Abstract
The aim of this study was to assess changes in indirect markers of muscle damage and type I collagen degradation, as well as, patellar and Achilles tendon morphological differences during nine daily drop-jumps sessions with constant load alternated with rapid increases in load to test the hypothesis that frequent drop-jump training results in negative muscular and tendon adaptation. Young men (n = 9) performed daily drop jump workouts with progression every 3 days in terms of number of jumps, platform height and squat amplitude. Voluntary and electrically evoked knee extensor torque, muscle soreness, blood plasma creatine kinase (CK) activity and carboxyterminal cross-linked telopeptide (ICTP), patellar and Achilles tendon thickness and cross-sectional area (CSA) were assessed at different time points during the training period and again on days 1, 3, 10 and 17 after the training. The findings were as follows: (1) steady decline in maximal muscle strength with major recovery within 24 hours after the first six daily training sessions; (2) larger decline in electrically induced muscle torque and prolonged recovery during last three training sessions; (3) increase in patellar and Achilles tendons CSA without change in thickness towards the end of training period; (4) increase in jump height but not in muscle strength after whole training period. Our findings suggest that frequent drop-jump sessions with constant load alternated with rapid increases in load do not induce severe muscle damage or major changes in tendons, nonetheless, this type of loading is not advisable for muscle strength improvement.
Key points.
Frequent drop jump training induces activation mode dependent muscle torque depression late in the training period.
No significant changes in the thickness of patellar and Achilles tendons are observed during frequent training, while CSA increases towards the end of training period.
Longitudinal effect for jump height but not for muscle strength is evident after the whole training period.
Key words: Stretch-shortening cycle, neuromuscular adaptation, electrical stimulation, Achilles tendon, patellar tendon
Introduction
Stretch-shortening cycle (SSC) exercise is defined as the eccentric contraction of an active muscle immediately followed by a concentric contraction (Komi, 1984). Previous studies have shown that high-force exercises with a large eccentric component impair muscle function (Byrne et al., 2004; Skurvydas et al., 2011), simultaneously inducing muscle swelling, stiffness, acute inflammation reaction, an increase of muscle enzymes such as creatine kinase (CK) activity in the bloodstream, delayed-onset of muscle soreness (DOMS, Chen et al., 2013; Nosaka and Clarkson, 1996), as well as changes in muscle fiber ultrastructural integrity (Hortobágyi et al., 1998; Féasson et al., 2002; Lauritzen et al., 2009). These changes are widely accepted as markers of exercise-induced muscle damage (Clarkson and Hubal, 2002) or represent muscle remodeling (Yu et al., 2002). Moreover, high-force rapid exercises place great stress on the tendons, which act as a buffering mechanism during eccentric contraction (Reeves and Narici, 2003; Roberts and Konow, 2013). Indicators of connective tissue injury are well documented histologically (Maffulli et al., 2000), biochemically (Waśkiewicz et al., 2012) and by imaging (Grigg et al., 2009).
SSC exercise such as drop jumping has been extensively used to enhance power abilities for athletes or healthy non-professionals for review see (Markovic, 2007). The positive effects are mainly associated with improvement in muscle and tendon strength and properties, as well as the ability to use elastic energy (Kubo et al., 2005). On the other hand, SSC exercise should be used with caution because high-volume training protocols lead to chronic fatigue and decreased exercise performance (Seene et al., 2008).
Our recent observations have shown that the neuromuscular system is sensitive to the strategy of load increase during SSC training (Kamandulis et al., 2011, 2012). Detrimental muscle damage can be avoided by gradual introduction of more demanding exercise induced by increasing the volume, intensity and muscle stretch amplitude (Kamandulis et al., 2011). However, the magnitude of improvement in muscle function is greater after constant loads are alternated with steep increases in load, than that induced by a continuous constant progression-loading pattern with small gradual load increments in each training session (Kamandulis et al., 2012).
The frequency of SSC exercise is another factor that should be taken into consideration because of the training effect on human neuromuscular system adaptation. In the field of athletic conditioning, maximal SSC exercise is performed no more than 2–3 times per week to provide sufficient time for muscle power generating machinery to recover completely. The concern is that more frequent SSC training loads might lead to muscle and tendon overuse and injuries. However, it is not always a case, especially in top level sports. By the end of conditioning phase, some coaches apply high intensity training more frequently than three times per week for short period of time (micro cycle of 5-9 days). Some coaches implements shock microcycles in pre-season training applying high intensity workouts more frequently than three times per week for short periods of time. This purposely induces great amounts of fatigue and damage with very challenging loads expecting superior positive supercompensation in subsequent phase of reduced loading. In addition, Chen and Hsieh (2001) have reported that daily repeated constant magnitude maximal isokinetic eccentric exercises did not exacerbate muscle damage and inflammation within a 7-day training period, suggesting that a short time between training stimuli does not lead to muscle damage accumulation. However, it is not known if observations made using constant loads are mirrored by applying a more demanding rapid load increase strategy. Also, there is a lack of data in the literature regarding the impact of frequent SSC training loads on structural alteration of tendons.
The aim of this study was to assess changes in indirect markers of muscle damage and type I collagen degradation, as well as patellar and Achilles tendon morphological differences during nine daily drop-jump sessions with constant load alternated with rapid increases in load magnitude. Based on data for recovery requirements, it was hypothesized that neuromuscular performance would decline during the period of frequently repeated SSC exercise training, which might consequently lead to negative muscular and connective tissue adaptation. In the present study, the training program used by Kamandulis et al. (2012) was performed daily instead of three times per week to simulate indirect comparison regarding training frequency.
