Abstract
Impaired visual context processing is closely linked with expression of symptoms in people with psychosis. However, to date, there have been no investigations of this phenomenon in the critical ultra high-risk (UHR) period immediately preceding the onset of psychosis. A total of 73 participants (33 UHR, 40 Control) were evaluated with clinical and global functioning interviews and with a computerized task that required comparing the size of two target circles. Targets could appear by themselves (no-context condition), or within a context that made size judgment easier (helpful condition) or more difficult (misleading condition). Susceptibility to illusion was measured as the accuracy differences between the no-context and the helpful (i.e., helpful-index) and the misleading (i.e., misleading-index) conditions. Both groups exhibited approximately the same percentage of accurate responses to the no-context condition. However, the UHR subjects exhibited significantly less susceptibility to the illusion, performing superior to controls on the misleading-index. The UHR group also showed less susceptibility on the helpful-index, but this was not significant. Lower susceptibility on the misleading-index was associated with increased negative symptoms and role functioning at a trend level. Lower susceptibility on the helpful-index was significantly associated with increased negative symptoms and both poor role and social functioning. These results indicate that visual context processing is impaired during the UHR period, and is related to core illness features, suggesting that this task may be a useful biomarker in studies of UHR participants.
Keywords: Ebbinghaus illusion, Perceptual Organization, Visual Context Processing, Prodrome, Socio-occupational Functioning
Introduction
In recent years there has been a renewed interest in visual perception in schizophrenia, in part because it is arguably the best understood domain of mental functioning from a neuroscience perspective. As such, it has the potential to accelerate our understanding of the cognitive neuroscience of schizophrenia (Silverstein & Thompson, in press). Studies of perception in schizophrenia may also shed light on symptom formation. For example, several theories based in Gestalt psychology or phenomenology (reviewed in Uhlhaas & Mishara, 2007), or experimental psychology (Maher, 1988; Maher, 2005; Coltheart, Langdon, & McKay, 2011; Gerrans, 2014) posit that delusions emerge, at least in part, as attempts to account for anomalous perceptual experiences. While these theories have subtle differences (e.g., in attributing delusion formation to phenomenological changes, cognitive biases, or normal reasoning about anomalous experiences), an implication of all of them is that abnormal visual experience precedes the development of delusions. Therefore, three predictions from these theories are that: 1) abnormal perception should be related to delusions (and perhaps other positive symptoms) in people with an established diagnosis of schizophrenia; 2) in people at high-risk for the disorder, perceptual abnormalities, if present, could exist in the absence of delusions; 3) in the same at high-risk sample, delusions would not be present in people who do not demonstrate perceptual abnormality. The first point has been addressed by past studies. However, despite studies demonstrating that specific perceptual differences appear to be independent of the generalized deficit in schizophrenia (Knight, Elliott, & Freedman, 1985; Silverstein et al., 1996; Dakin, Carlin, & Hemsley, 2005; Koethe et al., 2009; Tibber et al., 2013; Yoon et al., 2009), related to psychotic symptoms (Silverstein et al., 2013; Horton & Silverstein 2011) and closely tied to disease course, (Silverstein et al., 2013), there have been very few studies of perception in youth at ultra high-risk (UHR) for psychosis. As the UHR period is uniquely bereft of 3rd variable confounds that may influence findings (medications, side-effects, significant cognitive/attentional impairment, acute symptomatology) and represents a significantly promising period for viable intervention (McGorry, 2005), understanding etiological processes and highlighting novel biomarkers for these youth can have far reaching effects, highly relevant to the entire psychosis spectrum.
One promising mechanism for improved understanding of the UHR period is the modulation of size perception by visual context (also known as size contrast), as demonstrated in tasks involving the Ebbinghaus illusion. Prior studies in patients with schizophrenia showed that reduced susceptibility to the illusion (i.e., more accurate judgment of inner/target circle size when surrounded by larger or smaller circles that normally make the inner circle appear smaller or larger, respectively, than its actual size; see Figure 1) was related to increased symptoms (Silverstein et al., 2013; Uhlhaas, Phillips, Schenkel, & Silverstein, 2006a). Further, one study indicated that people with trait schizotypy and the presence of any formal thought disorder performed more accurately on an Ebbinghaus illusion task than either controls or high-schizotypy subjects without evidence of formal thought disorder. Prior theoretical work suggests that the Ebbinghaus illusion results from interpretation (‘unconscious inference’ in the sense of Helmholz) of the images based on prior knowledge of depth cues (Doherty, Campbell, Tsuji, & Phillips, 2010). In this view, large surrounding circles serve as a signal that the overall stimulus is in the foreground, and so perception of the inner circle (i.e., the target of fixation) is minimized; conversely, small surrounding circles are interpreted by the visual system as indicating that the stimulus is in the background, and so the inner circle is magnified. Therefore, the link between reduced illusion susceptibility and psychosis may indicate that size contrast is a biomarker of the process by which stored information modulates sensory input, an example of ‘Bayesian’ processing whose impairment has been theorized to be a core mechanism involved in the generation of hallucinations and delusions (Clark, 2013). However, as promising as visual context processing is as a biomarker of relapse and recovery in schizophrenia, we currently do not know if impairments occur in the UHR period, or how it may be tied to clinical presentation or functioning in this critical group.
