Abstract
Introduction
Scalable self-management interventions are necessary to address suboptimal diabetes control, especially among minority populations. The study tested the effectiveness of a telephone behavioral intervention in improving glycemic control among adults with diabetes in the New York City A1c Registry.
Design
RCT comparing a telephone intervention to print-only intervention in the context of the A1c Registry program.
Setting/participants
Nine hundred forty-one adults with diabetes and hemoglobin A1c (A1c) >7% from a low-income, predominantly Latino population in the South Bronx were recruited from the A1c Registry.
Intervention
All study participants were mailed print diabetes self-management materials at baseline and modest lifestyle incentives quarterly. Only the telephone participants received four calls from health educators evenly spaced over 1 year if baseline A1c was >7%–9%, or eight calls if baseline A1c was >9%. Medication adherence was the main behavioral focus and, secondarily, nutrition and exercise.
Main outcome measures
Primary outcome was difference between two study arms in change in A1c from baseline to 1 year. Secondary outcomes included diabetes self-care activities, including self-reported medication adherence. Data were collected in 2008–2012 and analyzed in 2012–2014.
Results
Participants were predominantly Latino (67.7%) or non-Latino black (28%), with 69.7% foreign-born and 55.1% Spanish-speaking. Among 694 (74%) participants with follow-up A1c, mean A1c decreased by 0.9 (SD=0.1) among the telephone group compared with 0.5 (SD=0.1) among the print-only group, a difference of 0.4 (95% CI=0.09, 0.74, p=0.01). The intervention had significant effect when baseline A1c was >9%. Both groups experienced similar improvements in self-care activities, medication adherence, and intensification.
Conclusions
A telephone intervention delivered by health educators can be a clinically effective tool to improve diabetes control in diverse populations, specifically for those with worse metabolic control identified using a registry. This public health approach could be adopted by health systems supported by electronic record capabilities.
ClinicalTrials.gov Registration
Introduction
The prevalence of diabetes in New York City (NYC) increased from 4.2% in the early 1990s to 9.2% in 2004.1 Faced with increasing diabetes prevalence and suboptimal control of diabetes,2,3 the NYC Department of Health and Mental Hygiene (DOHMH) expanded existing community programs and policy approaches to diabetes prevention and control by creating an A1c Registry (Registry). The Registry was created in 2006 following an amendment to the NYC Health Code mandating laboratories to report hemoglobin A1c (A1c) test results for NYC residents to the DOHMH. Using information in the Registry, the DOHMH planned to follow trends in glycemic control while providing broad-reaching, low-intensity Registry services to providers and patients across the city to support diabetes management.4 These services consisted of: quarterly mailing of reports to healthcare facilities and providers containing a summary of glycemic control in their population compared with a citywide benchmark, a list of their patients stratified by A1c level for panel management, and motivational letters mailed to patients with high A1c values or who were overdue for an A1c test. These free services were initially offered to providers serving the South Bronx because of the high prevalence of diabetes in this predominantly Latino and lower-income neighborhood. The program was expanded over time, ultimately reaching up to 150 healthcare facilities serving approximately 20% of residents with diabetes.
Although provider performance feedback, panel management, and patient reminders such as those provided in the Registry program are components of effective quality improvement interventions,5-7 self-management support is an aspect of diabetes management that is essential in any environment.8 Previous research3,9,10 has found that self-management support delivered in person or by telephone can improve diabetes outcomes, and may be delivered effectively by clinical11,12 or community13-16 providers. Telephone interventions are particularly appealing as they are relatively low cost, easily accommodated around individuals’ schedules, and may ameliorate health literacy challenges of print interventions. Given DOHMH’s interest in high-need South Bronx residents and under-representation of low-income Latino and black populations in RCTs, identification of a low-cost, community-based intervention to complement existing clinical management of diabetes and the DOHMH’s Registry program was important. We designed a study, “Bronx A1c,” to test the effectiveness of a tiered, tailored telephone intervention to improve glycemic control in adults with diabetes identified through the Registry. This report describes the main results of this study.
Methods
As previously described,17 eligible study participants were non-pregnant adults with type 1 or type 2 diabetes, aged >18 years, who had a recent A1c test >7.0%, had not opted out of receiving communications from the Registry, and lived in one of the ten ZIP codes of the South Bronx. The estimated number of people with diabetes living in the South Bronx at the start of recruitment was 35,000.18 Between September 2008 and October 2010, adults with an A1c test result >7.0% in the previous 3–6 weeks were contacted by telephone using recruitment lists generated by DOHMH Registry staff. Eligibility was confirmed by a self-reported diagnosis of diabetes. Those with plans to move from NYC within 12 months, inability to read or speak in English or Spanish, evidence of cognitive dysfunction, history of or intention to have bariatric surgery, or women who reported only having diabetes during pregnancy were excluded. Oral informed consent was documented, baseline surveys were completed, and participants were randomly assigned into the telephone (Tele/Pr) or print-only (PrO) intervention arm. Participants were randomized by opening a sealed opaque envelope containing a computer-generated sequence of random assignments. The sample size was chosen to provide at least 80% power to detect a 0.3 percentage point difference in mean decrease in A1c between arms using a two-tailed z-test at the 0.05 significance level, assuming an A1c SD of 1.6 percentage points and an intra-class correlation of 0.59 (Appendix S1 and S2). IRBs at Albert Einstein College of Medicine and the DOHMH approved study protocol and oral informed consent documents in English and Spanish. It was registered with ClinicalTrials.gov as NCT00797888 (clinicaltrials.gov/show/NCT00797888).
Interventions
In the environment of Registry services and following randomization, all study participants were mailed a “welcome” packet that included low-literacy print diabetes self-management materials. Retention incentives to promote healthy choices (e.g., pedometers to motivate walking) with their instructional materials were mailed every 3 months to all participants. All participants received a call at 6 months to complete mid-study surveys and a call at 12 months to complete post-intervention behavioral/psychosocial surveys. The control condition was called the PrO arm; they received print self-management materials and did not receive the intervention telephone calls.