Methods
Experimental approach to the problem
Upon arrival at the laboratory, each participant subjectively scored his level of muscle soreness and samples of venous blood were collected to measure CK activity before each training session and at 1, 3, 10 and 17 days after the final training session. After initial measurements, the participants completed warm-up exercises, and were then seated in an isokinetic dynamometer chair. Maximal isometric voluntary and electrically evoked knee extensor torque as well as countermovement jump height was measured before and within 2–5 minutes after each training session and again at 1, 3, 10 and 17 days after completion of the training. Biochemical analysis for markers of type I collagen degradation as well as patellar (PA) and Achilles (AT) tendons measurements by ultrasound were made before the experiment and at 24 h after training sessions 1, 4, 7 and 9 (upon arrival at the laboratory just before training session on days 2, 5, 8 and 10). The similar experimental protocol was used in previous study (Kamandulis et al., 2012).
Subjects
Nine healthy physically active untrained young men (mean ± standard deviation (SD), age 21.6 ± 3.2 years, body weight 80.5 ± 11.5 kg and height 1.80 ± 0.06 m) volunteered to participate in this study. They had not been involved in any strength or plyometric training programs in the 3 months before the experiment. The participants were instructed to abstain from any exercise 2 weeks before the experiment and were not allowed to participate in additional physical activities throughout the experimental period including 3-week follow-up phase. Each participant read and signed a written informed consent form consistent with the principles outlined in the Declaration of Helsinki. The Regional Ethics Committee approved this study.
Procedures
Familiarization: A familiarization session was performed 1 week before the initial exercise. Participants were seated in the isokinetic dynamometer chair and asked to activate their knee extensor muscles isometrically. Then participants’ tolerance to electrical stimulation was assessed. The intensity of electrical stimulation was determined on an individual basis by applying tetanic stimulation to the muscle. During this procedure, the voltage was increased until no increment in torque response was elicited by an additional 10% increase in voltage.
Warm-up: A warm-up preceded all test sessions. It comprised 8 min of stationary cycling with the power (W) approximately equal to the participant’s body weight (kg), followed by some light stretching exercises.
Training program: The participants performed intermittent (20 s interval between jumps) drop jumps (DJs) from a platform with immediate maximal rebound. The training sessions were performed daily for 9 days. Progression in terms of jump number, platform height and squat amplitude was applied every 3 days. During the first three training sessions (1–3), the jump number was 40, the platform height was 20 cm and the knee angle squat amplitude was up to 90° (0° = full knee extension). During training sessions 4–6, the jump number was 70, the platform height was 60 cm and the knee angle squat amplitude was up to 90°. In the final three training sessions (7–9), the jump number was 70, the platform height was 80 cm and the knee angle range was increased to exceed 110° knee flexion angle (the squat amplitude increased). The same training program with the lower frequency of training sessions was executed in previous study (Kamandulis et al., 2012). All jumps were performed on a contact mat (Newtest Powertimer Testing System, Oulu, Finland) and the jump height and contact time was monitored for all jumps. The knee joint angle was controlled visually by an experienced researcher, and if the angle diverged from the intended target angle during the exercise, the participant was instructed immediately to modify the way the jump was performed. The jumps were performed as quickly as possible during the ground-contact phase. The hands were placed on the waist. Each participant received verbal feedback about his performance after each jump and was in this way encouraged to jump as high as possible.
Dynamometry and electrostimulation: The isometric torque of the dominant leg knee extensor (KE) muscles was measured using an isokinetic dynamometer (System 3; Biodex Medical Systems, Shirley, USA). The participant sat upright in a dynamometer chair with their knee joint positioned at 70° (full knee extension = 0°). Shank, trunk and shoulders were stabilized by belts. Maximal voluntary isometric contraction (MVC) torque was measured twice, each separated by a 2 min rest interval. The participants were instructed to perform MVC as hard as possible and maintain the peak for 3 s before relaxation. The equipment and procedure for electrical stimulation were essentially the same as described previously (Skurvydas et al., 2010). Direct muscle stimulation was applied using three carbonized rubber electrodes covered with a thin layer of electrode gel (ECG–EEG Gel, Medigel, Modi’in, Israel). Two of the electrodes (6 cm i 11 cm) were placed transversely across the width of the proximal portion of the quadriceps femoris muscle next to the inguinal ligament. A third electrode (6 cm 11 cm) covered the distal portion of the muscle above the patella. A standard electrical stimulator (MG 440; Medicor, Budapest, Hungary) was used. The electrical stimulation was delivered in square-wave pulses, each 1 ms in duration. Tetanic torque of the KE muscles was evoked by 1 s trains of electrical stimulation at 20 Hz (P20) and 100 Hz (P100), separated by a 5 s rest interval. 20/100 Hz torque ratio decline indicates the presence of low-frequency fatigue (Warren et al., 2001). The intra-class correlation coefficient (ICC) of voluntary contraction force was 0.93, while ICC of electrically evoked force varied from 0.85 to 0.95 depending on stimulation frequency.
Counter-movement jump: The participants performed control jumps from an upright standing position with counter-movement to a knee angle of about 90° before the first training session and 1, 3, 10 and 17 days after the final training session. The same equipment as during the training was used. The jump height was calculated with the formula: jump height (cm) = 122.625 p (Tf)2, where Tf = time of flight (s) (Bosco et al., 1982). The highest of the five jumps was selected for analysis. The ICC of counter-movement jumps was 0.95.
Muscle soreness: Muscle soreness was reported daily throughout the study using a scale of 0–10, where 0 represented “no pain” and 10 represented “intolerably intense pain” (Skurvydas et al., 2010). Each participant indicated the severity of soreness in both legs quadriceps muscle separately during 2–3 squats performed slowly (2-3 sec each) to a 90° knee angle. The scores were almost identical for both legs, therefore dominant leg values were used for further analysis.
Biochemical measures: Samples of venous blood were collected, immediately centrifuged, and analyzed for plasma CK activity using a SpotchemTM EZSP-4430 biochemical analyzer (Menarini Diagnostics, Reading, UK) with soft reagent strips (ArkRay Factory Inc, Shiga, Japan). The normal reference range for human plasma CK activity using this method is between 56 and 244 IU·L–1 according to the manufacturer. Coefficient of variation for CK assay was less than 8 % as calculated from the ratio of standard deviation to the mean in repeated measurements (n = 8) of the same sample.
