Abstract
Objective
Barrett’s metaplasia might develop if GORD causes oesophageal squamous cells to convert into columnar cells. Acid and bile exposures upregulate columnar differentiation genes like CDX2 in oesophageal squamous cells, but it is not known if such exposures downregulate squamous differentiation genes like SOX2. In addition to acid and bile, GORD patients also have high oesophageal concentrations of nitric oxide (NO). This study aims to determine how acid, bile salts and NO affect genes that influence oesophageal cell phenotype.
Design
Oesophageal squamous cells from Barrett’s patients were exposed to acidic bile salts or NOC-9 (an NO donor). SOX2, p63 (squamous transcription factor), and CDX2 mRNAs were measured by quantitative RT-PCR. SOX2 and its regulatory Akt pathway proteins were evaluated by Western blotting. S-nitrosylation by NO was blocked by dithiothreitol. Immunohistochemistry for SOX2 was performed on the oesophagus of rats with surgically-induced GORD fed diets with and without nitrite supplementation.
Results
In oesophageal squamous cells, NO profoundly decreased SOX2 protein, and caused a significantly greater decrease in SOX2 mRNA than acidic bile salts. NO also decreased p63 and increased CDX2 expression. NO caused S-nitrosylation of Akt, blocking its phosphorylation. Akt pathway inhibition by LY294002 or Akt siRNA reduced SOX2 mRNA. Rats fed nitrite-supplemented diets exhibited weaker SOX2 oesophageal staining than rats fed normal diets.
Conclusion
In oesophageal squamous cells, NO blocks SOX2 expression through Akt S-nitrosylation. NO also increases CDX2 and decreases p63 expression. By triggering molecular events preventing squamous differentiation while promoting intestinal differentiation, NO might contribute to Barrett’s pathogenesis.
Keywords: p63, CDX2, metaplasia, Barrett’s esophagus, animal model
Introduction
Barrett’s oesophagus is the condition in which a metaplastic columnar epithelium predisposed to develop adenocarcinoma replaces oesophageal squamous epithelium that has been damaged by gastro-oesophageal reflux disease (GORD).1 Barrett’s oesophagus affects 2% to 7% of adults in Western countries, and is a major risk factor for oesophageal adenocarcinoma, a tumor whose rate of increase over the past 4 decades is among the highest reported for any cancer. 2–6
The cells of origin for Barrett’s oesophagus are not known. One hypothesis holds that Barrett’s metaplasia develops from columnar progenitor cells in the gastric cardia that migrate into the oesophagus to replace GORD-damaged squamous cells. 7, 8 Alternative hypotheses propose that Barrett’s metaplasia develops either as a result of transdifferentiation, in which mature oesophageal squamous cells convert into columnar cells, or transcommitment, in which immature progenitor cells in the oesophagus are reprogrammed to undergo aberrant columnar differentiation rather than normal squamous differentiation. 9, 10 For development of columnar metaplasia, both transdifferentiation and transcommitment involve alterations in cells that are native to the oesophagus, whereas the migration hypothesis involves cells that originate in the stomach.
Much indirect evidence suggests that transdifferentiation or transcommitment of oesophageal cells contributes to the pathogenesis of Barrett’s metaplasia. Columnar epithelium lines the early embryonic oesophagus and, in a mouse model, explants of embryonic oesophagus lose columnar markers and begin to express squamous markers after 5 days in culture, with some cells expressing both columnar and squamous markers simultaneously. 11 These data suggest that, in normal embryonic development, oesophageal squamous epithelium arises from columnar epithelium through a direct conversion process. By inference, a GORD-induced reversal of this process in adults could result in Barrett’s oesophagus.
In patients with Barrett’s oesophagus, biopsies of the junction between oesophageal squamous and metaplastic columnar epithelium can show cells with both squamous and columnar features by scanning electron microscopy 12, 13, and light microscopy can show a peculiar multilayered epithelium with a basal layer of squamous cells covered by a superficial layer of columnar cells. It has been proposed that these changes represent transitional stages in the squamous-to-columnar metaplasia of Barrett’s oesophagus. 14
Further evidence for transdifferentiation or transcommitment in the pathogenesis of Barrett’s metaplasia emerges from studies showing that oesophageal squamous cells and tissues exposed to gastro-oesophageal reflux in vivo or to acid and bile salts in vitro increase their expression of transcription factors involved in development of a columnar cell phenotype such as CDX2 and the Sonic Hedgehog pathway target genes, SOX9 and FOXA2. 15–20 In a rat model of surgically-induced reflux oesophagitis, oesophageal squamous cells in the basal layer express Cdx genes prior to the development of intestinal metaplasia. 20 Oesophageal squamous cell lines from patients with Barrett’s oesophagus increase their expression of CDX2 when they are exposed to acid and bile salts19, and CDX2 expression has been found in oesophageal squamous epithelium from patients with Barrett’s oesophagus. 19, 21
The studies discussed above have focused on how GORD might contribute to development of Barrett’s metaplasia by upregulating genes determining a columnar cell phenotype. However, it also seems likely that the pathogenesis of Barrett’s metaplasia involves downregulation of genes involved in squamous differentiation such as SOX2, a transcription factor that promotes development of stratified squamous epithelia. 22 Mice genetically engineered to express low levels of oesophageal Sox2 develop patchy columnar lining in the oesophagus, with areas resembling human multilayered epithelium. 14, 22 Little is known of how GORD influences the expression of squamous differentiation genes like SOX2.
Finally, studies on how GORD might underlie Barrett’s pathogenesis have focused largely on effects of acid and bile salts (the major noxious components of gastric juice) on oesophageal cells. In patients with GORD, however, high oesophageal concentrations of nitric oxide (NO) can be generated from dietary nitrate in green leafy vegetables and other foods. 23 This NO can react with oxygen to generate toxic, reactive nitrogen species (RNS).24 Relatively few studies have focused specifically on the role of NO in the pathogenesis of Barrett’s metaplasia. Now, we have explored mechanisms whereby acid, bile salts and NO might influence the expression of SOX2 in non-neoplastic oesophageal squamous cell lines, and have correlated our in vitro findings with in vivo data from a rat model of reflux oesophagitis. We have found that NO triggers molecular events in oesophageal squamous cells that can inhibit squamous differentiation while promoting intestinal differentiation, a combination that might lead to development of Barrett’s oesophagus.
Materials and Methods
Cell Lines
We used two non-neoplastic, telomerase-immortalised oesophageal squamous cell lines (NES-B3T, NES-B10T, created from endoscopic biopsy specimens of the distal, squamous-lined oesophagus of two patients with Barrett’s oesophagus) developed in our laboratory. 19, 25 All cell lines were maintained at 37°C in a 5% CO2 incubator.
Acidic Bile Salt and Nitric Oxide Exposure
For individual experiments, NES cells were cultured in neutral medium (pH 7.2) alone; neutral medium with periodic exposures to 500 µM of 6-(2-Hydroxy-1-methyl-2-nitrosohydrazino)-N-methyl-1-hexanamine (NOC-9; Sigma, St Louis, MO), which is a small molecule NO donor; or neutral medium with periodic exposures to acidic medium (pH 5.5) containing a mixture of conjugated bile acids as previously described. 26 NO measurements were performed using the ISO-NO Mark II nitric oxide meter and ISO-NO sensor (WPI Inc., FL, USA) as previously described. 27
Semiquantitative and Quantitative Real-Time Polymerase Chain Reaction (qRT-PCR)
Semiquantitative and qRT-PCR were performed as previously described. 28 The primer sequences (Table 1) were designed using Primer Express (Applied BioSystems, Foster City, CA) and manufactured by Integrated DNA Technologies (Coralville, IA).
Table 1.
Oligonucleotide primers
| Primer | Sequence (5’ to 3’) | Location | Use |
|---|---|---|---|
| SOX2-5’ SOX2-3’ |
CAG TTA CGC GCA CAT GAA CG CCC TGC TGC GAG TAG GAC AT |
sense antisense |
qPCR |
| CDX2-5’ CDX2-3’ |
CTC GGC AGC CAA GTG AAA AC GAT GGT GAT GTA GCG ACT GTA GTG A |
sense antisense |
qPCR |
| TAp63-5’ TAp63-3’ |
TGT ATC CGC ATG CAG GAC T CTG TGT TAT AGG GAC TGG TGGA C |
sense antisense |
qPCR |
| ΔNp63-5’ ΔNp63-3’ |
GAA AAC AAT GCC CAG ACT CAA TGC GCG TGG TCT GTG TTA |
sense antisense |
qPCR |
| β-actin-5’ β-actin-3’ |
CAT CCA CGA AAC TAC CTT CAA CTC C GAG CCG CCG ATC CAC ACG |
sense antisense |
qPCR |
| Akt-5’ Akt-3’ |
GTC TGA ACC TCT CTT TGT CTC C GCC AAC CCT CCT TCA CAA TA |
sense antisense |
RT-PCR |
| GAPDH-5’ GAPDH-3’ |
TCC CAC CTT TCT CAT CCA AG GTC TGC AAA AGG AGT GAG GC |
sense antisense |
RT-PCR |
Protein Extraction and Immunoblotting
Protein extracts were subjected to immunoblot analyses using primary antibodies (Supplemental Table 1) overnight at 4°C. As a loading control, membranes were stripped and re-probed with mouse anti-β-tubulin (Sigma, St. Louis, MO).
