Abstract
Non-enzymatic glycation (NEG) is an age-related process accelerated by diseases like diabetes, and causes the accumulation of advanced glycation end-products (AGEs). NEG-mediated modification of bone’s organic matrix, principally collagen type-I, has been implicated in impairing skeletal physiology and mechanics. Here, we present evidence, from in vitro and in vivo models, and establish a causal relationship between collagen glycation and alterations in bone fracture at multiple length scales. Through atomic force spectroscopy, we established that NEG impairs collagen’s ability to dissipate energy. Mechanical testing of in vitro glycated human bone specimen revealed that AGE accumulation due to NEG dramatically reduces the capacity of organic and mineralized matrix to creep and caused bone to fracture under impact at low levels of strain (3000–5000 μstrain) typically associated with fall. Fracture mechanics tests of NEG modified human cortical bone of varying ages, and their age-matched controls revealed that NEG disrupted microcracking based toughening mechanisms and reduced bone propagation and initiation fracture toughness across all age groups. A comprehensive mechanistic model, based on experimental and modeling data, was developed to explain how NEG and AGEs are causal to, and predictive of bone fragility. Furthermore, fracture mechanics and indentation testing on diabetic mice bones revealed that diabetes mediated NEG severely disrupts bone matrix quality in vivo. Finally, we show that AGEs are predictive of bone quality in aging humans and have diagnostic applications in fracture risk.
Introduction
Bone matrix is a composite of mainly type-I collagen and mineral, and smaller quantities of non-collagenous proteins [1]. The ability of bone to resist fracture is determined not only by bone mineral density, as previously thought, but also by the quality of its organic extracellular matrix [2, 3]. Type-I collagen, which comprises over 90% of the organic matrix, imparts ductility and toughness to bone. Collagen is built of tropocollagen triple helical molecules that self assemble into larger fibrils, a few hundred nanometers in diameter, and exhibit the characteristic 67 nm D-periodicity [4]. Self-assembly of collagen involves the formation of systematic enzymatic crosslinks such as pyrrole and pyridinoline, [5, 6]. Enzymatic collagen cross-links mature up to 15 years of age [7, 8] and are instrumental in providing collagen the necessary stability and mechanical competence to resist deformation. Various studies [9, 10, 11] have demonstrated that fibrils undergo periodic molecular deformation and stretching under force, which results in energy dissipation and retardation of crack growth within the bone matrix [12, 13, 14].
Aging or diseases like diabetes cause collagen type-I to crosslink through non-enzymatic glycation (NEG), resulting in the formation of advanced glycation endproducts (AGEs) [15, 16]. NEG-mediated crosslinking involves a reaction between an aldehyde of a reducing sugar (glucose or ribose) and amino groups of lysine or hydroxylysine present on collagen. The resultant aldimine complex rearranges to form a Schiff base or Amadori product, which subsequently undergoes reactions with other amino groups to form AGE crosslinks [17]. AGEs can form within the fibril and between individual collagen fibrils, and their number can increase up to five times with age [18, 19]. They have been correlated to reduced bone toughness [20, 21, 22]. Non-enzymatic glycation and AGE accumulation, due to aging, not only deteriorate bone quality and material properties [23, 24], but also increase stiffness and brittleness in other musculoskeletal tissues like cartilage [25] and tendon [26]. In spite of our growing understanding of glycation in bone tissue, there is no evidence to establish the mechanism by which molecular level modifications of bone collagen impair energy dissipation of bone and cause fracture. Furthermore, from a clinical perspective, it is unknown if NEG alters bone’s response to suddenly applied impact loading, typical of falls, and cause it to fracture. Cortical bone bears impact of loading during fracture but it is not established if effect of NEG is ubiquitous across age and in diseases such as diabetes that, despite higher bone mineral density, show increased incidence of bone fracture [27].
In this study, we hypothesized that glycation results in matrix level modifications across the scales of hierarchy in bone matrix, and these modifications cause, and are not simply correlative to, the reduction in bone mechanical properties observed in previous studies. Our objective was to use both, in vitro, and in vivo tests, to evaluate the hypothesis. Specifically, using in vitro mechanical studies on glycated collagen, glycated human bone specimens and their age matched controls, and in vivo diabetic animal models, we show how AGE accumulation in bone collagen causes an impairment in biomechanical properties. The use of age matched controls allows us to attribute NEG (glycation doubles AGE after 7 days of treatment, equivalent to 30 yrs of aging) as a cause of decreased bone fragility.
Materials and Methods
AFM studies on collagen type-I
Collagen fibrils were produced in-vitro using dissolved rat tail collagen (BD Biomedicals) and 1X phosphate buffered saline (PBS). The collagen solution was warmed up to room temperature and titrated with 1X PBS to achieve a physiological pH of 7.4. This procedure was followed by incubation of the resulting mixture for 18 hours at 37°C to allow formation of collagen fibrils. After their formation, the fibrils were centrifuged and removed as control specimen, or glycated using 1.5M ribose solution. Samples were dried on a glass cover slip at 37°C and imaged using MFP 3D Atomic Force Microscope (Asylum Research, Santa Barbara, CA) and AC 160 cantilevers (Asylum Research, k = 40 N/m, fo = 300 kHz). Fibril diameter (D) was measured using a section analysis tool (N = 8) (Igor). Force spectroscopy using cantilever approach and retraction rates of 5 μm/sec were carried out in PBS under ambient conditions using Olympus Biolever cantilevers (k = 0.025N/m). An intermittent dwell time of 4 seconds was incorporated to facilitate binding between the AFM tip and fibrils on sample surface. Area under the force-distance curves, a combined measure of interaction between collagen fibrils and tip-fibril interaction, was quantified (N = 5). Our peak forces and extensions were similar to those reported in other studies on collagen type-I [28, 29].
