Abstract
Cytokinins are plant hormones that play an integral role in multiple aspects of plant growth and development. The biological functions of cytokinins in mammalian systems are, however, largely uncharacterized. The naturally occurring cytokinin zeatin riboside has recently been demonstrated to activate the mammalian adenosine A2A receptor, which is broadly expressed by various cell types including immune system cells, with the activation of the A2AR playing a role in the regulation of cells involved in both innate and adaptive immunity. We show for the first time that zeatin riboside modulates mammalian immune system activity via an A2AR-dependent mechanism. Specifically, zeatin riboside treatment induces the production of cyclic adenosine monophosphate (cAMP) by T lymphocytes and inhibits the production by CD3+CD4+ T cells of interferon (IFN)-γ, IL-2, tumor-necrosis factor (TNF)-α, IL-4 and IL-13, and the production by CD3+CD8+ T cells of IFN-γ, IL-2 and TNF-α. Additionally, the upregulation of CD25, CD69 and CD40L by activated T lymphocytes is modulated by zeatin riboside. Zeatin riboside treatment also potently inhibits thioglycollate-induced peritoneal leukocytosis. The immunomodulatory activities of zeatin riboside are blocked by co-treatment with the selective A2AR antagonist ZM241385. These data suggest that zeatin riboside possesses therapeutic potential as a mammalian immunomodulatory agent.
Keywords: adenosine A2A receptors, T lymphocytes, zeatin riboside
Introduction
Cytokinins are a class of phytohormones that are involved in multiple aspects of plant growth and development.1 Naturally occurring cytokinins are predominantly adenine derivatives, and almost all cytokinins exist in plants as both a free base and corresponding nucleosides and nucleotides. Although cytokinin-binding proteins have been identified in mammalian sera, the biological functions of cytokinins and underlying mechanisms of action in mammalian systems remain largely uncharacterized.2,3,4 Interestingly, the cytokinin zeatin riboside has been shown to activate adenosine A2A receptor signaling in a mammalian neuronal cell line.5 Although the expression of the Gs-coupled A2AR varies widely by mammalian cell type, it is known to be broadly expressed by immune system cells, with the activation of the A2AR playing a role in terminating inflammation via the regulation of cells involved in innate and adaptive immunity.6 Given the adenosine-based structure of zeatin riboside, its activity as an A2AR agonist is not entirely surprising; however, it does endow the compound with significant therapeutic potential. We show for the first time that zeatin riboside modulates mammalian T lymphocyte activity in an A2AR-dependent manner via the inhibition of pro-inflammatory cytokine production and activation marker expression by CD4+ and CD8+ T lymphocytes.
Materials and methods
Mice
Eight- to tweleve-week-old female C57BL/6J mice were purchased from the Jackson Laboratory (Bar Harbor, ME, USA). All animal studies were approved by the Lebanon Valley College Animal Care and Use Committee.
T lymphocyte isolation and activation
Mice were killed and the spleens were removed. Splenocytes were passed through a 40 µM nylon cell strainer (BD Biosciences, Franklin Lakes, NJ, USA) and collected in phosphate-buffered saline. Red blood cells were removed with lysing buffer (Sigma-Aldrich, St. Louis, MO, USA), and CD3+CD4+ T lymphocytes were isolated via negative selection with mouse CD4 column kits and CD3+CD8+ T lymphocytes were isolated via negative selection with mouse CD8 column kits (R&D Systems, Minneapolis, MN, USA). Purified cells were resuspended at a concentration of 100 000 cells per 100 µl in RPMI 1640 medium supplemented with 10% heat-inactivated fetal bovine serum and 1% antibiotic-antimycotic (Invitrogen Life Technologies, Grand Island, NY, USA). T cells were activated by incubation on 2–10 µg/ml immobilized anti-CD3 monoclonal antibody (mAb) (BD Biosciences) for 24 h (or 6 h for the measurement of CD69 expression) at 37 °C in 5% CO2. Cells were cocultured with 31.25–1000 µM zeatin riboside, 1 µM CGS21680, or vehicle control and with 10 µM ZM241385 or vehicle control.
Thioglycollate-induced peritonitis
Two milliliters of a sterile 4% thioglycollate solution (in phosphate-buffered saline) or sterile saline was injected into the intraperitoneal cavities of C56BL/6 mice. Animals received either a 1 µg/kg i.p. bolus of CGS21680, a 1 mg/kg i.p. bolus of zeatin riboside or an equal volume i.p. bolus of vehicle immediately after thioglycollate injection, 1.5 h after injection and 3 h after injection. Mice were euthanized 5 h after initial thioglycollate injection and intraperitoneal cells were harvested via intraperitoneal wash with 6 ml cold phosphate-buffered saline.
