Abstract
Withdrawal is an essential component of classical addiction theory; it is a vital manifestation of dependence and motivates relapse. However, the traditional conceptualization of withdrawal as a cohesive collection of symptoms that emerge during drug deprivation and decline with either the passage of time or reinstatement of drug use, may be inadequate to explain scientific findings or fit with modern theories of addiction. This article expands the current understanding of tobacco withdrawal by examining: (1) withdrawal variability; (2) underlying causes of withdrawal variability, including biological and person factors, environmental influences, and the influence of highly routinized behavioral patterns; (3) new withdrawal symptoms that allow for enhanced characterization of the withdrawal experience; and (4) withdrawal-related cognitive processes. These topics provide guidance regarding the optimal assessment of withdrawal and illustrate the potential impact modern withdrawal conceptualization and assessment could have on identifying treatment targets.
Introduction
This article is based, in part, on the 2014 Russell–Jarvik Early Investigator talk given at the SRNT Annual Meeting in Seattle. The goal of this article is to explore the construct of withdrawal, identify how modern theories and research inform our understanding of the construct, and provide insights into how withdrawal should be assessed and treated. This article is not intended to be an exhaustive review of all extant withdrawal research, but rather a thought piece that examines a wide body of withdrawal research. For instance, this review focuses, to some degree, on two types of withdrawal symptoms (craving and negative affect) because these have the strongest relations with smoking motivation (eg, cessation outcomes) and because much of the extant research focuses on these two symptoms.
Withdrawal: Classical Addiction Theory
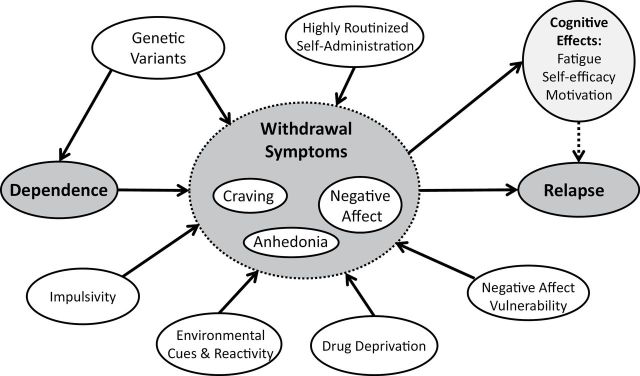
Classical addiction theory posits that: (1) the development of dependence produces withdrawal symptoms that appear following discontinuation of drug use and (2) those withdrawal symptoms motivate drug use reinstatement1–4 (Figure 1). In fact, withdrawal is a major criterion used to determine whether an agent is addictive.5 Scientists have identified self-reported nicotine withdrawal symptoms that meet this definition, including craving, negative affect, sleep disturbance, increased hunger and appetite and impaired concentration,6–9 all of which emerge among dependent smokers during decreased tobacco intake.10–13 However, despite use of valid self-report measures of withdrawal,14,15 data often do not conform to the classical definition of withdrawal as a set of symptoms that emerges following discontinuation of drug use, diminishes over time, motivates drug use, and is ameliorated by subsequent drug use. In other words, many studies have not found the expected relations among dependence, withdrawal, and cessation success.16–19 For instance, classical theory and some modern research suggest that withdrawal peaks within approximately 2 weeks and in some cases returns to baseline levels within that time frame.12,15,20,21 However, other studies find that withdrawal symptoms last more than 2 weeks22,23 or that dependence is not highly correlated with subsequent withdrawal during a cessation attempt24 or that withdrawal severity does not predict cessation success.25 While these discrepancies may be due to methodological issues (eg, studying motivated quitters vs. general population of smokers; prospective vs. retrospective data), these data suggest that there may be a larger problem with the current understanding and assessment of withdrawal.
Figure 1.
Classical addiction model.
It may be that the anomalous findings regarding withdrawal’s timecourse and its relations with dependence and relapse result from an insufficient understanding of withdrawal. The goal of this article is to discuss four areas intended to broaden understanding of withdrawal: (1) withdrawal variability, including symptom heterogeneity (ie, how symptoms can vary across dimensions such as timing, severity, type of symptom, and interrelations among symptoms) and individual variability (ie, how symptoms can vary across individuals) and how both types of variability are related to outcome; (2) underlying causes of withdrawal variability—this includes biological and person factors, environmental influences, and the influence of highly routinized behavioral patterns; (3) novel withdrawal symptoms that allow for an enhanced characterization of the withdrawal experience; and (4) withdrawal-related cognitive processes. Finally, this article will discuss how these areas inform the assessment of withdrawal.
Variability in Withdrawal Manifestation and Relations With Outcome
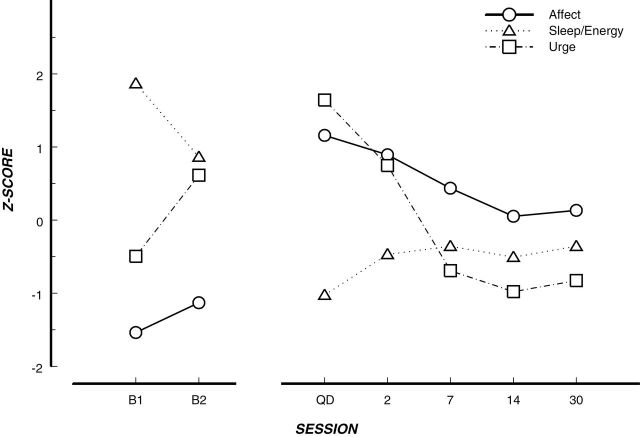
Withdrawal has long been conceptualized as a syndrome, a co-occurring set of symptoms, traditionally assessed via standard self-report withdrawal measures that tap the various symptoms. These ratings are averaged across symptoms to compute a single indicator of withdrawal.8,9 However, the withdrawal instruments themselves and subsequent research into individual withdrawal symptoms emphasize the multidimensional nature of withdrawal21,26 (although see8,9,27). Further, research has shown symptom heterogeneity in trajectory and severity over time, consistent with the variability of withdrawal symptoms for other drugs.28,29 For instance, one study found that negative affect decreased after quitting, but not as precipitously as craving (Figure 2).30
Figure 2.
Variable withdrawal symptom trajectory.30 Note. Aggregate time course of the individual symptom measures used in this article. Data points represent mean ratings from the whole sample for each factor score at the corresponding time point. Profiles are depicted in Z-score space to eliminate differences in scaling. B1 = 8 days precessation; B2 = 4 days precessation; QD = quit day. Remaining axis labels represent the number of days postquit.
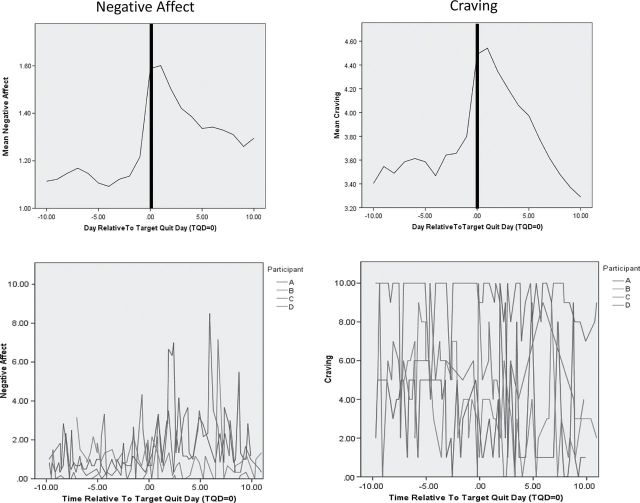
Research using ecological momentary assessment (EMA) data, which assess how participants are thinking, feeling, and behaving in real-time throughout the day, examines variability over time in withdrawal patterns even more closely. One EMA study found that although negative affect and craving may have similar average trajectories over the first 10 days postquit, they exhibit very distinct patterns over time (Figure 3)31: negative affect appears to be relatively low, peaking occasionally, especially postquit, whereas craving is considerably more variable, both pre-quit and postquit. EMA research has also illustrated individual variability in symptoms. For instance, one study found that craving and negative affect showed dramatically different profiles between individuals.32 Taken together, these findings illustrate the variability in withdrawal which could be due to both the heterogeneity of symptom time courses as well as individual variability among smokers attempting to quit.
