Abstract
Background
Underinflation of tracheal cuff is a risk factor for microaspiration of contaminated secretions and subsequent ventilator-associated pneumonia (VAP). The aim of this collaborative meta-analysis of individual participant data is to determine the impact of continuous control of Pcuff on the incidence of VAP.
Methods
Studies were identified by searching PubMed and references of relevant articles. Data from 3 prospective controlled trials (two randomized and one quasi-randomized), which evaluated the impact of continuous control of Pcuff on the incidence of VAP, were obtained and pooled together. Three different devices were used to continuously control Pcuff. VAP was diagnosed using clinical, radiologic, and quantitative microbiological criteria. The impact of continuous control of Pcuff on VAP was assessed by Cox regression analysis, stratified on trial.
Results
263 (48.4 %) patients received continuous control of Pcuff, and 280 (51.5 %) patients received routine control of Pcuff using a manometer. 36 (13.6 %) VAP were diagnosed in continuous control group, and 72 (25.7 %) in routine care group (HR 0.47, 95 % CI 0.31–0.71, p < 0.001). However, heterogeneity was apparent in continuous control effect size across trials (I2 = 58 %, p = 0.085). The number of patients needed to treat to prevent one VAP episode was 8. No significant impact of continuous control of Pcuff was found on duration of mechanical ventilation, ICU length of stay, or mortality.
Conclusion
Continuous control of Pcuff might be beneficial in reducing the risk for VAP. However, no significant impact of continuous control of Pcuff was found on duration of mechanical ventilation, ICU length of stay, or mortality.
Keywords: Cuff pressure, Pneumonia, Mechanical ventilation, Microaspiration, Critical care, Meta-analysis
Background
Prevention of ventilator-associated pneumonia (VAP) is an important strategy to improve the quality of care provided to critically ill patients [1–3]. While VAP-attributable mortality is still a matter for debate [4], this ICU-acquired infection is associated with increased antimicrobial use and duration of mechanical ventilation [5, 6]. Important progress has been achieved during the last two decades in the understanding of pathophysiology of VAP, resulting in improvement in prevention strategies and reduced incidence of VAP [7, 8].
Microaspiration of contaminated oropharyngeal and gastric secretions is the main mechanism of entry of bacteria into the lower respiratory tract [9, 10]. Local and general host defenses play an important role in the progression from colonization to VAP [11]. The quantity and virulence of bacteria are also important factors in this process [12]. Several measures have been studied for prevention of microaspiration in intubated critically ill patient. These could be classified into enteral nutrition, mechanical ventilation, tracheal tube, and patient-related factors [13]. With regards to tracheal tube, several recent studies investigated how sealing could be improved, in order to avoid microaspiration. Subglottic secretion drainage has been shown to significantly reduce VAP incidence, antimicrobial use, and duration of mechanical ventilation [14, 15]. Other preventive measures related to tracheal tube, such as polyurethane cuff and conical cuff shape have been suggested. However, a recent randomized controlled multicenter study did not report any significant impact of these measures regarding the rate of tracheobronchial colonization, or VAP [16].
Underinflation of tracheal cuff (<20 cmH2O) is a well-known risk factor for microaspiration and VAP [17]. Therefore, it is recommended to adjust cuff pressure (Pcuff) around 25 cmH2O using a manometer, to prevent complications related to underinflation and overinflation of tracheal cuff [18]. However, in spite of routine control of Pcuff using a manometer, underinflation and overinflation are very common in intubated patients [19, 20]. Continuous control of Pcuff has been suggested to improve tracheal sealing and to prevent VAP. Three prospective trials evaluated the impact of continuous control of Pcuff on the incidence of VAP [21–23]. However, all these studies were performed in single centers and reported different results. Therefore, we performed this collaborative-pooled meta-analysis to determine the impact of continuous control of Pcuff on the incidence of VAP in critically ill adult patients.
Methods
We established a collaboration to undertake this meta-analysis of individual patient data. We included all prospective trials, which evaluated the impact of continuous control of Pcuff on the incidence of VAP.
Ethical aspects
The three studies used for this pooled analysis have been approved by local institutional regulatory boards. Informed consent was obtained from all patients, or from their next of kin.
Search for eligible trials
We identified clinical prospective clinical trials that compared the continuous control of Pcuff and routine care regarding the incidence of VAP. We searched PubMed (from January 1995 through June 2015), using the terms “continuous control of tracheal cuff pressure,” “continuous control of endotracheal cuff pressure,” and the term “ventilator-associated pneumonia.” We also searched references of relevant articles. Studies comparing continuous control of Pcuff and another intervention versus routine care were excluded, because it is impossible to determine the exact impact of continuous control of Pcuff on VAP rate in these studies.
Outcomes
The primary outcome was the incidence of VAP. Secondary outcomes included duration of mechanical ventilation, mechanical ventilation-free days, duration of antimicrobial treatment, length of ICU stay, and ICU mortality.