In addition to the control condition, the Tele/Pr arm participants were assigned a health educator and could receive four phone calls, generally every 3 months, if their baseline A1c was in the >7.0% and ≤9.0% tier or eight phone calls if baseline A1c was in the >9.0% tier. In a previous telephone intervention study among individuals with a less optimal baseline A1c >7.5%, Walker et al.10 showed that six phone calls in 1 year was the minimum amount associated with a significant A1c decrease, compared with the control arm, and more than six phone calls did not add benefit. Because our glycemic threshold for inclusion in Bronx A1c was lower (i.e., better glycemic control) than that previous study (A1c >7.0% versus >7.5%), we created two A1c tiers for intervention. We speculated that participants with a lower A1c may not need as much support to decrease their A1c as those in the higher tier (A1c >9.0%), potentially minimizing the cost of the intervention. Tier assignment to the protocol was based on baseline A1c and did not change during the intervention year.
During these calls, bilingual health educators, who received at least 20 hours of training in delivering behavioral counseling by telephone, provided self-management support in the participant’s preferred language. These health educators used theory-based techniques such as problem solving19 and goal setting to increase self-efficacy20 in order to promote participant discussion of appropriate behavioral changes to improve their diabetes control. Medication adherence to their diabetes medications was addressed with participant in the Tele/Pr arm. Then, depending on participant preferences, health educators also tailored conversations to explore healthy eating and physical activity topics. During the scheduled phone calls, the Tele/Pr arm participants additionally received behavioral activation for all mailed items (e.g., prompting use of pedometer or to use a 7-day pill box). A multidisciplinary team (nurse certified diabetes educator, internal medicine physician, clinical health psychologist) shared supervision of health educators, primarily through weekly case management meetings.
Primary Outcome Data
The primary outcome for the study was change in A1c from baseline to post-intervention (1 year). For all participants, the baseline A1c was from the A1c Registry. Given the episodic nature of healthcare utilization, we defined the post-intervention primary outcome window as the time period from 6 weeks before to 4 months after the end of protocol anniversary (i.e., 12 months after randomization). For a few participants for whom a Registry test was not available, A1c test values within the primary outcome window were obtained from their healthcare provider with participant written permission.
Demographic and clinical characteristics (Table 1) collected at baseline included: race/ethnicity, country of origin, household income, marital status, educational attainment, insurance status, and BMI.17 Foreign-born was defined as being born outside of the U.S., not including Puerto Rico. Secondary outcome measures collected by self-report at baseline and at 1-year follow-up included: types of diabetes medications prescribed, medication adherence (Morisky Medication Adherence four-item scale21), diabetes self-care activities (Summary of Diabetes Self-Care Activities [SDSCA]22), depression screener (Patient Health Questionnaire, eight-item scale [PHQ-8]23,24), and the five-item general Well-Being scale of the WHO [WHO-5].25 Post-intervention data were collected by trained staff other than assigned intervention health educators. Diabetes medication regimen, self-reported by participants, was categorized as: pills only, insulin only, pills plus insulin, pills plus other injectable, or no medication. Participants were considered to have a change in diabetes medication regimen (yes/no) if they changed categories between baseline and follow-up. Additionally, change in intensity of the medication regimen was assessed by creating a three-level ordinal classification of diabetes medication regimen intensity (no medication, pills only or pills plus other non-insulin injectable, and insulin [with or without pills]) and counting the number of ranks the regimen intensity changed between baseline and end of study (potential range, −2 to +2).
Table 1.
Demographic and Baseline Participant Characteristics by Randomized Study Arm and Total
| Characteristic | Study arms |
Total (N=941) | |
|---|---|---|---|
| Telephone/Print (n=443) | Print only (n=498) |
||
| Age (years, mean ± s.d.) | 56.7 ± 11.3 | 56.0 ± 12.0 | 56.3 ± 11.7 |
| Female gender, % (n) | 64.8 (287) | 62.7 (312) | 63.7 (599) |
| Ethnicity/Race, % (n) | |||
| Latino | 66.1 (293) | 69.1 (344) | 67.7 (637) |
| Black (non-Latino) | 29.8 (132) | 26.3 (131) | 28.0 (263) |
| White (non-Latino) | 0.9 (4) | 1.0 (5) | 1.0 (9) |
| All other | 3.2 (14) | 3.6 (18) | 3.4 (32) |
| Foreign borna % (n) | 69.5 (308) | 69.9 (348) | 69.7 (656) |
| Spanish preferred, % (n) | 55.1 (244) | 56.4 (281) | 55.8 (525) |
| Marital status (%) | |||
| Married/Cohabitating | 35.2 (156) | 38.0 (189) | 36.7 (345) |
| Widowed | 11.5 (51) | 8.4 (42) | 9.9 (93) |
| Separated/Divorced | 24.4 (108) | 21.7 (108) | 23.0 (216) |
| Never married/Single | 28.9 (128) | 31.9 (159) | 30.5 (287) |
| Working status, % (n) | |||
| Works full time | 14.1 (62) | 18.4 (91) | 16.3 (153) |
| Works part time | 10.7 (47) | 6.1 (30) | 8.2 (77) |
| Unemployed | 22.7 (100) | 26.8 (133) | 24.9 (233) |
| Retired | 19.1 (84) | 13.9 (69) | 16.3 (153) |
| Disabled | 26.5 (117) | 30.0 (149) | 28.4 (266) |
| Other | 7.0 (31) | 4.8 (24) | 5.9 (55) |
| Household income, % (n) | |||
| <$20,000 | 78.0 (287) | 75.4 (316) | 76.6 (603) |
| $20-29,000 | 12.2 (45) | 11.0 (46) | 11.6 (91) |
| $30-39,000 | 4.6 (17) | 5.7 (24) | 5.2 (41) |
| $40-49,000 | 3.0 (11) | 4.5 (19) | 3.8 (30) |
| $50,000 + | 2.2 (8) | 3.3 (14) | 2.8 (22) |
| Education, % (n) | |||
| 8th Grade or less | 31.2 (138) | 31.4 (156) | 31.3 (294) |
| 9th–11th Grade | 19.0 (84) | 20.9 (104) | 20.0 (188) |
| Completed H.S. or GED | 26.0 (115) | 25.0 (124) | 25.4 (239) |
| Some college or beyond | 23.9 (106) | 22.7 (113) | 23.3 (219) |
| Any health care coverage, % (n) |
89.8 (387) | 91.6 (447) | 90.8 (834) |
| Reports diabetes education program, % (n) |
11.5 (51) | 16.1 (80) | 13.9 (131) |
| Years since diabetes diagnosis, (%) |
|||
| ≤5 | 33.6 (149) | 33.3 (166) | 33.5 (315) |
| 6-10 | 21.4 (95) | 21.7 (108) | 21.6 (203) |
| >10 | 44.9 (199) | 45.0 (224) | 45.0 (423) |
| HbA1c (mean ± s.d.) | 9.3 ± 2.1 | 9.1 ± 2.0 | 9.2 ± 2.0 |
| BMI (kg/m2, mean ± s.d.) | 32.3 ± 7.8 | 32.0 ± 7.5 | 32.1 ± 7.6 |
Note: Table 1 has been adapted with permission from reference 17, H.S., high school; GED, high school equivalency
Foreign born does not include those born in Puerto Rico.