FIGURE 1.

Ebbinghaus Illusion
Note: Examples of stimuli shown in each of the context conditions (the text was not present in the displays, only the circles). On each trial participants indicated whether the target on the left or right was bigger. In each case shown here the inner circle on the right is 2% larger than the one on the left. Figure reprinted with permission by John Wiley and Sons, from Doherty et al., (2010).
In the present study UHR and control participants were assessed with a computerized task based on the Ebbinghaus illusion as well as clinical and global functioning interviews to examine several questions. Based on findings from a recent study observing that visual illusions involving depth perception were reduced in schizophrenia patients and in a small UHR sample(Koethe et al., 2009), and our earlier work with distinct samples reporting that patients with schizophrenia exhibit reduced illusion effects on size perception tasks (Uhlhaas, Phillips, Mitchell, & Silverstein, 2006b; Horton & Silverstein, 2011; Silverstein et al., 2013), we predicted that UHR youth would exhibit less susceptibility in the illusion conditions than controls: specifically, we predicted that when compared with controls, the UHR group would perform more accurately in the misleading-index (i.e., not exhibiting a decline in accuracy from performance in the no-context condition indicates less susceptibility to misleading illusions) and less accurately in the helpful-index (i.e., not exhibiting improvement in accuracy from performance on no-context condition indicates lower susceptibility to helpful visual illusions). Further, based on prominent theories linking abnormal visual processing to delusional thinking (Coltheart, Langdon & McKay, 2011; Gerrans 2014) and several studies linking resistance to the illusion to clinical presentation in schizophrenia patients (Horton & Silverstein, 2011; Silverstein & Keane, 2011; Silverstein et al., 2013), we predicted that lower illusion susceptibility in both conditions would be associated with increased symptomatology. Finally, it is important to consider that there is evidence tying early visual processing to functional outcome in persons with schizophrenia (Sergi & Green, 2003; Sergi, Rassovsky, Nuechterlein, & Green, 2006; Rassovsky et al., 2011). Consistent with this evidence, we predicted that lower illusion susceptibility in both conditions would be associated with poorer socio-occupational functioning.
Methods
Participants
Adolescent and young adult UHR and control participants) were recruited by Craigslist, email postings, newspaper ads, and community professional referrals (see Table 1 for demographic characteristics). Exclusion criteria included history of head injury, the presence of a neurological disorder, and lifetime substance dependence. The presence of an Axis I psychotic disorder was an exclusion criterion for UHR participants. The presence of a psychotic disorder in a first-degree relative or meeting criteria for an Axis I disorder were exclusionary criteria for controls. Healthy control participants were recruited through announcements (advertised as a study of development for volunteers with no psychiatric symptoms and no family history of psychosis). Due to technical issues with the perception assessment software and administration, it was necessary to exclude 4 individuals (1 UHR and 3 control). A total of 35 UHR and 40 control individuals participated in this study. The protocol was approved by the University Institutional Review Board (IRB).
TABLE 1.
Participant Demographics, Symptoms, Functioning and No-Context Performance
| Healthy Control | Ultra High-Risk | Grand Total | Group Differences | |
|---|---|---|---|---|
| Gender | ||||
| Males | 19(47.5%) | 19(56.6%) | 38(52.1%) | N.S. |
| Females | 21(52.5%) | 14(42.4%) | 35(47.9%) | |
| Total | 40 | 33 | 73 | |
| Age | ||||
| Mean Years (SD) | 18.13(2.60) | 18.67(1.45) | 18.37(2.16) | N.S. |
| Parent Education | ||||
| Mean Years (SD) | 15.37(3.34) | 14.88(3.30) | 15.15(3.31) | N.S. |
| Symptoms | ||||
| Positive | ||||
| Mean (SD) | .48(1.18) | 13.03(4.48) | 6.15(7.02) | p ≤ 0.01 |
| Negative | ||||
| Mean (SD) | .40(1.19) | 8.93(6.83) | 4.26(6.31) | p ≤ 0.01 |
| Global Functioning | ||||
| Role | ||||
| Mean (SD) | 8.48(.68) | 6.97(1.57) | 7.77(1.49) | p ≤ 0.01 |
| Social | ||||
| Mean (SD) | 8.63(.77) | 6.73(1.49) | 7.79(1.37) | p ≤ 0.01 |
Note: not significant (N.S.); Positive and negative symptoms reflect total sums from domains from the Structured Interview for Prodromal Syndromes (SIPS). Global functioning was assessed with the Global Functioning Scale: Role (GFS-R) and the Global Functioning Scale: Social (GFS-S).
