Abstract
AIM: To investigate whether CTL vaccine therapy is suitable for primary hepatocellular carcinoma (HCC) from the viewpoint of HLA class I antigens expression.
METHODS: The immunocytochemistry, image analysis, flow cytometry, and labeled streptavidin biotin (LSAB) method of immunohistochemistry were applied respectively to study 4 HCC cell lines (e.g. Alexander, HepG2, SMMC-7721, and QGY-7703) cultured in vitro and 6 frozen tissue specimens of HCC.
RESULTS: The positive control cell line Raji had very strong positive staining. Most mitotic and nonmitotic cells of the 4 HCC cell lines had various intensity of HLA class I antigens expression. The negative control cell K562 and the control slides of all the cell lines had no positive staining. In the 6 HCC specimens immunohistochemically studied, histological normal hepatocytes had no or very weak positive staining and the liver sinus had very strong positive staining. Most HCC cells in the sections from the 6 HCC specimens had strong positive HLA class I antigens staining. The positive staining was located in the cytoplasm, the perinuclear area, and at the cell membrane of the liver cancer cells. Flow cytometry also revealed that Raji and those 4 HCC cell lines had strong HLA class I antigens expression, which was confirmed quantitatively by the image analysis. It showed that the objective grayscale values of Raji and those 4 HCC cell lines were significantly different from that of K562 (Raji 114.04 ± 10.94, Alexander 165.97 ± 5.35, HepG2167.02 ± 12.60, QGY-7703161.46 ± 7.13, SMMC-7721165.93 ± 5.21, K562244.89 ± 4.60, P < 0.01). Significant differences were also found between Raji and the 4 HCC cell lines.
CONCLUSION: HCC cells express HLA class I antigens strongly. From this point of view, the active specific immunotherapy of CTL vaccine is suitable and practicable for HCC.
INTRODUCTION
The immunity of cytotoxic T lymphocyte (CTL) is crucial for anti-tumor immune response. Tumor cells expressing MHC class I antigens is a necessary molecular basis and requirement for effective presentation, recognition, and cytotoxicity. Many tumors do decrease or lack MHC class I antigens to escape from immune surveillance. The monitoring of MHC class I antigens expression in tumors, therefore, is undoubtedly helpful to decide which immune strategy is suitable for a given tumor[1]. Therefore, it is necessary to start a preliminary investigation on the HLA class I antigens expression of HCC tissues in vivo and corresponding cell lines cultured in vitro before the design and construction of CTL vaccine against HCC.
MATERIALS AND METHODS
Main reagents
Mouse anti human HLA-ABC mAb, DAKO Corp; FITC labeled mouse anti human HLA-ABC mAb, Beckman Coulter Corp; biotin labeled goat anti mouse IgG and HRP-labeled streptavidin, from Beijing Zhongshan Corp; avidin biotin blocking system, Wuhan Boster Corp.
Cell lines and specimens
The erythromyeloid cell line K562 and the Burkitts’ lymphoma cell line Raji were used as HLA class I antigens negative and positive control cells respectively[2,3]. The human HCC cell lines Alexander[4], HepG2[5], SMMC-7721, and QGY-7703 were all kept and passed by our group. The cells above were maintained in complete PRMI 1640 medium at 37 °C in a incubator with 5% CO2. Fifteen pathological specimens were obtained from surgically resected tissues of patients with HCC in the First Affiliated Hospital of West China University of Medical Sciences. The specimens were frozen immediately in liquid nitrogen. Four μm consecutive frozen sections were prepared and stored at -70 °C. HE Staining was done by the routine method.