Collected data
The rationale for choice of factors was based on prior association with outcome. At ICU admission: age, acute physiology and chronic health evaluation (APACHE) II score, sequential organ failure assessment (SOFA) score, male gender, cause for ICU admission, type of admission, diabetes mellitus, chronic obstructive pulmonary disease (COPD), chronic heart failure, cirrhosis, chronic renal failure, and immunosuppression. During ICU stay: SOFA score at randomization, subglottic secretion drainage, duration of mechanical ventilation before randomization, sucralfate, proton-pump inhibitor, or H2 receptor antagonist use, reintubation, mean Pcuff, percentage of Pcuff measurements <20 cmH2O, underinflation of Pcuff (<20 cmH2O), overinflation of Pcuff (>30 cmH2O), mean positive end expiratory pressure (PEEP) level, sedation, Ramsay score, head of bed elevation, paralytic agent use, red blood cell transfusion, enteral nutrition, ventilator-associated tracheobronchitis (VAT) [24], and tracheostomy. At VAP diagnosis: polymicrobial VAP and type of microorganisms.
VAP definition
VAP was defined as the presence of new or progressive pulmonary infiltrate and at least two of the following criteria: (a) fever (≥38 °C) or hypothermia (<36 °C), (b) leukocytosis (>12 × 109/L) or leukopenia (<3.5 × 109/L), and (c) purulent respiratory secretions. Microbiological confirmation was required in all patients (positive bronchoalveolar lavage ≥104 cfu/mL, or positive tracheal aspirate ≥105 cfu/mL) [25]. Only first episodes of VAP diagnosed > 48 h after starting mechanical ventilation were taken into account.
Statistical analysis
Quantitative variables are expressed as mean [±standard deviation (SD)] in case of Gaussian distribution, or median [interquartile range (IQR)] otherwise. Normality was examined using histograms and Shapiro–Wilk test. Qualitative variables are expressed as numbers (percentage). Patient characteristics at ICU admission and during ICU stay; and secondary outcomes were compared between the two study groups using Student t-test for quantitative variables (Mann–Whitney U test was used for non-Gaussian distribution) and Chi-square test for qualitative variables (Fisher’s exact was used when the expected cell frequency was <5).
The probability of VAP occurrence over time was compared between the two study group using a Cox proportional hazard model stratified on trial. Heterogeneity across trials was examined by formal interaction test and quantified by calculating the I2. Patients were censored at the time of death or extubation. We performed an exclusion sensitivity analysis to evaluate the contribution of individual studies to the overall pooled estimate. A sensitivity analysis excluding patients receiving subglottic secretion drainage was also performed.
In both groups pooled together, we used a Cox proportional hazard model stratified on trial to identify factors associated with the occurrence of VAP. All variables with a p value < 0.20 were included into a backward-stepwise Cox regression analysis.
Statistical analysis was done at the 2-tailed α level of 0.05, except tests for the homogeneity in which an α level of 0.10 was chosen. Data were analyzed with the SAS software package, version 9.3 (SAS Institute, Cary, NC, USA).
Results
Study characteristics
Among the 30 identified studies, 23 studies were directly excluded (reviews 9, duplicates 5, out of scope 5, others 4). Among the 7 remaining studies, 2 were excluded because Pcuff control was not continuous, and 2 because other concomitant preventive measures were used in the intervention group (Fig. 1).
Fig. 1.
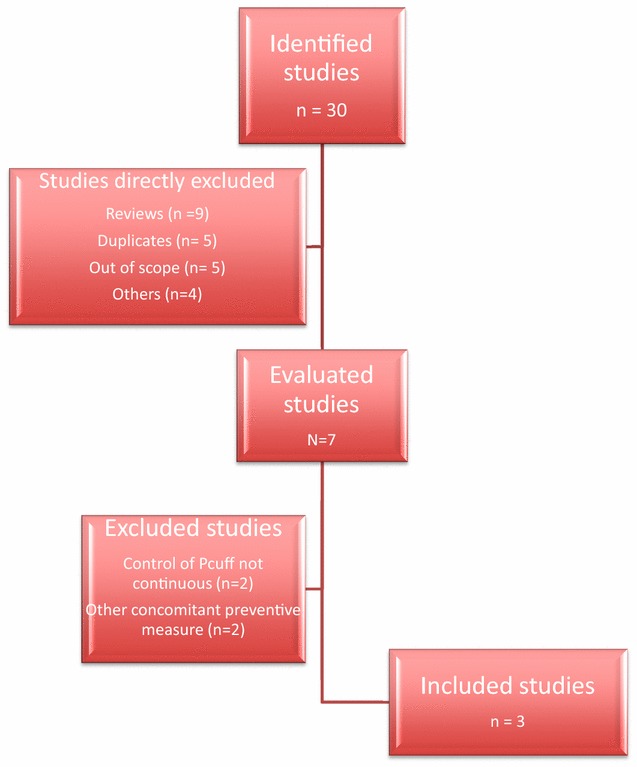
Studies evaluated for inclusion in this analysis
Data from 543 patients were obtained from the three included trials, representing 99 % of all included patients (5 patients were excluded for missing data). Two studies were randomized controlled and one was quasi-randomized controlled (patients who were admitted to an odd-numbered ICU cubicle received continuous control of Pcuff, and those admitted to an even-numbered ICU cubicle received routine care). Three different devices (Mallinckrodt Pressure Control®, VBM Medizintechnik GmbH, Sulz am Neckar, Germany; Nosten®, Leved, Saint Maur, France; and an electronic artisanal device) were used to continuously control Pcuff. Subglottic secretion drainage was used in some patients included in one trial. All tracheal tubes were polyvinyl chloride (PVC), standard shape—cuffed. Characteristics of the three studies are presented in Table 1.
Table 1.