Clinical Interviews
The Structured Interview for Prodromal Syndromes (SIPS) (Miller et al., 1999) was administered to diagnose a prodromal syndrome. Based on the widely used criteria for this interview, UHR participants were included in the study if they showed: 1) recent onset or escalation of moderate levels of attenuated positive symptoms (a score of 3–5) and/or 2) a decline in global functioning over the last 12 months accompanying the presence of schizotypal personality disorder (SPD), and/or 3) a decline in global functioning over the last 12 months accompanying the presence of a first-degree relative with a psychotic disorder such as schizophrenia (Miller et al., 1999). In addition, the SIPS was used to assess several distinct categories of prodromal symptom domains including positive and negative dimensions. The Structured Clinical Interview for Axis-I DSM-IV Disorders (SCID) (First, Spitzer, Gibbon, & Williams, 1995) was also administered to rule out formal psychosis (a noted exclusionary criterion). Training of interviewers (who were advanced doctoral students) was conducted over a 2-month period, and inter-rater reliabilities exceeded the minimum study criterion of Kappa ≥ .80.
Global Role and Social Functioning
Socio-occupational functioning was assessed with the Global Functioning Scale: Role (GFS-R) (Niendam et al., 2006), and the Global Functioning Scale: Social (GFS-S) (Auther, Smith, & Cornblatt, 2006). On the GFS-R, a score of 10 indicates “Superior Role Functioning”, whereas a low score of 1 reflects “Extreme Role Dysfunction”. A score of 10 on the GFS-S reflects “Superior Social/Interpersonal Functioning,” whereas the lowest score of 1 indicates “Extreme Social Isolation”. The scales were designed for adolescents/young-adults and have been found to be valid and reliable in assessing at-risk populations (Cornblatt et al., 2007).
Visual Context Processing Task
Stimuli were presented on an LCD monitor (resolution = 1600 × 900) with viewable dimensions of 48.3 by 26.7cm. Data were recorded and analyzed with a C++ program developed by Phillips and colleagues (2014) (Phillips, Chapman, & Berry, 2004). This task has been used in prior studies of the Ebbinghaus illusion, including two of patients with schizophrenia (Horton & Silverstein, 2011; Silverstein et al., 2013). On each trial, the task was to press a key to indicate whether the target on the left or the right half of the screen was larger. All circles were black and presented on a white background. The stimulus appeared on the screen until the subject responded or after 2 seconds. No feedback was provided informing the participant if an answer was correct or incorrect. If a response was not recorded within 2 seconds of stimulus onset, when computing mean accuracy in a condition, those trials were scored as 0.5 rather than 0 (incorrect); consistent with the other studies using this task (Silverstein et al., 2013), this strategy is designed to ensure that subjects who preferred to guess rather than time-out on a trial would not have an advantage. Trials were separated by 200ms. The targets were centered on either side of the screen and appeared either with or without surrounding circles (see below). The two target circles always differed in actual size. The center circle on one side was always 100 pixels (2.8° of visual angle) in diameter, while the center circle on the other side was always 2 pixels larger or smaller (1 pixel = .028° of visual angle). The side on which the larger circle appeared was randomized. This size comparison was presented in 3 conditions. In the misleading-condition, the target circles were always surrounded by 8 larger circles arranged in a square configuration (see Figure 1), with the larger central circle always surrounded by larger circles (3.5° in diameter) and the smaller central circle always surrounded by smaller circles (1.4° in diameter). In this condition, size contrast impairs discrimination by biasing the observer to perceive the larger target as smaller and the smaller target as larger (Doherty, Tsuji, & Phillips, 2008). In the helpful-condition, the target circles were also surrounded by 8 circles around the edges of a virtual square. Here, the smaller center circle was surrounded by circles 3.5° in diameter and the larger central circle was surrounded by circles 1.4° in diameter, so that size contrast increases accuracy. The 32 trials in the context conditions (16 in the misleading and 16 in the helpful condition) were presented in a different random order for each subject. In addition to these 32 trials, 32 additional trials were presented in a control (no-context) condition. In other words, the no-context trials were exactly the same as the block of trials with context, except that the surrounding circles were invisible. This block of trials was presented either before or after the trials containing context, with the order of context and no context blocks counterbalanced across subjects. It should be noted that a larger number of misleading stimuli and no-context stimuli, varying the sizes of target circles, were administered as part of the Ebbinghaus illusion task (Silverstein et al., 2013); however, for the purposes of this present report, we only include the smallest, most difficult size trials for the misleading context (n = 16) and the matching smallest no-context trials (n = 32). This decision was made because the helpful condition only contained trials at the smallest size difference (n = 16), and it was decided that the most sensitive test of the hypotheses would involve comparing performance in this, the most discriminating condition. The total task (including all conditions) takes approximately ten minutes to complete.