Inhibition of Akt Signaling
Inhibition of Akt signaling was performed using the highly selective pharmacologic inhibitor LY294002 (Cell Signaling, Danvers, MA) 29 or a specific Akt siRNA (Thermo Scientific, Waltham, MA) per manufacturer’s instructions.
Mechanistic target of rapamycin (mTOR)-Rictor Kinase Assay
To determine whether NOC-9 might reduce Akt phosphorylation by decreasing rictor-mTOR activity, we used an in vitro kinase assay. 30 Following immunoprecipitation of rictor, exogenous inactive Akt/PKB1 protein (that could be phosphorylated at Ser473 by the immunoprecipitated rictor) was added and Western blots performed for phospho-Akt Ser473 as an indicator of rictor-mTOR activity.
S-nitrosylation of Akt
During S-nitrosylation, NO converts protein cysteine thiols into S-nitrosothiols, which can be detected using a biotin-switch assay. S-nitrosylation can also be blocked by the reducing agent, dithiothreitol (DTT; Invitrogen, Carlsbad, CA). 31
Immunohistochemistry in a rat model of reflux oesophagitis
For immunohistochemical studies, we used previously collected oesophageal squamous tissue sections from rats with surgically-induced reflux oesophagitis that were fed diets with and without NO supplementation as described. 32 Tissue sections were obtained 4 weeks after the operation from animals with (n=6) or without (n=8) dietary NO supplementation.32 At this time point, the 6 animals given dietary NO supplementation developed intestinal metaplasia, while the 8 animals fed diets without NO supplementation did not. 32 Tissues were incubated with 1:100 dilution of SOX2 (Abcam, Cambridge, MA) and target protein was visualized using diaminobenzidine as substrate. Tissues were evaluated and scored by two gastrointestinal pathologists (AA, RDO) blinded to the treatment groups.
Data Analyses
Quantitative data are expressed as mean ± standard error of the mean (SEM). Statistical analyses were performed using an unpaired Student’s t-test or, for multiple comparisons, an ANOVA and the Student-Newman-Keuls multiple-comparisons test was used. P values ≤ 0.05 were considered significant for all analyses.
Additional details can be found in the Supplemental Material and Methods section.
Results
NOC-9 treatment profoundly suppresses SOX2 mRNA and protein expression in oesophageal squamous cells from patients with Barrett’s oesophagus
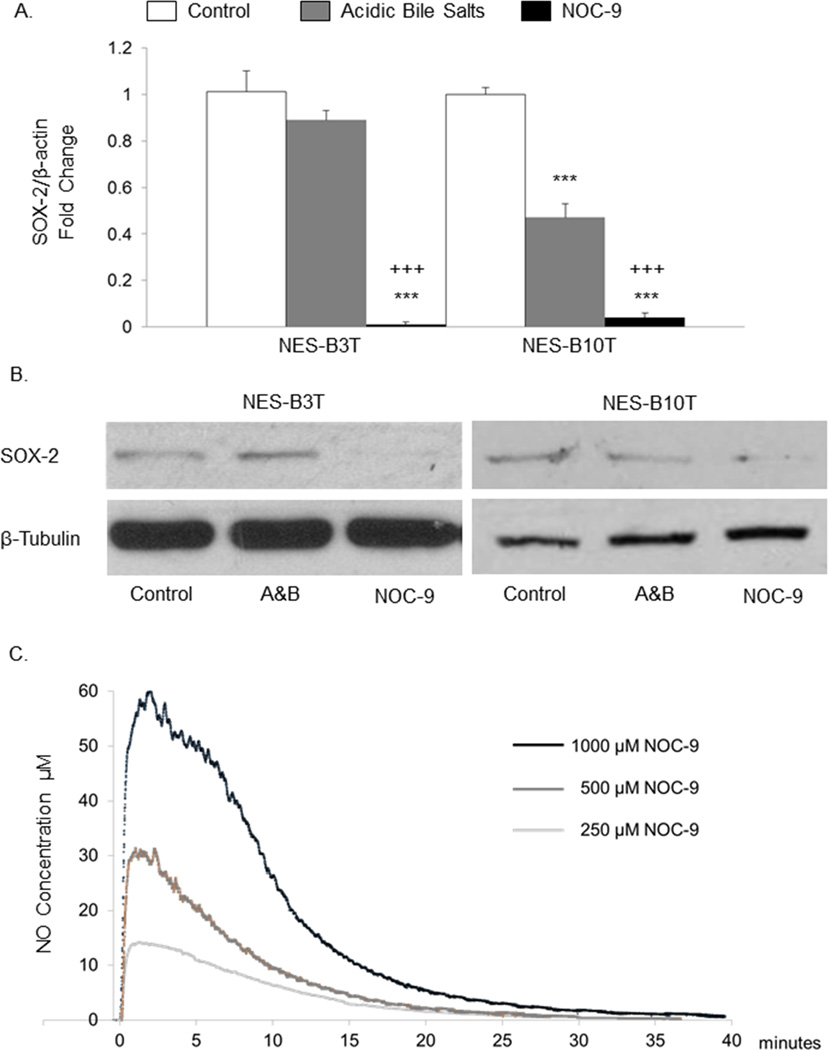
Exposure to acidic bile salts significantly reduced SOX2 mRNA expression in NES-B10T cells, but not NES-B3T cells (Figure 1A). In contrast, exposure to NOC-9 decreased SOX2 mRNA expression in both cell lines to levels significantly lower than in untreated control cells and in cells treated with acidic bile salts (Figure 1A). NOC-9 also decreased SOX2 protein levels in both oesophageal squamous cell lines (Figure 1B). After reviewing these data, we selected to use NOC-9 (rather than acidic bile salts) for all further experiments, given NOC-9’s consistent and profound suppression of SOX2 mRNA and protein.
Figure 1.
Treatment with NOC-9 suppresses SOX2 mRNA and protein expression in oesophageal squamous cell lines (NES-B3T and NES-B10T) from patients with Barrett’s oesophagus. Figures show representative experiments of (A) qRT-PCR for SOX2 mRNA and (B) Western blotting for SOX2 protein in NES cell lines following 5 days of treatment for 1 hour, 3 times daily with either with acidic bile salts (A&B) or 500 µM NOC-9; mRNA or protein was collected on the following day (day 6). Bar graphs represent the mean ±SEM. ***, p ≤ 0.001 compared to non-treated control; +++, p ≤ 0.001 compared to acidic bile salt-treated cells. (C) Continuous measurements of NO concentrations generated from media containing NOC-9 in doses of 250, 500 and 1,000 µM.
We measured NO concentrations generated from media containing NOC-9, and found that NOC-9 doses of 250, 500, and 1000 µM generated 15, 30, and 60 µM peak concentrations of NO, respectively (Figure 1C). Such concentrations have been detected in the oesophageal lumen of patients with GORD and Barrett’s oesophagus following nitrate ingestion. 33 Concentrations of NO peaked within 2 minutes after NOC-9 exposure (Figure 1C).