Characteristics of studies assessing the impact of continuous control of cuff pressure on the incidence of ventilator-associated pneumonia
| Valencia et al. [21] | Nseir et al. [22] | Lorente et al. [23] | |
|---|---|---|---|
| Number of included patients | 137 | 122 | 284 |
| Type of study | Randomized controlled | Randomized controlled | Quasi-randomized controlled |
| Primary objective | VAP | Microaspiration | VAP |
| Device | Electronic | Pneumatic | Electronic |
| Target P cuff (cmH2O) | 25 | 25 | 25 |
| Surgical patients | 28 | 0 | 28 |
| Chronic respiratory disorders | 38 | 27 | 15 |
| VAP preventive measures | |||
| Oral care | CHX 0.12 % X3/days | CHX 0.10 % X3/days | CHX 0.12 % X3/days |
| Semirecumbent position | Yes | Yes | Yes |
| Subglottic secretion drainage | No | No | Yes |
| VAP incidence in control group | 15 | 26 | 22 |
| Reduction in VAP rate | NS | 62 | 51 |
Results are %, unless otherwise specified
VAP ventilator-associated pneumonia, CHX chlorhexidine, NS not significant
Patient characteristics
Two hundred and sixty-three (48 %) patients received continuous control of Pcuff and 280 (52 %) received routine care using a manometer. No significant difference was found in patient characteristics at ICU admission between patients who received continuous control of Pcuff, and those who received routine care (Table 2). While mean Pcuff was significantly higher in patients who received continuous control of Pcuff compared with those who received routine care, rate of patients with underinflation of Pcuff, with overinflation of Pcuff, and percentage of Pcuff measurements <20 cmH2O were significantly lower in patients with continuous control of Pcuff compared with those who received routine care. Other patient characteristics during ICU stay were similar in the two groups (Table 3).
Table 2.
Patient characteristics at ICU admission
| Continuous control of P cuff | |||
|---|---|---|---|
| Yes (n = 263) | No (n = 280) | p value | |
| Age, years, mean ± SD | 61 ± 16 | 63 ± 15 | 0.141 |
| APACHE II score | 18 (13, 23) | 18 (13, 23) | 0.624 |
| SOFA score | 5 (3, 7) | 5 (3, 8) | 0.424 |
| Male gender | 177 (67) | 172 (61) | 0.181 |
| Direct admission | 86 (33) | 86 (31) | >0.999 |
| Cause for admissiona | |||
| Cardiac surgery | 23 (10) | 26 (9) | 0.944 |
| Cardiovascular failure | 52 (20) | 49 (17) | 0.569 |
| Respiratory failure | 98 (37) | 104 (37) | >0.999 |
| Digestive failure | 25 (9) | 27 (10) | >0.999 |
| Neurologic failure | 48 (18) | 52 (18) | >0.999 |
| Others | 18 (7) | 24 (8) | 0.554 |
| Type of admission | 0.868 | ||
| Surgical | 57 (22) | 62 (22) | |
| Medical | 182 (69) | 196 (70) | |
| Trauma | 24 (9) | 22 (8) | |
| Diabetes mellitus | 57 (22) | 71 (25) | 0.363 |
| COPD | 65 (25) | 66 (23) | 0.833 |
| Chronic heart failure | 48 (18) | 43 (15) | 0.431 |
| Cirrhosis | 22 (8) | 16 (6) | 0.298 |
| Chronic renal failure | 16 (6) | 23 (8) | 0.427 |
| Immunosuppression | 39 (15) | 44 (16) | 0.867 |
| Study | 0.828 | ||
| 1 | 68 (26) | 69 (25) | |
| 2 | 61 (23) | 61 (22) | |
| 3 | 134 (51) | 150 (53) | |
Data are number (%), or median (IQR); unless otherwise specified
APACHE acute physiology and chronic health evaluation, SOFA sequential organ failure assessment
aSome patients had more than one cause for ICU admission
Table 3.