Analyses
Independent t-tests and chi-square tests were employed to examine differences between groups in demographic and clinical status variables. One-tailed tests were used for directional hypotheses. Group differences in visual context sensitivity were examined with a 2 × 2 repeated measures ANOVA (control/UHR x helpful/misleading condition). To examine the extent to which participants are influenced by the illusion, both the misleading and helpful conditions were compared to the no-context condition. Specifically, a score representing ‘% correct in the misleading condition - % correct in no-context condition’ defined the misleading index (i.e., how much the misleading illusion led scores to decline from the non-context condition). Similarly, a score representing ‘% correct in helpful condition - % correct in no-context condition’ defined the helpful index (i.e., how much the helpful illusion improved scores from the no-context condition). Because the valences of these two indices are in opposite directions, in the 2 × 2 ANOVA with these indices defining the repeated measures factor, a significant group x condition interaction indicates an overall group difference in visual context sensitivity. The group x condition interaction was explored with planned contrasts to test the directional hypotheses that the UHR group would be more context sensitive in the misleading condition, and less context sensitive in the helpful condition. We also employed a series of regression analyses to examine if performance in the illusion conditions predicted clinical symptom or functional outcome in the clinical group.
Results
There were no significant between-group differences in demographic characteristics including age, t(71)= 1.12, p = .266, parental education, t(71) = −0.64, p = .527, or gender, χ2(1) = 0.74, p = .391. As expected, the UHR group showed significantly more positive, t(1,71) = 15.68, p ≤ .001, d = 3.84, and negative symptoms, t(1,71) = 7.09, p ≤ .001, d = 1.73, when compared with controls at baseline. Similarly, the UHR group showed significantly poorer role functioning t(71) = −5.18, p ≤ .001, d = 1.26, and lower social functioning as well, t(1,71) = −6.64, p ≤ .001, d = 1.60. There were no significant group differences for the no-context condition t(71) = −0.50, p = .622, indicating that both groups performed at roughly the same level of accuracy in the absence of the illusion-producing visual contexts (UHR: mean = 63.40%, SD = 10.93%; Control: mean = 64.49%, SD = 6.99%) (see Table 1). The target variables were not normally distributed (Kolmogorov-Smirnov tests were significant for each) and, as a result, we employed statistical strategies that are robust to violations to the assumption of normality (repeated-measures ANOVA and regression).
Group Differences for Visual Context Sensitivity
There was a significant interaction between group and condition, F(1,71) = 4.00, p = .049, ηp2 = .05. Planned comparisons indicated that the UHR group (mean = −44.46%; SD = 26.53%) was significantly more accurate than controls (mean = −53.63%, SD = 12.98%), t(71) = 1.82, p = .038, d = 0.44, on the misleading-index. In addition, the UHR group’s performance in the helpful-index (mean = 25.05%, SD = 17.22%) was in the predicted direction but did not approach statistical significance (mean = 27.85%, SD = 11.31%), t(71) = 0.84, p = .204. Taken together, these results suggest that the UHR group demonstrated less context sensitivity than controls (see Figure 2).
FIGURE 2.
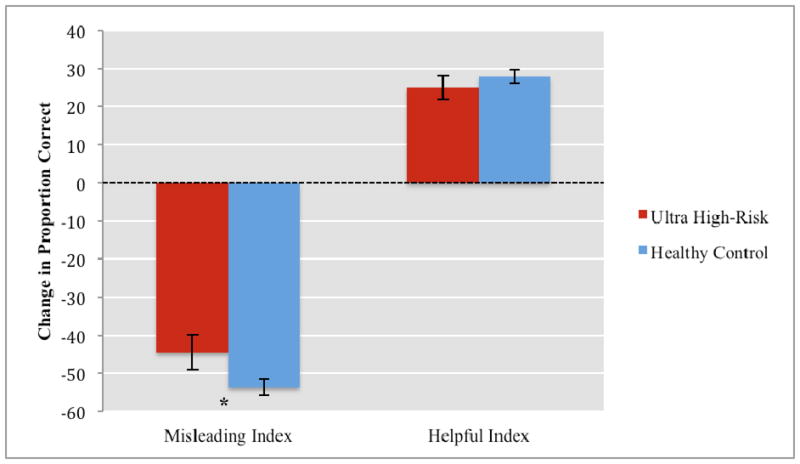
Sensitivity to Illusion Conditions by Group
Note: * p ≤ .05
Error bars represent standard error of mean;
Misleading Index = % correct on the misleading stimuli - % correct in the no-context stimuli;
Helpful index = % correct on helpful - % correct on no-context stimuli.
Relationships Between Visual Context Susceptibility, Symptoms, and Socio-occupational Functioning
The misleading-index performance was not related to positive symptoms, F(1,31) = 0.01, p = .462. Performance was associated with negative symptoms at a trend level, F(1,31) = 2.47, p = .063. In addition, performance on the misleading-index was associated with role functioning at a trend level, F(1,31) = 1.78, p = .096, and but not with social function, F(1,31) = 0.92, p = .172. As seen in Table 2, the direction of the Beta values suggests that reduced susceptibility on the misleading-index was associated at a trend level to elevated negative symptoms and role functioning. Results indicated that the helpful-index performance score was not associated with positive symptoms, F(1,31) = 0.76, p = .195, but was associated with negative symptom severity, F(1,31) = 3.35, p = .039, R2 = .10. These analyses also indicated that helpful-index scores were associated with poorer role, F(1,31) = 3.31, p = .039, R2 = .10, and poorer social functioning F(1,31) = 3.62, p = .034, R2 = .10. The direction of the Beta values suggests that reduced susceptibility on the helpful-index was tied to elevated negative symptoms and poor social-occupational functioning.