In vitro Non-Enzymatic Glycation
Aliquots (10 ml) of the precipitated collagen were sealed in dialysis tubing with a pore size of 300 nm. These were placed in ribosylation and control solutions for a period of 7 days at 37°C and pH was maintained between 7.2 and 7.4. The ribosylation solutions contain 1.5M of ribose, protease inhibitors to prevent enzymatic reactions (25 mM ε-amino-n-caproic acid, 5 mM benzamidine, 10 mM N-ethylmaleimide) and 30 mM HEPES in Hanks buffer [15]. The control solution had the same composition as the ribosylation solution but contains no ribose.
AGE Assay
For all samples, total fluorescent AGEs were measured by a fluorometric assay [20]. Cortical bone samples were freeze-dried and hydrolyzed in 6N HCl for 20 hours at 110°C to obtain sample hydrosylates. Hydrosylates were centrifuged at 12000 rpm for 30 min and the supernatant was obtained. Using an Infinite 200 microplate reader (Tecan; 2450 Zanker Road, San Jose, CA 95131, USA) fluorescence was measured for the hydrosylate supernatants and normalized against serially diluted quinine standards (stock: 10 μg/mL quinine per 0.1 N sulfuric acid) at 360/460 nm (excitation/emission). For the hydroxyproline measurements, chloramine-T, perchloric acid (3.15 M) and p-dimethylaminobenzaldehyde (PDB) solutions were made immediately before use. Chloramine-T was added to hydrosylate supernatants and serially diluted hydroxyproline standards (stock: 2000 μg/mL L-hydroxyproline per 0.001 N HCl). The mixture was incubated at room temperature for 20 minutes to allow for hydroxyproline oxidation. 3.15 M perchloric acid was added to quench residual chloramine-T and the mixture was incubated at room temperature for 5 minutes. P-dimethylaminobenzaldehyde solution was then added, and the mixture was further incubated for 20 minutes at 60°C. All samples including standards were cooled in the dark for 5 minutes, and the absorbance was measured at 570 nm using the same microplate reader. Collagen content in all the samples was obtained through hydroxyproline measurements. Fluorescence was normalized to the amount of collagen to calculate total fluorescent AGEs as ng of quinine sulfate fluorescence/mmol of collagen.
Multicyclic Creep and Stress Relaxation Tests
Mid-diaphyses of bilateral adult human tibiae (Age: 46yrs, Sex: Female), obtained from National Disease Research Interchange (NDRI), were cleaned and machined on a Denford CNC Microturn Lathe, into 18 dumbbell specimens (ASTM E 8M-88), under wet conditions. The specimens were machined into a 10mm gage length, with a diameter of 3mm, tapering off to a grip of 4mm diameter. The specimens were split into two groups of 9 each. The glycation group, was immersed in a 0.6M ribose solution at 37°C for 7 days as per the method described above. The control group was immersed in the same solution without any ribose. Specimens were subjected to standard stress controlled creep tests using a multiple load cycles, under physiological conditions (0.9% saline drip at 37°C) on a MTS 858 Mini Bionix II (MTS Systems Corporation, 14000 Technology Drive, Eden Prairie, MN 5344-2290). Each load cycle comprised a one-second ramp-up load, one-minute hold period under constant load, one-second ramp-down load to zero and one-minute hold at zero load. The above sequence was repeated several times on each specimen using progressively increasing loads.
For the stress relaxation tests, six millimeter long cylindrical specimen (n=12; six pairs) were demineralized in disodium EDTA (0.5M, pH 8.3, 20°C) for 40 days. Demineralization was verified by x-ray analysis and calcium end-point assay. Stress relaxation involved loading each cylinder under physiological conditions on an MTS servo-hydraulic testing machine under strain control to a maximum strain of 50% at a rate of 300 microstrain per second followed by a strain period under strain control, during which the specimen was held at a constant length allowing the stress to fall and stabilize. An equation with time dependence similar to the solution of a one-dimensional diffusion equation was fitted to stress (σ) versus time (t) data using the user defined equation function in TableCurve 2D v5 (Systat Software Inc. Canal Boulevard, Richmond, CA). The coefficients σeq, Δσ0, and D in the above equation describe stress at equilibrium, change in stress, and the characteristics of a specimen undergoing stress relaxation due to fluid diffusion, respectively. Stress at equilibrium (σeq) can be divided by hold strain to estimate equilibrium modulus (HA). Equilibrium modulus is a material property of a poroelastic material.
Compact Tension Testing and Microdamage Assessment
Longitudinal compact tension specimens (Width = 14 mm; Thickness = 3 mm, Initial Crack Length= 7mm) were prepared from the tibial mid-diaphysis of 9 male/female donors, aged 34–85yrs. Six specimens were machined from each donor and all specimens were divided into two groups (glycated and control) of 27 each, and subjected to fracture mechanics tests (ASTM E399). A crack propagation gage (Micromeasurement Ltd., TK-09-CPB02) was mounted on each compact specimen, and the specimen was loaded at 37°C on an MTS servo-hydraulic testing machine at 0.05 mm/min, under a constant irrigation of saline until a total crack extension of 2.25 mm was achieved. Subsequently, each specimen was sectioned for microdamage assessment and scanning electron microscopy (Carl Zeiss Supra SEM, Carl Zeiss Microscopy, Thornwood, U.S.A.).
Cohesive Modeling Studies
Crack growth in compact tension (CT) specimens (width = 14 mm, thickness = 3mm) was modeled using cohesive finite element modeling, which is a phenomenological traction-crack opening displacement relationship that captures the nonlinear fracture behavior of bone. Finite element models of CT specimens were created using a finite element program FEAP (v7.1, 1999). The model was meshed with plane strain quadrilateral elements. In order to elucidate the effects of experimental observations reported in this study on bone fracture resistance, three different finite element models were generated to investigate the effect of uncracked ligament overlap length on the toughening behavior of bone. The first model incorporated a straight crack where the crack propagation occurred along a single line with no uncracked ligament bridging. The second and third models included a crack with a fixed lateral shift of 20 μm and two different overlap lengths of 10 and 50 μm representing uncracked ligament bridges. The length of the crack segments, the overlap length, and the amount of lateral shift of the crack used in these simulations were deduced from experimental observations reported in this paper. Further details are provided in the Supplementary Methods.