Statistical Analysis
In all analyses presented here, study arm was based on intention to treat. The primary analysis and all other analyses examining the effect of the telephone intervention on variables measured at baseline and post-intervention used a random effects regression model, with indicators for study arm and time (baseline/post-intervention) and their interaction as predictors, and a random intercept at the person level. Model parameters were estimated by maximum likelihood. The coefficient of study arm X time interaction term is an estimator of the between-arm difference in within-person A1c change. It quantifies the intervention effect, and its significance is determined with a z-test.
Missing outcome data were handled in several ways (Appendix S3). For subjects who did not have an A1c test during the defined end of study window, we analyzed A1c test results before or after that window. A robustness analysis using this expanded definition of study outcome was carried out. Additional robustness analyses imputing the best or worst A1c available in the Registry for patients lacking an A1c within the defined end of study window were performed. Finally, multiple imputation using chained equations, basing imputation of outcome A1c on age, BMI, and baseline A1c, was carried out.
Analyses of modification of the intervention effect by demographic or psychosocial variables were conducted by including the corresponding covariate(s) and their interactions with the study arm, time, and arm X time interaction terms. Effect modification was considered detected if the coefficient of the covariate X study arm X time interaction(s) were (jointly) statistically significant. Analyses of mediation of the intervention effect by behavioral variables were carried out using path analysis and the Sobel test.26 Mediation by each behavior was considered detected if the product of path coefficients along the indirect path(s) from study arm to outcome through the behavioral variable(s) was statistically significant. The study design was not powered to detect effect modification or mediation effects, so these analyses are exploratory. Change in medication regimen intensity between study arms was contrasted using the Mann–Whitney U test. Data management and analyses were carried out using Stata, version 12.1MP.
Results
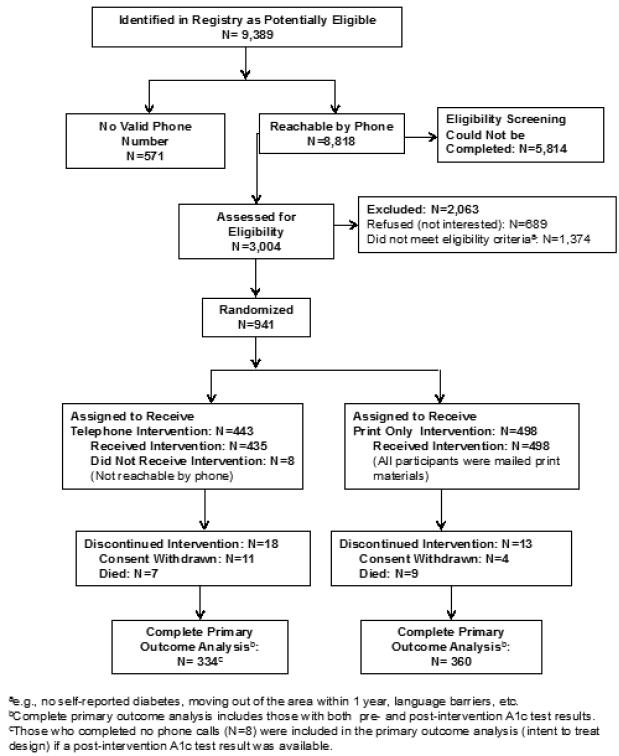
A total of 941 adults with diabetes were randomized, 31 of whom either died or withdrew informed consent (Figure 1). Study participants were: 67.7% Latino and 28.0% non-Latino black, with 69.7% being foreign-born (86.5% of Latinos and 31.5% of non-Latino blacks) and 55.1% Spanish-speaking; mean age was 56.3 years. The majority (77%) had an annual household income of <$20,000, and 91% of participants reported at least some healthcare coverage. There were no substantial differences at baseline in demographic characteristics between the two study arms17 (Table 1). Participants’ baseline A1c tests were ordered from 48 healthcare facilities. More than half (54.2%, n=26) of these facilities were receiving A1c Registry facility-level or provider-level reports at study start (September 2008) and 79.2% (n=38) were receiving the Registry patient letter service by September 2009. All data were collected between 2008 and 2012 and analyzed in 2012–2014.
Figure 1.
CONSORT diagram.