Figure 3.
Average trajectory and individual variability in negative affect and craving.
Laboratory research has also illustrated variability in withdrawal, namely in the timing of the emergence of withdrawal symptoms. For instance, one study using both objective and self-report measures of withdrawal found that heart rate decreased between baseline and 60 minutes of abstinence, whereas reaction time slowed among abstaining participants after 30 minutes of abstinence.33 Anger emerged after only 60 minutes, while anxiety didn’t emerge until 120 minutes of deprivation and concentration difficulties after 180 minutes of deprivation.33 These findings illustrate another important source of variability—negative affect. It is clear from this research, as well as affective neuroscience findings, that negative affect as a construct varies considerably in the degree of negative valence, the level of arousal, and neurobiological underpinnings.34 Current research is working to unpack the variability of negative affect in the context of drug use.35–38
Individual variability in the timing of the onset and severity of different withdrawal symptoms has been shown to be related to dependence indices and cessation outcome.33,39–41 These findings suggest that variability in symptom onset, time course, and phenomenology, as well as individual differences in symptom manifestation, may partially explain why using average estimates of withdrawal do not show consistent relations with dependence and cessation. For instance, in one study craving and negative affect each independently predicted cessation outcome in a multivariate model, suggesting that they offer unique insight into cessation success and researchers may not be well-served by averaging all withdrawal symptoms into a single withdrawal construct.31 Further, modern analytic techniques have revealed that symptoms’ relations with relapse are not consistent over time. Specifically, time-varying effects models revealed that craving consistently predicts outcome during the first 2 weeks postquit, but that the relation between negative affect and relapse increases over the course of the first 2 weeks postquit.41 These findings suggest that variability in withdrawal—both symptom heterogeneity and individual variability—is important to account for when considering withdrawal in a more comprehensive manner.
Taken together, these findings suggest that different withdrawal symptoms have different time courses. Even withdrawal symptoms that appear to have similar average time courses or trajectories may still show important variability in real-time symptom ratings. Symptom heterogeneity and individual variability have been related to cessation success, illustrating the importance of understanding withdrawal symptoms (eg, urge, negative affect) as individual constructs with unique onset timing, peaks, and variability as well as unique relations with dependence and cessation success. Understanding this variability may enhance our conceptualization of the relapse process and inform treatment for smokers trying to quit. For instance, understanding the variability in a smoker’s withdrawal symptoms (eg, low levels of negative affect with intermittent spikes) and how these manifest over time (eg, a spike in negative affect produces larger and larger spikes in craving over time) could inform adaptive treatment strategies.
Underlying Causes of Withdrawal Variability
Understanding the underlying causes of withdrawal variability can provide insight into how discontinuing drug use can produce such heterogeneous symptoms that vary across people and across time and could aid in treatment development and assignment. According to classical addiction theory, the elicitation of withdrawal is driven by internal cues (ie, falling drug levels, although Wikler and others suggested environmental elicitation of withdrawal more than 40 years ago42). That is, the body senses low levels of drug and produces withdrawal symptoms that motivate drug use. Within this context, variability in symptom manifestation and the timing of symptom onset would be consistent with different underlying biology. However, research suggests that classical withdrawal, conceptualized from a strictly biological perspective as evidenced when drug is removed and resolved over time or when when drug is reinstated (either through nicotine replacement or resumption of smoking), may be insufficient to account for the breadth of findings regarding the withdrawal phenomenon.22,23 Therefore, it may be important to consider underlying causes of withdrawal that go beyond symptoms that emerge in response to loss of the addictive molecules (ie, pharmacologic or biological withdrawal) to include the emergence of symptoms due to environmental influences (ie, cue reactivity) or to the absence of the drug self-administration ritual (ie, behavioral withdrawal). One does not have to define withdrawal in this broad manner, but retaining a classical definition of withdrawal may necessitate changes in experimental approaches such as developing new assessments given that self-report of withdrawal symptoms in real-life paradigms will to be influenced by cues and other aspects of the drug self-administration ritual in addition to pharmacologic withdrawal. This article will explore three potential causes of withdrawal symptoms and variability: biological and person factors, environmental, and behavioral.
Biological Causes and Person-Factor Modulators of Withdrawal
Research on the neurobiology of withdrawal suggests one source of withdrawal variability is that symptoms emerge via activation of different neurobiological pathways. Specifically, recent neural imaging work suggests that craving and other withdrawal symptoms are instantiated through distinct neural paths.43 This is consistent with the work of Curtin et al.44 who posited that motivational prods such as negative affect have been shown to be evidenced through “bottom up” processes involving the activation of subcortical structures, consistent with the motivational power of withdrawal symptoms such as negative affect, while cognitive or “top down” processes related to cognitive control were linked to craving.
Research studies with animals and humans have further demonstrated biological underpinnings of withdrawal symptom heterogeneity and individual variability in withdrawal. For instance, research using genetic knockout animals has shown that different nicotinic receptors are implicated in distinct withdrawal symptoms (eg, anhedonia, somatic signs, place aversion; see45 for review). Similarly, human research has shown that genetic variants coding for nicotinic receptors and dopamine receptors are differentially related to withdrawal severity and reward sensitivity during deprivation.46–48 These findings suggest that genetic variability is linked to withdrawal symptom variability and individual variability. Further, given the genetic underpinnings of dependence,49–52 it may be that connections between dependence and withdrawal could be better elucidated by examining the shared and independent genetic influences on specific dependence and withdrawal phenotypes.
Animal research has also demonstrated that person factors such as low impulsivity and gender are related to specific withdrawal symptom manifestation and differences in stress-related withdrawal symptoms, respectively.53–55 Research using two national samples of adult smokers found that while mental illness was associated with more severe withdrawal, in general, anxiety-related withdrawal symptoms were especially prevalent among those with internalizing disorders.56 Further, research has shown that smokers with a history of panic attacks, generalized anxiety disorder, or social phobia report more severe nicotine withdrawal.57,58 These findings suggest that smokers with internalizing disorders, especially anxiety disorders, are more vulnerable to experiencing elevated negative affect following a cessation attempt. Human laboratory research has also shown that gender and temperament facets such as harm avoidance and novelty seeking are related to differential withdrawal symptom manifestation during 12 hours of abstinence.59,60 Taken together, this body of research suggests that individual differences in gender, impulsivity, and negative affect vulnerability can influence a smoker’s withdrawal experience. It should be noted that other person factors might also moderate smokers’ response to withdrawal (eg, distress tolerance, abstinence expectancies61–63).
In sum, different biological and person factors appear to underlie some of the symptom heterogeneity and individual variability in withdrawal experience. However, it is unclear exactly how these factors interact with dependence and cessation outcome. It may be that biological and person factors moderate the relations between withdrawal symptoms and dependence and cessation success. For instance, an anxiety disorder may produce differential dependence manifestations (eg, more negative reinforcement motivation). Moreover, variability in negative affect withdrawal symptoms may differentially influence cessation success among smokers with internalizing disorders (eg, high levels of anxiety during abstinence may present more of a relapse risk for smokers with internalizing disorders). Further research is needed to understand how these underlying factors that influence withdrawal are related to smoker’s ability to quit.
Environmental Withdrawal
Another potential causal factor underlying withdrawal manifestation is environmental cues, which can elicit withdrawal symptoms (eg, negative affect, craving) independent of deprivation.64–66 Such cravings may be relatively unaffected by smoking cessation pharmacotherapy (nicotine replacement therapy, varenicline, or bupropion), which do alleviate cravings related to drug deprivation.67–75 This is consistent with research that suggests different neurobiological pathways for cue-induced versus deprivation-induced craving.76 Importantly, smokers cannot always differentiate whether an urge to smoke stems from a cue-induced versus deprivation-induced craving. Therefore, traditional withdrawal assessments could be enhanced and show stronger relations with dependence or cessation outcomes if the self-report measures take into account variability due to the craving source (ie, if the assessment is trying to capture changes in symptoms due to deprivation but is also able to account for the impact of environmental cues74).