Small Animal Bone Fracture Mechanics
Diabetes and hyperglycemia have been linked to increased AGE content in multiple tissues. In order to establish the relationship between glycation and bone fracture in vivo, femora from 6 months old non-diabetic and diabetic mice (n=5–6 in each case) were harvested and notched in the anterior mid-diaphyseal region using a slow speed diamond blade (Buehler). The anterior side was chosen to mimic natural loading conditions during three-point bending tests. Notched bones were soaked in saline for an hour prior to testing. The bending tests were performed on a custom made fixture, in the displacement feedback mode (Elf 3200, EnduraTEC), at cross-head rate of 0.001mm/s until fracture. The resulting load-displacement curves were used to compute propagation toughness. AGE quantification in these bones was done as described previously.
Reference Point Indentation
Reference point indentation (RPI) using BioDent (Active Life Scientific, Santa Barbara, California) purports to assess bone matrix quality by measuring the resistance that bone matrix offers crack formation. Indentation distance increase (IDI) is a measure of how far the indentor probe has traversed into bone matrix, and has been directly linked to fracture resistance in bone, in vivo, in animal models and humans (i.e. higher IDI, lower fracture toughness) [30]. RPI was employed to assess bone material quality of the non-diabetic and diabetic mice bones and verify the differences seen in the fracture toughness. Cyclic indentation tests were performed on the periosteal and anterior surface of the fractured bones at three points for each specimen. Care was taken to indent away from the fracture surface to avoid regions of damage. Indentations were performed with a maximum applied load of 3 N for 10 cycles with a frequency of 2 Hz.
Results
Glycation reduces interfibrillar energy dissipation in collagen
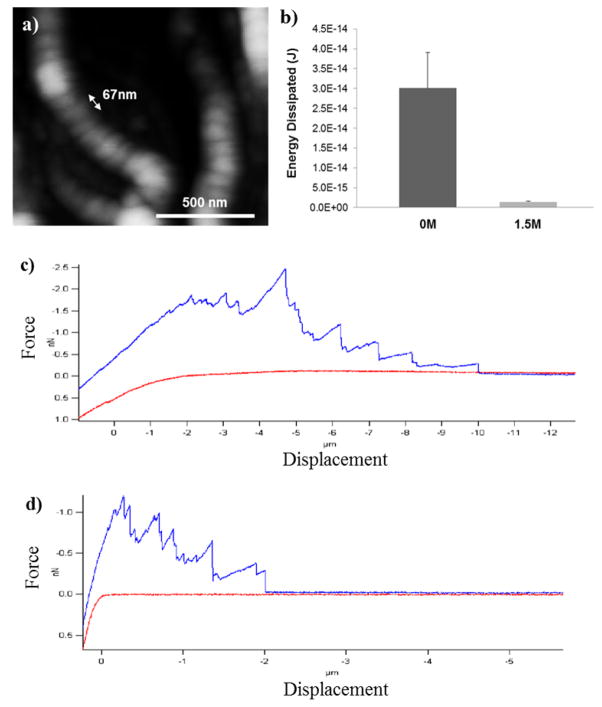
Atomic force microscopy (AFM) and spectroscopy was used to investigate the effect of NEG on collagen type-I energy dissipation. AFM images verified fibril formation (Fig. 1a). Quantification of AGEs in collagen, glycated with varying concentrations of ribose, showed an increase in AGEs with an increase in the molarity of the glycation solution (Supplementary Results). In contrast to large deflections associated with tip-sample interactions, force-extension curves revealed deformations in multiple microns (2–10μm) that are characteristic of fibrillar interactions [28]. Comparison of area under force-extension curve, obtained through force spectroscopy, demonstrated that the mean energy dissipated by the glycated fibrils (1.4E-15 ± 3.2E-16 Nm) during deformation was an order of magnitude smaller than that dissipated by control specimen (3.014E-14 ± 2.014E-14 Nm) (Fig. 1b; p=0.02). Force-extension curves also showed that the extent of deformation in the 1.5 M ribose glycated sample was lower than in controls, with the respective deformations being 2 and 10 micrometers, respectively (Fig. 1c–1d).
Figure 1.
(a) In vitro synthesized collagen fibrils showing 67nm banding. (b) Quantitative determination of energy dissipated by force spectroscopy. Energy dissipation decreases with the increased molarity of ribose in glycation solution (p<0.001). (c) Representative force curve on a control sample. Interactions between the fibrils are sustained for up to 10 microns, with peak forces reaching 2.5nN. (d) Representative force curve on a 1.5M glycated sample. Interfibrillar interactions terminate at about 2 microns, with peak forces peaking at 1nN.
NEG reduces ductility of organic and mineralized bone matrix and fracture toughness of bone
We performed multicyclic creep on multiple specimen derived from the tibiae of a 46 year old human female, to evaluate the effect of glycation on the organic and mineralized matrix. We also performed crack propagation tests on glycated and control samples from 9 human donors from ages 34 to 85yrs, to determine the effect of glycation on fracture properties of mineralized bone matrix. Fluorescence analysis to quantitatively determine NEG revealed a doubling of AGE content (p<0.00001) in the glycated bone (640±88ng quinine/mg collagen) as compared to the control group (314±37ng quinine/mg collagen). This is consistent with previously reported data on cancellous bone showing that a week of in-vitro glycation is equivalent to 3 decades of NEG in vivo [20].
Our mechanical testing data indicate that AGE accumulation alters organic matrix of bone. Stress relaxation tests were conducted on glycated and control demineralized cylindrical specimen. Stress relaxation data was fitted to a one-dimensional diffusion model and statistical data analyses with paired t-tests revealed that the stress in demineralized bone at equilibrium (σeq) was lower in control (1.177±0.487) than in the glycated group (2.486±0.234) (p<0.01). The control specimen showed lower equilibrium modulus (HA) than glycated group (control = 2.355±0.974; glycated = 4.168±0.467; p<0.01) suggesting stiffening and higher accumulation of residual stress in the organic matrix, with glycation.