Patient characteristics during ICU stay
| Continuous control of P cuff | |||
|---|---|---|---|
| Yes (n = 263) | No (n = 280) | p value | |
| SOFA score at randomization | 4 (1, 7) | 4 (2, 6) | 0.538 |
| Subglottic secretion drainage | 53 (20) | 65 (23) | 0.447 |
| Antimicrobial treatment | 237 (90) | 260 (93) | 0.321 |
| MV duration before randomization | 0 (0, 1) | 0 (0, 1) | 0.531 |
| Sucralfate | 42 (16) | 45 (16) | >0.999 |
| Proton-pump inhibitor | 182 (69) | 180 (64) | 0.732 |
| H2 receptor antagonists | 31 (12) | 47 (17) | 0.124 |
| Reintubation | 41 (15) | 30 (11) | 0.120 |
| Mean P cuff | 25 (24, 26) | 22 (21, 24) | <0.001 |
| Underinflation of P cuff | 2 (1) | 118 (42) | <0.001 |
| % P cuff measurements <20 cmH2O | 0 (0,0) | 16 (0, 18) | <0.001 |
| Overinflation of P cuff | 8 (3) | 82 (29) | <0.001 |
| Mean PEEP (cmH2O) | 5 (5, 5) | 5 (5, 5) | 0.358 |
| Sedation | 235 (89) | 253 (90) | 0.806 |
| Ramsay score | 4 (3, 4) | 4 (3, 4) | 0.432 |
| HOB elevation (°) | 37 (30, 40) | 35 (30, 40) | 0.508 |
| Paralytic agent use | 22 (8) | 32 (11) | 0.315 |
| Red blood cell transfusion | 77 (29) | 73 (26) | 0.359 |
| Enteral nutrition | 178 (68) | 195 (70) | 0.689 |
| Tracheostomy | 49 (19) | 41 (15) | 0.257 |
Data are number (%), or median (IQR)
SOFA sequential organ failure assessment, MV mechanical ventilation, P cuff cuff pressure, PEEP positive end expiratory pressure, HOB head of bed
Characteristics of VAP patients
One hundred and eight (19.8 %) patients developed at least one VAP episode. Early-onset and late-onset pneumonia represented 49 and 51 % of VAP episodes, respectively. Duration of mechanical ventilation [5 (2, 10) vs 6 (3, 10) days, p = 0.323] and percentage of patients with early-onset or late-onset VAP (8 vs 12 %, 6 vs 14 %, p = 0.153; respectively) were similar in patients with continuous control of Pcuff compared with patients with routine care. Ten patients (9 %) had polymicrobial VAP. Gram-negative bacteria and MDR bacteria represented 84 and 36 % of microorganisms responsible for VAP episodes, respectively. Pseudomonas aeruginosa was the most frequently identified microorganism (16 %), followed by Staphylococcus aureus (10 %), and Enterobacter spp. (10 %) (Table 4).
Table 4.
Microorganisms responsible for ventilator-associated pneumonia
| Continuous control of P cuff | ||
|---|---|---|
| Yes (n = 36) | No (n = 72) | |
| Microorganisms (n) | 38 | 80 |
| Polymicrobial VAP | 2 (5) | 8 (11) |
| MDR bacteria | 13 (36) | 30 (42) |
| Gram-negative | 33 (92) | 66 (92) |
| Pseudomonas aeruginosa | 5 (14) | 14 (19) |
| Enterobacter species | 3 (8) | 9 (12) |
| Escherichia coli | 6 (17) | 5 (7) |
| Citrobacter freundii | 1 (3) | 4 (5) |
| Acinetobacter baumannii | 2 (5) | 5 (7) |
| Haemophilus influenzae | 8 (22) | 8 (11) |
| Stenotrophomonas maltophilia | 1 (3) | 4 (5) |
| Klebsiella oxytoca | 2 (5) | 6 (8) |
| Serratia species | 2 (5) | 7 (10) |
| Others | 3 (8) | 4 (5) |
| Gram-positive | 5 (14) | 14 (19) |
| Methicillin-resistant S. aureus | 1 (3) | 7 (10) |
| Methicillin-sensitive S. aureus | 1 (3) | 3 (4) |
| Streptococcus pneumoniae | 1 (3) | 2 (3) |
| Others | 2 (5) | 2 (3) |
VAP ventilator-associated pneumonia, MDR multidrug resistant
Data are number (%)
p > 0.2 for all comparisons
Impact of continuous control of Pcuff on outcomes
Continuous control of Pcuff was associated with significantly reduced incidence of VAP, with a HR of 0.47 (95 % CI 0.31–0.71) (Fig. 2). However, heterogeneity in continuous control effect size across trial was apparent (I2 = 58 %, p = 0.085). The effect of continuous control of Pcuff to reduce the incidence of VAP remained significant in exclusion sensitivity analysis, with a lower effect after exclusion of the study of Nseir and colleagues (HR 0.58; 95 % CI 0.37–0.92, p = 0.019). In further sensitivity analysis, excluding patients receiving subglottic secretion drainage, the effect of continuous control of Pcuff on VAP occurrence was not modified with a HR of 0.52 (95 % CI 0.33–0.79).
Fig. 2.
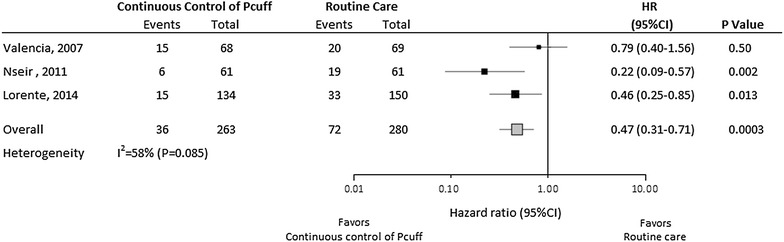
VAP-free survival curves for patients assigned to continuous control of cuff pressure and routine care groups. p value, and hazard ratio were calculated using Cox proportional hazard model stratified on trial
In multivariate cox regression analysis, continuous control of Pcuff remained significantly associated with decreased risk for VAP [HR 0.42, 95 % CI (0.27–0.64), p < 0.0001], independently of subglottic secretion drainage, chronic renal failure, respiratory, and digestive failures at ICU admission.
No significant difference was found in VAT rate between patients with continuous control of Pcuff and patients with routine care [13 of 263 (5 %) patients vs 22 of 280 (8 %), p = 0.227]. No significant impact of this procedure was found on duration of mechanical ventilation, mechanical ventilation-free days, antimicrobial treatment, length of ICU stay, or ICU mortality (Table 5).
Table 5.