Biochemical analysis for serum levels of carboxyterminal cross-linked telopeptide of type I collagen degradation (ICTP) was performed using venous blood. Serum samples were obtained by centrifugation (3500 rpm, 15 min) and were stored in a deep-freezer (–82°C) until use. ICTP levels were measured using an ELISA kit (TSZ ELISA, USA, coefficient of variation < 12.0%).
Ultrasonographic measurement: Patellar and Achilles tendon measurements were conducted with a with a General Electric Logiq 7 device (Solingen, Germany) using a 7–12 MHz linear probe. Before study, the overall joint condition of the participants was examined by ultrasound. Participants with pathological changes of PA and AT or fluid collections were excluded from the study. Repeated measurements using this technique have previously shown high reliability – the ICC for patella and Achilles tendons thickness and cross sectional area were 0.88 – 0.96 (Reeves et al., 2003; Neves et al., 2014). Ultrasonographic measurements were performed by a radiologist with nearly ten years of experience in musculoskeletal examination.
Patellar tendon examination was performed on the longitudinal and transverse axis of both knees while the participant was in a supine position with their knees supported and flexed. The examination was performed at the most appropriate degree of knee flexion (approximately 30° and 45°) at which the tendon is taut with well-defined margins and is homogeneous. The PA was assessed in three areas near (1 cm from) the apex of the patella, in the central part, and at the tibial attachment. The CSA and thickness at each location of PA were measured.
To assess the AT, participants were placed in a prone position and the feet were dorsiflexed to maximum to make the AT taut. AT thickness of both feet was scanned (longitudinally and transversely) approximately 1, 3 and 4.5 cm above the calcaneal insertion and the cross-sectional area of tendons at analogous sites was measured. Average of both legs thickness and CSA measurements were taken for analysis. Transverse images values are presented in the results.
Statistical analysis
The descriptive data are presented as mean ± SD. A Kolmogorov–Smirnov test was used to check normality of data distribution. Baseline differences were assessed using paired t test. A 2-way repeated-measures analysis of variance (ANOVA) was used to determine the effects of time (pre and post) and training sessions (1-9) on voluntary and electrically evoked torques. The same method was used to compare changes relative to baseline value for muscle torque measures of training sessions and activation mode (100 Hz vs. MVC), training sessions and stimulation frequency (20 Hz vs. 100 Hz) as well as time and training phase (1-3 vs. 7-9 training sessions). One-way repeated measures ANOVA was used to test the effect of training sessions on jump height, contact time, biochemical markers (CK and ICTP) and ultrasound measures (thickness and CSA). In all cases, changes during the nine training sessions (before and immediately after) were analyzed separately from recovery data after training period (baseline and 1, 3, 10 and 17 days after). When a significant main effect was identified by ANOVA, a pairwise Sidak test was applied to locate the significant difference between time points. The level of significance was set at P < 0.05. Muscle soreness was evaluated using a non-parametric Wilcoxon signed-rank test since values were not normally distributed. For a One-Tailed hypothesis with α = 0.05 and statistical power level 0.80, the number of subjects required to detect a change is 8 based on changes in the data of high frequency stimulation (100 Hz) evoked muscle torque of the KE muscles in a previous study (Kamandulis et al., 2012). All data analysis was performed using SPSS version 13.0 (SPSS Inc, Chicago, IL, USA).
Results
Baseline values
The mean baseline values were different between MVC torque and electrically evoked tetanic torques as well as between tetanic torques evoked by high and low stimulation frequencies (p < 0.001 for both).
Damaging exercise
The average height of the DJs was similar during training sessions (except session 7), while ground-contact time increased non-significantly (p > 0.05) in training sessions 4–6 and then increased significantly in training sessions 7–9 (F = 53.1, p < 0.001 for training phase effect; Figure 1) compared to baseline values.
Figure 1.
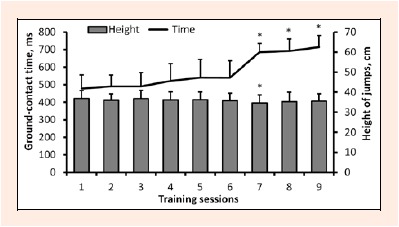
Drop jump height and ground contact time during each of nine training sessions (group mean ± SD). * p < 0.05 compared to value of the first training session.
Muscle strength
Both MVC torque and electrically evoked tetanic torques of the dominant leg KE muscles decreased significantly after each of the nine training sessions (p < 0.001; Figure 2). A significant effect of time by training session interaction on P100 (F = 2.8, p = 0.047) and P20 (F = 3.4, p = 0.004) but not on MVC (F = 1.8, p > 0.05) showed that electrically evoked strength changes were significantly different within the training period.
Figure 2.
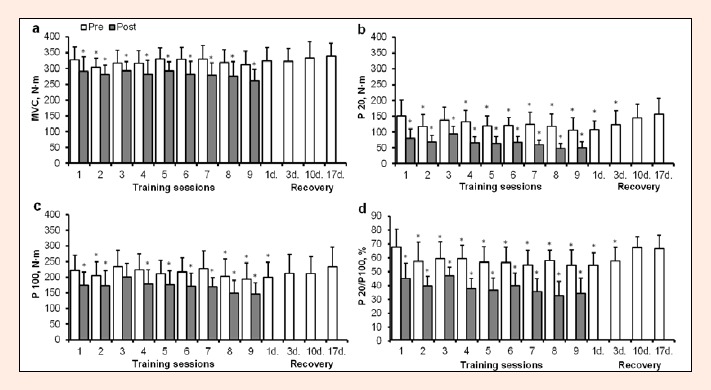
Maximal torque of dominant leg m. quadriceps obtained during maximal voluntary isometric contraction (MVC, a) and electrically evoked torque of m. quadriceps femoris using stimuli trains at 100 Hz (P100, c) and 20 Hz (P20, b) and expressed as 20/100 Hz torque (d) ratio immediately before (Pre) and 10 min after (Post) each of nine training sessions as well as 3, 10 and 17 days after cessation of training (group mean ± SD). * P < 0.05 compared to value before the first training session.