Immunocytochemistry and immunohistochemistry
HCC cell lines were grown on the glass slides until confluence and then fixed and washed. Some slides were coated with the fixed and washed Raji and K562 cells. The frozen sections were also fixed and washed. The endogenous peroxidase on the slides and sections were inactivated, and the slides and sections were blocked by normal goat serum. Additionally, the endogenous biotin in the frozen sections was blocked. The slides and sections were incubated with 50 μL of 1:100 diluted mouse anti-HLA-ABC mAb or 50 μL PBS as control at 37 °C in humid box for 1 h and then were washed twice with PBS for 5 min each time. Next, 50 μL of 1:200 diluted biotin labeled goat-anti mouse IgG was added. After being incubated for 30 min and washed as before, 50 μL of 1:200 diluted HRP labeled streptavidin was added. The following incubation and washing was exactly the same as above. Finally, 50 μL of DAB working solution was added for color reaction under the microscopical scrutiny. The reaction was stopped with tap water rinse. The slides and sections were then counterstained with hematoxylin as routine assay.
Image analysis
The slides were focused under photographic microscope and the 24 bit true color images at a resolution 768 × 76 were captured in real time by the JVC TK-C1381 digital camera. The captured images were read into the Mias-2000 Medical Images Analyzer. The objective grayscale was obtained through color HSV splitting and half binary quantifying. The data were assessed by Newman-Keuls method of q test using statistical package PEMS2.1 for Medical Statistics of Chinese Medical Encyclopedia. Values of P < 0.05 were considered statistically significant.
Flow cytometry
Cells were washed twice with PBS at 1200 r/min for 5 min per time. Then the cell concentration was adjusted to 1 × 106 Entries/mL and incubated with 10 μL of 1:10 diluted FITC labeled mouse anti HLA-ABC or mouse IgG as negative control at room temperature for 30 min. At last, cells were washed once and detected by Elite-ESP flow cytometer.
RESULTS
Routine HE stain
Raji and K562 cells were round in shape. QGY-7703, Alexander and HepG2 seem to be polygonal. SMMC-7721 cells were spindly. All the cell lines above had evident features of tumor cells, which were heterogeneous in nuclear size. Mononuclear tumor giant cells were commonly seen in these cell lines. In addition, binuclear or multinuclear, normal or abnormal mitotic tumor cells were frequent in QGY-7703, HepG2 and SMMC-7721. Ten of 15 specimens from patients clinically, were diagnosed as HCC pathologically in corresponding paraffin sections by the Pathological Department. The left 5 specimens were excluded from this study, which were confirmed to be combined hepatocholangiocellular carcinoma, cholangiocellular carcinoma, nodular cirrhosis, secondary liver cancer from nasopharyngeal carcinoma and colon cancer, respectively. In comparison between the HE stain of frozen sections with that of paraffin sections, HCC cells could be found in only 6 of the 10 paraffin sections corroborated specimens. The remained 4 frozen specimens without HCC cells, possibly due to the failure of sampling HCC tissue, were also excluded from this study.
Immunocytochemistry and immunohistochemsitry
Positive staining of HLA class I antigens appeared brown in color. Raji cells had very strong positive staining. Most mitotic and nonmitotic cells of the 4 HCC cell lines have various intensity of HLA class I antigens expression. The positive staining of HLA class I antigens was located in the cytoplasm, the perinuclear area, and at the cell membrane, but none in K562 and the control slides of all the cell lines (Figure 1, Figure 2). In the 6 HCC specimens studied immunohistochemically, histologically normal hepatocytes had no or very weak positive staining and the liver sinus had very strong positive staining. Most HCC cells in the sections from the 6 HCC specimens had strong positive staining of HLA class I antigens, which were located in the cytoplasm, the perinuclear area, and at the cell membrane of the liver cancer cells (Figure 3, Figure 4).
Figure 1.
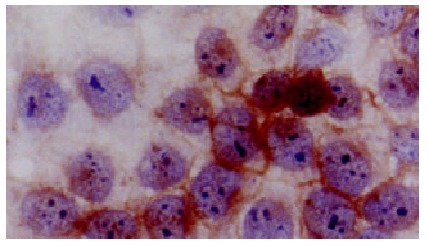
Positive staining of HLA-I antigens at QGY-7703 cell membrane. LSAB × 350
Figure 2.