NOC-9-mediated decrease in SOX2 is associated with decreased expression of mRNAs for TA and ∆N isoforms of p63, and increased expression of CDX2 mRNA
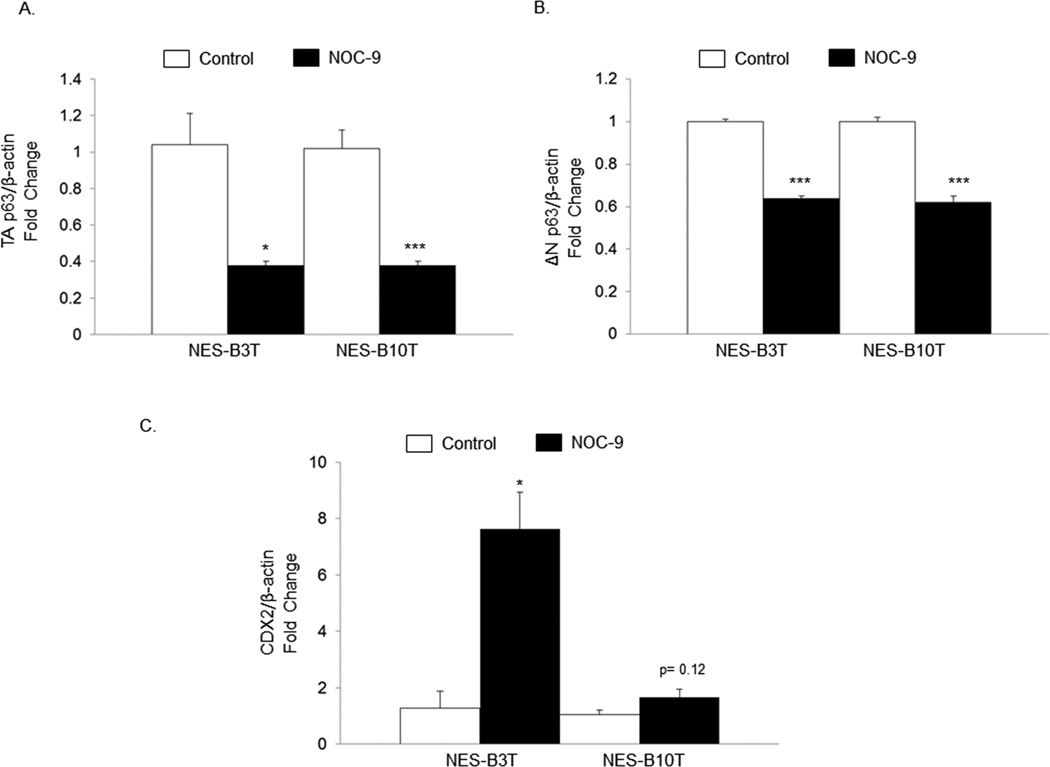
p63 is a transcription factor required for development and maintenance of oesophageal stratified squamous epithelium. 22, 34 The p63 gene encodes two major protein isoforms, TAp63 and ΔNp63, and p63 gene expression normally is linked with that of SOX2 in the oesophagus. To determine whether NOC-9 decreases p63 as well as SOX2, we determined mRNA expression for p63 isoforms using qRT-PCR. Treatment with NOC-9 significantly decreased both TAp63 and ∆Np63 mRNAs in NES-B3T and NES-B10T cells (Figure 2A and B). CDX2, a transcription factor that determines intestinal development, appears to have an inverse relationship with SOX2. 35, 36 Accordingly, NOC-9 treatment that caused a decrease in SOX2 caused an increase in CDX2 mRNA expression in both NES-B3T and NES-B10T cells, but the increase reached statistical significance only in NES-B3T cells (Figure 2C).
Figure 2.
NOC-9 suppresses mRNA expression of TA and ΔN isoforms of p63, and increases mRNA expression of CDX2 in NES cells. Figures show representative experiments for qRT-PCR of (A) TAp63, (B) ΔNp63, and (C) CDX2 mRNA expression in NES cell lines following 5 days of treatment with 500 µM NOC-9 for 1 hour, 3 times daily; mRNAs were collected on the following day (day 6). Bar graphs represent the mean ±SEM. *, p ≤ 0.05 compared to non-treated control; ***, p ≤ 0.001 compared to non-treated control.
In a rat model of surgically-induced reflux esophagitis, an NO-supplemented diet decreases SOX2 expression in the distal squamous oesophagus
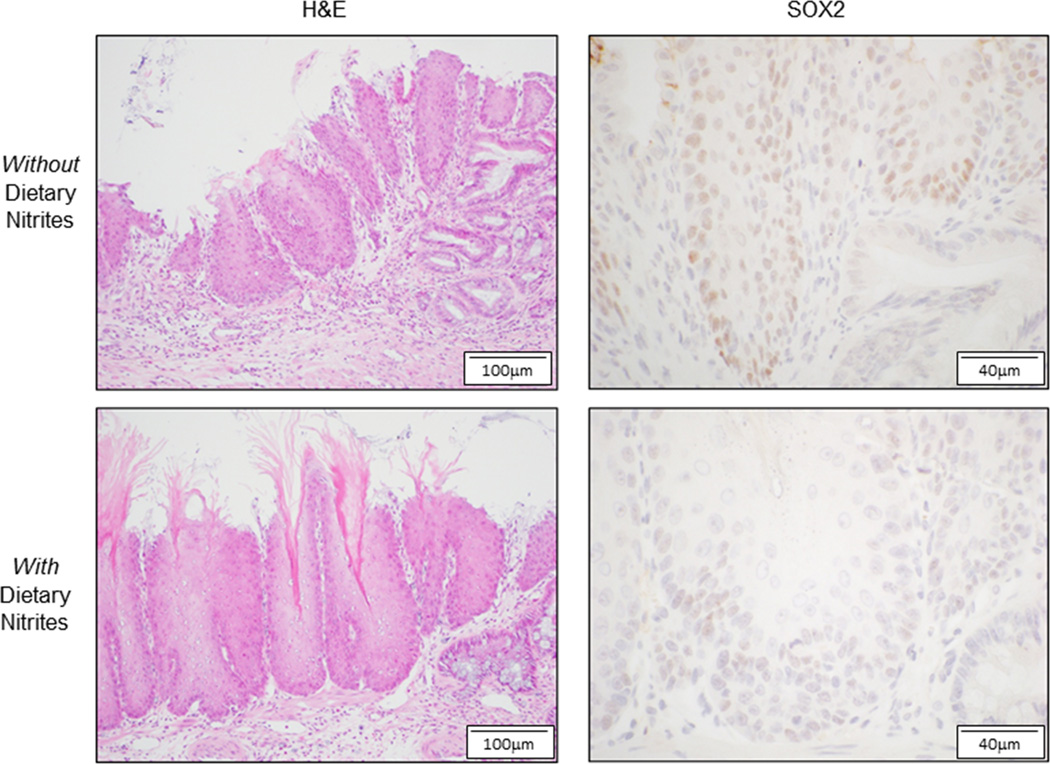
In a rat model, esophagojejunostomy and gastrojejunostomy were performed to induce gastro-duodenal-oesophageal reflux, and the rats develop reflux oesophagitis and columnar metaplasia similar to Barrett’s oesophagus. 32 The development of metaplasia was hastened by an NO-supplemented diet. To determine NO effects on oesophageal Sox2 expression in this model, we performed immunohistochemistry for Sox2 on the distal squamous-lined oesophagus of rats fed postoperative diets with and without NO supplementation.32 We found Sox2 staining predominantly in nuclei of the basal and suprabasal cells of squamous epithelium in all animals (Figure 3). However, the intensity of Sox2 staining was weaker in rats fed an NO-supplemented diet (0.83 ± 0.17 SEM) than in rats feed a normal diet (1.25 ± 0.16) (Figure 3).
Figure 3.
In a rat model of reflux oesophagitis, dietary NO supplementation reduces Sox2 expression in the distal squamous oesophagus. Representative photomicrographs of the distal squamous oesophagus of rats with surgically-induced reflux oesophagitis that were fed diets with (lower panels) or without (upper panels) NO supplementation at 4 weeks after surgery. Inflammatory changes are seen by H&E staining in both groups. Sox2 staining is seen predominantly in the nuclei of basal and suprabasal cells, and the intensity of Sox2 staining is weaker in the rats fed an NO-supplemented diet.
NOC-9 does not cause increased BMP signaling
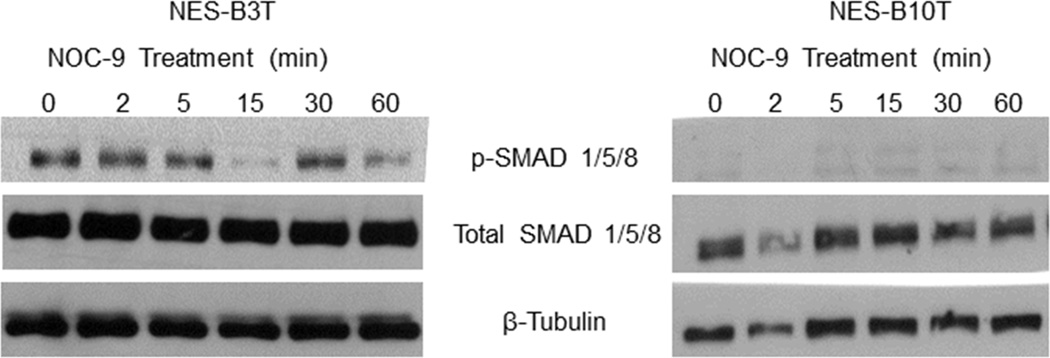
In non-oesophageal cells, activation of BMP pathway signaling has been linked with decreased SOX2 expression. 35, 37 Therefore, we investigated whether NOC-9 might decrease SOX2 expression in NES cells by activating BMP signaling. We performed Western blots for the phosphorylated form of SMAD1/5/8, an indicator of BMP pathway signaling. We found no increase in p-SMAD1/5/8 in either NES-B3T or NES-B10T cells following NOC-9 treatment (Figure 4). This suggests that NOC-9 does not decrease SOX2 expression by increasing BMP pathway signaling in oesophageal squamous cells.
Figure 4.
NOC-9 treatment does not increase BMP pathway signaling in NES cells. Representative experiments of Western blotting for phospho- and total SMAD 1/5/8. NOC-9 (500 µM) was administered at time 0, and the results show the effects at the indicated time points after NOC-9 administration.