Environmental cues can also interact with deprivation. EMA research has examined the impact of the environment (eg, cues, temptation events, smokers in the environment) on withdrawal symptoms among smokers trying to quit. One such study revealed that both deprivation and temptation events increased negative affect and craving, but smokers reported the greatest negative affective and craving responses when a temptation event was combined with deprivation (ie, during the postquit period31,32). In other words, drug deprivation increases reactivity to environmental cues, and results in increased withdrawal symptomatology, whether it manifests as negative affect or as craving (cf.,77). Brain imaging studies have also shown a larger response to cues during deprivation than when smokers were not deprived.78 These findings suggest that deprivation may further enhance susceptibility to drug cues and temptation events.
Research has also demonstrated that affective and urge reactivity to cues among smokers trying to quit are related to relapse risk.67,68,79 In addition, reactivity to temptation events or smoking cues is related to dependence motives, specifically secondary dependence motives (eg, reinforcement, cognitive enhancement, and social motives).80 These findings suggest that a broader account of withdrawal that includes enhanced cue reactivity during deprivation as a withdrawal phenomenon may enhance understanding of the relations among the classical addiction triad: dependence, withdrawal, and relapse risk. However, it is important to note that there is variability in cue reactivity response. For example, consistent with research on withdrawal symptoms produced by drug deprivation, symptoms prompted by drug cues appear to have distinct neural underpinnings (ie, different brain regions were associated with craving vs. negative affect when smokers were in withdrawal and exposed to smoking cues81). In addition, recent research suggests that individual differences in gender and personality traits may also influence reactivity to smoking cues.82,83 Therefore, it is important to consider how biological and environmental constructs can interact to influence both symptom heterogeneity and individual variability in withdrawal.
Behavioral Withdrawal
In addition to examining biological, person-factor, and environmental underpinnings of withdrawal variability, it may also be important to understand that when a person quits smoking, she/he is withdrawing not only from nicotine per se (ie, pharmacologic withdrawal) but from smoking, with all its attendant behaviors, cognitions, and rituals. In fact, the American Psychiatric Association Diagnostic and Statistical Manual (APA-DSM) system and others in the field have argued for the term tobacco withdrawal rather than nicotine withdrawal, in light of these issues.6,84 Behavioral withdrawal produces similar symptoms as pharmacologic withdrawal (eg, craving, negative affect, loss of concentration).85 It emerges when a smoker is unable to deliberately engage in the drug use ritual in situations where such use is highly routinized, such as in the presence of cues or stressors, or as a means of coping with negative affect.85 In other words, when the self-administration ritual has become an integral part of a daily routine and when an interoceptive or exteroceptive cue then elicits the self-administration routine via Pavlovian associations or classical conditioning (eg, talking on the phone or getting into an argument leads the person to automatically light a cigarette), prevention of the dominant response to use drug leads to behavioral withdrawal symptoms. For instance, not being able to use drug during an interpersonal conflict (a situation during which a smoker routinely uses drug) may heighten attention to the unavailability of drug use, thereby worsening frustration, hopelessness, irritability, and urges to use drug to alleviate these feelings. Behavioral withdrawal results from the conflict between the situation and the inability or resistance to engaging in the dominant response (drug use), thereby enlisting cognitive resources necessary to cope with the situation by either using drug or engaging in alternative behavior.85 It should be noted that behavioral withdrawal is still somewhat speculative and does not stem from as solid an evidence base as the construct of pharmacologic withdrawal, but it is supported by substantial theory and empirical evidence.86
Behavioral withdrawal might explain why smokers report continued craving well beyond the typical 2–4 week biological withdrawal window. For instance, epidemiology research has illuminated the issue of prolonged craving or behavioral withdrawal even among smokers who quit more than 6 months ago,22,23 despite biological evidence that suggests that some nicotine receptors have adapted to abstinence within 6–12 weeks.87 It may be that withdrawal symptoms precipitated by depleted nicotine receptors resolve early in a quit attempt but that behavioral withdrawal can manifest over much longer periods of time as it is elicited in situations in which the self-administration ritual is highly routinized or entrenched, but the smoker does not engage in drug self-administration in that situation.
The influence of behavioral withdrawal, independent of pharmacologic withdrawal, is further evident from research showing that smoking de-nicotinized cigarettes can reduce withdrawal symptoms.86,88–96 In fact, research has shown that the self-administration ritual, independent of drug administration, will activate brain reward and incentive systems, thereby alleviating negative affect.97 These findings suggest a critical role for the drug self-administration ritual that needs to be assessed and studied further to understand withdrawal in a more comprehensive manner. Incorporating behavioral withdrawal into a broader conceptualization of withdrawal may help make sense of findings that don’t appear to fit with the classical addiction time frame and explain more withdrawal variability.
In sum, viewing the causal underpinnings of withdrawal more comprehensively (rather than being restricted to symptoms that emerge solely as a response to a lack of drug in the system), and including what happens to people and their interaction with their environment when they discontinue drug use, might allow researchers and clinicians to explain more symptom heterogeneity and individual variability. We know that withdrawal symptoms can be elicited via biological, environmental, and behavioral sources and that withdrawal symptoms are motivationally significant regardless of the underlying causal mechanism. However, the causal underpinnings result in differential response to treatment, which would suggest that the source of withdrawal has treatment implications. In other words, behavioral or environmental withdrawal may need to be targeted with psychosocial or behavioral interventions rather than pharmacotherapy (ie, withdrawal emerging from not using drug in a situation where drug use is entrenched would be better served by developing alternative coping behaviors or avoiding such situations rather than replacing the missing addictive substance). Further, it is unclear whether the source of withdrawal influences the severity of the response and whether this also has important variability. Therefore, it is vital to be able to determine how much variance in withdrawal is due to the different causal factors.
New Withdrawal Symptoms
In addition to expanding the conceptualization of withdrawal, advances in theory and research methodologies have allowed researchers to explore the possibility of other withdrawal symptoms. For instance, modern addiction theory suggests that dependence modulates the reward value of drug and nondrug stimuli such that nicotine is associated with increased responsiveness to rewards (ie, incentive sensitization theory70). If increased reward modulation of behavior is a component of dependence, then one would expect to see a reduction in reward responsiveness during abstinence. Indeed, animal research has shown that animals have a shift in reward threshold during withdrawal.98,99 Recent research has also used analogous reward responsiveness tasks with rats and humans and found that both species reported withdrawal-induced reduction in reward responding.100 This would suggest that anhedonia—the inability to experience pleasure—is a withdrawal symptom. This would be consistent with smokers’ reports that, “nothing is as good when I’m not smoking.” Indeed, a reduction in the rewarding value of life activities would certainly be an aversive state that would motivate a return to drug use.
Recent research using EMA data from smokers attempting to quit reported anhedonia had a trajectory similar to a “traditional” withdrawal symptom trajectory. Specifically, smokers’ inability to experience pleasure in daily activities, or anhedonia, increased slightly prior to a quit attempt, increased precipitously on the quit day and then returned back to baseline levels within 2 weeks.101 The postquit increase in anhedonia was correlated with dependence level and predicted cessation outcome, independent of craving, negative affect, and depressed mood.101 Blunted hedonic response and a self-reported history of anhedonia prior to cessation have also been shown to predict relapse;102,103 cf.10. These findings suggest that anhedonia may be a key withdrawal symptom that has not been included in traditional withdrawal measures or targeted for either behavioral or pharmacological treatment (cf., behavioral activation treatment for smokers104).
Including anhedonia as a withdrawal symptom may also provide insight into the high rates of smoking and difficulty quitting among smokers with mental illness. Smokers with schizophrenia and depression diagnoses, two diagnoses that include anhedonia as a major index of underlying pathology, smoke at higher rates than the rest of the US population105,106 and research suggests that this higher rate is partially related to the reward value of cigarettes.107 Understanding anhedonia’s role in maintaining smoking behavior and treating this withdrawal symptom effectively, especially among these high-risk populations, could have important clinical and public health effects.