Despite the reduction magnitude, MVC torque had returned to the pre-training level before the next training session on each occasion except after training session 1 (Figure 2a). There was a tendency for a greater extent of MVC torque recovery up to training session 7, while an opposite tendency was observed after the final three training sessions (p > 0.05). At the same time, P100 recovered completely until the next training after sessions 2–6. However, the drop in P100 was largest after sessions 8 and 9 and peak torque did not recover during the 24 h after sessions 7–9 (F = 7.1, p < 0.001 for training phase and time interaction; Figure 2c). As predicted, the reduction was greater in tetanic torque evoked by 20 Hz compared with that evoked by 100 Hz stimulation (F = 12.9, p < 0.001 for training sessions and stimulation frequency interaction). In response to training, P20 decreased more than 60% after sessions 8 and 9 (F = 5.7, p = 0.002 for training phase and time interaction; Figure 2b), and did not recover during 24 h beyond training session 3. A similar tendency to P20 changes for the 20/100 Hz torque ratio was observed during the course of training (Figure 2d).
Following the training period, there was a need for 1, 3 and 10 days’ rest for complete MVC, P100 and P20 restoration, respectively. However, neither isometric peak torque exceeded pre-training values within 17 days after the end of training. By contrast, counter-movement maximal jump height increased from a pre-training value of 39.6 ± 4.8 cm to 42.3 ± 5.2 cm (7.0% increase, p = 0.031) and 42.7 ± 5.4 cm (7.9% increase, p = 0.007) at 10 and 17 days following the final training session, respectively.
Muscle soreness
Participants reported mild quadriceps muscle soreness (about 2 points on average) in dominant leg throughout the study (Figure 3a). The greatest value was at 24 h after training session 2 (3.4 points, p < 0.05 compared to training session 1), which was probably a cumulative effect of the first two training loads.
Figure 3.
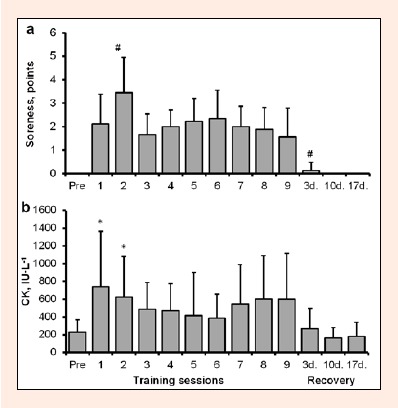
Soreness of dominant leg m. quadriceps (a) and plasma CK activity (b) immediately before (Pre) and at 24 h each of nine training sessions as well as 3, 10 and 17 days after cessation of training (group mean ± SD). * P < 0.05 compared to before the first training session; # P < 0.05 compared to value at 24 h after the first training session.
Biochemical markers
CK activity in the blood increased threefold at 24 h after the first session (p = 0.025; Figure 3b), then decreased slightly, and then had the tendency to increase again after sessions 8 and 9 (p > 0.05). The variability of CK response was large with the SD value almost reaching the average level. No significant changes were found in ICTP during the training period, measured at 24 h following exercise (Figure 4).
Figure 4.
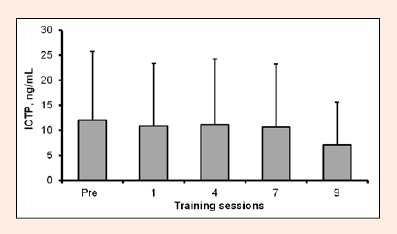
Serum levels of carboxyterminal cross-linked telopeptide of type I colagen degradation (ICTP) immediately before (Pre) and at 24 h after training sessions 1, 4, 7 and 9 (group mean ± SD).
Ultrasound measures
No significant changes were found in the thickness of the PT or AT during or after the training period (Figure 5a and 5b). However, a CSA increase was observed after 24 h following the final training session in both the PT and AT (p < 0.05; Figure 5c and 5d).
Figure 5.
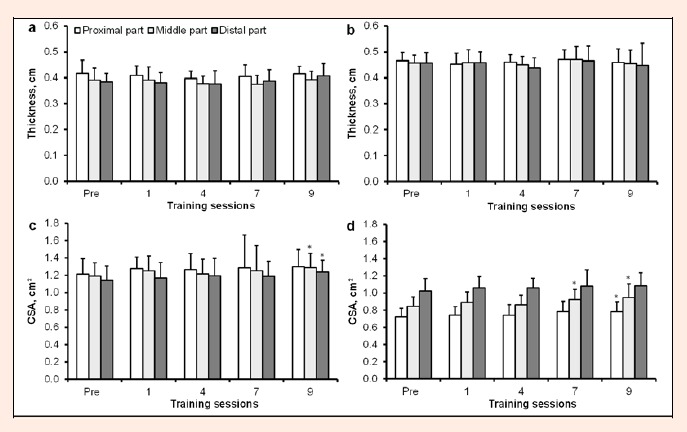
Patellar tendon (diagram a and c) and Achilles tendon (diagram b and d) ultrasonography measurements (group mean ± SD). * P < 0.05 compared to value before the first training session.
Discussion
In the present study, daily drop jumping was applied with constant load and alternated with rapid increases in load magnitude to test the hypothesis that neuromuscular performance would decline during the period of frequently repeated SSC training. The main findings were as follows: (1) steady decline in maximal muscle strength with major recovery within 24 hours after the first six daily training sessions; (2) larger decline in electrically induced muscle torque and prolonged recovery during last three training sessions; (3) increase in PA and AT tendons CSA without change in thickness towards the end of training period; (4) increase in jump height but not in muscle strength after whole training period. These findings support in part the present study hypothesis of performance deterioration during frequent SSC, together with a minor longitudinal effect for muscle function suggesting the minimal value of such a training strategy.
