Abstract
Background. Identification of patients at risk for kidney allograft (KAG) failure beyond the first posttransplant year is an unmet need. We aimed to determine whether serum beta-2-microglobulin (β2MG) in the late posttransplant period could predict a decline in KAG function. Methods. We assessed a value of single measurement of serum β2MG at one to seventeen years after transplantation in predicting the estimated glomerular filtration rate (eGFR) and the decline in eGFR over a period of two years in 79 recipients of KAG. Results. At baseline serum β2MG concentration was higher (P = 0.011) in patients with allograft dysfunction: 8.67 ± 2.48 µg/mL versus those with satisfactory graft function: 6.67 ± 2.13 µg/mL. Higher β2MG independently predicted the lower eGFR, the drop in eGFR by ≥25% after one and two years, and the value of negative eGFR slope. When combined with proteinuria and acute rejection, serum β2MG had excellent power in predicting certain drop in eGFR after one year (AUC = 0.910). In conjunction with posttransplant time serum β2MG had good accuracy in predicting certain eGFR drop after two years (AUC = 0.821). Conclusions. Elevated serum β2MG in the late posttransplant period is useful in identifying patients at risk for rapid loss of graft function.
1. Introduction
Kidney transplantation is the most effective method of treatment for patients with terminal kidney insufficiency. At present the rate of one-year kidney allograft survival exceeds 95% [1] and the preservation of allograft function in the late period after transplantation has become the main challenge [1, 2]. Identification of patients at risk for kidney graft failure beyond the first posttransplant year is a prerequisite for developing strategies for saving graft function and improving its survival [3–6]. Many recent studies have focused on higher serum creatinine and lower glomerular filtration rate (GFR) within the first posttransplant year as surrogate predictors of inferior graft survival [3, 7–10]. The rate of decline in GFR also predicts graft failure [3, 9] and can be even more useful surrogate endpoint [11, 12]. However, the ability of allograft function in the first year after transplantation to predict the long-term results is questionable [3, 13]. Furthermore, serum creatinine and estimated GFR (eGFR) are now considered by some authors as not quite sensitive and specific markers of kidney function [14–16]. Additional surrogate predictors of decline in allograft function in the late posttransplant period can help better identify patients at risk for failure in order to improve their monitoring and allow early intervention.
β2-microglobulin (β2MG) is a membrane protein associated with class I major histocompatibility complex proteins and is, therefore, found on the surface of all nucleated cells. Under physiological conditions, β2MG is produced at a constant rate and is eliminated through the kidney. The low molecular weight (11.800 Da) allows β2MG to pass through the glomerular membrane, but it is almost completely reabsorbed in the proximal tubules [17]. Several authors report that measurement of the serum β2MG concentration in native kidney diseases estimates GFR as serum creatinine does [16, 18, 19] and even supersedes it [15, 20, 21]. Serum β2MG level increases because of intensification of its extrarenal synthesis in conditions such as systemic inflammation, some acute viral infections, and a number of malignancies [17, 19, 22–25]. Data about β2MG as a marker of kidney graft function are limited and contradictory [26–28]. Astor et al. have recently demonstrated that high serum β2MG at discharge predicted kidney graft loss [29], but detailed retrospective information might not be available for all patients. The association between serum β2MG and the rate of decline in kidney allograft function remains obscure. The aim of the present study was to determine whether serum beta-2-microglobulin at different intervals of late posttransplant period could predict the decline in GFR over a follow-up period of two years.
2. Materials and Methods
2.1. Study Population
During the four-month period (from September till December, 2012), in total, 90 Caucasian patients receiving a kidney allograft in Zaporizhzhia transplantation center between January 1995 and September 2011 and willing to participate were recruited. A total of 79 patients, 47 males and 32 females, aged 16 to 59, who fulfilled the inclusion criteria, were enrolled in the study. The criteria of inclusion to the study were the following: the adult kidney allograft recipient, male or female, with primary transplantation from related or deceased donor, with allograft survival of at least one year and eGFR not less than 15 mL/min/1.73 m2. The criteria of exclusion were regular dialysis, acute kidney injury, diseases of immune system, solid tumours, and clinical signs of acute infections. All recipients received triple maintenance immunosuppressive therapy consisting of calcineurin-inhibitor (cyclosporine or tacrolimus), antiproliferative agent (mycophenolate mofetil or azathioprine), and steroid. All participants gave their informed written consent. This research was approved by local ethics committee and carried out in accordance with the ethical standards laid down in the Declaration of Helsinki (2013 version) and the Declaration of Istanbul.
2.2. Laboratory Methods
Serum was obtained from the venous blood taken in the morning from patients in a fasting condition. For measuring of β2MG serum had been frozen and stored at minus 40°С. Simultaneously, freshly voided morning urine samples were collected and centrifuged at 3000 rpm for 15 min. Urine specific gravity was measured with a urinometer. We measured serum concentration of urea by the urease method, urine protein level by the pyrogallol red-molybdate method, and serum and urine creatinine concentration by the Jaffe method. All kits were supplied by “Filisit-Diagnostics,” Ukraine. The absorbance was measured on spectrophotometer “APEL” (Japan). The results of total protein in urine spot were normalized to urinary creatinine. As a threshold of normalized proteinuria we considered the level of 15 mg/mmol [12]. For GFR estimation we used a four-variable equation derived from the Modification of Diet in Renal Disease (MDRD) Study [30]. Quantitative determination of β2MG in serum samples was performed by enzyme-linked immunosorbent assay (kit supplied by Orgentec GmbH, Germany) according to the manufacturer's protocol. The absorbance was measured using a plate reader “Tecan” Sunrise (Austria). Mean reference value given for serum β2MG is 0–3.0 μg/mL.