TABLE 2.
Associations Between Illusion Susceptibility, Symptoms and Functioning in Youth at Ultra High-Risk for Psychosis
| Misleading-Index | Helpful-Index | |||
|---|---|---|---|---|
| Beta Value | p-Value | Beta Value | p-Value | |
| Symptoms | ||||
| Positive | .02 | p = .462 | −.16 | p = .195 |
| Negative | .27 | p = .063 | −.31 | p = .039 |
| Global Functioning | ||||
| Role | −.23 | p = .096 | .31 | p = .039 |
| Social | −.17 | p = .172 | .32 | p = .034 |
Note: Symptom domains are scored utilizing the Structured Interview for Prodromal Syndromes (SIPS). Global functioning was assessed with the Global Functioning Scale: Role (GFS-R) and Social (GFS-S). Elevated scores on the SIPS reflect increased symptoms and increased scores on the GFS-R/S reflect superior functioning. Note: in the misleading-index, higher scores are equivalent to less susceptibility to illusion whereas in the helpful-index, lower scores are equivalent to less susceptibility to illusion.
Discussion
To our knowledge, this report represents the first published evidence for reduced susceptibility to the Ebbinghaus illusion in UHR individuals. Specifically, we observed that while the clinical and control groups performed comparably in the condition without an illusion component, UHR subjects demonstrated reduced overall context sensitivity, and the group difference was significant for the misleading context condition. Because the UHR participants performed comparably on the no-illusion condition and more accurately on the misleading condition than controls, this can be seen as promising support for a potential new series of biomarkers, as the findings are not likely to be attributable to a generalized deficit (Knight & Silverstein, 2001). Furthermore, results suggest that resistance to the illusion is closely tied with both negative symptoms as well as two key domains of global functioning. In the following, we discuss how these findings relate to past research and highlight implications for a novel biomarker in the visual domain.
Consistent with reports from patients with schizophrenia (Horton & Silverstein, 2011; Silverstein et al., 2013), we observed a decrease in illusion susceptibility in the UHR group. It is also important to consider that while investigators have reported large differences between chronic schizophrenia patients and controls in susceptibility to illusion (~60% when combining differences with controls in both the helpful and misleading conditions) (Silverstein et al., 2013), the present UHR sample differed at a rate of ~3% from control participants on the helpful-condition and ~9% on the misleading-condition. Interestingly, the difference between groups observed in the current study (combined in the two conditions = ~12%) is much closer to observations of the rates between first-episode schizophrenia patients and controls (Silverstein et al., 2013), suggesting that context processing impairments may worsen with disease progression.
Findings that resistance to the illusion in the UHR patients is associated with elevated negative symptoms are consistent with reports of similar correlations from studies of perceptual organization (Keri, Kiss, Kelemen, Benedek, & Janka, 2005) and backward masking (Green, Hellemann, Horan, Lee, & Wynn, 2012). In addition, Silverstein and colleagues (2013) observed that in first episode psychosis patients, increased visual context sensitivity on the Ebbinghaus illusion task over the course of a 2-week inpatient stay were associated with a reduction in negative symptoms during this same time period. There is a compelling theoretical rationale that is also consistent with these findings. One investigation observed that visual cortical GABA depletion in patients with schizophrenia was linked with decreased orientation-specific surrounding suppression (a measure of visual inhibition) (Yoon et al., 2010). Another study utilizing healthy volunteers reported ketamine-induced deficits in visual context-dependent processing, implicating a underlying role for deficient N-methyl-D-aspartate (NMDA) (Umbricht et al., 2000). Both sets of findings are in line with a GABA-glutamate hypothesis, whereby hypofunction in GABAergic interneurons in the prefrontal and hippocampal regions and hypofunction of a subpopulation of NMDA receptors contribute to negative symptoms (Coyle, 2004).
However it is also important to note that this is a nuanced area of the literature. Several related theoretical conceptualizations as well as investigations have noted relationships between abnormal visual processing, including illusion resistance, and positive symptoms (Coltheart, Langdon, & McKay, 2011; Keane, Silverstein, Wang, & Papathomas, 2013; Silverstein et al., 2013; Uhlhaas et al., 2006a; Yang et al., 2013; Gerrans 2014) but this was not detected in the present study. From a purely statistical perspective, this is likely to be due to range restriction, and more specifically, to the relatively low level of positive symptoms in the UHR group. This is supported by a study of clinically stable and relatively asymptomatic patients, which also did not find correlations between performance on a variant of the Ebbinghaus illusion task and symptoms (Yang et al., 2013). However, our data are also consistent with the implication of various theories (cited in the Introduction) that abnormal visual experiences would be present in patients where formal delusions have not yet developed, whereas delusions would not be found among patients who do not demonstrate perceptual impairment. What is now needed is to determine the extent to which delusions (or certain types of delusions) arise in people who demonstrate visual processing impairments, and do not arise in those who do not.