Isometric torque change recorded during the training period was used as a key marker of exercise-induced muscle damage in participants. There were quite long periods between jumps (20 s) to avoid metabolic fatigue. In addition, drop jumps with an active phase (subjects in contact with the mat) of 0.3–0.4 s are unlikely to induce any significant changes in energy metabolites (Sahlin and Ren, 1989). Absence of metabolic fatigue implies that peak torque reduction immediately after eccentric exercise is associated with muscle damage. In general, decline in muscle strength was observed after each training session in the present study, indirectly supporting the findings of other researchers that eccentric exercises induce damage to exercised muscles (Clarkson and Hubal, 2002; Lauritzen et al., 2009). However, the extent of muscle damage observed was quite small and remained stable during the first six training sessions despite rapid augmentation of load magnitude by an increase in jump number and intensity (platform height increase, training session 4). Changes in muscle torque within the first six training sessions were similar to those seen in previous studies, when participants underwent a similar load extending to 3 weeks of training, three times per week (Kamandulis et al., 2011; 2012). Our data are in agreement with those of other researchers who have concluded that because of adaptive response during the repair process after an initial bout of eccentric exercise muscle fibers become more resistive to damage in repeated bout of exercise (Chen and Nosaka, 2006). In the present study muscle torques have decreased to similar extent after training sessions 1 and 2 despite the fact that the second drop jumping was performed with the voluntary and electrically evoked torque still depressed. Similar trend was observed for tetanic force evoked by low frequency electrical stimulation changes. Protective effect have manifested itself before muscle full recovery signifying that adaptation is at least partly associated with neural factors (Chen and Hsieh, 2001; McHugh et al., 1999). Rests of training sessions are already influenced by previous jumping even though training load have been changed every three days.
However, the increase in range of motion during jumping and height of platform (exercise intensity) in training session 7 led to a decrease in electrically evoked tetanic peak torque (P100 and P20), which was not recoverable within 24 h, forcing participants to perform the next training session with incomplete muscle recovery. In addition, low-frequency fatigue remained similar within the six training sessions, but tended to increase later in the training. There was a trend for isometric voluntary and electrically evoked torque production to reach their lowest values after training sessions 8 and 9. All these findings suggest that training beyond training session 7 appeared to bring increased muscle damage. Possibly this damage could be associated with frequent load repetition without allowing sufficient time for participant recovery in the present study. It also might be related to the fact that muscle stretch amplitude and height of platform were greater during the final three training sessions compared with those earlier. However, this possibility is less likely because changes in isometric torque were more severe indirectly compared with the identical protocol performed less frequently in a previous study (Kamandulis et al., 2012). The authors of that study concluded that muscle damage did not increase even when more demanding exercise conditions were introduced by varying the volume, intensity, and stretch amplitude. Compared with these previous findings, increase in training frequency produced greater decline in isometric torque during final training phase, probably reflecting enlargement of muscle stress.
It is not surprising that a rapid increase in load during a drop-jump training program leads to a specific response of exercise-induced damage markers. Different markers address possibly unrelated sites of exercise-induced damage. In the present study, the effect of performance deterioration during frequent SSC was most noticeable for isometric torque, but not for soreness or plasma CK activity. Indeed, tetanic torque decreases observed in muscle contraction evoked by low-frequency (20 Hz) stimulation were greater and the recovery rate was smaller compared with high-frequency (100 Hz) stimulation, indicating a very strong manifestation of low-frequency fatigue, which is associated with disturbances in Ca2+ release from the sarcoplasmic reticulum to myoplasma, or decreased myofibrillar Ca2+ sensitivity (Allen et al., 2008). Our results indicate that SSC-induced long-lasting low-frequency fatigue tends to increase if exercise is performed frequently with a large range of motion. In view of this, frequent SSC exercise appears to induce additional stress on muscle fiber excitation–contraction coupling, which reflects the impairment of Ca2+ kinetics (Westerblad et al., 1993; Warren et al., 2001).
It is well established that an increase in CK activity is associated with ultrastructural skeletal muscle membrane damage (Brancaccio et al., 2007). CK activity increase was most significant after the first training session, while it remained elevated during all training periods in the present study. A lack of substantial CK activity increase during the training period suggests a reduction of ultrastructure damage to myofibril membranes and is consistent with the findings of previous studies (Kamandulis et al., 2011; 2012). The variables responsible for membrane strengthening remain unknown while upregulation of the matrix-related protein tenascin C (Mackey et al., 2011) and r771 integrin (Boppart et al., 2006) were both proposed to induce protection against further membrane damage. Moreover, it is interesting that previous studies have shown that muscles are protected by an initial exercise bout-induced adaptation and further training results in attenuation or absence of soreness even if more work is performed in the following training sessions (Lavender and Nosaka, 2008). In the present study, slight soreness was observed during all training periods, possibly compromising everyday functionality.
Serum ICTP is widely used as a marker of collagen degradation (Langberg et al., 2000), while it reflects bone degradation to a large degree because collagen type I is present mainly in bones (Rosenquist et al., 1998). Although it is assumed that the use of serum ICTP as tendon tissue collagen type I degradation marker is limited, increase in ICTP might be related with tendon/bone micro-damage. Overall, collagen degradation did not change from pre-exercise levels throughout the present study. In accordance to this observation, the ultrasound data revealed no changes in tendon thickness of either the PT or AT at 24 h after loading. These data suggest that chosen training loads and frequency provide rather minor stress to the tendons/bones and allow them sufficient time for recovery before a subsequent exercise bout. Changes seen in the present study are in general agreement with the opinion that only very prolonged loading is capable of changing the size and mechanical properties of human tendons (Couppé et al., 2008; Kjaer et al., 2009).