Impact of continuous control of cuff pressure on secondary outcomes
| Continuous control of P cuff | |||
|---|---|---|---|
| Yes (n = 263) | No (n = 280) | p value | |
| MV duration (day) | 8 (4, 16) | 8 (4, 16) | 0.681 |
| MV free days | 3 (0, 6) | 2 (0, 5) | 0.426 |
| ICU length of stay (day) | 11 (6, 24) | 12 (7, 21) | 0.440 |
| Duration of antibiotic treatment | 9 (6, 15) | 10 (6, 15) | 0.778 |
| ICU mortality | 86 (33) | 91 (32) | >0.999 |
Data are number (%), or median (interquartile range)
MV mechanical ventilation, VAP ventilator-associated pneumonia, ICU intensive care unit
Safety data and cost-effectiveness
Similar rates of tracheostomy and reintubation were found in patients who received in patients with continuous control of Pcuff compared with patients with routine care (Table 3). No significant difference was found in tracheal ischemic lesions between the two groups (n = 96).
The number of patients needed to treat to prevent one VAP episode was 8.
Discussion
Our results suggest that continuous control of Pcuff might be beneficial in reducing the incidence of VAP. However, continuous control of Pcuff had no significant impact on secondary outcomes such as duration of mechanical ventilation, mechanical ventilation-free days, antimicrobial treatment, ICU stay, or ICU mortality.
Strengths and limitations
The major strengths of this study are the large number of included patients (n = 543) and the collaborative-pooled design. The similarity of the trials, including intervention, disease, and study population, provided a strong rationale to pool individual patient-level data. However, some limitations of our study should be acknowledged. First, the number of included studies was small, one study was quasi-randomized, and different devices were used to continuously control Pcuff in these trials, which could probably explain the observed heterogeneity in estimates. Nevertheless, percentage of measurements of Pcuff < 20 cmH2O in continuous control of Pcuff group was quite similar in the three studies. Other differences between the three studies, including the lower rate of VAP in control group, and the higher percentage of patients with chronic respiratory disorders in one study [21] compared with the two others, could also explain the high heterogeneity. Second, no significant impact of continuous control of Pcuff was found on secondary outcomes, such as duration of mechanical ventilation, mechanical ventilation-free days, antimicrobial treatment, or ICU mortality. This could be explained by the fact that a larger study sample is required to demonstrate such an effect. A posteriori calculation, based on the results of the current meta-analysis, of number of patients required to demonstrate a significant impact of continuous control of Pcuff on outcomes indicates that 348 patients are required to demonstrate a reduction of VAP incidence of 12 % (from 26 to 14 %, p = 0.05, power 80 %), and 1132 patients are required to demonstrate a reduction of mechanical ventilation duration of 2 days (mean 14 vs 12 days, standard deviation 12, p = 0.05, power 80 %). Previous well-conducted randomized controlled studies aiming at evaluating a preventive measure of VAP also suffered from this limitation [14, 26, 27]. For example, several randomized controlled studies comparing subglottic secretion drainage to routine care demonstrated a significant reduction in VAP rate, but all failed to show any significant reduction in mortality rate or duration of mechanical ventilation. However, a large meta-analysis [15] performed on 2442 patients showed a significant reduction in mechanical ventilation duration in patients with subglottic secretion drainage compared to those with routine care. Third, subglottic secretion drainage was used in some patients of one trial, and might have influenced the results. However, the rate of patients who received subglottic secretion drainage was similar in patients who received a continuous control of Pcuff and those who received routine care. In addition, sensitivity analysis excluding patients who received subglottic secretion drainage did not modify the protective effect of continuous control of Pcuff on VAP incidence. Fourth, cost-effectiveness analysis could not be performed. However, the number of patients needed to treat to prevent one VAP episode was 8. Fifth, the safety of continuous control of Pcuff, regarding tracheal ischemic lesions, was evaluated in only one study. Sixth, the under-reporting of negative results could have biased our results. However, we have checked the abstract of the major international critical care congresses and did not find any additional study on the impact of continuous control of Pcuff on VAP incidence.
Explanations for study results
Rello et al. [17] have previously reported that underinflation of Pcuff was independently associated with VAP in a subgroup of patients without antimicrobial treatment. In addition, one of the three studies included in this analysis [22] has investigated the impact of continuous control of Pcuff on microaspiration of gastric content and tracheobronchial colonization. This study reported a significant (27 %) reduction of microaspiration of gastric contents, defined as the presence of pepsin in >65 % of tracheal aspirates, in patients who received continuous control of Pcuff, compared with those who received routine care. Further, a significant reduction in bacterial concentration in tracheal aspirates was also observed.
Several studies reported that routine care using a manometer was not efficient in continuously controlling Pcuff, and that risk factors for underinflation and overinflation of tracheal cuff were not modifiable [19, 28, 29]. Other studies clearly showed the efficiency of some devices in continuously controlling Pcuff, suggesting that their use might be beneficial in preventing intubation-related complications [30, 31].
Two randomized studies, excluded from this analysis, reported a beneficial effect of continuous control of Pcuff on VAP incidence [32, 33]. However, in these studies continuous control of Pcuff was not the only tested preventive measure. While, low-volume low-pressure cuff was used in intervention group, PVC-standard cuff was used in routine care group. Therefore, it is impossible to determine whether the reduced incidence of VAP is related to continuous control of Pcuff or to low-volume low-pressure cuff use. This is the reason why these studies were not included in our analysis. Similarly, two other studies showed a beneficial effect of implementing a bundle for VAP prevention, including routine care for tracheal cuff [34, 35]. However, whether this beneficial effect is related to Pcuff control is unknown.