2.3. Risk Factors and Outcomes Examined
In this study we assessed a prospective value of a cross sectional measurement of serum β2MG. Archival patient records and outpatient cards were used to obtain more information on major risk factors and evolution of allograft function. Data were collected on recipient's age, gender, and presence of chronic arterial hypertension, defined as a regular intake of antihypertensive drugs. We also obtained the information related to transplantation: donor source, type of immunosuppression, initial graft function, acute rejection episodes, and time after transplantation. At enrollment the arterial pressure was measured in a sitting position after a 10-minute rest period. For linear regression analysis the initial allograft function was classified as follows: immediate function (0 points), slow graft function, that is, serum creatinine reduction from transplantation to day seven <70% (1 point), delayed graft function (DGF), that is, need for at least one time dialysis in the first seven days after surgery (2 points), and DGF that required more than one dialysis procedure (3 points). Acute rejection was defined by the need for treatment, with or without biopsy confirmation. For linear regression analysis episodes of acute rejection were classified as follows: absence of acute rejection (0 points), early (<3 months) acute rejection successfully treated by steroid therapy (1 point), late acute rejection successfully treated by steroid therapy (2 points), acute rejection with need for antilymphocyte antibody therapy (3 points), and resistant to therapy acute rejection with noncomplete recovery (4 points). Patients, enrolled in this study, were followed for two years until death/return to dialysis or until December 2014. In the course of follow-up period GFR was estimated annually. During the 2nd year of follow-up, a total of three deaths with functioning graft occurred and five grafts failed. For five patients, who returned to dialysis, we imputed a GFR of 10 mL/min/1.73 m2. The annualized change (slope) in eGFR (mL/min/1.73 m2/year) over a period of two years was calculated for each patient, having three eGFR values, by the linear mixed effects model with varying intercept and slope. We determined the proportion of patients having an eGFR slope ≥ −1 mL/min/1.73 m2/year and the proportion of patients having an eGFR drop of ≥25% from baseline, since both measures indicate progressive loss of kidney function [12]. We also calculated the frequency of patients who showed improvement in GFR (increase in eGFR ≥ 1 mL/min/1.73 m2/year and increase in eGFR ≥25% from baseline). The endpoints of the study were the eGFR and the drop in eGFR of ≥25% from baseline after one and two years of follow-up, and the slope of eGFR.
2.4. Statistics
Normally distributed data are expressed as mean ± SD; the results were compared with Student's t-test; Pearson's coefficient of correlation (r) was determined where appropriate. Continuous nonparametric data are expressed as the median (interquartile range); for comparison we used Mann-Whitney's U-test; Spearman's correlation coefficient (R) was calculated where appropriate. Frequency data are expressed as percentages and for comparison we applied the Chi-square test. To identify predictors of eGFR after one and two years and those of eGFR slope, multiple linear regression was used. Normalized proteinuria and mean arterial pressure had non-Gaussian distribution and were natural log-transformed. The predictors that significantly correlated with dependent variable were tested for multicollinearity and excluded when appropriate. Predictive variables, if significantly associated (P < 0.05) with the dependent variable in simple linear regression analysis, were included in multivariate model with forward stepwise selection. To identify independent predictors of a certain drop in eGFR (≥25% from baseline [12]) after one and two years of follow-up we used univariate logistic regression analysis. Then multivariate analysis was performed with only those predictive variables that demonstrated individual P values <0.05. In addition, we calculated areas under the receiver operating characteristic curves (AUC) to assess the capability of serum β2MG and other variables of interest to discriminate patients with a drop in eGFR ≥25% after one and two years of follow-up. We used logistic regression models to estimate combinations of serum β2MG and other variables of interest and evaluated the discriminatory ability of the combinations with the AUC. AUCs were compared using the DeLong test. Statistica 7.0 (StatSoft Inc., Tulsa, USA), SPSS (version 19.0 SPSS Inc., Chicago, USA) and Medcalc V.14.8.1 (MedCalc Software bvba, Ostend, Belgium) packages were used for statistical analyses. Statistical significance was set at P < 0.05.
3. Results
3.1. Baseline Characteristics of Patients
The mean eGFR at baseline was 50.4 ± 19.7 mL/min/1.73 m2. Most patients were in the third (n = 49) or in the second (n = 21) stages of chronic kidney disease (CKD). We decided to categorize patients according to the median eGFR into “Satisfactory Function” and “Dysfunction” subsets with eGFR >44 mL/min/1.73 m2 and eGFR ≤44 mL/min/1.73 m2, respectively. We gained confidence in the correctness of the chosen approach since the eGFR of 44 mL/min/1.73 m2 is a threshold between CKD stages G3a and G3b. The demographics and clinical characteristics of patients are depicted in Table 1. The majority of patients received a kidney from a deceased donor and they were on CsA-based immunosuppression. No differences in gender, age, or type of immunosuppression were observed between subsets of recipients. Most of the patients (62.5% and 74.4% of the Dysfunction group and Satisfactory Function group, resp.) were transplanted from one to ten years ago. Time after transplantation exceeded 10 years in 35% of patients from the Dysfunction group and in 15.4% of patients with satisfactory function (P = 0.045). Accordingly, the time after transplantation was significantly longer in the Dysfunction group (Table 1). In the Dysfunction group impairment of initial function and acute rejection episodes were significantly more common. Only one patient had late acute rejection (at 3.5 years), whereas the remaining patients had early acute rejection episodes (within 3 months). The mean arterial pressure and the percentage of patients regularly receiving antihypertensive therapy were significantly higher in the Dysfunction group. The differences between the main laboratory parameters of graft status (serum creatinine, eGFR, and normalized proteinuria) were highly significant (Table 1).
Table 1.
Baseline characteristics of the study population.
| Parameter | Satisfactory Function subset | Dysfunction subset | P value |
|---|---|---|---|
| N = 39 | N = 40 | ||
| Recipient age at baseline, years | 37 ± 12∗ | 40 ± 11 | 0.329 |
| Recipient gender, n (%) | 0.573 | ||
| Male | 21 (54)‡ | 19 (48) | |
| Female | 18 (46) | 21 (52) | |
| CNI, n (%) | 0.105 | ||
| CsA | 34 (87) | 29 (73) | |
| Tacrolimus | 5 (13) | 11 (27) | |
| Antiproliferative agent, n (%) | 0.190 | ||
| Azathioprine | 3 (8) | 7 (18) | |
| Mycophenolate mofetil | 36 (92) | 33 (72) | |
| Type of donor, n (%) | 0.958 | ||
| Deceased | 32 (82) | 33 (83) | |
| Living related | 7 (18) | 7 (17) | |
| Impaired initial function, n (%) | 2 (5.1) | 10 (25) | 0.014 |
| Previous acute rejection, n (%) | 5 (12.8) | 14 (35) | 0.021 |
| Time after transplantation (months) | 72.6 ± 46.9 | 98.2 ± 57.7 | 0.034 |
| Serum creatinine (µmol/L) | 117.7 ± 29.5 | 195.7 ± 49.7 | <0.001 |
| eGFR at baseline (mL/min/1.73 m2) | 66.7 ± 13.4 | 34.3 ± 7.9 | <0.001 |
| Serum urea (mmol/L) | 7.4 ± 2.3 | 11.9 ± 3.4 | <0.001 |
| Proteinuria/creatinine (mg/mmol) | 3.6 (2.7–7.9)† | 11.8 (4.5–21.8) | <0.001 |
| Proteinuria/creatinine > 15 mg/mmol, n (%) | 2 (5.1) | 15 (37.5) | <0.001 |
| Diuresis (mL/day) | 1428 ± 243 | 1498 ± 372 | 0.331 |
| Urine specific gravity | 1012 ± 4 | 1007 ± 4 | <0.001 |
| Mean arterial pressure at baseline (mm Hg) | 103 ± 9 | 113 ± 9 | <0.001 |
| Treated hypertension at baseline, n (%) | 12 (30.8) | 28 (70.0) | <0.001 |
CNI: calcineurin inhibitors; CsA: cyclosporine A; eGFR: estimated glomerular filtration rate; N: number of patients investigated; ∗mean ± standard deviation, Student's t-test used for comparison; †median (interquartile range), Mann-Whitney's U-test used for comparison; ‡numbers (percentages), Chi-square tests used for comparison.