By contrast, an increase in tendon CSA of PT and AT was evident after the final training session in the present study. This increase may be explained by a higher water content and/or hyperemia in the tendons and serves as a sign of connective tissue damage (Shalabi et al., 2004). However, we noticed that despite the group average reaching statistical significance, only a few participants demonstrated a marked increase in total tendon volume. These findings indicate that some of the participants were more sensitive than others to SSC loads. Therefore, frequently repeated training sessions resulted in an increase in signs of connective tissue damage in their tendons. Results of previous studies have been inconsistent; for example, some studies found a decrease or no change in tendon dimensions (Grigg et al., 2009; Freund et al., 2011), while in other studies, tendon thickness and volume increase were observed (Shalabi et al., 2004; Van Drongelen et al., 2007). These discrepancies might be related to methodological differences, variance in study design and population (Tardioli et al., 2012).
It is important to note that in a similar study with the lower frequency of training sessions (Kamandulis et al., 2012) significant increases in muscle strength and jump height were found after completion of the full training period. Moreover, the performance of participants continued to increase within 3 weeks after training completion. In the present study, we did not find any increase in muscle strength, while a significant increase in jump height was evident. Jump height increase could possibly be explained by improved jump technique during the 9-day training. Further, it might be attributed to neural adjustments in motor unit recruitment and/or firing frequency, enhanced reflex potentiation or changes in muscle and connective tissue elastic properties (Kubo et al., 2007; Van Ingen Schenau et al., 1997). Lack of muscle strength gains advocates that a frequency of three times per week for SSC training is preferred to achieve adaptation to the human neuromuscular system.
There are some limitations to the present study. A study design that included more frequent ultrasound and biochemical measurements may have provided better vision of the changes related to connective tissue arrangement. Given the trends in tendon dimensions and CK activity changes it is possible that a type I error could be induced with such a small sample, but the significant results of the study compromise with data from the literature. Finally, the application of training loads was limited in previous studies to simulate comparison with regard to training frequency. However, more stress or training extended beyond nine training sessions possibly might lead to deeper muscle weakness and may expose further signs of high risk of muscle and tendon injury.
Conclusion
A modest degree of muscle damage with a substantial increase after rapid increases in movement range and exercise intensity late in the training and minor changes in tendons were found in young men during daily drop-jumping exercise. Jump height but not isometric voluntary and electrically evoked muscle torques increased noticeably during the post-training recovery period. Our findings suggest that frequent drop-jump sessions with constant load alternated with rapid increases in load does not induce severe muscle damage or major changes in tendons; nonetheless, this type of loading is not advisable for improvement in muscle strength.
Acknowledgements
The present study was supported by the funding from the Lithuanian Research Council. The experiments comply with the current laws of Lithuania. The authors declare that they have no conflicts of interest concerning this article.
Biographies
Vidas PALECKIS
Employment
PhD student, Institute of Sport Science and Innovations, Lithuanian Sports University, Sporto 6, LT-44221 Kaunas, Lithuania.
Degree
MSc
Research interests
The adaptation of human motor system to exercise induced muscle damage
E-mail: vidas.paleckis@lsu.lt
Mantas MICKEVIČIUS
Employment
PhD student, Institute of Sport Science and Innovations, Lithuanian Sports University, Sporto 6, LT-44221 Kaunas, Lithuania
Degree
MSc
Research interests
Muscle damage, tendons, motor performance in children
E-mail: mantas.mickevicius@lsu.lt
Audrius SNIEČKUS
Employment
Researcher, Institute of Sport Science and Innovations, Lithuanian Sports University, Sporto 6, LT-44221 Kaunas, Lithuania
Degree
PhD
Research interests
The adaptation of human motor system to exercise induced muscle damage
E-mail: audrius.snieckus@lsu.lt
Vytautas STRECKIS
Employment
Professor, Institute of Sport Science and Innovations, Lithuanian Sports University, Sporto 6, LT-44221 Kaunas, Lithuania
Degree
PhD
Research interests
The effect of age and gender on central and peripheral fatigue of motor system
E-mail: vytautas.streckis@lsu.lt
Mati PÄÄSUKE
Employment
Professor, Institute of Exercise Biology and Physiotherapy, University of Tartu, 5 Jakobi Street, 51014, Tartu, Estonia.
Degree
PhD
Research interests
Neuromuscular adaptation to exercise, musculoskeletal rehabilitation, motor performance in children, effect of aging on neuromuscular function, musculoskeletal biomechanics
E-mail: mati.paasuke@ut.ee
Saulius RUTKAUSKAS
Employment
Researcher, Department of Laboratory Medicine, Medical Academy, Lithuanian University of Health Sciences, Eiveniu 2, LT-50009, Kaunas, Lithuania.