Researchers have continued to identify other symptoms that may index nicotine withdrawal, including cold symptoms and mouth ulcers108 and constipation.109 Six experimental studies found that abstinence increased impatience and this increase had a time-limited effect consistent with a withdrawal symptom.110 Delay discounting and response inhibition have also been studied in experimental paradigms as potential withdrawal symptoms, but the evidence is unclear at this point.110 One internet survey examined the performance of 31 additional symptoms (not including anhedonia) but found that only mood swings worsened among those who abstained and sense of smell, sense of taste and sore throat improved with abstinence.111 While more research is needed on these potential withdrawal symptoms, it is clear that scientists in the field are thinking more broadly about the symptoms one would expect to see when one quits using an addictive substance.
Withdrawal’s Influence on Cognitive Processes
A broader perspective of withdrawal includes looking closely at reactions to withdrawal beyond cessation. For instance, enhanced understanding of how withdrawal can influence key cognitive processes linked to cessation, could provide a broader view of the full impact of withdrawal.
The novel cognitive reaction of cessation fatigue, or being tired of quitting, was put forth by Piasecki and colleagues in 2002 as an important construct in the cessation process—a reaction to withdrawal but not a withdrawal symptom per se. 112 One smoker trying to quit likened watching out for urges and temptation events to guard duty and noted that, “I can stand guard duty during a battle, but standing guard duty for the rest of my life is more than I can bear.” The construct of cessation fatigue is consistent with the strength model of self-regulation, wherein people are able to exert self-control over behavior, but, like a muscle, self-control eventually tires and gives out.113–116 In other words, the process of coping with withdrawal symptoms, regardless of their origin, exhausts self-regulatory resources and increases relapse risk. Further, cessation fatigue may also emerge in response to effort exerted to avoid or prevent withdrawal symptoms.
Results from time-varying effects models developed using EMA data from a cessation trial showed that cessation fatigue increases over time and smokers who reported greater craving or negative affect also reported more cessation fatigue.117 Interestingly, the nature of the relation between craving and negative affect and subsequent cessation fatigue varied over time (eg, elevated negative affect tended to affect fatigue considerably more early in the cessation process while the influence of craving on fatigue unfolded gradually over time). Cessation fatigue was negatively associated with cessation success. Interestingly, combination nicotine replacement therapy (NRT), which has been shown to reduce craving and produce higher abstinence rates relative to a single NRT, was also related to reduced fatigue.117,118 These data illustrate the connection between withdrawal, treatment, cessation outcome, and this related construct of cessation fatigue. However, more research is needed to replicate these findings and further explore the impact of cessation fatigue on cessation success and identify optimal treatments to ameliorate such fatigue.
Research has also examined the relations of withdrawal symptoms with more established cognitive processes such as self-efficacy and motivation. One study used EMA data to compare successful early quitters with relapsers and found a strong, consistent, negative association over time between confidence in the ability to abstain and urges to smoke among successful quitters but not among relapsers, even at the beginning of the quit attempt.119 This suggests that there may be variability in the ways in which withdrawal influences cognitive and motivational constructs and this variability may be related to relapse risk. It may be that to comprehensively understand the connection between withdrawal and cessation, researchers also need to understand how specific withdrawal symptoms influence cognitive and motivational constructs and whether these influences differ based on the underlying cause of the withdrawal symptoms (eg, do smokers experience greater fatigue when they are battling cue-reactive craving vs. deprivation-related craving).
Withdrawal Assessment
Overall, these findings illustrate the value of expanding our definition and evaluation of withdrawal. An expanded perspective may also be informative when considering withdrawal syndromes of other drugs (eg, alcohol, stimulants, and opioids). Withdrawal is not a monolithic construct that can easily be assessed via an average of self-report symptoms. Rather, the extant research reveals something that smokers have known for years—namely, that withdrawal experiences can vary widely, even within persons within days. This variability in phenomenology can result from individual differences in underlying neurophysiology and genetics, different environmental stimuli, or behavioral withdrawal. It is this variability, in part, that makes it difficult to develop a standard treatment that works for all smokers at all times.120
This research also reveals that scientists may not be using optimal measures of withdrawal, with respect to what is measured and how it is measured. With respect to what is measured, it could be that expanded withdrawal assessments that include reactivity to smoking cues, behavioral withdrawal, and reward functioning (ie, anhedonia), might provide additional insight into withdrawal and its consequences, including cognitive processes (eg, fatigue, motivation, and self-efficacy) and relapse as well as improving the ability to detect the relation between withdrawal and dependence.
How withdrawal is measured also merits further consideration. Historically, withdrawal assessment has been driven largely by observable symptoms (eg, decreased heart rate and reaction time, seizures, hypothermia, pupil size) and then by what people could report on (eg, cravings, negative affect). However, research has demonstrated that withdrawal symptoms such as craving and negative affect can be generated from different causes (eg, cues vs. deprivation), via different biological pathways, and respond differently to cessation pharmacotherapy. These findings speak to the need to develop assessments that can disentangle the source of the withdrawal symptoms—cravings stemming from nicotine deprivation versus craving induced by cue exposure; negative affect stemming from deprivation versus due to negative life events or difficulty regulating affect (as suggested by Addicott et al.76). For instance, modern neurocognitive processing research or information processing tasks in which the self-administration ritual is administered independent of drug administration would allow researchers to evaluate these processes in the laboratory context. Of course, such an assessment would not be practical for many research and clinical applications, but such an assessment might provide critical information that would allow researchers to refine or develop more targeted assessments. Further, if negative affect is a universal withdrawal symptom,121 and it exerts effects preconsciously, then researchers need to develop an appropriate information processing assessment instrument, given smokers’ inability to provide self-report on preconscious phenomena (eg, response time122). In sum, our assessment of withdrawal needs to move beyond what is observable or accessible via self-report to tap processes and effects one would expect to see based on theory and research.
The limitations of the current withdrawal assessments, both in content and approach, may also explain the difficulty in linking withdrawal to dependence and to cessation failure.24,25 For instance, some research has shown that withdrawal symptoms are related to relapse;123–125 however, some smokers relapse without reporting withdrawal symptoms.11,12,15,25,126 Further, many smokers experience significant withdrawal symptoms and yet manage to quit successfully. A more accurate assessment of withdrawal that goes beyond self-report would provide insight into the withdrawal phenomenon and thereby into the links between dependence and withdrawal, and between withdrawal and cessation. Optimal withdrawal assessments would be sensitive to: (1) different symptoms domains, (2) varying symptom trajectories over time, (3) underlying causes (eg, biology, person-factor influences, environmental effects, behavioral withdrawal), and (4) relations with key consequences (eg, fatigue) that may mediate clinical outcomes.
Summary
The classical theory of addiction posits that dependence results in withdrawal symptoms due to absence of drug in the system, and withdrawal symptoms, in turn, motivate a return to drug use (Figure 1). This article proposes that Figure 1 be expanded to account for discrepant findings regarding the relations among the addiction triad and incorporate new research and theoretical perspectives. In Figure 4, withdrawal symptoms have been expanded to include anhedonia as well as negative affect and craving—all of which may have variable timing, underlying neurobiology, and relations with outcomes. The different causal influences on withdrawal symptoms are illustrated in the unshaded circles and include constructs such as genetic variants, impulsivity, environmental cues and reactivity, highly-routinized self-administration rituals, and negative affect vulnerability as well as drug deprivation. These are included to illustrate how the causal underpinnings of withdrawal can influence variability in withdrawal expression, cessation success, and response to treatment. Finally, the effects of withdrawal on cognitive processes (ie, cessation fatigue, self-efficacy, and motivation; lightly shaded) are included to depict a broader impact of withdrawal and how such influences might subsequently influence cessation risk. By conceptualizing withdrawal more broadly, as what happens when a person tries to stop smoking, this more complex picture develops. The research reviewed here suggests the need for a shift in the conceptualization and assessment of withdrawal to allow researchers and clinicians to develop and tailor interventions to better treat this phenomenon that is so critical to successful cessation.
Figure 4.
Expanded withdrawal model.
Funding
MEP’s research has been funded by NIH grants 9P50CA143188-12, P50 DA019706, M01 RR03186, 1UL1RR025011, and the Wisconsin Partnership Program.
Declaration of Interests
None declared.