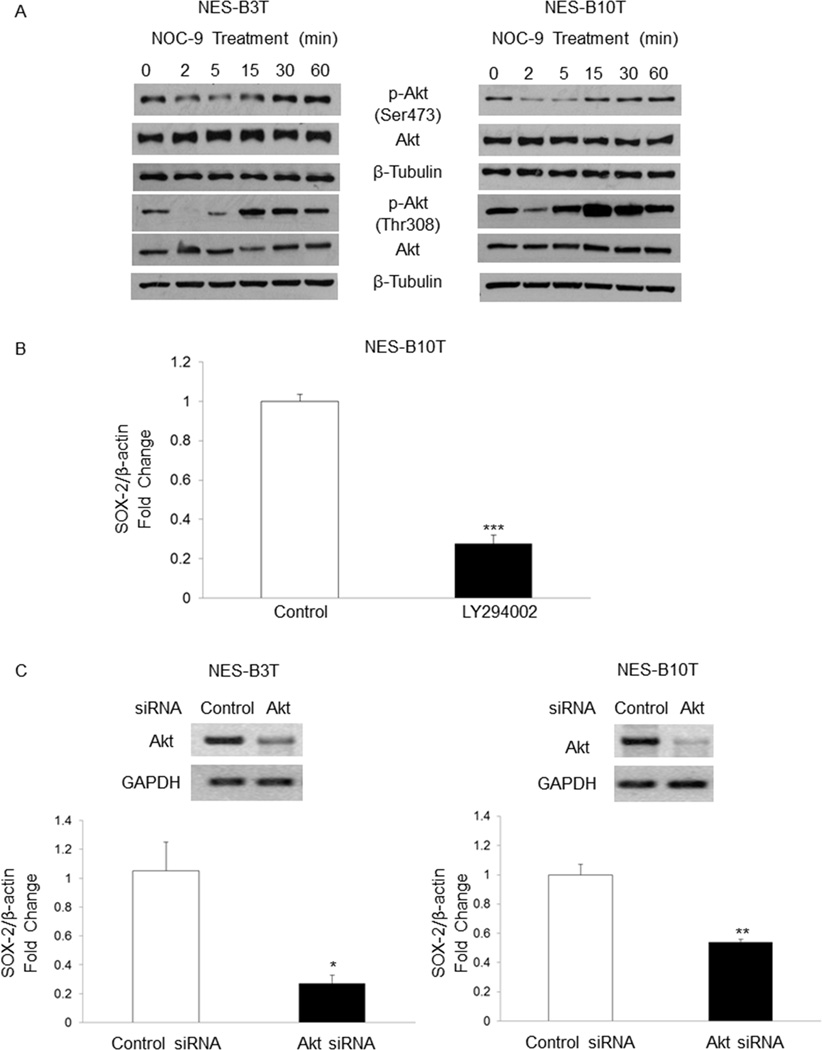
NOC-9 decreases phospho-Akt expression, and Akt signaling regulates SOX2 expression
Suppression of Akt signaling has been linked with decreased expression of SOX2 in non-oesophageal cells. 38, 39 For full activation, Akt must be phosphorylated at 2 distinct sites, i.e. threonine 308 (Thr308) and serine 473 (Ser473). 40 We treated NES cells with NOC-9 and determined effects on Akt phosphorylation at both sites. In both cell lines, NOC-9 decreased phospho-Akt at Ser473 and at Thr308, at 2 and 5 minutes after exposure (Figure 5A). To confirm that Akt signaling was indeed linked with SOX2 expression in oesophageal squamous cells, we inhibited Akt using the pharmacologic inhibitor LY294002 or a specific siRNA, and assessed SOX2 expression by qRT-PCR. Semiquantitative RT-PCR was performed to determine the efficiency of siRNA for inhibiting Akt expression. LY294002 significantly decreased SOX2 expression in NES-B10T cells (Figure 5B), a finding that was confirmed using siRNA against Akt in both NES-B3T and NES-B10T cells (Figure 5C). These data demonstrate that NOC-9 decreases phosphorylation of Akt at both Thr308 and Ser473, and that Akt signaling mediates expression of SOX2 in oesophageal squamous cells from patients with Barrett’s oesophagus.
Figure 5.
Treatment with NOC-9 decreases phosphorylation of Akt at Ser473 and Thr308, and inhibition of Akt signaling decreases SOX2 mRNA in NES cells. (A) Representative experiments of Western blotting for phospho-Akt Ser473 and Thr308. NOC-9 (500 µM) was administered at time 0, and the results show the effects at the indicated time points after NOC-9 administration. Representative experiment of qRT-PCR for SOX2 mRNA in NES cells treatedwith (B) LY294002 for 24 hours or (C) a specific Akt siRNA for 48 hours; gels demonstrate semiquantitative RT-PCR showing Akt knockdown by siRNA at 48 hours. Bar graphs represent the mean ±SEM. *, p ≤ 0.05 compared to non-treated control; **, p ≤ 0.01 compared to non-treated control; ***, p ≤ 0.001 compared to non-treated control.
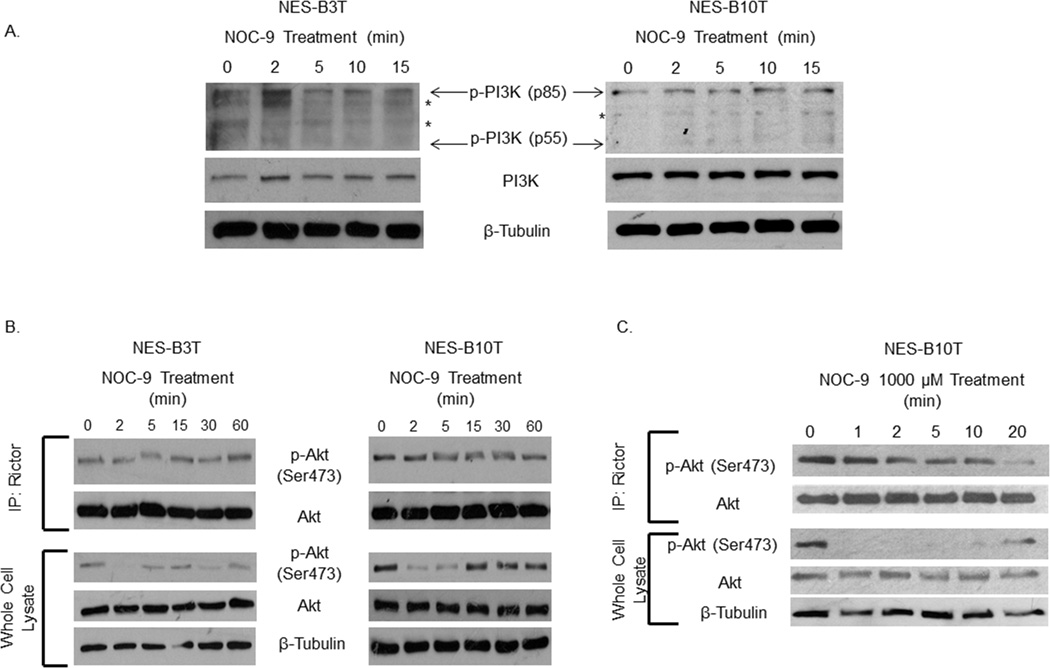
NOC-9 reduces Akt phosphorylation rapidly without affecting PI3 kinase or rictor-mTOR activity
PI3 kinase activity initiates Akt phosphorylation by recruiting Akt to the plasma membrane, along with PDK1 (3-phosphoinositide-dependent kinase-1), which phosphorylates Akt at Thr308. 30, 40 We explored whether NOC-9 might decrease Akt phosphorylation by inhibiting activation (phosphorylation) of PI3 kinase, and found that NOC-9 caused no apparent decrease in PI3 kinase phosphorylation in either cell line (Figure 6A).
Figure 6.
Early NOC-9-induced decreases in Akt phosphorylation are independent of effects on PI3 kinase or rictor-mTOR complex activity in NES cells. (A) Representative experiments of Western blotting for phospho- and total PI3K*, non-specific bands (B) Representative experiments of Western blotting for total and phospho-Akt Ser473 in a rictor-mTOR activity assay (upper blots) or in whole cell lysates (lower blots) from cells treated with 500 µM NOC-9 or (C) NES-B10T cells treated with 1000 µM NOC-9. NOC-9 (500 µM or 1000 µM) was administered at time 0, and the results show the effects at the indicated time points after NOC-9 administration.
Phosphorylation of Akt at Ser473 is accomplished by an mTOR complex that contains rictor (rapamycin-insensitive companion of�mTOR). 30 To determine whether NOC-9 might reduce Akt phosphorylation by decreasing rictor-mTOR activity, we used an in vitro kinase assay in which we immunoprecipitated rictor from NES cells treated with NOC-9, then added exogenous inactive Akt/PKB1 protein (that could be phosphorylated at Ser473 by the immunoprecipitated rictor), and performed Western blots for phospho-Akt Ser473 as an indicator of rictor-mTOR activity. We also compared the time course of NOC-9 effects on phosphorylation of exogenous Akt by immunoprecipitated rictor with those on phosphorylation of endogenous Akt in whole cell lysates. NOC-9 had little effect on phosphorylation of exogenous Akt at Ser473 by immunoprecipitated rictor at any time point (Figure 6B; upper blots). In contrast, a profound decrease in phosphorylation of endogenous Akt at Ser473 was apparent in the whole cell lysates after only 2 minutes of NOC-9 treatment (lower blots). These effects were even more apparent when the cells were treated with a higher concentration of NOC-9 (1000 µM rather than 500 µM, Figure 6C). This high-concentration NOC-9 virtually abolished Akt phosphorylation at Ser473 within 1 minute, whereas NOC-9 effects on immunoprecipitated rictor were not apparent until much later. These findings demonstrate that the rapid effect of NOC-9 in reducing Akt phosphorylation in NES cells is independent of effects on PI3 kinase or on rictor-mTOR activity.