Multicyclic creep tests on mineralized bone showed that glycated and control specimens fail at similar stress levels of 110 and 115 MPa, respectively. However, there was a 42% strain reduction in the glycated group (0.37% strain) at failure compared to the control group (0.64% strain) (Fig. 2a, p < 0.03). Significant differences in the steady creep behavior between the mineralized control and glycated groups were observed at all stress levels ≥ 95 MPa (p < 0.03). The control group’s creep rate (34.59 μstrain/s) was 3 times or more than the glycated group (9.61 μstrain/s) for all stress levels ≥ 110 MPa (Fig. 2b, p < 0.04). The glycated group also had significantly lower residual strain (700 ± 169 μstrain) than the control group (1163 ± 265 μstrain) for all stress levels ≥ 105 MPa (>40% reduction in residual strain upon glycation) (p < 0.01). Furthermore, there was absence of non-linearity, in the stress-strain curve of glycated bone (Supplementary Results). This result, in conjunction with lower creep rate and residual strains, indicated lesser energy dissipation and brittle nature of fracture of glycated bone.
Figure 2.
a) Glycated bone failed at 42% lower strain than control and at upper threshold of physiological loading of ~3000 μstrain. b) Control group’s creep rate was 3 times faster than the glycated group for all stress levels ≥ 110 MPa. c) No difference was observed in initiation fracture toughness between control and glycated. d) Average values of propagation fracture toughness (R-curve slope) in control and glycated groups demonstrate a highly significant difference (p < 0.003). (e) Average R-curves for control and glycated groups from a typical donor (62/F), and linear regression correlation coefficients (r2 = 0.85 and 0.63 for the control and glycated groups, respectively).
Fracture mechanics tests on mineralized bone specimen showed that there was no significant difference in the average initiation toughness (KIC) values of the control (2.26 ± 0.31) and glycated (2.27 ± 0.33) groups (Fig. 2c; p = 0.91). In contrast to initiation, the in vitro non-enzymatic glycation caused a 35% reduction in the average R-curve slope. The glycated group demonstrated a significantly lower average R-curve slope (0.41 ± 0.18 MPa) than the control group (0.63 ± 0.14 MPa) (Fig. 2d; p < 0.003). R-curve slopes for glycated and control specimens are shown in Fig. 2e.
NEG disrupts microcrack based toughening mechanisms in bone
Following mechanical testing, we performed a microscopic evaluation of 100 micron thick compact tension sections, with scanning electron microscopy (SEM), to quantify microdamage and evaluate toughening mechanisms. The results demonstrate that NEG significantly altered microcracking in bone. Control samples showed greater microcrack density (Fig. 3a; p<0.05) around the propagating start-stop type of microcracks than the glycated specimen. Glycation increased length of the individual cracks that form during the start-stop type of crack growth in bone and increased uncracked ligament length (Fig. 3b, c).
Figure 3.
A compromise in matrix toughening with NEG. a) Glycated samples exhibited lesser microcrack density associated with microcrack based toughening, b) an increase in crack segment length and c) an increase in uncracked ligament length, as compared to the controls. d) Schematics of the crack path in the model with no uncracked ligament bridging (top) uncracked ligament bridging with 10 and 50 μm overlap (bottom). (e) CT specimen geometry for fracture toughness simulations of human cortical bone (B = 3 mm, h = 8.4 mm, W = 14 mm and a/W = 0.52 before crack growth). The dotted arrows show the direction of osteonal orientation. Black line shows the location of the cohesive elements. The gray line denotes the starter notch. (f) Finite element mesh of the CT specimen. (g) Close up view of the deformed shape of the finite element model showing uncracked ligaments.
Because increase in uncracked ligament length with loss in fracture toughness is counter-intuitive to the concept of uncracked ligaments as a toughening mechanism, we performed finite element analysis (FEA) based cohesive modeling of the compact tension specimen to determine the contribution of uncracked ligaments overlap length on fracture toughness of glycated and control bone (Fig. 3d–3g). Our results revealed that a single microcrack resulted in a toughness of 2.45 MPa.m1/2. Uncracked ligaments with an overlap length of 10μm and 50μm, resulted in toughness values of 2.95 MPa m1/2 and 2.88 MPa m1/2 respectively. Hence, an increase in uncracked ligament overlap length (as seen in glycated bone, Fig. 3c) from 10μm to 50μm, decreased the resistance to crack propagation by ~15%. These results support the loss in propagation toughness of glycated bone observed experimentally, and confirm the detrimental role of glycation in crack propagation.
NEG impairs bone material properties in diabetic mice
Fracture mechanics data showed that propagation toughness of the non-diabetic group (4.72 ± 0.79 MPa.m0.5) was higher than the diabetic group (3.38 ± 0.58 MPa.m0.5, p=0.012). Reference point indentation measurements revealed the indentation depth increase (IDI) in non-diabetic group (6.84 ± 1.07μm) was greater than in diabetic group (9.04 ± 1.89μm, p=0.033). AGE measurements showed a near significant difference between the non-diabetic and diabetic groups (p=0.072).
Discussion
Aging and diseases like diabetes increase bone fragility in the elderly [14, 15, 16, 31]. Traditionally, dual energy x-ray absorptiometry (DXA) based bone mineral density (BMD) measurements have been the used to predict bone fracture risk [32, 33]. However, BMD alone is not a reliable predictor, as fracture risk increases with age, irrespective of BMD [34]. Bone resists fracture by dissipating energy at various scales of its hierarchy through microdamage formation, from dilatational bands at the nanoscale [35] to microcracking that spans multiple microns in dimension [36]. Microdamage formation, in turn, depends largely on the quality of the organic matrix, including collagen and non-collagenous proteins.