Acknowledgments
I would like to acknowledge the support of Jessica Cook in preparing this article, the support and effort of my colleagues at the University of Wisconsin Center for Tobacco Research and Intervention, my collaborators at the Pennsylvania State University Methodology Center, and a special thank you to Timothy Baker, a phenomenal mentor and colleague.
References
- 1. Edwards G, Gross MM. Alcohol dependence: provisional description of a clinical syndrome. BMJ. 1976;1(6017):1058–1061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Siegel S. Classical conditioning, drug tolerance, and drug dependence. In: Smart RG, Glaser FB, Israel Y, Kalant R, Popham E, Schmidt W, eds. Research Advances in Alcohol and Drug Problems. 7th ed. New York, NY: Plenum; 1983. [Google Scholar]
- 3. Solomon RL, Corbit JD. An opponent-process theory of motivation. I. Temporal dynamics of affect. Psychol Rev. 1974;81(2):119–145. 10.1037/h0036128. [DOI] [PubMed] [Google Scholar]
- 4. Wikler A. Opioid Dependence. New York, NY: Plenum; 1980. [Google Scholar]
- 5. U.S. Department of Health and Human Services. The Health Consequences of Smoking: Nicotine Addiction. Atlanta, GA: U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health; 1988. [Google Scholar]
- 6. American Psychiatric Association. Diagnostic and Statistical Manual. Vol V. Washington, DC: American Psychiatric Association; 2013. [Google Scholar]
- 7. Shiftman S. The tobacco withdrawal syndrome. In: Krasnegor NA, ed. Cigarette Smoking as a Dependence Process. Washington, DC: National Institute on Drug Abuse; 1979:158–184. [Google Scholar]
- 8. Welsch SK, Smith SS, Wetter DW, Jorenby DE, Fiore MC, Baker TB. Development and validation of the Wisconsin Smoking Withdrawal Scale. Exp Clin Psychopharmacol. 1999;7(4):354–361. 10.1037/1064-1297.7.4.354. [DOI] [PubMed] [Google Scholar]
- 9. Hughes JR, Hatsukami D. Signs and symptoms of tobacco withdrawal. Arch Gen Psychiatry. 1986;43(3):289–294. [DOI] [PubMed] [Google Scholar]
- 10. Dawkins L, Powell JH, Pickering A, Powell J, West R. Patterns of change in withdrawal symptoms, desire to smoke, reward motivation and response inhibition across 3 months of smoking abstinence. Addiction. 2009;104(5):850–858. 10.1111/j.1360-0443.2009.02522.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Hughes JR. Effects of abstinence from tobacco: etiology, animal models, epidemiology, and significance: a subjective review. Nicotine Tob Res. 2007;9(3):329–339. 10.1080/14622200701188927. [DOI] [PubMed] [Google Scholar]
- 12. Hughes JR. Effects of abstinence from tobacco: valid symptoms and time course. Nicotine Tob Res. 2007;9(3):315–327. 10.1080/14 622200701188919. [DOI] [PubMed] [Google Scholar]
- 13. Leventhal AM, Waters AJ, Moolchan ET, Heishman SJ, Pickworth WB. A quantitative analysis of subjective, cognitive, and physiological manifestations of the acute tobacco abstinence syndrome. Addict Behav. 2010;35(12):1120–1130. 10.1016/j.addbeh.2010.08.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. West R, Ussher M, Evans M, Rashid M. Assessing DSM-IV nicotine withdrawal symptoms: a comparison and evaluation of five different scales. Psychopharmacology (Berl). 2006;184(3–4):619–627. [DOI] [PubMed] [Google Scholar]
- 15. Hughes JR. Measurement of the effects of abstinence from tobacco: a qualitative review. Psychol Addict Behav. 2007;21(2):127–137. 10.1037/0893-164X.21.2.127. [DOI] [PubMed] [Google Scholar]
- 16. Payne TJ, Smith PO, McCracken LM, McSherry WC, Antony MM. Assessing nicotine dependence: a comparison of the Fagerstrom Tolerance Questionnaire (FTQ) with the Fagerstrom Test for Nicotine Dependence (FTND) in a clinical sample. Addict Behav. 1994;19(3):307–317. 10.1016/0306-4603(94)90032-9. [DOI] [PubMed] [Google Scholar]
- 17. Piper ME, McCarthy DE, Baker TB. Assessing tobacco dependence: a guide to measure evaluation and selection. Nicotine Tob Res. 2006;8(3):339–351. 10.1080/14622200600672765. [DOI] [PubMed] [Google Scholar]
- 18. Rios-Bedoya CF, Snedecor SM, Pomerleau CS, Pomerleau OF. Association of withdrawal features with nicotine dependence as measured by the Fagerstrom Test for Nicotine Dependence (FTND). Addict Behav. 2008;33(8):1086–1089. 10.1016/j.addbeh.2008.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Shiffman S, Waters A, Hickcox M. The nicotine dependence syndrome scale: a multidimensional measure of nicotine dependence. Nicotine Tob Res. 2004;6(2):327–348. 10.1080/1462220042000202481. [DOI] [PubMed] [Google Scholar]
- 20. Shiffman S, Patten C, Gwaltney C, et al. Natural history of nicotine withdrawal. Addiction. 2006;101(12):1822–1832. 10.1111/j.1360-0443.2006.01635.x. [DOI] [PubMed] [Google Scholar]
- 21. Shiffman S, West R, Gilbert D; SRNT Work Group on the Assessment of Craving Withdrawal in Clinical Trials. Recommendation for the assessment of tobacco craving and withdrawal in smoking cessation trials. Nicotine Tob Res. 2004;6(4):599–614. 10.1080/14622200410001734067. [DOI] [PubMed] [Google Scholar]
- 22. Herd N, Borland R. The natural history of quitting smoking: findings from the International Tobacco Control (ITC) Four Country Survey. Addiction. 2009;104(12):2075–2087. 10.1111/j.1360-0443.2009.02731.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Hughes JR. Craving among long-abstinent smokers: an Internet survey. Nicotine Tob Res. 2010;12(4):459–462. 10.1093/ntr/ntq009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Baker TB, Piper ME, Schlam TR, et al. Are tobacco dependence and withdrawal related amongst heavy smokers? Relevance to conceptualizations of dependence. J Abnorm Psychol. 2012;121(4):909–921. 10.1037/a0027889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Wray JM, Gass JC, Tiffany ST. A systematic review of the relationships between craving and smoking cessation. Nicotine Tob Res. 2013;15(7):1167–1182. 10.1093/ntr/nts268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Cappelleri JC, Bushmakin AG, Baker CL, Merikle E, Olufade AO, Gilbert DG. Revealing the multidimensional framework of the Minnesota nicotine withdrawal scale. Curr Med Res Opin. 2005;21(5):749–760. [DOI] [PubMed] [Google Scholar]
- 27. Toll BA, O’Malley SS, McKee SA, Salovey P, Krishnan-Sarin S. Confirmatory factor analysis of the Minnesota Nicotine Withdrawal Scale. Psychol Addict Behav. 2007;21(2):216–225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Dijkstra BA, Krabbe PF, De Jong CA, van der Staak CP. Prediction of withdrawal symptoms during opioid detoxification. J Opioid Manag. 2008;4(5):311–319. [DOI] [PubMed] [Google Scholar]
- 29. Hesse M, Thylstrup B. Time-course of the DSM-5 cannabis withdrawal symptoms in poly-substance abusers. BMC Psychiatry. 2013;13:258–269. 10.1186/1471-244X-13-258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Piasecki TM, Niaura R, Shadel WG, et al. Smoking withdrawal dynamics in unaided quitters. J Abnorm Psychol. 2000;109(1):74–86. 10.1037/0021-843X.109.1.74. [DOI] [PubMed] [Google Scholar]
- 31. Piper ME, Schlam TR, Cook JW, et al. Tobacco withdrawal components and their relations with cessation success. Psychopharmacology (Berl). 2011;216(4):569–578. 10.1007/s00213-011-2250-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. McCarthy DE, Piasecki TM, Fiore MC, Baker TB. Life before and after quitting smoking: an electronic diary study. J Abnorm Psychol. 2006;115(3):454–466. [DOI] [PubMed] [Google Scholar]
- 33. Hendricks PS, Ditre JW, Drobes DJ, Brandon TH. The early time course of smoking withdrawal effects. Psychopharmacology (Berl). 2006;187(3):385–396. [DOI] [PubMed] [Google Scholar]