3.2. β2MG Level and Its Correlation with Allograft Function at Baseline
Serum β2MG level remained in the reference ranges (0–3.0 μg/mL) only in one patient, whereas it was elevated in the other patients. β2MG concentration was significantly higher (P = 0.011) in the subset of patients with allograft dysfunction: 8.67 ± 2.48 μg/mL versus 6.67 ± 2.13 μg/mL in Satisfactory Function subset. The clinical variables: type of the donor, age and gender of the recipient, characteristics of acute rejection episodes, type of initial graft function, mean arterial pressure, and time after transplantation did not correlate with the serum levels of β2MG (P > 0.05). Only serum creatinine concentration (r = 0.466, P = 0.002), eGFR (r = −0.338, P = 0.033), and normalized proteinuria (r = 0.388, P = 0.013) significantly correlated with serum β2MG levels.
3.3. The Evolution of Allograft Function and the Predictive Variables
After one year of follow-up, the mean eGFR did not significantly change in the total group of patients: 49.2 ± 19.7 mL/min/1.73 m2 compared to eGFR at baseline (P = 0.289). Twenty percent of the patients from the Dysfunction group and only five percent of patients from the Satisfactory Function group (P = 0.047) demonstrated decline in eGFR by ≥25% from baseline after 1 year (Figure 1). Eight patients had chronic allograft dysfunction, but only in two cases the diagnoses were confirmed histologically. One patient had chronic active T-cell-mediated rejection and the second patient had chronic nephrotoxicity in combination with hypertensive nephropathy. We also observed progression of heart failure from New York Heart Association functional class II to class III in two other patients, which might cause worsening of graft function. Fifteen percent of patients from the Dysfunction group showed the increase in eGFR by ≥25% (Figure 1) after 1 year, whereas such patients were absent in the Satisfactory Function group (P < 0.001). The presumed causes of improving graft function were conversion of immunosuppression (from cyclosporine to tacrolimus in two patients and from mycophenolate mofetil to everolimus in two other patients), effective antibiotic treatment of urinary tract infection (1 patient), and increase of renal perfusion after coronary artery bypass (1 patient). During the 2nd year of follow-up, a total of three deaths with functioning graft occurred and a total of five grafts failed. The cause of allograft failure was verified histologically in selected patients and we found chronic active antibody-mediated rejection (1 patient), chronic pyelonephritis (1 patient), and de novo glomerulonephritis (1 patient). After two years of follow-up the mean eGFR in the total group of patients significantly decreased (46.3 ± 20.6 mL/min/1.73 m2) compared with that at baseline (P = 0.002). This decline was also significant compared to eGFR after one year (P < 0.001). 22.5% of patients from the Dysfunction group and 13% of patients from the Satisfactory Function group displayed the decrease in eGFR by ≥25% compared to baseline (P = 0.260) (Figure 1). We did not observe any new intercurrent events during this period. Only 10% of patients (all from the Dysfunction group) showed the increase in eGFR by ≥25% compared with that at baseline (P = 0.043) (Figure 1). The eGFR slope in the overall group of patients was −2.0 ± 5.7 mL/min/1.73 m2/year (median: −2.0 mL/min/1.73 m2/year). In Satisfactory Function subset the slope was −2.4 ± 6.3 mL/min/1.73 m2/year (median: −1.5 mL/min/1.73 m2/year), which did not significantly differ from Dysfunction subset (P = 0.519): −1.6 ± 5.2 mL/min/1.73 m2/year (median: −2.0 mL/min/1.73 m2/year).
Figure 1.
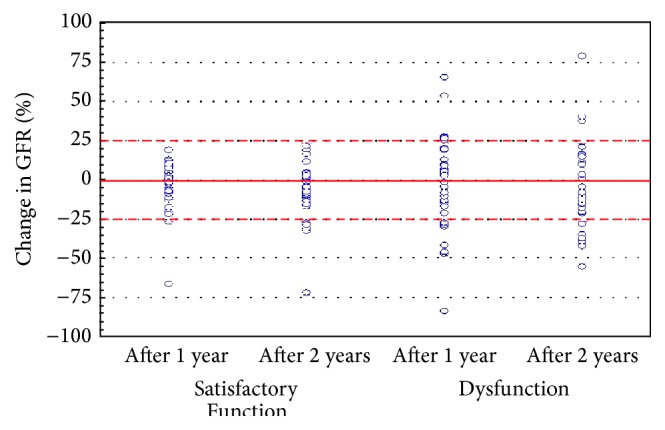
Scatterplot of patients from Satisfactory Function and Dysfunction subsets according to the change in glomerular filtration rate (GFR).
The eGFR decreased by ≥1 mL/min/1.73 m2/year in 60% of patients from the Dysfunction group and in 62% of patients with satisfactory graft function (P > 0.05). At the same time, the eGFR increased by ≥1 mL/min/1.73 m2/year in 30% of patients from the Dysfunction group and in 31% of patients with satisfactory function (P > 0.05). In the course of the study the relationship between the eGFR and serum β2MG was increasing: r = −0.338 (P = 0.033) at baseline to r = −0.538 (P < 0.001) at one year and r = −0.527 (P < 0.001) at two years after enrollment.