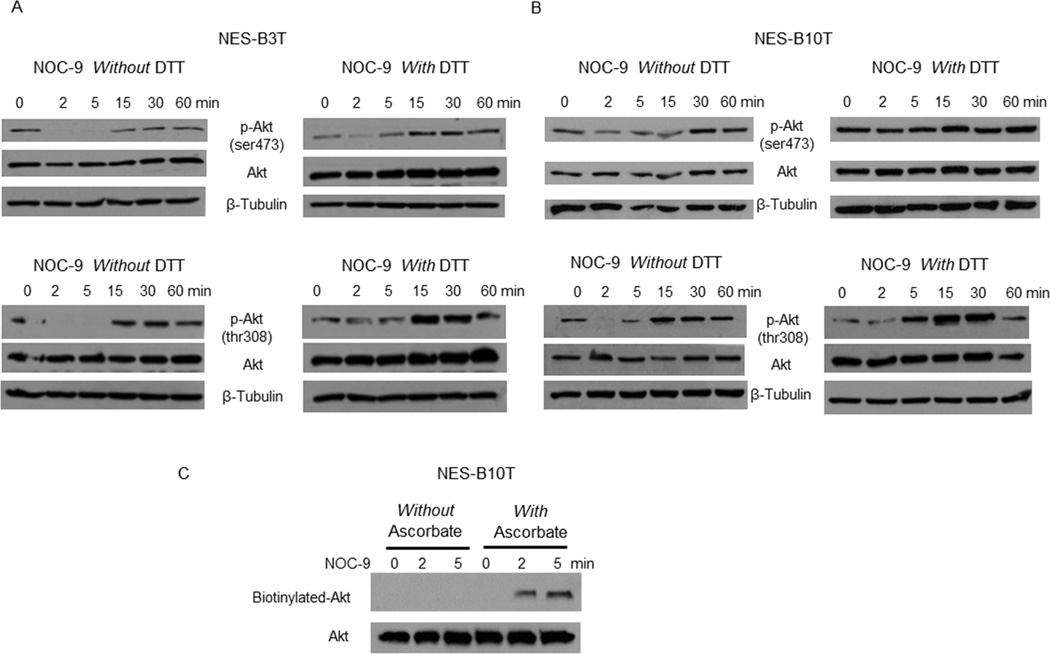
NOC-9 cause S-nitrosylation of Akt
It is known that NO can inactivate Akt protein through S-nitrosylation in which NO converts cysteine thiols into S-nitrosothiols. 31, 41 This S-nitrosylation can be blocked by the reducing agent DTT. 41 We treated NES-B3T and NES-B10T cells with NOC-9 in the presence or absence of DTT, and found that DTT attenuated or abolished the reduction in phospho-Akt Ser473 (Figure 7A&B, top panels) and Thr308 (Figure 7A&B, bottom panels) at 2 and 5 minutes. To confirm that these findings were the result of S-nitrosylation, we performed biotin-switch assays in NES-B10T cells treated with NOC-9. We found increased levels of biotinylated Akt in cells treated with NOC-9 in the presence of ascorbate (confirming S-nitrosylation) at 2 and 5 minutes after exposure (Figure 7C). These findings show that NO causes S-nitrosylation of Akt, which interferes with its phosphorylation at Ser473 and Thr308. A schematic model summarizing proposed mechanisms based on our findings is provided in Figure 8.
Figure 7.
Treatment with NOC-9 induces Akt S-nitrosylation, which interferes with its phosphorylation in NES cells. Representative experiments of Western blotting for total and phospho-Akt Ser473 and Thr308 without and with DTT in (A) NES-B3T and (B) NES-B10T cells. (C) Representative biotin-switch assays in NES-B10T cells. Note the appearance of biotinylated Akt in cells treated with ascorbate, confirming S-nitrosylation. NOC-9 (500 µM) was administered at time 0, and the results show the effects at the indicated time points after NOC-9 administration.
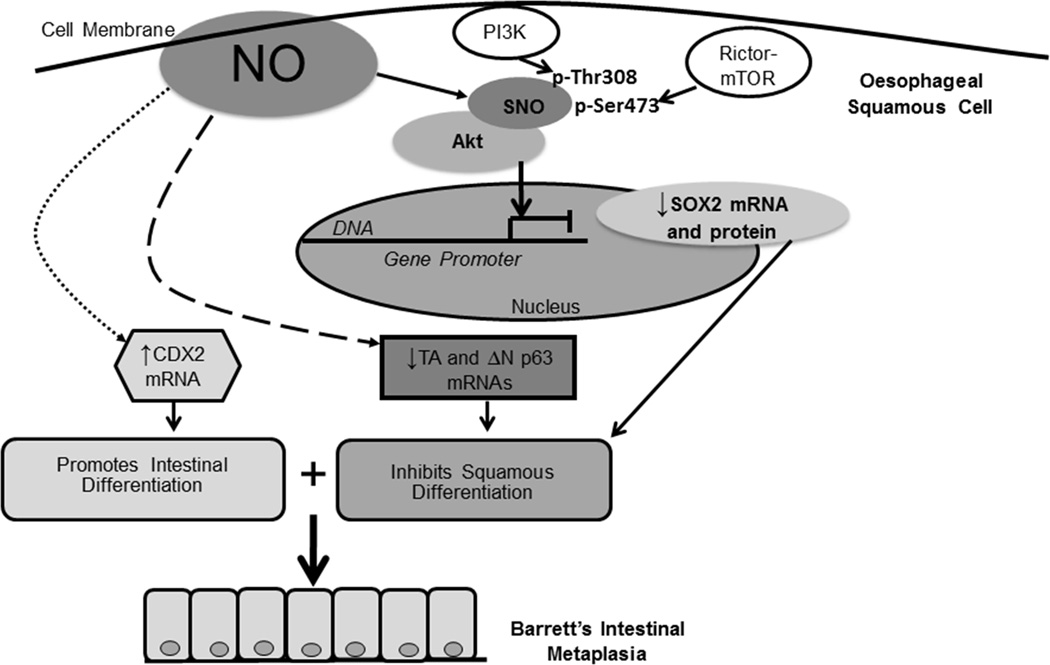
Figure 8.
Schematic model demonstrating the effects of NO on Akt, SOX2, p63 (dashed line), and CDX2 (dotted line) in oesophageal squamous cells. NO causes Akt S-nitrosylation (SNO), which blocks its activation by PI3K and rictor-mTOR, leading to decreased expression of SOX2 mRNA and protein. SOX2 is known to promote the development of stratified squamous epithelium. NO also decreases the expression of p63 isoforms, which also are required for the development of stratified squamous epithelium. At the same time, NO increases the expression of CDX2, which promotes intestinal differentiation. Thus, NO triggers molecular events that inhibit squamous differentiation while promoting intestinal differentiation, a combination that might lead to the development of the intestinal metaplasia of Barrett’s oesophagus.
Discussion
In oesophageal squamous epithelial cells from patients with Barrett’s oesophagus, we have demonstrated that SOX2 mRNA expression is suppressed by exposure to acidic bile salts or NO in physiologically relevant concentrations. These transcriptional suppressive effects are significantly greater with NO than with acidic bile salts, however, and NO also profoundly decreases expression of SOX2 protein. We have shown that NO causes S-nitrosylation of Akt protein, which blocks its phosphorylation at both Thr308 and Ser473, and that interference with Akt signaling in oesophageal squamous cells reduces their expression of SOX2. We have also shown that the squamous-lined distal oesophagus of rats with surgically-induced reflux oesophagitis exhibits diminished staining for SOX2 when the animals are fed a diet supplemented with NO. Finally, we have demonstrated that NO decreases mRNA expression of p63 isoforms, while increasing mRNA expression of CDX2 in oesophageal squamous cells. Thus, NO triggers molecular events that can inhibit squamous differentiation while promoting intestinal differentiation in the oesophagus. These observations elucidate molecular mechanisms whereby NO generated in the oesophagus from dietary nitrate might contribute to the development of Barrett’s metaplasia.
In earlier reports, we proposed that differences among individuals in oesophageal molecular pathways activated by GORD might determine whether that GORD is complicated by development of Barrett’s metaplasia. 16, 19 This proposal emerged from our observations that oesophageal squamous cells from patients with and without Barrett’s oesophagus responded differently to noxious stimuli. For example, acidic bile salts increased CDX2 expression in oesophageal squamous cells from patients with Barrett’s oesophagus, but not in those from patients who had GORD without Barrett’s oesophagus.19 We also observed differences between oesophageal squamous cells from patients with and without Barrett’s oesophagus in signaling through Hedgehog, a key developmental pathway known to influence genes that determine an intestinal phenotype.16 Based on these observations, we elected to use oesophageal squamous cell lines from patients with Barrett’s oesophagus for the studies described in this report.