- 34. Davidson RJ, Jackson DC, Kalin NH. Emotion, plasticity, context, and regulation: perspectives from affective neuroscience. Psychol Bull. 2000;126(6):890–909. [DOI] [PubMed] [Google Scholar]
- 35. Piper ME, Smith SS, Schlam TR, et al. Psychiatric disorders in smokers seeking treatment for tobacco dependence: relations with tobacco dependence and cessation. J Consult Clin Psychol. 2010;78(1):13–23. 10.1037/a0018065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Bradford DE, Magruder KP, Korhumel RA, Curtin JJ. Using the threat probability task to assess anxiety and fear during uncertain and certain threat. J Vis Exp. 2014(91):51905. 10.3791/51905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Bradford DE, Shapiro BL, Curtin JJ. How bad could it be? Alcohol dampens stress responses to threat of uncertain intensity. Psychol Sci. 2013;24(12):2541–2549. 10.3791/51905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Hefner KR, Moberg CA, Hachiya LY, Curtin JJ. Alcohol stress response dampening during imminent versus distal, uncertain threat. J Abnorm Psychol. 2013;122(3):756–769. 10.1037/a0033407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Rubinstein ML, Benowitz NL, Auerback GM, Moscicki AB. Rate of nicotine metabolism and withdrawal symptoms in adolescent light smokers. Pediatrics. 2008;122(3):e643–e647. 10.1542/peds.2007-3679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Kubota T, Nakajima-Taniguchi C, Fukuda T, et al. CYP2A6 polymorphisms are associated with nicotine dependence and influence withdrawal symptoms in smoking cessation. Pharmacogenomics J. 2006;6(2):115–119. [DOI] [PubMed] [Google Scholar]
- 41. Vasilenko SA, Piper ME, Lanza ST, Liu X, Yang J, Li R. Time-varying processes involved in smoking lapse in a randomized trial of smoking cessation therapies. Nicotine Tob Res. 2014;16(suppl 2):S135–143. 10.1093/ntr/ntt185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Wikler A. Dynamics of drug dependence. Implications of a conditioning theory for research and treatment. Arch Gen Psychiatry. 1973;28(5):611–616. [DOI] [PubMed] [Google Scholar]
- 43. Wang Z, Faith M, Patterson F, et al. Neural substrates of abstinence-induced cigarette cravings in chronic smokers. J Neurosci. 2007;27(51):14035–14040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Curtin JJ, McCarthy DE, Piper ME, Baker TB. Implicit and explicit drug motivational processes: a model of boundary conditions. In: Weirs RW, Stacy AW, eds. Handbook of Implicit Cognition and Addiction. Thousand Oaks, CA: Sage; 2006:233–250. [Google Scholar]
- 45. Stoker AK, Markou A. Unraveling the neurobiology of nicotine dependence using genetically engineered mice. Curr Opin Neurobiol. 2013;23(4):493–499. 10.1016/j.conb.2013.02.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Lazary J, Dome P, Csala I, et al. Massive withdrawal symptoms and affective vulnerability are associated with variants of the CHRNA4 gene in a subgroup of smokers. PLoS One. 2014;9(1):e87141. 10.1371/journal.pone.0087141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Jackson KJ, Sanjakdar SS, Chen X, Damaj MI. Nicotine reward and affective nicotine withdrawal signs are attenuated in calcium/calmodulin-dependent protein kinase IV knockout mice. PLoS One. 2012;7(11):e51154. 10.1371/journal.pone.0051154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Xu X, Clark US, David SP, et al. Effects of nicotine deprivation and replacement on BOLD-fMRI response to smoking cues as a function of DRD4 VNTR genotype. Nicotine Tob Res. 2014;16(7):939–947. 10.1093/ntr/ntu010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Weiss RB, Baker TB, Cannon DS, et al. A candidate gene approach identifies the CHRNA5-A3-B4 region as a risk factor for age-dependent nicotine addiction. PLoS Genet. 2008;4(7):e1000125. 10.1371/journal.pgen. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Berrettini W, Yuan X, Tozzi F, et al. Alpha-5/alpha-3 nicotinic receptor subunit alleles increase risk for heavy smoking. Mol Psychiatry. 2008;13(4):368–373. 10.1038/sj.mp.4002154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Saccone SF, Hinrichs AL, Saccone NL, et al. Cholinergic nicotinic receptor genes implicated in a nicotine dependence association study targeting 348 candidate genes with 3713 SNPs. Hum Mol Genet. 2007;16(1):36–49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Thorgeirsson TE, Geller F, Sulem P, et al. A variant associated with nicotine dependence, lung cancer and peripheral arterial disease. Nature. 2008;452(7187):638–642. 10.1038/nature06846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Kolokotroni KZ, Rodgers RJ, Harrison AA. Trait differences in response to chronic nicotine and nicotine withdrawal in rats. Psychopharmacology (Berl). 2014;231(3):567–580. 10.1007/s00213-013-3270-y. [DOI] [PubMed] [Google Scholar]
- 54. Kayir H, Semenova S, Markou A. Baseline impulsive choice predicts the effects of nicotine and nicotine withdrawal on impulsivity in rats. Prog Neuropsychopharmacol Biol Psychiatry. 2014;48:6–13. 10.1016/j.pnpbp.2013.09.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Torres OV, Gentil LG, Natividad LA, Carcoba LM, O’Dell LE. Behavioral, biochemical, and molecular indices of stress are enhanced in female versus male rats experiencing nicotine withdrawal. Front Psychiatry. 2013;4:38. 10.3389/fpsyt.2013.00038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Smith PH, Homish GG, Giovino GA, Kozlowski LT. Cigarette smoking and mental illness: a study of nicotine withdrawal. Am J Public Health. 2014;104(2):e127–e133. 10.2105/AJPH.2013.301502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Marshall EC, Johnson K, Bergman J, Gibson LE, Zvolensky MJ. Anxiety sensitivity and panic reactivity to bodily sensations: relation to quit-day (acute) nicotine withdrawal symptom severity among daily smokers making a self-guided quit attempt. Exp Clin Psychopharmacol. 2009;17(5):356–364. 10.1037/a0016883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Piper ME, Cook JW, Schlam TR, Jorenby DE, Baker TB. Anxiety diagnoses in smokers seeking cessation treatment: relations with tobacco dependence, withdrawal, outcome and response to treatment. Addiction. 2011;106(2):418–427. 10.1111/j.1360-0443.2010.03173.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Leventhal AM, Waters AJ, Boyd S, et al. Associations between Cloninger’s temperament dimensions and acute tobacco withdrawal. Addict Behav. 2007;32(12):2976–2989. 10.1016/j.addbeh.2007.06.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Leventhal AM, Waters AJ, Boyd S, Moolchan ET, Lerman C, Pickworth WB. Gender differences in acute tobacco withdrawal: effects on subjective, cognitive, and physiological measures. Exp Clin Psychopharmacol. 2007;15(1):21–36. 10.1037/1064-1297.15.1.21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Sirota AD, Rohsenow DJ, Mackinnon SV, et al. Intolerance for Smoking Abstinence Questionnaire: psychometric properties and relationship to tobacco dependence and abstinence. Addict Behav. 2010;35(7):686–693. 10.1016/j.addbeh.2010.02.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Hendricks PS, Leventhal AM. Abstinence-related expectancies predict smoking withdrawal effects: implications for possible causal mechanisms. Psychopharmacology (Berl). 2013;230(3):363–373. 10.1007/s00213-013-3169-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Brown RA, Lejuez CW, Strong DR, et al. A prospective examination of distress tolerance and early smoking lapse in adult self-quitters. Nicotine Tob Res. 2009;11(5):493–502. 10.1093/ntr/ntp041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Carter BL, Tiffany ST. Cue-reactivity and the future of addiction research. Addiction. 1999;94(3):349–351. [PubMed] [Google Scholar]