Table 2 summarizes the linear regression results for predictors of eGFR at different time points. The clinical variables, mean arterial pressure, normalized proteinuria, impaired initial graft function, and acute rejection, were associated with lower eGFR at baseline in univariate analysis. In multivariate analysis, only higher mean arterial pressure and higher normalized proteinuria were independent predictors of lower eGFR (Table 2). To determine the independent effect of β2MG on the evolution of kidney allograft function we also included clinical variables in the regression equation. The following variables significantly influenced the eGFR after one year in univariate analysis: eGFR at baseline, serum β2MG concentration, normalized proteinuria, mean arterial pressure, time after transplantation, and initial graft function (Table 2). The same variables and, in addition, acute rejection significantly influenced the eGFR after two years in univariate analysis (Table 2). In multivariate model, only lower eGFR at baseline and higher serum β2MG concentration were independent predictors of lower eGFR after one and two years of follow-up. The values of the adjusted coefficient of determination R 2 = 0.712 and Fisher statistics (F = 48.0, P < 0.001) indicated the good quality of models for prediction of the eGFR after one year as well as after two years: R 2 = 0.665, F = 26.2, and P < 0.001.
Table 2.
Significant predictors of eGFR at different time points of late posttransplant period∗.
| Predictive variables | Univariate linear regression | Multivariate linear regression | ||||
|---|---|---|---|---|---|---|
| Beta | SE | P value | Beta | SE | P value | |
| eGFR at baseline (mL/min/1.73 m2), N = 79 | ||||||
| Log proteinuria/creatinine† | −0.488 | 0.104 | <0.001 | −0.406 | 0.153 | 0.012 |
| Impaired initial function‡ | −0.235 | 0.112 | 0.040 | |||
| Log mean arterial pressure† | −0.502 | 0.099 | <0.001 | −0.332 | 0.153 | 0.037 |
| Previous acute rejection‡ | −0.241 | 0.112 | 0.035 | |||
|
| ||||||
| eGFR after 1 year of follow-up (mL/min/1.73 m2), N = 79 | ||||||
| eGFR at baseline | 0.870 | 0.057 | <0.001 | 0.721 | 0.091 | <0.001 |
| Log proteinuria/creatinine† | −0.476 | 0.105 | <0.001 | |||
| Serum β2MG | −0.538 | 0.137 | <0.001 | −0.291 | 0.091 | 0.003 |
| Impaired initial function‡ | −0.233 | 0.113 | 0.042 | |||
| Log mean arterial pressure† | −0.436 | 0.103 | <0.001 | |||
| Time after transplantation | −0.262 | 0.111 | 0.021 | |||
|
| ||||||
| eGFR after 2 years of follow-up (mL/min/1.73 m2), N = 71 | ||||||
| eGFR at baseline | 0.838 | 0.063 | <0.001 | 0.649 | 0.101 | <0.001 |
| Log proteinuria/creatinine† | −0.473 | 0.105 | <0.001 | |||
| Serum β2MG | −0.527 | 0.138 | <0.001 | −0.302 | 0.098 | 0.004 |
| Impaired initial function‡ | −0.230 | 0.113 | 0.030 | |||
| Log mean arterial pressure† | −0.447 | 0.103 | <0.001 | |||
| Time after transplantation | −0.332 | 0.108 | 0.003 | −0.148 | 0.097 | 0.135 |
| Previous acute rejection‡ | −0.261 | 0.112 | 0.023 | |||
β2MG: β2-microglobulin; eGFR: estimated glomerular filtration rate; beta: standardized regression coefficient; SE: standard error of beta; N: number of patients investigated; ∗only variables that significantly influenced the eGFR in univariate analysis are included; †natural log-transformed variables; ‡impaired initial function and acute rejection scored in points.
Only higher serum concentration of β2MG was a predictor of more negative slope of eGFR in linear regression analysis (β = −0.222, SE = 0.111, and P = 0.049). None of other variables, including the eGFR at baseline, correlated with the magnitude of eGFR slope.
Table 3 provides details of logistic regression analysis for the value of β2MG level and other laboratory and clinical parameters to predict the eGFR decline by ≥25% after one and two years of follow-up. Higher normalized proteinuria and serum β2MG as well as previous acute rejection episodes were individually predictive for decline in eGFR of ≥25% after one year of follow-up (Table 3), wherein only serum β2MG retained its predictive value in multivariate model. With respect to the graft function after two years of follow-up both higher serum β2MG and longer time after transplantation were associated with higher odds ratio of certain eGFR drop (Table 3).
Table 3.
Significant predictors of eGFR decline by ≥25% after one and two years of follow-up∗.
| Predictive variables | Univariate logistic regression | Multivariate logistic regression | ||||
|---|---|---|---|---|---|---|
| OR | CI | P value | OR | CI | P value | |
| eGFR decline by ≥25% after 1 year of follow-up, N = 10 | ||||||
| Proteinuria/creatinine | 1.03 | 1.00–1.07 | 0.038 | 1.03 | 0.99–1.07 | 0.158 |
| Serum β2MG | 1.51 | 1.10–2.07 | 0.010 | 1.54 | 1.05–2.26 | 0.025 |
| Previous acute rejection | 4.82 | 1.12–20.84 | 0.032 | 2.24 | 0.36–13.85 | 0.378 |
|
| ||||||
| eGFR decline by ≥25% after 2 years of follow-up, N = 12 | ||||||
| Serum β2MG | 1.36 | 1.06–1.76 | 0.016 | 1.41 | 1.06–1.87 | 0.016 |
| Time after transplantation | 1.02 | 1.00–1.03 | 0.006 | 1.02 | 1.00–1.03 | 0.006 |
β2MG: β2-microglobulin; eGFR: estimated glomerular filtration rate; OR: odds ratio; CI: 95% confidence interval; N: number of patients with certain decline in eGFR; ∗only variables that significantly influenced the eGFR decline in univariate analysis are included.
The discriminating ability of each variable, considered significant by simple logistic regression, for the drop in eGFR ≥25% after one and two years of follow-up was determined by the AUC analysis. As shown in Table 4, only serum β2MG was the significant predictor for the decline in eGFR by ≥25% after one year. The combination of normalized proteinuria, acute rejection, and serum β2MG resulted in an AUC of 0.910 in predicting certain eGFR drop after one year (Table 4), which indicated excellent discriminating ability. This AUC showed higher predictive power than AUC for normalized proteinuria (P = 0.0022) and acute rejection alone (P = 0.0003) but did not significantly differ from the AUC for serum β2MG (P = 0.070). Serum β2MG and time after transplantation exhibited only poor and fair discriminatory power, respectively, in predicting the eGFR decline by ≥25% after 2 years of follow-up (Table 4). The AUCs for these two variables were not significantly different from each other (P > 0.05). The combination of serum β2MG and time after transplantation yielded the AUC of 0.821, indicating good predictive power for certain eGFR drop after two years (Table 4). The AUC of this combined model was higher than the AUC for each predictor alone, but not significantly (P > 0.05).
