus and metabolic syndrome could predict the occurrence of low T. Finally, recent longitudinal data17 showed that weight changes are major determinants in modifying T levels. These data well highlight that the decline in T levels is not a definite process during aging, but is largely dependent on lifestyle factors and changes in health status. Nevertheless, aging in itself represents a risk factor for T reduction. Longitudinal studies agree that, in apparently healthy subjects, T reduction after age 40, although significantly attenuated with respect to those men co-morbidities, is 1%–2% per year.15
Based on these observations, it seems that making a diagnosis of LOH only when sexual symptoms are present11,18 might be misleading and inappropriate. Other signs might be suspicious of low T, and in particular, patients with clinical manifestations associated with insulin resistance (obesity, Type 2 diabetes mellitus, metabolic syndrome) and CVD risk factors should be screened for hypogonadism. Finding low T levels in these conditions helps in better management of these patients who will benefit from T substitution therapy, and finding normal T levels helps in having stricter follow-up of patients. On the other hand, finding low T levels in apparently healthy subjects might prevent development of such important clinical conditions by adequate treatment, whereas normal T levels in these subjects might represent a personalized reference starting point useful if the subject incurs a significant change in health status and/or develops symptoms.
CONCLUSION
We think that there is a general need for new research directions for a personalized medicine of LOH. Our study shows that overt and “subclinical” hypogonadism characterized by low levels of 25-hydroxyvitamin D should be supplemented preferably with calcidiol rather than cholecalciferol, but additional research is needed to better define in this context: (i) the clinical manifestation of hypogonadism and the screening procedures in conditions other than sexual symptoms; (ii) the impact of changes in health status in exacerbating the entity and rapidity of T decline; (iii) the impact of and therapeutic options for isolated LH elevation; (iv) the role of additional Leydig cell markers; (v) the biomarkers of T replacement therapy. In general, we hope for a more personalized medicine in this field owing to tailored diagnosis and therapy.
AUTHOR CONTRIBUTIONS
CF conceived the study, coordinated it and drafted the manuscript. AEC, FL and AL participated in study design and performed clinical analyses. AF participated in study design, performed clinical and statistical analyses, and wrote the final manuscript. All authors read and approved the final manuscript.
COMPETING INTERESTS
All authors declare no competing interests.
REFERENCES
- 1.Bhasin S, Cunningham GR, Hayes FJ, Matsumoto AM, Snyder PJ, et al. Testosterone therapy in men with androgen deficiency syndromes: an endocrine society clinical practice guideline. J Clin Endocrinol Metab. 2010;95:2536–59. doi: 10.1210/jc.2009-2354. [DOI] [PubMed] [Google Scholar]
- 2.Ferlin A, Selice R, Carraro U, Foresta C. Testicular function and bone metabolism – beyond testosterone. Nat Rev Endocrinol. 2013;9:548–54. doi: 10.1038/nrendo.2013.135. [DOI] [PubMed] [Google Scholar]
- 3.Tajar A, Forti G, O’Neill TW, Lee DM, Silman AJ, et al. Characteristics of secondary, primary, and compensated hypogonadism in aging men: evidence from the European Male Ageing Study. J Clin Endocrinol Metab. 2010;95:1810–8. doi: 10.1210/jc.2009-1796. [DOI] [PubMed] [Google Scholar]
- 4.Hyde Z, Norman PE, Flicker L, Hankey GJ, McCaul KA, et al. Elevated LH predicts ischaemic heart disease events in older men: the health in men study. Eur J Endocrinol. 2011;164:569–77. doi: 10.1530/EJE-10-1063. [DOI] [PubMed] [Google Scholar]