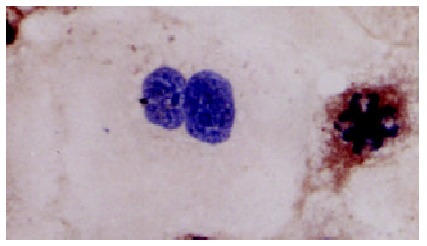
Positive staining of HLA-I antigens in cytoplasm of dividing Alexander cells•LSAB × 350
Figure 3.
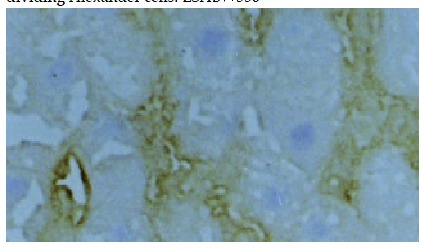
Negative staining of histologically normal liver cells and intensive positive staining within sinusoids. LSAB × 100.
Figure 4.
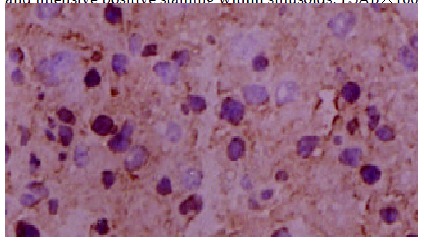
Positive staining of hepatocelluar carcinoma cells LSAB × 100.
Image analysis
The image analysis of HLA class I antigens expressions in the slides showed that the objective grayscale values of Raji and those 4 HCC cell lines were significantly different from that of K562 (P < 0.01). Significant differences were also found between Raji and the 4 HCC cell lines (Table 1).
Table 1.
Grayscale value comparison of HLA-I antigens among different cell lines
| Tumor cell lines | n | Grayscale value (¯x ± s) |
| Raji | 7 | 114.04 ± 10.94a |
| Alexander | 7 | 165.97 ± 5.35ab |
| HepG2 | 7 | 167.02 ± 12.60ab |
| QGY-7703 | 7 | 161.46 ± 7.13ab |
| SMMC-7721 | 7 | 165.93 ± 5.21ab |
| K562 | 7 | 244.89 ± 4.60 |
P < 0.01 vs K562;
P < 0.01 vs Raji.
Flow cytometry
The positive control Raji cells had strong fluorescence intensity, but Raji cells incubated with FITC labeled mouse IgG had very weak fluorescence. Fluorescence intensities of QGY-7703, Alexander, HepG2, and SMMC-7721 were very high. Some cells of the 4 cell lines had equal or even higher fluorescence intensities than those of Raji. The negative control cells K562 had very weak fluorescence intensity of HLA class I antigens, the irrelevant fluorescence stain of K562 was also weak (Figure 5, Figure 6).
Figure 5.
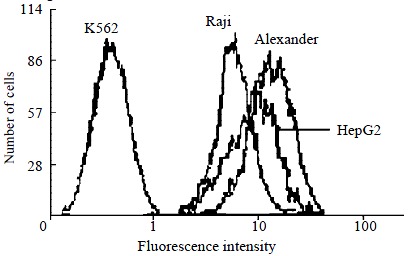
Flow cytometric histogram comparison of HLA-I antigens among K562, Raji, Alexander, HepG2 cell lines.
Figure 6.
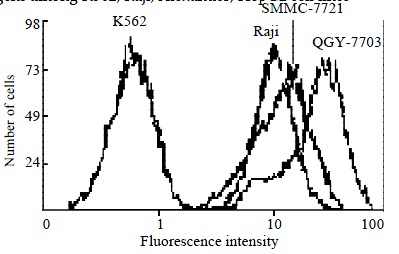
Flow cytometric histogram comparison of HLA-I antigens among K562, Raji, SMMC-7721, QGY-7703 cell lines.