Table 4.
AUC for potential markers of eGFR decline by ≥25% after one and two years of follow-up∗.
| Predictive variables | AUC | CI | P-level |
|---|---|---|---|
| eGFR decline by 25% after 1 year of follow-up, N = 10 | |||
| Proteinuria/creatinine | 0.724 | 0.606–0.823 | 0.056 |
| Previous acute rejection | 0.675 | 0.559–0.777 | 0.055 |
| Serum β2MG | 0.724 | 0.612–0.819 | 0.026 |
| Proteinuria/creatinine + previous acute rejection + serum β2MG | 0.910 | 0.818–0.965 | <0.001 |
|
| |||
| eGFR decline by 25% after 2 years of follow-up, N = 12 | |||
| Serum β2MG | 0.668 | 0.553–0.770 | 0.047 |
| Time after transplantation | 0.712 | 0.599–0.808 | 0.008 |
| Serum β2MG + time after transplantation | 0.821 | 0.719–0.899 | <0.001 |
β2MG: β2-microglobulin; eGFR: estimated glomerular filtration rate; AUC: areas under the receiver operating characteristic curve; CI: 95% confidence interval; N: number of patients with certain decline in eGFR; ∗only variables that significantly influenced the eGFR decline in univariate logistic regression are included.
4. Discussion
In this study we found the association between serum level of β2MG in the late posttransplant period and a subsequent decline in kidney allograft function within a short time-frame. Serum β2MG not only was associated with lower eGFR, but also predicted the value of negative slope in eGFR and the certain decline in eGFR of ≥25% from baseline. It is of importance that predicted drop in eGFR occurred in a short time-frame of two years and even one year. We also found the fair and poor accuracy of serum β2MG after one and two years, respectively, in discriminating patients with eGFR drop of ≥25%. However, in conjunction with normalized proteinuria at baseline and history of acute rejection, serum β2MG had excellent power in predicting certain drop in eGFR after one year. When combined with time after transplantation serum β2MG had good power in predicting the eGFR drop of ≥25% after two years.
As only higher serum creatinine, lower eGFR at baseline, and proteinuria were associated with higher serum β2MG concentration, we believe that the current allograft status is the main factor that influences serum β2MG. Several studies evaluating the role of serum β2MG as a marker of kidney function reported that serum β2MG concentration increased when renal function decreased both in transplanted patients [26, 27] and in patients with CKD [16, 17, 19, 31, 32]. Other authors stated that serum β2MG estimated GFR even more accurately compared to serum creatinine [15, 20, 21]. Woo et al. [33] concluded that the increase of serum β2MG concentration evidenced a glomerular pathology, which has recently been recognized as the main cause of chronic allograft dysfunction [6]. Meanwhile, observed weak correlation (r = 0.466) between serum β2MG and creatinine (though their sieving coefficients are close to unity [33]) suggests that another reason for the increase of serum β2MG concentration might be transplant complications and comorbidities leading to an increase of β2MG synthesis. The increase of serum β2MG after kidney transplantation can be a marker for cytomegalovirus infection [23], lymphoproliferative disease [24, 25], acute kidney allograft rejection [28, 34, 35], CsA-nephrotoxicity [35], and cardiovascular diseases [31]. These data imply that patients with elevated serum level of β2MG require closer monitoring.
Most of the patients displayed decline in the eGFR during the follow-up, whereas others exhibited increase or stabilization. After two years of follow-up the average eGFR decreased significantly compared to baseline. Our data are consistent with the conception of several patterns of graft function evolution over time [36, 37]: achieving optimal or suboptimal function with subsequent stabilization or decline. In addition, several papers reported the ability of grafts to increase GFR after six months or one year [3, 8, 38, 39]. The capacity to increase GFR in much later period after transplantation, as we have shown, suggests that even in late posttransplant period many allografts retain a substantial functional reserve and timely therapy can be effective.
Only lower eGFR at baseline and higher serum β2MG concentration were shown both as independent predictors of lower eGFR after one and two years of follow-up. The observed associations were independent from potential confounders. About 71% and 67% of the variances in the eGFR at one and two years after enrollment, respectively, can be explained by combined influence of the eGFR at baseline and serum β2MG concentration, which allows the use of this model in the future on other patients. Further, we evaluated the predictors of the slope of eGFR, which might be even more accurate and an earlier indicator of chronic allograft injury and better predict allograft function and survival [11, 12]. Patients in the whole group as well as in their subsets demonstrated the negative average slope of eGFR. A more negative slope of eGFR was typical for patients with higher concentration of serum β2MG, but it was independent from eGFR at baseline. The independence of the slope from the absolute level of GFR [38] and even opposite relationship between the GFR intercept and slope [39, 40] were reported earlier. At the same time, Sijpkens et al. [36] found a significant correlation between low creatinine clearance at 6 months and negative slope. But it should be mentioned that in the study by Sijpkens et al. [36] patients were transplanted between 1979 and 1983, before the introduction of CsA, when the deleterious effect of acute rejection episodes was more pronounced. Our findings along with the literature data imply that eGFR alone is not quite an accurate predictor of kidney graft failure. This is supported by the recent paper of Park et al. [3], describing that 41% of allografts with a satisfactory function at one year (eGFR ≥40 mL/min/1.73 m2) will progressively lose it and cease to function in the range of one to five years. In this regard β2MG, which accurately reflects graft function and provides information about additional injuries to the graft, might improve prediction [29].