Measurement ofcytokine production, cell surface markers and cell viability
Interferon (IFN)-γ, IL-2, tumor-necrosis factor (TNF)-α, IL-4 and IL-13 concentrations in supernatants of T-cell cultures were measured by ELISA according to the manufacturer's protocol (eBioscience, San Diego, CA, USA). Cell surface marker expression was measured via flow cytometry with fluorochrome-labeled anti-mouse CD3 (clone eBio500A2), anti-mouse CD4 (clone GK1.5), anti-mouse CD8 (clone eBioH35-17.2), anti-mouse CD25 (clone PC61.5), anti-mouse CD69 (clone H1.2F3) and/or anti-mouse CD40L (clone MR1) (eBioscience). All mAbs were utilized at a concentration of 0.5 µg/mL. Cell viability was measured via flow cytometry by labeling with Alexa Fluor 488 annexin V and propidium iodide. For all flow cytometry experiments, the fluorescence intensity was measured with a dual laser benchtop flow cytometer (FACSCalibur; Becton Dickinson, Franklin Lakes, NJ, USA) with a minimum of 20 000 events being collected. Analysis was performed with FlowJo software (Tree Star, Inc., Ashland, OR, USA).
Measurement of cyclic adenosine monophosphate (cAMP) production
Purified CD3+ T cells were resuspended at a concentration of 500 000 cells per 160 µl in RPMI 1640 medium supplemented with 10% heat-inactivated fetal bovine serum and 1% antibiotic-antimycotic (Invitrogen Life Technologies). Cells were treated with 1000 µM zeatin riboside, 1 µM CGS21680 or vehicle control for 30 min, after which total cellular cAMP levels were measured by enzyme immunoassay according to the manufacturer's protocol (GE Healthcare, Pittsburgh, PA, USA).
Statistical analysis
Prism software (GraphPad, San Diego, CA, USA) was used for all statistical analyses. One-way analysis of variance (ANOVA) with post-hoc Dunnett's multiple comparison or unpaired t-test (for single comparisons) was used.
Results
The anti-inflammatory activity of zeatin riboside is mediated by adenosine A2A receptor activation
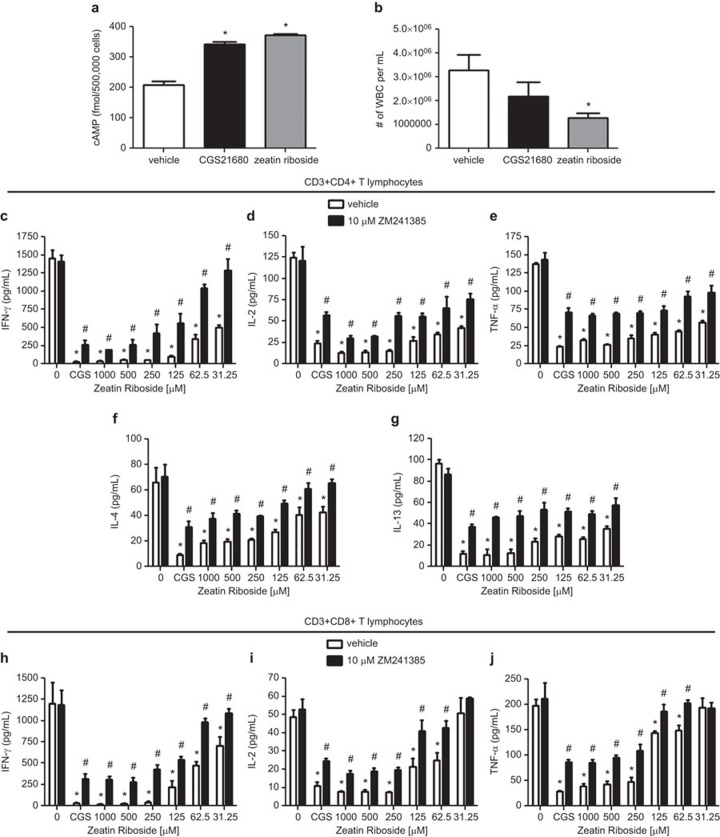
Treatment with 1000 µM of zeatin riboside induced the production of cAMP by purified CD3+ T lymphocytes to levels significantly above vehicle treated controls; cAMP production elicited by zeatin riboside exposure was similar to that induced by treatment of purified T cells with 1 µM of the well characterized, selective A2AR agonist, CGS21680 (Figure 1a). The intraperitoneal injection of sterile thioglycollate induces non-infectious peritonitis, with peritoneal exudates composed of neutrophils, lymphocytes and macrophages. Peritoneal leukocytosis elicited in C57BL/6 mice by thioglycollate challenge was significantly inhibited by the treatment with a 1 mg/kg i.p. bolus of zeatin riboside administered immediately after thioglycollate injection, 1.5 h after injection and 3 h after injection, with an approximate 60% decrease in white blood cell number in the peritoneal exudates of zeatin riboside treated mice as compared to vehicle-treated controls (Figure 1b). Treatment with a 1 µg/kg i.p. bolus of CGS21680 according to the same administration schedule as zeatin riboside treatment also reduced the number of white blood cells in the peritoneal cavities of thioglycollate challenged mice (with an approximate 33% reduction in white blood cell number in peritoneal exudates as compared to vehicle-treated mice), but the inhibition did not reach statistical significance (Figure 1b).