Different devices for continuous control of Pcuff
The heterogeneity between the three trials was high, probably reflecting the use of different devices for Pcuff control. An experimental study reported that electronic Pcuff controllers with rapid pressure correction interfere with the self-sealing mechanism of high-volume, low-pressure PVC–cuffed tracheal tubes and reduce their sealing characteristics [36]. Further, a recent prospective crossover study compared the efficiency of a pneumatic with an electronic device aiming at continuously controlling Pcuff [37]. The authors found underinflation of tracheal cuff to be more frequent using the electronic device than the pneumatic device (7 vs 0 %, respectively) and attributed this result to the over compensation of any elevated Pcuff.
Future studies
Future randomized controlled multicenter studies are required to confirm the beneficial effect of continuous control of Pcuff on VAP incidence, before recommending its routine use. In addition, the efficiency of different available devices should also be compared in critically ill patients.
Conclusions
Continuous control of Pcuff might be beneficial in reducing the incidence of VAP. However, no significant impact of this preventive measure was found on duration of mechanical ventilation, mechanical ventilation-free days, antimicrobial treatment, or ICU mortality. Further studies are required to confirm these results and to evaluate safety and cost-effectiveness of this preventive measure of VAP.
Authors’ contributions
SN, LL, and AT designed the study. SN, LL, MF, AR, OG, and GLB collected data. AD performed statistical analysis. All authors contributed in data analysis and manuscript preparation. SN is the guarantor of the paper, taking responsibility for the integrity of the work as a whole, from inception to published article. All authors read and approved the final version of the manuscript.
Acknowledgements
None.
Competing interests
The authors declare that they have no competing interests.
Abbreviations
- APACHE
Acute physiology and chronic health evaluation
- COPD
Chronic obstructive pulmonary disease
- ICU
Intensive care unit
- Pcuff
Cuff pressure
- PEEP
Positive end expiratory pressure
- PVC
Polyvinyl chloride
- SOFA
Sequential organ dysfunction assessment
- VAP
Ventilator-associated pneumonia
Contributor Information
Saad Nseir, Phone: 33 3 20 44 40 84, Email: s-nseir@chru-lille.fr.
Leonardo Lorente, Email: lorentemartin@msn.com.
Miquel Ferrer, Email: mferrer@clinic.ub.es.
Anahita Rouzé, Email: anahita.rouze@chru-lille.fr.
Oswaldo Gonzalez, Email: oswaldojglez@gmail.com.
Gianluigi Li Bassi, Email: glibassi@clinic.ub.es.
Alain Duhamel, Email: alain.duhamel59@gmail.com.
Antoni Torres, Email: atorres@clinic.ub.es.
References
- 1.Branch-Elliman W, Wright SB, Howell MD. Determining the ideal strategy for ventilator-associated pneumonia prevention: cost-benefit analysis. Am J Respir Crit Care Med. [DOI] [PubMed]
- 2.Craven DE, Hudcova J, Rashid J. Antibiotic therapy for ventilator-associated tracheobronchitis: a standard of care to reduce pneumonia, morbidity and costs? Curr Opin Pulm Med. 2015;21:250–259. doi: 10.1097/MCP.0000000000000158. [DOI] [PubMed] [Google Scholar]
- 3.Nair GB, Niederman MS. Ventilator-associated pneumonia: present understanding and ongoing debates. Intensive Care Med. 2014;41:34–48. doi: 10.1007/s00134-014-3564-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bekaert M, Timsit JF, Vansteelandt S, Depuydt P, Vésin A, Garrouste-Orgeas M, Decruyenaere J, Clec’h C, Azoulay E, Benoit D. Attributable mortality of ventilator-associated pneumonia: A reappraisal using causal analysis. Am J Respir Crit Care Med. 2011;184:1133–1139. doi: 10.1164/rccm.201105-0867OC. [DOI] [PubMed] [Google Scholar]
- 5.Melsen WG, Rovers MM, Groenwold RHH, Bergmans DCJJ, Camus C, Bauer TT, Hanisch EW, Klarin B, Koeman M, Krueger WA, Lacherade JC, Lorente L, Memish ZA, Morrow LE, Nardi G, van Nieuwenhoven CA, O’Keefe GE, Nakos G, Scannapieco FA, Seguin P, Staudinger T, Topeli A, Ferrer M, Bonten MJM. Attributable mortality of ventilator-associated pneumonia: a meta-analysis of individual patient data from randomised prevention studies. Lancet Infect Dis. 2013;13:665–671. doi: 10.1016/S1473-3099(13)70081-1. [DOI] [PubMed] [Google Scholar]
- 6.Jaillette E, Nseir S. Relationship between inhaled β2-agonists and ventilator-associated pneumonia: a cohort study. Crit Care Med. 2011;39:725–730. doi: 10.1097/CCM.0b013e318208ec61. [DOI] [PubMed] [Google Scholar]
- 7.Rouzé A, Nseir S. Continuous control of tracheal cuff pressure for the prevention of ventilator-associated pneumonia in critically ill patients: where is the evidence? Curr Opin Crit Care. 2013;19:440–447. doi: 10.1097/MCC.0b013e3283636b71. [DOI] [PubMed] [Google Scholar]
- 8.Klompas M, Branson R, Eichenwald EC, Greene LR, Howell MD, Lee G, Magill SS, Maragakis LL, Priebe GP, Speck K, Yokoe DS, Berenholtz SM. Strategies to prevent ventilator-associated pneumonia in acute care hospitals: 2014 update. Infect Control Hosp Epidemiol. 2014;35:915–936. doi: 10.1086/677144. [DOI] [PubMed] [Google Scholar]