Randomization assigned 443 to the Tele/Pr arm and 498 participants to the PrO arm. Mean baseline and available post-intervention A1c test results for all participants randomized to each arm and in each A1c tier are shown in Table 2. End of study A1c test results within the primary outcome time window were available for 334 in Tele/Pr arm and 360 in PrO arm (Figure 1), with only six and three results, respectively, obtained from non-Registry medical sources. The difference in change in A1c between the two arms estimated by random effects regression was 0.4 percentage points (95% CI=0.09, 0.74 percentage points, p=0.01). There was no evidence of effect modification by gender, language preference, race, ethnicity, income, educational attainment, or marital status. In addition to the decrease in the mean A1c being greater in the Tele/Pr arm, the percentage of participants in the Tele/Pr arm who had at least a 1% or 1.5% A1c decrease at end of protocol was 37.4% and 26.7%, respectively, compared with the PrO arm with 28.3% having a 1% decrease (p=0.01) and 19.2% with a 1.5% decrease (p=0.02) in A1c.
Table 2.
Mean A1c at Baseline and Post Intervention for Overall and Each A1c Tier by Study Arm
| Study arm | Overall | >7.0-9.0% A1c Tier | >9.0% A1c Tier | |||
|---|---|---|---|---|---|---|
|
| ||||||
| Baseline | Post | Baseline | Post | Baseline | Post | |
| Tele/Pr | N=443 | n=334 | n=26l | n=206 | n=182 | n=125 |
| A1c (%) | 9.3±2.1 | 8.4±1.9 | 7.9±0.5 | 7.9±1.3 | 11.3±1.9 | 9.2±2.4 |
| PrO | N=498 | n=360 | n=300 | n=229 | n=198 | n=131 |
| A1c (%) | 9.1±2.0 | 8.6±2.0 | 7.8±0.6 | 8.0±1.6 | 11.0±1.90 | 9.7±2.3 |
Note: Boldface indicates statistical significance (p<0.05). Between study arm difference in baseline to post change in A1c statistically significant in overall study sample (p=0.012), and in >9.0% A1c Tier (p=0.003), but not in <7.0-9.0% A1c Tier (p = 0.214). Tele/Pr, Telephone/Print Intervention; PrO, Print Only Intervention
The difference in change in A1c between the two arms was predominantly in the >9.0% A1c tier with a decrease of 2.1% A1c and 1.3% A1c in the Tele/Pr and PrO arms respectively, compared with the ≤9% A1c tier, in which there was no change in the Tele/Pr arm and an increase of 0.2% A1c in the PrO arm (Table 2). The difference in change between the >9.0% tier and ≤9.0% tier of –0.8% versus 0.2% A1c, respectively, was statistically significant (p=0.0005).
The proportion of participants without a follow-up A1c test within the primary outcome window was similar between the two arms, 24.6% for Tele/Pr and 27.7% for PrO (p=0.28). Overall, participants without a follow-up test had a higher baseline A1c compared with those with a follow-up test (9.6% vs 9.1% A1c, p=0.001), and were younger, less medication adherent, and more likely to be widowed, separated, or divorced (data not shown). When we expanded the outcome window to include any A1c test result 6 months before and 1 year after the 1-year randomization anniversary (Appendix S3), we found that the decrease in A1c of 1.1% among the Tele/Pr arm participants who were missing the primary outcome but had an expanded window test result was greater than that in the Tele/Pr arm with primary outcome data (0.7%). This A1c difference was not seen in the PrO arm (0.3% vs 0.4%, respectively). Additionally, the relationship between baseline A1c and a missing follow-up test was stronger in the Tele/Pr arm, such that Tele/Pr participants were more likely to have a follow-up test if their baseline A1c was lower. These findings suggest that our primary analysis is conservative.
Robustness analyses using multiple imputations and assorted single imputations (wider outcome window, last observation carried forward, best observation carried forward, and worst observation carried forward) all led to estimates of telephone intervention effect similar to the findings of the primary analysis and with similar levels of statistical significance. Details are provided in Appendix S3 for Tables 1–3.
Table 3.
Changes from Baseline to Post-Intervention in BMI, Medication Regimens, Behavioral and Psychosocial Measures
| MEASURES | Telephone/Print arm N=443 |
Print only arm N=498 |
||
|---|---|---|---|---|
|
| ||||
| BASELINE | POST | BASELINE | POST | |
| BMI (kg/m2) | 32.3 ± 7.8 (n=431) |
32.2 ± 6.7 (n=349) |
32.0 ± 7.5 (n=493) |
31.9 ± 7.1 (n=389) |
|
| ||||
| DIABETES MEDICATION REGIMENS, n (%) | ||||
|
| ||||
| Diabetes pills only | 212 (47.9) | 153 (34.6) | 242 (48.6) | 189 (38.0) |
| Insulin only | 71 (16.0) | 74 (16.7) | 83 (16.7) | 82 (16.5) |
| Pills and insulin | 113 (25.5) | 111 (25.1) | 112 (22.5) | 98 (19.7) |
| Pills and other injectable | 44 (9.9) | 9 (2.0) | 53 (10.6) | 7 (1.4) |
| None | 3 (0.7) | 9 (2.0) | 5 (1.0) | 18 (3.6) |
| Unknowna | 0 (0.0) | 87 (19.7) | 3 (0.6) | 104 (20.9) |
|
| ||||
| BEHAVIORAL AND PSYCHOSOCIAL MEASURES | ||||
|
| ||||
| Morisky scoreb, mean ± s.d. | 3.1 ± 1.1 (n=442) |