Figure 1.
The anti-inflammatory activity of zeatin riboside is mediated by adenosine A2A receptor activation. (a) Purified CD3+ T lymphocytes (500 000 per well) were treated with 1 µM CGS21680, 1000 µM zeatin riboside or vehicle for 30 min. Total cellular cAMP was measured by enzyme immunoassay. *P<0.05 vs. vehicle-treated control as assessed by one-way ANOVA followed by Dunnett's multiple comparison test. (b) C57BL/6 mice were subjected to thioglycollate challenge via intraperitoneal injection. Animals received a 1 µg/kg i.p. bolus of CGS21680, a 1 mg/kg i.p. bolus of zeatin riboside or an equal volume i.p. bolus of vehicle immediately after thioglycollate injection, 1.5 h after injection and 3 h after injection. Mice were euthanized 5 h after thioglycollate injection and intraperitoneal cells were harvested. *P<0.05 vs. vehicle-treated control as assessed by one-way ANOVA followed by Dunnett's multiple comparison test. (c–j) Purified CD3+CD4+ or CD3+CD8+ T cells (200 000 per well) were incubated on immobilized anti-CD3 mAb in the presence of 1 µM CGS21680 (CGS) or varying concentrations of zeatin riboside (or vehicle control) with 10 µM ZM241385 (or vehicle control). Supernatants were collected after 24 h and IFN-γ (c and h), IL-2 (d and i), TNF-α (e and j), IL-4 (f) and IL-13 (g) concentrations were measured by ELISA. Data are shown as the mean±s.e.m. from three independent experiments performed in quadruplicate. *P<0.05 vs. double vehicle-treated control (treated with neither zeatin riboside nor ZM241385) as assessed by one-way ANOVA followed by Dunnett's multiple comparison test. #P<0.05 vs. corresponding single vehicle samples (treated with equivalent concentrations of zeatin riboside, but no ZM241385) as assessed by unpaired t-test. ANOVA, analysis of variance; cAMP, cyclic adenosine monophosphate; IFN, interferon; mAb, monoclonal antibody; TNF, tumor-necrosis factor.