Deformation of collagen, formation of microdamage at the lamellar level and uncracked ligaments at osteonal levels are established mechanisms of energy dissipation and toughening of bone. In this study, using in vitro tests, we show that NEG has a causal relationship (i.e. directly alters) with each one of these. We investigated in detail how non-enzymatic glycation affects the structural and mechanical properties of collagen matrix, and fundamentally alters creep and microcrack based toughening mechanisms in bone.
In this study, we used atomic force spectroscopy to study the change in fibrillar diameter and energy dissipation characteristics of glycated and non-glycated collagen. Other studies, albeit on tendon collagen, have reported a change in diameter of fibrils on glycation [37]. Due to in vitro glycation, we observed a reduction in tip displacement by 4 times in the glycated specimens, and total energy dissipation by an order of magnitude. Although tip-sample interactions account for part of the energy dissipated, such interactions are not likely to be significantly different between the glycated and non-glycated specimens, and limited to lower displacements [38] than seen in our studies. Thus we conclude that the impaired dissipation of energy is likely due to AGE accumulation caused due to glycation of collagen.
Lack of effective load transfer in bone has been implicated in age related deterioration in bone quality [14], however the mechanisms of such reduction have not been elucidated. Previous work by the authors has implicated NEG to impair post-yield mechanical behavior. NEG is associated with a decline in the post-yield properties bone including a reduction in post-yield energy in glycated bone by 50% [23]. Results from creep testing of mineralized and demineralized specimen, in this study, suggest for the first time that creep, a time based mechanical property, is greatly influenced by glycation. These experiments were conducted on paired specimens from same donor to establish causality between alteration of organic matrix by NEG and creep based energy dissipation in bone. As reported in the manuscript, this involved a large number of specimens and allowed us to statically conclude the effect of AGEs on creep based energy dissipation mechanisms in bone. We did not repeat these tests across ages as AGE accumulation causes loss of energy dissipation (as measured by fracture toughness) across all ages.
The ability of the organic matrix to creep and sustain deformation dramatically was reduced upon glycation. We found no difference in the maximum stress values, indicating that glycation influences bone fracture through strain-based mechanisms. Glycated organic matrix creeps 3 times slower than control. Glycated mineralized bone accumulates more residual stress than control specimen and fails at ~42% lower strain. The failure happens at strain levels within the upper bounds of physiological loading (~3000 μstrain) and may explain the incidence of fracture, due to falls, in the elderly. Thus, the inability of the NEG cross-linked collagen matrix to accommodate deformation results in decreased residual strains and increased residual stress. Furthermore from this study, we surmise that by creeping slower, glycated bone is unable to dissipate energy as quickly as non-glycated bone. Due to impaired stress relaxation, glycated bone matrix potentially accumulates areas of high stress concentration that can lead to microcrack coalescence and increased microcrack length, both of which were observed in the study. These changes manifest in altered cracking behavior and a consequent reduction in propagation toughness of glycated bone (Fig. 2d).
The creep testing and fracture mechanics results substantiate the role of glycation in post-yield fracture behavior of bone as previously suggested [23, 24, 27]. Furthermore, they suggest that creep is a dominant mechanism in the dissipation of energy in bone matrix [35], and is altered due to glycation. The in vitro mechanical data, show that AGE accumulation within bone matrix impacts crack growth and propagation over crack initiation. NEG therefore regulates toughening mechanisms and bone fracture by altering creep deformation of the organic matrix.
It is noteworthy that we found no difference in initiation toughness between the control and glycated specimens. Burstein et al. (1975) [39] have shown that pre-yield behavior is affected by bone mineral and post-yield by the organic matrix. Since glycation only modifies the organic matrix, and not the mineral component, a difference in initiation toughness is not expected. Here, we also report that in vitro NEG causes accumulation of AGEs in bone, at levels similar to observed in vivo, and that such accumulation of AGEs only alters the propagation and not initiation toughness across a range of age in a paired study design. Next we measured levels of AGEs measured in cadaveric bone and found that, consistent with out findings from in vitro model, AGE accumulation only correlate to propagation and not initiation toughness across a range of ages.
Propagation toughness, the ability of a matrix to resist crack propagation and avoid catastrophic failure, is thus predominantly governed by organic matrix, which includes NEG modifications of collagen. Our results shows that non-linear creep behavior of bone, associated with submicroscopic cracking in the collagen fibril [40, 41, 42], is absent during the creep of glycated bone (Supplementary Results). Taken together these results further supports our claim that glycation directly affects fracture, through creep. Hence we conclude that collagen, like other non-collagenous proteins [35], regulates crack propagation and that the extent of glycation, as measured by AGE content, is likely to predict bone fracture properties.
In order to assess the predictive capacity of AGEs in humans, we performed correlative tests between AGEs and mechanical data obtained experimentally (Fig. 4). We found that AGE content showed a significant negative correlation with the creep rate (p = 0.003; r2 = 0.36) (Fig. 4a) and maximum strain reached at each stress level of mineralized bone (p = 0.004; r2 = 0.59) at 100 MPa (Fig. 4b). AGEs strongly correlated, negatively, with propagation toughness (r2 = 0.64) indicating that an increase in NEG-mediated cross-linking of collagen results in a greater fracture risk (Fig. 4c). Finally, we also found that propagation toughness, exhibited a strong negative correlation with donor age (Fig. 4d). Thus using the correlative data above, AGE assays can be used to better predict the bone quality and fracture risk in aging humans. More importantly, since BMD measurements are based exclusively on bone mineral, AGE assays can improve upon fracture prediction. The importance of high surface to volume ratio in microdamage is evident from diffuse damage, a superior toughening mechanism in bone that dissipates energy through the formation of many submicron cracks in a contained volume [43, 44]. Linear microcracks (LM) predominate in glycated bone. Thus, with aging, bone tends to accumulate more LM than diffuse damage. These linear microcracks can coalesce causing a deterioration of mechanical properties [15, 44, 45]. However, unlike the formation of LM, that increase the propensity of bone failure, microcracking, a fundamental energy dissipating mechanism in bone, allows for the dissipation of greater fracture energy through stress shielding ahead of the crack tip, and the formation of numerous cracks in addition to the main propagating crack. Whilst glycation affects the process zone dilation and consequently energy dissipation in microcracking, crack bridging mechanisms, as seen in uncracked ligaments, and distinct from stress shielding and process zone dilation, are also altered in glycation. Our cohesive modeling studies indicate that, unlike glycated bone, the formation of smaller, numerous cracks enhance bone toughness by 20% in control specimen with smaller propagating cracks and uncracked ligaments overlap zones. Furthermore, limited microcracking in glycated samples indicated existence of smaller and fewer creep zones and crack growth retardation.