3.1 ± 1.1 (n=366) |
3.1 ± 1.1 (n=493) |
3.2 ± 1.1 (n=404) |
| Morisky Scoreb = 4, n (%) | 225 (50.9) | 190 (51.9) | 235 (47.7) | 209 (51.7) |
|
| ||||
| Diabetes Self-Care (# days per week) c | ||||
|
| ||||
| General diet, mean ± s.d.** |
3.7 ± 2.6
(n=442) |
4.5 ± 2.5
(n=357) |
3.7 ± 2.7
(n=497) |
4.4 ± 2.4
(n=400) |
| Specific diet, mean ± s.d. ** |
4.1 ± 1.9
(n=441) |
4.5 ± 1.8
(n=360) |
4.1 ± 1.9
(n=497) |
4.4 ± 1.7
(n=400) |
| Exercise, mean ± s.d. ** |
2.6 ± 2.3
(n=442) |
3.2 ± 2.6
(n=360) |
2.6 ± 2.3
(n=498) |
3.3 ± 2.4
(n=398) |
| Blood Glucose tests, mean ± s.d. | 4.6 ± 2.8 (n=438) |
4.8 ± 2.6 (n=360) |
4.5 ± 2.9 (n=497) |
4.7 ± 2.6 (n=396) |
|
| ||||
| TV watching, n (%) | ||||
|
| ||||
| None | 8 (1.8) | 10 (2.7) | 10 (2.0) | 11 (2.7) |
| <2 hours/day | 119 (26.9) | 97 (26.5) | 136 (27.3) | 120 (29.4) |
| 2-4 hours/day | 119 (26.9) | 102 (27.9) | 148 (29.7) | 125 (30.6) |
| >4 hours/day | 196 (44.3) | 204 (42.9) | 204 (41.0) | 152 (37.3) |
|
| ||||
| PHQ-8 score, mean ± s.d. ** |
6.3 ± 5.4
(N=443) |
5.7 ± 5.7
(n=363) |
6.5 ± 5.5
(N=498) |
5.8 ± 5.3
(n=400) |
|
| ||||
| Well-being, mean ± s.d.* |
15.3 ± 6.7
(N=443) |
16.0 ± 6.9
(n=366) |
15.0 ± 6.7
(N=498) |
15.9 ± 6.6
(n=406) |
Note: Boldface indicates statistically significant contrasts between baseline and post-intervention values in both study arms
p<0.01;
p<0.005
The “unknown” category denotes a response of “I don’t know” at Baseline; at Post Intervention, the vast majority of the “unknown” (n=191) are participants who did not complete the surveys.
Morisky Medication Adherence survey with possible range 0-4, with 4 being most adherent to medication.
Data from the Summary of Diabetes Self Care Activities (SDSCA) survey.
Among the 774 participants (82.2%) who completed both baseline and post-intervention surveys, there were no significant group X time effects among the measured behavioral and psychosocial outcomes. Specifically, there were no significant changes in either study arm in measures of medication adherence, amount of TV watching, or blood glucose monitoring. Depression and well-being improved significantly in both study arms, as did some measures of dietary behaviors and exercise frequency (Table 3).
Among the 938 participants with self-reported medication information at baseline, 930 reported taking diabetes medication at the start of the study. Baseline diabetes medication regimen was not associated with change in A1c over the course of the study in either arm (p=0.61) and the effect of the intervention did not differ by baseline medication regimen (p=0.24). Among the 751 participants for whom we had diabetes medication information at baseline and follow-up, 30.2% had a change in their medication regimen over the course of the study (Appendix S4). This rate did not differ significantly between the two study arms (29.2% in Tele/Pr vs 31.1% in PrO, p=0.57), even among the >9.0% tier (32.7% in Tele/Pr vs 36.7% in PrO, p=0.47). Rate of change did differ significantly by A1c tier, irrespective of study arm (27.3% in >7%–9% tier vs 34.7% in >9% tier, p=0.03).
Although diabetes medication treatment intensity did not change for the majority (79%) of study participants, 7.6% of participants ended the study on a less intense medical regimen than they started and 13.4% ended on a more intense medical regimen than they started. These overall proportions did not differ significantly between study arms (p=0.298) or A1c groups (p=0.54). (Appendix S4, Tables 4–6).
Over the course of this study, the Tele/Pr participants engaged in a mean of 4.6 intervention phone calls over 12 months; this was a mean of 3.4 calls for those in the >7%–9% A1c tier (protocol maximum was four calls) and a mean of 6.3 calls completed for those in the >9% A1c tier (protocol maximum was eight calls). The total duration of intervention calls in the Tele/Pr arm averaged 109.8 minutes over 12 months and increased in relation to baseline A1c by study protocol. The average total minutes for those with baseline A1c <9% was 85.5 minutes, and for those with a baseline A1c >9%, it was 144.7 minutes.
Discussion
This report provides the main results of a telephone behavioral intervention to improve diabetes control in a predominately Latino, urban, low-income population living in the South Bronx, New York. The telephone intervention, incorporating print diabetes self-management materials, was associated with a significant decrease of 0.4% A1c more than the print intervention alone. These results compare favorably in terms of A1c lowering to monotherapy with some pharmacologic agents approved to treat type 2 diabetes.27,28 We found that those with higher baseline A1c experienced the greatest improvements in glycemic control, as seen in other studies,29 and a significantly larger proportion of those who were randomized to the Tele/Pr arm achieved decreases in A1c of at least 1% compared with the PrO arm.