We also demonstrated that after adjustment for confounding variables in logistic regression serum β2MG predicted a drop in eGFR by ≥25% after one and two years of follow-up. Thus, in multivariate model, for a one-unit increase in serum β2MG concentration, the odds ratio for certain drop in eGFR was 1.54 and 1.41 after one and two years, respectively. Serum β2MG alone showed a fair discriminatory power for certain decline in eGFR after one year. However, when combined with normalized proteinuria and acute rejection, serum β2MG further enhanced the quality of prognosis and displayed an excellent predictive performance. As long as the AUC for combination of variables did not significantly differ from that for β2MG alone, but it was higher than the AUCs for normalized proteinuria and acute rejection alone (P < 0.05), we conclude that serum β2MG provides absolutely essential data to the predictive model. Serum β2MG alone exhibited poor power to discriminate patients with certain decline in eGFR after two years. However, in conjunction with the time after transplantation serum β2MG enhanced the quality of prognosis and displayed good predictive performance. Thus, when combined with selected laboratory and clinical variables serum β2MG can predict a certain drop in GFR with high sensitivity and specificity. A negative impact of longer time after transplantation on the stability of graft function can be attributed to larger cumulative burden of injury with increasing time [41] and to the effect of transplant era. Thus, as reported in paper by Kasiske et al. [42], the rate of decline in kidney graft function significantly improved in recent years due to improvement in patients' care and treatment strategies.
We also identified proteinuria, higher mean arterial pressure, impaired initial function, and acute rejection episodes as clinical predictors of lower allograft function in the late posttransplant period. These variables are a combination of immunologic and nonimmunologic factors, associated with the early posttransplant events and chronic damage to the graft that fit into the conception of etiology and pathogenesis of chronic allograft dysfunction [11, 13, 37]. But their influence was significant only in univariate model, before adjustment for eGFR and serum β2MG. Our results imply that lower eGFR and higher serum β2MG in the late posttransplant period are surrogate markers of harmful effects on kidney allograft. None of clinical predictive variables (except time after transplantation) were independently associated with the rate of decline in kidney allograft function, whereas in several papers [36, 38, 40, 42] authors found the association of donor, recipient, and transplant variables with progressive loss of renal function in the course or after the 1st posttransplant year. Unlike these studies, our patients were in much later posttransplant period: from one to seventeen years. We followed recipients only for two years, but about 30% of patients exhibited positive eGFR slope. Also our results suggest that effects of early acute rejection and impaired initial function might not be so deleterious in the late posttransplant period. As highlighted by Meier-Kriesche et al. [43], two phases in evolution of allograft dysfunction exist. Technical problems and acute rejection have the greatest influence on the allograft function and survival in the early phase, whereas effects of chronic rejection, recurrent diseases, and nephrotoxicity predominate in the late phase [6]. Observed in recent years modest improvement in the allograft survival was achieved mainly due to improving of short-term outcomes, in particular, because of decrease in the incidence of acute rejection [1, 43]. Therefore, it is necessary to identify risk factors which are harmful for graft in the late posttransplant period as well as their surrogates. Also, since there are a number of variables that might not be available early after transplantation but, when available, can change the approach to therapy, we believe that information provided by late posttransplant tests, particularly, by serum β2MG, might be of use.
Our results suggest some more clinical and theoretical implications. Association of elevated serum β2MG with both low eGFR and decline in eGFR from baseline might have considerable clinical impact, since these surrogates are directly related to inferior allograft survival [3, 7, 11, 12] and, when combined, the negative effect is even more pronounced [9]. Predictably, a positive slope is associated with improved allograft survival [8]. Thus, the results lead us to believe that serum β2MG represents a link between low intercept and negative slope of GFR and can be an important surrogate marker for the progression of kidney allograft dysfunction. Thereby, our findings expand results of previous studies on the relationship between GFR intercept and slope and their predictive variables. It is also worthwhile to highlight that certain decline in GFR over one and two years, predicted by serum β2MG, indicates rapid progression of kidney allograft dysfunction [12]. To our knowledge, this is the first study to introduce late posttransplant serum β2MG for the prediction of decline in kidney graft function in the short time-frame. This study, however, has a number of limitations. First, it was a single-center study carried out on a small group of Caucasian patients and restricted to only two years of follow-up period. So our conclusions might have only preliminary character and should be confirmed with a larger cohort representative of the general kidney transplant population. Second, we did not measure GFR directly but relied on estimating GFR from MDRD equation, which, however, is allowable in accordance with the recommendations of recent guidelines [12]. Third, the creatinine measurements were not calibrated with respect to a reference laboratory. However, the effect of possible calibration error would equally influence the results of measurements in all patients and, therefore, cannot explain the observed differences in eGFR values. The fourth limitation is that GFR estimation was not paired with β2MG measurement at all time points. These data could help elucidate the processes leading to graft failure and understand whether the serum concentration of β2MG was mainly determined by the level of kidney function or increased protein synthesis. Finally, we can only assume the etiology of dysfunction, because allograft biopsy was performed only in individual patients. It is possible that future combination of serum β2MG measurement with biopsy results will improve diagnostics and prediction of kidney transplant pathology.
5. Conclusions
The overall results show that higher serum level of β2MG after a single measurement at different intervals of late posttransplant period independently predicts the lower eGFR and the drop in eGFR by ≥25% after one and two years of follow-up, as well as the value of negative annualized change in eGFR. With respect to its discriminative characteristics serum β2MG predicts the eGFR drop of ≥25% after one year and two years with fair and poor accuracy, respectively. However, when combined with normalized proteinuria at baseline and history of acute rejection serum β2MG demonstrated excellent power in predicting certain eGFR drop after one year. In conjunction with time after transplantation serum β2MG had good accuracy in predicting certain eGFR drop after two years. Thus, higher serum β2MG is a risk factor for decline in eGFR, particularly with respect to certain drop in eGFR after one year of follow-up. These observations highlight the potential importance of elevated serum β2MG in the late posttransplant period in identifying a group of transplant patients who are at risk for rapid loss of graft function and might benefit from early therapeutic interventions.
Acknowledgments
The authors gratefully acknowledge the assistance of the medical and nursing staff of the Department of Transplantation, Zaporizhzhia Regional Hospital. This study was supported in part by resources from the State Institution “Zaporizhzhia Medical Academy of Post-Graduate Education Ministry of Health of Ukraine.”
Disclosure
Part of this work was accepted for publication in the abstract format and was presented as e-poster at the 2015 ESOT Congress in Brussels, Belgium, 13–16 September, 2015.
Conflict of Interests
The authors declare that they have no competing interests.