To specifically investigate the effects of zeatin riboside on T lymphocyte activity, T cells (>91% pure CD3+CD4+ or CD3+CD8+ T-cell populations) were incubated on anti-CD3 mAb for 24 h, resulting in the production of pro-inflammatory TH1 and TH2 cytokines. The activation of purified CD4+ T cells and CD8+ T cells in the presence of zeatin riboside resulted in dose-dependent inhibitions of T-cell receptor-mediated IFN-γ (Figure 1c and h), IL-2 (Figure 1d and i), TNF-α (Figure 1e and j), IL-4 (Figure 1f) and IL-13 (Figure 1g) production. Zeatin riboside inhibited IFN-γ production by CD4+ and CD8+ T cells with EC50 values of 20.4±10.29 and 49.88±24.2 µM, respectively; IL-2 production by CD4+ and CD8+ T cells with EC50 values of 81.72±32.44 and 38.84±28.59 µM, respectively; TNF-α production by CD4+ and CD8+ T cells with EC50 values of 24.84±4.57 and 98.78±19.19 µM, respectively; IL-4 production by CD4+ T cells with an EC50 value of 98.27±49.65 µM; IL-13 production by CD4+ T cells with an EC50 value of 226.1±98.58 µM (EC50 values were sometimes obtained from extrapolation of data curves). Zeatin riboside doses of 5 µM or less were found to have no significant inhibitory effects on CD4+ or CD8+ T-cell cytokine production. In all cases, the maximum inhibitory activity of zeatin riboside to block pro-inflammatory cytokine production by purified T cells was comparable to the inhibitory activity of 1 µM of the selective A2AR agonist, CGS21680; comparable doses of CGS21680 have been previously shown to potently inhibit multiple manifestations of T-cell activation.7,8,9,10 The zeatin riboside (and CGS21680)-mediated inhibition of proinflamamtory cytokine production by both CD4+ and CD8+ T lymphocytes was potently blocked by cotreatment with 10 µM of the selective A2AR antagonist, ZM241385. Similar doses of ZM241385 have previously been established to inhibit A2AR-mediated effects in T lymphocytes.9,10,11,12 These data suggest that zeatin riboside modulates T-cell production of TH1 and TH2 cytokines via an adenosine A2AR-specific mechanism.
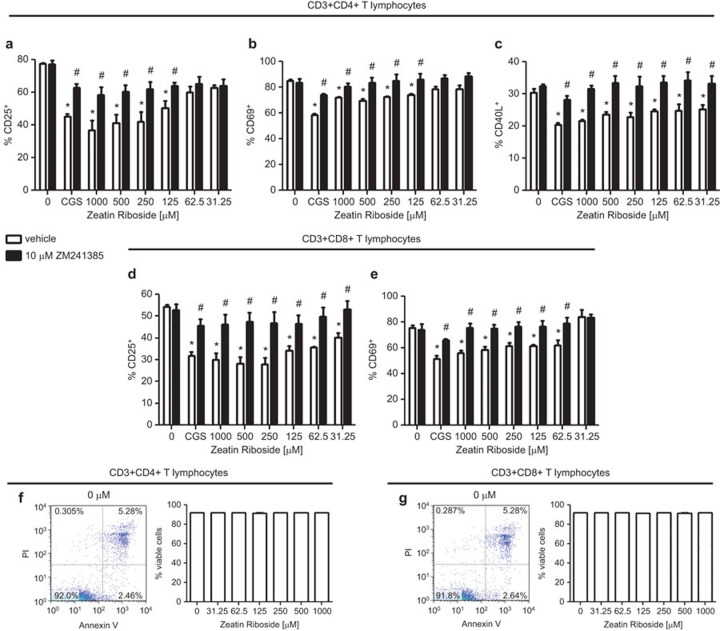
Zeatin riboside modulates the upregulation of activation maker expression by T cells
The incubation of purified murine T lymphocytes on immobilized anti-CD3 mAb triggered the upregulation of cell surface activation marker expression. Exposure to zeatin riboside reduced the expression of CD25, CD69 and CD40L by activated T cells in a dose-dependent manner. The cell surface expression by CD4+ T cells of CD25 was reduced by zeatin riboside by as much as 52.67%±7.70%, the expression of CD69 was reduced by as much as 19.0%5±2.57% and the expression of CD40L was reduced by as much as 29.1%±2.4% (Figure 2a–c). Furthermore, the cell surface expression by CD8+ T cells of CD25 was reduced by exposure to zeatin riboside by as much as 48.58±6.46 and the expression of CD69 was reduced by up to 21.54%±1.76% (Figure 2d and e). The inhibitory effects of zeatin riboside and CGS21680 on cell surface activation marker expression were potently blocked by cotreatment with 10 µM ZM241385, again suggesting that zeatin riboside modulates the pro-inflammatory activity of T lymphocytes via an A2AR-dependent mechanism. The maximum inhibitory activity of zeatin riboside to block activation marker expression by purified T cells was comparable to the inhibitory activity of 1 µM of the selective A2AR agonist, CGS21680. Notably, treatment with 31.25–1000 µM zeatin riboside had no significant effect on CD4+ or CD8+ T-cell viability, indicating that the zeatin riboside-generated, A2AR-mediated inhibition of TH1 and TH2 cytokine production and activation marker expression by T cells in this in vitro model represents immunomodulatory activity that is not simply a result of cytotoxicity (Figure 2f and g). These data are significant in that it establishes that zeatin riboside has a high degree of biocompatibility with murine lymphocytes.