Most investigations on how noxious agents in refluxed gastric juice might contribute to development of Barrett’s metaplasia have focused on how those agents increase expression of transcription factors that determine a columnar cell phenotype such as CDX2. However, it seems equally plausible that the pathogenesis of a squamous-to-columnar metaplasia also should involve decreased expression of transcription factors that determine a squamous cell phenotype, and few studies have addressed this issue. We have focused on how noxious agents affect SOX2, an endodermal transcription factor that plays a key role in formation of stratified squamous epithelium in the embryo. 22 In mice genetically engineered to express oesophageal Sox2 at levels <50% of normal, the embryonic oesophagus develops regions of columnar lining with foci of multi-layered epithelium similar to those described in Barrett’s oesophagus, and the oesophagus expresses genes normally expressed in the glandular stomach (e.g. trefoil factors 1 and 2, Muc5B). 22, 42 Thus, genetically-induced SOX2 reductions in the embryonic mouse oesophagus shift its gene expression patterns and histological features toward a columnar phenotype. Decreases in oesophageal SOX2 expression have been found in the setting of reflux oesophagitis in animal models and in patients with Barrett’s oesophagus.14 However, very little was known about the mechanisms whereby reflux might downregulate SOX2.
To study how noxious agents in refluxed gastric juice affect oesophageal expression of genes influencing cell phenotype, we treated oesophageal squamous cells with acidic bile salts or with the NO donor NOC-9, using concentrations and exposure durations designed to simulate typical reflux episodes. 23, 43–46 Although most studies on this issue have focused primarily on effects of acid and bile salts, NO in the oesophagus can react with oxygen to form RNS that can have considerable toxic effects. 24 NO is generated from nitrate (NO3−) ingested largely in green leafy vegetables. Some of this nitrate is concentrated in salivary glands and secreted into the mouth, where bacteria reduce the recycled nitrate to nitrite (NO2−). When swallowed nitrite encounters acidic gastric juice containing ascorbic acid, a powerful reducing agent, nitrite is converted rapidly to NO. In a study of healthy volunteers who ingested nitrate in an amount found in typical salad portions, potentially genotoxic concentrations of NO (up to 50 µM) were generated at the gastro-oesophageal junction, the location where nitrite in swallowed saliva normally encounters acidic gastric juice. 23 In patients with GORD and Barrett’s oesophagus, who frequently reflux acid into the oesophagus, high oesophageal concentrations of NO (up to 60 µM) are generated after nitrate ingestion.33 We found that both acidic bile salts and NO suppressed SOX2 expression in oesophageal squamous cells, but SOX2 suppression induced by NO was significantly greater than that for acidic bile salts.
The transcription factor p63, a homolog of the tumor suppressor gene p53, is required for development and maintenance of oesophageal stratified squamous epithelium. 22, 34, 47 Embryonic p63-knockout mice develop an oesophagus lined by columnar cells. 48 The p63 gene encodes two major, functionally-distinctive protein isoforms, TAp63 and ΔNp63. Studies in squamous carcinoma cell lines have demonstrated that SOX2 and ΔNp63 proteins interact physically and cooperate functionally to co-regulate scores of downstream target genes. 34 We found that exposure to NO, in addition to reducing SOX2 expression, also decreased mRNA expression of both the TA and ΔN isoforms of p63 in oesophageal squamous cells.
SOX2 and CDX2, a transcription factor that determines an intestinal-type columnar cell phenotype, appear to have an inverse relationship. 35, 36 Downregulation of SOX2 and upregulation of CDX2 have been associated with intestinal metaplasia in the stomach. 35, 49, 50 Further supporting an inverse relationship for these transcription factors, we found that exposure to NO significantly decreased SOX2 and increased CDX2 mRNA expression in our oesophageal squamous cell lines. We also analyzed tissue specimens of squamous-lined distal oesophagus in rats with surgically-induced GORD that were fed diets with or without NO supplementation. We found weaker nuclear staining for SOX2 in rats fed an NO-supplemented diet than in those on normal diets. At 4 weeks after surgery, furthermore, rats fed NO-supplemented diets developed intestinal metaplasia with Cdx2 expression significantly more often than rats fed normal diets. 32
In certain non-oesophageal cells, decreases in SOX2 expression have been linked either with activation of BMP pathway signaling or with suppression of Akt signaling. 35, 38, 39 We found that NO did not cause appreciable activation of BMP signaling, but did suppress Akt activation markedly. Akt is a serine/threonine kinase with critical regulatory roles in diverse cellular processes including cell differentiation. 51 Recent studies have shown that Akt signaling is a key regulator of SOX2 expression and function. 41, 52 In keeping with these reports, we have found that inhibition of Akt signaling using either a pharmacologic inhibitor or a specific siRNA decreases SOX2 mRNA expression in oesophageal squamous cells.
Akt is activated by phosphorylation both at Thr308 (located in the protein’s activation T-loop) and at Ser473 (located in the hydrophobic motif of the protein’s C-terminal tail). 30, 40 Phosphorylation at Thr308 is initiated by PI3K, which recruits Akt and PDK1 to the plasma membrane, where PDK1 phosphorylates Akt at Thr308. 40 Akt phosphorylation at Ser473 is accomplished through kinase activity of the rictor-mTOR complex, an event that might also depend on PI3K activity. 30, 40 We have found that NO disrupts Akt signaling in oesophageal squamous cells by causing S-nitrosylation of Akt protein. S-nitrosylation is the covalent binding of NO to protein cysteine residues, forming S-nitrosothiols that can interfere with protein function through effects on protein conformation. 53, 54 S-nitrosylation of Akt, which disrupts its kinase activity, can be blocked by the reducing agent DTT. 31, 41 We found that DTT blocks the NOC-9-induced decrease in phosphorylation of Akt at Thr308 and Ser473, indicating that S-nitrosylation of Akt interferes with its phosphorylation. Conceivably, S-nitrosylation of PI3K could interfere with Akt signaling, but we found that NOC-9 treatment caused no reduction in phosphorylated PI3K in oesophageal squamous cells. Using an in vitro kinase assay, we also found that NOC-9 had no appreciable early effect on the ability of rictor-mTOR to phosphorylate exogenous Akt at Ser473. Furthermore, NOC-9 in high concentration virtually abolished phosphorylation of endogenous Akt at Ser473 within 2 minutes, a time point at which NOC-9 did not affect the ability of rictor-mTOR to phosphorylate exogenous Akt at Ser473. These observations demonstrate that NO does not reduce Akt phosphorylation by decreasing PI3K or rictor-mTOR activity, and further support Akt protein S-nitrosylation as the mechanism whereby NO disrupts Akt phosphorylation and signaling.
In conclusion, in oesophageal squamous cells from patients with Barrett’s oesophagus, we have shown that NO causes Akt S-nitrosylation, which blocks its activation by PI3K and rictor-mTOR, and we have shown that NO decreases expression of SOX2, a transcriptional target of Akt. NO also decreases expression of p63 that, like SOX2, is required for the development of stratified squamous epithelium. At the same time, NO increases the expression of CDX2, which can promote intestinal differentiation. Thus, NO triggers molecular events that inhibit squamous differentiation while promoting intestinal differentiation, a combination that might lead to development of the intestinal metaplasia of Barrett’s oesophagus. These observations provide a rationale for research on interventions that might reduce nitric oxide production in the distal oesophagus as a way to prevent Barrett’s oesophagus.
Supplementary Material
Summary Box.
What is already known about this subject?
There is evidence to suggest that Barrett’s oesophagus develops when mature oesophageal squamous cells or their progenitors are converted into columnar cells under the influence of GORD.
A number of studies have shown that oesophageal squamous cells exposed to acid and bile salts increase their expression of genes involved in columnar cell differentiation such as CDX2, but little is known of how these noxious agents influence the expression of genes involved in squamous cell differentiation such as SOX2.
In addition to having abnormal oesophageal exposure to acid and bile, patients with GORD can have high concentrations of nitric oxide (NO) in the oesophagus, but it is not clear how NO might contribute to the pathogenesis of Barrett’s metaplasia.
What are the new findings?
In non-neoplastic oesophageal squamous cell lines derived from patients with Barrett’s oesophagus, NO increases expression of CDX2, decreases expression of p63, and profoundly decreases the expression of SOX2.
NO blocks Akt activation by causing S-nitrosylation of Akt protein, and reduced Akt signaling decreases SOX2 expression.
The in vitro observation that NO downregulates the expression of SOX2 in oesophageal squamous cells is supported in a rat model of reflux oesophagitis in which animals fed a diet supplemented with nitrite and ascorbic acid exhibit diminished staining for SOX2 in the squamous oesophagus.