Figure 4.
Correlations showing relationships between (a) creep rate, (b) maximum strain and (c) propagation toughness with AGEs, for 6 pairs of specimens. (d) Furthermore, propagation toughness (solid line) shows a stronger correlation with age than initiation toughness (broken line) suggesting that the use of propagation toughness with other measures like DXA can predict fractures better (n=15 donors).
A model explaining the effect of glycation on cracking was developed based on the experimental and modeling data (Fig. 5). According to this model, the initial application of loading on bone results in the formation of a microcrack zone around the notch, known as a frontal process zone [46]. As loading continues a microcrack originates from the notch and accelerates through the frontal process zone, accompanied by creep ahead of crack tip. Acceleration of the main crack results in a formation of microcracks behind the main crack tip, in what is known as the process zone wake, generating more creep zones, thereby shielding the main crack tip by redistributing the stresses in the crack tip region [23]. The redistribution of stresses consequently decelerates the main crack under continued loading. The creation of a large surface area, due to and the accompanying smaller micron scale microcracks, in the wake of the main crack allows the dissipation of more energy. This pattern of crack propagation and microcracking is repeated as the main crack continues to propagate through the bone [23]. In addition to the mechanism of microcracking, formation of uncracked ligament zones, of varying overlap lengths also contribute significantly to energy dissipation (Fig 3d). It is noteworthy that the initiation toughness only accounts for microcracks formed during the formation of a frontal process zone, whereas propagation approach includes microcracks formed during crack propagation and microcracking in the wake zone. Thus, in contrast to initiation, the propagation approach is able to quantify the full contributions of microcracks formed during fracture. Furthermore, it becomes clear that the contribution of creep during microcrack formation comes to play only during crack propagation.
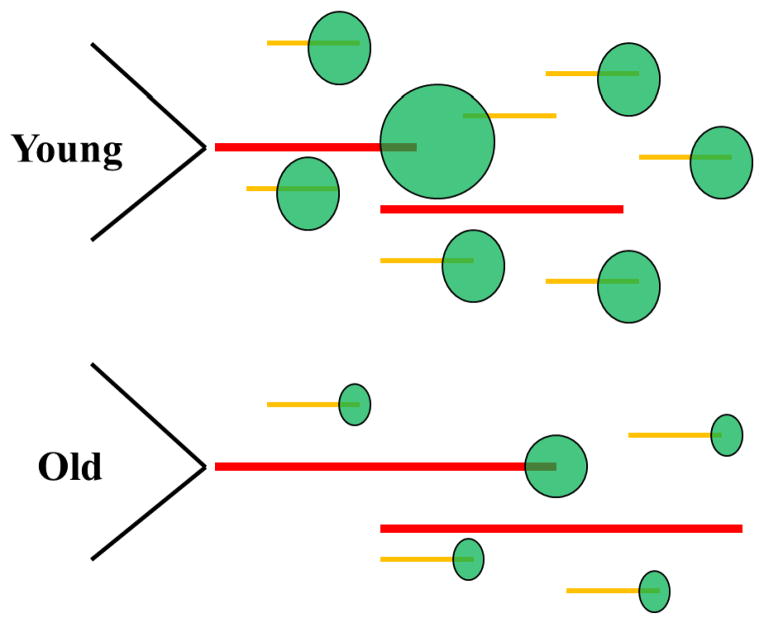
Figure 5.
Mechanistic model of microcrack toughening in glycated bone. Young bone (top) exhibits a larger number of short microcracks and uncracked ligaments, as compared to old/glycated bone (bottom). This increases creep based deformation (green circles) accommodated by the matrix in younger bone, thereby enhancing ductility and bone toughness.
It has previously been postulated that AGEs accumulate in bone matrix of aging and diabetic patients [16–21]. In the small animal study, we found a marked reduction in toughness and indentation measures (IDI) of the diabetic mice. The diabetic mice exhibited increased AGEs levels. It is noteworthy that NEG mediated 35% reduction in the crack propagation resistance of bone corresponds closely with the 37.2% reduction seen in vivo due to three decades of aging and 28.4% reduction in diabetic mice. The goal of this study was not to compare, validate or invalidate use of RPI as a tool to ascertain fracture toughness. We used fracture toughness testing and reference point indentation to evaluate the mechanical properties at the macro and micron scale respectively. Our results suggest that age- and diabetes- related changes in fracture resistance of bone in vivo are influenced mainly by the NEG-mediated modification of bone quality.
In conclusion, aging and diseases like diabetes result in accumulation of AGEs through non-enzymatic glycation, which is strongly correlated to a loss of bone toughness. In this study, we established a causal relationship between NEG and bone fracture. Furthermore, we demonstrated AGE measurement as a viable method that can be incorporated with existing fracture prediction modalities like DEXA, to allow for a more robust prediction of bone fragility.