References
- 1.Saran R., Li Y., Robinson B., et al. US Renal Data System 2014 annual data report: epidemiology of kidney disease in the United States. American Journal of Kidney Diseases. 2014;66(1, supplement 1):S1–S306. doi: 10.1053/j.ajkd.2015.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Morales J. M., Marcén R., del Castillo D., et al. Risk factors for graft loss and mortality after renal transplantation according to recipient age: a prospective multicentre study. Nephrology Dialysis Transplantation. 2012;27(4):iv39–iv46. doi: 10.1093/ndt/gfs544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Park W. D., Larson T. S., Griffin M. D., Stegall M. D. Identification and characterization of kidney transplants with good glomerular filtration rate at one year but subsequent progressive loss of renal function. Transplantation. 2012;94(9):931–939. doi: 10.1097/tp.0b013e3182692b70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Park W. D., Griffin M. D., Cornell L. D., Cosio F. G., Stegall M. D. Fibrosis with inflammation at one year predicts transplant functional decline. Journal of the American Society of Nephrology. 2010;21(11):1987–1997. doi: 10.1681/asn.2010010049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Issa N., Cosio F. G., Gloor J. M., et al. Transplant glomerulopathy: risk and prognosis related to anti-human leukocyte antigen class II antibody levels. Transplantation. 2008;86(5):681–685. doi: 10.1097/tp.0b013e3181837626. [DOI] [PubMed] [Google Scholar]
- 6.El-Zoghby Z. M., Stegall M. D., Lager D. J., et al. Identifying specific causes of kidney allograft loss. American Journal of Transplantation. 2009;9(3):527–535. doi: 10.1111/j.1600-6143.2008.02519.x. [DOI] [PubMed] [Google Scholar]
- 7.Kasiske B. L., Israni A. K., Snyder J. J., Skeans M. A. The relationship between kidney function and long-term graft survival after kidney transplant. American Journal of Kidney Diseases. 2011;57(3):466–475. doi: 10.1053/j.ajkd.2010.10.054. [DOI] [PubMed] [Google Scholar]
- 8.Magott-Procelewska M., Boratynska M., Janczak D., et al. Estimated glomerular filtration rate evolution between 6 and 24 months predicts long-term kidney transplant survival among patients with inferior graft function. Transplantation Proceedings. 2009;41(8):3028–3032. doi: 10.1016/j.transproceed.2009.07.105. [DOI] [PubMed] [Google Scholar]
- 9.Wu J., Li H., Huang H., et al. Slope of changes in renal function in the first year post-transplantation and one-yr estimated glomerular filtration rate together predict long-term renal allograft survival. Clinical Transplantation. 2010;24(6):862–868. doi: 10.1111/j.1399-0012.2009.01186.x. [DOI] [PubMed] [Google Scholar]
- 10.Hariharan S., McBride M. A., Cherikh W. S., Tolleris C. B., Bresnahan B. A., Johnson C. P. Post-transplant renal function in the first year predicts long-term kidney transplant survival. Kidney International. 2002;62(1):311–318. doi: 10.1046/j.1523-1755.2002.00424.x. [DOI] [PubMed] [Google Scholar]
- 11.Pascual J., Pérez-Sáez M. J., Mir M., Crespo M. Chronic renal allograft injury: early detection, accurate diagnosis and management. Transplantation Reviews. 2012;26(4):280–290. doi: 10.1016/j.trre.2012.07.002. [DOI] [PubMed] [Google Scholar]
- 12.KDIGO. KDIGO 2012 clinical practice guideline for the evaluation and management of chronic kidney disease. Kidney International Supplements. 2013;3(1):1–150. doi: 10.1038/ki.2013.243. [DOI] [PubMed] [Google Scholar]
- 13.Stegall M. D., Gaston R. S., Cosio F. G., Matas A. Through a glass darkly: seeking clarity in preventing late kidney transplant failure. Journal of the American Society of Nephrology. 2015;26(1):20–29. doi: 10.1681/asn.2014040378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Gera M., Slezak J. M., Rule A. D., Larson T. S., Stegall M. D., Cosio F. G. Assessment of changes in kidney allograft function using creatinine-based estimates of glomerular filtration rate. American Journal of Transplantation. 2007;7(4):880–887. doi: 10.1111/j.1600-6143.2006.01690.x. [DOI] [PubMed] [Google Scholar]
- 15.Herrero-Morín J. D., Málaga S., Fernández N., et al. Cystatin C and beta2-microglobulin: markers of glomerular filtration in critically ill children. Critical Care. 2007;11(3, article R59) doi: 10.1186/cc5923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Donadio C. Serum and urinary markers of early impairment of GFR in chronic kidney disease patients: diagnostic accuracy of urinary β-trace protein. The American Journal of Physiology—Renal Physiology. 2010;299(6):F1407–F1423. doi: 10.1152/ajprenal.00507.2009. [DOI] [PubMed] [Google Scholar]
- 17.Bianchi C., Donadio C., Tramonti G., Consani C., Lorusso P., Rossi G. Reappraisal of serum β2-microglobulin as marker of GFR. Renal Failure. 2001;23(3-4):419–429. doi: 10.1081/jdi-100104725. [DOI] [PubMed] [Google Scholar]
- 18.Donadio C., Lucchesi A., Ardini M., Giordani R. Cystatin C, β2-microglobulin, and retinol-binding protein as indicators of glomerular filtration rate: comparison with plasma creatinine. Journal of Pharmaceutical and Biomedical Analysis. 2001;24(5-6):835–842. doi: 10.1016/s0731-7085(00)00550-1. [DOI] [PubMed] [Google Scholar]
- 19.Jovanović D., Krstivojević P., Obradović I., Durdević V., Dukanović L. Serum cystatin C and β2-microglobulin as markers of glomerular filtration rate. Renal Failure. 2003;25(1):123–133. doi: 10.1081/JDI-120017475. [DOI] [PubMed] [Google Scholar]
- 20.Woitas R. P., Stoffel-Wagner B., Poege U., Schiedermaier P., Spengler U., Sauerbruch T. Low-molecular weight proteins as markers for glomerular filtration rate. Clinical Chemistry. 2001;47(12):2179–2180. [PubMed] [Google Scholar]
- 21.Ikezumi Y., Uemura O., Nagai T., et al. Beta-2 microglobulin-based equation for estimating glomerular filtration rates in Japanese children and adolescents. Clinical and Experimental Nephrology. 2015;19(3):450–457. doi: 10.1007/s10157-014-1015-9. [DOI] [PubMed] [Google Scholar]