It is also important to consider that while significant group differences were found for the misleading-index, it was the helpful-index (group difference were in the predicted direction but were not significant) that was significantly correlated with symptom and functioning variables. In this context, it is important to note that the regressions were conducted in the UHR group alone, and therefore it is possible that while the misleading-index scores most strongly differentiate the UHR participants, within group variability on performance on the helpful-index may be most closely tied to the clinical presentation. That being said, replication in larger studies is needed. It should also be noted that for this study we analyzed data only from the most difficult target size-difference condition (2 pixel difference), and this contained only 16 trials in each context condition. Use of a greater number of trials might have allowed for greater sensitivity in detecting between-group differences and correlational relationships.
Several studies have implicated the predictive value of visual processing deficits for determining later functional impairment. For example, Sergi and Colleagues (2006) noted a moderating relationship for social perception between early visual impairment and functional outcome and in a structural equation modeling (SEM) study, and Green and colleagues (2012) found a single significant path from perception to beliefs/motivation to outcome. In conjunction with the noted literature, the present study highlights the possibility that dysfunctional visual context processing is a core impairment and characteristic of psychosis, and perhaps an indication of a more widespread context processing disturbance in other sensory and cognitive domains (Phillips & Silverstein, 2003, 2013). Indeed, if efficient and accurate context processing is necessary for the acquisition and maintenance of real-world skills (Green, Waldron, Simpson, & Coltheart, 2008), and it is already impaired prior to the onset of psychosis, during the adolescent time-period when critical skill acquisition sets the foundation for function over the rest of the lifetime (Zarrett & Eccles, 2006), this could be an important target for cognitive remediation. However, significantly more research is required before more specific recommendations can be made about targeted remediation. With the caveat in mind, the available evidence is promising. In the present study, we utilized two functioning scales that were designed specifically for investigating UHR populations (Auther et al., 2006; Cornblatt et al., 2007) and found that consistent with the broader literature, poor susceptibility to helpful illusions was linked with declines in both role functioning and social functioning.
While this investigation includes several methodological strengths, there are still a number of noteworthy limitations. First, while the present sample size is comparable or larger than other visual processing studies in the psychosis spectrum (i.e., key papers have ranged from 16–37 in the clinical group) (Keane et al., 2013; Koethe et al., 2009; Silverstein et al., 2013; Uhlhaas et al., 2006a) there is currently no follow-up data. As one of the utilities of UHR research is to determine predictors of clinical course, it will be integral for future studies to include a longitudinal design. Second, it will be important to determine the degree to which visual context sensitivity in general, and size contrast in particular, is more sensitive to aspects of the UHR state, compared to other perceptual and cognitive indices. For example, an earlier study of perceptual organization in UHR individuals did not find any differences from controls (Silverstein et al., 2006) and so future studies that clarify which visual processing mechanisms are, and are not, impaired in UHR persons may bring clarity to pathophysiological mechanisms in the UHR group. Taken together with recent trends in the literature the present findings suggest that visual perception is closely tied to factors underlying the pathogenesis of psychosis, and future research in this area has significant potential to improve our etiological conceptions, and develop promising biomarkers in this critical area.
Lay Summary.
Persons with schizophrenia experience abnormalities with perception, but currently it is unknown if youth at-risk also show similar levels of dysfunction. This study reports findings that high-risk youth appear to be less affected by visual illusions than matched controls, and this altered visual perception is associated with negative symptoms as well as real-world social and occupational functioning.
Acknowledgments
Funding: This work was supported by National Institutes of Health Grants R01MH094650 and R21/R33MH103231 (Mittal).
Footnotes
Conflicts: The authors have no potential conflicts of interest to report.