Supplementary Material
Acknowledgments
This study is supported by NIH grant AG20618. The authors would like to thank the Center of Biotechnology and Interdisciplinary Studies for use of core facilities. We would also like to thank Ms. Dimple Goradia for her assistance with the AGE assays on collagen fibrils.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Zylberberg L. New data on bone matrix and its proteins. Comptes Rendus Palevol. 2004;3:591–604. [Google Scholar]
- 2.Hernandez CJ, Keaveny TM. A biomechanical perspective on bone quality. Bone. 2006;39:1173–1181. doi: 10.1016/j.bone.2006.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Burr DB. The Contribution of the Organic Matrix to Bone’s Material Properties. Bone. 2002;31(1):8–11. doi: 10.1016/s8756-3282(02)00815-3. [DOI] [PubMed] [Google Scholar]
- 4.Wang X, Bank RA, TeKoppele JM, Agrawal CM. The role of collagen in determining bone mechanical properties. Journal of Orthopaedic Research. 2001;19:1021–1016. doi: 10.1016/S0736-0266(01)00047-X. [DOI] [PubMed] [Google Scholar]
- 5.Knott L, Bailey AJ. Collagen cross-links in mineralizing tissues: A review of their chemistry, function, and clinical relevance. Bone. 1998;22(3):181–187. doi: 10.1016/s8756-3282(97)00279-2. [DOI] [PubMed] [Google Scholar]
- 6.Viguet-Carrin S, Garnero P, Delmas PD. The role of collagen in bone strength. Osteoporos Int. 2006;17:319–336. doi: 10.1007/s00198-005-2035-9. [DOI] [PubMed] [Google Scholar]
- 7.Eyre DR, Dickson IR, Vanness K. Collagen cross-linking in human-bone and articular-cartilage—age-related-changes in the content of mature hydroxypyridinium residues. Biochem J. 1988;252:495–500. doi: 10.1042/bj2520495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Saito M, Marumo K, Fujii K, Ishioka N. Single-column high-performance liquid chromatographic-fluorescence detection of immature, mature, and senescent crosslinks of collagen. Anal Biochem. 1997;253:26–32. doi: 10.1006/abio.1997.2350. [DOI] [PubMed] [Google Scholar]
- 9.Shen ZL, Dodge ML, Kahn H, Ballarini H, Eppel SJ. Stress-Strain Experiments on Individual Collagen Fibrils. Biophysical Journal. 2008;95(8):3956–3963. doi: 10.1529/biophysj.107.124602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gutsmann T, Fantner GE, Kindt JH, Venturoni M, Danielsen S, Hansma PK. Force Spectroscopy of Collagen Fibers to Investigate Their Mechanical Properties and Structural Organization. Biophysical Journal. 2004;86(5):3186–3193. doi: 10.1016/S0006-3495(04)74366-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Graham JS, Vomund AN, Phillips CL, Grandbois M. Structural changes in human type I collagen fibrils investigated by force spectroscopy. Experimental Cell Research. 2004;299:335–342. doi: 10.1016/j.yexcr.2004.05.022. [DOI] [PubMed] [Google Scholar]
- 12.Buehler MJ. Molecular nanomechanics of nascent bone: Fibrillar toughening by mineralization. Nanotechnology. 2007;18:295102. [Google Scholar]
- 13.Gupta HS, Wagermaier W, Zickler GA, Raz-Ben Aroush D, Funari SS, Roschger P, Wagner HD, Fratzl P. Cooperative deformation of mineral and collagen in bone at the nanoscale. Proc Natl Acad Sci USA. 2006;103:17741–17746. doi: 10.1073/pnas.0604237103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zimmermann EA, Schaible E, Bale H, Barth HD, Tang SY, Reichert P, Busse B, Alliston T, Ager JW, 3rd, Ritchie RO. Age-related changes in the plasticity and toughness of human cortical bone at multiple length scales. Proc Natl Acad Sci U S A. 2011;108:14416–21. doi: 10.1073/pnas.1107966108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Vashishth D. The role of the collagen matrix in skeletal fragility. Current Osteoporosis Reports. 2007;5(2):62–66. doi: 10.1007/s11914-007-0004-2. [DOI] [PubMed] [Google Scholar]
- 16.Saito M, Marumo K. Bone quality in diabetes. Front Endocrinol (Lausanne) 2013;4:72. doi: 10.3389/fendo.2013.00072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Vashishth D. Advanced glycation end-products and bone fractures. IBMS BoneKEy. 2009;6:268–278. doi: 10.1138/20090390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Sell DR, Monnier VM. Structure elucidation of a senescence cross-link from human extracellular matrix. Implication of pentoses in the aging process. J Biol Chem. 1989;264:21597–21602. [PubMed] [Google Scholar]
- 19.Odetti P, Rossi S, Monacelli F, Poggi A, Cirnigliaro M, Federici M, Federici A. Advanced glycation end-products and bone loss during aging. Ann N Y Acad Sci. 2005;1043:710–717. doi: 10.1196/annals.1333.082. [DOI] [PubMed] [Google Scholar]
- 20.Vashishth D, Gibson GJ, Khoury JI, Schaffler MB, Kimura J, Fyhrie DP. Influence of nonenzymatic glycation on biomechanical properties of cortical bone. Bone. 2001;28:195–201. doi: 10.1016/s8756-3282(00)00434-8. [DOI] [PubMed] [Google Scholar]
- 21.Wang X, Shen X, Li X, Agrawal CM. Age-related changes in the collagen network and toughness of bone. Bone. 2002;31:1–7. doi: 10.1016/s8756-3282(01)00697-4. [DOI] [PubMed] [Google Scholar]
- 22.Garnero P, Borel O, Gineyts E, Duboeuf F, Solberg H, Bouxsein ML, Christiansen C, Delmas PD. Extracellular post-translational modifications of collagen are major determinants of biomechanical properties of fetal bovine cortical bone. 2006;38(3):300–9. doi: 10.1016/j.bone.2005.09.014. [DOI] [PubMed] [Google Scholar]
- 23.Vashishth D, Koontz J, Qiu SJ, Lundin-Cannon D, Yeni YN, Schaffler MB, Fyhrie DP. In vivo diffuse damage in human vertebral trabecular bone. Bone. 2000;26(2):147–52. doi: 10.1016/s8756-3282(99)00253-7. [DOI] [PubMed] [Google Scholar]
