Dear Editor:
Glucose-dependent insulinotropic polypeptide (GIP) and proglucagon product glucagon-like peptide-1 (GLP-1) and their corresponding receptors promote secretion of glucose-dependent insulin[1] and may be responsible for up to 70% of postprandial insulin secretions[2]. A diminished incretin effect is typical for type 2 diabetes mellitus (T2DM) patients and their normoglycemic first-degree relatives due to reduced insulinotropic action of GIP[3]. Desensitization of the GIP/GIPR axis[4], down-regulation of GIP receptor (GIPR) on β cells[5], and variation in the GIPR gene[6] all contribute to reduced insulinotropic activity of GIP.
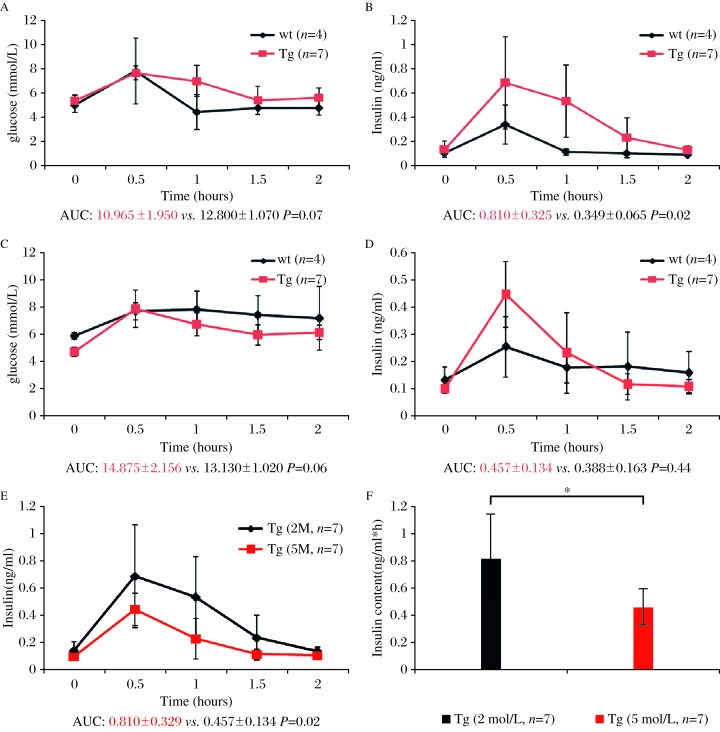
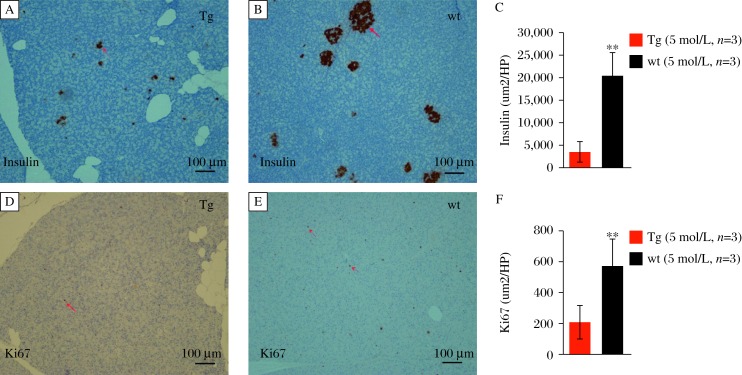
GIPRdn encodes human GIPR that is modified in the region of the third intracellular loop by a single mutation (Ala340→Glu340) and deletion of eight amino acids (residues 319–326) essential for signal transduction[8]. In this study, we produced porcine transgenic fibroblasts that overexpressed a GIPRdn under the control of rat Ins2 promoter in the pancreas. The study protocol was approved by the local institutional review board and animal study was done in accordance with established national and institutional guidelines. Oral glucose tolerance tests (OGTTs) in 2-month-old GIPRdn pigs and controls revealed no differences in area under the glucose response curve (AUCglucose) between the two groups (P = 0.07, Fig. 1A). However, the postprandial blood glucose declined slowly and the area under the insulin response curve (AUCinsulin) in GIPRdn pigs increased 2.3-fold versus the controls, implying impaired glucose tolerance (P = 0.02; Fig. 1B). OGTTs in 5-month-old GIPRdn pigs and controls showed no significant differences in glucose levels or insulin content between the groups (Fig. 1C, 1D). Additionally, we observed no significant differences in the insulin response profiles between 2-month-old and 5-month-old controls. However, AUCinsulin was reduced by 44% when the transgenic pigs grew from 2 months to 5 months of ages (Fig. 1E, 1F). To clarify the effect of GIPRdn expression on the proliferation of pancreatic cells, we measured insulin and Ki67 expression by immunohistochemistry in 5-month-old GIPRdn pigs and age-matched controls. Compared with the controls, the number and volume of islets in transgenic pigs decreased. Insulin expression in the islets was reduced by 83% (P<0.01) in 5-month-old GIPRdn pigs (Fig. 2A-2C). In addition, Ki67 expression in GIPRdn pigs decreased to 36% of controls, suggesting that GIPRdn expression impaired islet development and reduced insulin secretion (Fig. 2D-2F).
Fig. 1. Oral glucose tolerance test in GIPRdn transgenic (Tg) and wild type (wt) pigs.