- 9.Nseir S, Zerimech F, Jaillette E, Artru F, Balduyck M. Microaspiration in intubated critically ill patients: diagnosis and prevention. Infect Disord Drug Targets. 2011;11:413–423. doi: 10.2174/187152611796504827. [DOI] [PubMed] [Google Scholar]
- 10.Blot SI, Poelaert J, Kollef M. How to avoid microaspiration? A key element for the prevention of ventilator-associated pneumonia in intubated ICU patients. BMC Infect Dis. 2014;14:119. doi: 10.1186/1471-2334-14-119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Brusselaers N, Labeau S, Vogelaers D, Blot S. Value of lower respiratory tract surveillance cultures to predict bacterial pathogens in ventilator-associated pneumonia: systematic review and diagnostic test accuracy meta-analysis. Intensive Care Med. 2013;39:365–375. doi: 10.1007/s00134-012-2759-x. [DOI] [PubMed] [Google Scholar]
- 12.Messika J, Magdoud F, Clermont O, Margetis D, Gaudry S, Roux D, Branger C, Dreyfuss D, Denamur E, Ricard JD. Pathophysiology of Escherichia coli ventilator-associated pneumonia: implication of highly virulent extraintestinal pathogenic strains. Intensive Care Med. 2012;38:2007–2016. doi: 10.1007/s00134-012-2699-5. [DOI] [PubMed] [Google Scholar]
- 13.Jaillette E, Martin-Loeches I, Artigas A, Nseir S. Optimal care and design of the tracheal cuff in the critically ill patient. Ann Intensive Care. 2014;4:7. doi: 10.1186/2110-5820-4-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Damas P, Frippiat F, Ancion A, Canivet J-L, Lambermont B, Layios N, Massion P, Morimont P, Nys M, Piret S, Lancellotti P, Wiesen P, D’orio V, Samalea N, Ledoux D. Prevention of ventilator-associated pneumonia and ventilator-associated conditions. Crit Care Med. 2015;43:22–30. doi: 10.1097/CCM.0000000000000674. [DOI] [PubMed] [Google Scholar]
- 15.Muscedere J, Rewa O, McKechnie K, Jiang X, Laporta D, Heyland DK. Subglottic secretion drainage for the prevention of ventilator-associated pneumonia: a systematic review and meta-analysis. Crit Care Med. 2011;39:1985–1991. doi: 10.1097/CCM.0b013e318218a4d9. [DOI] [PubMed] [Google Scholar]
- 16.Philippart F, Gaudry S, Quinquis L, Lau N, Ouanes I, Touati S, Nguyen JC, Branger C, Faibis F, Mastouri M, Forceville X, Abroug F, Ricard JD, Grabar S, Misset B. Randomized intubation with polyurethane or conical cuffs to prevent pneumonia in ventilated patients. Am J Respir Crit Care Med. 2015;191:637–645. doi: 10.1164/rccm.201408-1398OC. [DOI] [PubMed] [Google Scholar]
- 17.Rello J, Soñora R, Jubert P, Artigas A, Rué M, Vallés J. Pneumonia in intubated patients: role of respiratory airway care. Am J Respir Crit Care Med. 1996;154:111–115. doi: 10.1164/ajrccm.154.1.8680665. [DOI] [PubMed] [Google Scholar]
- 18.Touat L, Fournier C, Ramon P, Salleron J, Durocher A, Nseir S. Intubation-related tracheal ischemic lesions: incidence, risk factors, and outcome. Intensive Care Med. 2013;39:575–582. doi: 10.1007/s00134-012-2750-6. [DOI] [PubMed] [Google Scholar]
- 19.Nseir S, Brisson H, Marquette C-H, Chaud P, Di Pompeo C, Diarra M, Durocher A. Variations in endotracheal cuff pressure in intubated critically ill patients: prevalence and risk factors. Eur J Anaesthesiol. 2009;26:229–234. doi: 10.1097/EJA.0b013e3283222b6e. [DOI] [PubMed] [Google Scholar]
- 20.Nseir S, Zerimech F, De Jonckheere J, Alves I, Balduyck M, Durocher A. Impact of polyurethane on variations in tracheal cuff pressure in critically ill patients: a prospective observational study. Intensive Care Med. 2010;36:1156–1163. doi: 10.1007/s00134-010-1892-7. [DOI] [PubMed] [Google Scholar]
- 21.Valencia M, Ferrer M, Farre R, Navajas D, Badia JR, Nicolas JM, Torres A. Automatic control of tracheal tube cuff pressure in ventilated patients in semirecumbent position: a randomized trial. Crit Care Med. 2007;35:1543–1549. doi: 10.1097/01.CCM.0000266686.95843.7D. [DOI] [PubMed] [Google Scholar]
- 22.Nseir S, Zerimech F, Fournier C, Lubret R, Ramon P, Durocher A, Balduyck M. Continuous control of tracheal cuff pressure and microaspiration of gastric contents in critically ill patients. Am J Respir Crit Care Med. 2011;184:1041–1047. doi: 10.1164/rccm.201104-0630OC. [DOI] [PubMed] [Google Scholar]
- 23.Lorente L, Lecuona M, Jiménez A, Lorenzo L, Roca I, Cabrera J, Llanos C, Mora ML. Continuous endotracheal tube cuff pressure control system protects against ventilator-associated pneumonia. Crit Care. 2014;18:R77. doi: 10.1186/cc13837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Nseir S, Di Pompeo C, Pronnier P, Beague S, Onimus T, Saulnier F, Grandbastien B, Mathieu D, Delvallez-Roussel M, Durocher A. Nosocomial tracheobronchitis in mechanically ventilated patients: Incidence, aetiology and outcome. Eur Respir J. 2002;20:1483–1489. doi: 10.1183/09031936.02.00012902. [DOI] [PubMed] [Google Scholar]
- 25.Focaccia R, Gomes Da Conceicao OJ. Pneumonia Hospitalar. Rev Bras Med. 1994;51(SPEC. ISS.):95–8.