- 24.Karim L, Vashishth D. Heterogeneous glycation of cancellous bone and its association with bone quality and fragility. PLoS One. 2012;7(4) doi: 10.1371/journal.pone.0035047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Chen AC, Temple MM, Ng DM, Verzijl N, DeGroot J, TeKoppele JM, Sah RL. Induction of advanced glycation end products and alterations of the tensile properties of articular cartilage. Arthritis Rheum. 2002;46(12):3212–7. doi: 10.1002/art.10627. [DOI] [PubMed] [Google Scholar]
- 26.Reddy GK. Cross-Linking in Collagen by Nonenzymatic Glycation Increases the Matrix Stiffness in Rabbit Achilles Tendon. Experimental Diabetes Research. 2004;5:143–153. doi: 10.1080/15438600490277860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Vestergaard P. Discrepancies in bone mineral density and fracture risk in patients with type 1 and type 2 diabetes--a meta-analysis. Osteoporos Int. 2007;18(4):427–44. doi: 10.1007/s00198-006-0253-4. [DOI] [PubMed] [Google Scholar]
- 28.Bozec L, Horton M. Topography and Mechanical Properties of Single Molecules of Type I Collagen Using Atomic Force Microscopy. Biophysical Journal. 2005;88(6):4223–4231. doi: 10.1529/biophysj.104.055228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Gutsmann T, Fantner GE, Kindt JH, Venturoni M, Danielsen S, Hansma PK. Force Spectroscopy of Collagen Fibers to Investigate Their Mechanical Properties and Structural Organization. Biophysical Journal. 2004;86(5):3186–3193. doi: 10.1016/S0006-3495(04)74366-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Diez-Perez A, Güerri R, Nogues X, Cáceres E, Peña MJ, Mellibovsky L, Hansma PK. Microindentation for in vivo measurement of bone tissue mechanical properties in humans. Journal of Bone and Mineral Research. 2010;25(8):1877–85. doi: 10.1002/jbmr.73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Catanese J, Bank RA, Tekoppele JM, Keaveny TM. Increased cross-linking by non-enzymatic glycation reduces the ductility of bone and bone collagen. Proceedings of the ASME 1999 Bioengineering Conference. 1999;42:267–268. [Google Scholar]
- 32.Lorente Ramos RM, Azpeitia Armán J, Arévalo Galeano N, Muñoz Hernández A, García Gómez JM, Gredilla Molinero J. Dual energy X-ray absorptimetry: fundamentals, methodology, and clinical applications. Radiologia. 2012;54(5):410–23. doi: 10.1016/j.rx.2011.09.023. [DOI] [PubMed] [Google Scholar]
- 33.Rothman MS, Miller PD, Lewiecki EM, Bilezikian JP. Bone Density Testing: Science, the Media, and Patient Care. Curr Osteoporos Rep. 2014 Mar 23; doi: 10.1007/s11914-014-0203-6. Epub ahead of print. [DOI] [PubMed] [Google Scholar]
- 34.Hui SL, Slemenda CW, Johnston CC., Jr Age and bone mass as predictors of fracture in a prospective study. J Clin Invest. 1988;81(6):1804–9. doi: 10.1172/JCI113523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Poundarik AP, Diab T, Sroga GE, Ural A, Boskey AL, Gundberg CM, Vashishth D. Dilatational Band Formation in Bone. Proceedings of the National Academy of Sciences. 2012;109(47):19178–83. doi: 10.1073/pnas.1201513109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Nalla RK, Kruzic JJ, Ritchie RO. On the origin of the toughness of mineralized tissue: microcracking or crack bridging? Bone. 2004;34(5):790–798. doi: 10.1016/j.bone.2004.02.001. [DOI] [PubMed] [Google Scholar]
- 37.Bai P, Phua K, Hardt T, Cernadas M, Brodsky B. Glycation alters collagen fibril organization. Connect Tissue Res. 1992;28(1–2):1–12. doi: 10.3109/03008209209014224. [DOI] [PubMed] [Google Scholar]
- 38.Gotsmann B, Anczykowski B, Seidel C, Fuchs H. Determination of tip–sample interaction forces from measured dynamic force spectroscopy curves. Applied surface science. 1999;140(3):314–319. [Google Scholar]
- 39.Burstein AH, Zika JM, Heiple KG, Klein L. Contribution of collagen and mineral to the elastic-plastic properties of bone. J Bone Jt Surg. 1975;57-A:956–961. [PubMed] [Google Scholar]
- 40.Fantner GE, Hassenkam T, Kindt JH, Weaver JC, Birkedal H, Pechenik L, Cutroni JA, Cidade GA, Stucky GD, Morse DE, Hansma PK. Sacrificial bonds and hidden length dissipate energy as mineralized fibrils separate during bone fracture. Nat Mater. 2005;4:612–616. doi: 10.1038/nmat1428. [DOI] [PubMed] [Google Scholar]
- 41.Zioupos P, Currey JD. The extent of microcracking and morphology of microcracks in damaged bone. Journal of Material Science. 1994;29:978–986. [Google Scholar]
- 42.Vashishth D, Tanner KE, Bonfield W. Contribution, development and morphology of microcracking in cortical bone during crack propagation. Journal of Biomechanics. 2000;33:1169–1174. doi: 10.1016/s0021-9290(00)00010-5. [DOI] [PubMed] [Google Scholar]
- 43.Diab T, Vashishth D. Morphology, localization and accumulation of in vivo microdamage in human cortical bone. Bone. 2007;40:612–8. doi: 10.1016/j.bone.2006.09.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Schaffler MB, Choi K, Milgrom C. Aging and matrix microdamage accumulation in human compact bone. Bone. 1995;17:521–525. doi: 10.1016/8756-3282(95)00370-3. [DOI] [PubMed] [Google Scholar]
- 45.Tang SY, Vashishth D. Non-enzymatic glycation alters microdamage formation in human cancellous bone. Bone. 2010;46:148–154. doi: 10.1016/j.bone.2009.09.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Vashishth D, Behiri JC, Bonfield W. Crack growth resistance in cortical bone: Concept of microcrack toughening. Journal of Biomechanics. 1997;30(8):763–769. doi: 10.1016/s0021-9290(97)00029-8. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.