- 22.Pertovaara M., Korpela M. ESSPRI and other patient-reported indices in patients with primary Sjögren's syndrome during 100 consecutive outpatient visits at one rheumatological clinic. Rheumatology. 2014;53(5):927–931. doi: 10.1093/rheumatology/ket476.ket476 [DOI] [PubMed] [Google Scholar]
- 23.Carvalho Matos A. C., Durão M. S., Jr., Pacheco-Silva A. Serial beta-2 microglobulin measurement as an auxilliary method in the early diagnosis of cytomegalovirus infection in renal transplant patients. Transplantation Proceedings. 2004;36(4):894–895. doi: 10.1016/j.transproceed.2004.03.110. [DOI] [PubMed] [Google Scholar]
- 24.Bökenkamp A., Grabensee A., Stoffel-Wagner B., et al. The beta2-microglobulin/cystatin C ratio—a potential marker of post-transplant lymphoproliferative disease. Clinical Nephrology. 2002;58(6):417–422. [PubMed] [Google Scholar]
- 25.Vassilakopoulos T. P., Nadali G., Angelopoulou M. K., et al. The prognostic significance of beta(2)-microglobulin in patients with Hodgkin's lymphoma. Haematologica. 2002;87(7):701–708. [PubMed] [Google Scholar]
- 26.Risch L., Blumberg A., Huber A. R. Assessment of renal function in renal transplant patients using cystatin C. A comparison to other renal function markers and estimates. Renal Failure. 2001;23(3-4):439–448. doi: 10.1081/jdi-100104727. [DOI] [PubMed] [Google Scholar]
- 27.Akbas S. H., Yavuz A., Tuncer M., et al. Serum cystatin C as an index of renal function in kidney transplant patients. Transplantation Proceedings. 2004;36(1):99–101. doi: 10.1016/j.transproceed.2003.11.024. [DOI] [PubMed] [Google Scholar]
- 28.Sonkar G. K., Usha, Singh R. G. A preliminary study on the significant value of beta-2-microglobulin over serum creatinine in renal transplant rejection and renal failure. Singapore Medical Journal. 2008;49(10):786–789. [PubMed] [Google Scholar]
- 29.Astor B. C., Muth B., Kaufman D. B., Pirsch J. D., Hofmann R. M., Djamali A. Serum β 2-microglobulin at discharge predicts mortality and graft loss following kidney transplantation. Kidney International. 2013;84(4):810–817. doi: 10.1038/ki.2013.172. [DOI] [PubMed] [Google Scholar]
- 30.Levey A., Greene T., Kusek J., Beck G. A simplified equation to predict glomerular filtration rate from serum creatinine. Journal of the American Society of Nephrology. 2000;11(supplement 2):p. 155. [Google Scholar]
- 31.Astor B. C., Shafi T., Hoogeveen R. C., et al. Novel markers of kidney function as predictors of ESRD, cardiovascular disease, and mortality in the general population. American Journal of Kidney Diseases. 2012;59(5):653–662. doi: 10.1053/j.ajkd.2011.11.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Juraschek S. P., Coresh J., Inker L. A., et al. Comparison of serum concentrations of beta-trace protein, beta2-microglobulin, cystatin C, and creatinine in the US population. Clinical Journal of the American Society of Nephrology. 2013;8(4):584–592. doi: 10.2215/cjn.08700812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Woo J., Floyd M., Cannon D. C. Albumin and β2-microglobulin radioimmunoassays applied to monitoring of renal-allograft function and in differentiating glomerular and tubular diseases. Clinical Chemistry. 1981;27(5):709–713. [PubMed] [Google Scholar]
- 34.Freue G. V. C., Sasaki M., Meredith A., et al. Proteomic signatures in plasma during early acute renal allograft rejection. Molecular & Cellular Proteomics. 2010;9(9):1954–1967. doi: 10.1074/mcp.m110.000554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Backman L., Ringden O., Bjorkhem I., Lindback B. Increased serum β2 microglobulin during rejection, cyclosporine-induced nephrotoxicity, and cytomegalovirus infection in renal transplant recipients. Transplantation. 1986;42(4):368–371. doi: 10.1097/00007890-198610000-00007. [DOI] [PubMed] [Google Scholar]
- 36.Sijpkens Y., Zwinderman A., Mallat M., Boom H., De Fijter H., Paul L. Intercept and slope analysis of risk factors in chronic renal allograft nephropathy. Graft. 2002;5(2):108–113. doi: 10.1177/1522162802005002010. [DOI] [Google Scholar]
- 37.Chapman J. R. Longitudinal analysis of chronic allograft nephropathy: clinicopathologic correlations. Kidney International. 2005;68(supplement 99):S108–S112. doi: 10.1111/j.1523-1755.2005.09920.x. [DOI] [PubMed] [Google Scholar]
- 38.Gourishankar S., Hunsicker L. G., Jhangri G. S., Cockfield S. M., Halloran P. F. The stability of the glomerular filtration rate after renal transplantation is improving. Journal of the American Society of Nephrology. 2003;14(9):2387–2394. doi: 10.1097/01.asn.0000085019.95339.f0. [DOI] [PubMed] [Google Scholar]
- 39.Gill J. S., Tonelli M., Mix C. H., Pereira B. J. G. The change in allograft function among long-term kidney transplant recipients. Journal of the American Society of Nephrology. 2003;14(6):1636–1642. doi: 10.1097/01.ASN.0000070621.06264.86. [DOI] [PubMed] [Google Scholar]
- 40.Marcén R., Morales J. M., Fernández-Rodriguez A., et al. Long-term graft function changes in kidney transplant recipients. Nephrology, Dialysis, Transplantation Plus. 2010;3(supplement 2):ii2–ii8. doi: 10.1093/ndtplus/sfq063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Halloran P. F., de Freitas D. G., Einecke G., et al. An integrated view of molecular changes, histopathology and outcomes in kidney transplants. American Journal of Transplantation. 2010;10(10):2223–2230. doi: 10.1111/j.1600-6143.2010.03268.x. [DOI] [PubMed] [Google Scholar]
- 42.Kasiske B. L., Gaston R. S., Gourishankar S., et al. Long-term deterioration of kidney allograft function. American Journal of Transplantation. 2005;5(6):1405–1414. doi: 10.1111/j.1600-6143.2005.00853.x. [DOI] [PubMed] [Google Scholar]
- 43.Meier-Kriesche H.-U., Schold J. D., Kaplan B. Long-term renal allograft survival: have we made significant progress or is it time to rethink our analytic and therapeutic strategies? American Journal of Transplantation. 2004;4(8):1289–1295. doi: 10.1111/j.1600-6143.2004.00515.x. [DOI] [PubMed] [Google Scholar]