DISCUSSION
HCC, one of the most common cancers, is the second cancer killer in China, and its mortality rate tends to increase year by year. About 11000 of Chinese die of HCC annually. This number covers approximately half of the world population who die of HCC. At present, the overall curative effect of HCC is far from satisfaction. More than 2/3 of the HCC patients were found inoperatable. The surgically treated patients have to face the challenge that how to clear away the residual cells to prevent metastasis and relapse. Because of serious side effects of radiotherapy and chemotherapy, new therapy is urgent clinically to be searched and developed so as to improve the curative effect of HCC. In recent years, studies of CTL vaccines against HCC have become frontiers of HCC immunotherapy[6-8]. The expression of MHC class I antigens is essential in CTL cytotoxicity. For example, the killing activity of mouse AFP specific CTL to mouse HCC cell lines Hepa1-6 and BWIC3, which express few MHC class I antigens, is much lower than that of MHC class I antigens positive EL4 cells[6]. Thus, whether HCC is suitable for CTL vaccine therapy depends to a great extent on the HLA class I antigens expression status of HCC cells.
Although a few studies have reported that liver cancer cells in HCC tissue or cultured in vitro have strong HLA class I antigens expression[2,3,9-12], few are aimed at Chinese. A Chinese research group, however, have initiated a study involving the expression of HLA-ABC on hepatocarcinoma. Our results of Alexander and HepG2 are consistent with results of other groups[2,3,10-13]. HCC cell lines QGY-7703 and SMMC-7721 have been established from HCC tissues of Chinese patients and frequently used in studies in China. Our results of these two cell lines indicate that both of them have strong HLA class I antigens expression. In addition, we found that mitotic HCC cells also strongly express HLA class I antigens. This phenomenon was not reported before, indicating that the corresponding specific CTL may kill the mitotic liver cancer cells. Our investigation on frozen specimens from 6 Chinese HCC patients shows that most liver cancer cells in the HCC tissue have strong HLA class I antigens expression. This is also consistent with the results of other groups[2,9,13]. It is worth paying attention to the articles of Paterson et al[9]. They reported that liver cancer cells in a handful of HCC specimens (4/70, 5.7%) did not express HLA class I antigens. However, this is not found in our study, possibly due to the limited amount of the specimens studied in our present study.
The results in our studies also indicate that in histologically normal liver HLA class I antigens were mainly expressed on liver sinusoidal lining cells rather than on hepatocytes. This is consistent with the results of several other groups[2,9,14]. The reason why the phenomenon that histologically normal hepatocytes have no or only weak HLA class I antigens expression though liver cancer cells strongly express HLA class I antigens, may be related to the unique lymphocytes distribution in liver and NK-cell escape[1,15,16]. About 31% of the liver resident lymphocytes are NK cells[15]. Therefore, re-expression or enhanced expression of HLA class I antigens on HCC cells would be helpful for inhibiting the nonspecific cytotoxicity of NK cells. However, the cause of above phenomenon and its immunobiological implications remain to be explained by further studies.
In brief, the results of this study and other research groups indicate, from viewpoint of HLA class I antigens expression, that CTL vaccine therapy is suitable and practicable for HCC. This would provide in vivo and in vitro experimental basis for the follow-up study of CTL vaccine design and construction.