References
- Auther AM, Smith CW, Cornblatt B. Global Functioning: Social Scale (GF: Social) New York: Zucker-Hillside Hospital; 2006. [Google Scholar]
- Clark A. Whatever next? Predictive brains, situated agents, and the future of cognitive science. Behavioral Brain Science. 2013;36(3):181–204. doi: 10.1017/S0140525X12000477. [DOI] [PubMed] [Google Scholar]
- Coltheart M, Langdon R, McKay R. Delusional belief. Annual Review of Psychology. 2011;62:271–298. doi: 10.1146/annurev.psych.121208.131622. [DOI] [PubMed] [Google Scholar]
- Cornblatt BA, Auther AM, Niendam T, Smith CW, Zinberg J, Bearden CE, Cannon TD. Preliminary findings for two new measures of social and role functioning in the prodromal phase of schizophrenia. Schizophrenia Bulletin. 2007;33(3):688–702. doi: 10.1093/schbul/sbm029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coyle JT. The GABA-glutamate connection in schizophrenia: Which is the proximate cause? Biochemical Pharmacology. 2004;68(8):1507–1514. doi: 10.1016/j.bcp.2004.07.034. [DOI] [PubMed] [Google Scholar]
- Dakin S, Carlin P, Hemsley D. Weak suppression of visual context in chronic schizophrenia. Current Biology. 2005;15(20):R822–824. doi: 10.1016/j.cub.2005.10.015. [DOI] [PubMed] [Google Scholar]
- Doherty MJ, Campbell NM, Tsuji H, Phillips WA. The Ebbinghaus illusion deceives adults but not young children. Developmental Science. 2010;13(5):714–721. doi: 10.1111/j.1467-7687.2009.00931.x. [DOI] [PubMed] [Google Scholar]
- Doherty MJ, Tsuji H, Phillips WA. The context sensitivity of visual size perception varies across cultures. Perception. 2008;37(9):1426–1433. doi: 10.1068/p5946. [DOI] [PubMed] [Google Scholar]
- First M, Spitzer R, Gibbon M, Williams J. Structured Clinical Interview for the DSM-IV Axis I Disorders (SCID-I), Patient Edition. Washington DC: American Psychiatric Press; 1995. [Google Scholar]
- Gerrans P. Pathologies of hyperfamiliarity in dreams, delusions and déjà vu. Frontiers in Psychology. 2014;5:97. doi: 10.3389/fpsyg.2014.00097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Green M, Hellemann G, Horan WP, Lee J, Wynn JK. From perception to functional outcome in schizophrenia: Modeling the role of ability and motivation. Archives of General Psychiatry. 2012;69(12):1216–1224. doi: 10.1001/archgenpsychiatry.2012.652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Green MJ, Waldron JH, Simpson I, Coltheart M. Visual processing of social context during mental state perception in schizophrenia. Journal of Psychiatry and Neuroscience. 2008;33(1):34–42. [PMC free article] [PubMed] [Google Scholar]
- Horton HK, Silverstein SM. Visual context processing deficits in schizophrenia: Effects of deafness and disorganization. Schizophrenia Bulletin. 2011;37(4):716–726. doi: 10.1093/schbul/sbr055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keane BP, Silverstein SM, Wang Y, Papathomas TV. Reduced depth inversion illusions in schizophrenia are state-specific and occur for multiple object types and viewing conditions. Journal of Abnormal Psychology. 2013;122(2):506–512. doi: 10.1037/a0032110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keri S, Kiss I, Kelemen O, Benedek G, Janka Z. Anomalous visual experiences, negative symptoms, perceptual organization and the magnocellular pathway in schizophrenia: A shared construct? Psychological Medicine. 2005;35:1445–1455. doi: 10.1017/S0033291705005398. [DOI] [PubMed] [Google Scholar]
- Knight RA, Elliott DS, Freedman EG. Short-term visual memory in schizophrenics. Journal of Abnormal Psychology. 1985;94(4):427–42. doi: 10.1037//0021-843x.94.4.427. [DOI] [PubMed] [Google Scholar]
- Knight RA, Silverstein SM. A process-oriented approach for averting confounds resulting from general performance deficiencies in schizophrenia. Journal of Abnormal Psychology. 2001;110(1):15–30. doi: 10.1037//0021-843x.110.1.15. [DOI] [PubMed] [Google Scholar]
- Koethe D, Kranaster L, Hoyer C, Gross S, Neatby MA, Schultze-Lutter F, Ruhrmann S, Klosterkotter J, Hellmich M, Leweke FM. Binocular depth inversion as a paradigm of reduced visual information processing in prodromal state, antipsychotic-naive and treated schizophrenia. European Archives of Psychiatry and Clinical Neuroscience. 2009;259:195–202. doi: 10.1007/s00406-008-0851-6. [DOI] [PubMed] [Google Scholar]
- McGorry PD. Early intervention in psychotic disorders: Beyond debate to solving problems. British Journal of Psychiatry. 2005;48:s108–110. doi: 10.1192/bjp.187.48.s108. [DOI] [PubMed] [Google Scholar]
- Maher B. Anomalous experience and delusional thinking: The logic of explanations. In: Oltmanns TF, Maher B, editors. Delusional Beliefs. New York: Wiley; 1998. pp. 15–33. [Google Scholar]
- Maher B. Delusional thinking and cognitive disorder. Integrative Physiology Behavioral Science. 2005;40:136–146. doi: 10.1007/BF03159710. [DOI] [PubMed] [Google Scholar]
- Miller TJ, McGlashan TH, Woods SW, Stein K, Driesen N, Corcoran CM, Hoffman R, Davidson L. Symptom assessment in schizophrenic prodromal states. Psychiatry Quarterly. 1999;70(4):273–287. doi: 10.1023/a:1022034115078. [DOI] [PubMed] [Google Scholar]