How might it impact on clinical practice in the foreseeable future?
We have demonstrated that NO triggers a series of molecular events in oesophageal squamous cells that can inhibit squamous differentiation while promoting intestinal differentiation, a combination that might contribute to the pathogenesis of Barrett’s metaplasia.
This study provides a rationale for research on interventions that might reduce NO production in the distal esophagus as a way to prevent the development of Barrett’s oesophagus.
Acknowledgments
This work was supported by Merit Review Award #BX002666 from the U.S. Department of Veterans Affairs Biomedical Laboratory Research Program (S.J.S.), the National Institutes of Health (R01-DK63621 and R01-CA134571 to R.F.S. and S.J.S.; K12 HD-068369-01 and K08-DK099383 to EC; R01-DK097340 to D.H.W.).
Abbreviations
- GORD
Gastro-oesophageal reflux disease
- NES
Normal oesophageal squamous
- SOX2
Sex determining region Y-box 2
- NO
Nitric oxide
- qRT-PCR
Quantitative reverse transcription polymerase chain reaction
- RT-PCR
Reverse transcription polymerase chain reaction
- Akt/PKB
Protein kinase B
- PI3K
Phosphatidylinositol-4,5-biphosphate 3-kinase
- mTOR
Mechanistic target of rapamycin
- Rictor
rapamycin-insensitive companion of mTOR
- p63
Transformation-related protein 63
- CDX2
Caudal type homeobox 2
- SOX9
Sex determining region Y-box 9
- FOXA2
Forkhead box protein A2
- NOC-9
6-(2-Hydroxy-1-methyl-2-nitrosohydrazino)-N-methyl-1-hexanamine
- BMP4
Bone morphogenetic protein 4
- DTT
Dithiothreitol
- SEM
Standard error of the mean
Footnotes
Financial Disclosures: No conflicts of interest exist for any of the authors.
Contributorship Statement:
All authors included on this paper fulfill the criteria of authorship and have approved the submission of this manuscript. Specific contributions are as follows:
Asanuma: study design; technical and material support; analysis and interpretation of data; critical revision of manuscript; important intellectual content; drafting of manuscript
Huo: study design; technical and material support; analysis and interpretation of data; critical revision of manuscript; important intellectual content; drafting of manuscript
Agoston: study design; technical and material support; analysis and interpretation of data; critical revision of manuscript; important intellectual content; drafting of manuscript
Zhang, X: technical and material support; important intellectual content
Yu: technical and material support; important intellectual content
Cheng: technical and material support; important intellectual content
Zhang, Q: technical and material support; important intellectual content
Dunbar: technical and material support; important intellectual content
Pham: technical and material support; important intellectual content
Wang: technical and material support; important intellectual content
Iijima: technical and material support; important intellectual content
Shimosegawa: technical and material support; important intellectual content
Odze: analysis and interpretation of data; critical revision of manuscript; important intellectual content
Spechler: study concept; analysis and interpretation of data; critical revision of manuscript; important intellectual content
Souza: study concept/design; analysis and interpretation of data; critical revision of manuscript; important intellectual content; drafting of manuscript
References
- 1.Spechler SJ, Souza RF. Barrett's esophagus. N Engl J Med. 2014;371:836–845. doi: 10.1056/NEJMra1314704. [DOI] [PubMed] [Google Scholar]
- 2.Rex DK, Cummings OW, Shaw M, et al. Screening for Barrett's esophagus in colonoscopy patients with and without heartburn. Gastroenterology. 2003;125:1670–1677. doi: 10.1053/j.gastro.2003.09.030. [DOI] [PubMed] [Google Scholar]
- 3.Ronkainen J, Aro P, Storskrubb T, et al. Prevalence of Barrett's esophagus in the general population: an endoscopic study. Gastroenterology. 2005;129:1825–1831. doi: 10.1053/j.gastro.2005.08.053. [DOI] [PubMed] [Google Scholar]
- 4.Spechler SJ. Barrett esophagus and risk of esophageal cancer: a clinical review. JAMA. 2013;310:627–636. doi: 10.1001/jama.2013.226450. [DOI] [PubMed] [Google Scholar]
- 5.Thrift AP, Pandeya N, Whiteman DC. Current status and future perspectives on the etiology of esophageal adenocarcinoma. Front Oncol. 2012;2:1–7. doi: 10.3389/fonc.2012.00011. Article 11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hayeck TJ, Kong CY, Spechler SJ, et al. The prevalence of Barrett's esophagus in the US: estimates from a simulation model confirmed by SEER data. Dis Esophagus. 2010;23:451–457. doi: 10.1111/j.1442-2050.2010.01054.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Quante M, Bhagat G, Abrams JA, et al. Bile acid and inflammation activate gastric cardia stem cells in a mouse model of Barrett-like metaplasia. Cancer Cell. 2012;21:36–51. doi: 10.1016/j.ccr.2011.12.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Wang X, Ouyang H, Yamamoto Y, et al. Residual embryonic cells as precursors of a Barrett's-like metaplasia. Cell. 2011;145:1023–1035. doi: 10.1016/j.cell.2011.05.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Shen CN, Burke ZD, Tosh D. Transdifferentiation, metaplasia and tissue regeneration. Organogenesis. 2004;1:36–44. doi: 10.4161/org.1.2.1409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Corbett JL, Tosh D. Conversion of one cell type into another: implications for understanding organ development, pathogenesis of cancer and generating cells for therapy. Biochem Soc Trans. 2014;42:609–616. doi: 10.1042/BST20140058. [DOI] [PubMed] [Google Scholar]
- 11.Yu WY, Slack JM, Tosh D. Conversion of columnar to stratified squamous epithelium in the developing mouse oesophagus. Dev Biol. 2005;284:157–170. doi: 10.1016/j.ydbio.2005.04.042. [DOI] [PubMed] [Google Scholar]
- 12.Boch JA, Shields HM, Antonioli DA, et al. Distribution of cytokeratin markers in Barrett's specialized columnar epithelium. Gastroenterology. 1997;112:760–765. doi: 10.1053/gast.1997.v112.pm9041237. [DOI] [PubMed] [Google Scholar]
- 13.Sawhney RA, Shields HM, Allan CH, et al. Morphological characterization of the squamocolumnar junction of the esophagus in patients with and without Barrett's epithelium. Dig Dis Sci. 1996;41:1088–1098. doi: 10.1007/BF02088224. [DOI] [PubMed] [Google Scholar]
- 14.Chen X, Qin R, Liu B, et al. Multilayered epithelium in a rat model and human Barrett's esophagus: similar expression patterns of transcription factors and differentiation markers. BMC Gastroenterol. 2008;8:1, 1–9. doi: 10.1186/1471-230X-8-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wang DH, Clemons NJ, Miyashita T, et al. Aberrant epithelial-mesenchymal Hedgehog signaling characterizes Barrett's metaplasia. Gastroenterology. 2010;138:1810–1822. doi: 10.1053/j.gastro.2010.01.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Wang DH, Tiwari A, Kim ME, et al. Hedgehog signaling regulates FOXA2 in esophageal embryogenesis and Barrett's metaplasia. J Clin Invest. 2014;124:3767–3780. doi: 10.1172/JCI66603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Milano F, van Baal JW, Buttar NS, et al. Bone morphogenetic protein 4 expressed in esophagitis induces a columnar phenotype in esophageal squamous cells. Gastroenterology. 2007;132:2412–2421. doi: 10.1053/j.gastro.2007.03.026. [DOI] [PubMed] [Google Scholar]
- 18.Mari L, Milano F, Parikh K, et al. A pSMAD/CDX2 complex is essential for the intestinalization of epithelial metaplasia. Cell Rep. 2014;7:1197–1210. doi: 10.1016/j.celrep.2014.03.074. [DOI] [PubMed] [Google Scholar]
- 19.Huo X, Zhang HY, Zhang XI, et al. Acid and bile salt-induced CDX2 expression differs in esophageal squamous cells from patients with and without Barrett's esophagus. Gastroenterology. 2010;139:194.e1–203.e1. doi: 10.1053/j.gastro.2010.03.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Tatsuta T, Mukaisho K, Sugihara H, et al. Expression of Cdx2 in early GRCL of Barrett's esophagus induced in rats by duodenal reflux. Dig Dis Sci. 2005;50:425–431. doi: 10.1007/s10620-005-2452-9. [DOI] [PubMed] [Google Scholar]