We had hypothesized that the telephonic self-management support delivered by health educators, which focused on medication adherence and healthy lifestyle, would result in A1c reduction through improved medication adherence and self-care activities such as healthier eating and increased exercise. However, among our measures of secondary outcomes, there were no mediators of glycemic changes identified from the behavioral or psychosocial surveys we used. Paradoxically, we saw no change in measures of medication adherence self-reported by participants. This may have been related to the fact that participants on average reported high medication adherence at baseline, even though their A1c levels were poor on average (mean baseline A1c, 9.3%), leaving little room for observed improvement in this self-reported, four-item measure. By study design, we did not have access to prescription claims data or pill counts, which can be a more reliable measure for medication adherence than self-report.10,30 Change in prescribed medication regimen could have driven the difference in A1c between the two arms, although we did not find a difference in the proportion of each arm that reported a change in medication regimen or regimen intensity from baseline to post-intervention. Significant improvements in other self-reported measures of diet, exercise, depression, and well-being were self-reported, although these occurred in both arms. Though we did not discern any major factors that explain the A1c improvement in the Tele/Pr arm, it is possible that these widely-used, brief behavioral measures (e.g., Morisky medication adherence scale21 and dietary or physical activity questions from SDSCA22) had limited sensitivity in our population to detect participant-specific behavior changes that may have contributed to our positive findings.31 Another study10 found similar results with these measures. Although a recent study by Treif et al.32 found the SDSCA adherence measure to be a significant predictor of A1c, perhaps other self-care measures, even if longer, should be considered in future research or clinical practice in similar populations.31,33-35
Keeping a scalable intervention as a priority, we designed our study to test the efficacy of a less-intensive telephone intervention for those with a lower A1c and more-intensive intervention with an A1c >9% at baseline. There appears to be no improvement in A1c in either arm for those with a baseline A1c between 7.1% and 9.0%. The improvement was seen in those with an A1c >9.0% at baseline in both arms, with participants receiving the Tele/Pr intervention having a significantly greater improvement. There are at least two potential reasons why we saw no change in A1c in the lower tier. First, a limit of four phone calls may not have been intense enough to promote behavior change for an A1c decline among those who received the Tele/Pr intervention. Second, as clinicians often report, it may be more difficult to lower an A1c that is closer to the standard goal of <7%3,29 because of perceived risk of hypoglycemia by patient or provider or more aggressive medical treatment when a baseline A1c is >9%, in partial response to a medical facility’s quality improvement measures. In addition, a pattern of greater decline in a subsample with higher baseline A1c values in both study arms is consistent with the effects of regression to the mean. Regression to the mean cannot, however, account for the differences between study arms.
Our goal was to test an affordable, effective, and scalable public health intervention implemented by a city department of health to improve glycemic control. Our telephone behavioral intervention by non-clinical health educators, including verbal activation of the standard print self-management materials, would usually be characterized as a modest-intensity intervention. Several recent self-management interventions have shown significant improvements in A1c; however, most of these interventions do so with a more labor-intensive approach implemented by interventionists with more advanced training and credentials, and often with face-to-face components.5,14,34,36,37 Presumably, these characteristics would contribute to higher intervention costs. The cost analysis for the Bronx A1c telephone intervention is currently in process.
Limitations
A major limitation of our study is that we were missing primary outcome data for 26.3% of the participants balanced in the two arms. Although participants were encouraged to receive quarterly A1c tests from their healthcare provider to assess diabetes control, this pragmatic telephonic trial was not designed to provide this A1c testing. We relied on participants and their providers to obtain an A1c test at the end of the 1-year intervention. This A1c test value would then be available in the A1c Registry. Additionally, our study population was almost 70% foreign-born and they may have received diabetes care outside the reach of the A1c Registry when visiting their country of origin. Though one quarter of participants were missing outcome data, the sensitivity analyses we conducted suggest that our findings may be conservative, underestimating the effect of the Tele/Pr intervention (Appendix S3). Another limitation to our study is that it is not generalizable to the overall diabetes population; instead, it is generalizable to a predominantly Latino, low-income population with consistent telephone access.
Trained and supervised bilingual health educators from the community16 were key to recruitment and retention of participants and implementation of the protocol. They provided telephonic self-management support using their knowledge of the Bronx socioeconomic–cultural environment for our diverse population of mostly Latino and black participants, knowledge which cannot be easily instilled. Ongoing training and supervision for health educators through case management meetings were crucial for success. We attempted to overcome health disparities in our South Bronx population compared with NYC in general by reaching out by telephone with support from health educators from similar cultural backgrounds.
Conclusions
This study and its findings have important public health implications. There have been significant gains in diabetes management over the past two decades as evidenced by increased prevalence of glycemic, blood pressure, and cholesterol control, and decreased rates of complications.38,39 However, there is still room for improvement in glycemic control, especially in low-income populations. Readily accessible, low-cost programs to support medication adherence and lifestyle behaviors are critical for improving glycemic control across all populations. A tailored telephone intervention such as the program we implemented has the potential for wide-scale use, particularly for those with poor metabolic control (A1c >9%), in the environment of a registry or other population health management system such as a patient-centered medical home. For those with an A1c between 7% and 9%, further research may be needed to understand what would be an effective telephone intervention.
Supplementary Material
Acknowledgments
This study was funded by R18 DK 078077 and partially by P60 DK 020541. We acknowledge the outstanding contributions of: our New York City Department of Health and Mental Hygiene (NYC DOHMH) health educators who contributed to study design and data collection, Giovanna DiFrancesca, BA, Danielle Powell, MPH, and Rosa Rosen, JD; NYC DOHMH Registry staff, Anita Richichi, BA, who contributed grant management, Angela Merges, MA, who supported recruitment activities, and Qun Jiang, MS, for assisting with data management; and at Albert Einstein College of Medicine, research associate Jennifer Lukin, BS, for assistance with data entry and manuscript preparation, and co-investigator Joel Zonszein, MD, for expert diabetes management consultations. All of the study participants gave generously of their time. Parts of this paper were presented at the American Diabetes Association Scientific Sessions, Chicago, IL; June, 21, 2013.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Author LS’s family holds stock in Abbott Laboratories and Abbvie due to spouse's previous employment. No other financial disclosures were reported by the authors of this paper.