- Niendam TA, Bearden CE, Johnson JK, McKinley M, Loewy R, O’Brien M, Nuechterlein KH, Green MF, Cannon TD. Neurocognitive performance and functional disability in the psychosis prodrome. Schizophrenia Research. 2006;84(1):100–111. doi: 10.1016/j.schres.2006.02.005. [DOI] [PubMed] [Google Scholar]
- Phillips WA, Chapman KL, Berry PD. Size perception is less context-sensitive in males. Perception. 2004;33(1):79–86. doi: 10.1068/p5110. [DOI] [PubMed] [Google Scholar]
- Phillips WA, Silverstein SM. Convergence of biological and psychological perspectives on cognitive coordination in schizophrenia. Behavioral Brain Science. 2003;26(1):65–82. doi: 10.1017/s0140525x03000025. [DOI] [PubMed] [Google Scholar]
- Phillips WA, Silverstein SM. The coherent organization of mental life depends on mechanisms for context-sensitive gain-control that are impaired in schizophrenia. Frontiers in Psychology. 2013;4:307. doi: 10.3389/fpsyg.2013.00307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rassovsky Y, Horan WP, Lee J, Sergi MJ, Green MF. Pathways between early visual processing and functional outcome in schizophrenia. Psychological Medicine. 2011;41(3):487–97. doi: 10.1017/S0033291710001054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sergi MJ, Green MF. Social perception and early visual processing in schizophrenia. Schizophrenia Research. 2003;59(2–3):233–241. doi: 10.1016/s0920-9964(01)00405-4. [DOI] [PubMed] [Google Scholar]
- Sergi MJ, Rassovsky Y, Nuechterlein KH, Green MF. Social perception as a mediator of the influence of early visual processing on functional status in schizophrenia. American Journal of Psychiatry. 2006;163(3):448–454. doi: 10.1176/appi.ajp.163.3.448. [DOI] [PubMed] [Google Scholar]
- Silverstein SM, Knight RA, Schwarzkopf SB, West LL, Osborn LM, Kamin D. Stimulus configuration and context effects in perceptual organization in schizophrenia. Journal of Abnormal Psychology. 1996;105(3):410–20. doi: 10.1037//0021-843x.105.3.410. [DOI] [PubMed] [Google Scholar]
- Silverstein S, Uhlhaas PJ, Essex B, Halpin S, Schall U, Carr V. Perceptual organization in first episode schizophrenia and ultra-high-risk states. Schizophrenia Research. 2006;83(1):41–52. doi: 10.1016/j.schres.2006.01.003. [DOI] [PubMed] [Google Scholar]
- Silverstein SM, Keane BP. Perceptual organization impairment in schizophrenia and associated brain mechanisms: Review of research from 2005 to 2010. Schizophrenia Bulletin. 2011;37(4):690–699. doi: 10.1093/schbul/sbr052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Silverstein SM, Keane BP, Wang Y, Mikkilineni D, Paterno D, Papathomas TV, Feigenson K. Effects of short-term inpatient treatment on sensitivity to a size contrast illusion in first-episode psychosis and multiple-episode schizophrenia. Frontiers in Psychology. 2013;4:1. doi: 10.3389/fpsyg.2013.00466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Silverstein SM, Thompson JL. A vision science perspective on schizophrenia. Schizophrenia Research: Cognition. doi: 10.1016/j.scog.2015.05.003. (in press) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tibber MS, Anderson EJ, Bobin T, Antonova E, Seabright A, Wright B, Carlin P, Shergill SS, Dakin SC. Visual surround suppression in schizophrenia. Frontiers in Psychology. 2013;4:88. doi: 10.3389/fpsyg.2013.00088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uhlhaas PJ, Phillips WA, Schenkel LS, Silverstein SM. Theory of mind and perceptual context-processing in schizophrenia. Cognitive Neuropsychiatry. 2006a;11(4):416–436. doi: 10.1080/13546800444000272. [DOI] [PubMed] [Google Scholar]
- Uhlhaas PJ, Phillips WA, Mitchell G, Silverstein SM. Perceptual grouping in disorganized schizophrenia. Psychiatry Research. 2006b;145(2):105–117. doi: 10.1016/j.psychres.2005.10.016. [DOI] [PubMed] [Google Scholar]
- Umbricht D, Schmid L, Koller R, Vollenweider FX, Hell D, Javitt DC. Ketamine-induced deficits in auditory and visual context-dependent processing in healthy volunteers: Implications for models of cognitive deficits in schizophrenia. Archives of General Psychiatry. 2000;57(12):1139–1147. doi: 10.1001/archpsyc.57.12.1139. [DOI] [PubMed] [Google Scholar]
- Yang E, Tadin D, Glasser DM, Hong SW, Blake R, Park S. Visual context processing in schizophrenia. Clinical Psychological Science. 2013;1(1):5–15. doi: 10.1177/2167702612464618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoon JH, Maddock RJ, Rokem A, Silver MA, Minzenberg MJ, Ragland JD, Carter CS. GABA concentration is reduced in visual cortex in schizophrenia and correlates with orientation-specific surround suppression. Journal of Neuroscience. 2010;30(10):3777–3781. doi: 10.1523/JNEUROSCI.6158-09.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoon JH, Rokem AS, Silver MA, Minzenberg MJ, Ursu S, Ragland JD, Carter CS. Diminished orientation-specific surround suppression of visual processing in schizophrenia. Schizophrenia Bulletin. 2009;35(6):1078–1084. doi: 10.1093/schbul/sbp064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zarrett N, Eccles J. The passage to adulthood: Challenges of late adolescence. New Directions for Youth Development. 2006;111:13–28. 17. doi: 10.1002/yd.179. [DOI] [PubMed] [Google Scholar]