Degree
PhD
Research interests
Knee and ankle injuries, ultrasound measurements, physical exercises
E-mail: saulius.rutkauskas@gmail.com
Rasa STEPONAVIČIŪTĖ
Employment
Researcher, Department of Laboratory Medicine, Medical Academy, Lithuanian University of Health Sciences, Eiveniu 2, LT-50009, Kaunas, Lithuania
Degree
PhD
Research interests
Exercise science, immune and stress markers
E-mail: rasastepkons@gmail.com
Albertas SKURVYDAS
Employment
Professor, Institute of Sport Science and Innovations, Lithuanian Sports University, Sporto 6, LT-44221 Kaunas, Lithuania
Degree
PhD
Research interests
Complex and dynamic adaptation in human motor system
E-mail: albertas.skurvydas@lsu.lt
Sigitas KAMANDULIS
Employment
Professor, Institute of Sport Science and Innovations, Lithuanian Sports University, Sporto 6, LT-44221 Kaunas, Lithuania
Degree
PhD
Research interests
The adaptation of human motor system to exercise induced muscle damage
E-mail: sigitas.kamandulis@lsu.lt
References
- Allen D.G., Lamb G.D., Westerblad H. (2008) Skeletal muscle fatigue: cellular mechanisms. Physiological Reviews 88(1), 287-332. [DOI] [PubMed] [Google Scholar]
- Boppart M.D., Burkin D.J., Kaufman S.J. (2006) Alpha7 beta1-integrin regulates mechanotransduction and prevents skeletal muscle injury. American Journal of Physiology - Cell Physiology 290, c1660-1665. [DOI] [PubMed] [Google Scholar]
- Bosco C., Ito A., Komi P.V., Luhtanen P., Rahkila P., Rusko H., Viitasalo J.T. (1982) Neuromuscular function and mechanical efficiency of human leg extensor muscles during jumping exercises. Acta Physiologica Scandinavica 114(4), 543-550. [DOI] [PubMed] [Google Scholar]
- Brancaccio P., Maffulli N., Limongelli M. (2007) Creatine kinase monitoring in sport medicine. British Medical Bulletin 81-82, 209-230. [DOI] [PubMed] [Google Scholar]
- Byrne C., Twist C., Eston R. (2004) Neuromuscular function after exercise-induced muscle damage: theoretical and applied implications. Sports Medicine 34(1), 49-69. [DOI] [PubMed] [Google Scholar]
- Chen T.C., Chen H.L., Lin M.J., Chen C.H., Pearce A.J., Nosaka K. (2013) Effect of two maximal isometric contractions on eccentric exercise-induced muscle damage of the elbow flexors. European Journal of Applied Physiology 113(6), 1545-1554. [DOI] [PubMed] [Google Scholar]
- Chen T.C., Hsieh S.S. (2001) Effects of a 7-day eccentric training period on muscle damage and inflammation. Medicine & Science in Sports & Exercise 33(10), 1732-1738. [DOI] [PubMed] [Google Scholar]
- Chen T.C., Nosaka K. (2006) Responses of elbow flexors to two strenuous eccentric exercise bouts separated by three days. The Journal of Strength and Conditioning Research 20(1), 108-116. [DOI] [PubMed] [Google Scholar]
- Clarkson P.M., Hubal M.J. (2002) Exercise-induced muscle damage in humans. American Journal of Physical Medicine & Rehabilitation 81(11), s52-869. [DOI] [PubMed] [Google Scholar]
- Couppé C., Kongsgaard M., Aagaard P., Hansen P., Bojsen-Moller J., Kjaer M., Magnusson S.P. (2008) Habitual loading results in tendon hypertrophy and increased stiffness of the human patellar tendon. Journal of Applied Physiology 105(3), 805-810. [DOI] [PubMed] [Google Scholar]
- Féasson L., Stockholm D., Freyssenet D., Richard I., Duguez S., Beckmann J.S., Denis C. (2002) Molecular adaptations of neuromuscular disease-associated proteins in response to eccentric exercise in human skeletal muscle. The Journal of Physiology 543(1), 297-306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Freund W., Billich C., Brambs H.J., Weber F., Schütz U.H. (2011) MRI changes of Achilles tendon and hindfoot in experienced runners and beginners during training and after a (half)-marathon competition. Zeitschrift für Orthopädie und Unfallchirurgie 149(4), 407-417. [DOI] [PubMed] [Google Scholar]
- Grigg N.L., Wearing S.C., Smeathers J.E. (2009) Eccentric calf muscle exercise produces a greater acute reduction in Achilles tendon thickness than concentric exercise. British Journal of Sports Medicine 43(4), 280-283. [DOI] [PubMed] [Google Scholar]
- Hortobágyi T., Houmard J., Fraser D., Dudek R., Lambert J., Tracy J. (1998) Normal forces and myofibrillar disruption after repeated eccentric exercise. Journal of Applied Physiology 84(2), 492-498. [DOI] [PubMed] [Google Scholar]
- Kamandulis S., Skurvydas A., Sniečkus A., Masiulis N., Aagaard P., Dargevičiutė G., Brazaitis M. (2011) Monitoring markers of muscle damage during a 3-week periodized drop-jump exercise program. Journal of Sports Sciences 29(4), 345-353. [DOI] [PubMed] [Google Scholar]
- Kamandulis S., Snieckus A., Venckunas T., Aagaard P., Masiulis N., Skurvydas A. (2012) Rapid increase in training load affects markers of skeletal muscle damage and mechanical performance. The Journal of Strength and Conditioning Research 26(11), 2953-2961. [DOI] [PubMed] [Google Scholar]
- Kjaer M., Langberg H., Heinemeier K., Bayer M.L., Hansen M., Holm L., Doessing S., Kongsgaard M., Krogsgaard M.R., Magnusson S.P. (2009) From mechanical loading to collagen synthesis, structural changes and function in human tendon. Scandinavian Journal of Medicine & Science in Sports 19(4), 500-510. [DOI] [PubMed] [Google Scholar]
- Komi P.V. (1984) Physiological and biomechanical correlates of muscle function: effects of muscle structure and stretch-shortening cycle on force and speed. Exercise and Sport Sciences Reviews 12, 81-121. [PubMed] [Google Scholar]
- Kubo K., Kanehisa H., Fukunaga T. (2005) Effects of viscoelastic properties of tendon structures on stretch-shortening cycle exercise in vivo. Journal of Sports Sciences 23(8), 851-860. [DOI] [PubMed] [Google Scholar]
- Kubo K., Morimoto M., Komuro T., Yata H., Tsunoda N., Kanehisa H., Fukunaga T. (2007) Effects of plyometric and weight training on muscle-tendon complex and jump performance. Medicine & Science in Sports & Exercise 39(10), 1801-1810. [DOI] [PubMed] [Google Scholar]