References
- 1.Gupta L, Olson C. Epi Data Brief: Diabetes in New York City. Vol. 26. New York City Department of Health and Mental Hygiene: Epi Data Brief; New York: Apr, 2013. [Google Scholar]
- 2.Thorpe LE, Upadhyay UD, Chamany S, et al. Prevalence and control of diabetes and impaired fasting glucose in New York City. Diabetes Care. 2009;32(1):57–62. doi: 10.2337/dc08-0727. http://dx.doi.org/10.2337/dc08-0727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.American-Diabetes-Association Standards of Medical Care in Diabetes - 2013. Diabetes Care. 2013;36(Suppl 1):S11–S66. doi: 10.2337/dc13-S011. http://dx.doi.org/10.2337/dc13-S011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Chamany S, Silver LD, Bassett MT, et al. Tracking diabetes: New York City's A1C Registry. Milbank Q. 2009;87(3):547–570. doi: 10.1111/j.1468-0009.2009.00568.x. http://dx.doi.org/10.1111/j.1468-0009.2009.00568.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Piatt GA, Orchard TJ, Emerson S, et al. Translating the chronic care model into the community: results from a randomized controlled trial of a multifaceted diabetes care intervention. Diabetes Care. 2006;29(4):811–817. doi: 10.2337/diacare.29.04.06.dc05-1785. http://dx.doi.org/10.2337/diacare.29.04.06.dc05-1785. [DOI] [PubMed] [Google Scholar]
- 6.Bodenheimer T, Wagner EH, Grumbach K. Improving primary care for patients with chronic illness. JAMA. 2002;288(14):1775–1779. doi: 10.1001/jama.288.14.1775. http://dx.doi.org/10.1001/jama.288.14.1775. [DOI] [PubMed] [Google Scholar]
- 7.Shojania KG, Ranji SR, McDonald KM, et al. Effects of quality improvement strategies for type 2 diabetes on glycemic control: a meta-regression analysis. JAMA. 2006;296(4):427–440. doi: 10.1001/jama.296.4.427. http://dx.doi.org/10.1001/jama.296.4.427. [DOI] [PubMed] [Google Scholar]
- 8.American-Diabetes-Association Foundations of Care: Education, Nutrition, Physical Activity, Smoking Cessation, Psychosocial Care, and Immunization. Diabetes Care. 2015;38(Supplement 1):S20–S30. doi: 10.2337/dc15-S007. http://dx.doi.org/10.2337/dc15-S007. [DOI] [PubMed] [Google Scholar]
- 9.Duke SA, Colagiuri S, Colagiuri R. Individual patient education for people with type 2 diabetes mellitus. Cochrane Database Syst Rev. 2009;(1):CD005268. doi: 10.1002/14651858.CD005268.pub2. http://dx.doi.org/10.1002/14651858.CD005268.pub2. [DOI] [PMC free article] [PubMed]
- 10.Walker EA, Shmukler C, Ullman R, Blanco E, Scollan-Koliopoulus M, Cohen HW. Results of a successful telephonic intervention to improve diabetes control in urban adults: a randomized trial. Diabetes Care. 2011;34(1):2–7. doi: 10.2337/dc10-1005. http://dx.doi.org/10.2337/dc10-1005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Davidson MB, Ansari A, Karlan VJ. Effect of a nurse-directed diabetes disease management program on urgent care/emergency room visits and hospitalizations in a minority population. Diabetes Care. 2007;30(2):224–227. doi: 10.2337/dc06-2022. http://dx.doi.org/10.2337/dc06-2022. [DOI] [PubMed] [Google Scholar]
- 12.Piette JD, Weinberger M, Kraemer FB, McPhee SJ. Impact of automated calls with nurse follow-up on diabetes treatment outcomes in a Department of Veterans Affairs Health Care System: a randomized controlled trial. Diabetes Care. 2001;24(2):202–208. doi: 10.2337/diacare.24.2.202. http://dx.doi.org/10.2337/diacare.24.2.202. [DOI] [PubMed] [Google Scholar]
- 13.Thom DH, Ghorob A, Hessler D, De Vore D, Chen E, Bodenheimer TA. Impact of peer health coaching on glycemic control in low-income patients with diabetes: a randomized controlled trial. Ann Fam Med. 2013;11(2):137–144. doi: 10.1370/afm.1443. http://dx.doi.org/10.1370/afm.1443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Philis-Tsimikas A, Fortmann A, Lleva-Ocana L, Walker C, Gallo LC. Peer-led diabetes education programs in high-risk Mexican Americans improve glycemic control compared with standard approaches: a Project Dulce promotora randomized trial. Diabetes Care. 2011;34(9):1926–1931. doi: 10.2337/dc10-2081. http://dx.doi.org/10.2337/dc10-2081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Heisler M, Vijan S, Makki F, Piette JD. Diabetes control with reciprocal peer support versus nurse care management: a randomized trial. Ann Intern Med. 2010;153(8):507–515. doi: 10.7326/0003-4819-153-8-201010190-00007. http://dx.doi.org/10.7326/0003-4819-153-8-201010190-00007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Gary TL, Bone LR, Hill MN, et al. Randomized controlled trial of the effects of nurse case manager and community health worker interventions on risk factors for diabetes-related complications in urban African Americans. Prev Med. 2003;37(1):23–32. doi: 10.1016/s0091-7435(03)00040-9. http://dx.doi.org/10.1016/S0091-7435(03)00040-9. [DOI] [PubMed] [Google Scholar]
- 17.Walker EA, Silver L, Chamany S, Schechter CB, Gonzalez JS, Carrasco J, et al. Baseline characteristics and Latino vs. non-Latino contrasts among Bronx A1c study participants. West J Nurs Res. 2014;36(9):1030–1051. doi: 10.1177/0193945913517947. http://dx.doi.org/10.1177/0193945913517947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.DOHMH-NYC Epiquery: NYC Interactive Health Data System - Community Health Survey. October 11, 2012; www.nyc.gov/health/epiquery.