- 65. Rose JE. Nicotine and nonnicotine factors in cigarette addiction. Psychopharmacology (Berl). 2006;184(3–4):274–285. [DOI] [PubMed] [Google Scholar]
- 66. Henningfield JE, Goldberg SR. Control of behavior by intravenous nicotine injections in human subjects. Pharmacol Biochem Behav. 1983;19(6):1021–1026. [DOI] [PubMed] [Google Scholar]
- 67. Cofta-Woerpel L, McClure JB, Li Y, Urbauer D, Cinciripini PM, Wetter DW. Early cessation success or failure among women attempting to quit smoking: trajectories and volatility of urge and negative mood during the first postcessation week. J Abnorm Psychol. 2011;120(3):596–606. 10.1037/a0023755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Waters AJ, Shiffman S, Sayette MA, Paty JA, Gwaltney CJ, Balabanis MH. Cue-provoked craving and nicotine replacement therapy in smoking cessation. J Consult Clin Psychol. 2004;72(6):1136–1143. 10.1037/0022-006X.72.6.1136. [DOI] [PubMed] [Google Scholar]
- 69. Teneggi V, Tiffany ST, Squassante L, Milleri S, Ziviani L, Bye A. Smokers deprived of cigarettes for 72 h: effect of nicotine patches on craving and withdrawal. Psychopharmacology (Berl). 2002;164(2):177–187. 10.1007/s00213-002-1176-1. [DOI] [PubMed] [Google Scholar]
- 70. Robinson TE, Berridge KC. The neural basis of drug craving: an incentive-sensitization theory of addiction. Brain Res Rev. 1993;18(3):247–291. [DOI] [PubMed] [Google Scholar]
- 71. Hughes JR, Lesmes GR, Hatsukami DK, et al. Are higher doses of nicotine replacement more effective for smoking cessation? Nicotine Tob Res. 1999;1(2):169–174. [DOI] [PubMed] [Google Scholar]
- 72. Hurt RD, Sachs DP, Glover ED, et al. A comparison of sustained-release bupropion and placebo for smoking cessation. N Engl J Med. 1997;337(17):1195–1202. [DOI] [PubMed] [Google Scholar]
- 73. Jorenby DE, Leischow SJ, Nides MA, et al. A controlled trial of sustained-release bupropion, a nicotine patch, or both for smoking cessation. N Engl J Med. 1999;340(9):685–691. [DOI] [PubMed] [Google Scholar]
- 74. Ferguson SG, Shiffman S. The relevance and treatment of cue-induced cravings in tobacco dependence. J Subst Abuse Treat. 2009;36(3):235–243. 10.1016/j.jsat.2008.06.005. [DOI] [PubMed] [Google Scholar]
- 75. Ravva P, Gastonguay MR, Faessel HM, Lee TC, Niaura R. Pharmacokinetic-pharmacodynamic modeling of the effect of varenicline on nicotine craving in adult smokers. Nicotine Tob Res. 2015;17(1):106–113. 10.1093/ntr/ntu154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Addicott MA, Froeliger B, Kozink RV, et al. Nicotine and non-nicotine smoking factors differentially modulate craving, withdrawal and cerebral blood flow as measured with arterial spin labeling. Neuropsychopharmacology. 2014;39(12):2750–2759. 10.1038/npp.2014.108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77. Shiffman S, Dunbar M, Kirchner T, et al. Smoker reactivity to cues: effects on craving and on smoking behavior. J Abnorm Psychol. 2013;122(1):264–280. 10.1037/a0028339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78. Engelmann JM, Versace F, Robinson JD, et al. Neural substrates of smoking cue reactivity: a meta-analysis of fMRI studies. Neuroimage. 2012;60(1):252–262. 10.1016/j.neuroimage.2011.12.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79. Lam CY, Businelle MS, Aigner CJ, et al. Individual and combined effects of multiple high-risk triggers on postcessation smoking urge and lapse. Nicotine Tob Res. 2014;16(5):569–575. 10.1093/ntr/ntt190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80. Piper ME, Bolt DM, Kim SY, et al. Refining the tobacco dependence phenotype using the Wisconsin Inventory of Smoking Dependence Motives. J Abnorm Psychol. 2008;117(4):747–761. 10.1037/a0013298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81. McClernon FJ, Kozink RV, Rose JE. Individual differences in nicotine dependence, withdrawal symptoms, and sex predict transient fMRI-BOLD responses to smoking cues. Neuropsychopharmacology. 2008;33(9):2148–2157. 10.1038/sj.npp.1301618. [DOI] [PubMed] [Google Scholar]
- 82. Michalowski A, Erblich J. Reward dependence moderates smoking-cue- and stress-induced cigarette cravings. Addict Behav. 2014;39(12):1879–1883. 10.1016/j.addbeh.2014.07.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83. Doran N. Sex differences in smoking cue reactivity: craving, negative affect, and preference for immediate smoking. Am J Addict. 2014;23(3):211–217. 10.1111/j.1521-0391.2014.12094.x. [DOI] [PubMed] [Google Scholar]
- 84. Fagerström K. Determinants of tobacco use and renaming the FTND to the Fagerstrom Test for Cigarette Dependence. Nicotine Tob Res. 2012;14(1):75–78. 10.1093/ntr/ntr137. [DOI] [PubMed] [Google Scholar]
- 85. Baker TB, Japuntich SJ, Hogle JM, McCarthy DE, Curtin JJ. Pharmocologic and behavioral withdrawal from addictive drugs. Curr Dir Psychol Sci. 2006;15(5):232–236. 10.1111/j.1467-8721.2006.00442.x. [Google Scholar]
- 86. Perkins KA, Ciccocioppo M, Conklin CA, Milanak ME, Grottenthaler A, Sayette MA. Mood influences on acute smoking responses are independent of nicotine intake and dose expectancy. J Abnorm Psychol. 2008;117(1):79–93. 10.1037/0021-843X.117.1.79. [DOI] [PubMed] [Google Scholar]
- 87. Cosgrove KP, Batis J, Bois F, et al. beta2-Nicotinic acetylcholine receptor availability during acute and prolonged abstinence from tobacco smoking. Arch Gen Psychiatry. 2009;66(6):666–676. 10.1001/archgenpsychiatry.2009.41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88. Butschky MF, Bailey D, Henningfield JE, Pickworth WB. Smoking without nicotine delivery decreases withdrawal in 12-hour abstinent smokers. Pharmacol Biochem Behav. 1995;50(1):91–96. [DOI] [PubMed] [Google Scholar]
- 89. Gross J, Lee J, Stitzer ML. Nicotine-containing versus de-nicotinized cigarettes: effects on craving and withdrawal. Pharmacol Biochem Behav. 1997;57(1–2):159–165. [DOI] [PubMed] [Google Scholar]
- 90. Donny EC, Houtsmuller E, Stitzer ML. Smoking in the absence of nicotine: behavioral, subjective and physiological effects over 11 days. Addiction. 2007;102(2):324–334. 10.1111/j.1360-0443.2006.01670.x. [DOI] [PubMed] [Google Scholar]
- 91. Buchhalter AR, Acosta MC, Evans SE, Breland AB, Eissenberg T. Tobacco abstinence symptom suppression: the role played by the smoking-related stimuli that are delivered by denicotinized cigarettes. Addiction. 2005;100(4):550–559. 10.1111/j.1360-0443.2005.01030.x. [DOI] [PubMed] [Google Scholar]
- 92. Rose JE, Behm FM. Extinguishing the rewarding value of smoke cues: pharmacological and behavioral treatments. Nicotine Tob Res. 2004;6(3):523–532. 10.1080/14622200410001696501. [DOI] [PubMed] [Google Scholar]
- 93. Domino EF, Ni L, Domino JS, et al. Denicotinized versus average nicotine tobacco cigarette smoking differentially releases striatal dopamine. Nicotine Tob Res. 2013;15(1):11–21. 10.1093/ntr/nts029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94. Juliano LM, Brandon TH. Effects of nicotine dose, instructional set, and outcome expectancies on the subjective effects of smoking in the presence of a stressor. J Abnorm Psychol. 2002;111(1):88–97. [DOI] [PubMed] [Google Scholar]