- 21.Moons LM, Bax DA, Kuipers EJ, et al. The homeodomain protein CDX2 is an early marker of Barrett's oesophagus. J Clin Pathol. 2004;57:1063–1068. doi: 10.1136/jcp.2003.015727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Que J, Okubo T, Goldenring JR, et al. Multiple dose-dependent roles for Sox2 in the patterning and differentiation of anterior foregut endoderm. Development. 2007;134:2521–2531. doi: 10.1242/dev.003855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Iijima K, Henry E, Moriya A, et al. Dietary nitrate generates potentially mutagenic concentrations of nitric oxide at the gastroesophageal junction. Gastroenterology. 2002;122:1248–1257. doi: 10.1053/gast.2002.32963. [DOI] [PubMed] [Google Scholar]
- 24.Jaiswal M, LaRusso NF, Gores GJ. Nitric oxide in gastrointestinal epithelial cell carcinogenesis: linking inflammation to oncogenesis. Am J Physiol Gastrointest Liver Physiol. 2001;281:G626–G634. doi: 10.1152/ajpgi.2001.281.3.G626. [DOI] [PubMed] [Google Scholar]
- 25.Zhang HY, Zhang X, Chen X, et al. Differences in activity and phosphorylation of MAPK enzymes in esophageal squamous cells of GERD patients with and without Barrett's esophagus. Am J Physiol Gastrointest Liver Physiol. 2008;295:G470–G478. doi: 10.1152/ajpgi.90262.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Liu T, Zhang X, So CK, et al. Regulation of Cdx2 expression by promoter methylation, and effects of Cdx2 transfection on morphology and gene expression of human esophageal epithelial cells. Carcinogenesis. 2007;28:488–496. doi: 10.1093/carcin/bgl176. [DOI] [PubMed] [Google Scholar]
- 27.Iijima K, Fyfe V, McColl KE. Studies of nitric oxide generation from salivary nitrite in human gastric juice. Scand J Gastroenterol. 2003;38:246–252. doi: 10.1080/00365520310000708a. [DOI] [PubMed] [Google Scholar]
- 28.Cheng E, Zhang X, Huo X, et al. Omeprazole blocks eotaxin-3 expression by oesophageal squamous cells from patients with eosinophilic oesophagitis and GORD. Gut. 2013;62:824–832. doi: 10.1136/gutjnl-2012-302250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Vlahos CJ, Matter WF, Hui KY, et al. A specific inhibitor of phosphatidylinositol 3-kinase, 2-(4-morpholinyl)-8-phenyl-4H-1-benzopyran-4-one (LY294002) J Biol Chem. 1994;269:5241–5248. [PubMed] [Google Scholar]
- 30.Sarbassov DD, Guertin DA, Ali SM, et al. Phosphorylation and regulation of Akt/PKB by the rictor-mTOR complex. Science. 2005;307:1098–1101. doi: 10.1126/science.1106148. [DOI] [PubMed] [Google Scholar]
- 31.Yasukawa T, Tokunaga E, Ota H, et al. S-nitrosylation-dependent inactivation of Akt/protein kinase B in insulin resistance. J Biol Chem. 2005;280:7511–7518. doi: 10.1074/jbc.M411871200. [DOI] [PubMed] [Google Scholar]
- 32.Endo H, Iijima K, Asanuma K, et al. Exogenous luminal nitric oxide exposure accelerates columnar transformation of rat esophagus. Int J Cancer. 2010;127:2009–2019. doi: 10.1002/ijc.25227. [DOI] [PubMed] [Google Scholar]
- 33.Suzuki H, Iijima K, Scobie G, et al. Nitrate and nitrosative chemistry within Barrett's oesophagus during acid reflux. Gut. 2005;54:1527–1535. doi: 10.1136/gut.2005.066043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Watanabe H, Ma Q, Peng S, et al. SOX2 and p63 colocalize at genetic loci in squamous cell carcinomas. J Clin Invest. 2014;124:1636–1645. doi: 10.1172/JCI71545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Camilo V, Barros R, Sousa S, et al. Helicobacter pylori and the BMP pathway regulate CDX2 and SOX2 expression in gastric cells. Carcinogenesis. 2012;33:1985–1992. doi: 10.1093/carcin/bgs233. [DOI] [PubMed] [Google Scholar]
- 36.Raghoebir L, Bakker ER, Mills JC, et al. SOX2 redirects the developmental fate of the intestinal epithelium toward a premature gastric phenotype. J Mol Cell Biol. 2012;4:377–385. doi: 10.1093/jmcb/mjs030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Lee MY, Lim HW, Lee SH, et al. Smad, PI3K/Akt, and Wnt-dependent signaling pathways are involved in BMP-4-induced ESC self-renewal. Stem Cells. 2009;27:1858–1868. doi: 10.1002/stem.124. [DOI] [PubMed] [Google Scholar]
- 38.Dogan I, Kawabata S, Bergbower E, et al. SOX2 expression is an early event in a murine model of EGFR mutant lung cancer and promotes proliferation of a subset of EGFR mutant lung adenocarcinoma cell lines. Lung Cancer. 2014;85:1–6. doi: 10.1016/j.lungcan.2014.03.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Singh S, Trevino J, Bora-Singhal N, et al. EGFR/Src/Akt signaling modulates Sox2 expression and self-renewal of stem-like side-population cells in non-small cell lung cancer. Mol Cancer. 2012;11:73, 1–15. doi: 10.1186/1476-4598-11-73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Scheid MP, Woodgett JR. Unravelling the activation mechanisms of protein kinase B/Akt. FEBS Lett. 2003;546:108–112. doi: 10.1016/s0014-5793(03)00562-3. [DOI] [PubMed] [Google Scholar]
- 41.Numajiri N, Takasawa K, Nishiya T, et al. On-off system for PI3-kinase-Akt signaling through S-nitrosylation of phosphatase with sequence homology to tensin (PTEN) Proc Natl Acad Sci U S A. 2011;108:10349–10354. doi: 10.1073/pnas.1103503108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Glickman JN, Chen YY, Wang HH, et al. Phenotypic characteristics of a distinctive multilayered epithelium suggests that it is a precursor in the development of Barrett's esophagus. Am J Surg Pathol. 2001;25:569–578. doi: 10.1097/00000478-200105000-00002. [DOI] [PubMed] [Google Scholar]
- 43.Vaezi MF, Richter JE. Role of acid and duodenogastroesophageal reflux in gastroesophageal reflux disease. Gastroenterology. 1996;111:1192–1199. doi: 10.1053/gast.1996.v111.pm8898632. [DOI] [PubMed] [Google Scholar]
- 44.Kauer WK, Peters JH, DeMeester TR, et al. Composition and concentration of bile acid reflux into the esophagus of patients with gastroesophageal reflux disease. Surgery. 1997;122:874–881. doi: 10.1016/s0039-6060(97)90327-5. [DOI] [PubMed] [Google Scholar]
- 45.Nehra D, Howell P, Williams CP, et al. Toxic bile acids in gastro-oesophageal reflux disease: influence of gastric acidity. Gut. 1999;44:598–602. doi: 10.1136/gut.44.5.598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Boeckxstaens GE, Smout A. Systematic review: role of acid, weakly acidic and weakly alkaline reflux in gastro-oesophageal reflux disease. Aliment Pharmacol Ther. 2010;32:334–343. doi: 10.1111/j.1365-2036.2010.04358.x. [DOI] [PubMed] [Google Scholar]
- 47.Pellegrini G, Dellambra E, Golisano O, et al. p63 identifies keratinocyte stem cells. Proc Natl Acad Sci U S A. 2001;98:3156–3161. doi: 10.1073/pnas.061032098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Yang A, Schweitzer R, Sun D, et al. p63 is essential for regenerative proliferation in limb, craniofacial and epithelial development. Nature. 1999;398:714–718. doi: 10.1038/19539. [DOI] [PubMed] [Google Scholar]
- 49.Tsukamoto T, Inada K, Tanaka H, et al. Down-regulation of a gastric transcription factor, Sox2, and ectopic expression of intestinal homeobox genes, Cdx1 and Cdx2: inverse correlation during progression from gastric/intestinal-mixed to complete intestinal metaplasia. J Cancer Res Clin Oncol. 2004;130:135–145. doi: 10.1007/s00432-003-0519-6. [DOI] [PubMed] [Google Scholar]
- 50.Tsukamoto T, Mizoshita T, Mihara M, et al. Sox2 expression in human stomach adenocarcinomas with gastric and gastric-and-intestinal-mixed phenotypes. Histopathology. 2005;46:649–658. doi: 10.1111/j.1365-2559.2005.02170.x. [DOI] [PubMed] [Google Scholar]
- 51.Franke TF, Kaplan DR, Cantley LC. PI3K: downstream AKTion blocks apoptosis. Cell. 1997;88:435–437. doi: 10.1016/s0092-8674(00)81883-8. [DOI] [PubMed] [Google Scholar]
- 52.Jeong CH, Cho YY, Kim MO, et al. Phosphorylation of Sox2 cooperates in reprogramming to pluripotent stem cells. Stem Cells. 2010;28:2141–2150. doi: 10.1002/stem.540. [DOI] [PubMed] [Google Scholar]
- 53.Qu Z, Meng F, Bomgarden RD, et al. Proteomic quantification and site-mapping of S-nitrosylated proteins using isobaric iodoTMT reagents. J Proteome Res. 2014;13:3200–3211. doi: 10.1021/pr401179v. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Gu Z, Nakamura T, Lipton SA. Redox reactions induced by nitrosative stress mediate protein misfolding and mitochondrial dysfunction in neurodegenerative diseases. Mol Neurobiol. 2010;41:55–72. doi: 10.1007/s12035-010-8113-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.