A) Serum glucose, and (B) insulin levels, in 2-monthold pigs (AUC glucose P=0.07, AUC insulin P=0.02). (C) Serum glucose, and (D) insulin levels in 5-month-old pigs (AUC glucose P=0.06, AUC insulin P=0.44). (E) (F) Insulin levels in 2-month-old and 5-month-old GIPRdn transgenic pigs (P=0.02). * P=<0.05.
Fig. 2. Insulin and Ki67 expression in the pancreas.
A: transgenic pigs (Tg); B: wild type (wt) pigs. Insulin is expressed in islet. C: transgenic pigs (Tg); B: wild type (wt) pigs. Ki67 expression is decreased in GIPRdn transgenic pigs. (n=3 per group, **P=<0.01).
Transgenic mice overexpressing a dominant-negative GIPR were found to develop diabetes accompanied by a marked fasting hypoinsulinemia and severe reduction of β-cell mass by 10 days of age[7]. Similarly, transgenic pigs overexpressing a dominant-negative GIPR exhibited a pre-diabetic phenotype of hyperglycemia and hyperinsulinemia in OGTT[8]. However, no transgenic pigs had developed fasting hyperglycemia. In this study, 2-month-old GIPRdn pigs exhibited a slow decline in postprandial blood glucose and an increase in insulin secretion following an oral glucose load. This increase was approximately 2.3 times higher than in the control, thus imitating insulin secretion in the prodromal stage of T2DM. As the pigs grew up, pancreas development was significantly impaired due to the compromised GIP/GIPR axis, which resulted in reduced insulin secretion and islet dysfunction. Studies indicated that GIP and GLP-1 induced the transcription of beta cell proliferation, differentiation, survival and transcription of some genes[9]. GIP was believed to be the mitogen of beta cells, resulting in insulin secretion and inhibiting the apoptosis of beta cells. In this study, there were no significant differences in insulin content between the 5-month-old groups. However, the oral glucose load of 5-month-old transgenic pigs resulted in a 44% decrease in insulin secretion compared with the 2-month-old transgenic pigs. This is consistent with previous studies and implies progression of T2DM. Defective pancreatic proliferation led to fewer and smaller islets, which were excessively activated to produce more insulin by compensatory mechanism. Overload pressure from the compensation mechanism and impaired proliferation for inactivated GIP/GIPR axis further deteriorated islet function. Similar to the previous study[9], the transgenic pigs did not develop diabetes until 2 years of age. Diabetes is caused by many factors; a single gene change would not lead to the occurrence of the disease.
In summary, GIPRdn pigs exhibit impaired glucose metabolism and beta cell function and simulated the phenotype of the prodromal stage in diabetes and progression of the disease.
References
- [1].Drucker DJ. The biology of incretin hormones[J] Cell Metab. 2006;3(3):153–165. doi: 10.1016/j.cmet.2006.01.004. [DOI] [PubMed] [Google Scholar]
- [2].Holst JJ, Vilsbøll T, Deacon CF. The incretin system and its role in type 2 diabetes mellitus[J] Mol Cell Endocrinol. 2009;297(1–2):127–136. doi: 10.1016/j.mce.2008.08.012. [DOI] [PubMed] [Google Scholar]
- [3].Nauck MA, Heimesaat MM, Orskov C, et al. Preserved incretin activity of glucagon-like peptide 1 [7-36 amide] but not of synthetic human gastric inhibitory polypeptide in patients with type-2 diabetes mellitus[J] J Clin Invest. 1993;91(1):301–307. doi: 10.1172/JCI116186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [4].Meier JJ, Hücking K, Holst JJ, et al. Reduced insulinotropic effect of gastric inhibitory polypeptide in first-degree relatives of patients with type 2 diabetes[J] Diabetes. 2001;50(11):2497–2504. doi: 10.2337/diabetes.50.11.2497. [DOI] [PubMed] [Google Scholar]
- [5].Vilsbøll T, Krarup T, Madsbad S, et al. Defective amplification of the late phase insulin response to glucose by GIP in obese Type II diabetic patients[J] Diabetologia. 2002;45(8):1111–1119. doi: 10.1007/s00125-002-0878-6. [DOI] [PubMed] [Google Scholar]
- [6].Saxena R, Hivert MF, Langenberg C, et al. Genetic variation in GIPR influences the glucose and insulin responses to an oral glucose challenge[J] Nature Genet. 2010;42(2):142–148. doi: 10.1038/ng.521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [7].Herbach N, Goeke B, Schneider M, et al. Overexpression of a dominant negative GIP receptor in transgenic mice results in disturbed postnatal pancreatic islet and beta-cell development[J] Regul Pept. 2005;125(1-3):103–117. doi: 10.1016/j.regpep.2004.08.021. [DOI] [PubMed] [Google Scholar]
- [8].Renner S, Fehlings C, Herbach N, et al. Glucose intolerance and reduced proliferation of pancreatic beta-cells in transgenic pigs with impaired glucose-dependent insulinotropic polypeptide function[J] Diabetes. 2010;59(5):1228–1238. doi: 10.2337/db09-0519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [9].Ehses JA, Pelech SL, Pederson R, et al. Glucose-dependent insulinotropic polypeptide activates the Raf-Mek1/2-ERK1/2 module via a cyclic AMP/cAMP-dependent protein kinase/Rap1-mediated pathway[J] J Biol Chem. 2002;277(40):37088–37097. doi: 10.1074/jbc.M205055200. [DOI] [PubMed] [Google Scholar]