Figure 2.
Zeatin riboside inhibits activation marker expression by CD4+ and CD8+ T cells; T-cell viability is unaffected by zeatin riboside exposure. Purified CD3+CD4+ or CD3+CD8+ T cells (200 000 per well) were incubated on immobilized anti-CD3 mAb for 6 (b and e) or 24 (a, c and d) h in the presence of 1 µM CGS21680 (CGS) or varying concentrations of zeatin riboside (or vehicle control) and 10 µM ZM241385 (or vehicle control). Cell surface expression of CD25 (a and d), CD69 (b and e) and CD40L (c) was assessed by FACS. CD3+ T cells were gated on for analysis. Data are shown as the mean±s.e.m. from three independent experiments performed in quadruplicate. *P<0.05 vs. double vehicle-treated control (treated with neither zeatin riboside nor ZM241385) as assessed by one-way ANOVA followed by Dunnett's multiple comparison test. #P<0.05 vs. corresponding single vehicle samples (treated with equivalent concentrations of zeatin riboside, but no ZM241385) as assessed by unpaired t-test. Viability of cells treated with 0, 31.25, 62.5, 125, 250, 500 or 1000 µM zeatin riboside was assessed via staining with Alexa Fluor 488 annexin V and propidium iodide (f and g); viable cells are defined as annexin V-negative/PI-negative cell populations. Data are shown as the mean±s.e.m. from three independent experiments; one representative dot plot for each cell subset is included to illustrate characteristic fluorescence intensity levels. ANOVA, analysis of variance; mAb, monoclonal antibody; PI, propidium iodide.
Discussion
Selective A2AR agonists have garnered increasing attention in recent years due to their potent anti-inflammatory activities. Not only is the A2AR broadly expressed by immune system cells, but its expression is upregulated during periods of inflammation, and A2AR activation modulates the activity of virtually all inflammatory cells including T lymphocytes, macrophages, monocytes, polymorphonuclear leukocytes and platelets.6,9,13 Selective A2AR agonists have been shown to inhibit multiple manifestations of inflammatory cell activation including cytokine production, chemotaxis, respiratory burst and adhesion molecule expression.14,15,16,17 Furthermore, A2AR activation limits the severity and progression of multiple inflammatory pathologies, including the T lymphocyte-driven ischemia–reperfusion injury, graft-versus-host disease and colitis.18,19,20 For these reasons, the development and/or discovery of selective A2AR agonists is of great interest. We show for the first time that the plant-derived hormone zeatin riboside modulates mammalian T lymphocyte activity via the activation of the A2AR. Zeatin riboside treatment induces the production of cAMP by purified CD3+ T lymphocytes, potently inhibits the production of both TH1 and TH2 cytokines by CD4+ and CD8+ T cells and limits the expression of CD25, CD69 and CD40L. Additionally, zeatin riboside treatment limits thioglycollate-induced peritoneal leukocytosis. The A2AR-mediated immunomodulatory activities of zeatin riboside observed in these in vitro and in vivo models are significant because they suggest that zeatin riboside may have immunosuppressive effects that directly or indirectly modulate both T cell-mediated and humoral immune responses. It will be interesting to investigate the in vivo immunomodulatory activity of zeatin riboside, to examine its pharmacological and toxicological profiles and to determine if zeatin riboside proves to be more efficacious that other A2AR agonists in the treatment of acute or chronic inflammatory disorders.
The authors declare that there are no conflicts of interest.