- 5.Hyde Z, Norman PE, Flicker L, Hankey GJ, Almeida OP, et al. Low free testosterone predicts mortality from cardiovascular disease but not other causes: the health in men study. J Clin Endocrinol Metab. 2012;97:179–89. doi: 10.1210/jc.2011-1617. [DOI] [PubMed] [Google Scholar]
- 6.Brandi ML, Minisola S. Calcidiol [25(OH)D3]: from diagnostic marker to therapeutical agent. Curr Med Res Opin. 2013;29:1565–72. doi: 10.1185/03007995.2013.838549. [DOI] [PubMed] [Google Scholar]
- 7.Foresta C, Strapazzon G, De Toni L, Perilli L, Di Mambro A, et al. Bone mineral density and testicular failure: evidence for a role of vitamin D 25-hydroxylase in human testis. J Clin Endocrinol Metab. 2011;96:E646–52. doi: 10.1210/jc.2010-1628. [DOI] [PubMed] [Google Scholar]
- 8.Foresta C, Selice R, De Toni L, Di Mambro A, Carraro U, et al. Altered bone status in unilateral testicular cancer survivors: role of CYP2R1 and its luteinizing hormone-dependency. J Endocrinol Invest. 2013;36:379–84. doi: 10.3275/8650. [DOI] [PubMed] [Google Scholar]
- 9.Holick MF, Binkley NC, Bischoff-Ferrari HA, Gordon CM, Hanley DA, et al. Evaluation, treatment, and prevention of vitamin D deficiency: an Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab. 2011;96:1911–30. doi: 10.1210/jc.2011-0385. [DOI] [PubMed] [Google Scholar]
- 10.Cianferotti L, Marcocci C. Subclinical vitamin D deficiency. Best Pract Res Clin Endocrinol Metab. 2012;26:523–37. doi: 10.1016/j.beem.2011.12.007. [DOI] [PubMed] [Google Scholar]
- 11.Wu FC, Tajar A, Beynon JM, Pye SR, Silman AJ, et al. Identification of late-onset hypogonadism in middle-aged and elderly men. N Engl J Med. 2010;363:123–35. doi: 10.1056/NEJMoa0911101. [DOI] [PubMed] [Google Scholar]
- 12.Pye SR, Huhtaniemi IT, Finn JD, Lee DM, O’Neill TW, et al. Late-onset hypogonadism and mortality in aging men. J Clin Endocrinol Metab. 2014;99:1357–66. doi: 10.1210/jc.2013-2052. [DOI] [PubMed] [Google Scholar]
- 13.Lerchbaum E, Pilz S, Boehm BO, Grammer TB, Obermayer-Pietsch B, et al. Combination of low free testosterone and low vitamin D predicts mortality in older men referred for coronary angiography. Clin Endocrinol (Oxf) 2012;77:475–83. doi: 10.1111/j.1365-2265.2012.04371.x. [DOI] [PubMed] [Google Scholar]
- 14.Holm AC, Fredrikson MG, Theodorsson E, Palmefors LG, Karlsson PS, et al. Change in testosterone concentrations over time is a better predictor than the actual concentrations for symptoms of late onset hypogonadism. Aging Male. 2011;14:249–56. doi: 10.3109/13685538.2011.582527. [DOI] [PubMed] [Google Scholar]
- 15.Feldman HA, Longcope C, Derby CA, Johannes CB, Araujo AB, et al. Age trends in the level of serum testosterone and other hormones in middle-aged men: longitudinal results from the Massachusetts male aging study. J Clin Endocrinol Metab. 2002;87:589–98. doi: 10.1210/jcem.87.2.8201. [DOI] [PubMed] [Google Scholar]
- 16.Haring R, Xanthakis V, Coviello A, Sullivan L, Bhasin S, et al. Clinical correlates of sex steroids and gonadotropins in men over the late adulthood: the Framingham Heart Study. Int J Androl. 2012;35:775–82. doi: 10.1111/j.1365-2605.2012.01285.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Camacho EM, Huhtaniemi IT, O’Neill TW, Finn JD, Pye SR, et al. Age-associated changes in hypothalamic-pituitary-testicular function in middle-aged and older men are modified by weight change and lifestyle factors: longitudinal results from the European Male Ageing Study. Eur J Endocrinol. 2013;168:445–55. doi: 10.1530/EJE-12-0890. [DOI] [PubMed] [Google Scholar]
- 18.Huhtaniemi I. Late-onset hypogonadism: current concepts and controversies of pathogenesis, diagnosis and treatment. Asian J Androl. 2014;16:192–202. doi: 10.4103/1008-682X.122336. [DOI] [PMC free article] [PubMed] [Google Scholar]