- 19.Hill-Briggs F, Gemmell L. Problem solving in diabetes self-management and control: a systematic review of the literature. Diabetes Educ. 2007;33(6):1032–1050. doi: 10.1177/0145721707308412. http://dx.doi.org/10.1177/0145721707308412. [DOI] [PubMed] [Google Scholar]
- 20.Bandura A. Self-efficacy: The exercise of control. W.H. Freeman; New York: 1997. [Google Scholar]
- 21.Morisky DE, Green LW, Levine DM. Concurrent and predictive validity of a self-reported measure of medication adherence. Med Care. 1986;24(1):67–74. doi: 10.1097/00005650-198601000-00007. http://dx.doi.org/10.1097/00005650-198601000-00007. [DOI] [PubMed] [Google Scholar]
- 22.Toobert DJ, Hampson SE, Glasgow RE. The summary of diabetes self-care activities measure: results from 7 studies and a revised scale. Diabetes Care. 2000;23(7):943–950. doi: 10.2337/diacare.23.7.943. http://dx.doi.org/10.2337/diacare.23.7.943. [DOI] [PubMed] [Google Scholar]
- 23.Kroenke K, Strine TW, Spitzer RL, Williams JB, Berry JT, Mokdad AH. The PHQ-8 as a measure of current depression in the general population. J Affect Disord. 2009;114(1-3):163–173. doi: 10.1016/j.jad.2008.06.026. http://dx.doi.org/10.1016/j.jad.2008.06.026. [DOI] [PubMed] [Google Scholar]
- 24.Kroenke K, Spitzer RL, Williams JB. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med. 2001;16(9):606–613. doi: 10.1046/j.1525-1497.2001.016009606.x. http://dx.doi.org/10.1046/j.1525-1497.2001.016009606.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Bonsignore M, Barkow K, Jessen F, Heun R. Validity of the five-item WHO Well-Being Index (WHO-5) in an elderly population. Eur Arch Psychiatry Clin Neurosci. 2001;251(Suppl 2):II27–31. doi: 10.1007/BF03035123. http://dx.doi.org/10.1007/BF03035123. [DOI] [PubMed] [Google Scholar]
- 26.Sobel ME. Asymptotic confidence intervals for indirect effects in structural equation models. Sociol Methodol. 1982;13:290–312. http://dx.doi.org/10.2307/270723. [Google Scholar]
- 27.Kahn SE, Haffner SM, Heise MA, et al. Glycemic durability of rosiglitazone, metformin, or glyburide monotherapy. N Engl J Med. 2006;355(23):2427–2443. doi: 10.1056/NEJMoa066224. http://dx.doi.org/10.1056/NEJMoa066224. [DOI] [PubMed] [Google Scholar]
- 28.Inzucchi SE, Bergenstal RM, Buse JB, et al. Management of Hyperglycemia in Type 2 Diabetes, 2015: A Patient-Centered Approach. Update to a Position Statement of the American Diabetes Association and the European Association for the Study of Diabetes. Diabetes Care. 2015;38(1):140–149. doi: 10.2337/dc14-2441. http://dx.doi.org/10.2337/dc14-2441. [DOI] [PubMed] [Google Scholar]
- 29.Tricco AC, Ivers NM, Grimshaw JM, et al. Effectiveness of quality improvement strategies on the management of diabetes: a systematic review and meta-analysis. Lancet. 2012;379(9833):2252–2261. doi: 10.1016/S0140-6736(12)60480-2. http://dx.doi.org/10.1016/S0140-6736(12)60480-2. [DOI] [PubMed] [Google Scholar]
- 30.Walker EA, Molitch M, Kramer MK, et al. Adherence to preventive medications: predictors and outcomes in the Diabetes Prevention Program. Diabetes Care. 2006;29(9):1997–2002. doi: 10.2337/dc06-0454. http://dx.doi.org/10.2337/dc06-0454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Gonzalez JS, Schneider HE, Wexler DJ, et al. Validity of medication adherence self-reports in adults with type 2 diabetes. Diabetes Care. 2013;36(4):831–837. doi: 10.2337/dc12-0410. http://dx.doi.org/10.2337/dc12-0410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Trief PM, Izquierdo R, Eimicke JP, et al. Adherence to diabetes self care for white, African-American and Hispanic American telemedicine participants: 5 year results from the IDEATel project. Ethn Health. 2013;18(1):83–96. doi: 10.1080/13557858.2012.700915. http://dx.doi.org/10.1080/13557858.2012.700915. [DOI] [PubMed] [Google Scholar]
- 33.Weinger K, Beverly EA, Lee Y, Sitnokov L, Ganda OP, Caballero AE. The Effect of a Structured Behavioral Intervention on Poorly Controlled Diabetes: A Randomized Controlled Trial. Arch Intern Med. 2011;117:1990–1999. doi: 10.1001/archinternmed.2011.502. http://dx.doi.org/10.1001/archinternmed.2011.502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Safren SA, Gonzalez JS, Wexler DJ, et al. A randomized controlled trial of cognitive behavioral therapy for adherence and depression (CBT-AD) in patients with uncontrolled type 2 diabetes. Diabetes Care. 2014;37(3):625–633. doi: 10.2337/dc13-0816. http://dx.doi.org/10.2337/dc13-0816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Mayberry LS, Osborn CY. Empirical validation of the information-motivation-behavioral skills model of diabetes medication adherence: a framework for intervention. Diabetes Care. 2014;37(5):1246–1253. doi: 10.2337/dc13-1828. http://dx.doi.org/10.2337/dc13-1828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Davis RM, Hitch AD, Salaam MM, Herman WH, Zimmer-Galler IE, Mayer-Davis EJ. TeleHealth improves diabetes self-management in an underserved community: diabetes TeleCare. Diabetes Care. 2010;33(8):1712–1717. doi: 10.2337/dc09-1919. http://dx.doi.org/10.2337/dc09-1919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Weinstock RS, Teresi JA, Goland R, et al. Glycemic control and health disparities in older ethnically diverse underserved adults with diabetes: five-year results from the Informatics for Diabetes Education and Telemedicine (IDEATel) study. Diabetes Care. 2011;34(2):274–279. doi: 10.2337/dc10-1346. http://dx.doi.org/10.2337/dc10-1346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Casagrande SS, Fradkin JE, Saydah SH, Rust KF, Cowie CW. The Prevalence of Meeting A1C, Blood Pressure, and LDL Goals Among People with Diabetes, 1988-2010. Diabetes Care. 2013;36(8):2271–2279. doi: 10.2337/dc12-2258. http://dx.doi.org/10.2337/dc12-2258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Gregg EW, Li Y, Wang J, et al. Changes in Diabetes-Related Complications in the United States, 1990-2010. N Engl J Med. 2014;370(16):1514–1523. doi: 10.1056/NEJMoa1310799. http://dx.doi.org/10.1056/NEJMoa1310799. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.