- 26.Bouza E, Granda MJP, Hortal J, Barrio JM, Cercenado E, Muñoz P. Pre-emptive broad-spectrum treatment for ventilator-associated pneumonia in high-risk patients. Intensive Care Med. 2013;39:1547–1555. doi: 10.1007/s00134-013-2997-6. [DOI] [PubMed] [Google Scholar]
- 27.Bergan EH, Tura BR, Lamas CC. Impact of improvement in preoperative oral health on nosocomial pneumonia in a group of cardiac surgery patients: a single arm prospective intervention study. Intensive Care Med. 2014;40:23–31. doi: 10.1007/s00134-013-3049-y. [DOI] [PubMed] [Google Scholar]
- 28.Lizy C, Swinnen W, Labeau S, Poelaert J, Vogelaers D, Vandewoude K, Dulhunty J, Blot S. Cuff pressure of endotracheal tubes after changes in body position in critically ill patients treated with mechanical ventilation. Am J Crit Care. 2014;23:e1–e8. doi: 10.4037/ajcc2014489. [DOI] [PubMed] [Google Scholar]
- 29.Sole ML, Su X, Talbert S, Penoyer DA, Kalita S, Jimenez E, Ludy JE, Bennett M. Evaluation of an intervention to maintain endotracheal tube cuff pressure within therapeutic range. Am J Crit Care. 2011;20:109–118. doi: 10.4037/ajcc2011661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Duguet A, D’Amico L, Biondi G, Prodanovic H, Gonzalez-Bermejo J, Similowski T. Control of tracheal cuff pressure: a pilot study using a pneumatic device. Intensive Care Med. 2007;33:128–132. doi: 10.1007/s00134-006-0417-x. [DOI] [PubMed] [Google Scholar]
- 31.Farré R, Rotger M, Ferrer M, Torres A, Navajas D. Automatic regulation of the cuff pressure in endotracheally-intubated patients. Eur Respir J. 2002;20:1010–1013. doi: 10.1183/09031936.02.02692001. [DOI] [PubMed] [Google Scholar]
- 32.Young PJ, Pakeerathan S, Blunt MC, Subramanya S. A low-volume, low-pressure tracheal tube cuff reduces pulmonary aspiration. Crit Care Med. 2006;34:632–639. doi: 10.1097/01.CCM.0000201406.57821.5B. [DOI] [PubMed] [Google Scholar]
- 33.Gopal S, Luckraz H, Giri R, Nevill A, Muhammed I, Reid M, Bickerton S, Jenkins D. Significant reduction in ventilator-associated pneumonia with the Venner-PneuX System in high-risk patients undergoing cardiac surgery: the low ventilator-associated-pneumonia study. Eur J Cardio Thoracic Surg. 2015;47:e92–e96. doi: 10.1093/ejcts/ezu483. [DOI] [PubMed] [Google Scholar]
- 34.Rello J, Afonso E, Lisboa T, Ricart M, Balsera B, Rovira A, Valles J, Diaz E. A care bundle approach for prevention of ventilator-associated pneumonia. Clin Microbiol Infect. 2013;19:363–369. doi: 10.1111/j.1469-0691.2012.03808.x. [DOI] [PubMed] [Google Scholar]
- 35.Bouadma L, Mourvillier B, Deiler V, Le Corre B, Lolom I, Régnier B, Wolff M, Lucet J-C. A multifaceted program to prevent ventilator-associated pneumonia: impact on compliance with preventive measures. Crit Care Med. 2010;38:789–796. doi: 10.1097/CCM.0b013e3181ce21af. [DOI] [PubMed] [Google Scholar]
- 36.Weiss M, Doell C, Koepfer N, Madjdpour C, Woitzek K, Bernet V. Rapid pressure compensation by automated cuff pressure controllers worsens sealing in tracheal tubes. Br J Anaesth. 2009;102:273–278. doi: 10.1093/bja/aen355. [DOI] [PubMed] [Google Scholar]
- 37.Brisson H, Bouhamed B, Lu Q, Rouby JJ. Comparison of two automated endotracheal cuff pressure regulators devices in intubated critically ill patients: mechanical and electronic (abstract) Intensive Care Med. 2011;37:296. [Google Scholar]