References
- 1Mok DW, Mok MC. Cytokinin metabolism and action. Annu Rev Plant Physiol Plant Mol Biol 2001; 52: 89–118. [DOI] [PubMed] [Google Scholar]
- 2Honma Y, Ishii Y. Differentiation of human myeloid leukemia cells by plant redifferentiation-inducing hormones. Leuk Lymphoma 2002; 43: 1729–1735. [DOI] [PubMed] [Google Scholar]
- 3Rattan SI, Clark BF. Kinetin delays the onset of ageing characteristics in human fibroblasts. Biochem Biophys Res Commun 1994; 201: 665–672. [DOI] [PubMed] [Google Scholar]
- 4Verbeke P, Siboska GE, Clark BF, Rattan SI. Kinetin inhibits protein oxidation and glycoxidation in vitro. Biochem Biophys Res Commun 2000; 276: 1265–1270. [DOI] [PubMed] [Google Scholar]
- 5Lee YC, Yang YC, Huang CL, Kuo TY, Lin JH, Yang DM et al. When cytokinin, a plant hormone, meets the adenosine A2A receptor: a novel neuroprotectant and lead for treating neurodegenerative disorders? PLoS ONE 2012; 7: e38865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6Lappas CM, Sullivan GW, Linden J. Adenosine A2A agonists in development for the treatment of inflammation. Expert Opin Investig Drugs 2005; 14: 797–806. [DOI] [PubMed] [Google Scholar]
- 7Chehata VJ, Domeier PP, Weilnau JN, Lappas CM. Adenosine A2A receptor activation limits chronic granulomatous disease-induced hyperinflammation. Cell Immunol 2011; 267: 39–49. [DOI] [PubMed] [Google Scholar]
- 8Erdmann AA, Gao ZG, Jung U, Foley J, Borenstein T, Jacobson KA et al. Activation of Th1 and Tc1 cell adenosine A2A receptors directly inhibits IL-2 secretion in vitro and IL-2-driven expansion in vivo. Blood 2005; 105: 4707–4714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9Lappas CM, Rieger JM, Linden J. A2A adenosine receptor induction inhibits IFN-gamma production in murine CD4+ T cells. J Immunol 2005; 174: 1073–1080. [DOI] [PubMed] [Google Scholar]
- 10Raskovalova T, Lokshin A, Huang X, Su Y, Mandic M, Zarou HM. Inhibition of cytokine production and cytotoxic activity of human antimelanoma specific CD8+ and CD4+ T lymphocytes by adenosine-protein kinase A type I signaling. Cancer Res 2007; 67: 5949–5956. [DOI] [PubMed] [Google Scholar]
- 11Bruzzese L, Fromonot J, By Y, Durand-Gorde JM, Condo J, Kipson N et al. NF-kappaB enhances hypoxia-driven T-cell immunosuppression via upregulation of adenosine A receptors. Cell Signal 2014; 26: 1060–1067. [DOI] [PubMed] [Google Scholar]
- 12Himer L, Csoka B, Selmeczy Z, Koscso B, Pocza T, Pacher P et al. Adenosine A2A receptor activation protects CD4+ T lymphocytes against activation-induced cell death. FASEB J 2010; 24: 2631–2640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13Murphree LJ, Sullivan GW, Marshall MA, Linden J. Lipopolysaccharide rapidly modifies adenosine receptor transcripts in murine and human macrophages: role of NF-kappaB in A2A adenosine receptor induction. Biochem J 2005; 391: 575–580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14Cronstein BN. Adenosine, an endogenous anti-inflammatory agent. J Appl Physiol 1994; 76: 5–13. [DOI] [PubMed] [Google Scholar]
- 15Ohta A, Sitkovsky M. Role of G-protein-coupled adenosine receptors in downregulation of inflammation and protection from tissue damage. Nature 2001; 414: 916–920. [DOI] [PubMed] [Google Scholar]
- 16Sullivan GW, Rieger JM, Scheld WM, Macdonald TL, Linden J. Cyclic AMP-dependent inhibition of human neutrophil oxidative activity by substituted 2-propynylcyclohexyl adenosine A2A receptor agonists. Br J Pharmacol 2001; 132: 1017–1026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17Sullivan GW, Lee DD, Ross WG, DiVietro JA, Lappas CM, Lawrence MB et al. Activation of A2A adenosine receptors inhibits expression of alpha 4/beta 1 integrin (very late antigen-4) on stimulated human neutrophils. J Leukoc Biol 2004; 75: 127–134. [DOI] [PubMed] [Google Scholar]
- 18Lappas CM, Day YJ, Marshall MA, Engelhard VH, Linden J. Adenosine A2A receptor activation reduces hepatic ischemia reperfusion injury by inhibiting CD1d-dependent NKT cell activation. J Exp Med 2006; 203: 2639–2648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19Lappas CM, Liu PC, Linden J, Kang EM, Malech HL. Adenosine A2A receptor activation limits graft-versus-host disease after allogenic hematopoietic stem cell transplantation. J Leukoc Biol 2010; 87: 345–354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20Naganuma M, Wiznerowicz EB, Lappas CM, Linden J, Worthington MT, Ernst PB. Cutting edge: critical role for A2A adenosine receptors in the T cell-mediated regulation of colitis. J Immunol 2006; 177: 2765–2769. [DOI] [PubMed] [Google Scholar]