Footnotes
Edited by Ma JY
Supported by the National Natural Science Foundation of China, No.30070855
References
- 1.Ruiz-Cabello F, Garrido F. HLA and cancer: from research to clinical impact. Immunol Today. 1998;19:539–542. doi: 10.1016/s0167-5699(98)01349-8. [DOI] [PubMed] [Google Scholar]
- 2.Sung CH, Hu CP, Hsu HC, Ng AK, Chou CK, Ting LP, Su TS, Han SH, Chang CM. Expression of class I and class II major histocompatibility antigens on human hepatocellular carcinoma. J Clin Invest. 1989;83:421–429. doi: 10.1172/JCI113900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Wadee AA, Paterson A, Coplan KA, Reddy SG. HLA expression in hepatocellular carcinoma cell lines. Clin Exp Immunol. 1994;97:328–333. doi: 10.1111/j.1365-2249.1994.tb06089.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Alexander JJ, Bey EM, Geddes EW, Lecatsas G. Establishment of a continuously growing cell line from primary carcinoma of the liver. S Afr Med J. 1976;50:2124–2128. [PubMed] [Google Scholar]
- 5.Aden DP, Fogel A, Plotkin S, Damjanov I, Knowles BB. Controlled synthesis of HBsAg in a differentiated human liver carcinoma-derived cell line. Nature. 1979;282:615–616. doi: 10.1038/282615a0. [DOI] [PubMed] [Google Scholar]
- 6.Vollmer CM, Eilber FC, Butterfield LH, Ribas A, Dissette VB, Koh A, Montejo LD, Lee MC, Andrews KJ, McBride WH, et al. Alpha-fetoprotein-specific genetic immunotherapy for hepatocellular carcinoma. Cancer Res. 1999;59:3064–3067. [PubMed] [Google Scholar]
- 7.Butterfield LH, Koh A, Meng W, Vollmer CM, Ribas A, Dissette V, Lee E, Glaspy JA, McBride WH, Economou JS. Generation of human T-cell responses to an HLA-A2.1-restricted peptide epitope derived from alpha-fetoprotein. Cancer Res. 1999;59:3134–3142. [PubMed] [Google Scholar]
- 8.Butterfield LH, Meng WS, Koh A, Vollmer CM, Ribas A, Dissette VB, Faull K, Glaspy JA, McBride WH, Economou JS. T cell responses to HLA-A*0201-restricted peptides derived from human alpha fetoprotein. J Immunol. 2001;166:5300–5308. doi: 10.4049/jimmunol.166.8.5300. [DOI] [PubMed] [Google Scholar]
- 9.Paterson AC, Sciot R, Kew MC, Callea F, Dusheiko GM, Desmet VJ. HLA expression in human hepatocellular carcinoma. Br J Cancer. 1988;57:369–373. doi: 10.1038/bjc.1988.84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Paroli M, Carloni G, Franco A, De Petrillo G, Alfani E, Perrone A, Barnaba V. Human hepatoma cells expressing MHC antigens display accessory cell function: dependence on LFA-1/ICAM-1 interaction. Immunology. 1994;82:215–221. [PMC free article] [PubMed] [Google Scholar]
- 11.Tatsumi T, Takehara T, Katayama K, Mochizuki K, Yamamoto M, Kanto T, Sasaki Y, Kasahara A, Hayashi N. Expression of costimulatory molecules B7-1 (CD80) and B7-2 (CD86) on human hepatocellular carcinoma. Hepatology. 1997;25:1108–1114. doi: 10.1002/hep.510250511. [DOI] [PubMed] [Google Scholar]
- 12.Kurokohchi K, Carrington M, Mann DL, Simonis TB, Alexander-Miller MA, Feinstone SM, Akatsuka T, Berzofsky JA. Expression of HLA class I molecules and the transporter associated with antigen processing in hepatocellular carcinoma. Hepatology. 1996;23:1181–1188. doi: 10.1002/hep.510230537. [DOI] [PubMed] [Google Scholar]
- 13.Zhai SH, Liu JB, Zhu P, Wang YH. CD54, CD80, CD86 and HLA-ABC expressions in liver cirrhosis and hepatocarcinoma. Shijie Huaren Xiaohua Zazhi. 2000;8:292–295. [Google Scholar]
- 14.Franco A, Barnaba V, Natali P, Balsano C, Musca A, Balsano F. Expression of class I and class II major histocompatibility complex antigens on human hepatocytes. Hepatology. 1988;8:449–454. doi: 10.1002/hep.1840080302. [DOI] [PubMed] [Google Scholar]
- 15.Doherty DG, O'Farrelly C. Innate and adaptive lymphoid cells in the human liver. Immunol Rev. 2000;174:5–20. doi: 10.1034/j.1600-0528.2002.017416.x. [DOI] [PubMed] [Google Scholar]
- 16.Luo DZ, Vermijlen D, Ahishali B, Triantis V, Plakoutsi G, Braet F, Vanderkerken K, Wisse E. On the cell biology of pit cells, the liver-specific NK cells. World J Gastroenterol. 2000;6:1–11. doi: 10.3748/wjg.v6.i1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]