- Langberg H., Skovgaard D., Asp S., Kjaer M. (2000) Time pattern of exercise-induced changes in type I collagen turnover after prolonged endurance exercise in humans. Calcified Tissue International 67(1), 41-44. [DOI] [PubMed] [Google Scholar]
- Lauritzen F., Paulsen G., Raastad T., Bergersen L.H., Owe S.G. (2009) Gross ultrastructural changes and necrotic fiber segments in elbow flexor muscles after maximal voluntary eccentric action in humans. Journal of Applied Physiology 107(6), 1923-1934. [DOI] [PubMed] [Google Scholar]
- Lavender A.P., Nosaka K.A. (2008) Light load eccentric exercise confers protection against a subsequent bout of more demanding eccentric exercise. Journal of Science and Medicine in Sport 11, 291-298. [DOI] [PubMed] [Google Scholar]
- Mackey A.L., Brandstetter S., Schjerling P., Bojsen-Moller J., Qvortrup K., Pedersen M.M., Doessing S., Kjaer M., Magnusson S.P., Langberg H. (2011) Sequenced response of extracellular matrix deadhesion and fibrotic regulators after muscle damage is involved in protection against future injury in human skeletal muscle. The FASEB Journal 25(6), 1943-1959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maffulli N., Barrass V., Ewen S.W. (2000) Light microscopic histology of Achilles tendon ruptures. A comparison with unruptured tendons. The American Journal of Sports Medicine 28(6), 857-863. [DOI] [PubMed] [Google Scholar]
- Markovic G. (2007) Does plyometric training improve vertical jump height? A meta-analytical review. British Journal of Sports Medicine 41(6), 349-355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McHugh M.P., Connolly D.A., Eston R.G., Gleim G.W. (1999) Exercise-induced muscle damage and potential mechanisms for the repeated bout effect. Sports Medicine 27(3), 157-170. [DOI] [PubMed] [Google Scholar]
- Neves K.A., Johnson A.W., Hunter I., Myrer J.W. (2014) Does Achilles tendon cross sectional area differ after downhill, level and uphill running in trained runners? Journal of Sports Science and Medicine 13(4), 823-828. [PMC free article] [PubMed] [Google Scholar]
- Nosaka K., Clarkson P.M. (1996) Changes in indicators of inflammation after eccentric exercise of the elbow flexors. Medicine & Science in Sports & Exercise 28(8), 953-961. [DOI] [PubMed] [Google Scholar]
- Reeves N.D., Maganaris C.N., Narici M.V. (2003) Effect of strength training on human patella tendon mechanical properties of older individuals. The Journal of Physiology 548(3), 971-981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reeves N.D., Narici M.V. (2003) Behavior of human muscle fascicles during shortening and lengthening contractions in vivo. Journal of Applied Physiology 95(3), 1090-1096. [DOI] [PubMed] [Google Scholar]
- Roberts T.J., Konow N. (2013) How tendons buffer energy dissipation by muscle. Exercise and Sport Sciences Reviews 41(4), 186-193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosenquist C., Fledelius C., Christgau S., Pedersen B.J., Bonde M., Qvist P., Christiansen C. (1998) Serum CrossLaps One Step ELISA. First application of monoclonal antibodies for measurement in serum of bone-related degradation products from C-terminal telopeptides of type I collagen. Clinical Chemistry 44(11), 2281-2289. [PubMed] [Google Scholar]
- Sahlin K., Ren J.M. (1989) Relationship of contraction capacity to metabolic changes during recovery from a fatiguing contraction. Journal of Applied Physiology 67(2), 648-654. [DOI] [PubMed] [Google Scholar]
- Seene T., Umnova M., Kaasik P., Alev K., Pehme A. (2008) Overtraining injuries in athletic population. Skeletal muscle damage and repair. Ed: Tiidus P.M. Champaign: Human Kinetics; 173-184. [Google Scholar]
- Shalabi A., Kristoffersen-Wiberg M., Aspelin P., Movin T. (2004) Immediate Achilles tendon response after strength training evaluated by MRI. Medicine & Science in Sports & Exercise 36(11), 1841-1846. [DOI] [PubMed] [Google Scholar]
- Skurvydas A., Brazaitis M., Kamandulis S., Sipaviciene S. (2010) Peripheral and central fatigue after muscle-damaging exercise is muscle length dependent and inversely related. Journal of Electromyography and Kinesiology 20(4), 655-660. [DOI] [PubMed] [Google Scholar]
- Skurvydas A., Brazaitis M., Venckūnas T., Kamandulis S. (2011) Predictive value of strength loss as an indicator of muscle damage across multiple drop jumps. Applied Physiology, Nutrition, and Metabolism 36(3), 353-360. [DOI] [PubMed] [Google Scholar]
- Tardioli A., Malliaras P., Maffulli N. (2012) Immediate and short-term effects of exercise on tendon structure: biochemical, biomechanical and imaging responses. British Medical Bulletin 103(1), 169-202. [DOI] [PubMed] [Google Scholar]
- Van Drongelen S., Boninger M.L., Impink B.G., Khalaf T. (2007) Ultrasound imaging of acute biceps tendon changes after wheelchair sports. Archives of Physical Medicine and Rehabilitation 88(3), 381-385. [DOI] [PubMed] [Google Scholar]
- Van Ingen Schenau G.J., Bobbert M.F., DeHaan A. (1997) Mechanics and energetics of the stretch–shortening cycle: a stimulating discussion. Journal of Biomechanics 13, 484-496. [Google Scholar]
- Warren G.L., Ingalls C.P., Lowe D.A., Armstrong R.B. (2001) Excitation-contraction uncoupling: major role in contraction-induced muscle injury. Exercise and Sport Sciences Reviews 29(2), 82-87. [DOI] [PubMed] [Google Scholar]
- Waśkiewicz Z., Kłapcińska B., Sadowska-Krępa E., Czuba M., Kempa K., Kimsa E., Gerasimuk D. (2012) Acute metabolic responses to a 24-h ultra-marathon race in male amateur runners. European Journal of Applied Physiology 112(5), 1679-1688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Westerblad H., Duty S., Allen D.G. (1993) Intracellular calcium concentration during low-frequency fatigue in isolated single fibers of mouse skeletal muscle. Journal of Applied Physiology 75(1), 382-388. [DOI] [PubMed] [Google Scholar]
- Yu J.G., Malm C., Thornell L.E. (2002) Eccentric contractions leading to DOMS do not cause loss of desmin nor fibre necrosis in human muscle. Histochemistry and Cell Biology 118(1), 29-34. [DOI] [PubMed] [Google Scholar]