- 95. Pickworth WB, Fant RV, Nelson RA, Rohrer MS, Henningfield JE. Pharmacodynamic effects of new de-nicotinized cigarettes. Nicotine Tob Res. 1999;1(4):357–364. [DOI] [PubMed] [Google Scholar]
- 96. Tidey JW, Rohsenow DJ, Kaplan GB, Swift RM, Ahnallen CG. Separate and combined effects of very low nicotine cigarettes and nicotine replacement in smokers with schizophrenia and controls. Nicotine Tob Res. 2013;15(1):121–129. 10.1093/ntr/nts098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97. Balfour DJ. The neurobiology of tobacco dependence: a preclinical perspective on the role of the dopamine projections to the nucleus accumbens [corrected]. Nicotine Tob Res. 2004;6(6):899–912. [DOI] [PubMed] [Google Scholar]
- 98. Kenny PJ, Markou A. Nicotine self-administration acutely activates brain reward systems and induces a long-lasting increase in reward sensitivity. Neuropsychopharmacology. 2006;31(6):1203–1211. [DOI] [PubMed] [Google Scholar]
- 99. Kenny PJ, Markou A. Conditioned nicotine withdrawal profoundly decreases the activity of brain reward systems. J Neurosci. 2005;25(26):6208–6212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100. Pergadia ML, Der-Avakian A, D’Souza MS, et al. Association between nicotine withdrawal and reward responsiveness in humans and rats. JAMA Psychiatry. 2014;71(11):1238–1245. 10.1001/jamapsychiatry.2014.1016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101. Cook JW, Piper ME, Leventhal AM, Schlam TR, Fiore MC, Baker TB. Anhedonia as a component of the tobacco withdrawal syndrome. J Abnorm Psychol. 2015;124(1):215–225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102. Leventhal AM, Piper ME, Japuntich SJ, Baker TB, Cook JW. Anhedonia, depressed mood, and smoking cessation outcome. J Consult Clin Psychol. 2014;82(1):122–129. 10.1037/a0035046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103. Versace F, Lam CY, Engelmann JM, et al. Beyond cue reactivity: blunted brain responses to pleasant stimuli predict long-term smoking abstinence. Addict Biol. 2012;17(6):991–1000. 10.1111/j.1369-1600.2011.00372.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104. MacPherson L, Tull MT, Matusiewicz AK, et al. Randomized controlled trial of behavioral activation smoking cessation treatment for smokers with elevated depressive symptoms. J Consult Clin Psychol. 2010;78(1):55–61. 10.1037/a0017939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105. Pratt LA, Brody DJ. Depression and smoking in the U.S. household population aged 20 and over, 2005–2008. NCHS Data Brief. 2010;(34):1–8. [PubMed] [Google Scholar]
- 106. McClave AK, McKnight-Eily LR, Davis SP, Dube SR. Smoking characteristics of adults with selected lifetime mental illnesses: results from the 2007 National Health Interview Survey. Am J Public Health. 2010;100(12):2464–2472. 10.2105/AJPH.2009.188136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107. Spring B, Pingitore R, McChargue DE. Reward value of cigarette smoking for comparably heavy smoking schizophrenic, depressed, and nonpatient smokers. Am J Psychiatry. 2003;160(2):316–322. [DOI] [PubMed] [Google Scholar]
- 108. Ussher M, West R, Steptoe A, McEwen A. Increase in common cold symptoms and mouth ulcers following smoking cessation. Tob Control. 2003;12(1):86–88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109. Hajek P, Gillison F, McRobbie H. Stopping smoking can cause constipation. Addiction. 2003;98(11):1563–1567. [DOI] [PubMed] [Google Scholar]
- 110. Hughes JR, Dash M, Callas P. Is impulsivity a symptom of initial tobacco withdrawal? a meta-analysis and qualitative systematic review [published online ahead of print 2014]. Nicotine Tob Res. 2014. 10.1093/ntr/ntu220. [DOI] [PubMed] [Google Scholar]
- 111. Etter JF, Ussher M, Hughes JR. A test of proposed new tobacco withdrawal symptoms. Addiction. 2013;108(1):50–59. 10.1111/j.1360-0443.2012.03981.x. [DOI] [PubMed] [Google Scholar]
- 112. Piasecki TM, Fiore MC, McCarthy DE, Baker TB. Have we lost our way? The need for dynamic formulations of smoking relapse proneness. Addiction. 2002;97(9):1093–1108. [DOI] [PubMed] [Google Scholar]
- 113. Muraven M, Tice DM, Baumeister RF. Self-control as limited resource: regulatory depletion patterns. J Pers Soc Psychol. 1998;74(3):774–789. [DOI] [PubMed] [Google Scholar]
- 114. Hagger MS, Wood C, Stiff C, Chatzisarantis NL. Ego depletion and the strength model of self-control: a meta-analysis. Psychol Bull. 2010;136(4):495–525. 10.1037/a0019486. [DOI] [PubMed] [Google Scholar]
- 115. Vohs KD, Heatherton TF. Self-regulatory failure: a resource-depletion approach. Psychol Sci. 2000;11(3):249–254. [DOI] [PubMed] [Google Scholar]
- 116. Inzlicht M, Schmeichel B. What is ego depletion? Toward a mechanistic revision of the resource model of self-control. Perspectives Psychol Sci. 2012;7(5):450–463. 10.1177/1745691612454134. [DOI] [PubMed] [Google Scholar]
- 117. Liu X, Li R, Lanza ST, Vasilenko SA, Piper M. Understanding the role of cessation fatigue in the smoking cessation process. Drug Alcohol Depend. 2013;133(2):548–555. 10.1016/j.drugalcdep.2013.07.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118. Bolt DM, Piper ME, Theobald WE, Baker TB. Why two smoking cessation agents work better than one: role of craving suppression. J Consult Clin Psychol. 2012;80(1):54–65. 10.1037/a0026366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119. Shiyko MP, Lanza ST, Tan X, Li R, Shiffman S. Using the time-varying effect model (TVEM) to examine dynamic associations between negative affect and self confidence on smoking urges: differences between successful quitters and relapsers. Prev Sci. 2012;13(3):288–299. 10.1007/s11121-011-0264-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120. Panksepp J, Knutson B, Burgdorf J. The role of brain emotional systems in addictions: a neuro-evolutionary perspective and new ‘self-report’ animal model. Addiction. 2002;97(4):459–469. [DOI] [PubMed] [Google Scholar]
- 121. Baker TB, Piper ME, McCarthy DE, Majeskie MR, Fiore MC. Addiction motivation reformulated: an affective processing model of negative reinforcement. Psychol Rev. 2004;111(1):33–51. [DOI] [PubMed] [Google Scholar]
- 122. Germeroth LJ, Wray JM, Tiffany ST. Response time to craving-item ratings as an implicit measure of craving-related processes [published online ahead of print 2014]. Clin Psychol Sci. 2014. 10.1177/2167702614542847 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123. Javitz HS, Brigham J, Lessov-Schlaggar CN, Krasnow RE, Swan GE. Association of tobacco dependence and quit attempt duration with Rasch-modeled withdrawal sensitivity using retrospective measures. Addiction. 2009;104(6):1027–1035. 10.1111/j.1360-0443.2009.02540.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124. Piasecki TM, Jorenby DE, Smith SS, Fiore MC, Baker TB. Smoking withdrawal dynamics: I. Abstinence distress in lapsers and abstainers. J Abnorm Psychol. 2003;112(1):3–13. 10.1037/0021-843X.112.1.3. [PubMed] [Google Scholar]
- 125. Zhou X, Nonnemaker J, Sherrill B, Gilsenan AW, Coste F, West R. Attempts to quit smoking and relapse: factors associated with success or failure from the ATTEMPT cohort study. Addict Behav. 2009;34(4):365–373. 10.1016/j.addbeh.2008.11.013. [DOI] [PubMed] [Google Scholar]
- 126. Ferguson SG, Shiffman S, Gwaltney CJ. Does reducing withdrawal severity mediate nicotine patch efficacy? A randomized clinical trial. J Consult Clin Psychol. 2006;74(6):1153–1161. 10.1037/0022-006X.74.6.1153. [DOI] [PubMed] [Google Scholar]